Abstract
Previously, we investigated the higher incidence of hyperplastic lesions and thymomas and histopathological resemblance of cortex–medullary structures between thymomas and normal thymuses in Wistar Hannover (WH) rats. Thymomas had pale-staining cell foci (PA) similar to medulla but without lymphocytes. Here, we focused on the differences in cytokeratin (CK) expression in the thymic epithelia of the cortex and medulla and compared the structures of thymomas and normal thymuses. Thymomas, hyperplastic lesions, and normal thymuses obtained from background studies of WH rats were stained with antibodies against CK14, CK18, and CD20. In normal thymuses, the epithelial cells were positive for CK14 in the medulla and subcapsular area and for CK18 in the cortex, B-cells were positive for CD20 in the medulla. In thymomas, the epithelial cells were positive for CK14 in the medullary differentiation (MD) areas and for CK18 in the cortex-like lymphocyte rich and PA, and B-cells were positive for CD20 in the MD areas.
Introduction
Thymoma, a prevalent tumor in Wistar Hannover (WH) rats, has an incidence of 5% in males and 15% in females as shown in our previous report. 1 In that study, we reported a higher incidence of thymic hyperplasia and thymomas in WH rats compared to Sprague Dawley rats and presented the histopathological features of hyperplasia and thymomas. Moreover, we found that the structure of the thymomas in WH rats consisting of the cortex and medullary differentiation (MD) areas resembled the normal thymic architecture. The cortex-like area was lymphocyte-rich (LyR) and exhibited a large number of lymphocytes among the epithelial cells. The MD area was of variable size and consisted of epithelial cells accompanied by lymphocytes resembling the normal medulla of the thymus. Other than the MD area, many foci of pale-staining cells containing few or no lymphocytes were detected in thymomas. The histological differences between hyperplastic lesions and thymomas were based on the size of the proliferative areas and the number of LyR and MD areas and pale-staining cell foci. 1 In the thymic proliferative lesions, the proliferative cells originated from the thymic epithelial cells in both cortex-like LyR and MD areas; however, it was difficult to recognize the proliferative thymic epithelial cells in the cortex-like LyR area due to the prominently increased number of lymphocytes observed in hematoxylin and eosin–stained specimens. 2 There are various types of thymic epithelia in the normal thymus, such as cortical, medullary, and subcapsular types. 3 In humans, the cortical thymic epithelium is positive for cytokeratin (CK) 8, CK18, and CK19; medullary thymic epithelium is positive for CK5, CK7, CK8, CK14, CK18, and CK19 4,5 ; and subcapsular epithelium is positive for CK14. 4 Frith et al reported that thymic epithelial cells in thymoma of rats are positive for Keratin. 6 Elmore et al and Hojo et al reported that thymic epithelial cells in the LyR thymomas of WH rats are positive for CK18. 7,8 Additionally, Terayama et al reported that medullary thymic epithelial cells in the normal thymus of a Royal College of Surgeons rat are positive for CK14. 9 Brelińska et al reported that CK16 positive for thymic epithelial cells in the outer region of medulla of thymus in Wistar rats. 10 We focused on the differences in CK expression in the thymic epithelium of the cortex and medulla, investigated the type of thymic epithelium present in the cortex-like LyR, MD areas, and pale-staining cell foci observed in thymomas, and compared the structure of thymomas with the normal thymic cortex and medulla. Additionally, we revealed the existence of B-cells in thymomas based on CD20 expression in the MD areas.
Materials and Methods
Animals
WH rats from background-collecting studies (4, 26, and 104 weeks) were used. The RccHanTM: WIST strain was purchased from Japan Laboratory Animals, Inc (Saitama, Japan) and Crl: WI (Han) strain was purchased from Charles River Laboratories Japan, Inc (Yokohama, Japan). The number of animals examined is shown in Table 1.
Number of Animals Examined for Immunohistochemical Analysis.

Rats were individually housed in hanging-type stainless steel wire mesh cages (195 mm[w] × 325 mm[d] × 180 mm[h]; Tokiwa Kagaku Kikai Co, Ltd, Tokyo, Japan) or polycarbonate cages (265 mm[w] × 426 mm[d] × 200 mm[h]; Tokiwa Kagaku Kikai Co, Ltd) at 22 ± 3°C and 55 ± 20% humidity, with 6 to 20 air changes/h and 12-h light/dark cycle. Radiation-sterilized pellet diet (CR-LPF; Oriental Yeast Co, Ltd, Tokyo, Japan) and tap water passed through a 5-µm filter and irradiated with ultraviolet light were supplied to the rats ad libitum. The animals were cared for and euthanized according to the principles outlined in Guidelines for the Care and Use of Laboratory Animals by the Japanese Association of Laboratory Animal Science and the guidelines of our institution.
Pathological Evaluation
Thymuses collected at necropsy were fixed in 10% phosphate buffer formalin for 2 to 3 months, embedded in paraffin, and sectioned at thickness of 4 µm for light microscopic examination. Hematoxylin and eosin staining was performed according to the standard methods. Number of animals examined for immunohistochemical analysis including normal thymuses, hyperplastic lesions, and thymomas are shown in Table1. The paraffin sections were analyzed immunohistochemically using antibodies against CK18 (Ks18.04, Mouse monoclonal, PROGEN Biotechnik, GmbH, Heidelberg, Germany) for cortical epithelium, 7 CK14 (EPR17350, Rabbit monoclonal; Abcam, Cambridge, MA, USA) for medullary epithelium, 9 and CD20 (M-20, Goat polyclonal; Santa Cruz Biotechnology, Inc., Santa Cruz, CA, USA) for B lymphocytes. Secondary antibody used the polymer kit (HISTOFINE Simple Stain Mouse MAX-PO; Nichirei Bioscience, Inc., Tokyo, Japan). The 3,3’-diaminobenzidine (liquid DAB+ substrate chromogen system, DAKO) was used for visualized the target antigen. 11 The procedures and primary antibodies used for immunohistochemical examination are shown in Table 2.
Procedure and Primary Antibodies Used in Immunohistochemical Analysis.
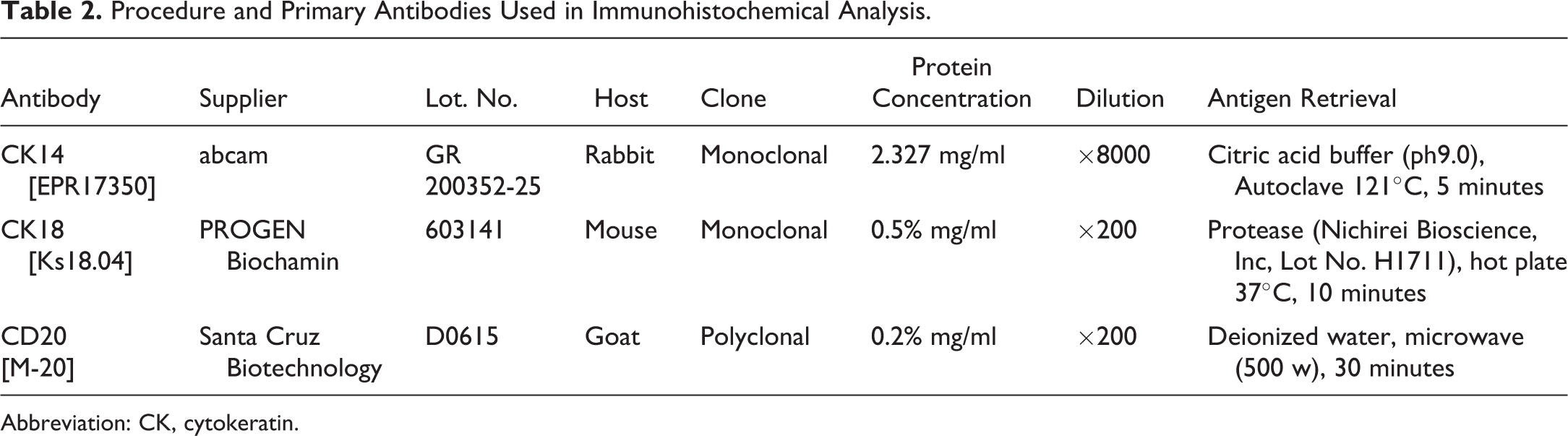
Abbreviation: CK, cytokeratin.
Results
Results of immunohistochemical staining for normal and proliferative lesions of thymus are shown in Table 3. In the normal thymuses of all 10 cases, cortical thymic epithelium showed positive reaction for CK18 (Figure 1A), and Medullary thymic epithelium and subcapsular thymic epithelium showed positive reaction for CK14 (Figure 1B). A few CK18-positive thymic epithelial cells were observed in the medulla. B-cells scattered in the medulla were recognized by CD20-positive reaction (Figure 1D). The distribution of CK14- and CK18-positive thymic epithelium and B-cells was similar among the normal thymuses in the 4-, 26-, or 104-week studies.
Results of Immunohistochemical Staining for Normal and Proliferative Lesions of Thymus in Wistar Hannover Rats.
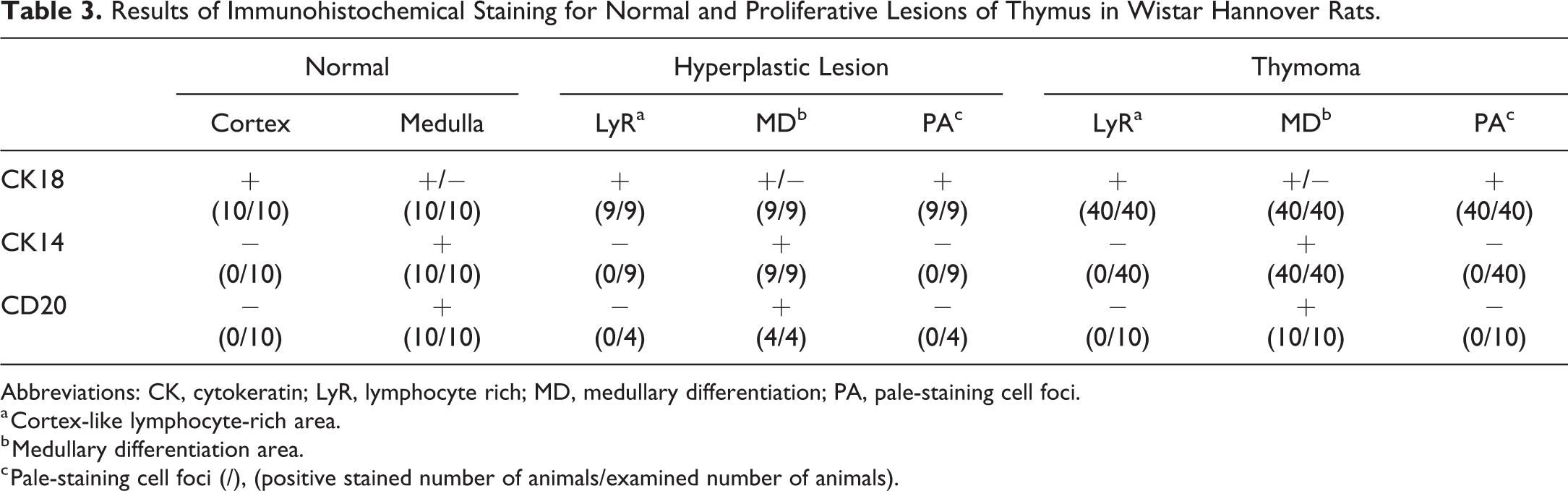
Abbreviations: CK, cytokeratin; LyR, lymphocyte rich; MD, medullary differentiation; PA, pale-staining cell foci.
a Cortex-like lymphocyte-rich area.
b Medullary differentiation area.
c Pale-staining cell foci (/), (positive stained number of animals/examined number of animals).
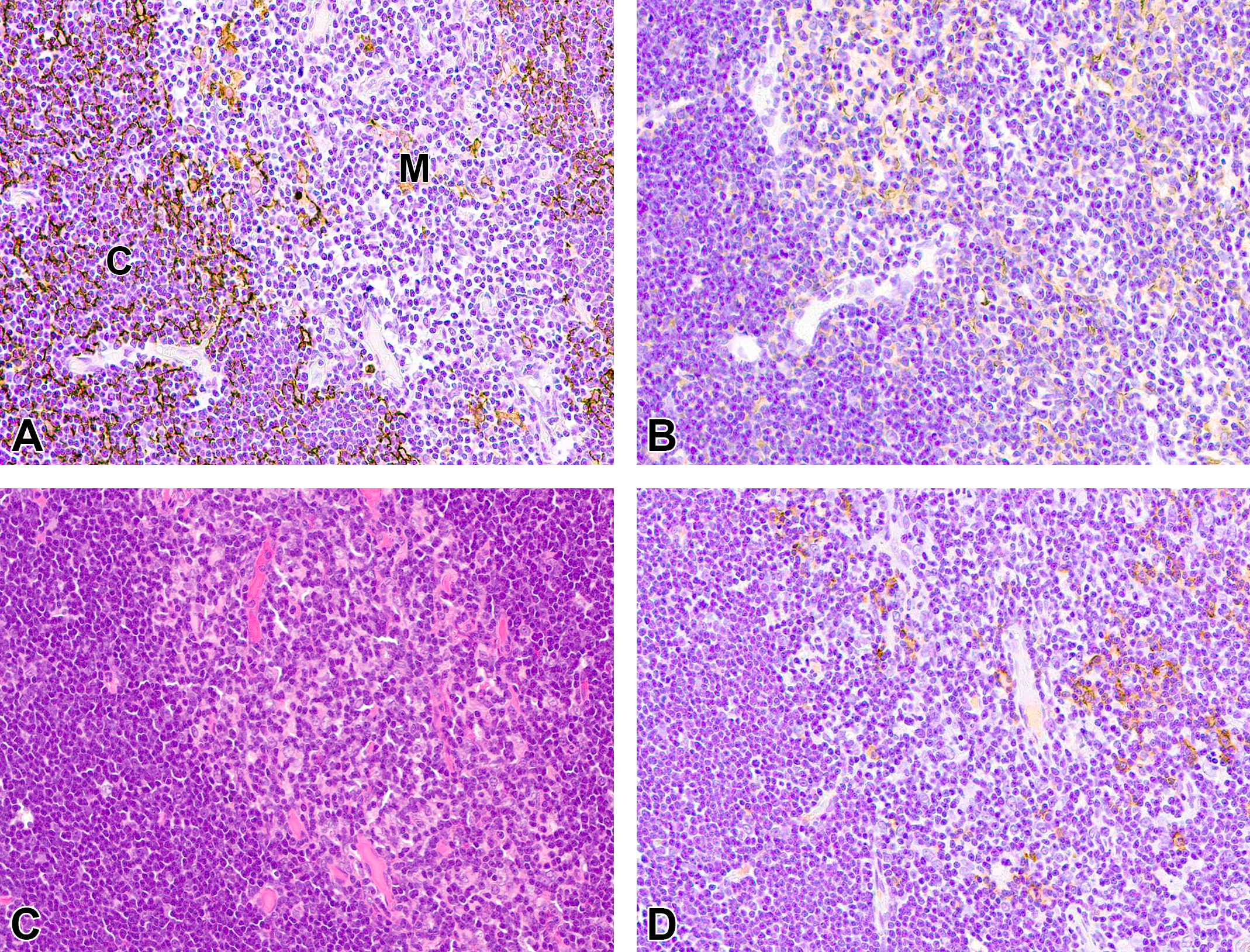
Immunohistochemical and histopathological feature of normal thymus. A, Cortical thymic epithelial cells are positive for CK18. B, Medullary thymic epithelia are positive for CK14. C, Hematoxylin and eosin staining. D, CD20-positive B-cells are observed in the medulla. CK indicates cytokeratin; C, cortex; M, medulla.
In thymoma, the cortex-like LyR area had large numbers of lymphocytes among epithelial cells and the variable sized MD areas consisted of epithelial cells accompanied by lymphocytes. Thymic epithelial cells of the cortex-like LyR area were positive for CK18 (Figure 2A), and those of the MD area were positive for CK14 (Figure 2B). Thymic epithelial cells showing positive reaction for CK18 in the cortex-like LyR area were densely present among abundant lymphocytes and a small number of them were found in the MD area. Thymic epithelial cells positive for CK14 were scattered in the MD area and were not observed in the cortex-like LyR area. Moreover, thymomas had small foci similar to the MD area that contained pale-staining cells and little or no lymphocytes. These pale-staining cells were all positive for CK18 (Figure 2E) and negative for CK14 (Figure 2F). Positive reaction for both CKs was observed in the perinuclear cytoplasm and fine cytoplasmic processes, resembling normal thymic epithelial cells in the cortex and medulla. Similar to the normal thymuses, CD20-positive B-cells were observed in the MD area (Figure 2D), but not in the cortex-like LyR area or pale-staining cell foci.
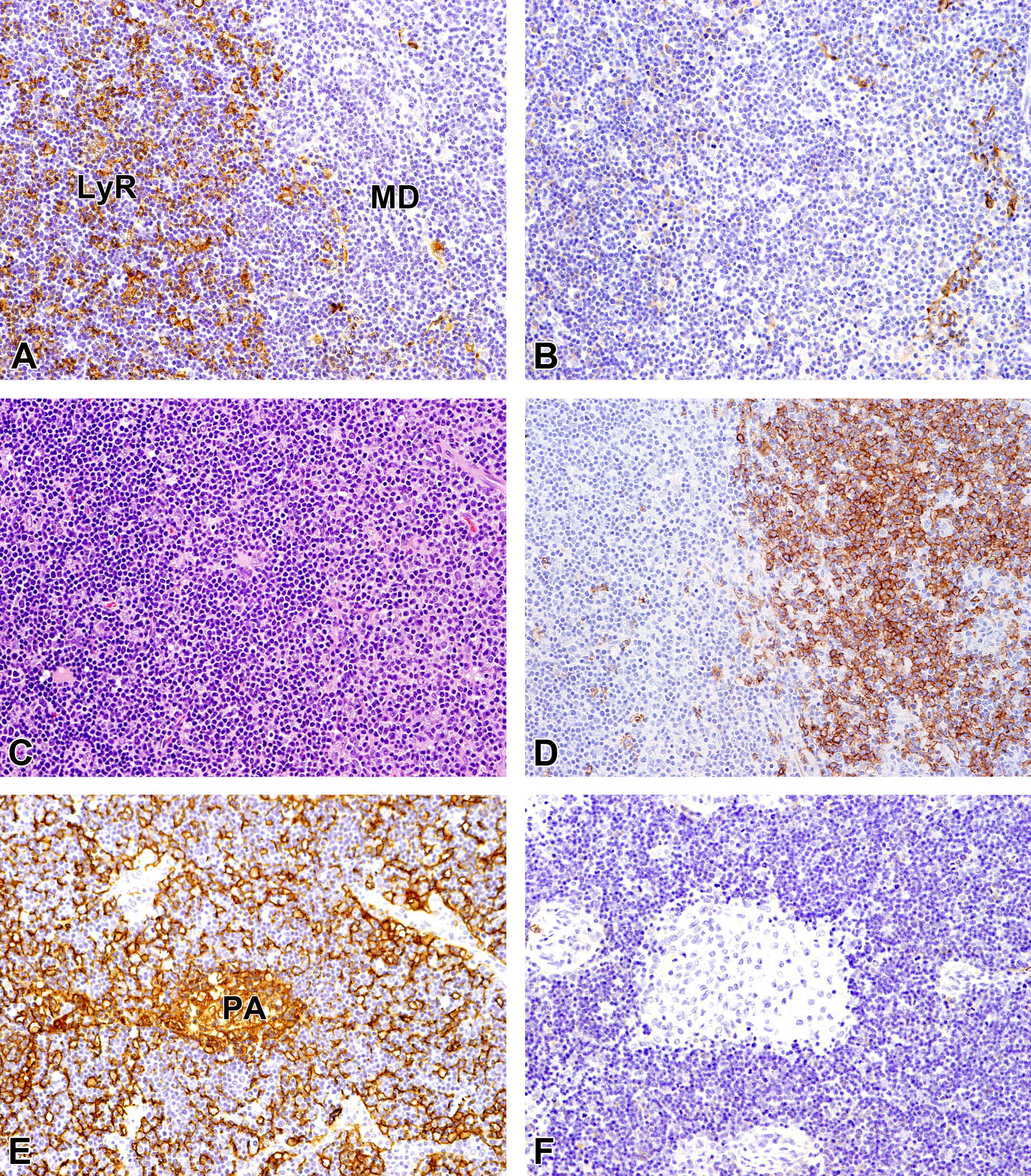
Immunohistochemical and histopathological features of thymoma (LyR/MD areas). A, Thymic epithelial cells of the LyR area are positive for CK18. B, Thymic epithelial cells of the MD area are positive for CK14. C, Hematoxylin and eosin staining. D, CD20-positive B-cells are observed in the MD area. E, Pale-staining cells are positive for CK18. F, Pale-staining cells are negative for CK14. CK indicates cytokeratin; LyR, lymphocyte rich; MD, medullary differentiation; PA, pale-staining cell focus.
In hyperplastic lesions, immunohistochemical features of the thymic epithelial cells in each area of lesions in the thymus were similar to those of thymomas (Figure 3). There was not any difference between males and female in immunohistochemical reaction of normal thymus and proliferative lesions.
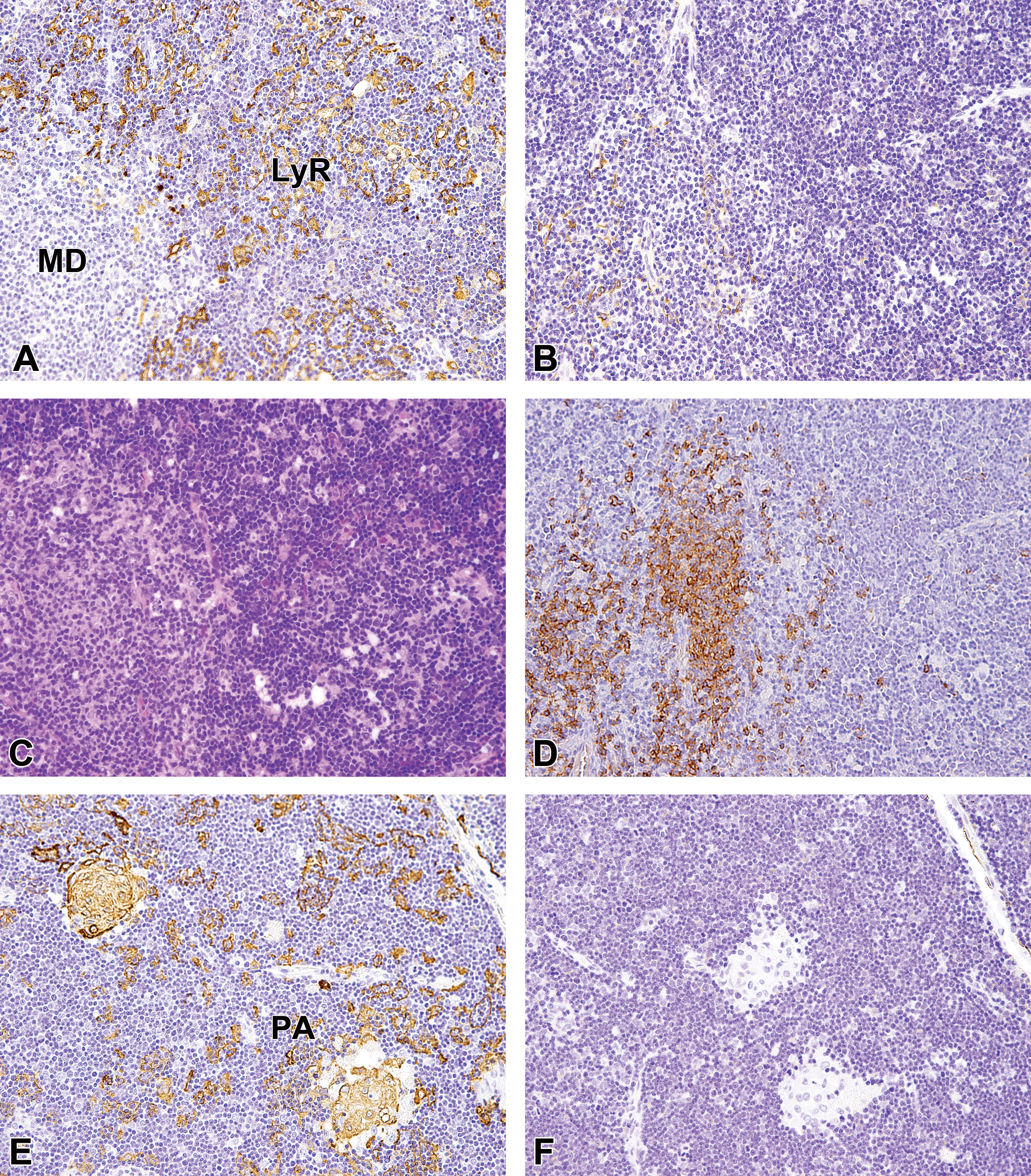
Immunohistochemical and histopathological features of hyperplastic lesion (LyR/MD areas). A, Thymic epithelial cells of the LyR area are positive for CK18. B, thymic epithelial cells of the MD area are positive for CK14. C, Hematoxylin and eosin staining. D, CD20-positive B-cells are observed in the MD area. E, pale-staining cells are positive for CK18. F, pale-staining cells are negative for CK14. CK indicates cytokeratin; LyR, lymphocyte rich; MD, medullary differentiation; PA, pale-staining cell focus.
Discussion
In thymomas and hyperplastic lesions of the thymus in WH rats, CK14 and CK18 were used to study the thymic epithelial type (cortical or medullary). The thymic epithelial cells in the cortex-like LyR area were CK18-positive similar to the cells of the normal cortex, while the thymic epithelial cells in the MD area were CK14-positive similar to the cells of the normal medulla. The type of proliferative thymic epithelial cells in the cortex-like LyR and MD areas was found to be different. This epithelial difference may lead to morphological differences in each area within the thymic lesion.
In World Health Organization classification of human thymomas, thymomas have been classified into 6 types (A, B1, B2, AB, B3, and C) according to the growth pattern of thymic epithelium and accompanying lymphocytes. Among these types, B1 type is a tumor imitating the normal thymus and has a distinct differentiation between cortex and medulla. 12 The common type of WH rat thymomas containing medullary-like structure are similar to the B1 type thymomas in humans. 12 In type B1 thymomas, CK19, and B5T are reacted at the thymic epithelium in the cortical-like area, and CK14, CK19, Claudin-4, CD40, and AIRE are reacted at the thymic epithelium in the medullary-like differentiation area. 4,13 Additionally, normal cortical thymic epithelium in humans showed positive reaction for CK8, CK18, CK19, and B5T, while medullary thymic epithelium showed positive reaction for CK5, CK7, CK8, CK14, CK18, CK19, Claudin-4, CD40, and AIRE. 4,5,13 Some of the expression of CK in the LyR and MD areas of type B1 thymomas of humans correspond to that in the normal cortical and medullary thymic epithelia, respectively. Analysis of CK expression in WH rat thymomas also suggested that the cortex-like LyR and MD areas consisted of thymic epithelial cells having the characterization of cortical and medullary thymic epithelial cells similar to that in human B1 type thymoma.
The presence of foci of pale-staining cell aggregates in the cortex-like LyR area is not clearly described in human thymomas. Kuper et al described the foci of thymic epithelial cell aggregates as of pale epithelial cells in Wistar rats; however, they did not make a clear distinction between foci of pale epithelial cells and the MD area immunohistocheically. 14 Our results indicate that the pale-staining cell foci can be distinguished from the MD area since thymic epithelial cells in the pale-staining cell foci were positive for CK18 as well as those in LyR area.
Thymic medulla in humans, cynomolgus monkeys and dogs sometimes showed the presence of normal lymphoid follicles including B lymphocytes. 15,16 B-cells were present in the medulla of normal thymus in WH rats as same in these species. In the MD area, there were fewer T lymphocytes than the cortex-like LyR area; however, it contained some CD20 positive cells suggesting B lymphocyte similar to the normal medulla. The thymic epithelium is mainly divided into 3 types: subcapsular epithelium, cortical epithelium, and medullary epithelium, and each epithelium contributes to the maturation and proliferation of lymphocytes. 4,5,14 It is unknown whether neoplastic thymic epithelium is responsible for the differentiation of lymphocytes in normal thymus; however, at least from its structure, it is considered that lymphocytes correlating to the type of thymic epithelium exist in each area of thymomas.
A single progenitor differentiates into thymic cortical and medullary epithelial cells. Differentiation includes CK expression and establishment of desmosomal intercellular linkages, after which the thymic epithelial cells form an open network that enables a close contact with thymocytes. 17 It has been reported that both cortical and medullary thymic epithelia develop and become activated during the regeneration of an acutely damaged thymus. 18 It is considered that in the proliferative lesions such as thymomas and hyperplasia, cortical or medullary structures develop due to different CK expression in the proliferating thymic epithelium.
In conclusion, thymomas and hyperplastic lesions of WH rats were found to be related to the distribution of lymphocytes present in both cortex-like LyR and MD areas, suggesting that the proliferation of different types of thymic epithelia leads to the formation of cortical–medullary structure. Additionally, the pale-staining cell foci were focal aggregate of cortex-like epithelial cells. We believe further research into CK expression in thymic epithelial cells of thymomas consisting of a predominant epithelial type will be both informative and fascinating.
Supplemental Material
Supplemental Material, sj-docx-1-tpx-10.1177_0192623320922849 - Immunohistochemical Characteristics of Thymomas and Hyperplastic Lesions in Wistar Hannover Rats
Supplemental Material, sj-docx-1-tpx-10.1177_0192623320922849 for Immunohistochemical Characteristics of Thymomas and Hyperplastic Lesions in Wistar Hannover Rats by Yuki Tomonari, Junko Sato, Naoaki Yamada, Tetsuro Kurotaki, Takuya Doi, Takeshi Kanno and Minoru Tsuchitani in Toxicologic Pathology
Supplemental Material
Supplemental Material, sj-tif-1-tpx-10.1177_0192623320922849 - Immunohistochemical Characteristics of Thymomas and Hyperplastic Lesions in Wistar Hannover Rats
Supplemental Material, sj-tif-1-tpx-10.1177_0192623320922849 for Immunohistochemical Characteristics of Thymomas and Hyperplastic Lesions in Wistar Hannover Rats by Yuki Tomonari, Junko Sato, Naoaki Yamada, Tetsuro Kurotaki, Takuya Doi, Takeshi Kanno and Minoru Tsuchitani in Toxicologic Pathology
Footnotes
Acknowledgments
The authors gratefully acknowledge Ms. Kanae Tamatsukuri and Mr. James Harada for language editing of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
