Abstract
The Göttingen minipig is recognized by the scientific community and by health authorities as the animal model of choice to evaluate dermally applied drugs under development. Young adults of approximately 4 months of age are most generally chosen to participate in dermal pharmacology and toxicology studies, and recently, minipigs have been proved to be also suitable for juvenile studies. A complete anatomical cartography of the skin morphology of juvenile male and female minipigs from postnatal day 1 (PND1) to twelve weeks of age was performed measuring the thickness of skin layers for each anatomical location and time point. Overall, the neonatal skin of minipigs (PND1 and PND8) shows prominent cellularity, similar to that seen in human neonates, and the morphology of the skin of older animals is considered similar to that of young adult minipigs. Epidermal thickness varies only minimally over the period; whereas, the dermal and more markedly, the subcutaneous thicknesses increase over time.
The Göttingen minipig is considered as the animal model of choice to investigate percutaneous absorption, wound healing, dermal tolerance, and generally for dermal pharmacology and toxicology studies (Stricker-Krongrad et al. 2017; Ganderup et al. 2012; Bode et al. 2010). This model is also recognized and accepted by the authorities in regulatory drug development.
The minipig is well suited for skin studies as they allow the use of a large application surface with low hair density, an easy evaluation of skin reactions and feature only few background cutaneous findings. The minipig skin shares many common features with the human skin, in particular the low density of hair follicles, the properties of the dermoepidermal junction, and the epidermal morphology and thickness. When compared to the other nonrodent animal models, the skin thickness of the minipig is the most similar to human’s. Overall, among the animal species, the minipig is the most suitable species for dermal preclinical studies.
In addition to the conventional repeat-dose toxicity studies performed in adult animals of 4 months of age during drug development, juvenile animal toxicity studies are required by the authorities for the development of pediatric drugs. As stated by Tassinari (2013) or De Schaepdrijver (2010, 2013) and in line with the ICH guidance for industry on the “nonclinical safety evaluation of pediatric drug products” (2006), the main objectives of juvenile animal studies are to determine potential toxicities unique to immature animals. Over the last ten years, the number of juvenile animal toxicity studies, including all age groups from neonatal to young animals, increased drastically in the context of pediatric investigative plans. The species selection depends on several concurrent factors such as metabolic and physiological relevance of the model but also practical aspects such as feasibility of the intended route of administration.
Due to the relatively recent use of the minipigs in juvenile studies, only sparse background data are available (Feyen et al. 2016), and no information is available on the skin morphology characteristics in the juvenile minipig. In addition, the use of juvenile minipigs in dermal studies has long been considered as not feasible due to husbandry and behavioral constraints. However, it has been recently demonstrated that juvenile studies by dermal route can be performed with animals from postnatal day (PND1) onward (Penard 2016). Practical aspects such as the dressing materials, jackets, and estimation of the total body surface area were slightly adapted to the constraints of suckling piglets; and overall, it was demonstrated that the brief periods during which the piglets were removed from the mother did not impact the behavior, growth, and the health status of the young animals. In this study, sequential necropsies were performed and skin samples were taken in order to document the skin morphology at various anatomical location and time points from PND1 to 12 weeks of age. This article summarizes the main morphological characteristics of the skin at each time point.
Materials and Methods
Juvenile Animals and In-Life Follow-up
Male and female Göttingen minipigs were received from Ellegaard, Soroe Landevej 302, DK-4261 Dalmose, Denmark, and housed in a conventional and GLP-compliant “minipig unit” with restricted entry and controlled environmental conditions at Charles River, Les Oncins, France. Housing and handling of animals were performed in accordance with the European directive 2010/63/EU for animal experiments and national regulations, and the study design and technical procedures were approved by the relevant ethical committee.
Three multiparous Göttingen female minipigs were mated at the Test Facility (Charles River, Les Oncins, France) to obtain 20 piglets (10 males and 10 females).
Piglets from the first litter were used uniquely to establish the technical procedures and materials without the application of any test item and to document the skin morphology of juvenile minipigs.
Piglets from the two other litters were treated dermally with a placebo gel (standard volume of 2 ml/kg/day) applied on both flanks in areas defined using anatomic landmarks (rectangles between the scapulae and hipbones), in order to assess the feasibility of dermal administration in the juvenile minipig (not part of this article, results described in Penard L., Ellegaard’s Newsletter 47). Piglets from each litter were socialized and kept with the mother.
Sampling
Skin morphology documentation in juvenile minipigs from PND1 to week 12 of age was performed on skin samples taken at necropsy from the first litter (i.e., without application of any test item) at Charles River, Les Oncins, France. All samples were fixed in 10% neutral buffered formaldehyde, thereafter sent and processed at the Sophia Antipolis site of Galderma R&D. Twelve samples from one male and one female were obtained per time point from PND1 to 3 months of age (PND1, PND8, PND15, PND29, and W12) as described in Figure 1. The selected anatomical areas allow an extensive documentation of the skin morphology variability and included two samples taken from the lateral region (flank posterior area and flank anterior area), two samples taken on the dorsal line around the spine (rachis posterior area and rachis anterior area), one sample behind the right ear, one sample on the snout, one sample on the chin, one sample taken at the internal face of the forelimb and one on the internal face of the hind limb, two samples taken at the abdomen (anterior and posterior area), and finally one sample taken in the pelvic region (ischium–pelvis). It is to be noted that the term “flank” was used in this study to describe the entire lateral region (region defined using anatomic landmarks: rectangles between the scapulae and hipbones) although this anatomical correct term for the anterior part of this region should be “thoracic region.” This region as defined here corresponds to the area commonly used for dermal application in the dermal toxicology. The other anatomical regions have been selected to document the histological characteristics of areas that could potentially be used for injections or topical administrations.
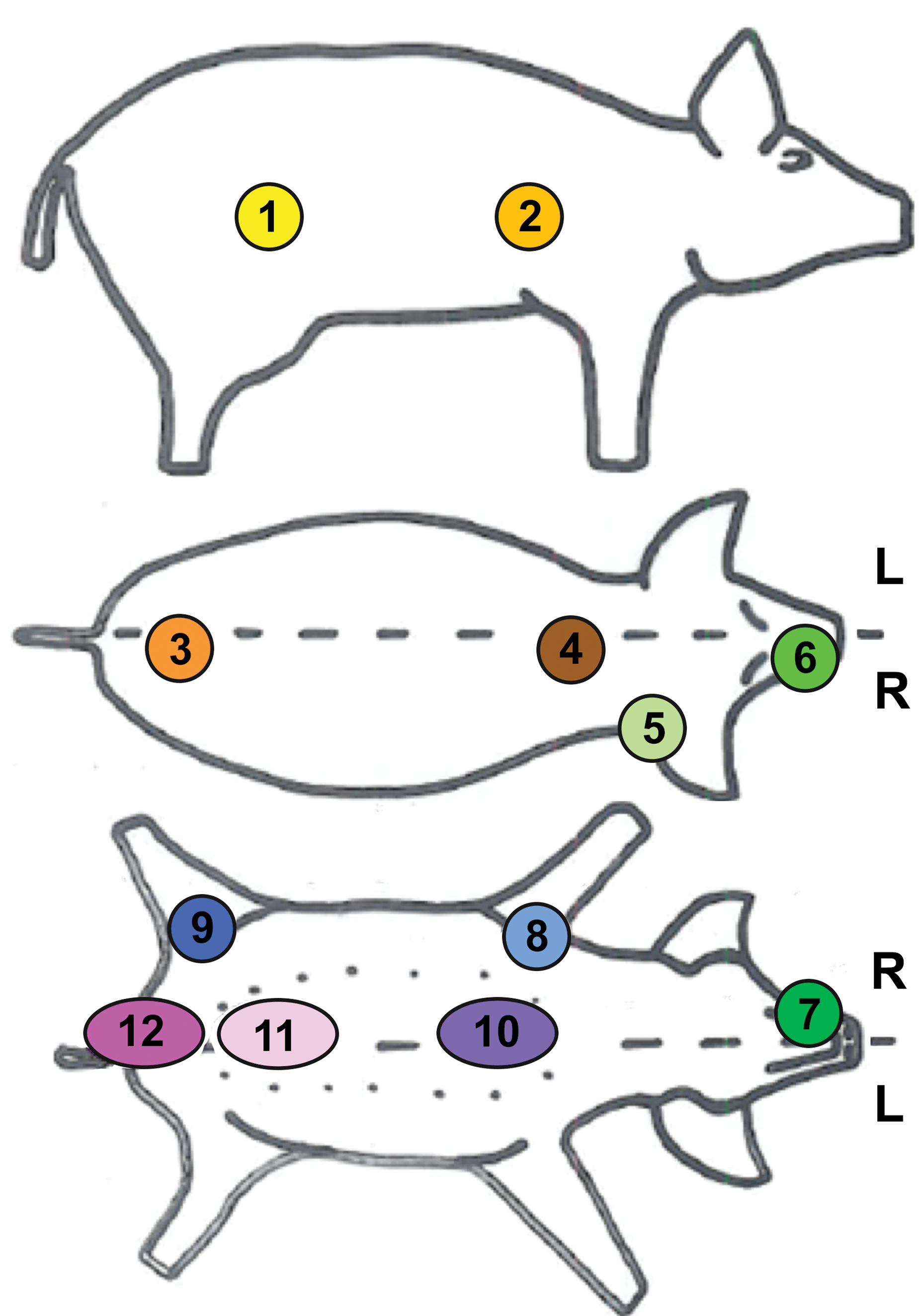
Anatomical location of the skin samples. Twelve skin samples were taken from each animal from the following anatomical regions: 1: flank (posterior area), 2: flank (anterior area), 3: around the spine (rachis, posterior area), 4: around the spine (rachis, anterior area), 5: behind the ears, 6: snout, 7: chin, 8: internal face of the forelimb, 9: internal face of the hind limb, 10: abdomen (anterior area), 11: abdomen (posterior area), and 12: ischium—pelvis area.The term flank was used to describe the entire lateral region, which is commonly used for dermal application (region defined using anatomic landmarks: rectangles between the scapulae and hip bones).
Sample Processing and Skin Thickness Measurements
All samples were fixed in 10% neutral buffered formalin. Samples were paraffin-embedded, and sections of 4-µm thickness were stained with hematoxylin and eosin. The slides were scanned using the Nanozoomer (Hamamatsu, Japan), and virtual slides were imported and stored in the internal image database (IMS from Imagic, Switzerland).
In addition to the descriptive evaluation at each time point, skin thickness measurements were performed manually using the Arivis viewer (provided by Imagic Switzerland) as follows: – For each skin layer ( – For the – Special attention was paid to avoid areas of oblique cutting incidence or other artifacts that may affect skin thickness measurements. – Comments were added to the database when necessary, in particular when the subcutis exceeded the margins of the available section. Data are reported but represent an underestimation of the actual data. – Considering that the most superficial part of the
All the results were exported as an excel file and compiled together with animal information.
Results
Skin Morphology and Histomorphometric Data in Minipigs from PND1 to Week 12 of Age
Morphology of juvenile skin at PND1
Regardless of the anatomical region or sex, samples taken shortly after birth (PND1) showed the typical cutaneous multilayered structure consisting of the epidermis (a keratinized, stratified, and squamous epithelium), the dermis (a dense connective tissue consisting of different types of fibers, cells within a gel matrix), and the hypodermis (loose connective tissue with adipocytes). When compared with the skin of mature animals, the skin samples at PND1 showed a thick epidermis as well as a very high cellular density in dermis and hypodermis layers (see Figure 2 for epidermis and dermis morphology: sample taken at the anterior region of the spine). No immunohistochemistry was performed in this study; however, the morphology of these cells is evocative of active fibroblasts with an abundant cytoplasm and round nucleus.
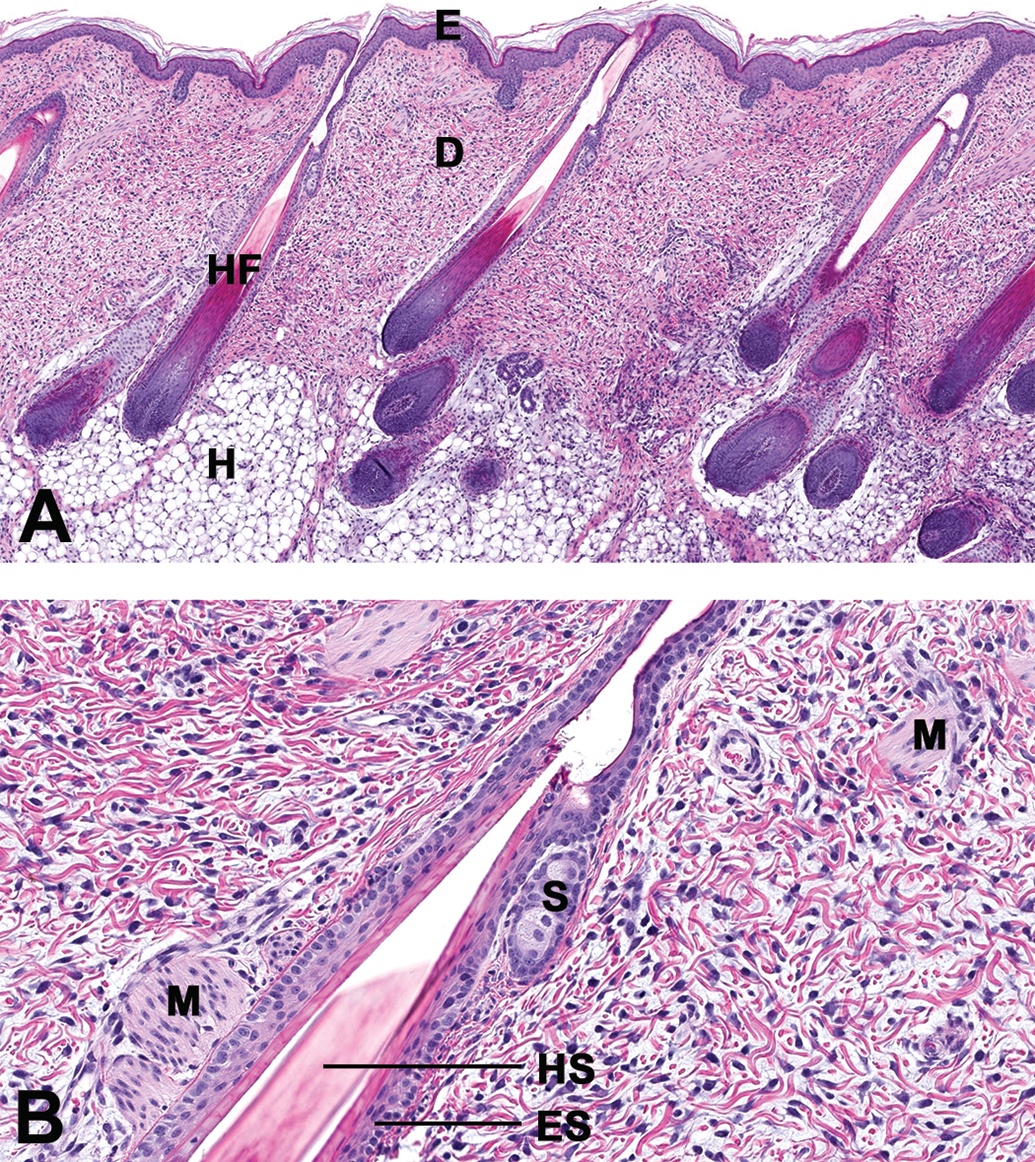
(A and B) Morphology of the epidermis and dermis in a juvenile male at PND1. Samples taken near the spine (rachis anterior) in a male juvenile minipig at PND1. All samples were fixed in 10% neutral buffered formaldehyde. Samples were paraffin-embedded and 4-µm thick sections were stained with hematoxylin and eosin. The slides were scanned using the Nanozoomer (Hammamatsu, Japan) and virtual slides were imported and stored in the internal image database (IMS from Imagic, Switzerland). (A) Low magnification (scan magnification ×5) and (B) high magnification (×20). E = epidermis; D = dermis; H = hypodermis (subcutis); HF = hair follicle in anagen phase; HS = hair shaft; ES = epithelial sheath; M = muscle, S = sebaceous gland; PND = postnatal day.
The subcutis was found to be very thin when compared with the week 12 samples (see Figure 3 for hypodermis at anterior spine), with the adipocytes present in this layer showing typical large optically empty vacuoles. All epidermal appendices were already present and well developed in all regions. Specifically, sebaceous glands were found as very small glands appended to hair follicles with a low number of differentiated sebaceous cells and prominent stem cells. Sweat glands were seen adjacent to hair follicles (apocrine glands) and showed a thick wall with cubic epithelial cells. When compared with young adult skin, sweat glands were less convoluted as they mostly appeared only as 3 to 4 transversal sections.
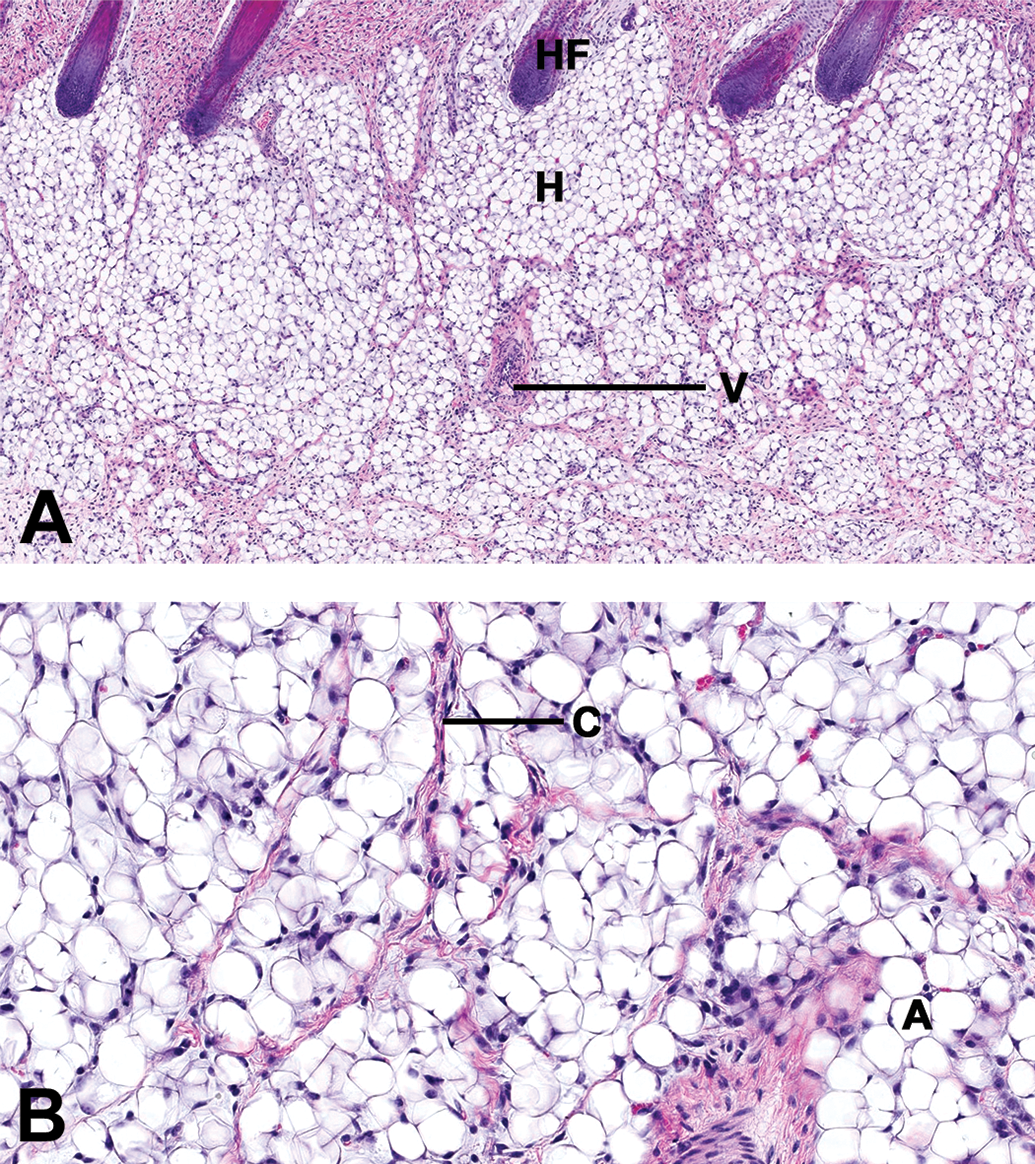
(A and B) Morphology of the hypodermis in a male juvenile minipig at PND1. Samples taken near the spine (rachis anterior) in a male juvenile minipig at PND1: hematoxylin and eosin stain. (A) Low magnification (scan magnification ×5) and (B) high magnification (×20). HF = hair follicle; H = hypodermis (subcutis); A = adipocytes; C = collagen strands; and B = blood vessel. The hypodermis consisted of a dense adipocytes population. The adipocytes showed a unique optically empty vacuole but were smaller than in mature animals. Thin strands of collagen were seen between the cells. PND = postnatal day.
Interestingly, vibrissae were seen at the chin or snout sections in the male and the female at PND1. Although the structure was easily recognized on the skin section as a very large hair follicle (about 2-fold larger than the other hair follicles in the same section; diameter of at least 150 µm), the peripheral blood sinus did not have its mature pattern and showed a highly cellular population surrounding the hair follicle (see Figure 4).
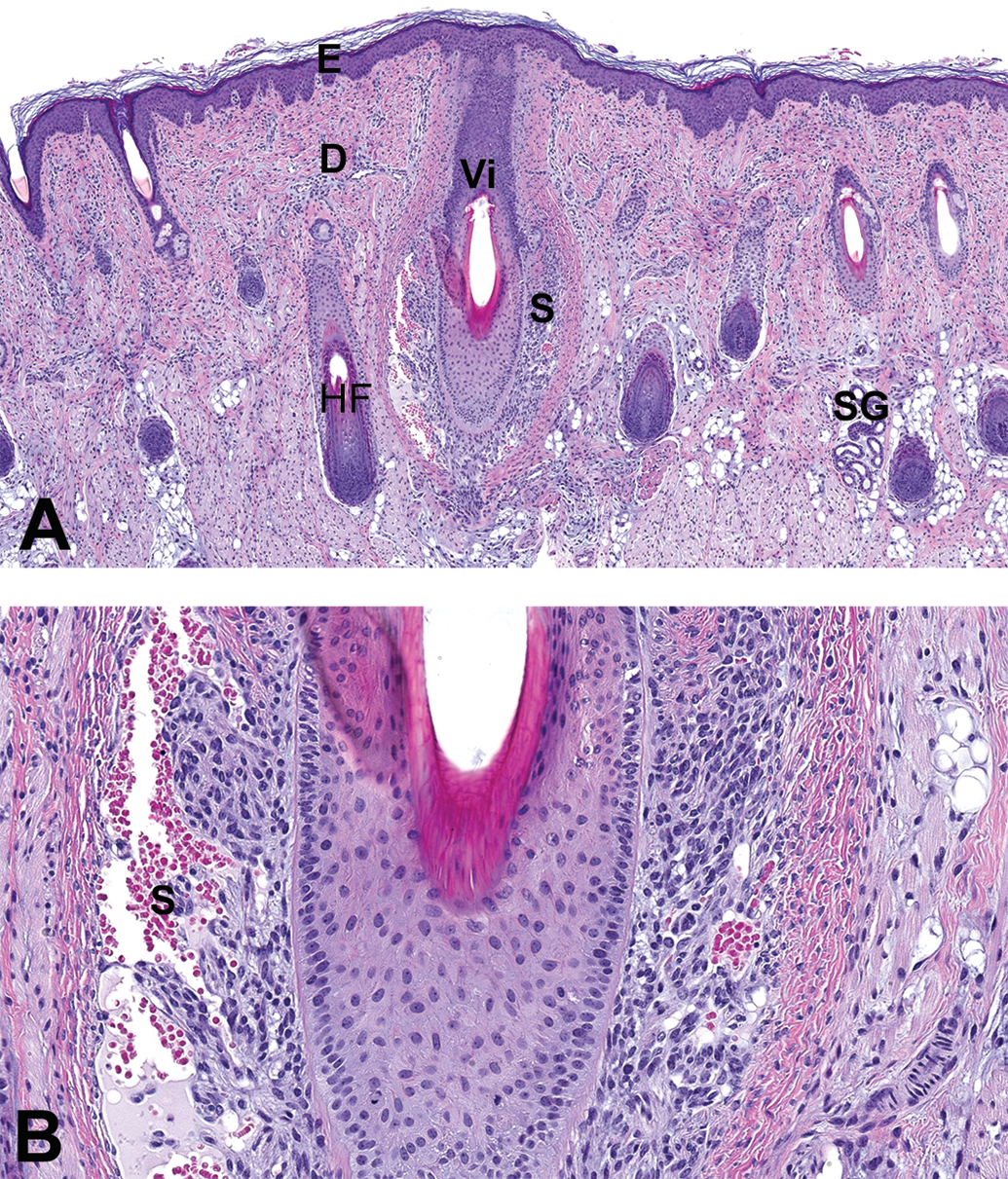
Morphology of the vibrissa in a juvenile male at PND1. Samples taken from the snout of a juvenile male at PND1: hematoxylin and eosin stain. (A) Low magnification (scan magnification ×5) and (B) high magnification (×20). PND = postnatal day.
Skin maturation from PND1 to week 12
At PND8 and PND15, the morphology of the skin of both the male and the female still differed from that observed at week 12 in all anatomical regions. As illustrated in Figure 5 (skin maturation of the flank from PND1 to PND15), the dermis still showed a diffusely dense cell population, although the cellular density appeared clearly lower than that of the neonatal skin at PND1.
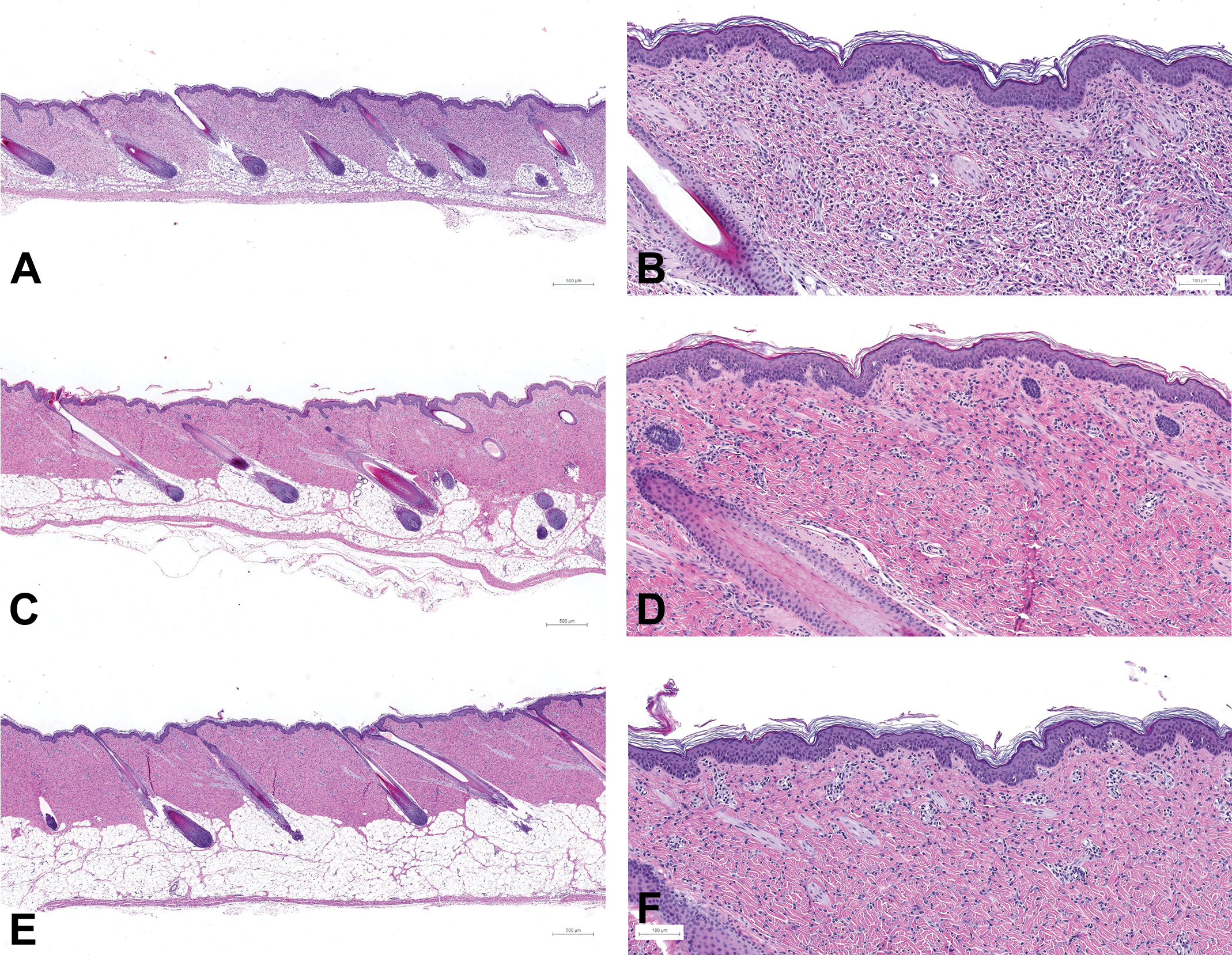
Skin maturation of the flank (posterior area) in females from PND1 to PND15. Samples taken from the flank posterior area of female juvenile minipigs at PND1 (A: scan magnification×5 and B: ×10), PND8 (C and D), PND15 (E: ×5 and F: ×10): hematoxylin and eosin stain. PND = postnatal day.
At PND29, all regions showed a morphology identical to what was seen in animals at week 12 (see Figure 6 for the skin morphology at PND29 and W12).
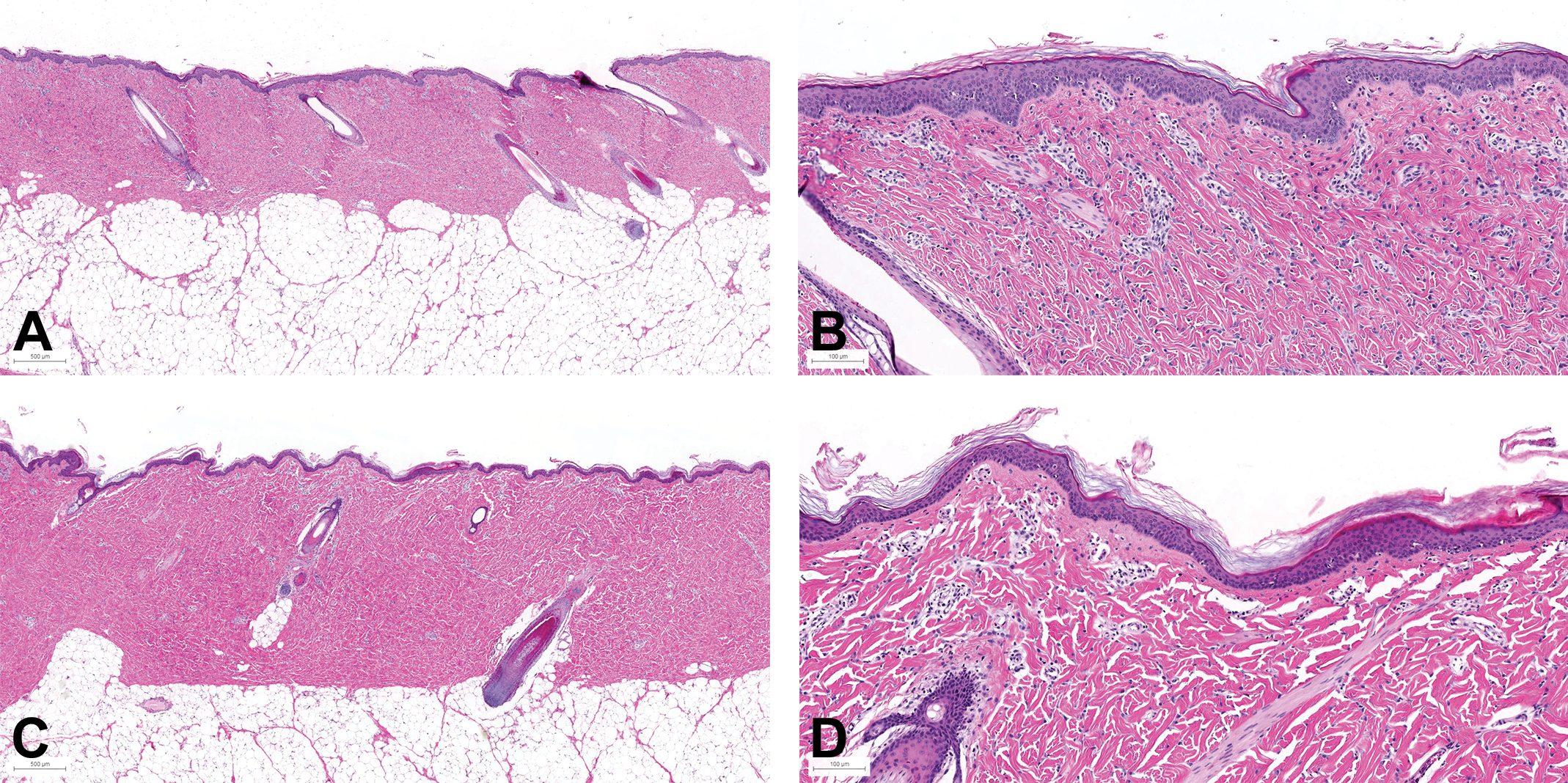
Skin morphology of the flank (posterior area) in female at PND29 and W12. Samples taken from the flank posterior area of female juvenile minipigs at PND29 (A and B: scan magnification×10) and at week 12 (C: ×5 and D: ×10): hematoxylin and eosin. PND = postnatal day.
At W12, the presence and morphology of appendices (sweat glands, hair follicles, and sebaceous glands), as well as the thickness of the three skin layers (epidermis, dermis, and hypodermis), differ according to the anatomical location. Apocrine glands, tubular glands with a coiled secretory duct related to the hair follicle lumen, are widely distributed in the minipig.
Sebaceous glands were seen in association with hair follicles in almost all samples, regardless of the location and age. In almost all locations, including the flanks where the formulations are commonly applied in dermal toxicity studies, the sebaceous glands were very small (transverse section width less than 40 µm with about four differentiated sebocytes). They were found to be slightly larger at the snout and at the chin.
Among the different locations evaluated, the snout and the chin had a very specific morphology. They showed a thicker epidermis and a thicker hypodermis when compared to other anatomical regions. In addition, large muscular bands crossing through the dermis were observed at both locations. Vibrissae, a very unique structure, was also found in snout and chin sections of minipigs. These specialized hair follicles were larger than other hair follicles and were surrounded by a sinus.
Skin Layer Thickness Maturation over Time from PND1 to Week 12
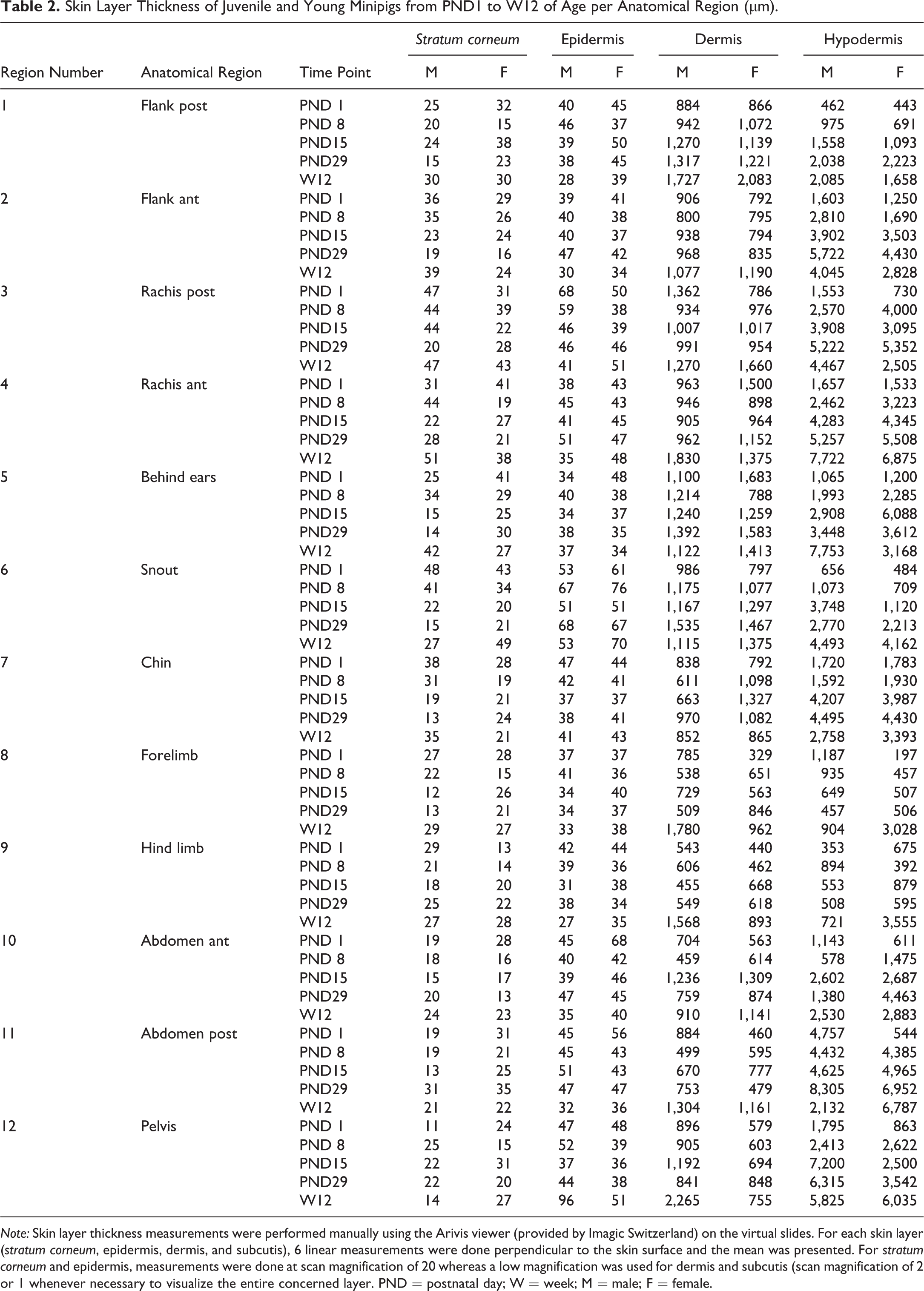
The thickness of each layer according to the anatomical location and time point is shown in Table 1 (presented by time point) and Table 2 (presented by anatomical region). The anatomical variations are illustrated for animals from PND1 to week 12 in Figures 7 to 11.
Skin Layer Thickness of Juvenile and Young Minipigs from PND1 to W12 of Age (µm).
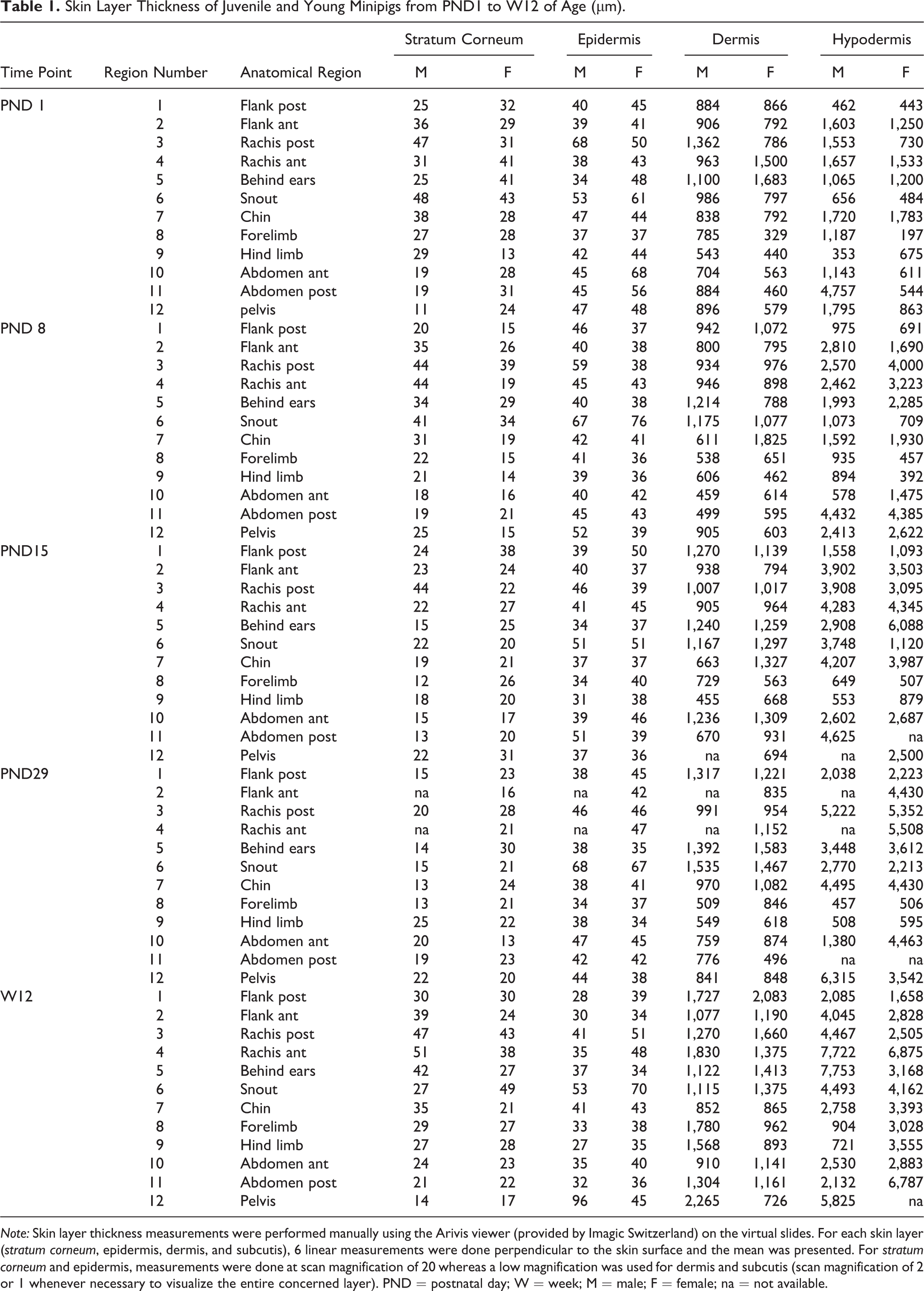
Skin Layer Thickness of Juvenile and Young Minipigs from PND1 to W12 of Age per Anatomical Region (µm).
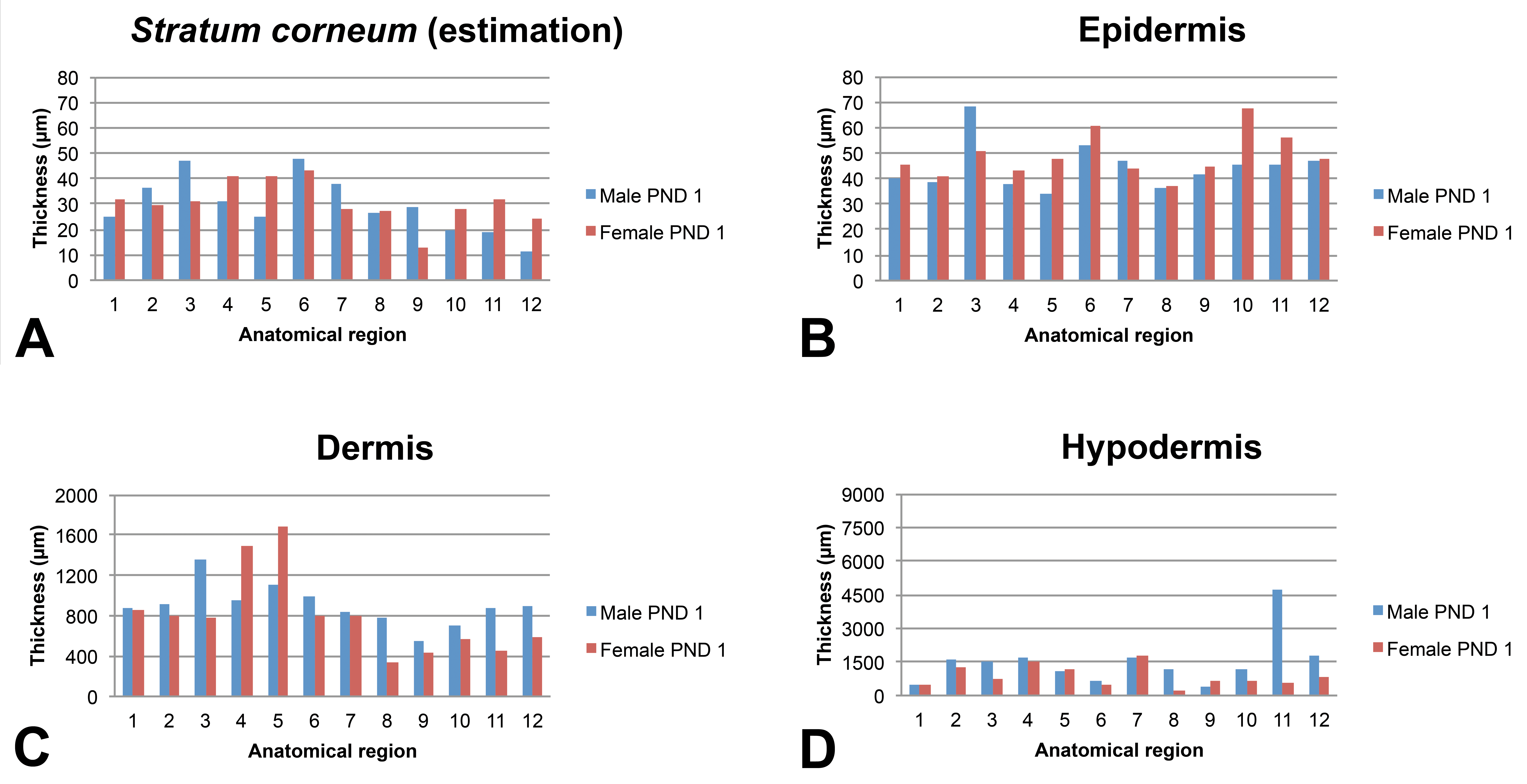
Skin layer thickness (µm) in juvenile minipigs at PND1 according to the anatomical location. 1: flank (posterior area), 2: flank (anterior area), 3: around the spine (rachis, posterior area), 4: around the spine (rachis, anterior area), 5: behind the ears, 6: snout, 7: chin, 8: internal face of the forelimb, 9: internal face of the hind limb, 10: abdomen (anterior area), 11: abdomen (posterior area), and 12: ischium–pelvis area. Skin thickness measurements were performed manually using the Arivis viewer (provided by Imagic Switzerland) on the virtual slides. For each skin layer (
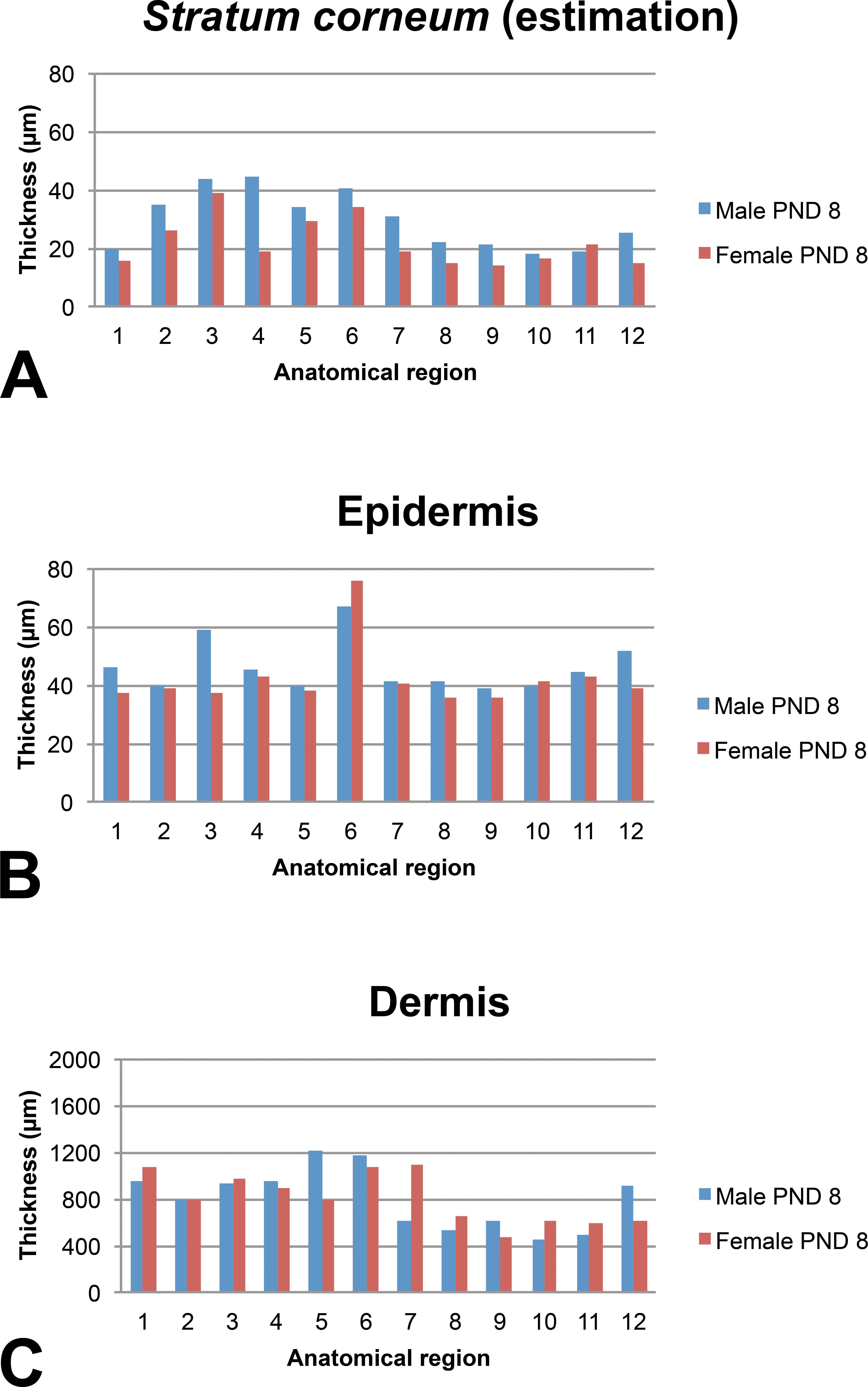
Skin layer thickness (µm) in juvenile minipigs at PND8 according to the anatomical location. 1: flank (posterior area), 2: flank (anterior area), 3: around the spine (rachis, posterior area), 4: around the spine (rachis, anterior area), 5: behind the ears, 6: snout, 7: chin, 8: internal face of the forelimb, 9: internal face of the hind limb, 10: abdomen (anterior area), 11: abdomen (posterior area), and 12: ischium–pelvis area. Skin thickness measurements were performed manually using the Arivis viewer (provided by Imagic Switzerland) on the virtual slides. For each skin layer (
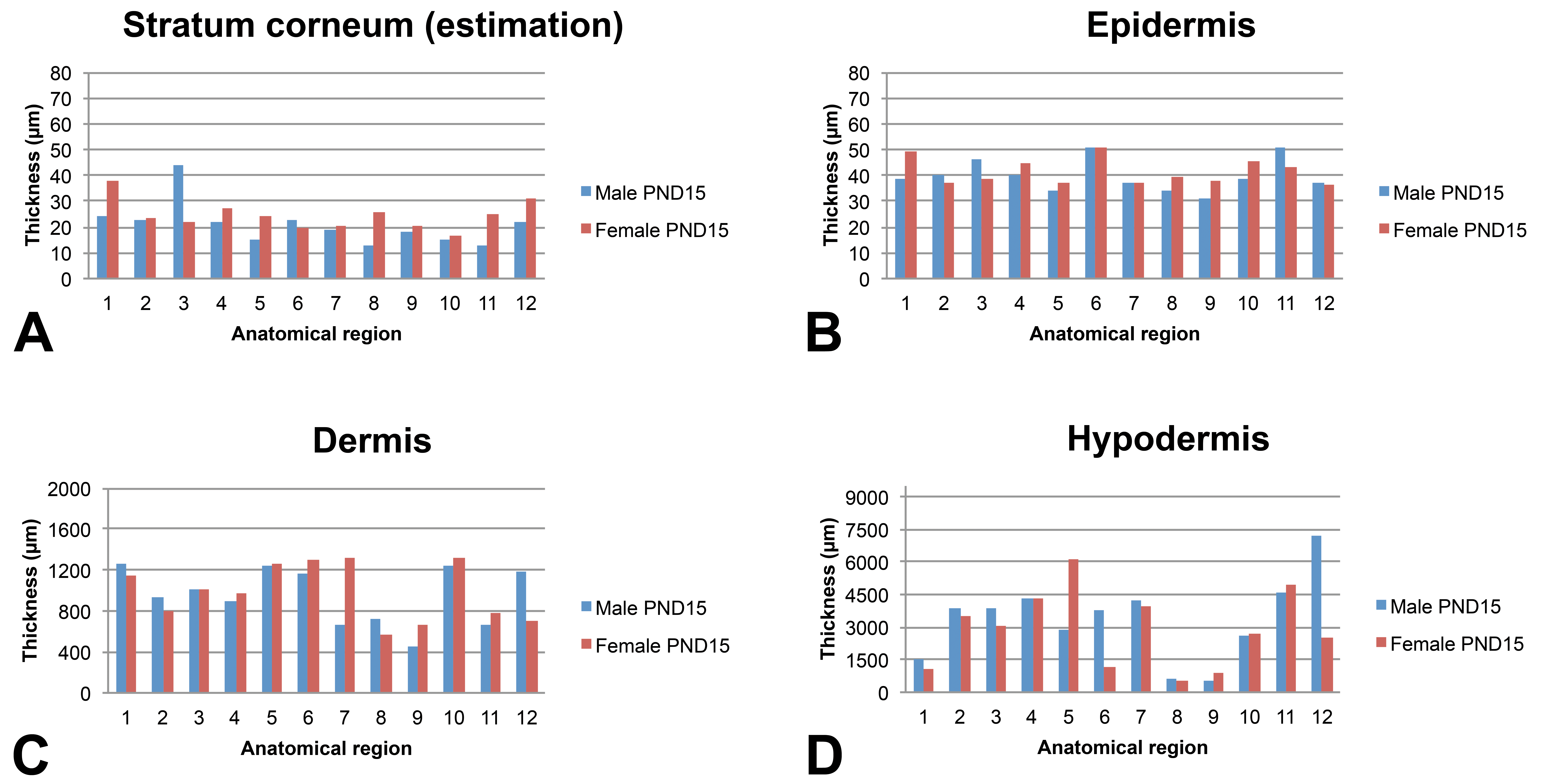
Skin layer thickness (µm) in juvenile minipigs at PND15 according to the anatomical location. 1: flank (posterior area), 2: flank (anterior area), 3: around the vertebral column (rachis, posterior area), 4: around the vertebral column (rachis, anterior area), 5: behind the ears, 6: snout, 7: chin, 8: internal face of the forelimb, 9: internal face of the hind limb, 10: abdomen (anterior area), 11: abdomen (posterior area), and 12: Ischium–pelvis area. Skin thickness measurements were performed manually using the Arivis viewer (provided by Imagic Switzerland) on the virtual slides. For each skin layer (
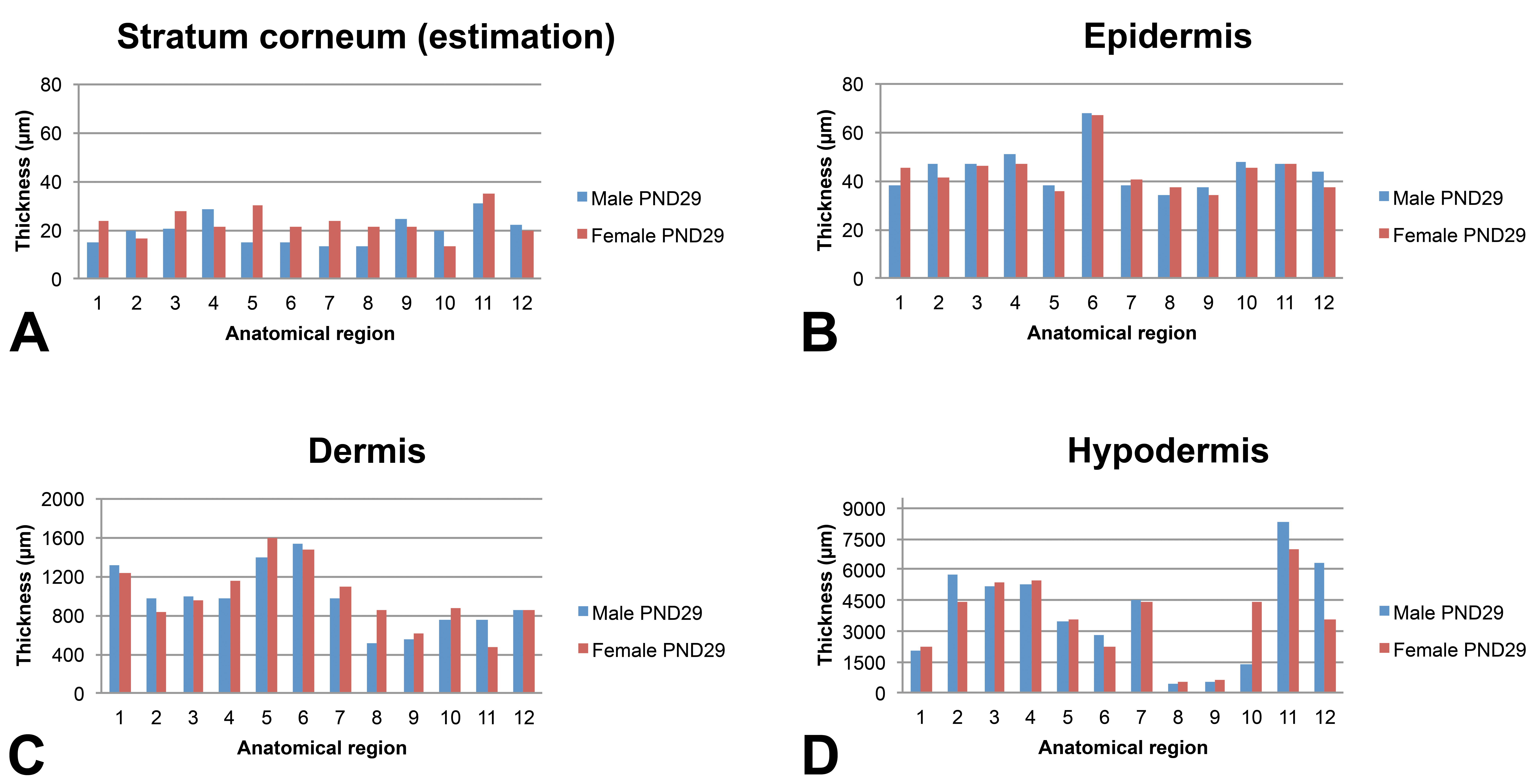
Skin layer thickness (µm) in juvenile minipigs at PND29 according to the anatomical location. 1: flank (posterior area), 2: flank (anterior area), 3: around the spine (rachis, posterior area), 4: around the spine (rachis, anterior area), 5: behind the ears, 6: snout, 7: chin, 8: internal face of the forelimb, 9: internal face of the hind limb, 10: abdomen (anterior area), 11: abdomen (posterior area), and 12: ischium–pelvis area. Skin thickness measurements were performed manually using the Arivis viewer (provided by Imagic Switzerland) on the virtual slides. For each skin layer (
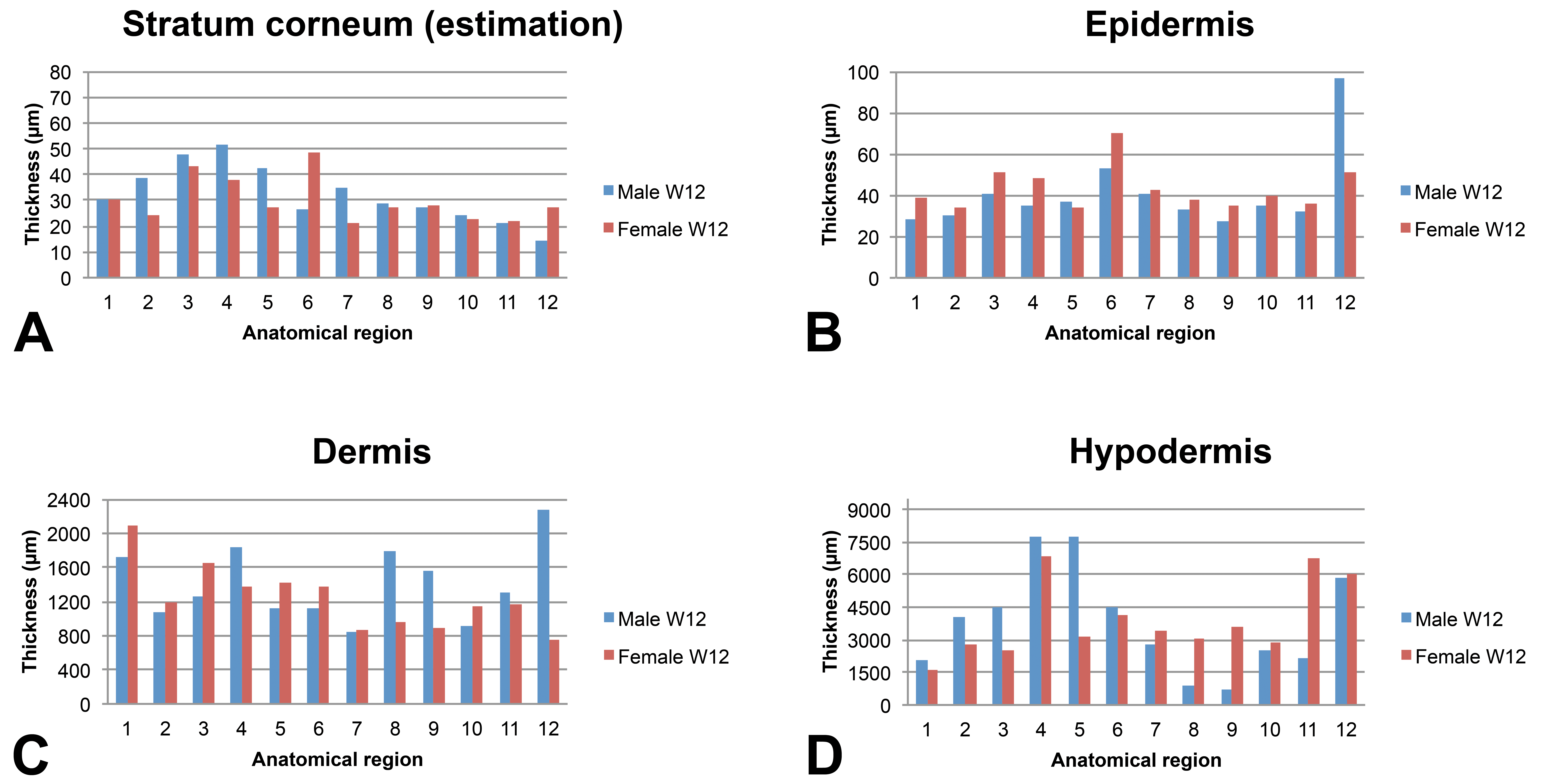
Skin layer thickness (µm) in juvenile minipigs at W12 according to the anatomical location. 1: flank (posterior area), 2: flank (anterior area), 3: around the spine (rachis, posterior area), 4: around the spine (rachis, anterior area), 5: behind the ears, 6: snout, 7: chin, 8: internal face of the forelimb, 9: internal face of the hind limb, 10: abdomen (anterior area), 11: abdomen (posterior area), and 12: ischium–pelvis area. Skin thickness measurements were performed manually using the Arivis viewer (provided by Imagic Switzerland) on the virtual slides. For each skin layer (
In the very young animals (juvenile at PND1 or PND8), the estimated thickness of the
The epidermis thickness in very young animals shows marked variations depending on the anatomical region at PND1 (ranging from about 35 µm in the internal face of the forelimb and behind the ears to about 60 µm for the snout). Individual very high values were noted for the spine region of the male at PND1 and for the abdomen region of the female at PND1. Conversely, the epidermal thickness appeared very homogeneous in all anatomical regions (approximately 40 µm) except the snout at PND8 and in older animals.
Regardless of the age of the animals, the thickness of the dermis showed marked variations depending on the anatomical region. As a general rule, the thinnest dermal compartments were seen for the internal face of the forelimb and hind limb and for the abdomen and for the pelvis area whereas a thicker dermis was seen for the samples taken behind the ears and the snout.
The subcutis was found to be highly variable depending on the anatomical region and differences tended to increase over time. In all animals, the thinnest regions were always the internal face of the forelimb and of the hind limb (region numbers 8 and 9, respectively).
Two samples were taken from the flank region, from the spine region, and from the abdomen: one sample at the anterior part and one located at the posterior part of each anatomical region (numbers 1 and 2 for the flank, 3 and 4 for the rachis, and 10 and 11 for the abdomen). Overall, regardless of the time point and sex, the anterior and posterior samples were almost identical from a morphology or thickness point of view, except for the hypodermis thickness in the flank and abdomen regions. The anterior area always showed a thicker hypodermis than the posterior area for the flank region (approximately 2-fold) whereas the thickness of the posterior part of the abdominal skin (closest to the pelvic area) was always found to be thicker than the anterior part (closest to the thoracic region).
Maturation of the flank region is illustrated in Figure 12. The estimated thickness of the
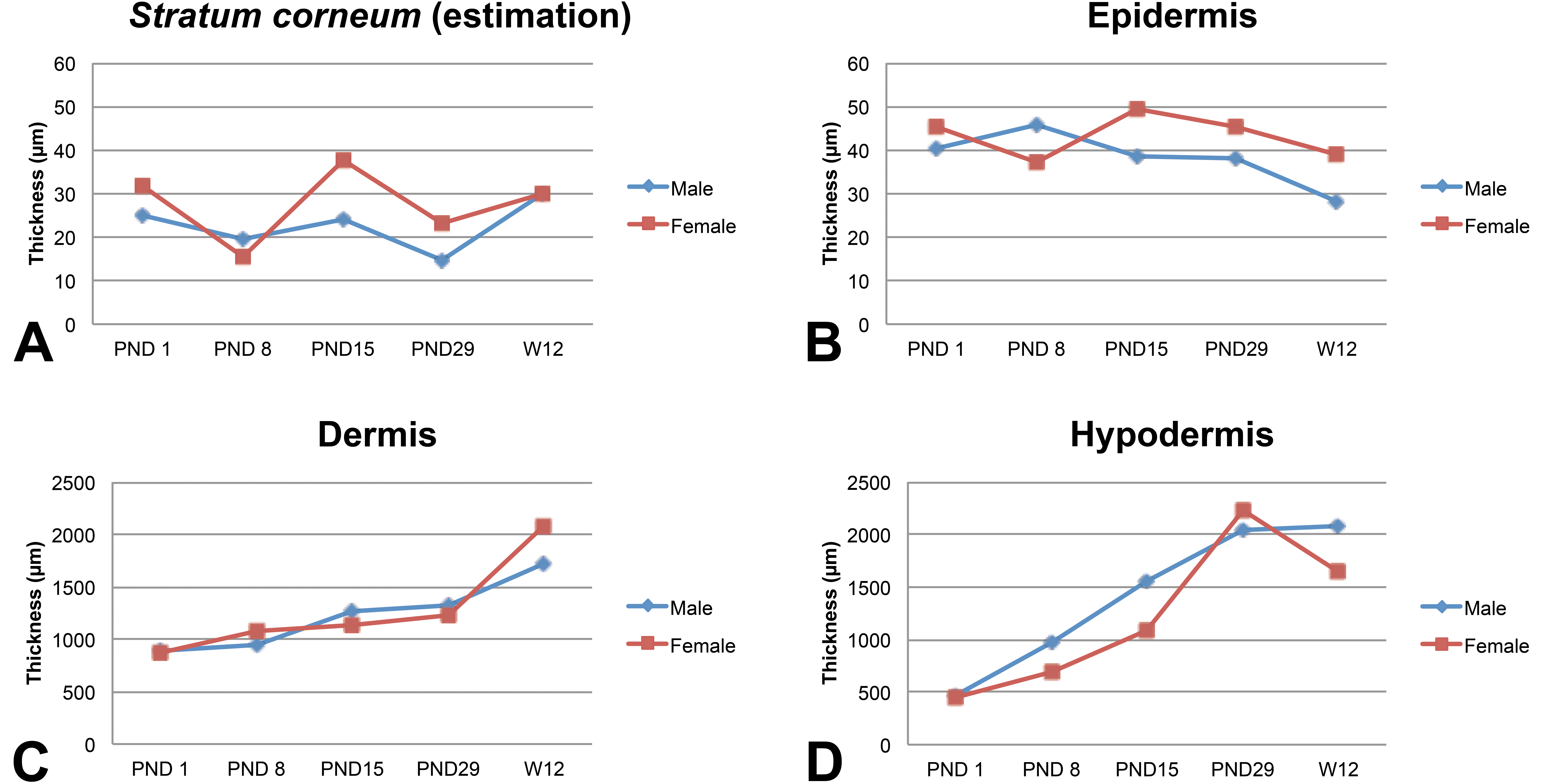
Maturation changes of the skin layer thickness (µm) in juvenile animals (flank, posterior area). Skin layer thickness measurements were performed manually using the Arivis viewer (provided by Imagic Switzerland) on the virtual slides. For each skin layer (
Discussion
In the minipig, as in humans, the morphology of the skin layers (
Regardless of the anatomical location, the morphology of the juvenile minipig skin samples taken at PND1 differs dramatically from that of young animals at 12 weeks of age, the main feature being the very high cellularity observed in the dermis. No specific literature data of dermal morphology was found for juvenile minipigs; however, this feature corresponds to what is well known and described for human neonates.
The age range of the animals in this study covered the skin maturation between PND1 and week 12. No specific translational information was found in the literature regarding minipig skin maturation, and therefore, we used the comparative diagram of age categories across species reproduced in the review article from De Schaepdrijver in 2010, although it was based on the central nervous system and the reproductive system development. The age categories used in our study may correspond to the evolution between neonate and child (from birth to approximately 12 years old). More specifically, the period from birth to 2 weeks in minipigs was thought to correspond to the term/neonatal period in humans (i.e., birth to 0.08 years); the period from 2 to 4 weeks in minipigs is considered to correspond to the infant/toddler period in humans (i.e., from 0.08 to 2 years), and the period from 4 to 14 weeks in minipigs was considered as equivalent to the childhood in humans (from 2 to 12 years).
From an embryologic point of view (reviewed in Loomis 2001), epidermis derives exclusively from ectoderm while dermal origins in humans are known to vary depending on the body site: neural crest ectoderm for the dermal mesenchyme of the face and anterior scalp, dermomyotome for the back, and mesoderm for limbs and ventral trunk. During the embryonic period, a presumptive epidermis forms as a single cell layer associated after 6 weeks, but for a short period of time, with a second more superficial layer called periderm. Stratification occurs later during pregnancy, approximately between 11 and 15 weeks for the skin appendages and between 22 and 24 weeks for the interfollicular epidermis. The fetal epidermis consists of a well-differentiated stratified epithelium resembling the adult skin by the middle of the third trimester. The fetal mesenchymal part of the skin is mainly cellular with few fibers and an extracellular gel-like matrix consisting of hydrated proteoglycans. The collagen and elastics fibers assembled in the second half of the pregnancy and the amount of fibers increases over time. In humans, the skin of the neonate shows a thick and well-organized dermis, but when compared to the skin of adults, the cellular component that consists mainly of fibroblasts is still prominent. This corresponds to our observations in neonatal minipigs.
Although several references could be found to support the interest of Göttingen minipig for dermal studies, only scant information was found on the potential impact of the age and/or bodyweight of the minipig on skin thickness. Qvist et al. (2000) documented the thickness of the skin layers according to age ranges in the minipig and compared them to human skin, but these data are based on a limited number of animals (
To our knowledge, this is the first time that an extensive skin anatomical morphology documentation is performed in the minipig, and it demonstrates marked anatomical differences in morphology and skin layer thickness. Some areas on the body, such as the chin and snout, have a very specific morphology, but it is also noted that skin layer thickness may also differ within the anatomical regions that are classically considered as homogeneous. For example, our assessment emphasized the flank region, which was defined using anatomic landmarks, that is, as a rectangle between the scapulae and hipbones, and corresponds to the area commonly used for dermal application in dermal toxicology studies. In this documentation work, two samples were performed taken, and it was noted in all animals that the anterior and posterior samples were almost identical in morphology or thickness except for the subcutis thickness. Indeed, the anterior area consistently showed a thicker subcutis than the posterior area (approximately 2-fold). This example highlights the fact that definition and standardization of skin samples are of major importance in dermal toxicology studies in order to provide adequate controls and reproducible results.
Overall, the neonate skin of minipigs showed the typical 3-layer structure of a mature skin and, as observed in neonate humans, the cellular component of the dermis appeared prominent and dermis and hypodermis thicknesses were smaller when compared to young adult skin. Over time, these unique neonatal characteristics tended to disappear progressively and one month after birth, the skin sections were very similar to those taken at week 12. The utility of the adult Göttingen minipig as an animal model in regulatory toxicology is already recognized for dermal application studies, and our results demonstrate that the minipig may also be the animal of choice for juvenile studies.
Footnotes
Acknowledgment
We thank Dr. Marie-France Perron Lepage for her very valuable review.
Author Contribution
Authors contributed to conception or design (BG, LP, JB, BR); data acquisition, analysis, or interpretation (BG, LP, NB); drafting the manuscript (BG); and critically revising the manuscript (LP, NB, JB, BR). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
