Abstract
Thymomas occur prevalently in aged Wistar Hannover (WH) rats, along with hyperplastic lesions that cannot be categorized as thymomas. We compared the histological features of hyperplastic lesions and thymomas in WH rats, the incidences of these lesions, and the relationship of these lesions to the degree of thymic involution and also compared these lesions with those of Sprague Dawley (SD) rats in 4-, 13-, 26-, and 104-week studies. There were no morphological differences between hyperplastic cells and benign tumor cells in thymomas. The histological difference between hyperplastic lesions and thymomas was the size of the proliferative areas and the number of medullary differentiation areas. The hyperplastic lesions of the thymus in WH rats might have a potential for progression to thymomas due to the observed multiple hyperplastic lesions or mixed lesions with thymomas. The incidence of these proliferative lesions in the thymus was higher in females than in males. Further, the incidence of these proliferative lesions was higher in WH rats than in SD rats. Thymic involution was more severe in males than in females and more severe in SD rats than in WH rats. The differences in involution progression may have been reflected in the incidence of thymic proliferative lesions in SD and WH rats.
Thymomas are prevalent tumor in Wistar Hannover (WH) rats, with an incidence of 1.31% to 2.5% in male and 3.28% to 13.4% in female Crl:WI(Han) and RccHan:WIST strains (Blankenship et al. 2016; Weber et al. 2011; Weber 2017). Thymomas with medullary differentiation are commonly seen in WH rats (Iwata et al. 2013; Elmore et al. 2015; Kuper, Beems, and Hollanders 1986), while in Fisher rats or Sprague Dawley (SD) rats used frequently in toxicological studies, the incidence of thymomas is lower, specifically, 0.16% in male and 0.41% in female Fisher rats and 0.42% in male and 0.42% in female Crl:CD(SD) rats (Weber 2017). Certain strains of rats such as (ACI/NMs × BUF/Mna)F1, W/Nhg, and WAB (castrated or spayed) have a high incidence of thymomas and an early onset of tumor development (Hinsull and Bellamy 1977; Matsuyama et al. 1988; Murray et al. 1985). Interestingly, (ACI/NMs × BUF/Mna)F1 rats exhibit lesions that occur as multiple small nodules and further develop into medium to large overt thymomas as a result of growth and fusion (Matsuyama et al. 1988).
We have often noted hyperplastic lesions that cannot be categorized as either thymomas or as persistent thymuses in carcinogenicity studies in WH rats. A persistent thymus has a normal architecture but is considerably larger than expected for the animal’s age (Kuper et al. 1992). We have histopathologically observed focal or lobular hyperplastic lesions with distorted cortex–medullary structures in the thymus with or without thymomas. To our knowledge, however, such hyperplastic lesions of the thymus in WH rats have not been previously described.
To characterize these unidentified lesions, we examined the incidence of hyperplastic lesions, the number of hyperplastic foci per section and the histological features of the hyperplastic lesions compared to those of thymomas, and whether a hyperplastic lesion has any significance in the pathogenesis of thymomas. We further hypothesized that the thymus of aged WH rats, compared to that of SD rats, exhibits delayed involution and has a greater tendency to be affected by proliferative conditions. We therefore examined the progression of thymic involution in WH and SD rats to explain the strain difference in thymoma incidence.
Materials and Methods
Animals
WH rats were used in 4-, 13-, 26-, and 104-week background-collecting studies. The RccHan™:WIST strain was purchased from Japan Laboratory Animals, Inc. (Saitama, Japan), and Crl:WI(Han) was purchased from Charles River Laboratories Japan (Yokohama, Japan; Table 1). To compare thymic lesions among rat strains, the thymuses of SD rat strains, namely, Crl:CD(SD) and Crj:CD(SD)IGS, used in 4-, 13-, 26-, and 104-week background-collecting studies were examined. SD rats were purchased from Charles River Laboratories Japan. The number of animals of each strain is shown in Table 1.
Number of Animals Examined for Histopathology in Wistar Hannover (WH) and Sprague Dawley (SD) Rats.
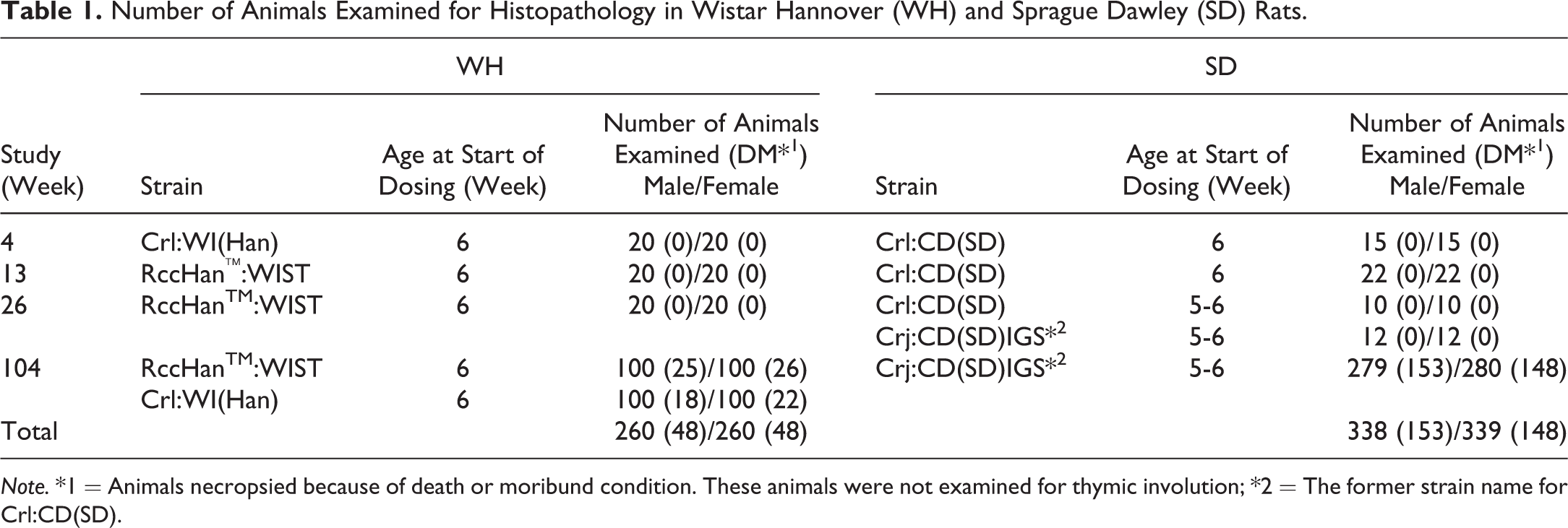
Note. *1 = Animals necropsied because of death or moribund condition. These animals were not examined for thymic involution; *2 = The former strain name for Crl:CD(SD).
Rats were individually housed in hanging-type stainless steel wire mesh cages (195 mm[w] × 325 mm[d] × 180 mm[h]; Tokiwa Kagaku Kikai Co., Ltd., Tokyo, Japan) or polycarbonate cages (265 mm[w] × 426 mm[d] × 200 mm[h]; Tokiwa Kagaku Kikai Co., Ltd., Tokyo, Japan) in an animal room kept at a temperature of 22°C ± 3°C with 55% ± 20% humidity, 6 to 20 air changes/hr, and a 12-hr light/dark cycle. A pellet diet for experimental animals (radiation sterilized, CR-LPF; Oriental Yeast Co., Ltd, Japan) and tap water passed through a 5-µm filter and irradiated with ultraviolet light were supplied ad libitum. The animals were anesthetized by intraperitoneal injection of sodium thiopental (Ravonal®; Mitsubishi Tanabe Pharma Corporation) and euthanized by exsanguination from the abdominal aorta and then subjected to necropsy. The animals were cared for and euthanized according to the principles outlined in the Guidelines for the Care and Use of Laboratory Animals by the Japanese Association of Laboratory Animal Science and the guidelines of our institution.
Thymic Weight
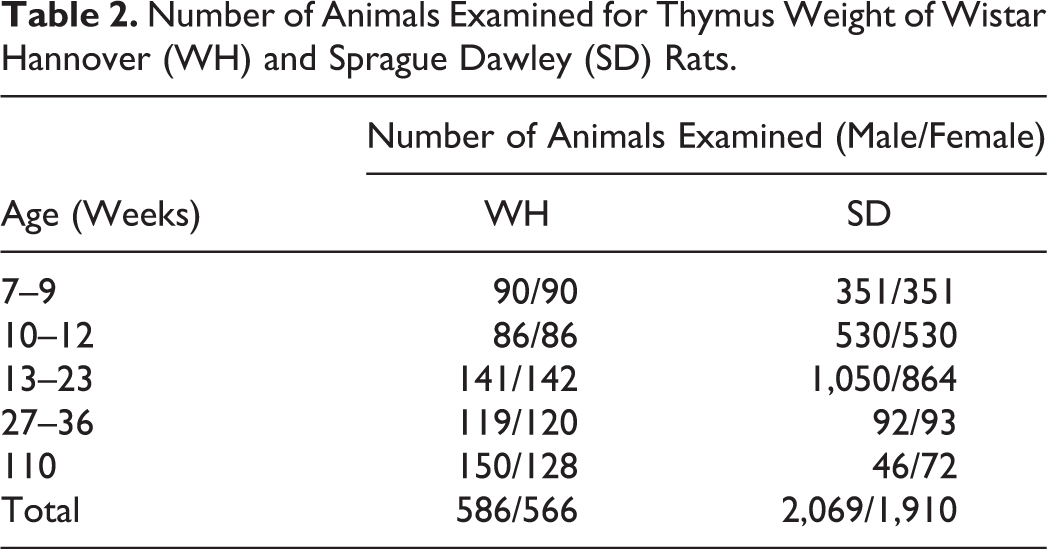
The number of rats in each age-group is shown in Table 2. To compare the thymic weights of sacrificed animals without thymic nodules, we collected the background data of control animals obtained in toxicological studies, 586 male and 566 female WH rats and 2,069 male and 1,910 female SD rats at 7 to 9, 10 to 12, 13 to 23, 27 to 36, and 110 weeks of age. However, the thymic weight measured at 110 weeks of age included the thymic interparenchymal fatty tissue because it was impossible to separate these tissues once thymic involution had progressed.
Number of Animals Examined for Thymus Weight of Wistar Hannover (WH) and Sprague Dawley (SD) Rats.
Pathological Evaluation
Thymuses sampled at necropsy were fixed in 10% phosphate buffer formalin, trimmed as cross section through the center of the thymus, embedded in paraffin, and sectioned at 4 μm. Hematoxylin and eosin (HE) staining was performed according to standard methods for light microscopic examination. The incidence of thymomas, hyperplastic lesions of the thymus, and thymic involution were examined in each strain. Animals that died or were sacrificed in moribund conditions were excluded from the thymic involution data because animal condition has been shown to influence thymic involution (Greaves 2012; Haley 2013).
The criteria used to define hyperplastic lesions, thymomas, and involution are described below. A hyperplastic lesion was defined as a focus of an increased number of thymocytes with a cortical–medullary structure. However, the lesion showed little to no expansive growth or compression of adjacent tissues; the size did not exceed the width of the minor axis of the normal thymus at the same age. The architecture of the cortex and medulla was distorted. Thymomas exhibited a distorted thymic architecture and were divided into two main types: those containing abundant lymphocytes with or without “medullary differentiation” (Kuper et al. 1992). The size of the tumor was larger than the width of a normal thymus. Thymic involution was divided into three grades (mild, moderate, and severe) according to the size of the remaining cortical and medullary areas (Figure 1). “No significant change” indicated the absence of any obvious involution, maintaining the lobular pattern with cortical and medullary architecture as observed in a normal thymus of the same age. Mild (+) indicated that the thymus and lobule size were slightly decreased, but that the cortex and medullary structures were maintained; moderate (++) indicated that the thymus and lobule size were smaller than half of the normal thymus and that the number of lymphocytes was significantly decreased; and severe (+++) indicated that the thymus was very small, that the lobule, cortex, and medullary structures were not defined, and that fat infiltration into the thymic parenchyma was prominent.
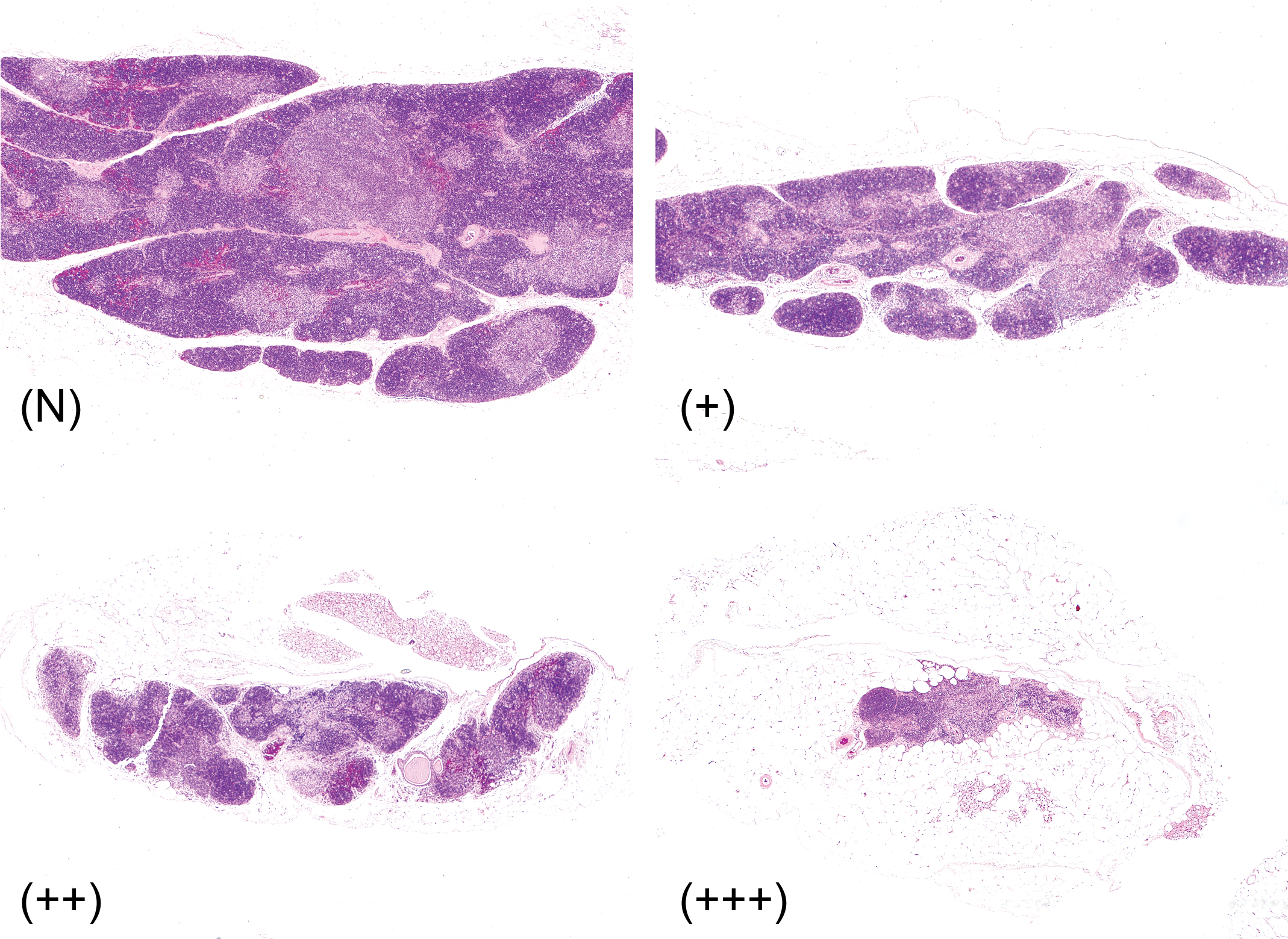
Involution of the thymus. No significant change (N), mild (+), moderate (++), severe (+++). The images with N to moderate grade changes were obtained from sacrificed female Wistar Hannover rats in a 104-week background-collecting study. The image with severe grade changes was obtained from a sacrificed female Sprague Dawley rat in a 104-week background-collecting study. Hematoxylin and eosin.
Morphometrical Analysis
Histopathological pictures of the thymus diagnosed with thymomas and hyperplastic lesions were inputted into a computer system (Digital pathology whole slide scanner: Aperio AT2, S. No. 7277, Leica Microsystems GmbH, Wetzlar, Germany), and the maximum length of the minor and major axes of each lesion was calculated. For comparison, the same calculations were performed on histopathological images of thymuses diagnosed with no significant change. The number of hyperplastic lesions and thymomas per thymus specimen was also calculated.
Results
Hyperplastic Lesion
Hyperplastic lesions could not be observed macroscopically because they were smaller or equivalent to the width of the minor axis of a normal thymus of the same age (Figure 2). Hyperplastic lesions were noted as areas with slight borders separated from normal thymic tissue and showed little to no expansive growth pattern but had a distorted cortex and medulla, such as an increase in the ratio of the cortical area to the medullary area (Figure 3). Lobules tended to be round, with a cortical area larger than that of a normal thymus cortex. Other than the medullary area, a few foci containing pale-staining cells were detected in hyperplastic lesions. The foci were smaller than the medullary area (Figure 3, arrows). The number of lesions per thymus is shown in Table 3. Six of the nine cases of hyperplastic lesions showed single lesions, while the remaining three occurred as multiple lesions (Figure 3). In 12 cases, hyperplastic lesions were found to coexist with thymomas (Figure 4).
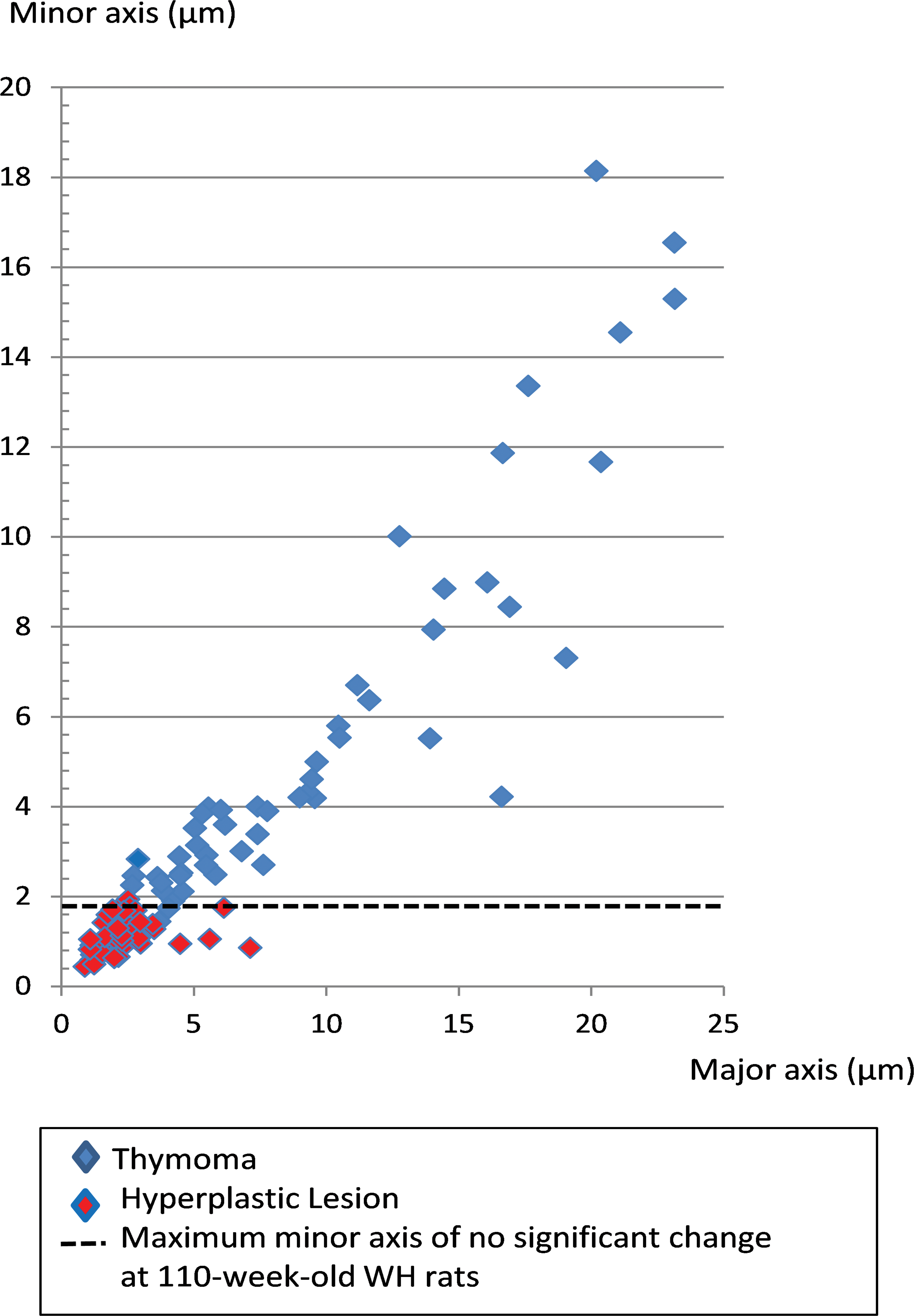
Minor and major axes of hyperplastic lesions and thymomas in Wistar Hannover (WH) rats. Hyperplastic lesions were smaller or equivalent to the width of the minor axis of a normal thymus (no significant change) of 110-week-old WH rats. Thymomas were equivalent to or larger than the width of the minor axis of a normal thymus.
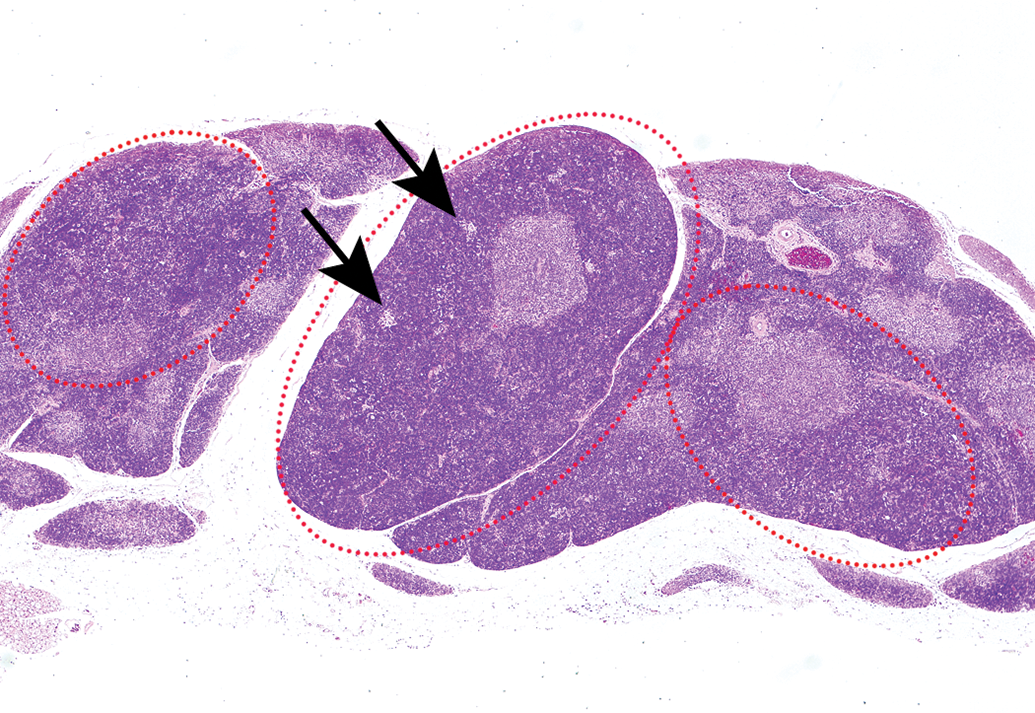
Multiple hyperplastic lesions were found in the thymus of 110-week-old female Wistar Hannover rats (red-dotted circles). The lesions showed little to no expansive growth pattern but had a distorted cortex and medulla, such as an increase in the ratio of the cortical area to the medullary area. A few foci containing pale-staining cells were detected in the hyperplastic lesions (arrows). Hematoxylin and eosin.
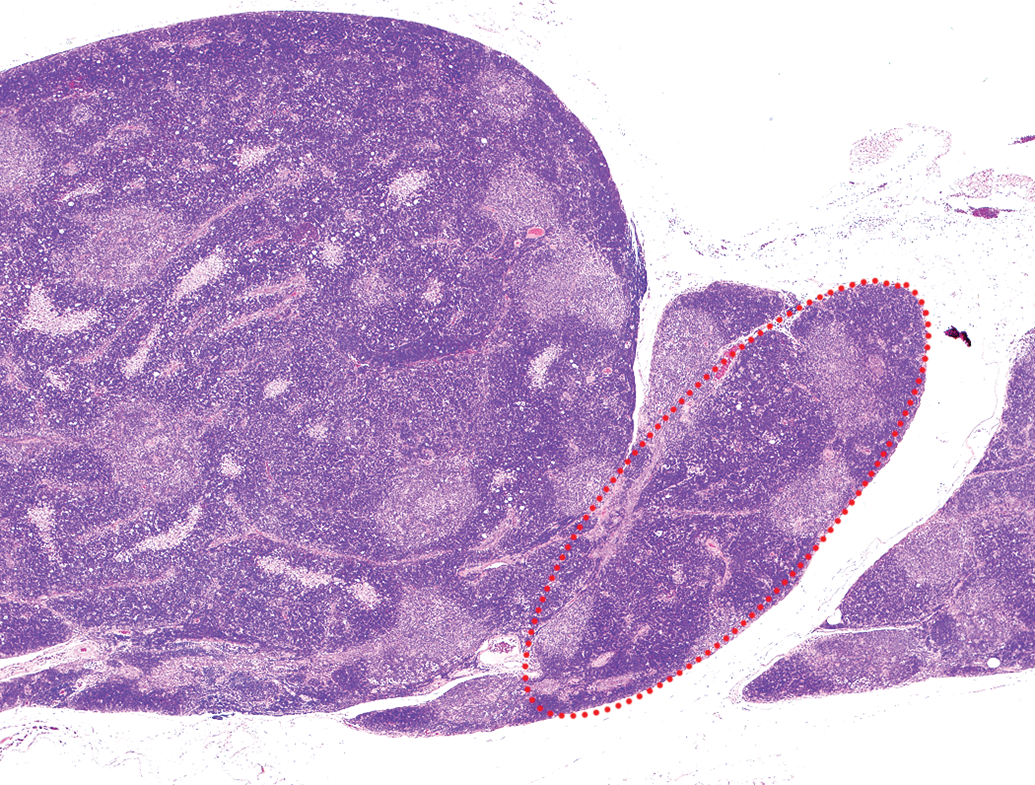
Hyperplastic lesion (red dotted circle) that was coexisting and continuous with a thymoma. Wistar Hannover rat, female, 110-week-old. Hematoxylin and eosin.
Animals Classified by the Number of Lesions in the Thymus of Wistar Hannover Rats.

Hyperplastic lesions were noted in 9/200 (4.5%) of female WH rats in a 104-week study, while none were observed in males (Table 4). Hyperplastic lesions were first noted histopathologically at 102-week-old female WH rats (Figure 5).
The Incidence of Thymomas and Hyperplastic Lesions of Wistar Hannover (WH) and Sprague Dawley (SD) Rats.

anumber of males/females examined.
bIncidence (%) = number of animals with lesions/number of animals examined.
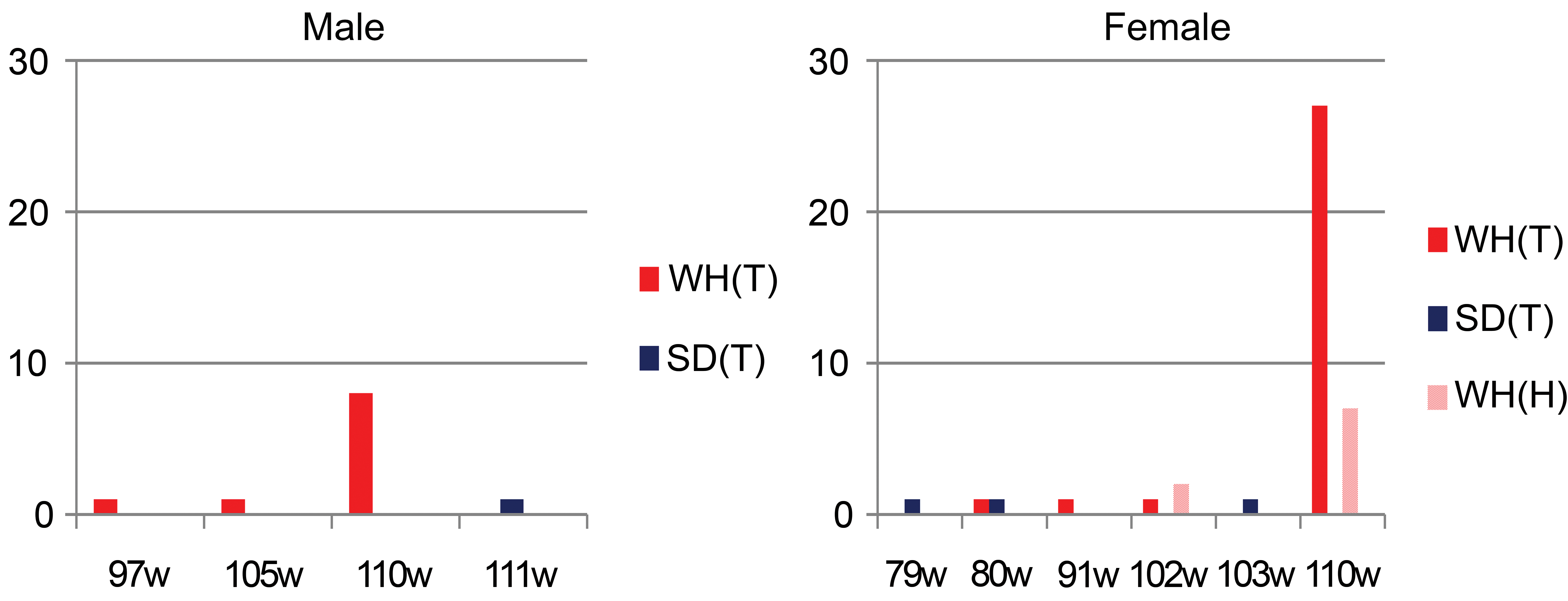
Chronological incidence of thymomas (T) and/or hyperplastic lesions (H) in Wistar Hannover (WH) and Sprague Dawley (SD) rats. The y-axis indicates the number of animals with thymomas or hyperplastic lesions, and the x-axis indicates the age of the animals. The earliest occurrence of thymomas was at 97-week-old male and 80-week-old female WH rats. Hyperplastic lesions were histopathologically noted at 102-week-old female WH rats.
Thymoma
Thymic masses in WH rats were found in 9/200 (4.5%) males and 30/200 (15%) females including moribund and dead animals at necropsy following a 104-week study. All nine masses in males and 28/30 masses in females were diagnosed as thymomas. The remaining two masses in females were diagnosed as histiocytic sarcoma and metastasis of malignant schwannoma, respectively. The masses of thymomas expanded with clear margins and variable size.
The incidence of thymomas is shown in Table 4. Microscopically, thymomas were not detected in 4-, 13-, or 26-week studies of WH and SD rats. In a 104-week study, thymomas occurred more frequently in females than in males, with an incidence of 10/200 (5.0%) in male and 30/200 (15.0%) in female WH rats and an incidence of 1/338 (0.3%) in male and 3/339 (0.9%) in female SD rats. The incidence of thymomas in both sexes was markedly higher in WH rats than in SD rats. The chronological incidence of thymomas and/or hyperplastic lesions in WH or SD rats indicated that these proliferative lesions occurred only in aged animals in both strains (Figure 5). The earliest occurrence of thymomas in WH rats, identified by incidental findings in moribund-sacrificed or dead animals, was at 97-week-old males and 80-week-old females (Figure 5).
Thymomas in WH rats showed expansive growth with single or multiple proliferative areas that sometimes exhibited lobular or nodular patterns separated by thin fibrous tissues (Figure 6a). Among the forty cases of thymomas, twenty-one tumors consisted of a single nodule, while seven tumors consisted of a few nodules (Figure 6a; Table 3). As the most characteristic feature, the structure of the WH thymomas consisting of the cortex and medullary differentiation areas resembled normal thymic architecture (Figure 6b). The cortex-like area was a lymphocyte-rich area, exhibiting a large number of lymphocytes among the epithelial cells (Figure 6c). No tumors showed invasion and/or metastasis to other tissues to indicate malignancy, although multiple lymphocytic infiltrations were noted in the surrounding adipose tissues. The area of medullary differentiation was of variable size and consisted of epithelial cells accompanied by lymphocytes, resembling the normal medulla of the thymus (Figure 6b and c). Some Hassall bodies were observed in the areas of medullary differentiation. Other than the area of medullary differentiation, many foci containing pale-staining cells were detected in thymomas. The foci were smaller than the area of medullary differentiation and comprised large round cells with a clear cytoplasm, containing little or no lymphocytes, distinguishing them from the medullary area (Figure 6b and d).
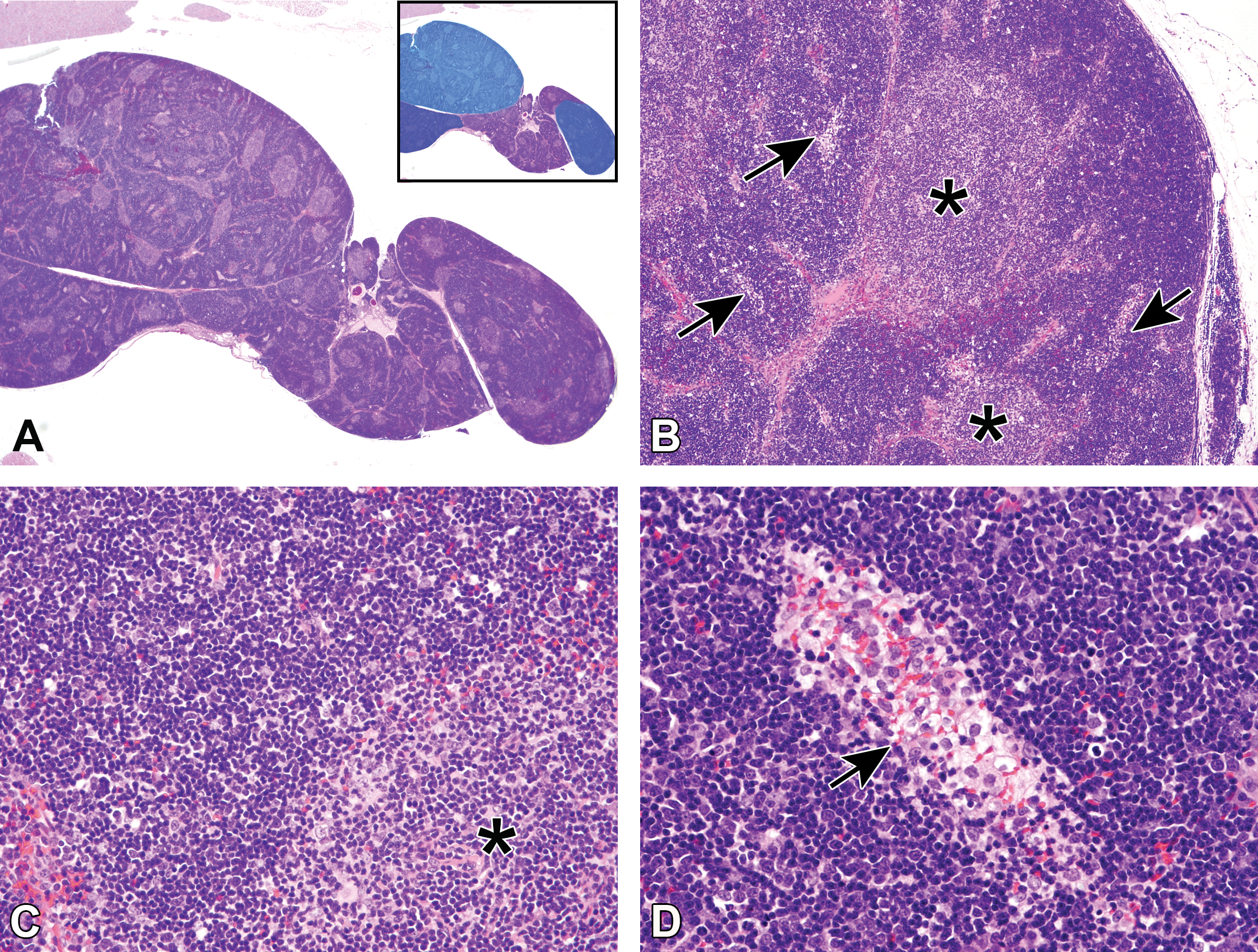
Histopathological features of thymomas of 110-week-old female Wistar Hannover rats. Hematoxylin and eosin. (a) Thymomas showed expansive growth with multiple proliferative areas that exhibited lobular or nodular patterns separated by thin fibrous tissue. This thymoma consisted of three nodules (blue areas, inset). (b) The cortical and medullary (asterisks) differentiation areas imitated the normal thymic architecture. The area of medullary differentiation was of variable size and resembled the normal medulla of the thymus. Foci of pale-staining cells were found in lymphocyte-rich areas (arrows). These foci were smaller than the area of medullary differentiation. (c) The cortex-like area was a lymphocyte-rich area, exhibiting a large number of lymphocytes among the epithelial cells. The area of medullary differentiation (asterisk) consisted of epithelial cells accompanied by lymphocytes, resembling the normal medulla of the thymus. (d) This focus of pale-staining cells (arrow) was composed of large round cells with a clear cytoplasm and contained few or no lymphocytes.
There were no morphological differences between hyperplastic cells and benign tumor cells in thymomas. The histological difference between hyperplastic lesions and thymomas was the size of the proliferative areas and the number of medullary differentiation areas and pale-staining cell foci.
Thymic Involution
Thymic involution in WH rats started later and was less severe than that in SD rats (Figure 7). Involution was noted in 27.2% of male and 4.5% of female SD rats but was not observed in WH rats at 26-week-old. At 104-week-old, involution was observed in almost all SD rats of both sexes and in 86.6% of male and 65.8% of female WH rats. The severity of involution in WH rats progressed to grade (+) and (++) while that in SD rats progressed to grade (+) to (+++) at 104-week-old. In both strains, thymic involution was more severe in males than in females.
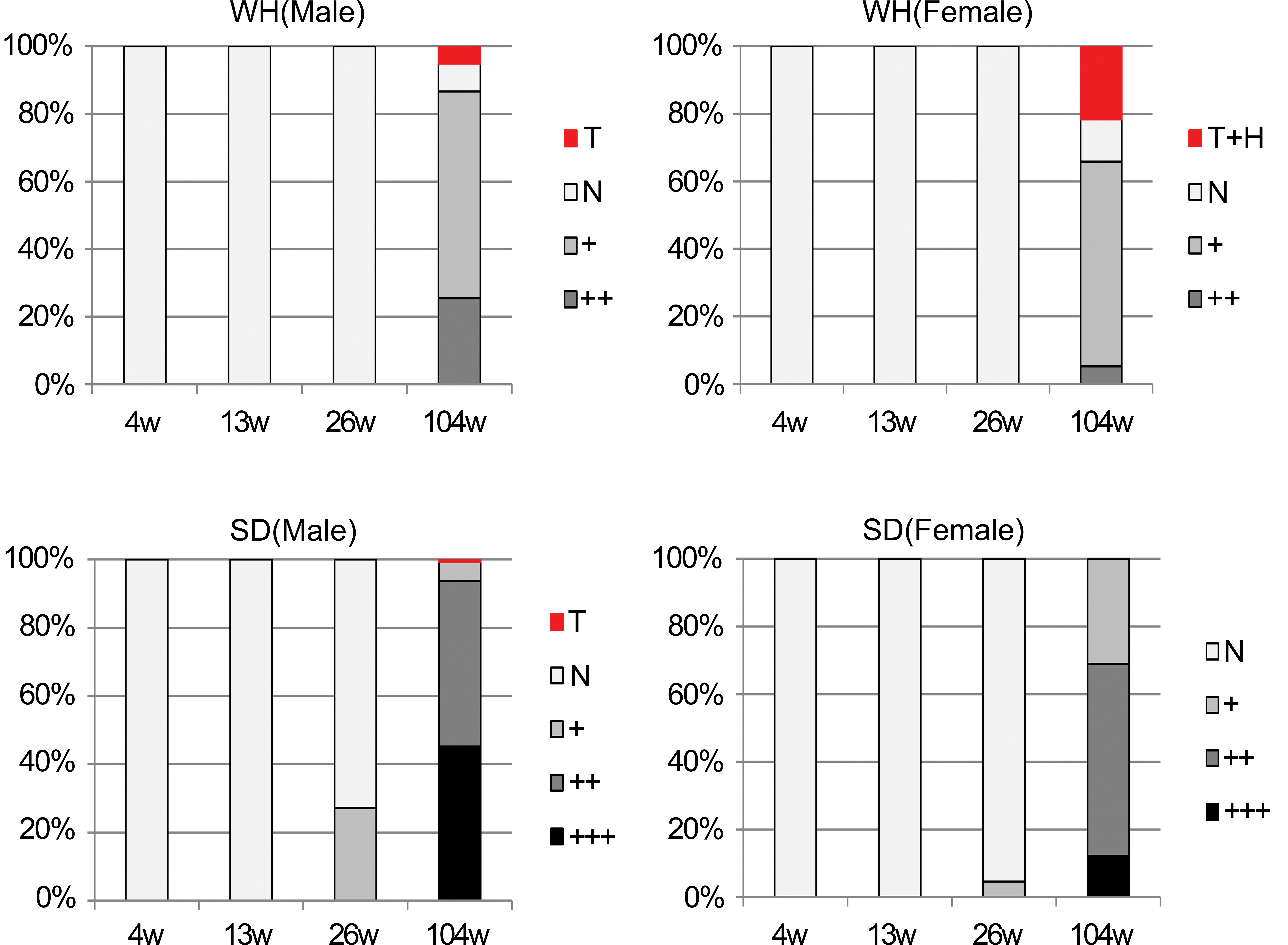
Incidence of thymic involution in Wistar Hannover and Sprague Dawley rats. The y-axis shows the incidence (%), calculated by the number of animals with findings/number of examined animals. The x-axis shows the period of the study. Data were obtained from the sacrificed animals at each of the scheduled periods. T = thymomas; T + H = thymomas and/or hyperplastic lesions; N = no significant change; + = mild; ++ = moderate; +++ = severe.
Thymic Weight
From 7- to 36-week-old, there was no difference in the mean thymic weight between WH rats and SD rats. However, the mean relative thymic weight in WH rats was greater than that in SD rats because WH rats are smaller than SD rats (Figure 8). At 110-week-old, the relative thymic weight in SD rats became equivalent to that in WH rats because the thymic weight in SD rats included that of replaced fatty tissue due to involution.
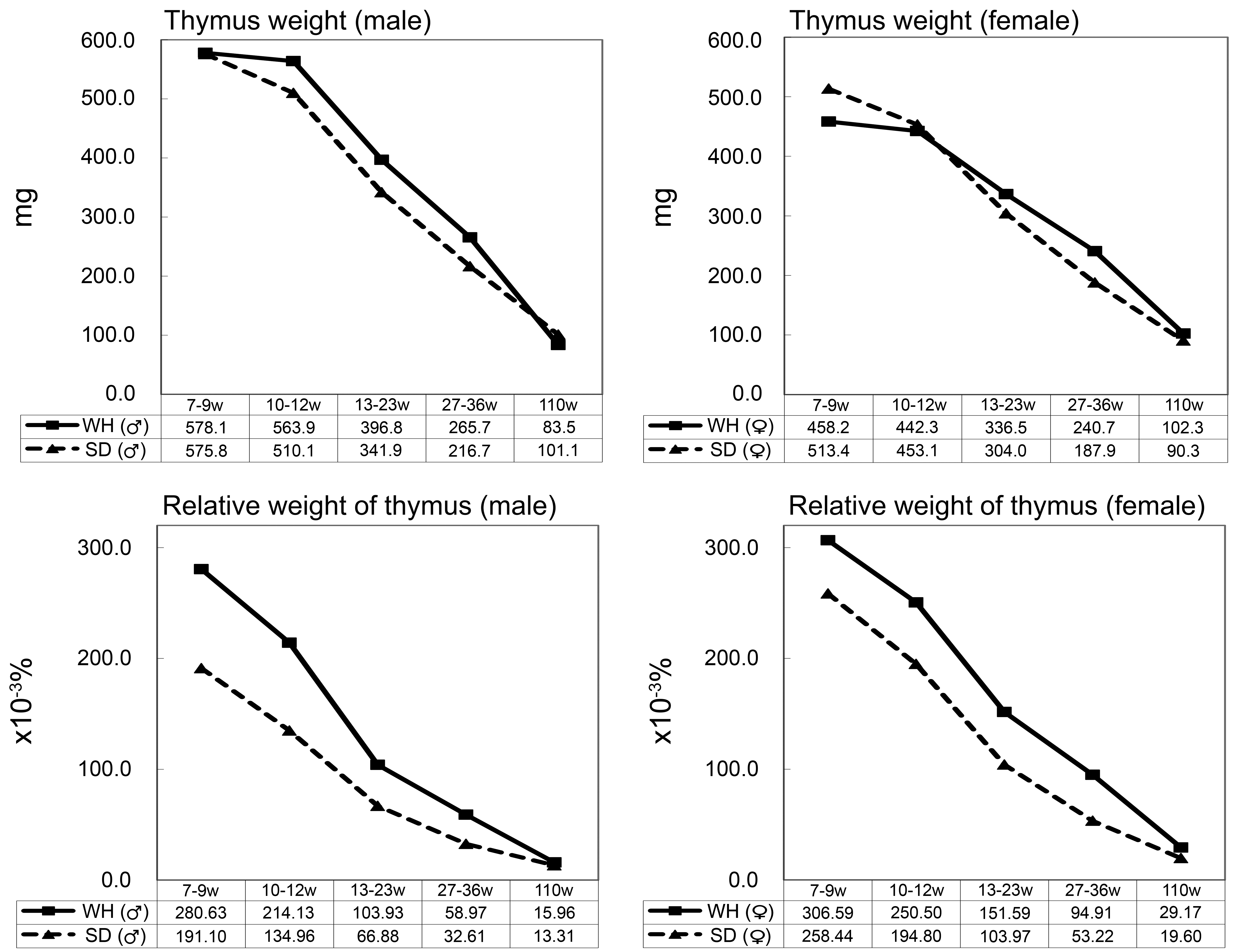
Thymic weight and relative thymic weight of Wistar Hannover (WH) and Sprague Dawley (SD) rats. From 7-week-old onward, the relative weight of the thymus of WH rats was greater than that of SD rats, although there was no difference in mean weights.
Discussion
Many hyperplastic lesions and thymomas were observed at approximately 110-week-old WH rats. The incidences of both hyperplastic lesions and thymomas were higher in females than in males. Multiple hyperplastic lesions were sometimes observed. Additionally, thymomas consisting of nodules and those with hyperplastic lesions comprising mixed lesions were observed in one thymus. These morphological features suggest that hyperplastic lesions occur as multiple lesions in aged WH rats, and some of these lesions progress to form thymomas after expanded growth or fusion with other hyperplastic lesions. The hyperplastic lesions in aged WH rats might have a potential for progression to thymomas. BUN/Mna rats have been reported to have a high incidence of thymomas that occur as multiple small nodules that further develop into thymomas as in WH rats. BUN/Mna rats harbor the thymoma susceptibility gene (Tsr-1; Matsuyama et al. 1988); however, this genetic factor was not examined in the present study.
We have often noted the presence of thymic hyperplastic lesions that could not be categorized as thymomas in carcinogenicity studies in WH rats; however, there have been few reports on hyperplasia in the thymus of WH rats, although it has been reported in other rat strains and mice (Greaves 2012; Kuper et al. 1992; Kuper and Beems 1990; Kuper, Beems, and Hollanders 1986; Pearse 2006; Stewart and Snell 1968). Pearse examined various types of thymomas in rodents and characterized a number of hyperplastic lesions including epithelial hyperplasia, lymphoid hyperplasia, and atypical hyperplasia; epithelial hyperplasia refers to lesions with a thymic epithelium or duct proliferation with thymic atrophy; lymphoid hyperplasia refers to lesions with lymph follicle formation in the medulla or localized lymphoid hyperplasia extending from the medulla into the adjacent cortex in mice; and atypical hyperplasia is a precursor to induced thymic lymphoma but not thymomas (Pearse 2006). However, none of these categories is applicable to the thymic hyperplasia observed in WH rats (Blankenship et al. 2016). Kuper and Beems (1990) described a persistent or hyperplastic thymus in aged Wistar rats; the lesions were larger than the normal thymus of adult or aged rats involving the whole organ and had a normal cortical and medullary architecture. These descriptions likewise do not suitably describe the hyperplastic lesions observed in our investigation of WH rats.
The relative organ weight of the thymus was greater in WH rats than in SD rats at all stages, and thymic involution was slower in WH rats than in SD rats. Additionally, thymic involution was slower in females than in males. These differences may underlie the high incidence of hyperplastic lesions and thymomas in WH rats, especially in females, compared to SD rats. In the investigation of thymic spontaneous lesions in Wistar (Cpb:WU) rats, the incidence of thymomas is higher in females than males; additionally, severe thymic involution occurs more frequently in males than in females (Kuper, Beems, and Hollanders 1986). Persistent thymic tissue in aged animals might constitute a contributing factor to thymic epithelium proliferation.
The thymic epithelium contributes to the maturation (and proliferation) of lymphocytes regulated by binding of sex hormones (Seiki and Sakabe 1997). Sex steroid receptors are expressed on thymic stromal cells (McMurray et al. 2000). Prepubertal ovariectomy induces thymomagenesis, which is almost completely inhibited by estrogen compensation in BUF/Mna rats (Seiki and Sakabe 1997). On the other hand, estrogen has been shown to induce thymic atrophy, similar to that of glucocorticoids (McMurray et al. 2000). The cause of the higher incidence of hyperplastic lesions and thymomas in female WH rats than SD rats may be related to hormonal differences.
A large population of epithelial cells has been observed in thymomas (except for areas of medullary differentiation) in WH rats by using immunohistochemical staining for cytokeratin-18 (Elmore et al. 2015). We tried to count the incidence of Ki67-positive cells including thymic epithelial cells and lymphocytes. There was no difference between the incidences of no significant change, hyperplastic lesions, and thymomas (data not shown). Further investigation of immunohitochemical staining for cytokeratin is needed, because proliferation of the epithelium was not evident in HE-stained sections due to the large number of lymphocytes within thymoma-masked epithelial cells.
In conclusion, hyperplastic lesions of the thymus observed in WH rats might have a potential for progression to thymomas. The incidence of these proliferative lesions in the thymus was higher in female than in male WH rats. Furthermore, the incidence of these proliferative lesions was higher in WH rats than in SD rats. Thymic involution was more severe in males than in females and more severe in SD rats than in WH rats. The differences in involution progression may have been reflected in the incidence of thymic proliferative lesions in SD and WH rats.
Footnotes
Acknowledgment
We gratefully acknowledge the language editing by Ms. Kanae Tamatsukuri and James Harada.
Author Contribution
Authors (YT, JS, TK, YW, TKan, MT) contributed to conception or design; data acquisition, analysis, or interpretation (YT, JS, TK, YW, TKan, MT); drafting the manuscript (YT, JS, TK, YW, TKan, MT); and critically revising the manuscript (YT, JS, TK, MT). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
