Abstract
Both a rodent and a nonrodent species are required for evaluation in nonclinical safety studies conducted to support human clinical trials. Historically, dogs and nonhuman primates have been the nonrodent species of choice. Swine, especially the miniature swine or minipigs, are increasingly being used in preclinical safety as an alternate nonrodent species. The pig is an appropriate option for these toxicology studies based on metabolic pathways utilized in xenobiotic biotransformation. Both similarities and differences exist in phase I and phase II biotransformation pathways between humans and pigs. There are numerous breeds of pigs, yet only a few of these breeds are characterized with regard to both xenobiotic-metabolizing enzymes and background pathology findings. Some specific differences in these enzymes based on breed and sex are known. Although swine have been used extensively in biomedical research, there is also a paucity of information in the current literature detailing the incidence of background lesions and differences between commonly used breeds. Here, the xenobiotic-metabolizing enzymes are compared between humans and pigs, and minipig background pathology changes are reviewed with emphasis on breed differences.
Keywords
Introduction
Pigs have been models in biomedical research for decades, and there are numerous breeds of minipigs in use in research worldwide. The use of the pig as a nonrodent model in toxicological studies continues to grow. It has been reported that in 2008, over 500 Center for Drug Evaluation and Research (CDER) reviews using minipigs had been filed within the previous 5 years (Brown, Stricker-Kongrad, and Bouchard 2013). The advent of the increased availability of minipigs, coupled with the knowledge that the physiology of the pig is more similar to humans than is the mouse, has led to an increased utilization of pigs in the biomedical sciences (Freeman et al. 2012).
We have learned from other species, including mice and rats, that colonies originating from a small population which is subsequently separated and then continually bred diverge both genetically and phenotypically from the parent population over time. We also know that there are strain-specific differences in background lesion incidence among rodents. Breed or strain differences have been addressed in other species of biomedical importance, especially rodents, but they have not been examined in depth in the minipig. While the Göttingen minipig is a specific breed, there are several reproductively separate commercial and research populations, which may have some degree of variance. The Göttingen minipig is available in Europe from Ellegaard, in North America from Marshall Bioresources, and recently became available in Japan from Oriental Yeast Company (Ellegaard Minipigs 2010). Additionally, much of the comparative research that has been performed on the Göttingen minipig evaluating cytochrome p450s (CYP) has been performed on a separate group of animals derived from a colony maintained at Brno University in the Czech Republic, which are not commercially available (Anzenbacher et al. 2002; Anzenbacher et al. 1998; Anzenbacherova et al. 2003; Anzenbacherova et al. 2005; Soucek et al. 2001). We examined the literature available as well as data collected from different breeds and these separate breeding populations of minipigs to evaluate differences in xenobiotic-metabolizing enzymes and background pathological findings. Although the genomes of several pig breeds have been sequenced, to date, no sequence comparisons of the drug-metabolizing enzymes and receptors has published (Choi et al. 2015; Groenen et al. 2012). There are CYP idiosyncrasies among minipig breeds, which may result in differences in drug metabolism. Differences in background pathological findings are present among the breeds. Although 70 breeds of pig are native to China alone (Fang et al. 2012), this report covers background pathology data only from breeds of biomedical importance that are readily available in North America and Europe, namely, the Göttingen, Hanford, and Yucatan breeds.
Drug Entry Pathways
Drugs are administered using numerous routes in toxicological studies, the most common being oral, intravenous, and dermal. Consideration of administration routes is important depending on whether the drug is active in the native state, or whether a metabolite is the active moiety, and metabolic activity in the gastrointestinal (GI) tract (oral) and skin (dermal) may determine which route is utilized.
The GI tract and skin are the most common sites of drug administration and both show metabolically active capabilities. In the GI tract, passive cellular diffusion is the primary mechanism of intestinal drug absorption (Lennernas 2007; Sugano et al. 2010; Thorn 2012). Profound species differences exist in the GI tract, and all facets of the GI tract are important in drug absorption and metabolism. Factors that contribute to the differences include presence and amount of salivary amylase; the pH of the stomach and small and large intestines; rate of gastric emptying and GI transit time; and the age of the animal. For instance, young pigs more readily absorb large molecules because of the physiological ability to absorb colostrum. In addition, frequency of feeding affects gastric emptying and a twice-daily regimen results in a more human-like gastric emptying (Suenderhauf and Parrott 2013). Bile salts that solubilize lipophilic drugs are also present in the GI tract. There is similar bile flow in the pig, dog, and human, all of which are lower than in rodents (Martinez et al. 2002). The GI tract is the most important extrahepatic site of drug biotransformation with CYP enzymes within enterocytes, the most common being CYP3A (Ding and Kaminsky 2003; Paine et al. 1997; Thorn 2012). Overall, pigs have a similar gut physiology to humans with similar salivary amylase amounts, gastric pH and emptying, and GI transit time.
The skin of the pig is morphologically similar to that of humans; however, differences in anatomy and physiology exist and can vary between anatomic locations. In the pig, the skin on the flank (Figure 1) and behind the ear (Figure 2) is thinnest and is most similar to human skin. Skin on the porcine dorsal neck and over the shoulders thickens as animals age and is thicker in males than in females. Males may also display glandular dilatation of apocrine glands. There are also increased numbers of sebaceous glands associated with the hair follicles in this region. These changes need to be considered when applying drugs and normal anatomic variations need to be taken into consideration when interpreting porcine skin sections. Although less is currently known about the drug-metabolizing enzymes in the skin compared with the liver, kidney, and intestine in pigs (Oesch et al. 2007), metabolizing enzymes are documented in keratinocytes (Baron et al. 2001). There is a recent comprehensive review comparing xenobiotic-metabolizing enzymes in the skin among species (Oesch et al. 2014). Juveniles are often used in toxicity studies, as they have thinner skin than adult animals. In Bama minipigs, the porcine CYP3A4 analog (CYP3A29) is increased in the skin with age. Since pigs have a reduced capability for sulfation, glucuronidation compensates in some instances in the skin, whereas in humans, only sulfate conjugation occurs (Toutain, Ferran, and Bousquet-Melou 2010). It has been reported that pigs are deficient at sulfate conjugation of phenols, naphthols, acetaminophen, and possibly others (Capel, Millburn, and Williams 1974; Smith and Timbrell 1974). Thus, when evaluating a compound that is reliant on sulfation for inactivation and elimination, the pig is not the best choice as a model organism.
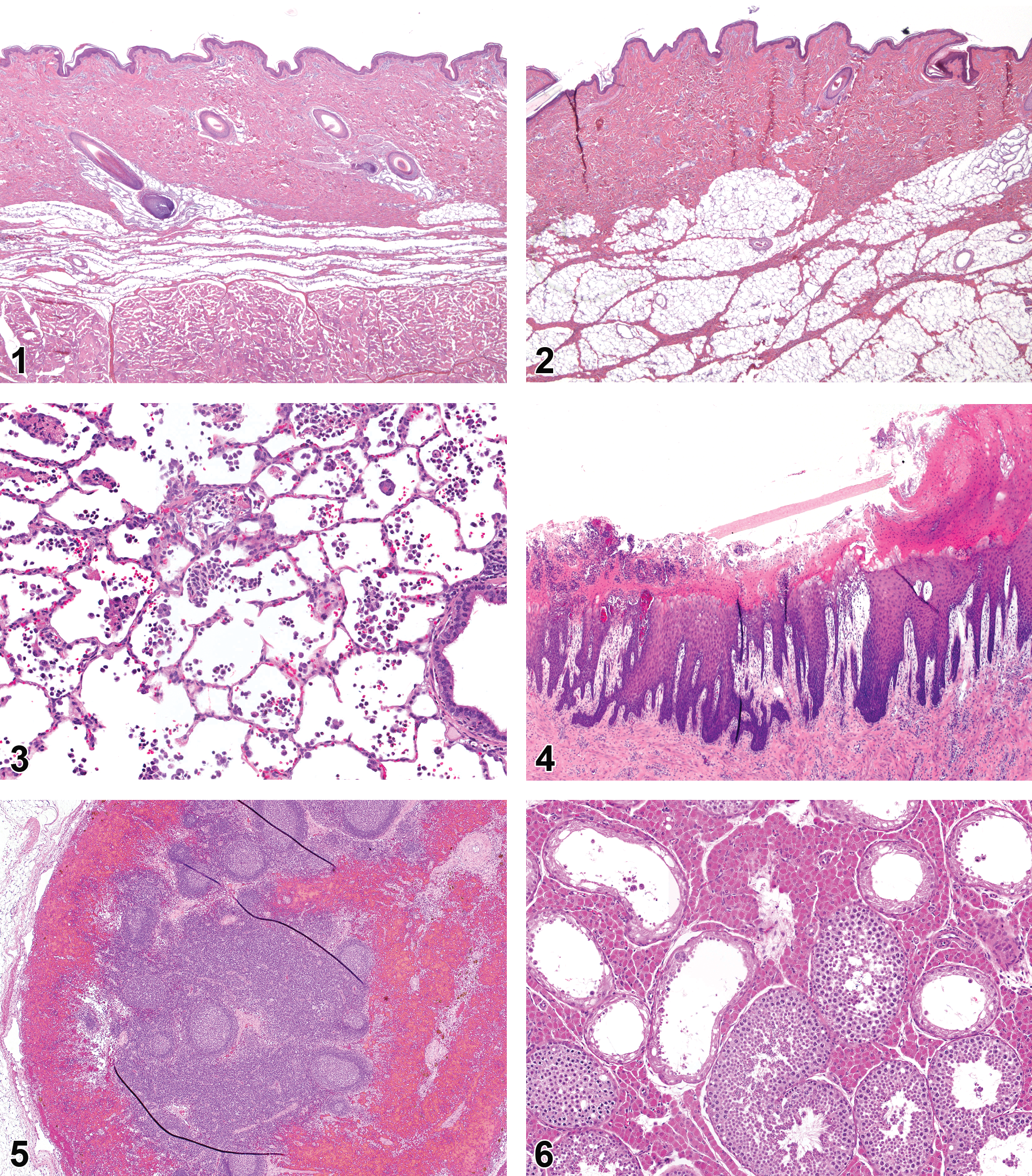
The kidney is also metabolically active and in some cases surpasses the metabolic activity of the liver (Anders 1980; Benet, Kroetz, and Sheiner 1996). The metabolizing enzymes found in the kidney are primarily CYP. The renal proximal convoluted tubules contain the highest levels of CYP enzymes. Renal drug metabolism enzymes have not been extensively studied in the pig. The kidney is the most important organ for elimination of drugs and their metabolites.
Drug Metabolism Reactions
In addition to the metabolizing capabilities of the GI tract and skin, primary or presystemic extraction or metabolism of agents occurs in the liver, where both phase I and phase II enzymes are present. Phase I reactions typically expose functional groups of the parent compound, which may result in increased activity or loss of activity. Phase I reactions provide exposure of functional groups for phase II reactions to take place (Benet, Kroetz, and Sheiner 1996). Phase I reactions are mediated by CYPs, flavin monooxygenases, monoamine oxidases, and molybdenum hydroxylases as well as many others (Antonovic and Martinez 2011). The phase I reactions consist of oxidative, reductive, hydrolytic, and dealkylation reactions (Antonovic and Martinez 2011; Lock and Reed 1998).
Phase II reactions result in the formation of covalent linkage between the functional group via glucuronidation, sulfation, glutathione conjugation, or acetylation (Antonovic and Martinez 2011; Benet, Kroetz, and Sheiner 1996). The phase II reactions result in increased polarity of the molecule to aid in excretion.
The most important phase I enzymes in humans belong to the CYP enzyme family. The CYPs are functionally conserved in all mammals and are found in all organisms including bacteria. In humans, primarily 3 CYP families are involved in the majority of all drug biotransformation, CYP1, CYP2, and CYP3 (Toutain, Ferran, and Bousquet-Melou 2010). Within the cells, CYPs are present primarily in the endoplasmic reticulum and to a lesser extent in the mitochondria (Antonovic and Martinez 2011). Total CYP has been measured in the liver in numerous species, and among different breeds within a species. Humans, dogs, and farm pigs have equivalent amounts of CYP within the liver, whereas minipigs have greater than twice the amount and nonhuman primates have up to 3× the amount found in humans (Dalgaard 2015). Not only are there different amounts of CYP among species, but there are polymorphisms which result in both interspecies and intraspecies variations. Allelic variations are also common, and some individuals carry multiple copies of certain CYPs resulting in increased metabolism of certain xenobiotics (Fink-Gremmels 2008; Vaclavikova et al. 2004).
A challenge when examining the current literature is the heterogeneity in interpreted study end points: some studies measure messenger RNA (mRNA), some measure protein levels, others measure activity levels of the protein, and still others measure both protein and activity. All parameters are important to measure since posttranscriptional regulation of CYP is an important regulatory mechanism. Another variation is the way in which the activity of the CYP is measured. In humans, transformation of specific substrates is used to define the activity of a specific CYP. In the pig, evidence indicates that these specific substrates may be metabolized by more than 1 porcine CYP and different substrates from those commonly used for human CYP testing may be more appropriate for measuring porcine CYP (Skaanild and Friis 2007). Buffer strength, type, and pH can all significantly affect the metabolism reaction, and while standardized assay conditions are recommended, none have been adhered to for CYP activity assays. Different pig breeds have been shown to have differing amounts of CYP and different activity levels of CYP (Helke and Swindle 2013). CYP mRNA levels can change with age and may vary between sexes (Sakuma et al. 2004; Skaanild and Friis 1997; Vaclavikova et al. 2004). Other variables in CYP diversity include epigenetics and diet (Antonovic and Martinez 2011), with dietary fat shown to modulate CYP in pigs via transcriptional and posttranscriptional mechanisms (Puccinelli et al. 2013).
Much of the work characterizing cytochromes in pigs has been done to better understand “boar taint.” Boar taint is a characteristic of pork produced by the metabolism products skatole and androstenone by CYP and results in unpalatable pork. These studies have focused primarily on CYPs associated with boar taint, specifically CYP1A1, CYP2A19, CYP2C33v4, CYP2C49, CYP2E1, CYP2D6, CYP2C49, and CYP3A (Wiercinska, Lou, and Squires 2012). Most of these studies were done on farm pigs, not minipigs, and many focused on males, as they are more likely to be affected.
Cytochrome Families
There are over 2,000 cytochromes defined. Approximately 60 of these are found in humans, of which only a few metabolize approximately 75% of drugs (National Institutes of Health 2015). Different CYP isoforms are present in different species, although there is homology among species. Since the sequences do vary between species, some activity differences are expected.
CYP1
In humans, the CYP1 family metabolizes about 13% of substances tested including carcinogens, estrogens, mycotoxins, xanthenes, some antidepressants, and analgesics (Nebbia et al. 2003; Rendic 2002; Skaanild 2006). This cytochrome family is induced by the same agents across species. In minipigs and in humans, CYP1A activity is sex related, however, not similarly. In minipigs, females have 2 to 4× higher activity than males, whereas in humans, Caucasian males have 2 to 4× higher activity than females (Shimada et al. 1994; Skaanild and Friis 1999). One study examining wild pigs showed lower levels of CYP1A in dams compared to piglets, suggesting the amount of CYP1A in pigs decreases with age (Mizukawa et al. 2015). In humans, CYP1B1 is the predominant isoform outside the liver, yet is not well characterized in the minipig (Chirulli et al. 2007; Puccinelli, Gervasi, and Longo 2011).
CYP2
The CYP2 family metabolizes up to 50% of all prescription medications as well as nicotine, nitrosamines, and aflatoxin (Al Omari and Murry 2007; Skaanild 2006). Porcine CYP2A19 represents 34% of hepatic CYP in the pig and has 90.1% DNA sequence homology to human CYP2A13 and 87.2% homology to human CYP2A6 (Achour, Barber, and Rostami-Hodjegan 2011). Porcine CYP2A19 is 99% homologous between Göttingen and conventional breed pigs. Female Göttingens have 70× higher activity than males, but when males are castrated, the activity of CYP2A in these animals increases 10× (Gillberg, Skaanild, and Friis 2006; Skaanild 2006). Sex differences have also been reported in Yucatans, where the females have 5× higher activity than males (Bogaards et al. 2000). Sex differences in CYP activity have not been reported in humans, although gender differences in protein levels have been reported in both pigs and humans (Skaanild and Friis 1999), and differences between human races are also evident (Al Omari and Murry 2007). There are marked species and sex differences in CYP2A. In minipigs, the CYP2A gene is influenced by sex hormones, and large interindividual variations in CYP2A activity occur due to transcriptional regulation (Gillberg, Skaanild, and Friis 2006; Skaanild 2006). In contrast, CYP2A variations in humans are primarily due to polymorphisms (Skaanild and Friis 2005).
CYP2B enzymes metabolize diazepam, lidocaine, cyclophosphamide, and tamoxifen (Skaanild 2006). There is 81.1% DNA sequence homology between human CYP2B6 and porcine CYP2B22 (Kojima and Morozumi 2004). Overall, pigs have higher levels of CYP2B than humans (Bogaards et al. 2000; Myers et al. 2001). Piglets have higher protein levels of CYP2B than their dams (Mizukawa et al. 2015). No sex differences in protein amounts have been found in Yucatan pigs (Bogaards et al. 2000). Numerous inconsistencies in the literature exist regarding CYP2B in the pig likely due to different breeds, ages, genders, and testing methods being used (Helke and Swindle 2013). Some groups report 7-pentoxyresofufin dealkylation reaction due to CYP2B, but other groups have been unsuccessful in producing this reaction with porcine samples (Anzenbacher et al. 1998; Behnia et al. 2000; Desille et al. 1999; Monshouwer et al. 1998; Skaanild and Friis 1999). This may be due to breed differences, sex differences, different source of hepatocytes or microsomes, or reaction assay differences.
In humans, CYP2C metabolizes 25% of drugs including losartan, propofol, estrogens, testosterone, and methadone (Rendic 2002). Porcine CYP2C33v4 has only 62.6% homology to human CYP2C9 (Achour, Barber, and Rostami-Hodjegan 2011). Porcine CYP2C enzymes show some cross-reactivity toward many human test substrates, but not those specific for human CYP2C, making extrapolation between pigs and humans for CYP2C difficult (Skaanild 2006; Skaanild and Friis 2008; Thorn 2012).
Antidepressants, antipsychotics, and beta-blockers are metabolized by CYP2D in humans (Rendic 2002). In humans, there are high interindividual variances in CYP2D due to polymorphisms. CYP2D25 comprises 25.5% of hepatic CYP in the pig and is 77.6% homologous to human CYP2D6 (Achour, Barber, and Rostami-Hodjegan 2011). CYP2D has not been fully examined in the pig, and many of the substrates that are specific for human CYP2D are also metabolized by porcine CYP2B (Skaanild 2006). A study was performed that examined clearance of specific drugs in pigs, and CYP2D6 substrate metabolism occurred faster in the pig than other species (Mogi et al. 2012; Thorn 2012).
The CYP2E enzyme family metabolizes alcohols, ketones, anesthetics, and nitrosamines (Skaanild 2006). Metabolism by CYP2E can lead to production of highly reactive toxic or carcinogenic metabolites. There is 75% homology between human and porcine protein sequences (Rendic 2002; Skaanild 2006). This family of CYP can be induced by alcohol and high-fat diets (Skaanild 2006). CYP2E is also induced physiologically with stress with up to an 8-fold increase in protein with no change in mRNA levels, indicating a posttranscriptional method of protein upregulation (Antonovic and Martinez 2011). In both the Göttingen and the Yucatan breeds of minipigs, the females have higher CYP2E activity than the males (Zamaratskaia and Zlabek 2009).
CYP3
The CYP3 family is the most important drug-metabolizing family. It metabolizes approximately 34% of compounds and represents 30 to 40% of total CYP in the human liver (Omiecinski, Remmel, and Hosagrahara 1999; Rendic 2002). This family is involved in steroid hydroxylation, conversion of sex hormones, and metabolization of polycyclic aromatic hydrocarbons and pesticides (Lock and Reed 1998; Omiecinski, Remmel, and Hosagrahara 1999; Rendic 2002). Expression of CYP3A is found primarily in the liver, intestine, and kidney but is found in other sites as well. With expression in intestine and liver, it plays a role in both presystemic and systemic drug disposition (Dresser, Spence, and Bailey 2000). There are differences in transcriptional regulation of CYP3A between humans and pigs, and while tissue expression patterns are similar, interindividual variances have been detected (Shang et al. 2009). Yucatan pigs have higher activity of CYP3A compared to Göttingen and conventional pigs (Bogaards et al. 2000; Skaanild and Friis 1997, 1999). Tissue expression patterns also change with age. In Bama minipigs, piglets showed higher expression of CYP3A29 in duodenum than in liver, while the reverse was true for adults (Shang et al. 2009). A recent finding shows that the diet can differentially affect CYP3A between the sexes. Ingestion of chicory root, which acts as a ligand for receptors regulating CYP, results in decreased enzyme activity in males, whereas in females, activity is increased (Rasmussen and Ekstrand 2014; Rasmussen et al. 2012).
Phase II Reactions
Phase II reactions result in covalent linkage between a functional group and glucuronic acid, sulfate, glutathione, amino acids, or acetate. These reactions increase the polarity of the compound to aid in excretion. In drug biotransformation, glucuronidation and sulfation are the most important phase II reactions. Not much research has been done on phase II reactions in the pig; however, it is known that sulfate conjugation in swine is absent or slower than in other species. Sulfation is the major conjugation pathway for phenols and contributes to biotransformation of alcohols, amines, and thiols (Caldwell 1986; Lohr, Willsky, and Acara 1998). Pigs compensate for this deficiency by using other phase II pathways in order to metabolize enzymes (Merrifield et al. 2011). It has been shown that there are similar levels of glutathione transferase and uridine-diphosphate (UDP)-glucuoronosyl transferase found in both humans and pigs, although the pig is more efficient than the human at glucuronidation (Merrifield et al. 2011; Thorn 2012). In contrast to the dog, pigs have a high acetylating capability (Toutain, Ferran, and Bousquet-Melou 2010). Not much is known about Uridine 5’-diphospho-glucuronosyltranssferase (UGT) in the pig (Thorn 2012). There is gene duplication of UGT in humans, rodents, and Duroc pigs, but there are 4 copies in Göttingen minipigs (Vamathevan et al. 2013).
Background Pathology
To better define the current knowledge of background lesions in minipigs, data were collected from 3 breeds: Göttingen, Hanford, and Yucatan. The Göttingen data originated from animals that were bred at 2 different sites (United States and Denmark). Only data from animals born after 2000 were included to minimize the potential effects of genetic drift. Data were separated into animal age-groups; less than 2 months, 2–6 months, and greater than 6 months. In North America, data from 1,078 Göttingen, 118 Hanford, and 88 Yucatan minipigs were gathered from several biopharmaceutical contract research laboratories and combined with data from 881 Göttingen minipigs in the European Union (EU; Helke et al. 2015; Jeppesen and Skydsgaard 2015). Entities found in greater than 1% of the population are presented for each system (Table 1).
Overview Table of Lesions Found in Minipigs: Differences Between Breeds and Geographical Locations.
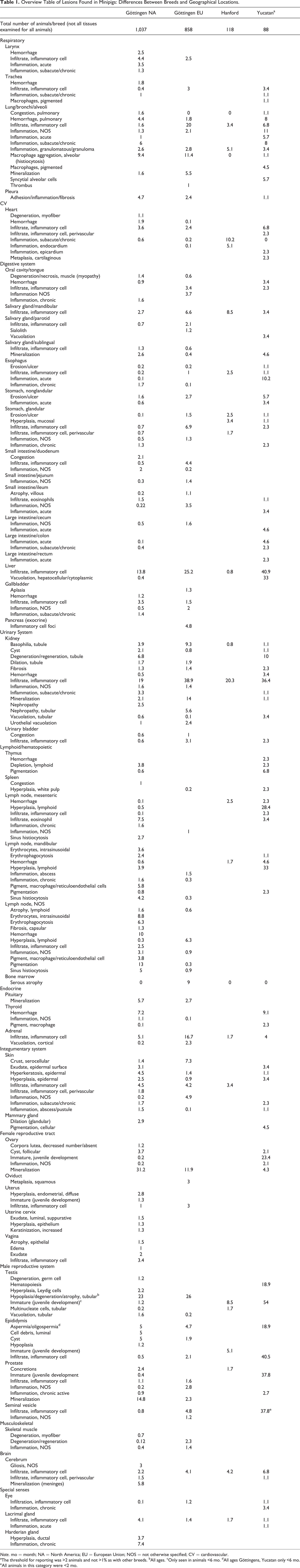
aThe threshold for reporting was >2 animals and not >1% as with other breeds. bAll ages. cOnly seen in animals <6 mo. dAll ages Göttingens, Yucatan only <6 mo. eAll animals in this category were <2 mo.
Many terms were used to represent similar changes; these have been rationalized for the purposes of this article. For example, morphologically similar inflammatory cell infiltrates might be identified as chronic, subacute/chronic, mononuclear, lymphocytic, and mixed cell. Since these all represent a similar entity, they were combined into a single morphological finding of subacute/chronic inflammation for the purpose of this report.
Systemic
Only 2 animals of 2,155 total minipigs had systemic diseases. One animal had thrombocytopenic purpura and 1 had porcine stress syndrome. Both of these animals were in the EU group of Göttingen minipigs (Helke et al. 2015).
Respiratory System
The most common background findings in the larynx were in the Göttingen minipigs, in which inflammatory cell infiltrates were reported. In the North American groups, inflammation was both acute and chronic, and hemorrhage in the larynx was also commonly reported. There were no laryngeal findings in the Hanford or Yucatan minipigs. In the trachea, inflammatory cell infiltrate was present in Göttingen and Yucatan minipigs less than 2 months old. Hemorrhage (which may be iatrogenic) was also noted in the trachea of North American animals.
Within the lungs, Göttingen minipigs commonly had the findings of alveolar macrophages, the incidence of which decreased with age (12.4% of animals <2 months, 9.9% of animals 2–6 months, and 5.6% of animals >6 months). Pulmonary hemorrhage, inflammatory cell infiltrate, and inflammation (Figure 3), specifically granulomatous and granulomas, were also common in Göttingens. Pulmonary mineralization, pleural adhesions, and associated fibrosis were seen in Göttingen minipigs (Helke et al. 2015; Jeppesen and Skydsgaard 2015). The Hanford breed commonly displayed granulomatous inflammation or granulomas and inflammatory cell infiltrate (Helke et al. 2015). Yucatan minipigs also had pulmonary granulomatous inflammation or granulomas but also displayed acute, subacute, and chronic inflammation. In Yucatans less than 2 months of age, syncytial alveolar cells and pigmented macrophages were common, which were not reported in animals greater than 6 months old. Inflammatory cell infiltrate increased with age in the Yucatan breed (Helke et al. 2015).
Cardiovascular System
Inflammatory cell infiltrates were reported in the heart of both Göttingen and Yucatan minipigs (Helke et al. 2015; Jeppesen and Skydsgaard 2015). Göttingens also displayed mesothelial hyperplasia and hypertrophy more commonly in animals >6 months of age (Helke et al. 2015). Hemorrhage and cardiomyofiber degeneration were common in North American Göttingen minipigs (Helke et al. 2015). The Hanford breed commonly had subacute to chronic myocardial inflammation and endocardial inflammation (Helke et al. 2015). Epicardial inflammation and cartilaginous metaplasia of the epicardium were reported in Yucatan minipigs (Helke et al. 2015).
Digestive System
Both Göttingens and Yucatans had inflammatory cell infiltrate of the tongue (Helke et al. 2015; Jeppesen and Skydsgaard 2015). Göttingens also had lingual inflammation and myocyte degeneration and necrosis which were more commonly seen in males (Helke et al. 2015; Jeppesen and Skydsgaard 2015). The Yucatan breed had hemorrhage of the tongue, and no tongue lesions were noted in Hanfords (Helke et al. 2015). In salivary glands, inflammatory cell infiltration was commonly seen in both Göttingen and Hanford minipigs (Helke et al. 2015; Jeppesen and Skydsgaard 2015). Mineralization was reported in the Göttingen breed, and in EU, sialoliths were reported (Helke et al. 2015; Jeppesen and Skydsgaard 2015). Salivary gland findings were rare in the Yucatan minipigs (Helke et al. 2015). Esophageal inflammation was seen in the Göttingens and in Yucatans younger than 2 months (Helke et al. 2015; Jeppesen and Skydsgaard 2015). The nonglandular portion of the stomach of both Göttingens and Yucatans displayed erosions and ulcers (Figure 4), whereas the glandular portion of the stomach in the Hanford displayed erosions and ulcers (Helke et al. 2015; Jeppesen and Skydsgaard 2015). Inflammatory cell infiltrates and inflammation were seen in the glandular portion of the stomach of Göttingens and Yucatans (Helke et al. 2015; Jeppesen and Skydsgaard 2015). In all 3 breeds examined, small intestinal background lesions were minimal and were composed of inflammatory cell infiltrates and inflammation, and large intestinal background findings were rare (Helke et al. 2015; Jeppesen and Skydsgaard 2015).
Hepatic infiltration of inflammatory cells was common in the Göttingens and Yucatans (Helke et al. 2015; Jeppesen and Skydsgaard 2015). Yucatans also displayed vacuolation of hepatocytes in animals less than 2 months old (Helke et al. 2015). Adhesions and fibrosis of the hepatic capsule were common in Hanfords (Helke et al. 2015). The gall bladder in Göttingen minipigs displayed hypoplasia or aplasia of the gall bladder, which was more common in EU animals (Helke et al. 2015; Jeppesen and Skydsgaard 2015). Inflammation and inflammatory cell infiltrate in the gall bladder were both common in Göttingens; and in the U.S. population, hemorrhage was observed (Helke et al. 2015; Jeppesen and Skydsgaard 2015).
Urinary System
Inflammatory cell infiltrates were the most common finding in the kidney of all breeds (Helke et al. 2015; Jeppesen and Skydsgaard 2015). Göttingens also displayed inflammation, mineralization, tubular degeneration and regeneration, tubular dilation, and tubular basophilia (Helke et al. 2015; Jeppesen and Skydsgaard 2015). The mineralization of the renal papilla in the Göttingen is phenotypically different than mineral in other organs in that it is eosinophilic on hematoxylin and eosin staining (Helke et al. 2015). The Yucatans also displayed inflammatory cell infiltrates, hemorrhage, fibrosis, and vacuolation of renal tubular epithelium (Helke et al. 2015). Females of all breeds were overrepresented for inflammatory cell infiltration of the kidney, and female Göttingens had increased tubular dilatation and fibrosis compared to males, while Yucatan females had increased tubular degeneration and regeneration compared to males (Helke et al. 2015; Jeppesen and Skydsgaard 2015).
Lymphoid/Hematopoietic
In the Göttingen minipigs, sinus histiocytosis, inflammation, pigmented macrophages, and lymphoid atrophy were all seen in the lymph nodes (Helke et al. 2015; Jeppesen and Skydsgaard 2015). Hemorrhage in the lymph nodes (Figure 5) was observed in all breeds. Serous atrophy of bone marrow is only reported in the Göttingen breed and was more common in EU animals as well as males, with 5× higher incidence in males versus females (15.5% males, 2.6% females; Helke et al. 2015; Jeppesen and Skydsgaard 2015). This change was also more common in animals as they age (1.6% in <2 months, 6.3% in 2–6 months, and 21.4% in animals >6 months old; Helke et al. 2015; Jeppesen and Skydsgaard 2015).
Musculoskeletal
Degeneration and/or regeneration of skeletal muscle as well as inflammatory cell infiltrate in the skeletal muscle also occurred in Göttingen minipigs (Helke et al. 2015; Jeppesen and Skydsgaard 2015).
Integumentary System
Göttingen minipigs displayed many dermal background findings consisting of hyperkeratosis, hyperplasia, exudate, serocellular crust or scab, abscesses or pustules, inflammation, and inflammatory cell infiltrates (Helke et al. 2015; Jeppesen and Skydsgaard 2015). Inflammatory cell infiltration was common in Hanfords (Helke et al. 2015). Yucatans also showed hyperplasia, exudate, and inflammation (Helke et al. 2015). Mammary glands of both male and female Göttingens displayed glandular dilatation (Helke et al. 2015; Jeppesen and Skydsgaard 2015). Cellular pigmentation of Yucatan mammary glands was present in 2- to 6-month-old females (Helke et al. 2015).
Female Reproductive Tract
Mineral deposits were found in ovaries of Göttingen and Yucatan minipigs (Helke et al. 2015; Jeppesen and Skydsgaard 2015). Inflammatory cell infiltrates were present in Göttingen uteri (Helke et al. 2015; Jeppesen and Skydsgaard 2015). No changes were found in the female reproductive tract of Hanfords (Helke et al. 2015).
Male Reproductive Tract
Testicular hypoplasia, tubular degeneration, and tubular atrophy (Figure 6) were found in 25% of Göttingen males (Helke et al. 2015; Jeppesen and Skydsgaard 2015). This was the most common finding in the male reproductive tract and increased mildly with age, occurring in 18% of animals less than 2 months old, 28% of those 2–6 months old, and in 30% of those greater than 6 months old (Helke et al. 2015; Jeppesen and Skydsgaard 2015). All Hanford males were reported to be sexually mature at 6 months of age (Helke et al. 2015). Yucatan males less than 2 months old were found to be immature, and 47% displayed hematopoiesis in the testicle (Helke et al. 2015). The epididymis of Göttingens revealed aspermia or oligospermia (Figure 7), luminal cell debris, and epididymal cysts (Figure 8; Helke et al. 2015; Jeppesen and Skydsgaard 2015). The epididymides of the Hanford minipigs were immature in 7% of 2- to 6-month-olds (Helke et al. 2015). The epididymis of Yucatans was immature and had inflammatory cell infiltrates in 100% of animals less than 2 months old, and in animals 2–6 months old, aspermia or oligospermia was common (31% of animals; Helke et al. 2015). Inflammatory cell infiltration was present in the seminal vesicle of Göttingens and Yucatans (Helke et al. 2015; Jeppesen and Skydsgaard 2015). Göttingens also had inflammation in the seminal vesicle (Helke et al. 2015; Jeppesen and Skydsgaard 2015). The prostate of Göttingens displayed mineralization, acinar dilatation, inflammatory cell infiltrate, and inflammation (Figure 9; Helke et al. 2015; Jeppesen and Skydsgaard 2015). Overall, many changes were present in the Göttingen male reproductive organs, whereas most changes in the Yucatan were in animals less than 2 months of age and associated with immaturity (Helke et al. 2015; Jeppesen and Skydsgaard 2015). The only findings in the male reproductive tract of Hanford minipigs were signs of immaturity in animals less than 6 months of age.
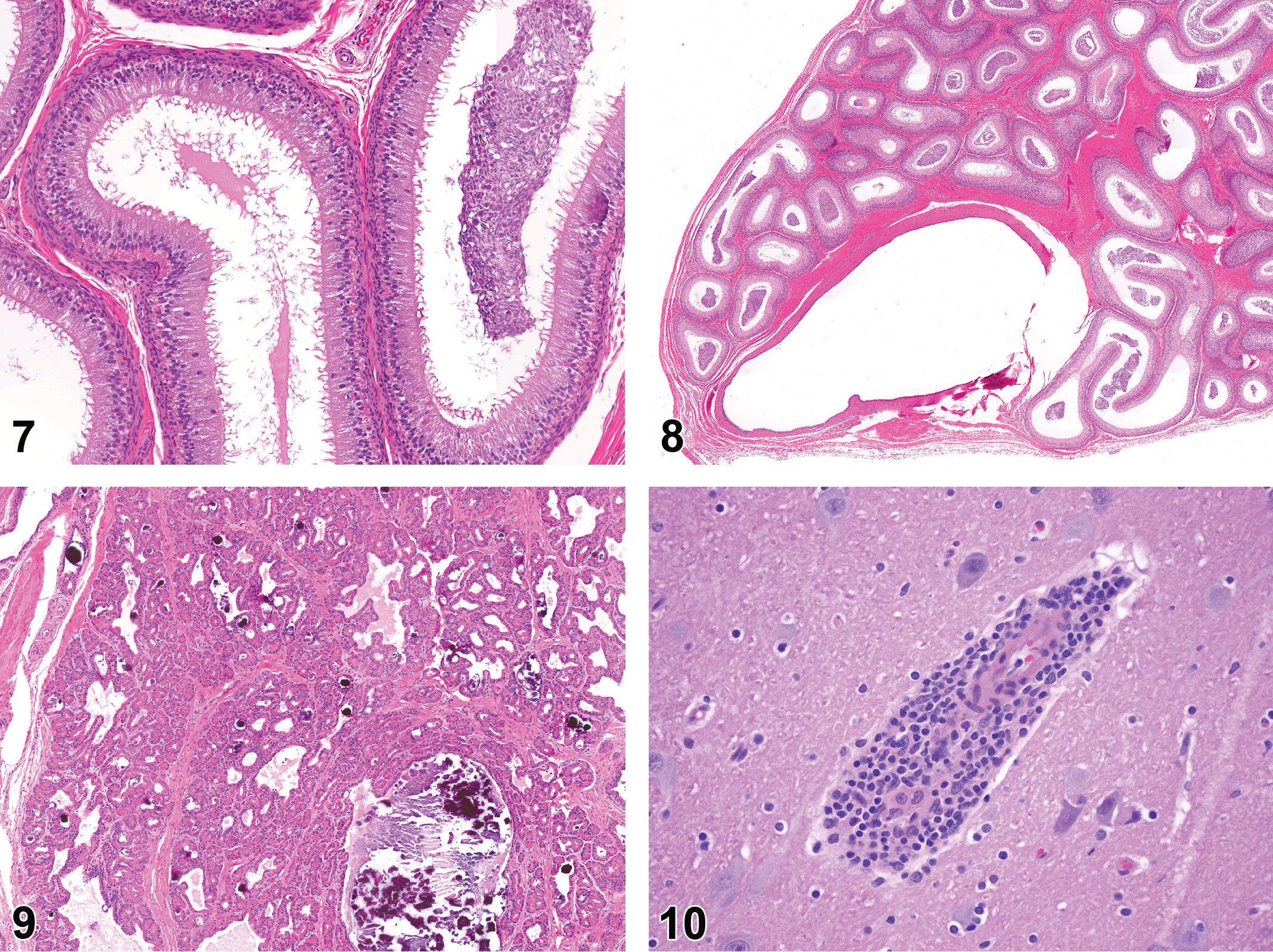
Nervous System
Within the brain, all 3 breeds had inflammatory cell infiltrates noted (Figure 10). Göttingens also displayed perivascular inflammation, and those in North America had mineralization of the meninges (Helke et al. 2015; Jeppesen and Skydsgaard 2015).
Endocrine
Hemorrhage (which may be iatrogenic) was noted in the thyroid gland of all 3 breeds (which may be iatrogenic; Helke et al. 2015; Jeppesen and Skydsgaard 2015). Mineralization of the pituitary gland was found in the Göttingen and Yucatan breeds, and incidence increased with age (Helke et al. 2015; Jeppesen and Skydsgaard 2015). Pituitary mineralization was not seen in any Göttingens less than 2 months old, 0.4% of animals 2 to 6 months old, and in 9.5% of animals >6 months old (Helke et al. 2015; Jeppesen and Skydsgaard 2015). Inflammatory cell infiltrates were present in the adrenal gland of all 3 breeds (Helke et al. 2015; Jeppesen and Skydsgaard 2015).
Special Senses
Inflammatory cell infiltrates were reported in the eyes of Göttingens and Yucatan minipigs (Helke et al. 2015; Jeppesen and Skydsgaard 2015). In the lacrimal gland of Göttingens and Hanfords, inflammatory cell infiltrates were reported (Helke et al. 2015; Jeppesen and Skydsgaard 2015).
Discussion
Göttingen minipigs have higher CYP content overall relative to 3 breeds of conventional pigs and 2 races of humans (Dalgaard 2015). Both CYP content and activity differ among breeds of pig and minipig. There are significant discrepancies in interpretation of CYP levels and substrate specificities. Both inducibility and the magnitude of induction differ across tissues and cell types even when exposed to the same inducer. CYP protein levels may be affected by androgen levels, which are inherited in an autosomal dominant manner (Brunius et al. 2012; Kojima and Degawa 2013). Transcriptions of CYP1A1, 2A19, and 2E1 are all decreased with testicular steroids in pigs (Zamaratskaia et al. 2012). Both phase I and phase II drug-metabolizing enzyme levels can be differentially affected by exposure to specific chemicals or dietary components (Meadus et al. 2014; Mizukawa et al. 2015; Rasmussen et al. 2012). A study evaluating differential expression of cytochrome b5A among pig breeds found that 3 single nucleotide polymorphisms in the promoter were responsible, emphasizing how minor changes in genetic code of the different breeds result in differing phenotypes (Bai et al. 2015). Other variables that affect phase I/II enzymes include not only specific nutrients but the amount of nutrients. Obese animals have been shown to have decreased levels of CYP, although not all CYPs are affected (Ghose et al. 2011), and variations exist between studies (Puccinelli et al. 2013). The maternal diet also has been shown to have an effect on cytochrome levels via epigenetic regulation (Cai et al. 2014; Cong et al. 2012).
Studies of the cytochromes and xenobiotic-metabolizing enzymes are important, but conclusions from the current literature are limited. Overall, there seem to be no major variations in substrates, inducers, inhibitors, and tissue distribution between humans and pigs in CYP1A1, CYP1A2, and CYP3A. Species differences are much more pronounced in phase I versus phase II metabolism. Much of the previous work has been done in microsomes (Yang et al. 2015) and will need to be expanded and completed in vivo. The current literature uses a range of isolation and testing techniques, which may not always be directly comparable. Studies of the cytochromes and xenobiotic-metabolizing enzymes are important for understanding the use of the minipig in biomedical research and safety assessment, but much of the available literature on minipigs focuses on the Brno Göttingen, which is not commercially available. Thus, variables affecting the interpretation of CYP levels include background genetics, gender, diet, epigenetic factors, transcriptional regulation, and circadian variation.
As seen with the results from the background data, there are differences in the Göttingen minipigs between the North American and European populations. Some background pathology findings are seen in both North America and European Göttingen animals, but differences in current protocols among CROs may result in variance between organizations. This may account for some of the differences seen between geographically separated Göttingen populations. Between organizations, there may even be varying guidelines for determining which findings should be recorded. For example, there are differences as to whether what is commonly known to be iatrogenic, such as venipuncture-associated hemorrhage of the trachea or thyroid, is routinely recorded. Although it has not been examined, it would not be surprising if there are differences between the Brno Göttingen population and other Göttingen populations that are commercially available. Many breed differences have been reported, although differences within a breed have not yet been examined with respect to cytochrome levels and activities.
The sample size for the Göttingen animals was robust, while the data from Yucatans and Hanfords represented fewer animals and could have been more affected by sampling bias. The prominence of background findings in these breeds may not be as notable once the sample size is increased.
In the male reproductive system, Hanfords appeared sexually mature at 6 months of age, and most findings in the Yucatan were found in animals less than 2 months of age and were associated with immaturity. There are numerous findings in the Göttingen male reproductive tract, which could confound interpretation of sexual maturity and testicular toxicity. Overall, females had a higher occurrence of renal lesions than males, the cause of which is undetermined.
Background pathology findings suggest Göttingens are more likely to have mineralization in tissues than Hanfords or Yucatans, and mineralization or concretions were more commonly reported in the male and female reproductive systems, and the central nervous system (CNS) in North American than in European Göttingen minipigs. In contrast, mineralization within the urinary tract and lung was more common in the European Göttingens. Whether these differences are due to genetics, husbandry, or other factors remains to be determined.
Pig biology must be considered when interpreting background findings as well. Pigs explore their environment with their snouts, and rooting is common behavior. If enthusiastic rooting takes place, foreign bodies in the respiratory tract and lungs with associated granulomatous inflammation should not be surprising.
The need for International Harmonization of Nomenclature and Diagnostic Criteria for the minipig was made clear during the conduct of this project. Making raw data available when publishing manuscripts of this type allows comparisons and addition of data sets as was done here thus increasing the power of following literature.
Age, sex, and breed of pig models are among the variables that need to be considered in pharmacological and toxicological studies. Breed differences need to continue to be accounted for when performing studies, and the most appropriate breed should be chosen based on what is known. There are also sex differences that need to be accounted for when planning and performing any study in minipigs.
No one species best models the human. Human responses can be better predicted when using a model that replicates the anatomy, physiology, and known metabolic reactions specific to the compound in question. Quantification of known background findings allows more careful analysis of the safety assessment and the relevance of given findings to the model species as well as to humans. Knowledge of factors such as CYP and other xenobiotic-metabolizing enzymes in model species and strains will further the relevant and focused use of these models for biomedical research purposes and preclinical safety evaluation. While genetic factors are important, other variables such as womb environment, diet, and body condition must also not be overlooked.
Footnotes
Acknowledgments
We would like to thank Dr. Lydia Andrews-Jones for providing images of some entities.
Author Contribution
Authors contributed to conception or design (KH, CH, KN, AMS); data acquisition, analysis, or interpretation (KH, CH, KN, AMS, BJ, SM, JH, VV, MG, AS, NN, DB); drafting the manuscript (KH); and critically revising the manuscript (KH, CH, KN, AMS, BJ, SM, JH, VV, MG, AS, NN, DB). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
