Abstract
A nodule was observed in the adrenal medulla of a twenty-week-old male Wistar Hannover rat. The nodule was predominantly (over 80%) composed of neural components, with ganglion cells scattered in sparse supporting tissue containing nerve fibers and Schwann cells. In the peripheral area of the tumor, atypical chromaffin cells were also observed. Accumulation of eosinophilic serous fluid was also noted in the stromal tissue. There were neither mitotic figures in the ganglion cells nor necrotic foci. In immunohistochemistry, the ganglion cells were positive for neuronal nuclei (NeuN), and negative for proliferating cell nuclear antigen, S-100, and chromogranin A. There were some NeuN-positive small cells in the peripheral area of the tumor. These findings indicate that this tumor was a ganglioneuroma. This seems to be an extremely rare case, as the spontaneous occurrence of ganglioneuroma in rats is very low, even in two-year carcinogenicity studies.
Introduction
Ganglioneuroma is composed of ganglion cells, nerve fibers, and neural supporting cells such as stellate cells and Schwann cells (Melvin and Deborah 1990). These tumors rarely occur (0.05–0.5%) in the adrenal medulla in rats, whereas pheochromocytoma is common (Haseman et al. 1998; Pace et al. 2002; Reznik et al. 1980). As far as we know, the earliest appearance of a ganglioneuroma in the adrenal medulla in rats was observed in a fifty-eight-month-old Fischer rat (Reznik et al. 1980). In this report, we describe a spontaneous ganglioneuroma in the adrenal medulla that occurred in a twenty-week-old Wistar Hannover rat.
Case Report
The rat was a twenty-week-old male Wistar Hannover rat (RccHan: WIST; Japan Laboratory Animals, Inc., Tokyo, Japan). All procedures were performed under the Rules for Animal Experiments approved by the institutional Committee of Animal Experiments. The animal had been dosed daily with 0.5% methyl cellulose by gavage administration for thirteen weeks and was individually housed in a taper-type bracket cage in a barrier-sustained room at a controlled temperature of 23°C ± 2°C, relative humidity of 55% ± 10%, illumination of twelve hours per day at about 200 luces, and ventilation of ten to fifteen cycles per hour. The animal had free access to a radiosterilized pellet diet (CRF-1: Oriental Yeast Co., Ltd., Tokyo, Japan) and tap water supplied through the nozzle of an automatic water-supplying apparatus. The animal was euthanized by exsanguination under anesthesia.
At necropsy, the adrenals and other routinely collected systemic organs were fixed in neutral-buffered 10% formalin solution and embedded in paraffin by routine procedures. Each paraffin-embedded section was stained with hematoxylin and eosin. For the left adrenal, Bodian staining and immunohistochemistry were additionally performed. Immunohistochemistry was performed using a Dako Envision system (Dako Japan, Tokyo, Japan). The antibodies were purchased from Dako Japan with the exception of those for neuronal nuclei (NeuN), which were purchased from Millipore (Billerica, MA, USA). The sections were incubated with antibodies against proliferating cell nuclear antigen (PCNA; dilution 1:400), S-100 (dilution 1:1,600), chromogranin A (CA; dilution 1:200), or NeuN (dilution 1:100). The antibodies for S-100, CA, and NeuN were used to detect Schwann cells, chromaffin cells, and neurons, respectively. Antigen retrieval for the immunohistochemical staining was performed using an autoclave (PCNA and NeuN), proteinase K digestion (S-100), or a microwave (CA). The reaction products were visualized with 3,3′-diaminobenzidine tetrahydrochloride.
No abnormal changes in any organs, including the adrenals, were noted at necropsy. The absolute organ weight of the left adrenal was slightly increased compared to that of a control animal (approximately 20%). During the histopathological examination, a nodule slightly compressing the zona reticularis cells was observed in the left adrenal medulla (Figure 1). The nodule was predominantly (over 80%) composed of ganglion cells and sparse supporting tissue containing nerve fibers and Schwann cells. The ganglion cell had large cytoplasm, which was stained amphophilic centrally and basophilic peripherally.
Figure 1. Histopathological appearance of the tumor. Proliferation of ganglion cells was observed in the center. Atypical chromaffin cells were noted in the peripheral area of the tumor (a), adjacent to normal adrenal medulla (b). The accumulation of eosinophilic serous fluid was also observed (c). Hematoxylin and eosin stain. Bar = 200 µm.
Figure 2. Histopathological appearance of the peripheral area of the tumor. The detailed morphological features of ganglion cells, stellate cells (arrow), and atypical chromaffin cells are shown. The intermediate cells (black arrowhead) are also shown. The detailed appearance of the intermediate cells (asterisked black arrowhead) is shown in the inset in the right upper corner. A mitotic figure is also noted (white arrowhead). Hematoxylin and eosin stain. Bar = 50 µm.
Figure 3. Histopathological appearance of the area where eosinophilic serous fluid accumulated. The fluid accumulated between the stromal cells slightly (d) and severely (e). Bar = 50 µm.
Figure 4. Immunohistochemistry of neoplastic cells stained for NeuN. The intermediate cells (arrowhead) in the peripheral area of the tumor and the ganglion cells were positive for NeuN. Bar = 50 µm.
Figure 5. Immunohistochemistry of neoplastic cells stained for S-100. The Schwann cells and stellate cells were positive for S-100. Bar = 50 µm.
Figure 6. Immunohistochemistry of neoplastic cells stained for chromogranin A. The atypical chromaffin cells were positive, and the intermediate cells (arrowhead) in the peripheral area of the tumor were negative. Bar = 50 µm.
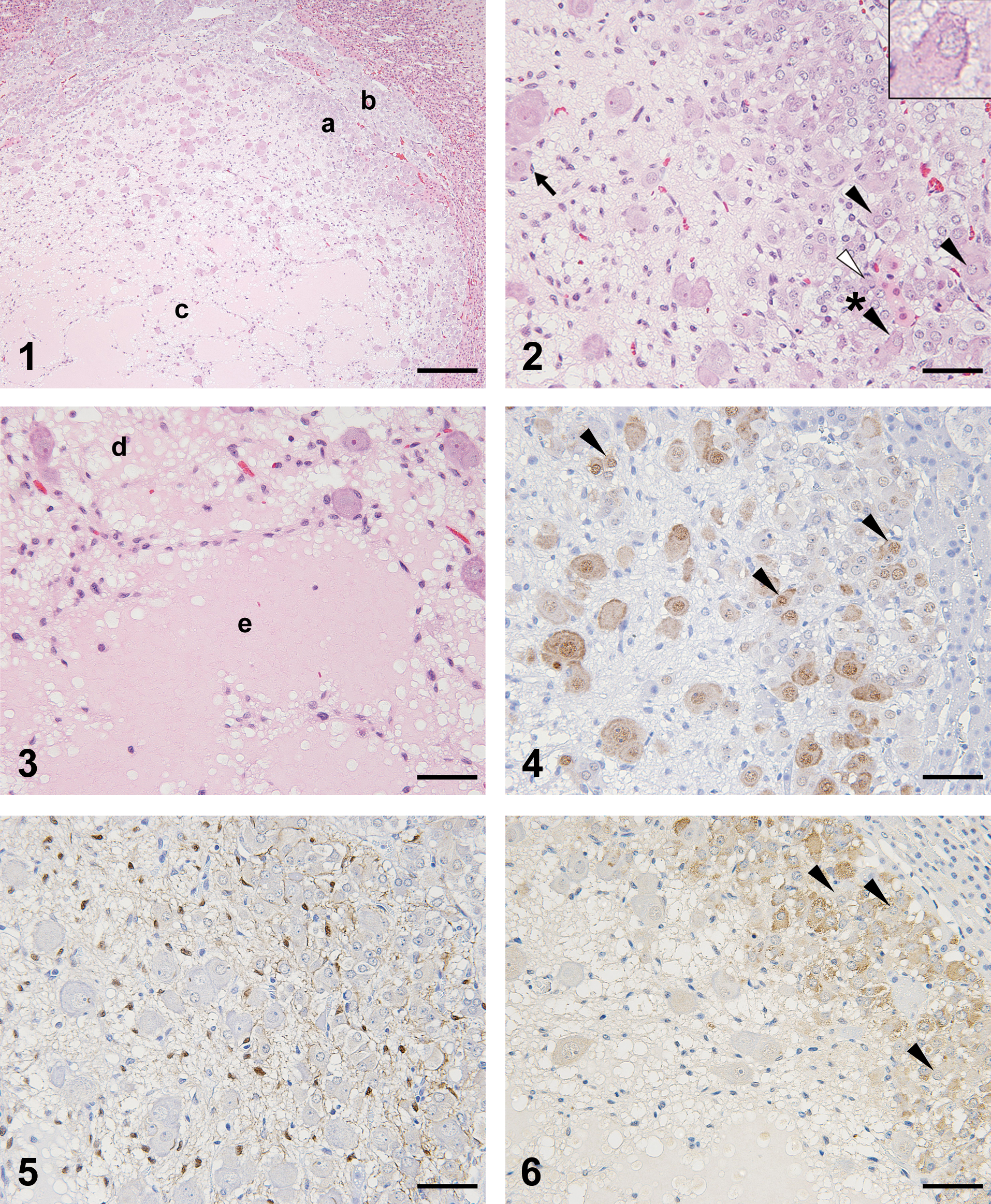
The nucleus of the ganglion cell was large and oval, and it had prominent eosinophilic nucleoli (Figure 2). The ganglion cells existed separately or in clusters composed of a few cells. The ganglion cells were surrounded mainly by spindle-shaped cells, which were considered to be stellate cells. In the peripheral area of the tumor, atypical chromaffin cells surrounding the neural compartment were also observed (Figures 1 and 2). These cells had small basophilic cytoplasm, oval nuclei, and amphophilic to eosinophilic nucleoli. Ganglion cells also were observed in the peripheral area, but they tended to be smaller than those in the central area. In the area where atypical chromaffin cells were observed, there were some “intermediate” cells whose cytoplasm was amphophilic and resembled that of ganglion cells, but the nuclei were small and resembled those of atypical chromaffin cells (Figure 2).
In addition, a varying amount of eosinophilic serous fluid was observed in the stromal tissue. Where the fluid accumulation was severe, the fluid separated the stromal cells (Figure 3). A few mitotic figures were observed in the atypical chromaffin cells (Figure 2), but not in the ganglion cells. This nodule in the left adrenal did not invade any adjacent tissues and did not contain necrotic foci. No similar lesions were found in the right adrenal or any other organs that we examined.
In the Bodian-stained sections, black-stained fibrous materials were observed in the supporting tissue, which confirmed the existence of nerve fibers. In the immunohistochemical examinations, the ganglion cells were positive for NeuN and negative for PCNA, S-100, and CA. The Schwann cells and stellate cells were positive for S-100, partly positive for PCNA, and negative for CA and NeuN, including in the place where the serous fluid accumulated. The atypical chromaffin cells and normal chromaffin cells were positive for CA, partly positive for PCNA, and negative for S-100 and NeuN (Figures 4 –6). In the peripheral area of the nodule, the intermediate cells stained positively for NeuN (Figure 4) and negatively for CA (Figure 6).
Ganglioneuroma, pheochromocytoma, and complex pheochromocytoma must be differentiated from each other. These adrenal tumors can often contain neural structures. In this case, there were atypical chromaffin cells. In a textbook, it is recommended that the neoplasm consisting almost entirely (over 80%) of neural components should be diagnosed as ganglioneuroma (Melvin and Deborah 1990). In this case, in addition to the presence of atypical chromaffin cells, the proportion of neuronal structures was considered to be over 80%. Therefore, we diagnosed this tumor as ganglioneuroma, not (complex) pheochromocytoma.
Neither mitotic figures nor PCNA-positive figures were observed in the ganglion cells. In cells in the peripheral area, mitotic figures and PCNA staining were observed. Therefore, proliferative precursor ganglion cells might be expected to exist in this area. In this case, the intermediate cells, whose morphological features resembled both ganglion cells and chromaffin cells, were positive for NeuN, a marker of differentiated neurons (Mullen et al. 1992), and negative for CA. Therefore, the intermediate cells among the atypical chromaffin cells were considered to be precursors of the mature ganglion cells noted in this tumor. Chromaffin cells and ganglion cells are generated from a common undifferentiated cell, the sympathoadrenal cell (Huber et al. 2009), and dedifferentiation or differentiation into another cell sharing the same cell lineage is known to occur in tumors. In fact, almost all of the ganglioneuromas in the rat adrenal are accompanied by pheochromocytoma cells (Glaister et al. 1997; Pace et al. 2002; Reznik et al. 1980). These previous reports and our results suggest that the ganglion cells in ganglioneuroma originate from dedifferentiated and differentiated atypical chromaffin cells of the adrenal medulla in rats.
In this case, accumulation of eosinophilic serous fluid was observed in the stromal tissue. The details were unclear, but the fluid was considered to originate from serum because no cells that could secrete it were detected histopathologically, and there was a rich supply of capillary vessels. There have been no reports regarding such fluid in ganglioneuroma, as far as we know.
In conclusion, a nodule observed in the adrenal medulla of a twenty-week-old male Wistar Hannover rat was diagnosed as a ganglioneuroma. This was thought to be an extremely rare case, especially because of the young age of this animal, considering the very low spontaneous occurrence of ganglioneuroma in older rats such as those in two-year carcinogenicity studies (Haseman et al. 1998; Pace et al. 2002; Reznik et al. 1980).
Footnotes
Acknowledgments
The authors would like to thank our colleagues for their extended care of the rats in our facility, as well as Ms. Takako Kitajima for her histotechnological assistance with the pathological examinations.
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
