Abstract
Drug-induced phospholipidosis is characterized by intracellular accumulation of phospholipids with lamellar bodies in cells exposed to xenobiotics. Demonstration of the lamellar bodies by transmission electron microscopy (TEM) is the hallmark for a definitive diagnosis of phospholipidosis. However, the preparation of tissue samples for TEM and their ultrastructural evaluation are technically challenging and time consuming. Paraphenylenediamine (PPD) is essentially a fat stain, and the staining mechanism is based upon the osmication of unsaturated lipids. Thus, the application of PPD staining to osmicated tissue samples is considered an optimal way to identify lipids. We evaluated the potential of PPD staining to localize phospholipid accumulations on osmium-fixed semi-thin tissue sections of the lung, kidney, and liver, which were affected with phospholipidosis, under a light microscope. PPD staining revealed the presence of PPD positive dark fine granular material in the cytoplasm for all affected tissues examined, which correlated ultrastructurally with lamellar bodies as well as a light microscopic finding of cytoplasmic vacuolation. The great advantage of PPD is that it can be incorporated into the protocol for standard TEM tissue preparation and significantly improve the efficiency of TEM work. In conclusion, PPD provides a simple, sensitive, and reliable method for visualizing phospholipid accumulation on light microscopy and represents an easy tool to study drug-induced phospholipidosis.
Phospholipids are structural components of mammalian plasma and intracellular membranes. The metabolism of these components is regulated by the individual cell and may be altered by drugs that interact with phospholipids or the lysosomal enzymes that affect their metabolism. Drug-induced phospholipidosis is characterized by intracellular accumulation of phospholipids. A wide range of chemical entities including marketed drugs have been reported to cause phospholipidosis, many of which contain cationic amphiphilic structure (Anderson and Borlak 2006; Halliwell 1997; Nonoyama and Fukuda 2008; Shayman and Abe 2013). While drug-induced phospholipidosis can occur in various organs/tissues, pulmonary macrophages, bile ducts, and renal tubules are common tissues/cells involved (Nonoyama and Fukuda 2008; Shayman and Abe 2013). In affected cells, presence of membrane-bound lamellar bodies of primarily lysosomal origin is a definitive morphologic hallmark.
During nonclinical safety assessment phase of drug development, a number of different techniques have been used to confirm the presence of drug-induced phospholipidosis when the characteristic cytoplasmic vacuolation is seen in multiple tissues during routine light microscopic examination. For instance, special stains such as Nile red on frozen sections or immunohistochemistry utilizing formalin-fixed, paraffin-embedded tissue samples (Obert et al. 2007) for a lysosome-associated membrane protein (LAMP-2), have been shown to differentiate cytoplasmic vacuolation due to phospholipidosis. However, transmission electron microscopy (TEM) has been called the gold standard for determinations of phospholipidosis because it provides direct confirmation by demonstrating lamellar bodies. While TEM can be performed on routine formalin-fixed tissue samples (Fagerland et al. 2012), TEM is optimal when the tissue samples are fixed with glutaraldehyde or paraformaldehyde. Regardless of the initial fixative, tissue processing for TEM typically begins with postfixation in osmium tetroxide, which reacts with lipids, particularly phospholipids.
The dye, paraphenylenediamine (PPD), was first reported by Schultze in 1917 as a myelin staining technique for the central nervous system, and was found by Ledingham and Simpson in 1972 to function effectively for the visualization of osmium-treated neutral lipid. Also its application to osmicated tissues embedded in resin or plastic has been introduced by Estable-Puig, Bauer, and Blumberg (1965) and Snipes (1977). Guyton and Klemp described the use of PPD staining technique to discriminate lipids in atherosclerosis (Guyton and Klemp 1988). This staining technique has been also shown to provide a high-resolution visualization of myelin sheaths in osmicated nerve tissues (Braund, McGuire, and Lincoln 1982; Sadun, Smith, and Kenyon 1983). PPD chelates the osmium in tissues fixed with osmium tetroxide, leaving a permanent marker on osmicated lipid elements. Thus, the application of PPD staining to osmicated tissues during processing these samples for TEM evaluation is considered an optimal way to identify lipids by light microscopy on semi-thin sections prior to proceeding to the electron microscope. PPD staining methodology, however, is not routinely applied today. We report here the usefulness of PPD staining on TEM semi-thin tissue sections for a determination of drug-induced phospholipidosis in general toxicology studies.
Materials and Methods
Formalin-fixed wet tissues (kidney, liver, and lung) with phospholipidosis were selected from general toxicology studies of 2 different novel compounds in rats (Sprague-Dawley) or monkeys (cynomolgus). Similar wet tissues from vehicle-administered rats and monkeys were used as controls. Tissue samples included kidneys (renal tubules and glomeruli) from 2 monkeys administered a test compound and 1 control monkey; and livers (bile ducts) and lungs (alveolar macrophages) from 4 rats administered either 1 or the other of 2 compounds and 2 control rats. Considering a morphologic similarity between neutral lipids and phospholipids on routine hematoxylin and eosin (H&E) tissue sections, small intestines (epithelium) with neutral lipid deposition that had been identified in 4 monkeys were selected for comparison from toxicology studies as well. Formalin-fixed, paraffin-embedded sections from each tissue sample were also stained with H&E and examined by light microscopy to confirm the light microscopic feature of the changes.
Tissue samples that had been preserved in 10% neutral buffered formalin for up to 2 months were postfixed in 1% osmium tetroxide for 2 hr. The samples were then processed and embedded in epoxy resin for routine electron microscopy. Serial step semi-thin (0.5–1.5 uM) tissue sections were cut, mounted on glass slides, and stained with PPD as well as standard toluidine blue (TB) for light microscopy evaluation. For PPD staining, the slides were immersed in 1% PPD in methanol for 10 min at room temperature, followed by rinsing in 100% ethanol for 5 min. After that, the slides were dried and coverslipped with mounting media. Corresponding ultrathin sections were also prepared from each epoxy resin block, stained with uranyl acetate and lead citrate, and examined under a Hitachi H7100 transmission electron microscope at 75 kV. Digital images were captured using a digital charged-coupled device camera system (Advanced Microscopy Techniques, Danvers, MA).
Results
Light Microscopic Observations on Paraffin Embedded H&E Sections
Phospholipidosis was characterized light microscopically by cytoplasmic enlargement with multivesicular vacuolation of bile ductular epithelium in the liver (Figure 1C) or the presence of a large clear vacuole occupying the cytoplasm of distal tubular epithelium of the kidney (Figure 2C). Glomeruli of the affected kidney appeared to have a few small clear-appearing vacuoles in the capillary tuft, but it was difficult to distinguish them from capillary spaces. In the lung, phospholipidosis was characterized by accumulation of alveolar macrophages which had abundant multivesicular foamy cytoplasm. Neutral lipid deposition in the small intestine was seen as variably sized, round-shaped vacuoles within cytoplasm of enterocytes (Figure 2E).
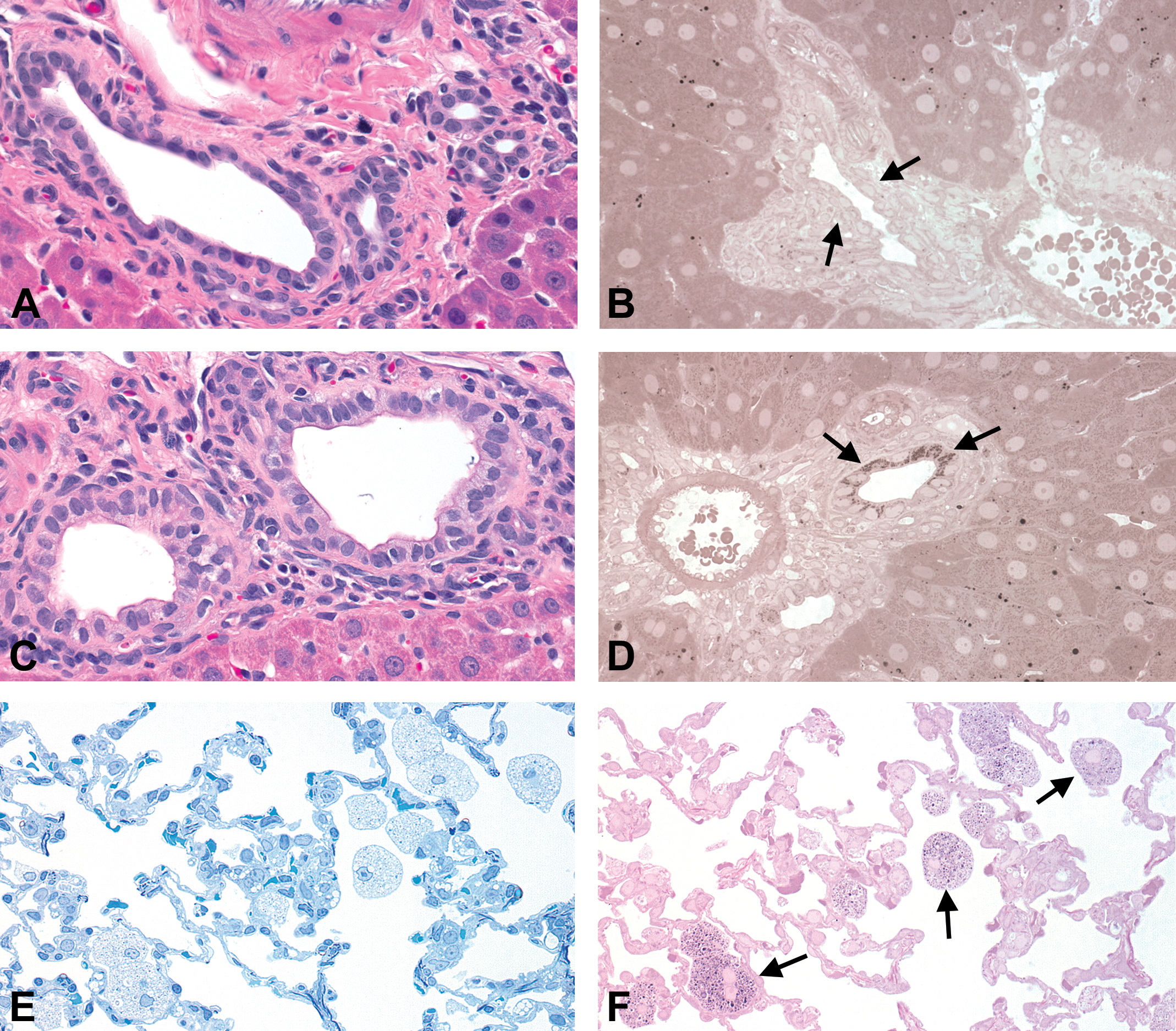
Representative photomicrographs of phospholipidosis. Sprague-Dawley rat. Original objective lens magnification 40×. (A and B) Intrahepatic bile duct of a vehicle control rat. Intrahepatic bile duct (arrows) is negative for paraphenylenediamine (PPD) staining. Sparsely scattered PPD-positive dots in hepatocytes and Ito cells represent neutral lipids. (A) Hematoxylin and eosin (H&E), paraffin section; (B) PPD staining, resin section. (C and D) Phospholipidosis in the intrahepatic bile duct showing cytoplasmic enlargement with occasional multivesicular vacuolation of the epithelium. Enlarged cytoplasms are positive for PPD staining (arrows). (C) H&E, paraffin section; (D) PPD staining, resin section. (E and F) Phospholipidosis in the lung exhibiting accumulation of alveolar foamy macrophages. Foamy macrophages contain PPD-positive fine granular material (arrows). (E) Toluidine blue staining, resin section; (F) PPD staining, resin section.
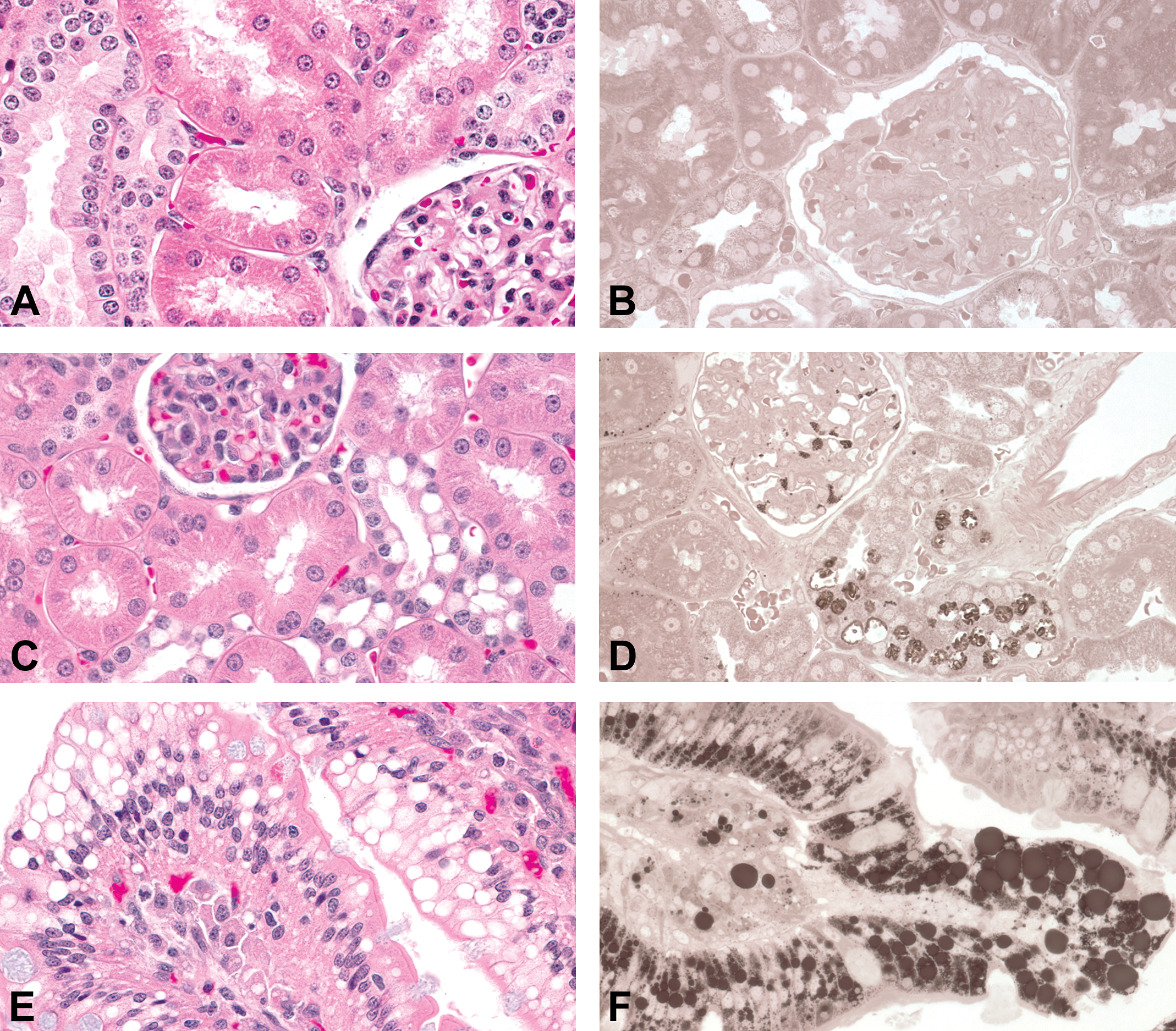
Representative photomicrographs of phospholipidosis versus neutral lipid deposition. Cynomolgus monkey. Original objective lens magnification 40×. (A and B) Kidney of a vehicle control monkey. Both glomerulus and tubule are negative for paraphenylenediamine (PPD) staining. (A) Hematoxylin and eosin (H&E), paraffin section; (B) PPD staining, resin section. (C and D) Phospholipidosis in the renal tubule showing a large clear vacuole occupying the cytoplasm of each cell in the distal tubular epithelium. Note the presence of PPD-positive fine granular material in the glomerulus as well as distal tubular epithelium. (C) H&E, paraffin section; (D) PPD staining, resin section. (E and F) Jejunum with enterocyte vacuolation due to neutral lipid deposition. PPD staining reveals variably sized amorphous lipid droplets in the enterocyte. (E) H&E, paraffin section; (F) PPD staining, resin section.
Light Microscopic Observations on PPD and TB Semi-thin Sections
Phospholipidosis was identified by the presence of PPD positive, dark, fine granular material in the glomerulus (Figure 2D) as well as bile duct epithelium (Figure 1D), renal tubule (Figure 2D), and pulmonary macrophage (Figure 1F), which correlated with light microscopic findings of cytoplasmic vacuolation or foamy appearance. Neutral lipids in intestinal epithelium (Figure 2F) were characterized by variably sized, amorphous and homogenous, PPD positive droplets while phospholipids primarily exhibited granular appearance. In general, TB staining provided no further information over the original paraffin embedded H&E-stained sections, although PPD positive material was occasionally recognizable on TB sections as it exhibited higher staining intensity in comparison with the background.
TEM Observations
In all tissues with phospholipidosis, TEM demonstrated the presence of multiple, uni- or multicentric laminated membraneous structures (lamellar bodies) in the cytoplasm of bile duct epithelium (Figure 3A), alveolar foamy macrophages (Figure 3B), podocytes (Figure 3C), and renal tubular epithelium (Figure 3D), which are consistent with phospholipid accumulation. Neutral lipids were visualized ultrastructurally by small to large variably sized, amorphous, homogenous, and moderately electron-dense globular droplets in the cytoplasm of enterocytes (Figure 4).
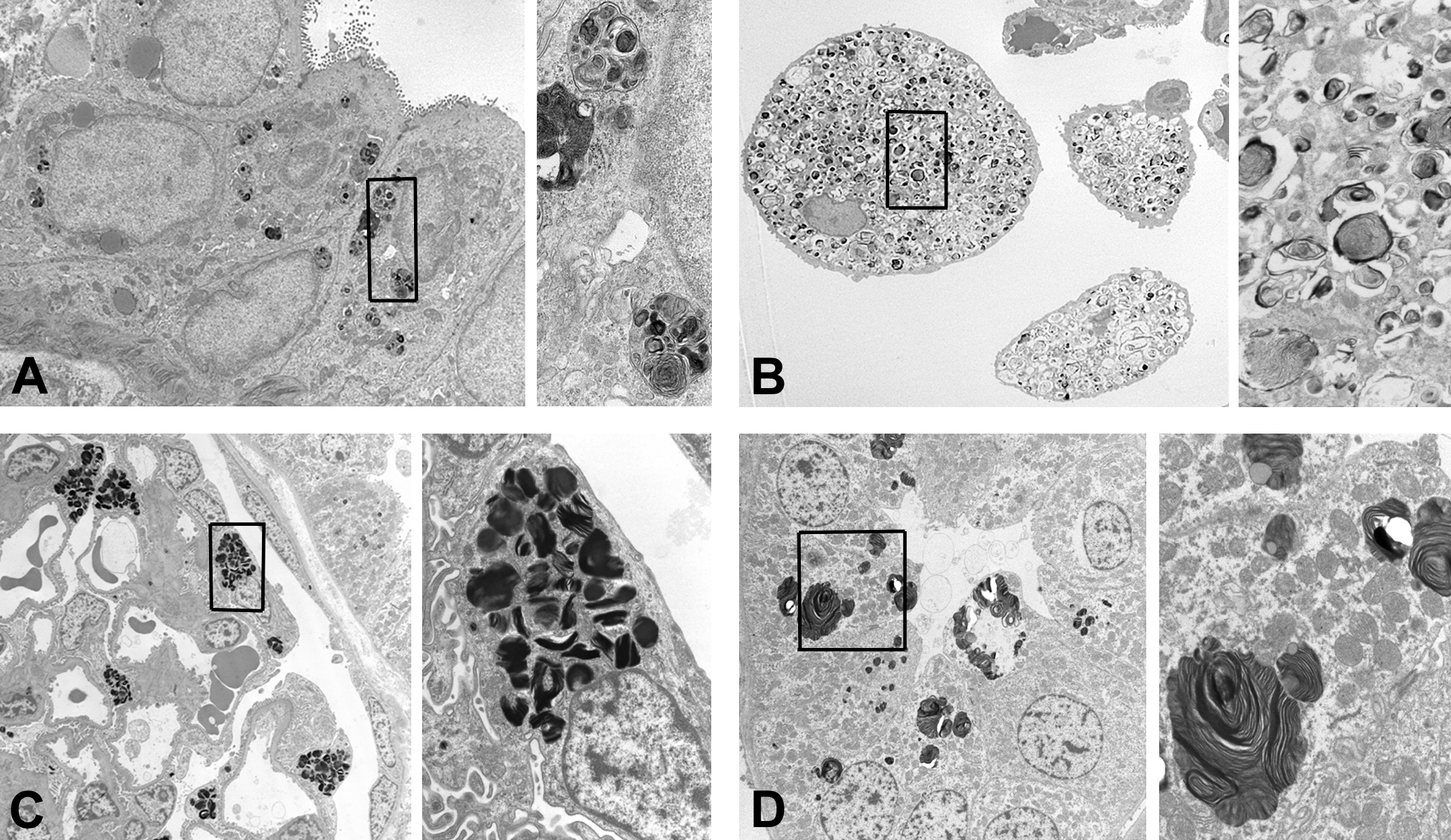
Transmission electron microscopy images of phospholipidosis. The box in a lower magnification image denotes the field shown by a higher magnification. (A) Intrahepatic bile duct epithelium. Sprague-Dawley rat. Multicentric laminated membranous structures in the cytoplasm of epithelial cells. Original magnification: 2000× (left) and 9000× (right). (B) Alveolar foamy macrophage. Sprague-Dawley rat. Numerous laminated membranous structures in the cytoplasm of macrophage. Original magnification: 1000× (left) and 5000× (right). (C) Glomerulus in the kidney. Cynomolgus monkey. Densely packed multilamellar bodies in the podocyte. Original magnification: 1000× (left) and 5000× (right). (D) Distal convoluted tubule in the kidney. Cynomolgus monkey. Multiple multilamellar bodies in the cytoplasm. Original magnification: 1500× (left) and 5000× (right).
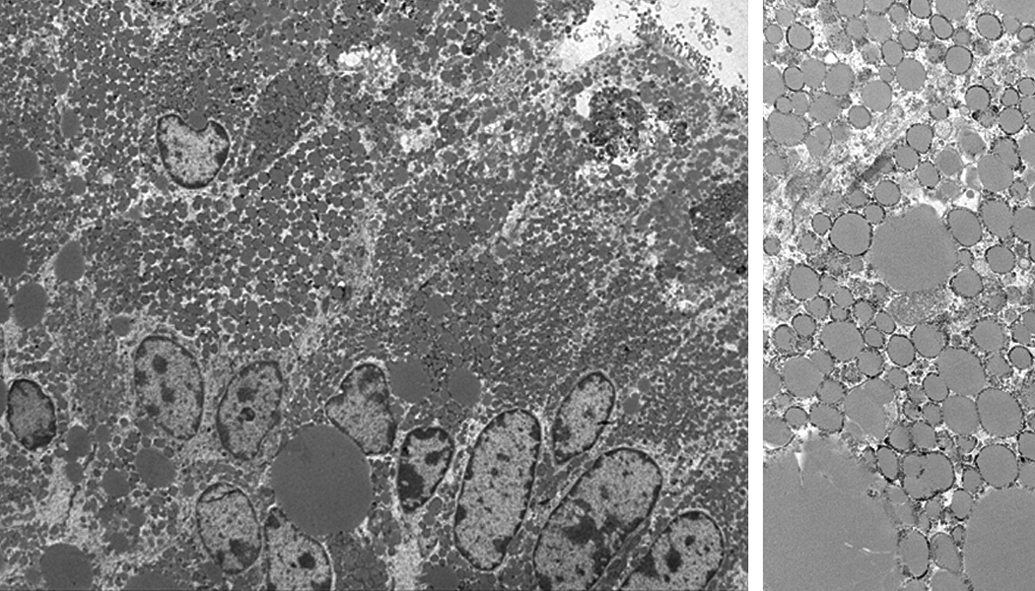
Transmission electron microscopy images of neutral lipid deposition. Enterocytes in the jejunum. Cynomolgus monkey. Variably sized, moderately electron dense, amorphous lipid droplets, consistent with neutral lipids. Original magnification: 1000× (left) and 5000× (right).
Discussion
Our study demonstrates that PPD is an outstanding stain to detect intracellular lipids on osmicated resin-embedded semi-thin tissue sections by light microscopy. Tissue samples used in this study were formalin-fixed (up to 2 months) livers, kidneys, lungs, and intestines from rats and monkeys. These tissue samples were treated with 1% osmium tetroxide for PPD staining and TEM, and provided adequate preservation of phospholipids and neutral lipids as shown by TEM images. The great advantage of PPD is that it can be easily incorporated into the protocol for standard TEM examination and allows ultrastructural characterization of PPD positive areas on corresponding sections from the same tissue block. Furthermore, application of PPD staining to TEM semi-thin sections is reasonable since the sections have not been subjected to the lipid extraction that occurs during the course of alcoholic dehydration and embedding procedures for routine histologic processing of tissue samples for paraffin sections. Once stained, PPD-positive areas are easily visualized, thus permitting rapid localization of the area for TEM. Then precise ultrathin sections can be selected and mounted on TEM grids, and the ultimate ultrastructural characterization can be reliably made. Consequently, iterative use of TEM and light microscopy can be eliminated. If the areas of interest were negative for PPD, then further ultrastructural examination might not be needed or can be limited to a representative sample for determinations of phospholipidosis. This approach will significantly improve the efficiency of TEM work.
PPD staining was a sensitive method to detect lipids at the light microscopy level using resin-embedded semi-thin sections based especially on the fact that it visualized phospholipid accumulation in renal glomeruli as shown in Figure 2D and allowed a subsequent ultrastructural determination, which would not have been possible based on H&E slides alone. Interestingly, phospholipid accumulation in podocytes has been reported in association with administration of chloroquine, a cationic amphiphilic drug, in humans (Bracamonte et al. 2006; Muller-Hocker et al. 2003: Romansky and Cohen 2005). While both neutral lipids and phospholipids were positively stained with PPD, phospholipids were distinguishable from neutral lipids because of their fine granular features. As previously mentioned, the same tissue blocks are immediately available for further subcellular differentiation of lipids by TEM when necessary.
All major classes of lipids can accumulate in cells: triglycerides and cholesterol/cholesterol esters (neutral lipids), and phospholipids (Kumar, Abbas, and Aster 2015). Accumulation of neutral lipids can be associated with diseases that alter lipid metabolism or a manifestation of cell damage especially in the liver. Both unesterified and esterified cholesterol accumulate in human atherosclerotic lesions. Hydrophobic lipids like neutral lipid and esterified cholesterol are demonstrated typically by lipid stain such as oil red O or Sudan III. These stains require frozen sections and often yield suboptimal results due to extraneous dye precipitate. Nile red, a fluorescent hydrophobic probe, has been shown to be an excellent stain for visualization of phospholipids in cultured cells or macrophages in pulmonary lavage fluids (Brown, Sullivan, and Greenspan 1992). Nile red is also reported to be capable of staining tissue lipids (Fowler and Greenspan 1985). Nile red staining, however, requires frozen tissue sections, which often provide poor resolution at higher magnifications and limited chance to study retrospectively. PPD staining could be an alternative tool to study lipid accumulation on tissue sections because it provides good resolution and contrast as well as permanence.
In conclusion, PPD provides a simple, sensitive, and reliable method for visualizing lipids on semi-thin resin sections under a light microscope and facilitates determination of phospholipidosis by TEM.
Footnotes
Authors’ Contribution
Authors contributed to conception or design (NS, FG, WB, CO); data acquisition, analysis, or interpretation (NS, FG, WB, CO); drafting the manuscript (NS); and critically revising the manuscript (NS, FG, WB, CO). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
