Abstract
Intranuclear and cytoplasmic inclusions in the renal proximal tubular epithelium were observed in nontreated male and female Wistar Hannover rats in a 26-week study (32 weeks of age) and a 104-week study (110 weeks of age). The incidence rates were less than 5% in these two studies. In affected animals, the inclusions were observed in more than 60% of proximal tubular epithelium as various sized (approximately 1–8 μm in diameter) round and eosinophilic materials, but not in distal tubules, Henle’s loop, or collecting ducts. Ultrastructurally, inclusions appeared finely granular, homogenous with middle-electron density, and without a limiting membrane. These inclusions were determined to be protein histochemically stained by Azan-Mallory and immunoreactive with an antibody against
Introduction
There are few reports about the spontaneous formation of intranuclear and cytoplasmic inclusions in the renal tubular epithelium of laboratory animals. Naturally occurring acidophilic crystalline intranuclear inclusions in the renal tubular epithelium and hepatocytes in dogs are well known, but the pathogenesis or clinical significance is still unexplained (Thompson, Cook, and Hoey 1959).
Recently, it was reported that administration of two different types of compounds, propiverine hydrochloride (1-methylpiperid-4-yl 2,2-diphenyl-2-(1-propoxy)acetate hydrochloride: propiverine) and norepinephreine (NE)/serotonin (5-HT) reuptake inhibitor (NSRI), induced intranuclear and cytoplasmic inclusions in the renal proximal tubular epithelium in male and female rats as a result of accumulation of
A few years ago, RccHanTM:WIST (Wistar Hannover) rats were introduced to Japan as experimental animals for toxicity testing of pharmaceuticals. However, relatively little background data are available on Wistar Hannover rats (Okamura et al. 2011; Yamatoya et al. 2012). In a background data study gathered from a 26-week study and carcinogenicity study that used Wistar Hannover rats, we found spontaneous intranuclear and cytoplasmic inclusions in the proximal tubular epithelium. Affected rats included one 32-week-old male, two 110-week-old males, and one 110-week-old female. There were no abnormalities related to general condition or in clinical pathological data suggestive of renal dysfunction. Morphologically, renal proximal tubular inclusions in Wistar Hannover rats, which occurred spontaneously, resembled propiverine and NSRI-induced inclusions in F344 rats and SD rats, respectively. We therefore suspected that spontaneously occurring inclusions in Wistar Hannover rats might also consist of DAO and undertook histochemical, ultrastructural, and immunohistochemical methods to investigate the hypothesis.
Materials and Methods
Animals
Two background data collection studies in Wistar Hannover rats (RccHanTM:WIST) were conducted by Astellas Pharma Inc. in 2009 (26-week study) and BoZo Research Center Inc. from 2008 to 2010 (104-week study). The animals were obtained from Japan Laboratory Animals, Inc. (Hanno, Japan) at 4 weeks of age (104-week study) and 5 weeks of age (26-week study), and the studies began at 6 weeks of age. Animals were kept and observed for 26 weeks or 104 weeks without any treatment and subjected to pathological examination at the age of 32 weeks and 110 weeks. In the 104-week study, the animals were divided into two groups: a pellet diet group and a powdered diet group. The numbers of animals were 20 per sex (the total number of animals was 40) in the 26-week study and 50 per sex per group (the total number of animals was 200) in the 104-week study.
Animal Husbandry
The animals were housed 2 rats per aluminum cage with stainless steel floor mesh in stainless steel racks in the 26-week study and housed individually in suspended stainless steel wire mesh cages in the 104-week study. The animal rooms were maintained with a room temperature of 23°C ± 3°C, relative humidity of 50% ± 20%, and air ventilation 10 to 20 times/hr and 12-hr illumination (7:00–19:00). Animals were allowed free access to CR-LPF pellet or powdered diet (irradiation sterilized CR-LPF, Oriental Yeast Co., Ltd., Tokyo, Japan) and tap water via an automatic water supply system. The experiment was conducted in compliance with the laws and guidelines relating to animal welfare, including the “Act on Welfare and Management of Animals” (Law No. 105, October 1, 1973, Finally Revised on June 22, 2006, Law No. 50) and “The Standards Relating to the Care and Management of Laboratory Animals and Relief of Pain” (Notification No. 88 of the Ministry of the Environment, Japan, April 28, 2006) and “Guidelines for Proper Conduct of Animal Experiments” (Science Council of Japan, June 1, 2006).
In-life Observation and Clinical Pathology
Animals were observed twice daily for clinical signs and once daily for body weight. One-day food consumption per animal was determined by measuring the cumulative food consumption for 7 days on every seventh day. Hematology and blood chemistry parameters were evaluated at the end of the study periods. Animals were anesthetized with isoflurane (26-week study) or ether (104-week study) and euthanized by exsanguination via the abdominal aorta, after blood sampling. Blood samples were collected into blood collection tubes containing EDTA-2K and analyzed for the following hematology parameters: red blood cell count, hemoglobin, hematocrit, mean corpuscular volume, mean corpuscular hemoglobin, mean corpuscular hemoglobin concentration, reticulocyte count, platelet count, white blood cell (WBC) count, differential WBC count, and microscopic examination. Serum samples were analyzed for the following blood chemistry parameters: alkaline phosphatase, total bilirubin, blood urea nitrogen, creatinine, sodium, potassium, chloride, and albumin. Plasma samples were also analyzed for the following blood chemistry parameters: aspartate aminotransferase, alanine aminotransferase, lactate dehydrogenase, and creatine phosphokinase.
Anatomic Pathology
External appearance and all the organs/tissues in the cephalic, thoracic, and abdominal cavities were carefully examined and the results recorded. Then, kidneys were weighed.
Histopathology
After complete necropsy, organs and tissues were fixed in 10% neutral-buffered formalin, embedded in paraffin, sectioned at 4 μm, and stained with H&E. Additional sections from the kidneys were stained with Azan-Mallory to detect proteins (Yamate et al. 1998), periodic acid Schiff (PAS) reaction, Feulgen reaction, and methylgreen pyronin to examine whether inclusions contain polysaccharide, DNA, and RNA, respectively.
Ultrastructural Pathology
For electron microscopic examination, kidneys that were originally fixed with 10% neutral formalin were refixed with 0.5% glutaraldehyde and 1.5% paraformaldehyde, postfixed with 1% osmium tetroxide, and embedded in epoxy resin (Oken Shoji, Tokyo, Japan). Ultrathin sections were stained with uranyl acetate and lead citrate and examined under a JEM-100 CXII transmission electron microscope (Nippon Denshi, Tokyo, Japan).
Immunohistochemistry for DAO
To analyze the protein expression of DAO, 4 μm sections were cut from formalin-fixed, paraffin-embedded blocks and stained immunohistochemically using VECTASTAIN ABC Sheep IgG kit (Vector Laboratories, Burlingame, CA) according to the manufacturer's procedure with 3,3′-diaminobenzidine (Wako Pure Chemical Industries Ltd., Osaka, Japan) as the chromogen and counterstaining with hematoxylin. Slides were deparaffinized and then rehydrated. Enzyme antigen retrieval was completed with protease for 10 min, then, to block endogenous biotin, sections were incubated for 10 min each in avidin and biotin solution (Dako, Tokyo, Japan). The primary antibody used was anti-DAO (pig kidney) antibody (Rockland Inc., Limerick, PA) diluted to 100 μg/ml with phosphate-buffered saline (PBS) and incubated at room temperature for 120 min. As negative control reactions, sections were incubated with PBS or normal sheep immunoglobulin G (IgG; 100 μg/ml) instead of the primary antibody and processed as discussed previously. Additionally, a kidney section from a 110-week-old Wistar Hannover rat that did not have inclusions was stained according to the method described previously as a normal control kidney.
Results
Histopathology and Ultrastructural Pathology
The intranuclear and cytoplasmic inclusions were observed in 1 of the 20 male rats in the 26-week study (32 weeks of age) and 2 of the 100 male and 1 of the 100 female rats in the 104-week study (110 weeks of age) sacrificed at the end of the scheduled study period. During the test period, these animals showed no abnormality in in-life observations or clinical test values suggestive of renal dysfunction. In addition, there were no distinct macroscopic findings in the kidneys and no significant change in kidney weight. Histopathological examination revealed that there was no remarkable change other than inclusions in the kidney in a 32-week-old male rat, and chronic progressive nephropathy, urothelial hyperplasia, or renal pelvic mineralization were observed in the 110-week-old male and female rats. As for the organs/tissues other than the kidney, minimal histopathological changes were observed in the heart, spleen, lung, and liver in the 32-week-old male rat, and tumors, proliferative changes (e.g., follicular cell and C-cell adenoma in thyroid, adenoma of pars distalis in pituitary, lipoma in subcutis, and fibroadenoma or adenocarcinoma in mammary gland), and age-related changes were observed in the 110-week-old male and female rats. In inclusion-bearing rats, there was no commonly observed macroscopic or histopathological finding.
Bilateral intranuclear and cytoplasmic inclusions were observed in more than 60% of proximal convoluted and proximal straight tubular epithelium as homogenous eosinophilic round bodies, and their sizes were approximately 1 to 8 μm in diameter. There was one inclusion per nucleus, whereas there were one or several inclusions per cytoplasm. Intranuclear inclusions existed in nearly the center of the nucleus surrounded by a clear halo with marginated chromatin. These inclusions were not observed in distal tubules, Henle’s loops, or collecting ducts. There were no associated degenerative changes in the kidneys, and similar inclusions were not observed in the other organs (Figure 1).
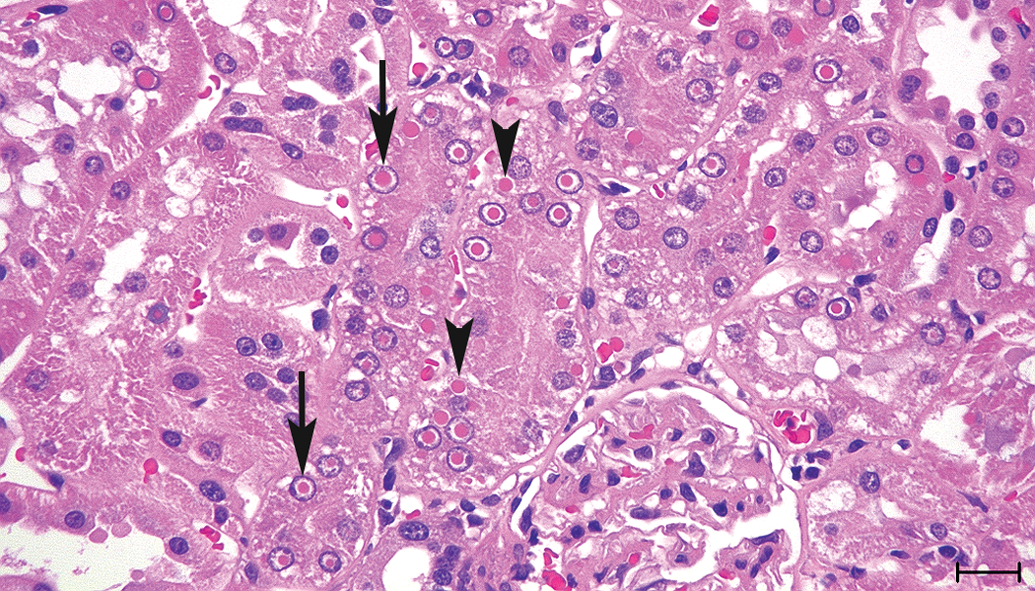
Kidney sections from the 110-week-old male Wistar Hannover rat stained with H&E. Intranuclear (arrow) and cytoplasmic (arrowhead) inclusions were observed in the proximal tubular epithelium as homogenous eosinophilic round bodies, and intranuclear inclusions were surrounded by a clear halo consistent with chromatin margination. Bar = 20 μm.
Ultrastructurally, both intranuclear and cytoplasmic inclusions consisted of finely granular and amorphous material of middle-electron density without a limiting membrane. Intranuclear inclusions displaced nucleoli and surrounding chromatin to the periphery. There were no obvious changes to any of the cellular organelles (Figure 2).
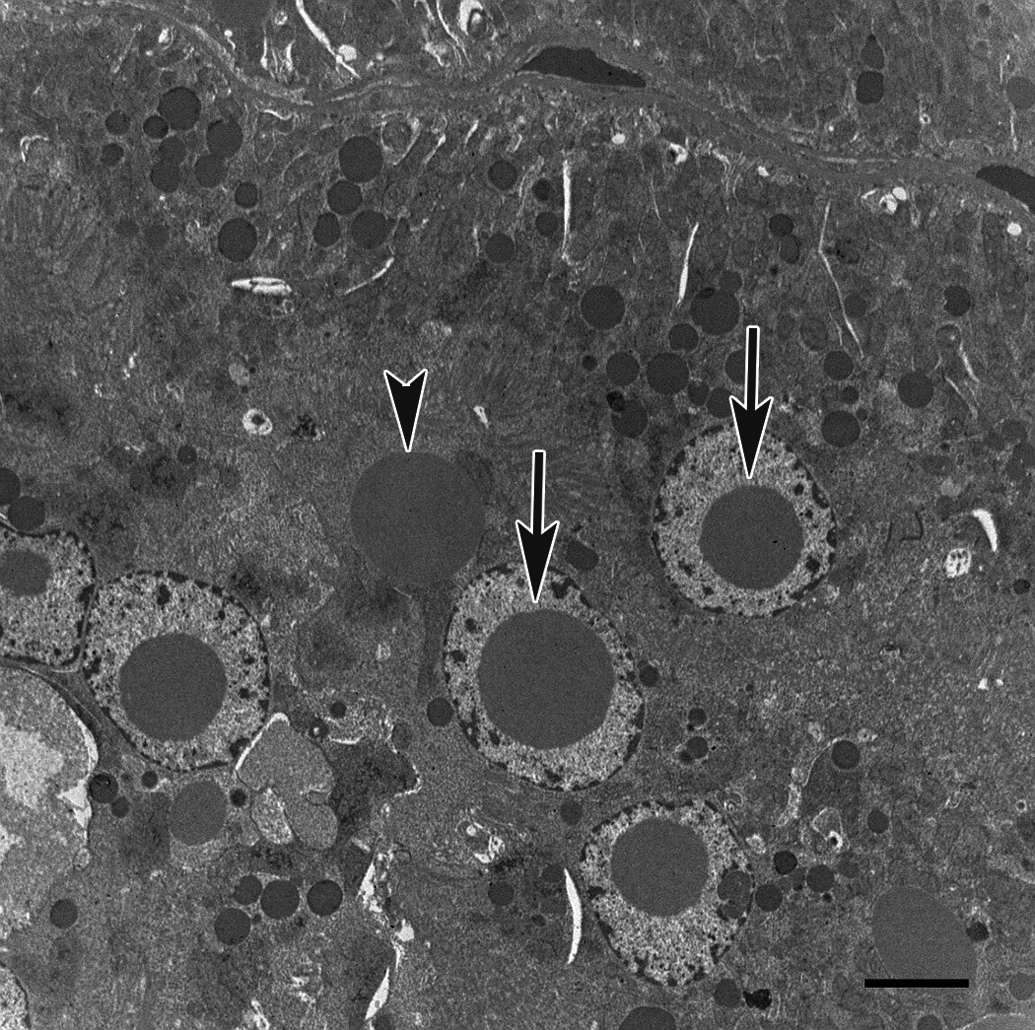
Electron micrograph of renal proximal tubular epithelium from the 32-week-old male Wistar Hannover rat. There were no obvious changes in any of the cellular organelles. Intranuclear (arrow) and cytoplasmic (arrowhead) inclusions were nonmembrane bound, finely granular, and composed of amorphous material of middle-electron density. Bar = 4 μm.
Special staining revealed that inclusions were stained red-orange to dark red by Azan and were negative for PAS, Feulgen reaction, and methylgreen pyronin (Figure 3). The morphology, distribution, and chemical features of the inclusions were similar in all affected rats.
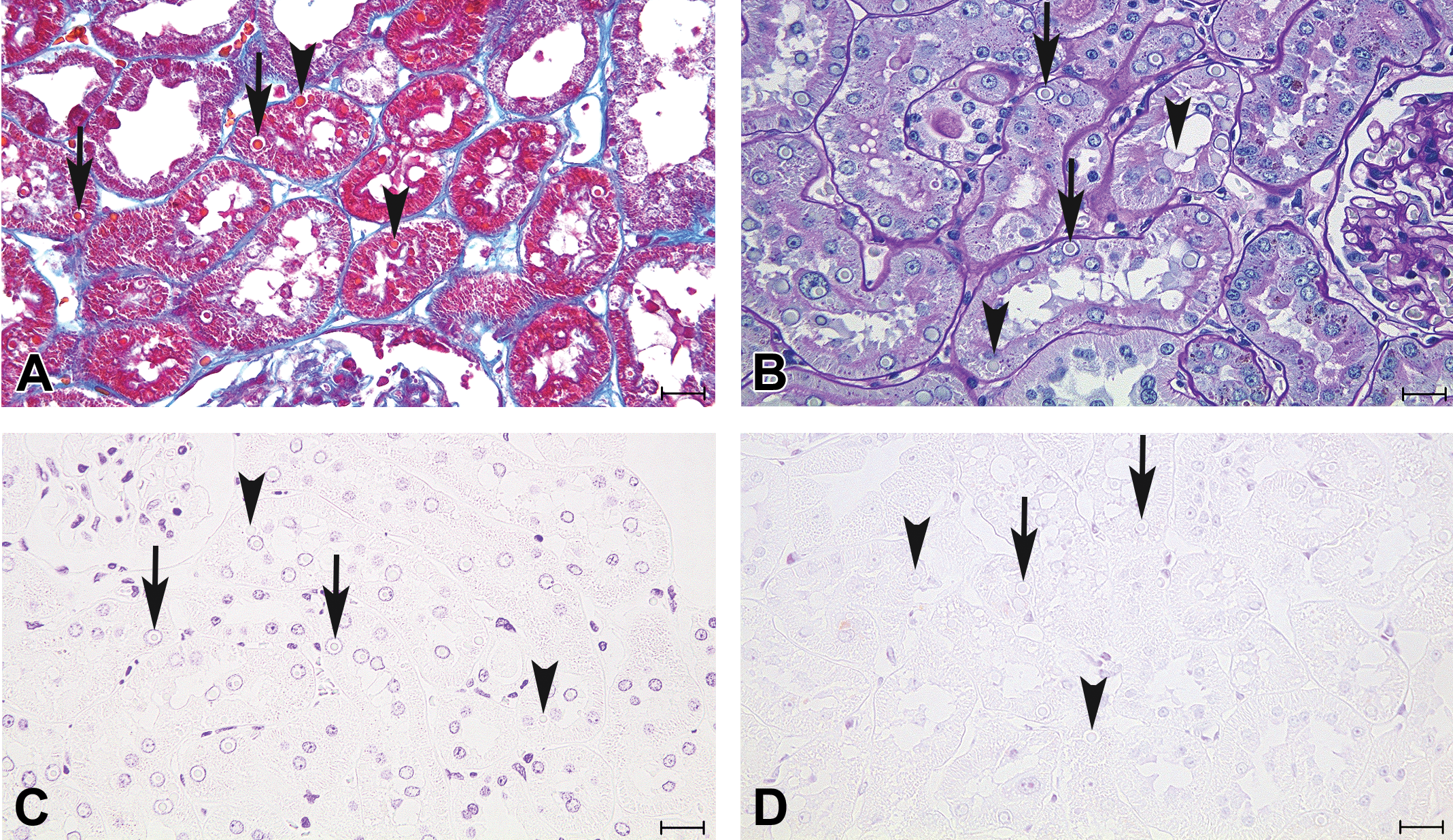
Kidney sections from the 110-week-old male Wistar Hannover rat stained with Azan-Mallory (A), PAS (B), Feulgen reaction (C), and methylgreen pyronin (D). Intranuclear (arrow) and cytoplasmic (arrowhead) inclusions were red-orange to dark red with Azan-Mallory staining and negative for PAS, Feulgen reaction, and methylgreen pyronin stains. PAS = periodic acid Schiff. Bars = 20 μm.
Immunohistochemistry for DAO
Almost all intranuclear and cytoplasmic inclusions were strongly positive for DAO protein by immunohistochemistry (Figure 4). There was no positive reaction for DAO in regions other than proximal tubules in these inclusion-bearing rats.
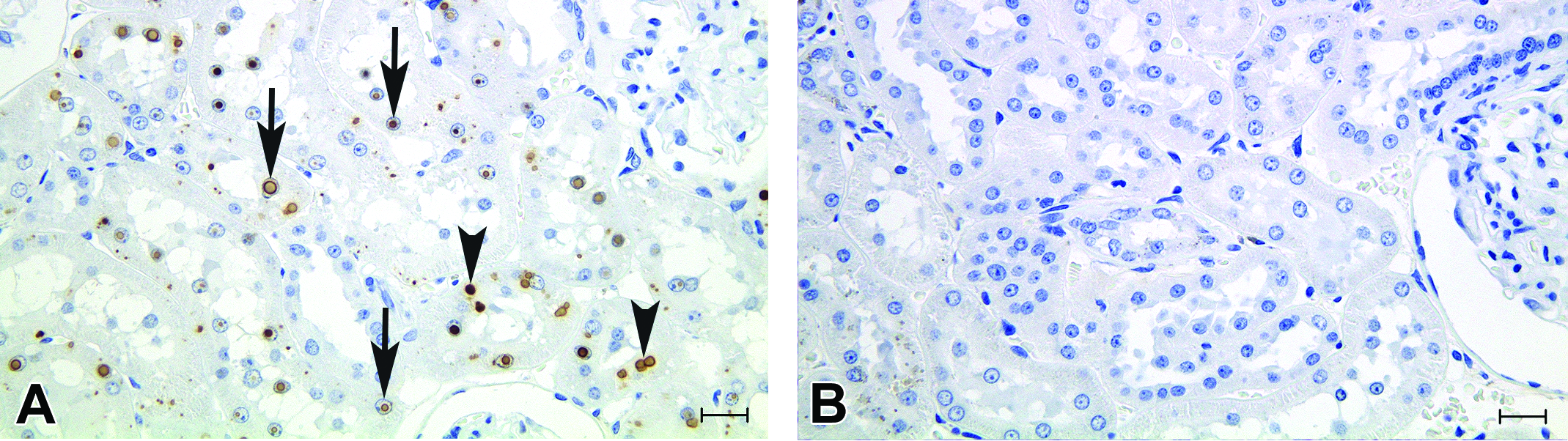
Immunohistochemistry for
Discussion
Intranuclear and cytoplasmic inclusions in the kidney of Wistar Hannover rats were observed by H&E staining to be amorphous, variably sized, round eosinophilic bodies. Intranuclear inclusions were not pseudoinclusions but “true” inclusions because of their ultrastructural feature of nonmembrane-bound structure. Special stains of renal specimens suggested that the inclusions did not contain polysaccharide, DNA, or RNA as demonstrated by PAS, Feulgen reaction, and methylgreen pyronin, respectively, and were instead likely composed of protein by Azan-Mallory stain. All of the animals that had these inclusions were not associated with changes in kidney weights or with any significant adverse renal function effects based on the limited clinical chemistry parameters analyzed. There was no histopathological change that seemed to be related to inclusions in the kidneys, and no similar inclusions were observed in the other organs.
Young adult male rats normally have hyaline droplets in the cytoplasm in the P2 region of the proximal tubular epithelium but not immature males or females of any age. Hyaline droplet accumulation associated with α-2u-globulin nephropathy in male rats is a relatively frequent response to certain hydrocarbons when tested in subchronic assays for toxicity (Hard 2008; Uwagawa et al. 1992). Inclusions in this study differed from hyaline droplets or α-2u-globulin because they were observed in both the nucleus and cytoplasm of proximal convoluted and straight tubular epithelium in male and female rats. In addition, hyaline droplets and α-2u-globulin should appear as electron-dense phagolysosomes (Uwagawa et al. 1992), whereas the inclusions did not include a limiting membrane nor involve secondary lysosomes by ultrastructural evaluation.
In animals and humans, intranuclear or cytoplasmic inclusions in the renal tubular epithelium are observed in cases of metallic poisoning and virus infection. Lead is a nephrotoxic metal, which causes renal tubular damage characterized by development of intranuclear inclusions in the tubular epithelium (Fowler 1992; Misson, Slimani, and Aoues 2010; Fowler, Kahng, and Smith 1994). Intranuclear inclusions in the tubular epithelium are caused by canine herpesvirus and BK-virus in puppies and humans, respectively (Schulze and Baumgärtner 1998; Nickeleit et al. 2000), and porcine circovirus 2 causes multiple botryoid cytoplasmic inclusions in infected pigs (Huang et al. 2008). Absence of any degenerative changes in the tubular epithelium, glomerulus, or interstitial inflammation suggested that these inclusions were not an effect of lead poisoning. The possibility of virus infection was ruled out because these inclusions lacked DNA and RNA and characteristic viral ultrastructural features.
Immunohistochemical staining suggested that these inclusions consisted of DAO. These inclusions were also suggested to be spontaneously occurring lesions because they were observed in animals without any treatment. The incidence rates of the inclusions in these two studies were less than 5%. The unique incidence of the inclusions, that they were observed only in Wistar Hannover rats, both male and female rats, both relatively young and aged rats, and only in the proximal tubular epithelium, whereas the liver and brain also have high DAO content, can be the key to their pathophysiologic mechanism.
Formation of intranuclear and cytoplasmic inclusions in the renal proximal tubules due to accumulation of DAO was induced by the administration of propiverine in F344 rats and NSRI in SD rats during test periods of 4 to 104 weeks (Dietrich et al. 2008; Radi et al. 2013). Although they had not been proved to be accumulation of DAO, it was reported that similar intranuclear and cytoplasmic inclusions were observed in male and female Jcl:Wistar rats in toxicity studies of propiverine (Nakano et al. 1989; Yamashita et al. 1990). The inclusions observed in these two studies were reported to be increased in severity and appeared at lower doses in male rats than in female rats. Additionally, these inclusions became smaller and paler in H&E-stained specimens, and ultrastructurally the boundary between inclusion and karyoplasm became indistinct at the end of the recovery period. Although the mechanism of accumulation of DAO and formation of inclusions in the proximal tubules remained unclear, peroxisome proliferation and enzyme induction mediated via PPARα were thought to be related to the pathogenesis. This hypothesis is supported by the fact that DAO expression is under the control of PPARα (Peters et al. 1996), treatment of rats with clofibrate, a PPARα agonist, led to a marked increase in DAO expression in liver and kidney (Stefanini et al. 1999), and PPARα is found predominately in renal cortex proximal convoluted tubules (Yang et al. 1999). In addition, propiverine and NSRI-induced DAO accumulation and formation of inclusions occurred especially in rats but not in mice or beagle dogs, despite the fact that mice DAO is highly homologous (94%) to that of rats (Dietrich et al. 2008). Thus, the mechanism of accumulation of DAO appears to be related to a species-specific event. In the presence of
Spontaneously occurring formation of intranuclear and cytoplasmic inclusions due to accumulation of DAO is very similar in morphological features to propiverine and NSRI-induced inclusions, although the spontaneous incidence is much lower. There are no previous reports of similar spontaneous inclusions in other preclinical toxicity species (mice, dogs, and monkeys) or in humans. Although propiverine and NSRI-induced inclusions have been observed in several rat strains, these spontaneous inclusions have only been noted in Wistar Hannover rats.
As an unusual background lesion in Wistar Hannover rats, there were eosinophilic intranuclear and cytoplasmic inclusions in the renal proximal tubular epithelium due to DAO accumulation, irrespective of age and sex. These inclusions do not appear to have any biologic or toxicologic significance based on the lack of association with degeneration, necrosis, or inflammation in these kidneys, and it may be a species- and strain-specific phenomenon. The pathogenesis and mechanism of formation of these intranuclear and cytoplasmic inclusions remain undetermined.
Footnotes
Acknowledgments
The authors gratefully appreciate Professor Masahiro Morimoto (Yamaguchi University) and Dr. John Curtis Seely (Experimental Pathology Laboratories, Inc.) for their kind advice and the members of the Veterinary Pathology Laboratory (Yamaguchi University) for technical assistance.
Author Contribution
Authors contributed to conception and design (N.S., S.N., and T.H.); data acquisition, analysis, and interpretation (R.A., Y.Y., and K.T.); drafting the article (N.S.); and critically revising the article (S.N., T.H., R.A., Y.Y., and K.T.). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
