Abstract
Trimethyltin (TMT) is widely used as a plastic heat stabilizer and can cause severe toxicity. Here, the effects of TMT on testosterone production by adult Leydig cells and the related mechanisms of action were investigated. Eighteen adult male Sprague Dawley rats (56 days old) were randomly divided into 3 groups and given intraperitoneal injection of TMT for 21 consecutive days at the doses of 0 (vehicle control), 5, or 10 mg/kg/d. After treatment, trunk blood was collected for hormonal analysis. In addition, related gene and protein expression in testes was detected. At 10 mg/kg, TMT significantly reduced serum testosterone levels but increased serum luteinizing and follicle-stimulating hormone levels. The messenger RNA and protein levels of luteinizing hormone/chorionic gonadotropin receptor, steroidogenic acute regulatory protein, cytochrome P450 17-hydroxylase/17,20-lyase, follicle-stimulating hormone receptor, and SRY box 9 were significantly lower in the TMT-treated testes than in controls. Immunohistochemical study showed that TMT decreased adult Leydig cell number. In conclusion, these findings indicate that TMT reduced adult Leydig cell testosterone production in vivo by directly downregulating the expression of steroidogenic enzymes and decreasing adult Leydig cell number in the testis.
Introduction
Trimethyltin (TMT) is an organotin compound that is widely used in industrial and agricultural fields as a plastic heat stabilizer, in antifouling paints for ship hulls, and as a fungicide. 1,2 However, TMT is highly toxic in its free form and can exert toxic effects in both animals and humans. It has been reported that TMT causes neurodegeneration, particularly in the hippocampus. 1,2 Depending on the dose, occupational TMT exposure causes headaches, impaired memory, disorientation, psychotic behavior, attacks of rage, and coma. 3 -6 Trimethyltin may cause Alzheimer disease by increasing the expression of important pathophysiological factors such as amyloid precursor protein, presenilin, and c-fos in the limbic system. 7
Leydig cells can be found in the testicular interstitial compartment. They undergo 4 stages of development: progenitor Leydig cells appear at 21 days postpartum from the commitment of stem Leydig cells, undergo transition to immature Leydig cells at 35 days postpartum, and are fully mature adult Leydig cells at 56 days postpartum. Adult Leydig cells contribute to 95% to 99% of circulatory testosterone in the adult stage. 8 Testosterone is essential for many important physiological processes such as spermatogenesis and reproduction. 9
The biosynthesis of testosterone is an enzyme-controlled steroidogenic process that requires cholesterol. Extracellular cholesterol is transported into the mitochondria by 2 transporter proteins, namely cell membrane scavenger receptor class B type 1 (SCARB1, encoded by
The luteinizing hormone (LH, LH subunit is encoded by
Although extensive studies on the toxicities of TMT have been carried out, 1,2,19 -21 none have investigated the effects of TMT on testosterone production by rat Leydig cells. Therefore, the aim of the present study was to investigate the in vivo effects of TMT on rat Leydig cell testosterone production and to characterize the related mechanisms of action.
Materials and Methods
Chemicals
Trimethyltin was provided by Sigma-Aldrich (St Louis, Missouri). The Immulite 2000 Total Testosterone Kit were provided by Sinopharm Group Medical Supply Chain Services Co, Ltd (Hangzhou, Zhejiang, China). TRIzol were obtained from Invitrogen (Carlsbad, California), while the SYBR Green real-time quantitative polymerase chain reaction (qPCR) kit together with the bicinchoninic acid (BCA) Protein Assay Kit were provided by Takara (Otsu, Japan). Radioimmunoprecipitation (RIPA) buffer was purchased from Bocai Biotechnology (Shanghai, China).
Animals
Eighteen male Sprague Dawley rats (49 days old) were obtained from the Laboratory Animal Center of Lanzhou University (Lanzhou, China) and raised on a controlled light/dark cycle of 12-hour/12-hour (light cycle:7:00
All rats were sacrificed at the end of TMT treatment by asphyxiation with CO2. The trunk blood was collected, placed in a gel glass tube, and centrifuged at 1,500
Measurement of Serum Testosterone Concentration
Immulite 2000 Total Testosterone Kit (Sinopharm, Hangzhou, China) was used to measure the concentration of testosterone in the serum in accordance with the manufacturer’s protocol. The minimal detection limit for testosterone was 0.1 ng/mL. The intra- and interassay coefficients of variation were within 10%.
Measurement of Serum LH and FSH Levels
The levels of serum LH and FSH were detected using enzyme-linked immunosorbent assay kits according to the manufacturer’s instruction (Chemicon, Temecula, California) as previously described. 25 Briefly, aliquots of the samples and assay diluents were added to precoated wells and incubated at room temperature for 2 hours. Peroxidase-labeled immunoglobulin G anti-LH or anti-FSH solution was added into all wells to incubate for 2 hours under ambient temperature. Eventually, substrate solution was added to all wells and incubated for 30 minutes in dark under ambient temperature. The enzyme reaction was terminated by adding the stop solution. Quantification of LH and FSH levels was obtained by a microplate reader at 550 nm with correction wavelength at 450 nm.
Quantitative Polymerase Chain Reaction Analysis
Total RNA was separated from the TMT-treated rat testes by the TRIzol kit in accordance with the manufacturer’s protocol (Invitrogen). Concentration of total RNA was detected through measuring the OD value at 260 nm by the NanoDrop2000 (ThermoFisher Scientific) . RNA purity was evaluated through measuring the OD 260/280 by the NanoDrop2000, and the samples were considered qualified only if the value turned out to be at around 2.0. The integrity of RNA samples was assessed in a randomly chosen subset of samples (at least one replicate for all doses) using agarose gel electrophoresis. The first strand complimentary DNA (cDNA) was reverse-transcribed using a Reverse-Transcriptase Reagents Kit according to the manufacturer’s instructions (Invitrogen). Complimentary DNA was used as the template for subsequent qPCR measurement as previously described.
26
The levels of Leydig cell-specific messenger RNAs (mRNAs;
Western Blotting
Testes were homogenized with ice-cold phosphate-buffered saline buffer and then lysed with RIPA assay buffer (Bocai Biotechnology) to obtain protein samples. The protein concentrations in the supernatants were measured using the BCA Assay kit (Takara), according to the manufacturer’s instructions; 10% sodium dodecyl sulphate polyacrylamide gel electrophoresis was carried out to separate the protein samples (50 g), and the isolated protein was then transferred to the polyvinylidene fluoride membrane. The membrane was blocked with 5% free-fat milk in Tween 20-containing Tris-buffered saline for 1 to 2 hours to avoid nonspecific binding. After blocking, the membrane was incubated overnight at 4°C with primary antibodies against the following antigens: LHCGR, STAR, SCRAB1, CYP11A1, 3β-HSD1, CYP17A1, 17β-HSD3, FSHR, AMH, DHH, SOX9, and ACTIN, respectively. The membrane was washed and incubated with a 1:5,000 dilution of goat antirabbit antiserum conjugated with horseradish peroxidase for 2 hours under ambient temperature (Bioword, MN, USA). After washing, the protein bands were visualized with an enhanced chemiluminescence kit (Pierce Chemical Co, Rockford, Illinois). The intensity of proteins was quantified using ImageJ software and adjusted to that of ACTIN, the house-keeping protein. Related antibody information is displayed in Supplementary Table S2.
Immunohistochemical Staining and Cell Counting
3-Hydroxysteroid dehydrogenase 1 was taken as the adult Leydig cell biomarker. To enumerate the number of 3β-HSD1-positive Leydig cells, the testes were subjected to the fractionator technique, as previously described. 10 In brief, 6 testis samples were randomly collected from each group at every time point. Each testis was cut into 8 sections as disks, and 2 parts were randomly selected. The 2 parts were further cut into 4 pieces, and 1 piece was randomly selected from total 8 pieces. All testicular samples were subjected to paraffin embedding within the tissue array, which were then sliced to sections 6 µm in thickness. About 10 sections were randomly screened from every testis of each rat. Sections were used for immunohistochemical staining. Avidin–biotin immunohistochemical staining was performed according to the manufacturer’s instructions (Vector, Burlingame, California). Antigen retrieval was carried out in citrate buffer (10 mM, pH 6.0) in a microwave heated for 10 minutes. Afterward, endogenous peroxidase would be subjected to 30 minutes of blocking by 0.5% H2O2 in methanol. Sections were incubated in 3β-HSD1 antibody (1:1,000 dilution, vol/vol) for 1 hours at room temperature. The antibody–antigen complexes were visualized with diaminobenzidine as brown cytoplasmic staining for positively labeled adult Leydig cells. The sections were counterstained with Mayer hematoxylin, dehydrated in graded concentrations of alcohol, and coverslipped with resin. Images were made to count the total number of microscopic fields in each section. The total number of adult Leydig cells was calculated by multiplying the number of adult Leydig cells counted in a known fraction of the testis by the reciprocal probability of sampling.
Statistical Analysis
All data were expressed as mean ± standard error of the mean. GraphPad Prism (version 6; GraphPad Software Inc, San Diego, California) was employed for statistical analysis through one-way analysis of variance with post hoc Turkey multiple comparisons. A difference of
Results
Effects of TMT on Testes and Body Weights
To analyze the general toxicity of TMT, the body and testis weights of the rats were measured and recorded (Table 1). The results showed that TMT had no effects on body weight and testis weights even at the highest dose tested (10 mg/kg). No death was reported in this study.
Body and Testis Weights.a
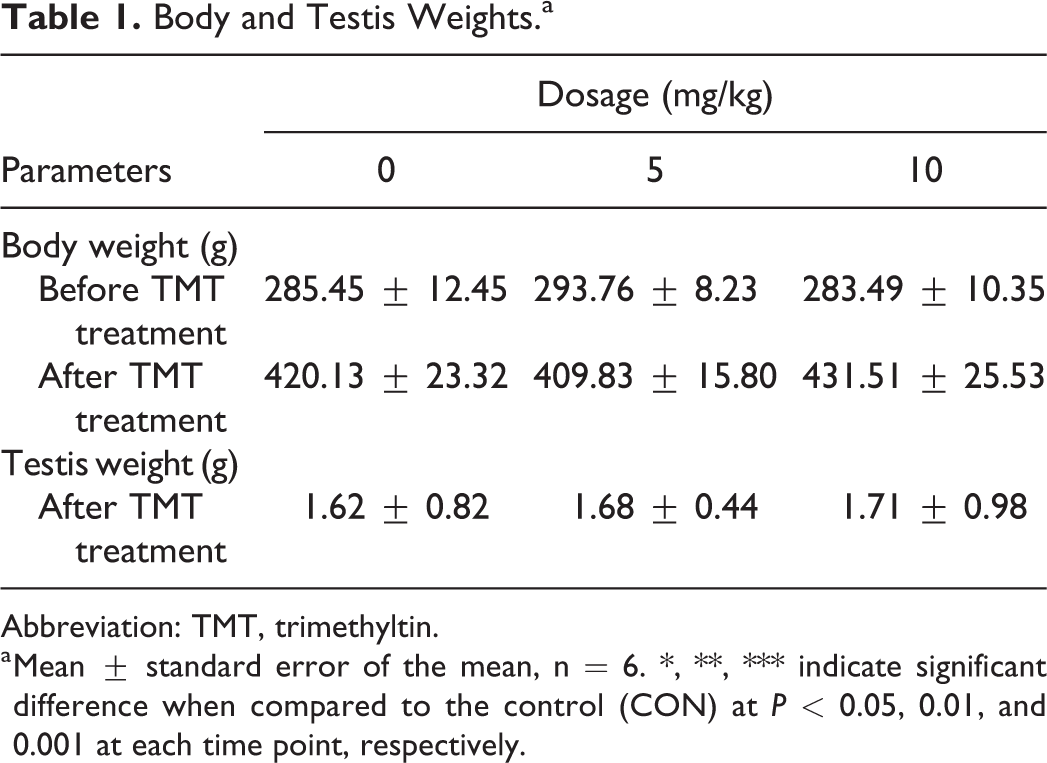
Abbreviation: TMT, trimethyltin.
a Mean ± standard error of the mean, n = 6. ∗, ∗∗, ∗∗∗ indicate significant difference when compared to the control (CON) at
Effects of TMT on Serum Hormone Levels
We found that TMT significantly reduced serum testosterone levels at 5 mg/kg (Figure 1A). In contrast, TMT significantly elevated serum LH and FSH levels at 10 mg/kg (Figure 1B, C). These results indicate that TMT inhibited the production of testosterone in rat adult Leydig cells and also influenced the related regulatory mechanism.
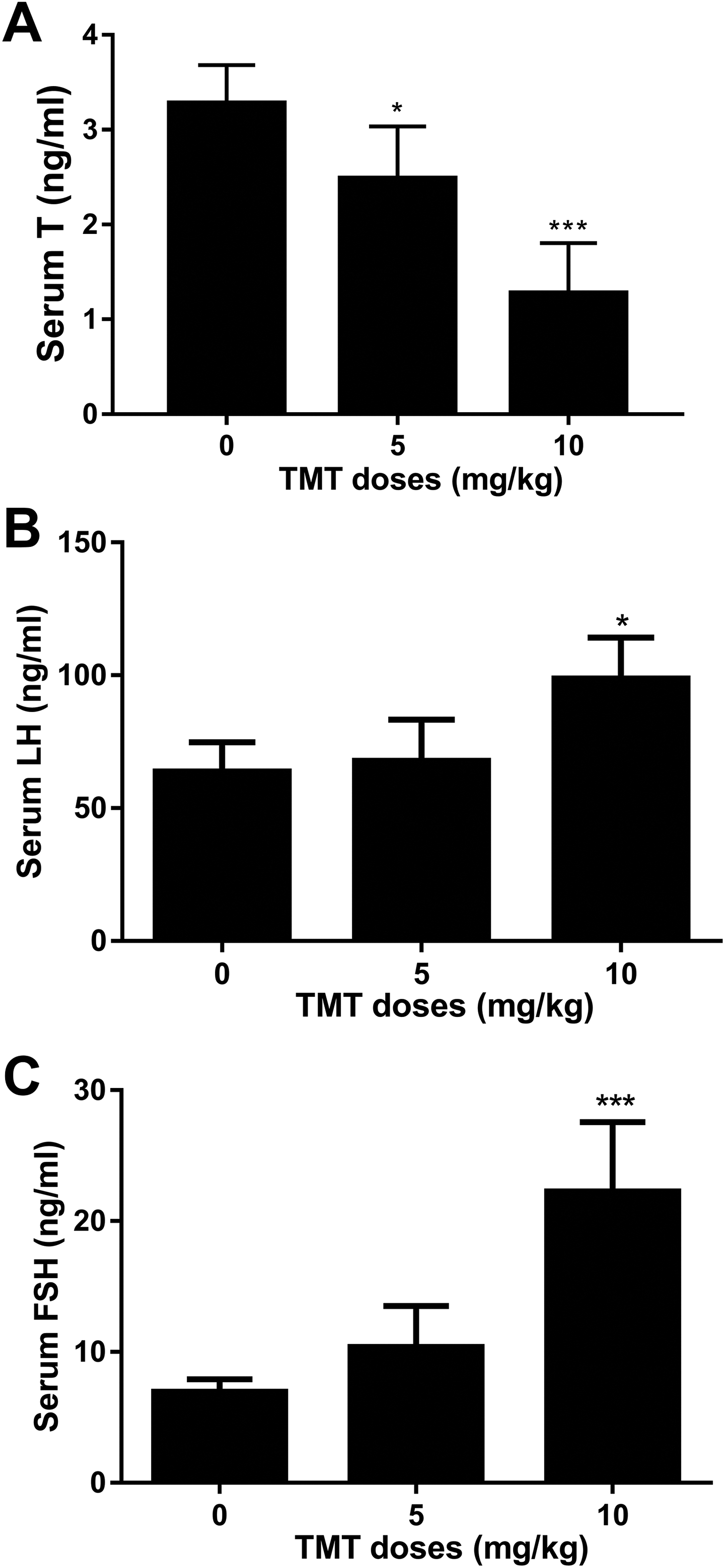
Effects of TMT on hormones affecting male reproduction. A, Serum testosterone levels after TMT treatment. B, Serum luteinizing hormone levels after TMT treatment. C, Serum follicle-stimulating hormone levels after TMT treatment. Mean ± standard error of the mean, n = 6. ∗, ∗∗, ∗∗∗ indicate significant difference when compared to the control (0 mg/kg TMT) at
Trimethyltin Downregulates Gene Expression in Leydig and Sertoli Cells
For the adult Leydig cell-specific genes, we found that TMT significantly downregulated the expression of
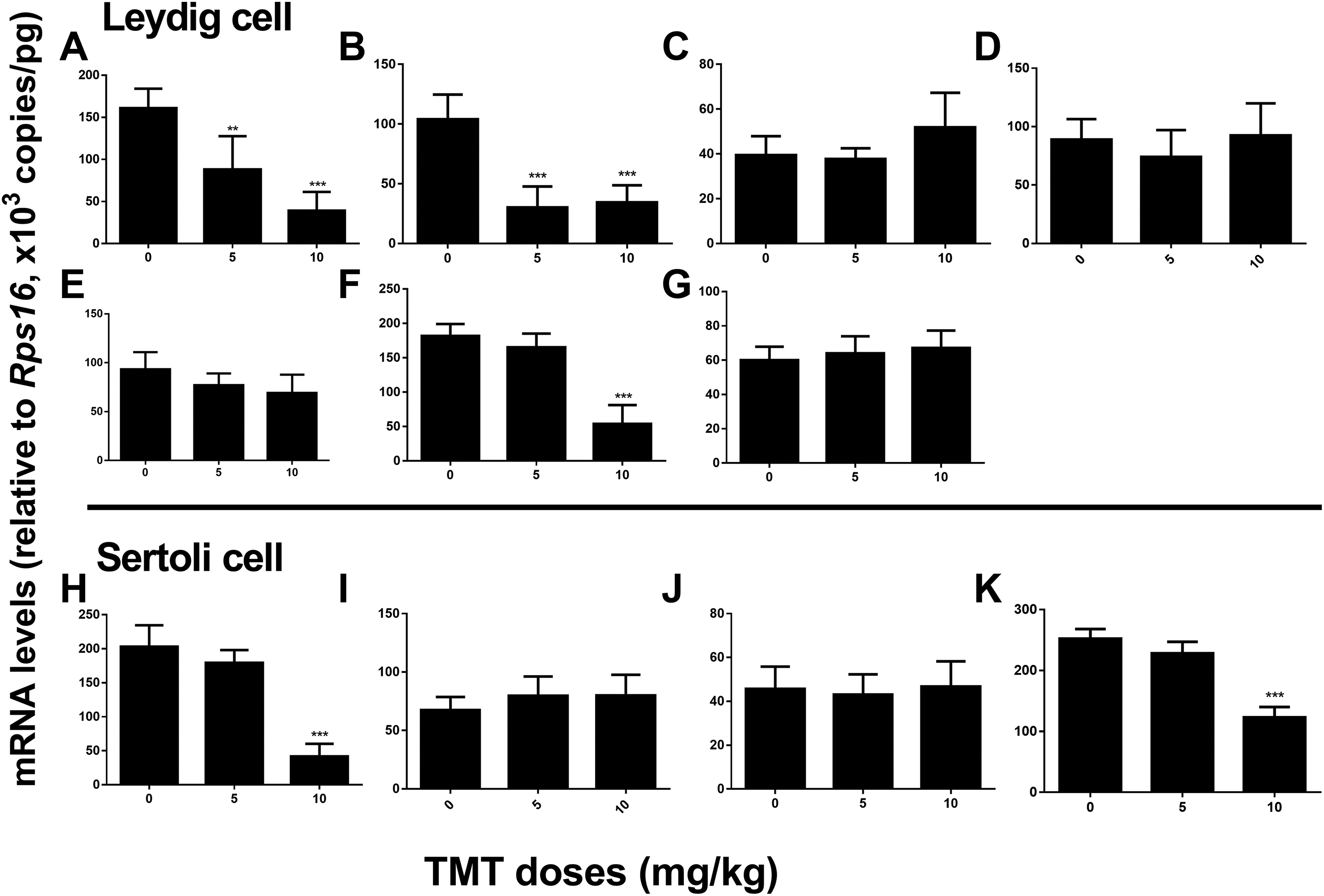
Expression levels of Leydig and Sertoli cell genes in the rat testes after TMT treatment. Leydig cell genes: (A)
Trimethyltin Decreases Protein Expression in Leydig and Sertoli Cells
As shown in Figure 3, for the Leydig cell-specific proteins, we found that TMT significantly decreased the expression of STAR at 5 and 10 mg/kg as well as that of LHCGR and CYP17A1 at 10 mg/kg, but had no effects on the expression of SCARB1, CYP11A1, 3β-HSD1, and 17β-HSD3 even at the highest dose (10 mg/kg). For the Sertoli cell-specific proteins, we found that TMT significantly decreased the expression of FSHR and SOX9 at 10 mg/kg, but had no effects on the expression of AMH and DHH even at the highest dose (10 mg/kg). Changes in protein expression were related to those in gene expression, which suggested that TMT selectively exerted negative effects on Leydig and Sertoli cell protein expressions.
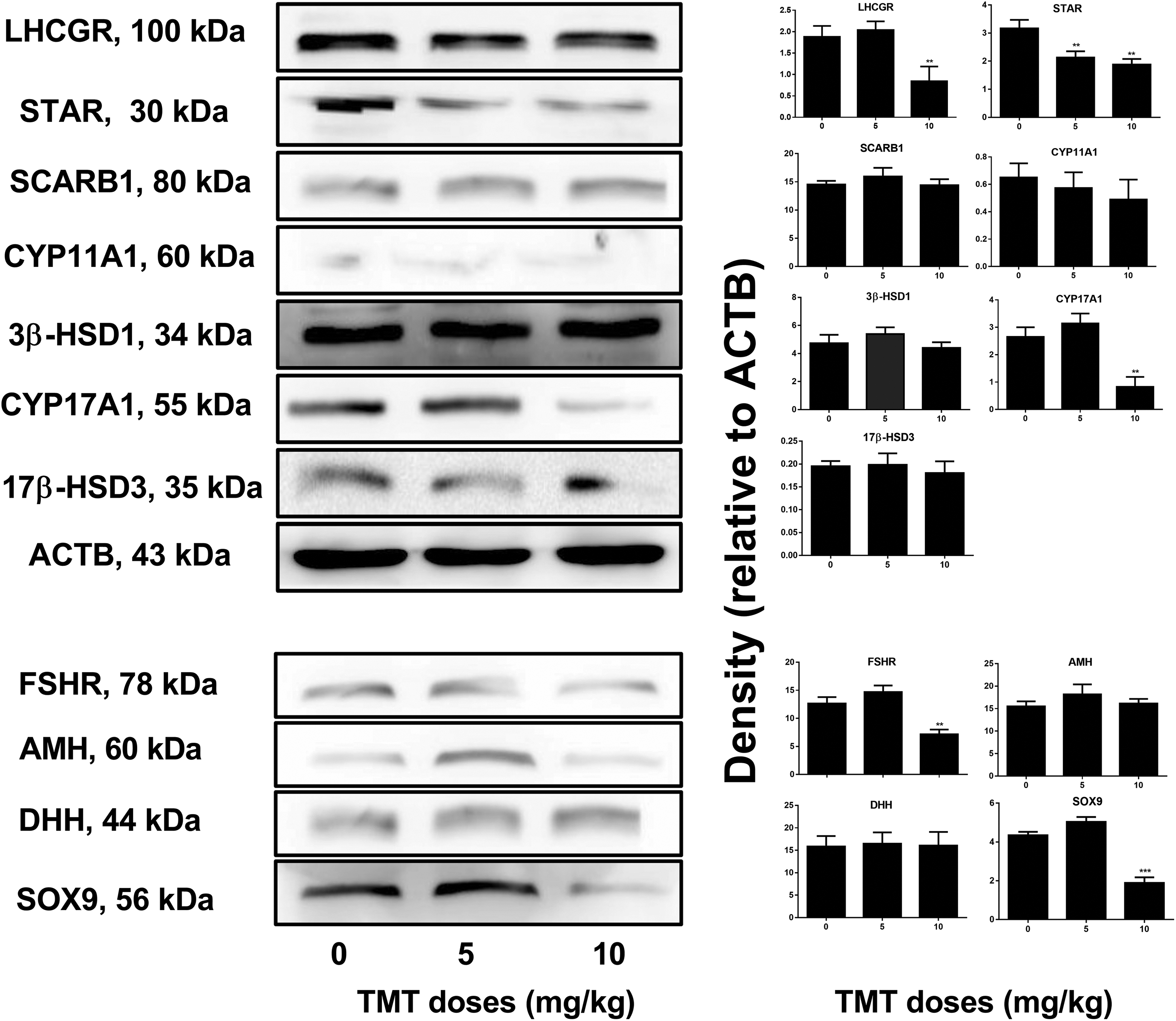
Expression levels of Leydig and Sertoli cell proteins in the rat testes after TMT treatment. A, Gel images. B, Quantitative results. Leydig cell proteins: LHCGR, STAR, SCARB1, CYP11A1, 3β-HSD31, CYP17A1, and 17β-HSD3. Sertoli cell proteins: FSHR, AMH, DHH, and SOX9. Mean ± standard error of the mean, n = 6. ∗, ∗∗, ∗∗∗ indicate significant difference when compared to the control (0 mg/kg TMT) at
Trimethyltin Decreases Adult Leydig Cell Number in the Testis
Leydig cells were stained for the biomarker 3β-HSD1 (Guo et al, 2013). As shown in Figure 4, TMT treatment significantly reduced 3-HSD1-positive cells in rat testis at 5 and 10 mg/kg. These results suggest that TMT decreases adult Leydig cell number in the testis.
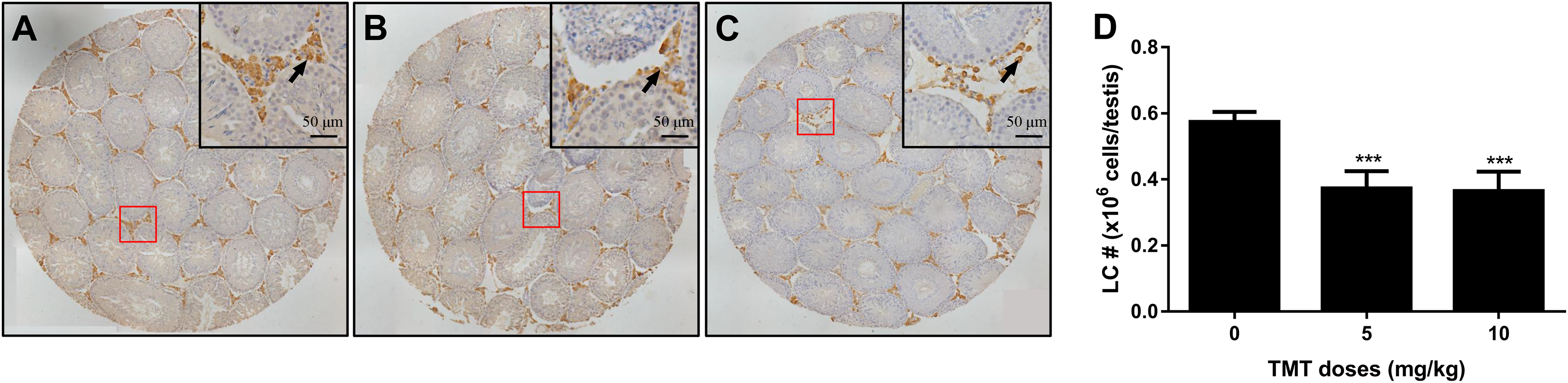
The effects of trimethyltin (TMT) treatment on Leydig cell (LC) number. Leydig cells were identified by immunohistochemical staining of 3β-HSD1 and LC number was enumerated by the stereological method: (A) for control group, (B) for 5 mg/kg TMT group, and (C) for 10 mg/kg TMT group. Bar = 50 μm. Black arrow points to Leydig cells. D, Quantification of LC number (#). Mean ± standard error of the mean, n = 6. ∗, ∗∗, ∗∗∗ indicate significant difference when compared to the control group at
Discussion
The testis interstitial cells called Leydig cells originate from stem Leydig cells which commit to progenitor Leydig cells, transform to immature Leydig cells, and finally to adult Leydig cells at 56 days postpartum. The cells secrete 95% to 99% of circulatory testosterone, 8 which is essential for many important physiological processes such as spermatogenesis and reproduction. 9 In adult Leydig cells, testosterone is synthesized from cholesterol through the action of many steroidogenic enzymes including LHCGR, SCABR1, STAR, CYP11A1, 3β-HSD1, CYP17A1, and 17β-HSD3, with STAR and CYP11A1 involved in the rate-limiting steps. 10 The pituitary hormone LH binds to its receptor LHCGR in adult Leydig cells to stimulate the expression and activities of steroidogenetic enzymes, which lead to increased testosterone production through the cAMP-PKA pathway. 11 -13 Leydig-supporting cells known as Sertoli cells promote the development and function of Leydig cells by secreting DHH and AMH. 14 -17 Follicle-stimulating hormone binds to FSHR on Sertoli cell surface and stimulates the Leydig cell-supporting function of Sertoli cells. SRY box 9 is a critical transcription factor involved in the developmental and maturation of Sertoli cells. 27
Trimethyltin is an organotin compound that is widely used in the industrial and agricultural fields as a plastic heat stabilizer, in antifouling paints for ship hulls, and as a fungicide. 1,2 Although lines of evidence have pointed to the severe toxicity of TMT 1,2,19 -21 and the higher TMT accumulation in the testes than in other tissues (3-5 g/g body weight), 28 there are no reports on the effects of TMT on Leydig cell steroidogenic enzyme expression and testosterone production. In the present study, the in vivo effects of TMT on rat Leydig cell testosterone production and the related regulatory mechanisms of action were investigated.
Trimethyltin at doses of 0 (vehicle control), 5, and 10 mg/kg were administered to 56-day-old male Sprague Dawley rats for 21 days consecutively. We found that TMT treatment not only downregulated the gene and protein expression levels of LHCGR, STAR, and CYP17A1 in adult Leydig cells but also decreased adult Leydig cell number in the testis. The decrease in LHCGR could render the adult Leydig cells incapable of responding to LH stimulus, and the downregulation of STAR and CYP17A1 could lead to the decrease in testosterone production of the Leydig cells under the same stimulus. Both the downregulation of these enzymes and the decrease in adult Leydig cell number contributed to the decrease in serum testosterone levels. Of particular note is the downregulation of STAR, an enzyme involved in the rate-limiting step of the transmitochondrial-membrane translocation during the biosynthesis of testosterone. 10 The decreased level of testosterone production provided negative feedback to increase the release of LH and FSH into the circulatory system through the hypothalamic–pituitary–gland axis. In addition to the inhibitory effects of TMT on Leydig cells, TMT also downregulated the FSHR and SOX9 expression levels, which are critical for Sertoli cell function and development. However, it is quite interesting to note that levels of AMH and DHH remained unaffected. This means that the support for Leydig cell development and function from Sertoli cells did not change after TMT treatment. In conclusion, the present study demonstrated for the first time that TMT reduced rat Leydig cell testosterone production in vivo by downregulating the expression of some steroidogenic enzymes and adult Leydig cell number in testis.
However, there may be other mechanisms through which TMT affects adult Leydig cell testosterone production. For example, TMT accumulation has also been found in kidney in mice, though the accumulation is less severe than it is in testis, 28 TMT in kidney caused hypokalemia, 29 and hypokalemia was demonstrated to decrease testosterone production in male mice. 30 Previous studies have confirmed the toxicity of TMT in rats at doses of 5 to 8 mg/kg, 23,24 and a study reported that the median lethal dose (LD50)
of TMT in rats is 12.6 mg/kg. 31 In the present study, doses of 0, 5, and 10 mg/kg were administered and the in vivo toxic effects of TMT on Leydig cells were evaluated for the first time. The administration of TMT to rats at a dose of 5 mg/kg resulted in whole blood and plasma accumulation of TMT with concentrations as high as 323 and 4.7 nmol/mL, respectively. 31 Trimethyltin accumulation in humans typically occurs by inhalation; therefore, workers should use special masks to avoid occupational exposure. 32 Further studies are required to clarify the other possible effects of TMT on Leydig cells. Some studies have demonstrated that organotins such as dibutyltin dichloride and tributyltin retard the regeneration of rat adult Leydig cells 8,27 ; thus, a similar study is worth conducting on TMT, which is also a widely used organotin. 1,2
Supplemental Material
Supplemental Material, DS1_IJT_10.1177_1091581819870719 - Trimethyltin (TMT) Reduces Testosterone Production in Adult Leydig Cells in Rats
Supplemental Material, DS1_IJT_10.1177_1091581819870719 for Trimethyltin (TMT) Reduces Testosterone Production in Adult Leydig Cells in Rats by Derong Ma, Nengqin Luo and Guoqiang Xue in International Journal of Toxicology
Supplemental Material
Supplemental Material, DS2_IJT_10.1177_1091581819870719 - Trimethyltin (TMT) Reduces Testosterone Production in Adult Leydig Cells in Rats
Supplemental Material, DS2_IJT_10.1177_1091581819870719 for Trimethyltin (TMT) Reduces Testosterone Production in Adult Leydig Cells in Rats by Derong Ma, Nengqin Luo and Guoqiang Xue in International Journal of Toxicology
Footnotes
Acknowledgments
The authors would like to thank Xiande Huang for critical comments and advice on this article.
Author Contributions
Derong Ma and Nengqin Luo contributed equally to this work. X. Guoqiang substantially contributed to conception or design and critically revised the manuscript for important intellectual content. M. Derong substantially contributed to conception or design, contributed to acquisition, analysis, or interpretation of data, drafted the manuscript, and critically revised the manuscript for important intellectual content. L. Nengqin contributed to acquisition, analysis, or interpretation of data and drafted the manuscript. All authors gave final approval and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by National Natural Science Foundation of China (number 81460456).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
