Abstract
Osteoporosis is a very common skeletal disorder characterized by reduced bone mass and altered trabecular microarchitecture that leads to bone fragility and fractures. Such disease is due to alterations of the remodeling process that occurs in the basic multicellular units that are transitory cellular complexes including an osteoclastic phase (osteoclast activation and resorption of microscopic portions of bone), a reversion phase (osteoclast replacement by so-called postosteoclastic cells), and an osteoblastic phase (osteoblastic reconstruction of the resorbed bone matrix till the initial volume is regained). Bone remodeling is regulated by a number of systemic and local factors; among the former, besides physical activity and mechanical stresses, a primary role is played by hormones such as parathyroid hormone, vitamin D metabolites, estrogens, calcitonin, and glucocorticoids; among the latter, several growth factors (macrophage colony-stimulating factor, transforming growth factor β, platelet-derived growth factor, fibroblast growth factor 1, bone morphogenetic protein, and insulin-like growth factor 1), as well as the osteoprotegerin-receptor activator of nuclear factor-κ B ligand system and the sclerostin, play a primary function. The remodeling phases can be evaluated by static and dynamic histomorphometry. Their abnormalities may lead to several osteopathies, the most common of which is osteoporosis (above all senile and postmenopausal), a rather elusive disease chiefly due to its slow development. The use of animal models in its study is emphasized.
Introduction
Osteoporosis is one of the most frequent diseases in humans and one of the most expensive in economic terms, largely on account of its physical, psychological, and social consequences. It arises out of abnormal forms of bone remodeling that lead to a gradual fall in bone strength and to fractures. At least in its primary forms (postmenopausal and senile), osteoporosis develops clinically over years and often only becomes evident because of the pain and fractures experienced once the skeletal changes have reached a very advanced stage. This trend, and the difficulty of obtaining bone samples from otherwise healthy people, makes the study of the changes occurring in bone remodeling at their initial phase of development a demanding, if not impossible, task and severely hinders the understanding of the pathogenesis of the disease. This situation has led to the need to thoroughly examine bone remodeling and the osteoporotic changes that can be detected in bone biopsies by evaluating the static and dynamic histomorphometric variables in bone but also to assess the use of experimental animals as a source of bone samples showing the earliest spontaneous or induced osteoporotic changes. The present overview aims to inquire into the bone remodeling process and its possible effects on bone physiology and pathology, to examine bone histomorphometry as a means of studying bone remodeling phases together with the parallel changes in osteoporosis, and to evaluate the correctness of the use of experimental animals as a source of bone specimens with experimentally induced, early osteoporotic changes.
Bone Remodeling
The term “bone remodeling” refers to a process characterized by the demolition and subsequent reconstruction of microscopic portions of calcified bone matrix. This apparently useless process plays fundamental physiological roles, and its alteration can lead to severe pathological skeletal changes (reviewed by Del Fattore, Teti, and Rucci 2012).
Bone is not a stable, biologically inert tissue, as its solid, stone-like appearance might suggest; on the contrary, it fulfills several fundamental physiological functions, in some of which it plays only a passive role. For instance, the rigid osseous segments of the skeleton give rise, together with the articulations, to levers that permit movements and passively transmit the mechanical activity of muscles. Moreover, bone passively holds and protects soft tissues (e.g., the brain), while containing the bone marrow and its stem cells. It has an active role in other functions, which demands cell participation and mainly involves the bone matrix destruction–reconstruction cycle that characterizes bone remodeling. First, this process contributes to the maintenance of the structural integrity of the skeletal segments and to their renewal. Altered portions of mineralized matrix, such as fatigue lesions and microfractures, can, in fact, be removed and replaced with new bone tissue, so that, in a broad sense, the age of the skeleton does not reflect—more precisely, is younger than—that of the individual. The same destruction–reconstruction process can change and reorganize the microanatomy of the tissue (hence, the term bone remodeling) in such a way that its microstructures (chiefly the collagen fibrils) can better respond to changing mechanical demands.
Through the same process of bone remodeling, many extraneous, potentially dangerous, circulating substances (Pb, Al, tetracyclines, and radioactive isotopes) can be removed from the bloodstream by their incorporation in, and consequent neutralization by, the mineralized matrix during the reconstruction phases, to be gradually released and eliminated during the destruction phases. Moreover, the demolition of the mineralized matrix and its subsequent reconstruction induce the release of ions, such as calcium and phosphorus, into the bloodstream, and they later recapture from it, respectively, so contributing to the regulation of two vital parameters—specifically calcemia and phosphoremia. Bone remodeling is thus a process that not only deeply influences skeletal physiology but also markedly affects the whole organism. It occurs at the level of what are known as “basic multicellular units (BMUs).”
Basic Multicellular Units
BMUs are transitory functional entities that develop in multiple microscopic areas of the bone surface (BS) and imply a succession of different cell activities, inducing first the resorption and then the reconstruction of a quantum of bone matrix (Bonucci 1990a; Parfitt 1979; Figure 1). A BMU is, therefore, characterized by the succession of different cellular types. To be able to develop on a steady BS, it initially requires the activation of one or several osteoclasts (phase of activation) that induce the resorption of a quantum of bone matrix, so producing a resorption lacuna, otherwise called Howship’s lacuna, in trabecular bone, or a cutting cone, in compact bone (phase of resorption). When this phase is complete, all osteoclasts disappear and are replaced by other cells that are functionally not yet fully defined and for this reason are called “postosteoclastic cells” (phase of reversion). At the end of this phase, these postosteoclastic cells disappear and are replaced by active osteoblasts (phase of ossification) that synthesize as much bone matrix as that previously resorbed by osteoclasts. At the end of the process, therefore, the osseous surface gains its initial volume with the activity of osteoclasts being coupled with that of osteoblasts in such a way that the degree of bone resorption is balanced by the degree of bone formation. The new bone synthesized by osteoblasts, that is, the end product of each BMU, corresponds to the bone stuctural unit (BSU) that can be viewed, using polarized light, as a quantum of bone with a parallel (trabecular bone) or concentrical (cortical bone) lamellar orientation bounded on one side by a thin cement line, which points to the limit of Howship’s lacuna (trabecular bone) or cutting cone (cortical bone), and on the other by a quiescent BS. The microanatomy of the rebuilt bone may, however, be changed if the area has been impacted by new but different mechanical forces.
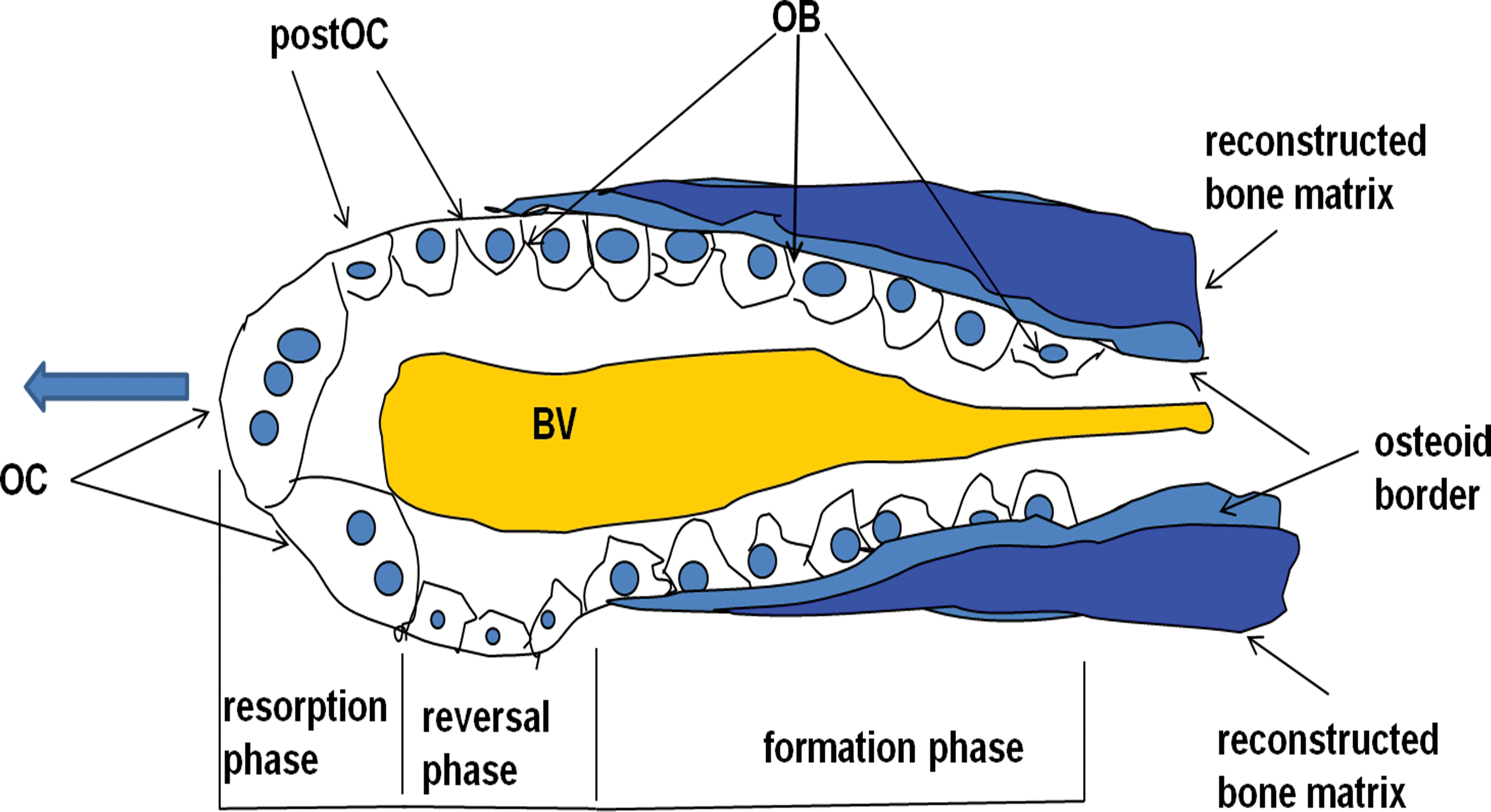
Schematic representation of a basic multicellular unit of compact bone (so-called cutting cone). The light blue arrow points to the direction of the osteoclastic resorption. OC = osteoclasts; post-OC = postosteoclasts; OB = osteoblasts; BV = blood vessel.
The BMU Activation Phase
Characterized as it is by the differentiation and development of one or many osteoclasts, this phase can occur in every segment of the skeleton, although it develops preferentially on the endosteal surface of the spongy bone, probably because the numerous blood vessels present in this type of tissue facilitate cell proliferation and differentiation. BMUs are, however, also activated in compact bone, either on the subperiosteal surface or in the context of the cortex, where osteoclasts resorb the mineralized matrix according to a tunnel-like cavity (the cutting cone).
Many different factors induce the activation phase and the development of a BMU; they can be divided into systemic (i.e., those that affect the whole skeleton) and local factors. The former are mainly related to the hormones that regulate the blood calcium concentration (see section on the BMU Resorption Phase) and to the physical activity of the individual, while the latter include both cellular products (growth factors) and structural microscopic changes. These latter factors are probably directly responsible for the location of a BMU in a specific microscopic area of a bone segment rather than in another one. They are, however, poorly known. Microdamage to cells or to calcified matrix might induce the cellular reactions that are at the basis of bone remodeling (Mulcahy et al. 2011). Changes in osteocyte integrity (apoptosis) associated with osseous microdamage can stimulate osteoclast activity (Verborgt, Gibson, and Schaffler 2000). The osteocyte is the cell that transduces normal mechanical stimuli, which are indispensable for the maintenance of skeletal integrity, into factors that stimulate bone cell activity (see the section on Osteocytes and Bone Remodeling). They can, therefore, initiate the physiological cycle of any BMU. However, BMU is a highly coordinated process that can involve a number of different, still poorly known activation factors.
The numbers of BMUs that may be active in the skeleton at any given time obviously depend on the intensity of the activation stimuli but also on that of the maintaining factors, which in their turn are responsible for the life span of BMUs. The duration of the various BMU phases is still only partly known and can change with the stimuli that cause BMU activation and maintenance (Ballanti et al. 2001); the mean normal values are reported in the section on Bone Histomorphometric Variables. The speed of advancement of the cutting cone through the compact bone of the ribs was 43.61 µm/day in normal dogs and 27.97 µm/day in uremic dogs (Jaworski, Lok, and Wellington 1975).
The BMU Resorption Phase
This phase of the BMU is distinguished by the osteoclastic resorption of a microscopic portion of bone matrix, with the formation of a Howship’s lacuna (Figure 2A). One or many osteoclasts can be active in the same place, and their number, as well as the intensity and duration of their activity, depends on systemic and local stimuli (reviewed by Mellis et al. 2011). The former are mainly connected with the regulation of calcemia. It has long been known that the Ca ion concentration in the blood is maintained constant above all through the equilibrium between intestinal absorption and urinary excretion of calcium. In addition, its value can be increased or reduced by the resorption or deposition, respectively, of the calcified bone matrix. The resorption of the hydroxyapatite-like inorganic substance leads to its dissolution into calcium and phosphate ions that penetrate the bloodstream and increase calcemia and phosphoremia. On the other hand, the formation of new bone implies the calcification of its matrix and a consequent reduction in the calcium concentration within blood.
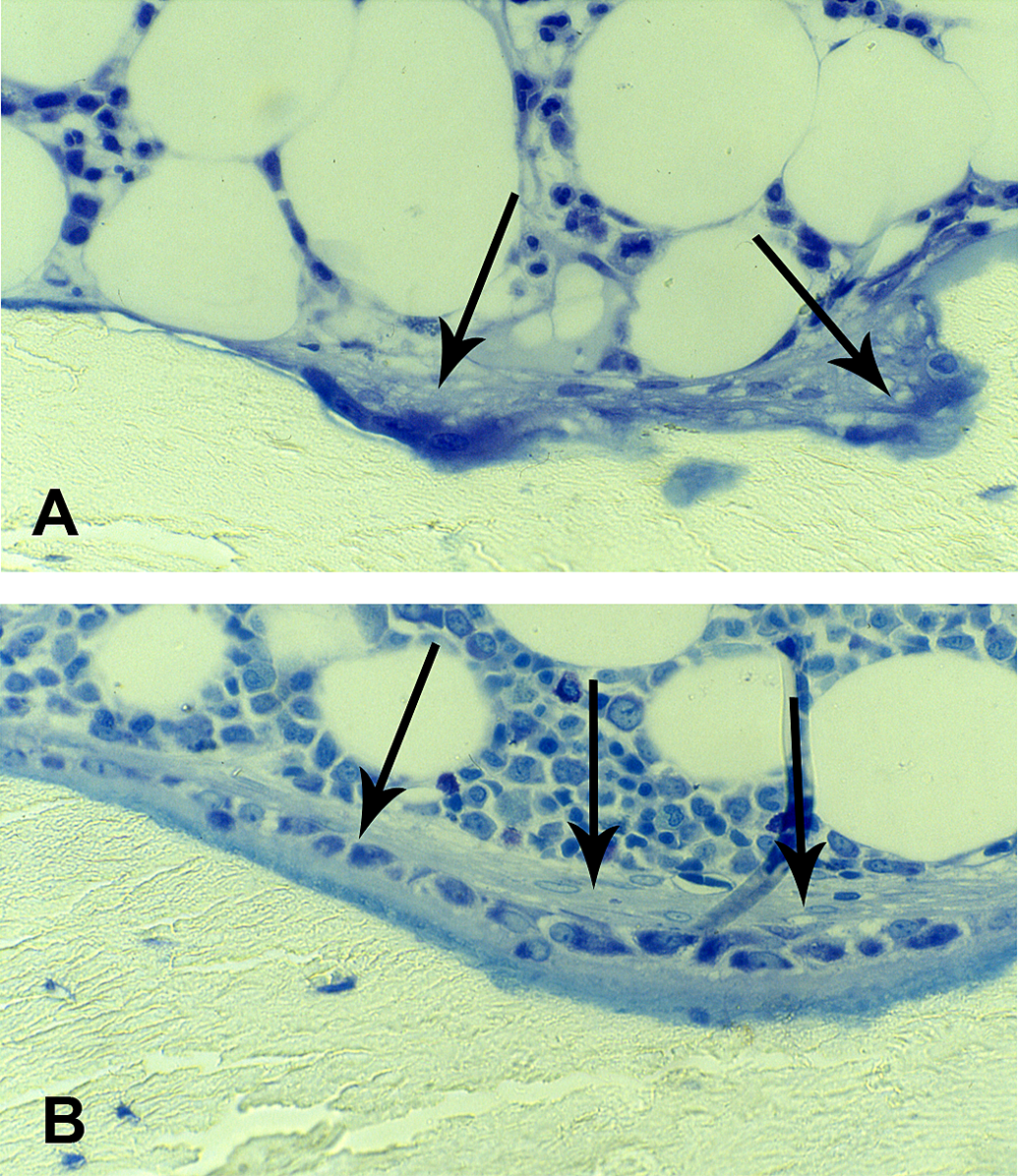
Human trabecular bone. (A) A Howship’s lacuna with osteoclasts (arrows). (B) An osteoid seam lined by osteoblasts (arrows). Undecalcified, 2-µm thick sections stained by methylene blue—azure II; original magnifications ×40.
The stimulation of osteoclast activity is mainly due to the parathyroid hormone (PTH) and 1,25-dihydroxy-vitamin D3 (1,25(OH)2D3) and the inhibition to calcitonin. Other factors, like cytokines, especially interleukin (IL) 1 and IL-6, and growth factors, especially the transforming growth factor β (TGF-β), the bone morphogenetic protein (BMP), and the insulin-like growth factor (IGF), can have either a systemic or a local stimulating effect, or both. It is important to note that osteoclast differentiation is regulated by the osteoprotegerin (OPG) system. The preosteoclasts of the bone marrow express receptor activator of nuclear factor-κB (RANK), an intrinsic hematopoietic cell surface receptor whose interaction with its ligand (RANKL) promotes, in the presence of the macrophage colony-stimulating factor, the differentiation and development of preosteoclasts into osteoclasts; OPG, a glycoprotein of the TNF receptor superfamily produced by osteoblasts and marrow stromal cells, functions as a soluble decoy receptor for RANKL, so preventing its binding to RANK and inhibiting osteoclastogenesis (reviewed by Aubin and Bonnelye 2000; Boyce and Xing 2008; Kostenuik and Shalhoub 2001).
The BMU Reversal Phase
The resorption phase has a variable but limited duration, depending on the persistence or decay of the stimuli that induce osteoclast differentiation and activity. It is followed by the reversal phase that is characterized by the disappearance of all osteoclasts, their substitution by postosteoclastic cells, and the synthesis of the “reversal” or “cementing” line. Postosteoclasts are mononuclear cells that lack osteoclastic characteristics; in particular, they do not show a ruffled border or any endocytosis of inorganic particles. An electron microscope study by Domon et al. (2001) showed that, before the formation of postosteoclastic cells, the surface of Howship’s lacunae in cultured dentine slices was coated by exposed, sharply pointed collagen fibrils but not by cells or by the reversal (or cementing) line. Conversely, after the formation of postosteoclasts, the resorption surfaces in rat mandibles showed no collagen fibrils but an evident reversal line. The authors conclude that postosteoclastic cells remove the residues of degraded and disrupted matrix that may remain in the resorption lacunae after the disappearance of osteoclasts and produce the reversal line on the lacunar surface.
The resorption phase may come to an end simply because the stimuli that originally generated it have come to an end, too. In any case, some local factor is probably active in determining the event, because otherwise all BMUs would reach the reversal phase at the same moment. Starting from the observation that the bone matrix is a store of immobilized growth factors, and that these are mobilized during bone resorption in amounts proportional to the intensity of osteoclastic activity, it has been suggested that endocytosis by osteoclasts of these mobilized components might constitute, once an adequate intracellular concentration is reached, the signal that ends osteoclast activity (discussed by Baron, Tran Van, and Vignery 1982; Zhao 2012). The IGF could be one of these factors (Hayden, Mohan, and Baylink 1995).
The BMU Formation Phase
This phase is marked by the disappearance of postosteoclastic cells and the development of osteoblasts in their place (Figure 2B). Osteoblasts are large, polygonal, mononucleated cells whose cytoplasm is very rich in granular endoplasmic reticulum, which makes them strongly basophilic. Moreover, active osteoblasts develop a strong alkaline phosphatase activity. They form a continuous layer first on the “cementing” line, then on the osteoid tissue that they themselves have synthesized. They are therefore responsible both for the synthesis of the bone matrix that is going to fill the Howship’s lacuna and for its mineralization.
The differentiation and activity of osteoblasts are regulated by numerous factors that, as in the case of osteoclasts, can be separated into systemic and local ones. Among the former, estrogens are the major hormonal regulator of the bone metabolism; they have a direct inhibitory effect on osteoclasts but a stimulatory one on osteoblasts, so that they contribute to the maintenance of bone formation (reviewed by Khosla, Oursler, and Monroe 2012). Glucocorticoids, on the other hand, have an inhibitory effect on osteoblast activity and bone matrix synthesis (reviewed by Canalis 1996; Reid 1997), and their administration induces rapid bone loss (Lo Cascio et al. 1995). Among the local factors, the components of the BMP family (comprising at least 15 morphogens involved in intracellular messages), which are members of the TGF-β superfamily, play a critical role in the differentiation of osteoblasts from mesenchymal cells (reviewed by Wan and Cao 2005; Zuo et al. 2012). The BMPs’ intracellular signals (and those pertinent to other members of TGF-β superfamily) are transduced from the cell membrane to the nucleus via specific type I and type II receptors and Smad proteins and are regulated by other factors (Osterix, Runx-2) that are essential to the induction of osteoblast differentiation (Katagiri and Takahashi 2002; Miyazono 1999; Sakou et al. 1999; Wan and Cao 2005). The IGF and the platelet-derived growth factor are other local factors that stimulate osteoblast differentiation and activity (Ogino et al. 2006).
The differentiation of mesenchymal cells toward mature osteoblasts is induced by Wnt proteins, too. This large family of growth factors mediates fundamental biological processes related to cellular growth, differentiation, function, and death (reviewed by Bodine 2008; Piters, Boudin, and Van Hul 2008). They bind to membrane receptors consisting of frizzled protein (Fz) and low-density lipoprotein receptor–related proteins 5 (LRP5), so initiating a number of intracellular signaling cascades that lead to the inhibition of the phosphorylation of β-catenin by glycogen synthase kinase 3β. As a consequence, β-catenin is stabilized and can enter the nucleus of mesenchymal cells, where it activates specific transcription factors that induce osteoblastic differentiation (canonical Wnt/β-catenin pathway). The binding of Wnt with Fz and LRP5 is moderated and regulated by the competing cofactors frizzled-related proteins and Dickkopf1. Moreover, it has been shown that the Wnt/β-catenin pathway can be controlled through the production by loaded osteocytes either of sclerostin (encoded by the SOST gene), which has an inhibitory function by linking to LRP5, or of prostaglandin E2 (PGE2), which can induce cyclic adenosine monophosphate (cAMP)-dependent protein kinase A (PKA) activation and the stabilization of β-catenin (see the section on Osteocytes and Bone Remodeling).
Osteocytes and Bone Remodeling
Until recently osteocytes were considered as cells that, being embedded within the mineralized bone matrix, have a low level of activity. One possible osteocytic function is referred to as “periosteocytic osteolysis,” that is, an osteoclast-like resorption confined to the border of the osteocytic lacunae (reviewed by Bonucci 1990b). Criticized as being a misinterpretation of the fact that in primary bone, the osteocyte lacunae are normally enlarged with respect to those found in secondary bone, as well as being a misreading of the possibility that lacunae may appear enlarged when the matrix is poorly calcified (Marotti et al. 1990), periosteocytic osteolysis has recently received new support (reviewed by Bonucci 2009; Teti and Zallone 2009). There are now several proofs that osteocytes are not merely “static lacunar-dwelling cells” but are metabolically very active, so much so that they have been defined as the “underestimated conductors of the bone orchestra” (Bonucci 2009), a role that mainly includes a reaction to load and other forms of physiological stimulation.
It has long been known that physical activity is indispensable to maintenance of bone mass and that the lack of skeletal loads (immobilization, prolonged bed rest, and lack of gravity) induces osteopenia or even osteoporosis. Mechanical forces, therefore, are able to modify BMU activity (Frost 1987). This consolidated concept is not corroborated by any precise knowledge about the way strain is transduced into cell stimulation or about what type of cell, if any, is able to function as the mechanosensory cell of bone. In this connection, the best candidates are osteocytes because of their high numbers in the bone matrix and the numerous connections they establish with one another and with the other bone cells through their many processes. The mechanical deformation of cell lacunae and the consequent variations in intralacunar hydrodynamic pressure, as well as the shear stress generated by fluid flow in their canaliculi (Anderson et al. 2005), may well be the stimuli that enable osteocytes to convert mechanical energy into biochemical signals. Reviewing this topic, Civitelli (2008) points to the possibility that cell–cell interaction occurs by cell–cell communication via gap junctions, that is, via transmembrane channels that permit cytoplasmic continuity between communicating cells. Moreover, osteocytes can express functional superficial hemichannels (halves of gap junctions) that permit cell–matrix communication. Both channels and hemichannels consist of connexins (Cx), whose main component, called Cx43, might be the transducer of the mechanical signal, because its expression increases proportionally with mechanical strain (see Bonucci 2009) and its deletion attenuates the loss of trabecular bone formation during unloading (Lloyd et al. 2012). The osteocyte reaction to stress also includes the production of nitric oxide and PGE2. This can play a primary role in stimulating osteoblast differentiation, because through the PGE2 receptor EP2 subtype it can activate the cAMP-dependent PKA signaling (Cherian et al. 2003), which itself leads to the same results as the canonical Wnt/β-catenin pathway, that is, the inhibition of phosphorylation of β-catenin, its stabilization, and its translocation into the nucleus (reviewed by Bonewald and Johnson 2008).
Osteocytes take part in bone remodeling not only through the activation of the Wnt/β-catenin and cAMP/PKA pathways but also through the production of various substances, the most interesting of which appears to be sclerostin. Secreted as it is by old osteocytes, sclerostin binds to LRP5, so antagonizing the canonical Wnt/β-catenin pathway and inhibiting osteoblast differentiation (Bonewald and Johnson 2008; Piters, Boudin, and Van Hul 2008; ten Dijke et al. 2008). Its production is reduced by skeletal loading and increased by unloading (Robling et al. 2010), thereby representing a point of contact between physical activity and bone physiology. Its effects are controlled by PTH, which inhibits SOST transcription in vivo and in vitro (Bellido, Saini, and Pajevic 2013).
Coupling of Bone Resorption With Bone Formation
The maintenance of skeletal integrity requires a perfect balance between the destructive activity of osteoclasts and the reparative function of osteoblasts. The coupling between these processes presupposes an intimate form of cross-talk between the cells and the existence of autocrine/paracrine mechanisms of interaction that, however, still need to be defined. This lack of knowledge prompts the hypothesis of the existence of osteoclast-derived “coupling factors” that include the numerous signaling substances reported previously as well as molecules released from the resorbed bone matrix (BMP and IGF). Moreover, the interaction between membrane-bound ephrin ligands (Ephrin B2) expressed in osteoclasts and Eph tyrosine kinase receptors (EphB4) expressed in osteoblast precursors could constitute bidirectional signals that facilitate the transition in BMUs from the resorption to the formation phase (Matsuo 2010; Matsuo and Irie 2008).
The coupled osteoclast–osteoblast activity, however it is regulated, can be altered, whether by an excessive level of bone resorption (hyperparathyroidism) or by an insufficient level of bone formation (glucocorticoid administration, hypogonadism) or else by a combination of both (low turnover osteoporosis). The evaluation of BMU activity therefore becomes an important means for controlling the bone metabolism and the development or the course of a metabolic bone disease.
The Evaluation of BMU Activity
A number of biochemical and physical methods and techniques are available to study and control the skeletal metabolism and to follow the course of a skeletal disease. However, direct measurement of the activity of BMUs and their phases can only be achieved by histomorphometry.
Bone Histomorphometry
This method makes it possible to measure the value of a number of microscopic variables from which the activity of BMUs can be calculated. It is carried out on histological slides whose preparation implies the need for a bioptic bone sample. Due to its invasive procedure, bone biopsy is not commonly used in the diagnosis of osteoporosis. The usual diagnostic tests are dual energy X-ray absorptiometry, for assessment of fracture risk, and some simple first-level exams (serum calcium, phosphate, alkaline phosphatase, creatinine, protein electrophoresis, complete blood cell count, erythrocyte sedimentation rate, and 24 hr urine calcium) for discrimination between primary and secondary forms of osteoporosis. However, there are some conditions in which bone biopsy can be indicated: suspicion of osteomalacia (e.g., osteoporomalacia); evaluation of adherence to treatment (e.g., malabsorption syndrome); excessive skeletal fragility in young patient; and suspicion of a rare bone disease. Bone biopsy with histomorphometry has a fundamental role as research tools, which is the only method that allows a direct and precise evaluation of BMU activity, that is crucial in the assessment of the mechanism of action and efficacy of new bone-active agents (Recker and Barger-Lux 2003).
Bone biopsy is an outpatient form of surgery carried out by means of special trephines—whether manual or automatic—that are thrust through the iliac crest in a vertical or horizontal, transiliac direction. A small bone cylinder is obtained in both cases, but the second method is preferable because it yields samples that show both the inner and the outer cortical plates and the intermediate cancellous bone. The samples must be processed without decalcification to facilitate the microscopic distinction between calcified and uncalcified areas and must be embedded in methacrylates or other synthetic resins to maintain the microscopic structures and their relationship as little changed as possible. Sections of about 1- to 2-µm thick can be obtained with relative ease, although 5-µm thick sections are much more commonly used. They can be stained with various histological stains or histochemical methods. Electron microscope studies can be carried out too, if needed. Moreover, to evaluate the speed of the mineralization process, before the biopsy, patients may be given 2 cycles of tetracyclines separated by a known time interval: the tetracyclines bind to ionic calcium and are deposited along the mineralization front during the mineralization process, becoming components of the calcified matrix, where they can be made visible as 2 fluorescent yellow lights if excited by ultraviolet light. The distance between the 2 labels varies with the speed of mineralization. The measurement of this distance, as well as those of the other histomorphometric variables, is usually carried out using an interactive image analyzer, which includes a computer with a dedicated software program; the analyzer is linked to a microscope by a camera. A review concerning some issues in conducting histomorphometry on bone biopsies taken from humans, or specimens taken from animals, was recently published (Recker et al. 2011). In the review, in reference to the laboratory animals, the anatomical sites most frequently used for histomorphometry in rodents (see also Animal Models for Postmenopausal Osteoporosis: The Rat section), as well as in nonhuman primates, are described. Moreover, the in vivo double labeling schedule usually applied to the various animal models, where calcein is the much more frequently used fluorochrome in comparison to tetracycline, is reported.
Bone Histomorphometric Variables
The activity of BMUs may be assessed by combining numerous histomorphometric results that are deduced from linear and surface measurements. This is possible on the basis of the stereological principle that in thin sections measurements of length and area are equal to measurements of areas and volume, respectively. The following variables are those most frequently used to evaluate the type and degree of bone remodeling in trabecular bone; the nomenclature adopted here is that suggested by a Committee of the American Society of Bone and Mineral Research (Parfitt et al. 1987). The structural variables of bone, which express the end product of the bone remodeling process, are as follows:
Bone volume/tissue volume (BV/TV, %), is the percentage of TV (trabeculae plus bone marrow) that is occupied by trabeculae. In the case of trabecular bone, it corresponds to the area occupied by trabeculae (calcified and uncalcified bone matrix) with respect to the whole area occupied by trabeculae plus the soft structures of the bone marrow cavities (normal value 19.91 ± 4.55%). The value for the BV/TV in normal young people is about 20%, but it falls with advancing age, especially in women after the menopause, and can reach values lower than 12%, which is considered the threshold for fractures (Meunier 1995).
It must be noted that bone strength depends not only on BV/TV but also, and above all, on bone quality, which, in turn, depends mainly on trabecular connectivity. The connectivity is correlated with the thickness of the trabeculae and of their interconnections (microarchitecture), which, according to the measurement method proposed by Parfitt et al. (1983), are expressed by the variables reported below.
Trabecular thickness (Tb.Th, µm), the thickness of bone trabeculae (normal value 129.69 ± 22.70 µm).
Trabecular number (Tb.N, no./mm), number of trabeculae intersected by a scanning line (normal value 1.46 ± 0.23 no./mm).
Trabecular separation (Tb.Sp, µm), distance between adjacent trabeculae (normal value 574.50 ± 130.50 µm).
A more direct evaluation of BMU activity can be obtained by measuring the amount of bone erosion and of bone formation that normally occur on about 20% of the trabecular BS. The variables of bone erosion include:
eroded surface (ES/BS, %), percentage of trabecular surface consisting of resorption lacunae (normal value 1.52 ± 1.28%);
osteoclast surface (Oc.S/BS, %), percentage of trabecular surface covered by osteoclasts and undergoing resorption (normal value 0.18 ± 0.18%);
osteoclast number (Oc.N/BS, n/mm2), number of osteoclasts/mm2 of trabecular surface (normal value 0.05 ± 0.05/mm2); and
erosion depth (E.De, µm), the mean depth of Howship’s lacunae, that is, the depth of bone eroded in each BMU (normal values 46.3–66.1 µm; Eriksen, Mosekilde, and Melsen 1985).
The variables of bone formation include:
osteoid volume (OV/BV, %), percentage of trabecular BV consisting of osteoid tissue (normal value 1.39 ± 1.08%);
osteoid thickness (O.Th, µm), the thickness of the osteoid seams (normal value 9.55 ± 3.32 µm);
osteoid surface (OS/BS, %), percentage of trabecular surface covered by osteoid tissue (normal value 9.58 ± 6.88%);
osteoblast surface (Ob.S/BS, %), percentage of trabecular surface covered by osteoid tissue lined by active osteoblasts (normal value 0.20 ± 0.49%);
wall thickness (W.Th, µm), the mean width of the BSU, that is, the thickness of new bone synthesized by osteoblasts in each BMU (normal value 48.23 ± 5.17 µm); measured under the polarization microscope; gradually falls with increasing age (Lips, Courpron, and Meunier 1978);
mineralizing BS (MS/BS, %), percentage of trabecular surface labeled by tetracycline (normal value 9.60 ± 3.86%); and
mineralizing osteoid surface (MS/OS, %), percentage of osteoid surface labeled by tetracycline (normal value 69.55 ± 20.64%).
After defining the variables reported earlier, which are called “static,” a series of other bone formation variables can be added (called “dynamic”); they can be calculated by starting from the value of the mineral appositional rate (MAR).
MAR (µm/day), the distance between 2 contiguous tetracycline labels (µm) divided by the time interval between the 2 cycles of tetracycline (days); this variable indicates the osteoblast mineralization activity at the single cell level (normal value 0.62 ± 0.12 µm/day);
adjusted apposition rate (Aj.AR, µm/day), the osteoblast mineralization activity at the BMU level (Aj.AR = MAR × MS/OS; normal value 0.42 ± 0.11 µm/day);
bone formation rate (BFR/BS, µm3/µm2/year), the volume of newly mineralized bone per unit of trabecular BS per year (BFR/BS = MAR × MS/BS × 365; normal value 20.07 ± 8.39 µm3/µm2/year); and
mineralization lag time (Mlt, day), time interval between deposition of osteoid matrix and its mineralization (Mlt = O.Th/Aj.AR; normal value 33.85 ± 10.18 days).
The following dynamic variables, indicative of the length of each phase of the total remodeling period, can be calculated too:
Erosion Period (EP, day) the mean time required for a BMU to complete a lacunar erosion (EP = ES/BS / OS/BS × formation period (FP); normal value 15.66 ± 9.00 days);
FP (day), the mean time required for a BMU to rebuild a new BSU (FP = W.Th/Aj.AR; normal value 121.66 ± 28.50 days);
quiescent period (QP, day), the average time during which no remodeling takes place at a given trabecular surface (QP = [100 − (OS/BS + ES/BS)]/OS/BS × FP; normal value 882.47 ± 458.68 days);
The reciprocal of the total remodeling period corresponds to the activation frequency, the only variable that includes the whole bone remodeling process:
Activation frequency (Ac.f), the number of times when a new remodeling cycle is initiated at the same point of a trabecular surface in a year (Ac.f = 365/EP (days) + FP (days) + QP(days); normal value 0.43 ± 0.21 events/year).
All these variables used in the analysis of bone biopsy specimens from humans can be measured and have similar meaning in animal species used as models for osteoporosis (Recker et al. 2011). However, normal values may vary, even considerably, in relation to animal species, strain, and skeletal site. The variability in normal histomorphometric values also occurs with age, as shown in rats and nonhuman primates, the 2 most commonly used species for final safety and efficacy evaluation of bone-active agents (Lees and Ramsay 1999; Li et al. 1991). Thus, it is recommended that in each study using laboratory animals a normal control group is included to obtain normal values to be compared with those of treated animals.
Osteoporosis and Its Histomorphometric Characteristics
Although its distinctive traits are reduced bone strength and a predisposition to fracture (National Institute of Health 2001), osteoporosis is not a unique disease from an etiopathogenetic point of view. It is classified as primary or secondary. Primary osteoporosis can occur in both sexes at any age; it includes idiopathic juvenile osteoporosis and involutional osteoporosis (postmenopausal and senile). Secondary osteoporosis accounts for about 10% of all osteoporotic conditions; it may be the result of medications (e.g., glucocorticoids), endocrine disorders (e.g., hypogonadism), hematologic diseases (e.g., systemic mastocytosis), gastrointestinal disorders (e.g., celiac disease), lifestyle factors (e.g., alcoholism), prolonged immobilization (e.g., prolonged bed rest), or other conditions (e.g., lack of gravity). The main microscopic bone change consists of a fall in BV/TV. In reference to trabecular connectivity, different patterns of bone loss have been reported in defining the various types of osteoporosis: postmenopausal osteoporosis is mainly due to a fall in Tb.Ns, while the chief feature of senile osteoporosis is a decrease in Tb.Th. These differences are a result of changes in bone remodeling. However, fragmentary, sometimes conflicting values are reported in osteoporosis for the histomorphometric variables of remodeling, because bone biopsies obtained from patients are examined at different, unpredictable stages of the disease (the loss in bone density has mostly already taken place when a patient first asks for medical care) and because of the obvious difficulties in finding normal human bone samples as controls. They do, however, maintain their diagnostic value; in particular, histomorphometry is the only method that makes possible the evaluation of remodeling balance (Re.Ba), that is dependent on the quantity of bone eroded and formed within each BMU. This is calculated as the difference between the W.Th (the thickness of new bone synthesized by osteoblasts in each BMU) and the E.De (the mean depth of Howship’s lacunae). In each BMU in a normal young subject, the amount of bone resorbed by osteoclasts is equivalent to that formed by osteoblasts, so that the value of Re.Ba is 0, and BV/TV is maintained. Conversely, BV/TV falls whenever the value of Rm.Ba becomes negative, as happens when E.De is excessive (with osteoclast activity too great), or when the W.Th is lower than normal (poor osteoblast activity), because in both cases the erosion lacuna cannot be completely replenished. If this happens, the trabeculae inevitably become thinner than normal and the degree of connectivity falls.
The total amount of bone depends not only on the value of the Re.Ba in each BMU but also on the total number of BMUs at a given moment, that is, on the rate of bone turnover. In this connection, useful information can be drawn from the histodynamic variables, although some controversy exists on their values. Thus, the value of Ac.f has been reported to increase by about 33% after the menopause (Han et al. 1997), and it is supposed that the increase is even higher in women with postmenopausal osteoporosis, whereas no consistent data are available on senile osteoporosis, where what is reported is either a lower level of remodeling or an actual increase, according to the unitary model of involutional osteoporosis (Riggs, Khosla, and Melton 1998).
It should be concluded that postmenopausal osteoporosis is a high turnover disease, whereas senile osteoporosis is predominantly a low turnover disease, although this conclusion has been challenged by some authors. Because of the great therapeutic importance that the true pathogenesis of the disease has come to acquire, there is a strong need for further studies on the changes occurring in osteoporotic bone, especially in the initial phases of osteoporosis. Several problems arise, though, when human bone biopsies should be studied. They can only be obtained through an invasive procedure; normal control values are fragmentary because of the obvious practical difficulties in studying normal bone samples that can only be obtained at autopsy or during orthopedic surgery. In patients, bone biopsy is generally performed when the osteoporotic changes are already in an advanced stage of development, and when the alterations in bone remodeling that lead to the lowering of bone mass and the disruption in trabecular microarchitecture have already taken place. Additionally, ethical limits rule out the testing of new potential therapies in humans. These difficulties can only be overcome by the use of animal models that offer a precise knowledge of the state of the skeleton and its changes over time and do not impose limitations on the availability of bone tissue.
Animal Models for Postmenopausal Osteoporosis: The Rat
Because it is easy to manage and not very expensive, the laboratory rat has often been used in many experimental protocols leading to bone loss and osteoporosis. Immobilization, dietary manipulations, and hormonal deprivation or administration are the methods of study that are usually chosen (Lelovas et al. 2008). Of these, ovariectomy is frequently adopted, and the ovariectomized rat (OVX) is a well-established model for postmenopausal osteoporosis (Ballanti et al. 1993; Kalu 1991). In this context, 2 different animal types can be used: the aged rat and the mature rat. The former is not a senescent animal, as the name suggests, but rather an animal whose skeletal properties have been stabilized and no longer subject to age changes, that is, similar to the skeletal characteristics of the women who are susceptible to postmenopausal osteoporosis. The use of this type of rat brings with it sufficient confidence that the bone changes observed after OVX are really due to estrogen deficiency and are not complicated by continued rapid bone growth as occurs in younger animals or by age-related bone loss and diseases that can occur later in the life of the rat. The aged rat is usually sacrificed several months after OVX to permit the loss of a significant amount of bone. As a consequence, the aged rat is expensive and its availability is limited, so that the less expensive and more easily available mature rat is preferentially used. The mature rat is an approximately 3-month-old animal, and the adjective “mature” is only used to indicate that it is reproductively (i.e., sexually) mature and capable of responding appropriately to the sex hormone deficiency induced by OVX, whose effects, in fact, become manifest in a month or less (Kalu 1999).
The bone changes that occur in the 2 types of animals are similar. It must be noted that in osteoporotic women the first and most severe bone changes occur in the spongy bone of the vertebral body, whereas in aged and mature rats they predominantly involve the trabecular bone of the metaphyseal region of the long bones and, more specifically, of the distal femur and proximal tibia. Histomorphometric measurements performed in these sites in rats should preferentially be drawn from frontal histological sections within a conventional rectangular area at least 1 mm far from the central point of the growth plate–metaphyseal junction, so as to avoid the primary bone of the growth zone (Figure 3).
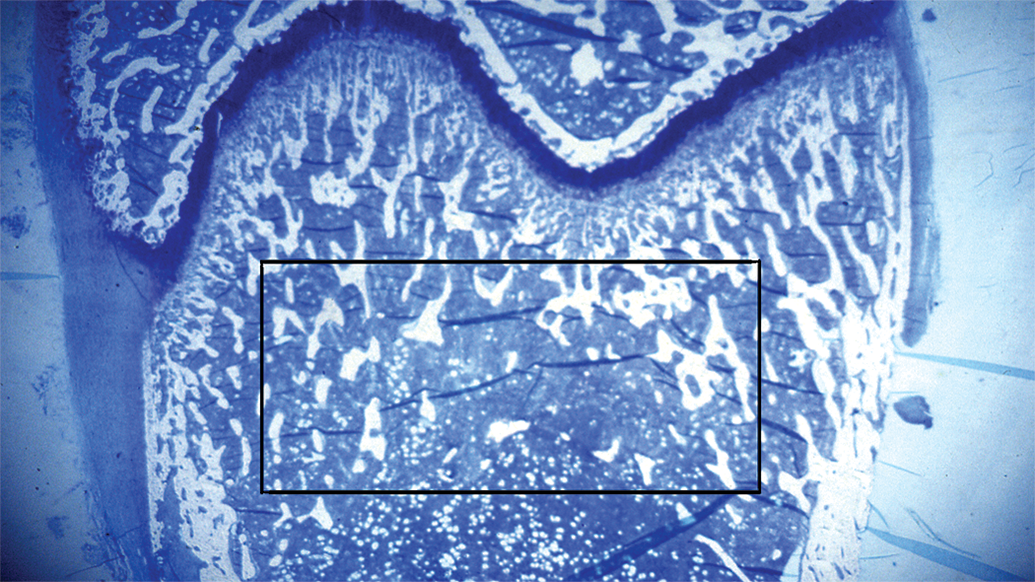
Frontal section of a rat distal femur: the conventional rectangular zone for histomorphometric measurements, about 1 mm far from the central point of the growth plate–metaphyseal junction, is shown. Undecalcified, 2-µm thick section stained by methylene blue—azure II; original magnification ×2.5.
In this area, the value of the BV/TV decreases rather rapidly during the first 30 days after OVX, with a total final fall of about 50% of the initial value (Figure 4A and 4B). These changes must obviously be evaluated by comparison with the BV/TV of a basal group of rats (sacrificed at the beginning of the study) and of sham-operated rats (Kalu 1991). The bone loss chiefly depends on a reduction in numbers of trabeculae and an increase in Tb.Sp due to a dramatic increase in osteoclastic bone resorption. This is clearly shown by the histochemical demonstration on the trabecular endosteal surface of a great number of mono- and polynucleated cells that give a positive reaction for tartrate-resistant acid phosphatase activity (Figure 4C and D). Actually, as shown by the MAR values, osteoblastic bone formation increases too after OVX, but the increase is lower than that of osteoclastic resorption and is insufficient to compensate for the loss of bone (Bonucci et al. 1995; Figure 4E and 4F).
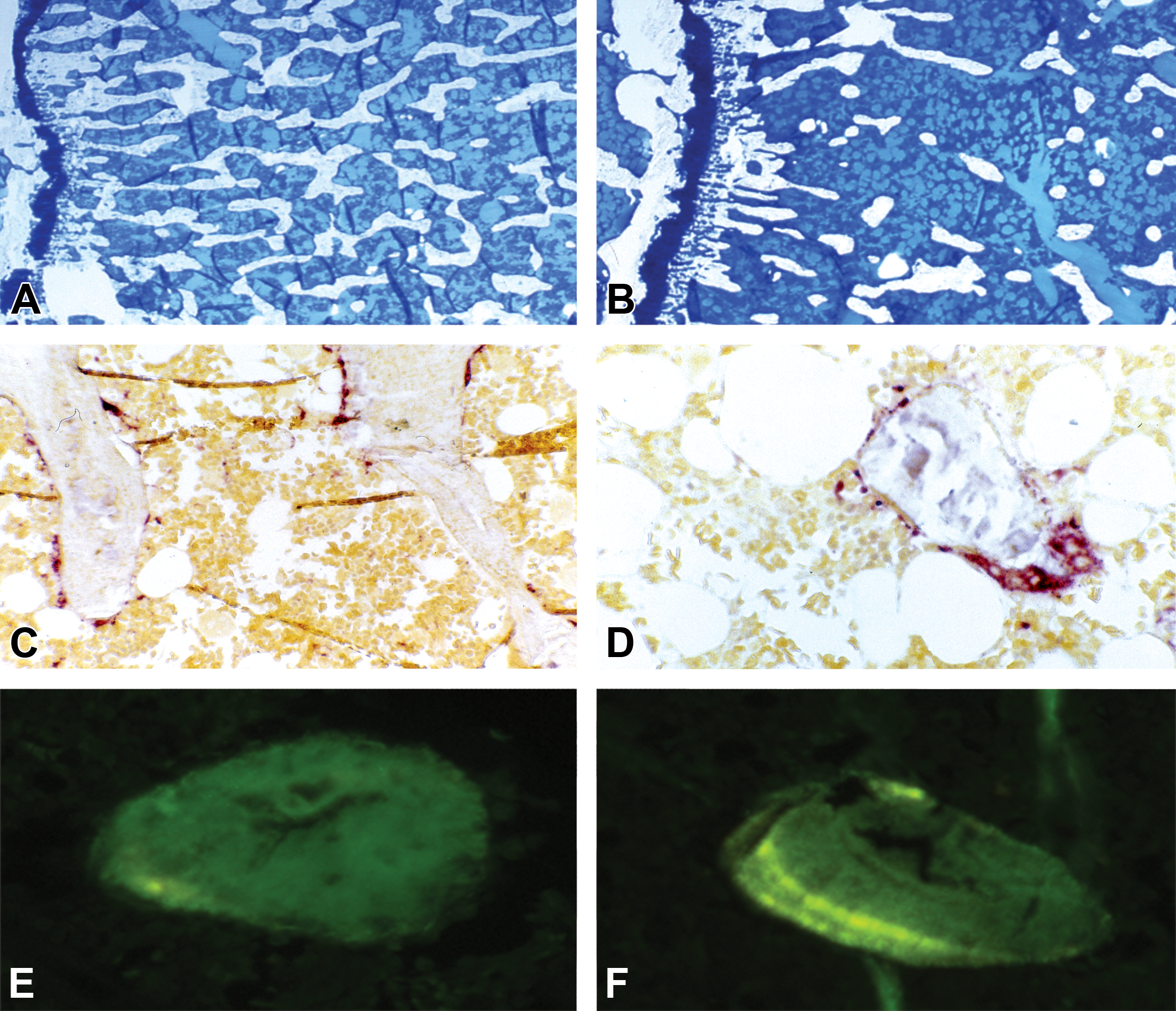
Metaphyseal bone sections from sham operated control (A, C, and E) and ovariectomized rat (B, D, and F). (A and B) In comparison with the control, the ovariectomized animal shows considerable trabecular bone loss (undecalcified, 2-µm thick sections stained by methylene blue—azure II; original magnifications ×4). (C and D) In the control rat, only few and little osteoclasts are found along the trabecular surfaces, while in the ovariectomized rat numerous and large osteoclasts containing many nuclei are visible (undecalcified, 3-µm thick sections treated for the cytochemical demonstration of tartrate-resistant acid phosphatase activity; original magnifications ×25 and ×40, respectively). (E and F) A single tetracycline fluorescent label of a control rat is shown in contrast with a double, well-spaced label found in an ovariectomized animal (undecalcified, 5-µm thick unstained sections examined under UV light; original magnifications ×40). UV = ultraviolet.
Besides those produced in trabecular bone, the skeletal changes caused by OVX can be evaluated using cortical bone, too, although they are less pronounced at this site. In humans, two variables of cortical bone are usually measured at the iliac crest:
cortical thickness (Ct.Th, mm), the thickness of cortical bone, from the periosteal to the endosteal surface (normal value 1.1 ± 064 mm; Brockstedt et al. 1993); and
cortical porosity (Ct.Po, %), percentage of cortical bone occupied by Haversian canals (normal value 5.8 ± 3%; Brockstedt et al. 1993).
In humans, cortical bone is lost with aging at the endocortical endosteum. In normal postmenopausal women, the fall in Ct.Th is attributed to an increase in the E.De value, which could be due to a delay in osteoclast apoptosis; in women with postmenopausal osteoporosis, the fall in Ct.Th can be as great as 30%, leading to fragility and fractures, especially in the diaphysis of long bones. In men, the loss of cortical bone is less pronounced and, in both sexes, it usually clinically manifests only in elderly people. In both sexes, increased intracortical remodeling (most of it closely related to the Haversian canals) leads to an increase in Ct.Po, which, in any case, does not exceed 10% even in osteoporotic patients and does not affect bone strength. In rats, the cortical bone is examined in coronal sections of the diaphysis of long bones. In mature rats, which are still growing, the OVX does not greatly change the Ct.Th, because endosteal resorption is balanced by periosteal bone formation, as it is shown in cross-sections of either the midpoint of the femur or the region located 1 to 2 mm proximal to the tibiofibular junction, the sites where cortical bone studies are preferably performed. Moreover, Ct.Po is not measurable because rats lack well-developed Haversian systems.
For these reasons, limitations have been introduced on the use of rats as models of postmenopausal osteoporosis. The Guidelines for Preclinical and Clinical Evaluation of Agents Used in the Prevention or Treatment of Postmenopausal Osteoporosis introduced by the FDA Division of Metabolism and Endocrine Drug Products (Washington, DC, 1994) recommended to perform preclinical studies of bone quality (i.e., bone mass and architecture) in two animal species, one of which corresponds to the ovariectomized rat; and, because no single animal species duplicates all the characteristics of human osteoporosis, it was suggested to include a second study on a nonrodent model (i.e., larger, remodeling species). More recently, these statements were further considered by the Guideline on the Evaluation of New Medicinal Products in the Treatment of Primary Osteoporosis elaborated by the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency (London, 2006).
Animal Models for Postmenopausal Osteoporosis: Nonrodent Animals
Several large animals might be used as models of osteoporosis, but the most suitable are the dog, the pig, the sheep, and the nonhuman primates (Hayashi and Fotovati 2003; Kalu 1999; Rodgers, Monier-Faugere, and Malluche 1993). The dog offers several advantages: dogs possess Haversian systems, and the intracortical remodeling activity in dogs is similar to that found in the human skeleton; their epiphyses fuse following maturity; they lose bone with aging; and repeat biopsies may be taken from the ilium. There are, however, disadvantages, too: available reports on the nature of the skeletal response of the dog skeleton to OVX are conflicting, probably because these animals have only 1 estrous cycle per year; in addition, because of social pressures, their availability is decreasing and their management is becoming difficult.
The use of pigs, too, has several advantages: these animals have well-developed Haversian systems; in a way similar to women, pigs experience a continuous estrous cycle lasting about 20 days; they have similar feeding system, that is, are omnivorous; serial biopsies can be easily obtained; miniature laboratory pigs are available. There is no lack of disadvantages, either: information on ovariectomized pigs used for bone studies is limited; commercial farm pigs are large in size and are difficult to manage; and minipigs are relatively expensive.
Sheeps, likewise, are seasonally polyestrous animals, experiencing several estrous cycles (14–21 days in length) during their breeding season; the ≥7-month-old sheep have well-developed Haversian systems; aged sheep display bone loss; sheep can easily be handled. Their skeleton, however, undergoes seasonal variations in bone loss and bone mass: bone formation is severely depressed in winter and animals can spontaneously lose and regain bone during an experimental period. Moreover, due to their large body size, they require large spaces to be housed. Reviewing this topic, Oheim et al. (2012) conclude that although different ewe models for osteoporosis have been successfully established, a large animal model that perfectly mimics the human disease is still to be found.
Nonhuman primates (old-world monkeys), too, can be advantageously used for the study of bone remodeling in postmenopausal osteoporosis. They, are, in fact, genetically very close to humans; they have menstrual cycles and a “menopause” similar to human females, although their “menopause” occurs much later chronologically; some of them maintain an upright body posture with a bone biomechanical loading pattern similar to that of humans; they lose bone mass at an advanced age; their immune system is similar to humans. Currently, in consideration of all these aspects, the nonhuman primate is the most widely used large animal model to evaluate the effects on bone of new drug entities, especially with the increase in testing new biologic agents (Smith, Jolette, and Turner 2009). Unfortunately, their use has various disadvantages: they must be ovariectomized to induce ovarian hormone deficiency and bone loss of postmenopausal type; large numbers of animals are necessary to achieve adequate statistical significance; special facilities are required and high expenses must be covered.
Animal Models for Senile Osteoporosis
While the ovariectomized rat has an established status as a powerful and acceptable model of postmenopausal osteoporosis, and other large animals can be used in its place, equally reliable models of senile osteoporosis are not available, although aged animals have been used for this research (Hayashi and Fotovati 2003). As a result, special types of animals have been developed.
Senescence-accelerated mouse (SAM) strains have been developed using an inbreeding system (Takeda, Hosokawa, and Higuchi 1997); of these, the SAMP3 and, above all, the SAMP6 strains are considered valid models for the study of senile osteoporosis. These animals reach a low-peak bone mass at their fourth to fifth month, after which they show a slow, constant bone loss that leads to severe osteopenia and spontaneous fractures. Compared with their controls, they show a reduction in trabecular BV in lumbar vertebrae and distal femur, associated with lower osteoblast numbers and a falling BFR; the reported osteoclast reduction has been explained as a secondary effect of reduced osteoblastogenesis (Jilka et al. 1996). The phenotype of SAM rats depends on the mutations and polymorphism of many genes, so that their study is quite complicated.
Another model of senile osteoporosis has been developed using the Klotho mouse, a transgenic mouse model obtained by an insertion mutation that disrupts the Klotho gene locus. The Klotho gene encodes a transmembrane protein that forms a complex that includes multiple fibroblast growth factor (FGF) receptors and functions as coreceptor for FGF23, an osteocyte-derived hormone that induces negative phosphate balance (Kuro-o 2009). Defects in either Klotho or Fgf23 gene expression cause not only phosphate retention but also a premature-aging syndrome in mice (Kuro-o 2012) that comprises atherosclerosis, emphysema, hypogonadism, ectopic calcifications, osteopenia, and premature death. Eight-week-old Klotho mice show an approximately 40% fall in the cortical thickness of the tibia diaphysis and a reduction in the metaphysis of histomorphometric variables both in bone formation (Ob.S/BS, BFR/BS) and in its resorption (Oc.N/BS, ES/BS), so indicating a condition of low bone turnover (Kawaguchi 2006). The reduction in bone formation (80–90% of the control value) is much sharper than that (60%) of bone resorption, a relationship which approaches that of human senile osteoporosis and makes the Klotho mouse a good model for this pathologic condition.
Conclusions
The maintenance of the mechanical and metabolic efficiency of the skeleton depends on a normal bone remodeling activity and on the coupling between its resorption and formation phases. A knowledge of the many systemic and local factors that regulate the activity of osteoclasts and osteoblasts, and of the increasing importance of osteocytes as the pivot of the whole metabolic process, permits a progressively more comprehensive approach to a thorough understanding of the pathogenesis of bone diseases and their therapy. The discovery of new osteotropic factors (OPG and sclerostin) paves the way to new cures for bone diseases, in particular, for the various types of osteoporosis. Bone histomorphometry is a powerful tool for the assessment of the microstructure of bone and its metabolism and offers a unique way of attaining a precise evaluation of the phases of bone remodeling. By providing information that is not made available by any other investigative approach, it has become the technique of choice for studying the pathogenesis of osteopathies which, like osteoporosis, offer no other investigative means for recognizing the earliest pathological changes. It has, however, the serious handicap of requiring a bone biopsy, an invasive procedure that is not easily accepted by patients. This has led to the extensive use of the bone of experimental animals instead of human bone, although there is no ideal animal model that perfectly reproduces osteoporotic human conditions. The ovariectomized rat has similarities with, and can be used for the study of bone in, postmenopausal osteoporosis; larger animals (dogs, pigs, sheeps and nonhuman primates) can be advantageously used with the same aim. Some conflicting results are inevitable, but the animal models developed so far have given valuable information on the pathogenesis of osteoporosis as well as on other pathological conditions of the skeleton, and bone histomorphometry can be considered the gold standard in these studies. The development of new animal models (SAM, Klotho mouse) paves the way to an understanding of what have previously been poorly known skeletal changes and their therapy.
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
