Abstract
The potential for neurogenesis in the cranial (superior) cervical ganglia (SCG) of the sympathetic nervous system was evaluated. Eleven consecutive daily doses of guanethidine (100 mg/kg/d) were administered intraperitoneally to rats in order to destroy postganglionic sympathetic neurons in SCG. Following the last dose, animals were allowed to recover 1, 3, or 6 months. Right and left SCG from guanethidine-treated and age-matched, vehicle-treated control rats were harvested for histopathologic, morphometric, and stereologic evaluations. Both morphometric and stereologic evaluations confirmed neuron loss following guanethidine treatment. Morphometric analysis revealed a 50% to 60% lower number of tyrosine hydroxylase (TH)-positive neurons per unit area of SCG at both 3 and 6 months of recovery, compared to ganglia of age-matched controls, with no evidence of restoration of neuron density between 3 and 6 months. Reductions in TH-positive neurons following guanethidine treatment were corroborated by unbiased stereology of total hematoxylin and eosin-stained neuron numbers in SCG. Stereologic analyses revealed that total neuron counts were lower by 37% at 3 months of recovery when compared to age-matched vehicle controls, again with no obvious restoration between 3 and 6 months. Thus, no evidence was found that postganglionic neurons of the sympathetic nervous system in the adult rat have a neurogenic capacity.
Introduction
Today, it is generally well accepted that the brain of most adult mammalian species can generate new neurons. 1,2 Using combinations of proliferative and neuron-specific stains in a variety of animal models, 3 -5 scientists have generated robust evidence for the phenomenon of neurogenesis within specific regions of the adult mammalian central nervous system (CNS), especially the subventricular zone of the lateral ventricles and the subgranular zone of the hippocampal dentate gyrus. Neurogenesis has been reported in these regions of adult human brain as well. 6 However, more recent reports suggest that neurogenesis in higher primates may decline rapidly in juveniles and young adults. 7 Neurogenesis occurs under both noninjury 3 and injury conditions. 8 Additionally, other areas of the brain traditionally not considered neurogenic, such as the cortex and the cerebellum, have also been reported to give rise to new neurons under conditions of injury 9 and may in fact be neurogenic in the absence of injury. 10
Neurogenesis has also been reported in the somatic portion of the peripheral nervous system of adult animals. A number of studies have provided evidence of neurogenesis in peripheral sensory ganglia with increasing age in adult rodents. 11,12 Cells with a neuroprogenitor phenotype (eg, expressing the progenitor marker nestin) have been identified in dorsal root ganglia (DRG) in adult rats. 13,14 More recently, it has been shown that capsaicin-induced neuronal loss in the nodose ganglia and DRG was restored over 2- to 3-month period from a local pool of proliferating progenitor cells, 14 -16 perhaps satellite glial cells. 17
There are no reports of neurogenesis in the autonomic portion of the peripheral nervous system. However, there appear to be progenitor cells in perinatal mouse and adult rat sympathetic cranial (superior) cervical ganglion (SCG), 18,19 suggesting a potential capacity for neurogenesis. In the work described here, we hypothesized that postganglionic neuron loss in the ganglia of the sympathetic division of the autonomic nervous system may be replaced over time, similar to what has been reported for neurons in the sensory nodose and DRG ganglia. However, the basic anatomy and physiology of the sympathetic ganglia is different from sensory ganglia. Neurons in the sympathetic ganglia are primarily multipolar, adrenergic, and mediate involuntary functions, whereas neurons in the sensory ganglia are primarily pseudo-unipolar and transmit sensory information from the periphery to the CNS using a variety of chemical transmitters. Observations of neurogenesis in the sensory ganglia thus may not necessarily translate to the autonomic nervous system. This question is important because there are a number of diseases associated with the sympathetic nervous system (eg, Horner Syndrome and Familial Dysautonomia) for which a better understanding of neuronal plasticity and regenerative capacity in autonomic ganglia is important. Additionally, neurogenesis within the autonomic nervous system would be an important consideration when developing drugs with potential adverse effects on this portion of the peripheral nervous system. For example, the new class of anti-nerve growth factor therapies being developed for pain has been associated with nonadverse, reversible neuronal atrophy in the sympathetic ganglia. 20
To test our hypothesis that neuron loss in the sympathetic ganglia may be replaced over time, we used guanethidine, an antihypertensive drug, 21 as a model toxicant that at high doses cause selective destruction of postganglionic adrenergic neurons within the sympathetic ganglia in the rat. 22 We have shown previously that daily administration of high-dose guanethidine (100 mg/kg/d) to rats for 11 days causes significant reductions in sympathetic neurons in T3 23 and cranial (superior) cervical 24 ganglia following a 3- to 4-week recovery period. The objective of the work described here was to determine whether any restoration of neurons occurs after a 3- or 6-month recovery following administration of high-dose (100 mg/kg/d) guanethidine to rats. To answer this question, we used a combination of morphometric and stereology techniques to estimate neuron losses and recovery in the SCG. Because guanethidine kills postganglionic adrenergic neurons, we used tyrosine hydroxylase (TH), the rate-limiting enzyme in for catecholamine synthesis and commonly used marker for adrenergic neurons, in our morphometric analyses. In case we were killing nonadrenergic neurons, we used hematoxylin and eosin (H&E) to visualize and count all neurons in the stereologic analysis.
Methods
Design
Sixty adult male Sprague-Dawley (Crl: CD® [SD]) rats were obtained from Charles River Laboratories (Morrisville, North Carolina) and randomly assigned to 6 groups of 10 rats each (Table 1). Rats in groups 2, 4, and 6, approximately 13 to 14 weeks old and 350 and 450 g at initiation of dosing were administered guanethidine sulfate (BOC Sciences, Shirley, New York) by intraperitoneal (IP) injection at 100 mg/kg once daily for 11 days. At the same time, age- and weight-matched control rats in groups 1, 3, and 5 received daily IP injections of vehicle (0.9% saline 1 mL/kg/d) for 11 days. Rats were individually housed in polycarbonate cages in animal rooms under controlled light–dark cycle (12/12 hours), temperature (20°C-26°C), and relative humidity (30%-70%) levels. Rats had free access to food and water and were monitored daily for signs of dehydration, malaise, or general discomfort. Body weights and food consumption were monitored predose on treatment day 1, on treatment days 3, 5, 7, and 11, and weekly during recovery phases. After the final guanethidine treatment, rats were allowed to recover for 1 (groups 1 and 2), 3 (groups 3 and 4), and 6 (groups 5 and 6) months prior to euthanasia and necropsy. All procedures were approved by the Pfizer Institutional Animal Care and Use Committee and were consistent with all state and federal laws and National Institutes of Health regulations.
Experimental Design.
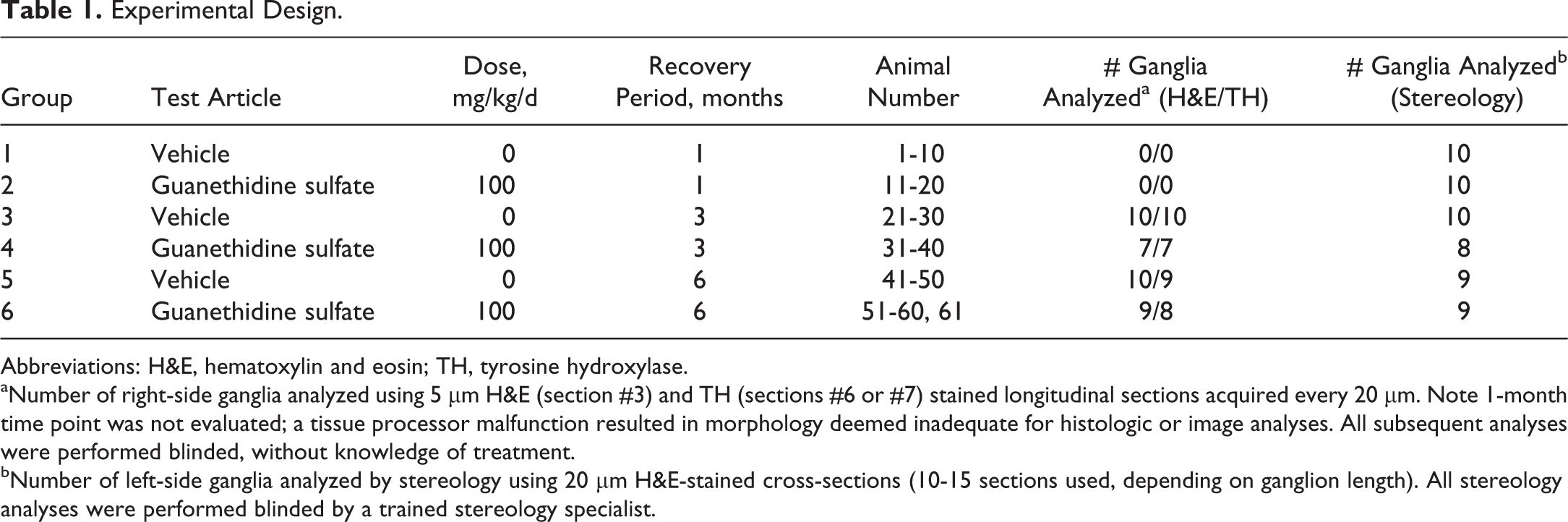
Abbreviations: H&E, hematoxylin and eosin; TH, tyrosine hydroxylase.
aNumber of right-side ganglia analyzed using 5 µm H&E (section #3) and TH (sections #6 or #7) stained longitudinal sections acquired every 20 µm. Note 1-month time point was not evaluated; a tissue processor malfunction resulted in morphology deemed inadequate for histologic or image analyses. All subsequent analyses were performed blinded, without knowledge of treatment.
bNumber of left-side ganglia analyzed by stereology using 20 µm H&E-stained cross-sections (10-15 sections used, depending on ganglion length). All stereology analyses were performed blinded by a trained stereology specialist.
Necropsy
Rats were anesthetized with sodium pentobarbital (100 mg/kg, IP), perfused transcardially with saline, and then 4% methanol-free paraformaldehyde in 0.1 M phosphate buffer. The head was separated from the carcass and immersed in 4% paraformaldehyde at ∼4°C. After fixation, both the left and the right SCG were harvested from the neck under a dissecting microscope. Briefly, the muscle layers were removed from the trachea to expose the vagosympathetic trunk, including the carotid artery. The sympathetic trunk was followed cranially until the ganglion was exposed at the level of bifurcation of the internal and external carotid artery. The intact SCG were then carefully dissected free of surrounding tissues while minimizing tension placed on the tissue to avoid morphological artifacts. 25 Right-side ganglia were then used for microscopic and morphometric analyses, and left-side ganglia were used for stereology analysis.
Histopathology
Right-side SCG were dehydrated through graded ethanols and xylenes using a Sakura Tissue-Tek VIP 6 Vacuum Infiltration Processor for automated processing (Sakura Finetek, Torrence, California) and then individually embedded along the long axis in paraffin and used for H&E staining and TH immunohistochemistry (IHC). Briefly, 5-µm sections were cut using a rotary microtome. Once the first full section of ganglion was located, a 5-µm section was then acquired every 20 µm and mounted on glass slides. Hematoxylin and eosin staining was done using a Sakura Tissue-Tek Prisma Automated Slide Stainer (Sakura Finetek, Torrence, California). For TH IHC, slides were deparaffinized and rehydrated and then immersed into Rodent Decloaker (Biocare Medical, Concord, California) and loaded into a Biocare Decloaking Chamber NxGENTM (Biocare Medical) set at 110°C for 15 minutes. Immunohistochemistry staining was completed on the Biocare IntelliPATH automated IHC instrument (Biocare Medical). For each TH assay, a subset of duplicate SCG slides were run as negative controls. These slides were incubated with a rabbit IgG isotype control (polyclonal; Vector Labs, Burlingame, California) in place of the primary antibody. Briefly, endogenous peroxidase was blocked by incubating with a 3% solution of hydrogen peroxide for 10 minutes, and nonspecific binding sites were blocked by applying Rodent Block R (Biocare Medical) for 20 minutes. Anti-TH (ab112, 0.15 mg/mL; Abcam, Cambridge, Massachusetts) was incubated at a dilution of 1:5000, 0.03 µg/mL for 60 minutes at room temperature and detected by applying a Rabbit-on-Rodent HRP Polymer (Biocare Medical) for 30 minutes followed by visualization with the diaminobenzidine (Dako, Carpinteria, California) for 5 minutes. The slides were then removed from the IntelliPATH and counterstained with Mayer hematoxylin (Dako), dehydrated, and mounted with a permanent mounting medium.
Image Analysis
Hematoxylin and eosin sections
Microscopic examinations were conducted on one H&E-stained longitudinal section (section #3) from the right-side SCG of each animal. Changes were graded as minimal, mild, moderate, or marked by a board-certified veterinary pathologist. The total number of ganglia scored per treatment group is shown in Table 1.
Tyrosine hydroxylase sections
Tyrosine hydroxylase IHC slides were scanned on the Leica/Aperio AT2 whole slide digital scanner (Leica Biosystems, Vista, California) using the 40× magnification setting. Images were saved in .svs format and stored using Aperio eSlide Manager Digital Pathology Management Software (Leica Biosystems Inc, Buffalo Grove, Illinois). Digital images of SCG were opened in Aperio ImageScope software (Leica Biosystems Inc). One longitudinal section of SCG (section 6 or 7) was analyzed per animal. Manual outlines of the SCG (blue) and the neurons (red) were drawn (Figure 1A). A neuron was defined as a cell with TH-positive cytoplasm containing at least 1 distinct nucleolus within a nucleus. These criteria were consistently applied to all neurons manually outlined (Figure 1B). Quantitative image analysis was performed on the TH immunostained sections from control and guanethidine-treated samples at the 3- and 6-month recovery times. HALO image analysis (Indica Labs, Corrales, New Mexico) software was used to export the area of SCG (square millimeter of SCG area), the area of the neurons that are TH-positive (neuron profile area), and the total number of circled neurons per SCG sectional area (neuron count) for each image to a Microsoft Excel (Redmond, Washington) spreadsheet. To evaluate whether the number of neurons per cross section of SCG were different between the groups, total numbers of TH-positive neurons per square millimeter of SCG sectional area were calculated. To evaluate whether the average cross-sectional area of each neuron profile (as an indication of neuronal size) was different between the groups, the mean TH-positive neuron profile area (µm2) per animal was calculated. The total number of ganglia analyzed per treatment group is shown in Table 1.
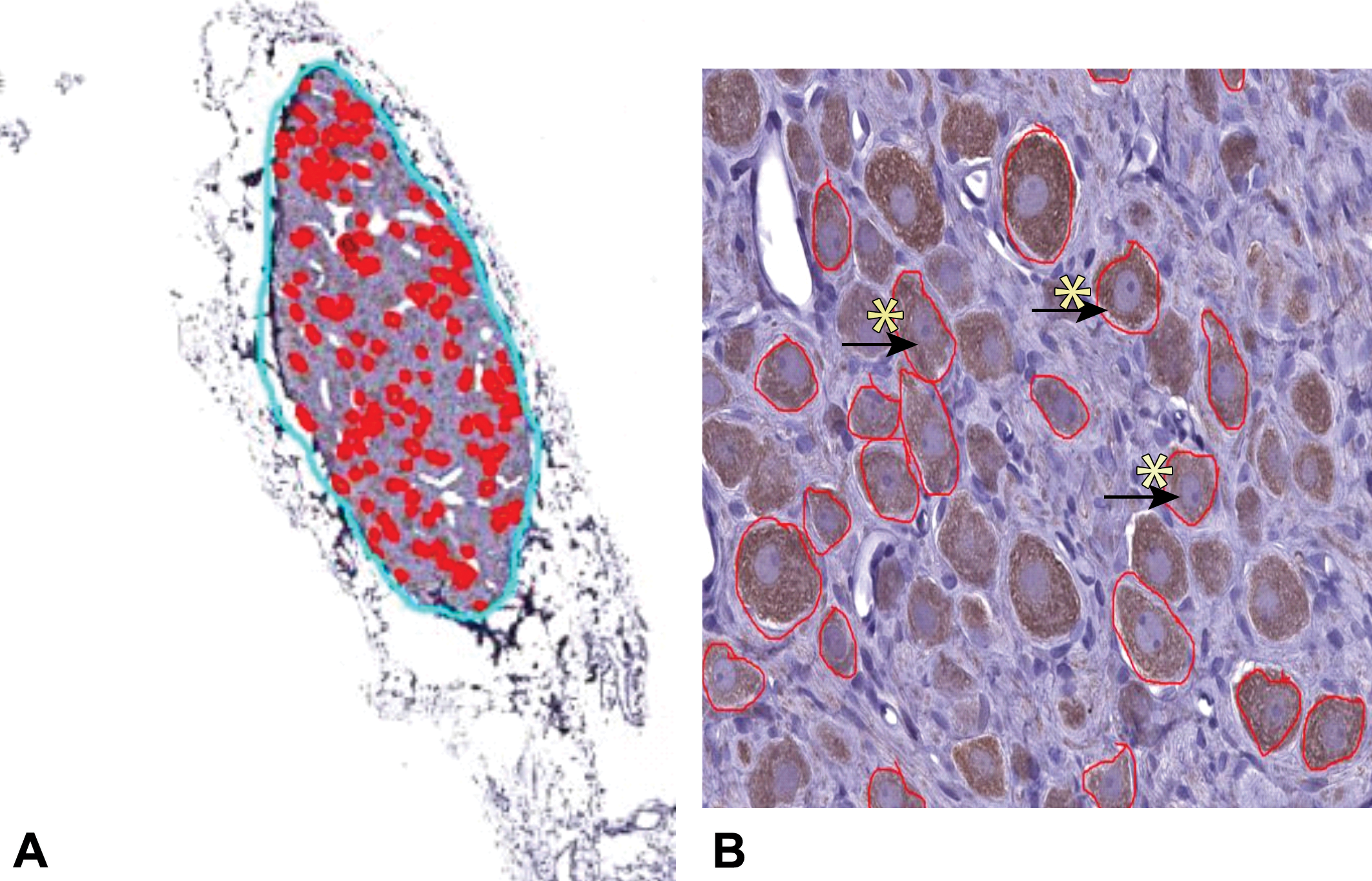
Morphometric methods for TH-stained sections of rat SCG. One longitudinal section of SCG (as per Table 1) was analyzed per animal. A manual outline of neurons was performed in ImageScope viewing software (A). To consistently measure neurons in a central plane of section, we also identified and manually outlined only neurons (red circles) that met the following criteria: A cell with TH-positive cytoplasm (yellow stars) containing at least 1 distinct nucleolus (arrows) within a nucleus (B). SCG = superior cervical ganglia; TH = tyrosine hydroxylase.
Stereology
Tissue processing
Left-side SCGs were dehydrated through graded alcohols and xylenes, and then manually embedded in paraffin. Serial sections (20 μm) were cut transversely through the entire SCG from cranial to caudal direction (tower orientation). Systematic-random sections of every
Stereology analyses
An initial pilot study was performed to determine optimal sampling parameters to achieve mean coefficient of error per group between 0.05 and 0.15.
26
Total volume (V) was estimated for each ganglion by the unbiased Cavalieri principle.
27
Total number of neurons in each ganglion (N) was estimated by the unbiased optical fractionator.
28
These studies were done with assistance from an integrated computerized stereology microscope system (
Statistics
For each end point, a 2-way analysis of variance model was fit, with treatment, time, and their 2-way interaction included as factors. A log transformation was used to stabilize the variance and compare geometric means. Follow-up pairwise comparisons were conducted using prespecified contrast statements. For each comparison, an estimate (stated as a % change between 2 groups, based on geometric means) and accompanying 95% confidence interval (CI) were computed, in addition to a
Results
In-Life Observations
Of the 30 rats treated with high-dose (100 mg/kg/d) guanethidine, 2 from group 4 (3-month time point) and 1 from group 6 (6-month time point) died during the dosing phase of the study. The rat from group 6 died immediately after the first dose, possibly due to a dosing error and was replaced on day 2; the replacement rat (#61) received 10 (rather than 11) daily doses of guanethidine. A fourth rat (from group 6) died during the recovery phase, and no cause of death could be determined. These 4 animals were not examined further. Clinical signs in surviving animals included partially closed eyes, decreased skin turgor, soft feces, and decreased activity. Several treated animals also exhibited lesions in the abdominal area likely related to IP dosing of guanethidine sulfate. Some of the lesions were treated with a topical antibacterial cream. There was a decrease in food consumption (0.60-0.65× controls) and body weights (0.89-0.91× mean day 1) in guanethidine-treated animals during the dosing phase. During the recovery phase, body weights of guanethidine treated rats returned to control values by approximately 3 months.
Morphometry
Due to a tissue processor malfunction, right-side ganglia from the 1-month time point (groups 1 and 2) were not suitable for morphometric analysis. However, the 1-month time point was assessed by stereology in left-side ganglia (see below). Additionally, due to early animal death (described above) or loss of tissue during collection, only 7 and 8 ganglia from the guanethidine-treated groups were evaluated at 3 and 6 months of recovery, respectively, and 9 ganglia were evaluated from the vehicle-treated rats at 6 months. The complete set of 10 ganglia was evaluated from vehicle-treated animals at 3 months. Hematoxylin and eosin–stained sections of ganglia from guanethidine-treated rats showed a diffuse, minimal-to-mild decrease in neuron density at both 3 and 6 months of recovery when compared to time matched controls (Figure 2). Occasionally, a minimal increase in cellularity of the stroma was observed. Infiltration of mononuclear cells was seen in both control and guanethidine-treated groups and was therefore not considered related to guanethidine treatment. Ganglia sections from the 3- and 6-month groups looked morphologically similar (Figure 2).
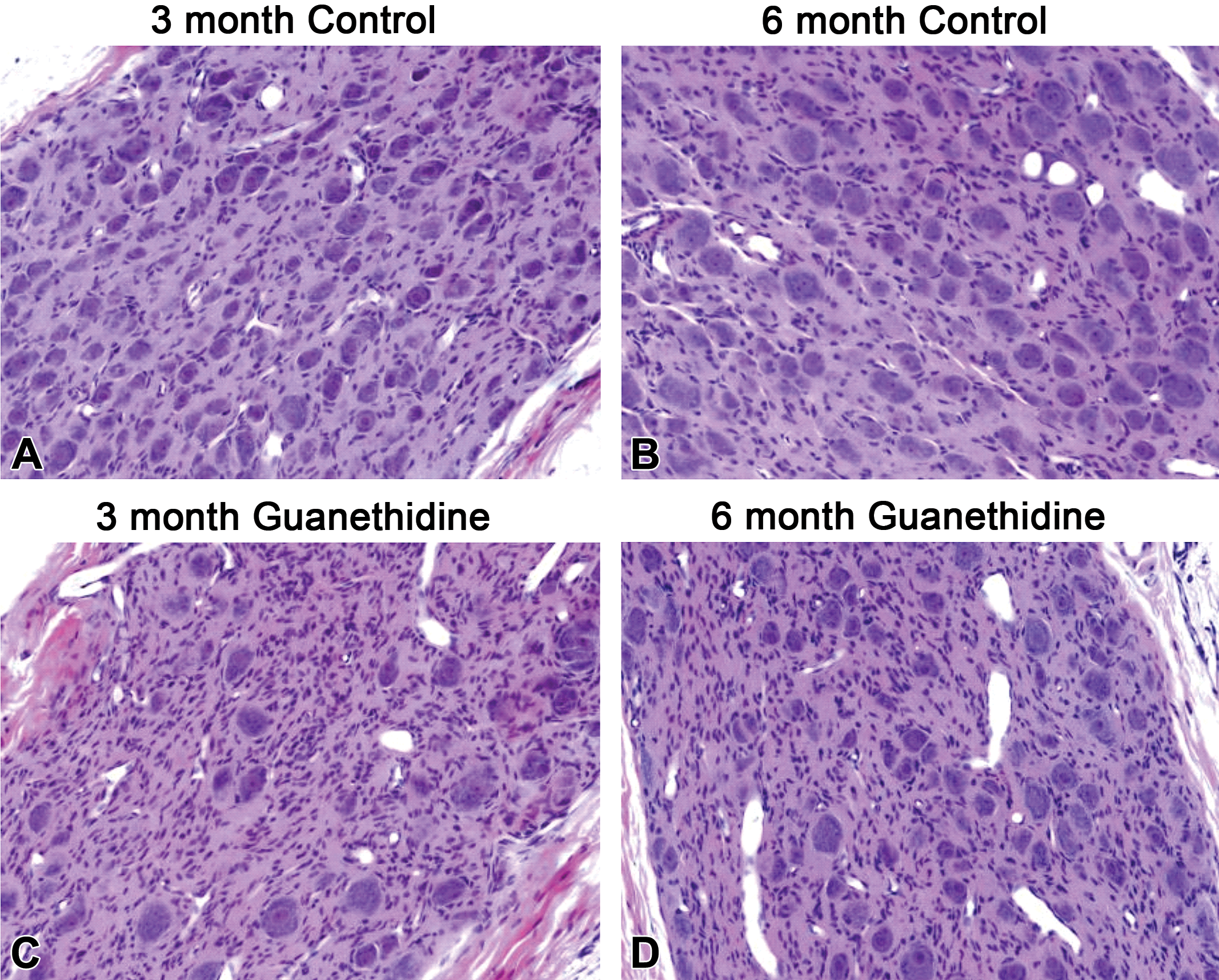
Representative images of H&E-stained sections of rat SCG. Diffuse minimal to mild, decrease in neuronal density can be seen comparing vehicle control (A, B) to guanethidine (C, D) treated images at both 3 (A, C) and 6 (B, D) months. SCG = superior cervical ganglia.
Following immunostaining for TH, the rate-limiting enzyme in catecholamine synthesis and a marker for noradrenaline-containing SCG neurons, a clear decrease in the density of TH-positive neurons was observed in sections from ganglia of guanethidine-treated animals (Figure 3A and B). In vehicle-treated rats, quantitative image analysis of the TH immunostained sections showed a mean neuron count/square millimeter of SCG area of 179 ± 12.0 and 171 ± 26.4 neurons/mm2 (± SEM) at 3 and 6 months, respectively. In guanethidine-treated rats, neuron density was reduced to 85.0 ± 10.9 and 71.6 ± 5.8 neurons/mm2 at 3 and 6 months, respectively. Statistical analysis of log-transformed data indicated a significant effect of treatment (
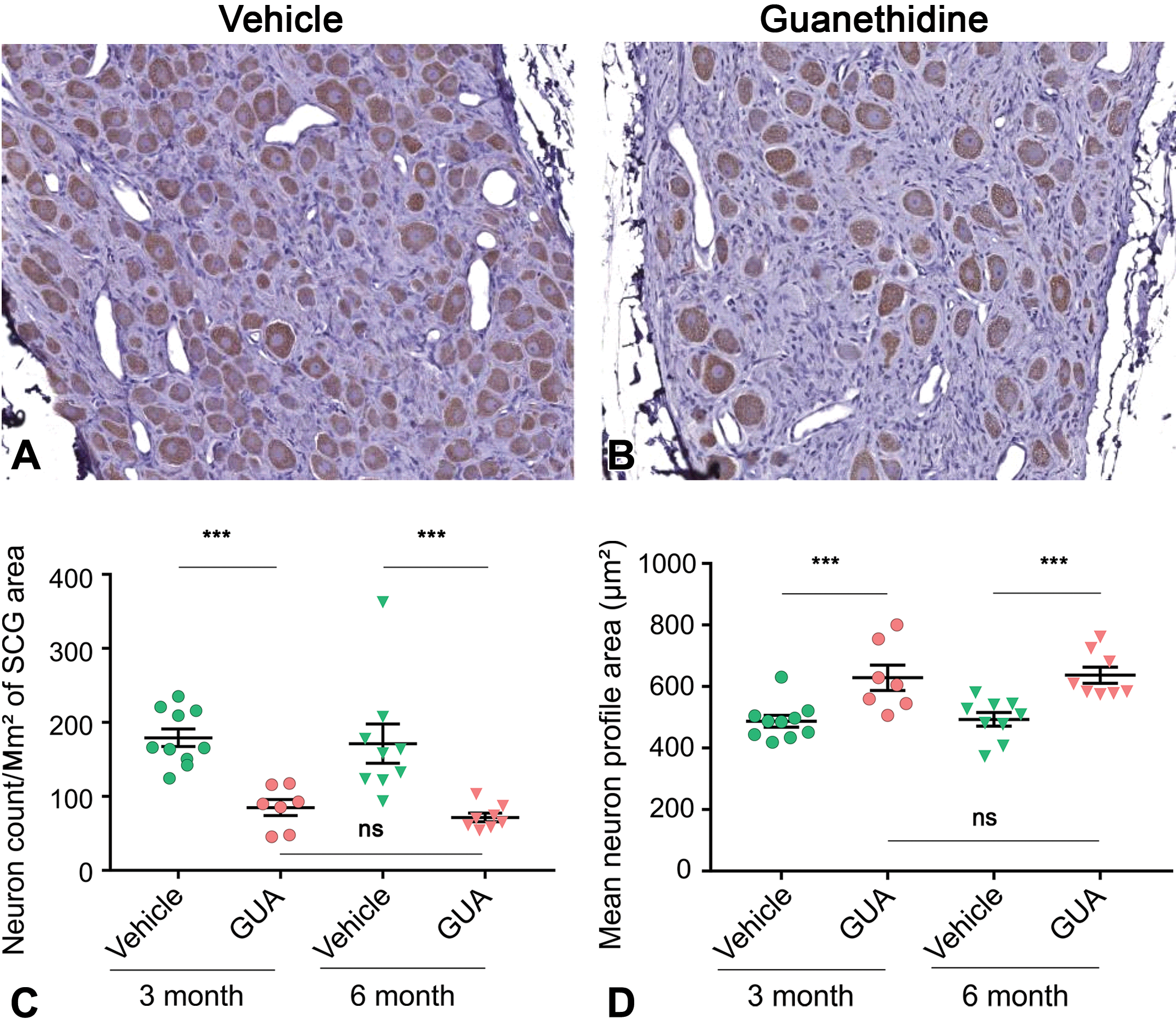
Morphometric evaluation of TH-stained sections of rat SCG. Neurons staining positive for tyrosine hydroxylase (TH) are brown. There are more TH-positive neurons in the SCG sections from a control rat (A) compared with a representative section from a guanethidine-treated rat after 6 months recovery (B). Following 11 daily doses of 100 mg/kg guanethidine, there was a statistically significant (
Stereology
Neuron numbers were estimated in left-side ganglia collected from vehicle- and guanethidine-treated rats using unbiased stereology. The total number of left-side ganglia evaluated from treated rats was 27, since 3 animals were lost and not replaced during the dosing phase. The number of left-side ganglia evaluated from control rats was 29 because 1 ganglion volume was more the 5 standard deviations below the group mean of the remaining cohort (6 month vehicle-treated group) and was excluded. In vehicle-treated rats, neuron counts were 48 146 ± 7701, 43 529 ± 2430, and 48 867 ± 4833 neurons at 1, 3, and 6 months, respectively. In guanethidine-treated rats, these numbers were reduced to 39 789 ± 3344, 30 582 ± 4933, and 37 207 ± 3171 neurons at 1, 3, and 6 months, respectively. Statistical analysis of log-transformed data indicated a significant effect of treatment (
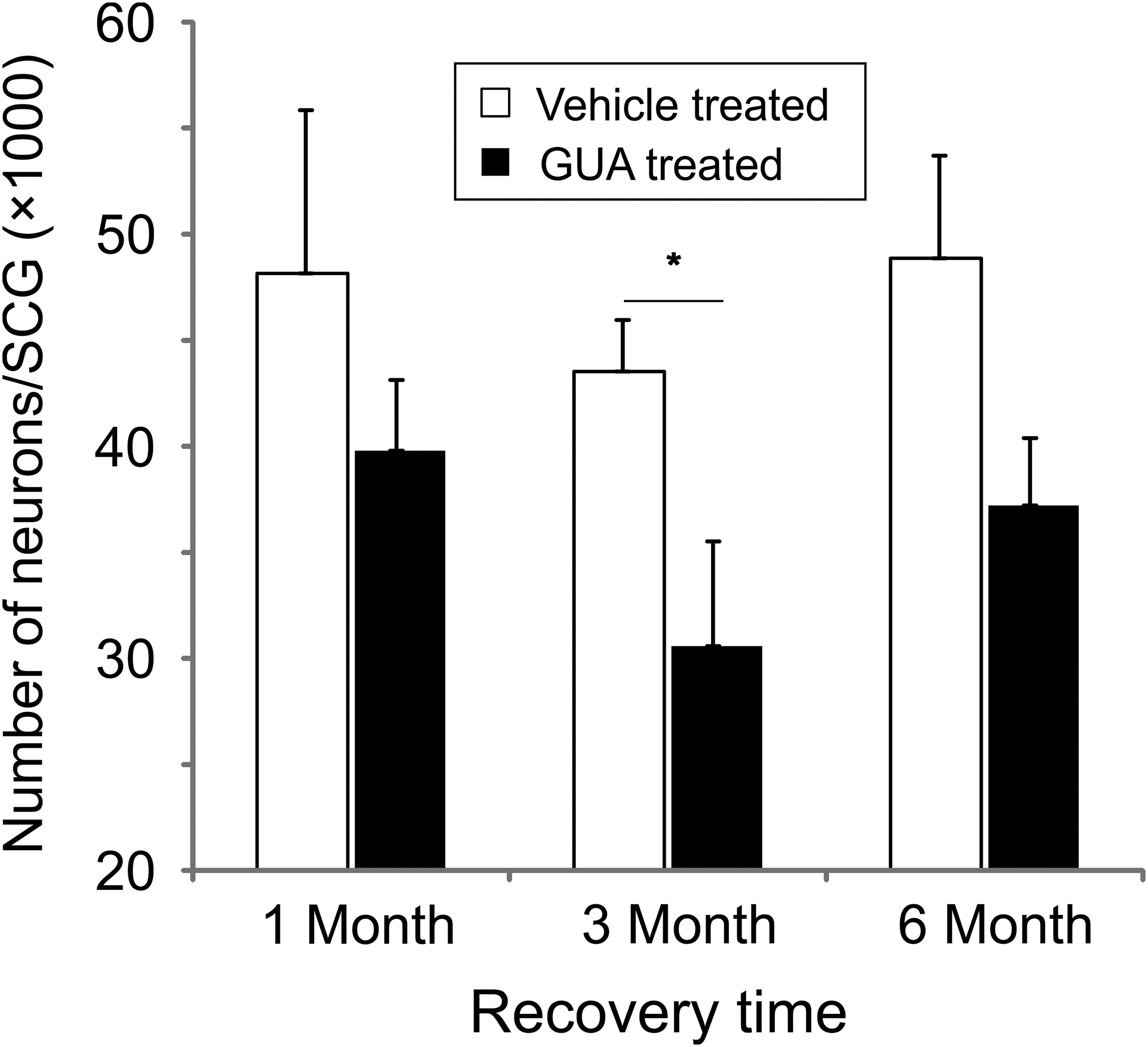
Stereology evaluation of H&E-stained sections of rat SCG. Neuron counts were significantly decreased following guanethidine treatment (
Discussion
In this study, we evaluated the potential for neurogenesis in the SCG of the rat. Eleven consecutive daily doses of guanethidine (100 mg/kg/d) were administered IP to rats in order to kill postganglionic sympathetic neurons as described previously. 22 -24 Following the last dose, animals were allowed to recover for 1, 3, or 6 months. These time points were chosen based on studies in sensory ganglia 14,15 in which peak neuron losses following chemical treatment were observed at 1 month with recovery by 2 to 3 months. Right and left SCG from guanethidine-treated and age-matched, vehicle-treated rats were harvested for histopathologic, morphometric, and stereologic evaluations. Both morphometric and stereologic evaluations confirmed neuron loss following guanethidine treatment. Morphometric analysis revealed a 50% to 60% lower neuron count/square millimeter of SCG area of TH-positive neurons in SCG at both 3 and 6 months of recovery when compared to ganglia from age-matched, vehicle-treated controls, with no evidence of restoration of neuron density between 3 and 6 months. Remaining TH-positive neurons had a larger mean neuron profile area than concurrent vehicle controls, consistent with previous reports. 24 The reason for this finding is unknown but could indicate a compensatory response of surviving neurons or a bias of guanethidine toward killing smaller neurons. Reductions in TH-positive neuron count/square millimeter of SCG area were corroborated by unbiased stereology of total H&E-stained neuron numbers in SCG. Stereologic analyses revealed that total neuron counts were lower by 37% when compared to counts from concurrent controls at 3 months, again with no clear evidence of number restoration by 6 months.
The reductions in neuron counts estimated using unbiased stereology in the present work were less than observed previously in our lab with this model. 23,24 Previously, guanethidine treatment of 100 mg/kg/d for 11 days resulted in a 50% to 60% reduction in total H&E-stained neurons in the SCG after a 1-month recovery 24 compared to 7.4% (95% CI: −32 to 35) reduction in this study. Additionally, our previous estimate of total neuron numbers within the SCG of vehicle-treated rats was ∼30 000 neurons compared to ∼45 000 estimated in this study. The reason for these discrepancies in stereology-based statistical estimates is unclear. Animal age, strain, and gender were unchanged from earlier work, as were the dose and dosing regimen, as well as the methods used for SCG dissection and harvesting. Notably in past studies, the coefficient of variation (CV) was below 0.3 (30%), while in the current study these CVs ranged to as high as 0.52 (52%) in the 1-month vehicle-treated group. This increased CV may reflect a change in the quality of ganglion dissections and trimming in the current study. In fact, one ganglion volume was more the 5 standard deviations below the group mean of the remaining cohort (6-month vehicle-treated group), and the neuron count for this ganglion was removed from our statistical analyses as a consequence. In this case, it appeared that a complete, intact ganglion was not acquired for analysis. Such variability may have affected neuron count and ganglion volume estimates in the current study and highlights a critical aspect of any stereology-based estimation of quantitative data such as neuron counts, namely, the need for consistent harvesting and counting in a clear volume of interest. That being said, a known concern for stereology analyses is the need for careful tissue harvesting to ensure study repeatability.
Both morphometric and stereologic assessments of the SCG from guanethidine-treated rats revealed a loss of neurons that was not restored after 3 to 6 months of recovery. The morphometric analyses of right-side ganglia revealed a reduction in the TH-positive neuron count/square millimeter of SCG area of 50% to 60%. This reduction in neuron density, and lack of recovery following guanethidine treatment, was corroborated by our unbiased stereology assessments of the left-side ganglia. The difference in magnitude of apparent neuron loss when comparing the 2 assessment methods may reflect, in part, the different types of neurons being counted; in the case of the morphometric assessment, the counts were an estimate of TH-positive neurons, while in the case of the stereology assessments the neurons counted were an estimate of total number of neurons in the SCG. Given that catecholamine-containing neurons represent only about 80% of the total neurons present within the SCG 30 and that guanethidine affects only catecholamine (noradrenergic) neurons, 21,22 it makes sense that the effect would be somewhat larger when focused on just TH-positive neurons as done in the morphometric analysis. It is also important to keep in mind that our morphometric assessment quantifies changes in density (neurons per unit area), and differential shrinkage between control and injured SCG 31 could account in part for the difference in magnitude of apparent neuron loss when comparing the 2 assessment methods. Finally, quantitative differences in neuron loss estimates between the 2 assessments are unlikely to reflect the different SCG sides used in each assessment. Although some ganglia in the sympathetic chain may be more sensitive to injury than others, 32 there are no reports in the literature, nor would it make sense from a toxicodynamic perspective, of an asymmetric (left vs right side) SCG response to systemically delivered guanethidine.
Evidence for neurogenic potential has been reported in the peripheral nervous system of adult rodents, including age-related increases in neuron numbers in aging rats, 11,12 the presence of neuronal progenitor cells in peripheral sensory, 13,14 and sympathetic 18,19 ganglia as well as reports of neuron replacement following chemical depletion in nodose 15,33 and dorsal root 14 ganglia. Given these reports, we hypothesized that the peripheral sympathetic nervous system of the adult rat may be capable of neurogenesis and therefore able to replace postganglionic neurons destroyed by high doses of guanethidine. However, when we quantified neurons 1, 3, and 6 months after guanethidine treatments and compared them against age-matched concurrent controls, we saw no clear evidence of neuron recovery following initial losses. We employed 2 independent techniques for this evaluation; a 2-D morphometric analysis and a 3-D unbiased stereology analysis and neither technique revealed neuron replacement over a 6-month recovery time period. Previous studies using capsaicin to kill neurons in sensory ganglia 14,15,33 showed neuron count recovery after 60 days. Therefore, if sympathetic neurons were capable of regenerating following guanethidine treatment, we believe we should have seen recovery in the SCG within the 3- to 6-month period examined.
It is possible that the methods and model used in the current study were not sufficiently sensitive to detect neurogenesis in the sympathetic ganglia. Progenitor cells have been reported in both sensory 13 and sympathetic 18 ganglia of adult animals. Slight age differences may be important. While the capsaicin sensory ganglia studies in which neurogenesis was observed 14,15,33 were conducted in sexually mature, adult rats (∼7 weeks at study start), our study used slightly older rats (13-14 weeks at study start). It has been reported that neurogenesis in the CNS in higher primates declines rapidly in juveniles and young adults. 7 It is also possible that guanethidine, administered at the high doses used here, destroys not only mature neurons in the SCG but also neuronal precursor cells, removing the SCG capacity for neurogenesis. However, this seems unlikely given that guanethidine killed only a portion of SCG neurons in this study and thus would be unlikely to knockout all progenitor cells capable of proliferation and neuron restoration. Additionally, in unpublished studies, we have observed nestin-positive satellite and stromal cells in SCG at the ∼1-month time point, suggesting a neurogenic potential remains at this time. It is more likely that the sympathetic portion of the autonomic nervous system of the adult rat does not have that same neurogenic capacity as the sensory portion of the peripheral nervous system. To the best of our knowledge, neurogenesis has never been demonstrated for the postganglionic neurons of the sympathetic nervous system of any adult species.
In the work described here, we hypothesized that postganglionic neuron loss in the ganglia of the sympathetic division of the autonomic nervous system may be replaced over time following destruction with high doses of the sympatholytic compound guanethidine. However, despite evaluating neuron restoration in the rat SCG 1, 3, and 6 months following treatment, neuron numbers were not restored to the levels of age-matched controls. This is in contrast to reports for neurons of the nodose and dorsal root sensory ganglia which are reported to recover to control levels within 60 days of destruction with capsaicin treatment. Thus, we found no compelling evidence that neurons of the sympathetic portion of the autonomic nervous system in the adult rat have a neurogenic capacity.
Footnotes
Acknowledgments
We would like to thank Drs Mike Mirsky and Carlin Okerberg for providing input and expertise, David Potter for statistical support, Sarah Anderson for QC of all raw data, Brad Hirakawa for QC of the manuscript, and Natalie Bajorin, Sarah Crowell, Laura Kearney, and Kim Kowsz for study technical support.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: All work described in this publication was sponsored and funded by Pfizer.
