Abstract
It is important to know whether the animals used in toxicology studies are sexually mature. As minipigs are being used increasingly in toxicity studies, we reviewed published data on the age of sexual maturity in the minipig. Maturity in females was assessed on the basis either of normal cycles of progesterone secretion or of the histological presence of corpora lutea and, in males, was assessed on the histological appearance of the seminiferous tubules and epididymides. In female Göttingen minipigs, the first progesterone peak was at 3.7 to 4.2 or 6.1 to 6.5 months of age. These animals were in the presence of a boar. In female Göttingen minipigs in toxicology studies, which were not in the presence of a boar, at least 1 corpus luteum in the ovaries was present in only 50% of the females by 6.5 months of age, while all were mature by 7.7 months of age. Histological maturity in the male Yucatan minipig is reported to be attained at about 4.4 months old, but in male Göttingen minipigs at about 2 months old, although the definition of maturity may have been different in the 2 studies.
Introduction
It is important to know whether the animals used in toxicology studies are sexually mature because (1) if they are to be used for breeding in reproductive and developmental toxicity studies, they should be of the optimal age for reproduction to avoid poor fertility being misinterpreted as an effect of the test item; (2) in nonreproductive studies, the effects of the test item on the mature reproductive system should be examined where possible; and (3) it is important not to misinterpret incomplete sexual maturity as an effect of the test item. This is especially important for the testis, which is 1 of the organs most frequently affected by candidate pharmaceuticals in toxicology studies in nonrodents (Horner et al. 2013). Minipigs are a useful alternative to rabbits in reproductive and developmental toxicity studies and are being used increasingly in other toxicity studies.
Complete sexual maturity requires the production of gametes in sufficient amounts, the ability to mate, and the ability of the female to maintain pregnancy and give birth. In the data we reviewed here, maturity in females is assessed on the basis either of the presence of cycles of progesterone secretion or of the histological appearance of the ovaries and, in males, is assessed on the basis of the histological appearance of the seminiferous tubules and epididymides.
We review published data on the age of sexual maturity in the minipig that were presented during the 2015 Society of Toxicologic Pathology Symposium and make additional comments on the question of male maturity.
Method and Results
Details of the procedures for animal care and, where appropriate, the guidelines followed for the conduct of the work can be found in the original publications that are cited. Terminology used in this review for the histological appearance of the various stages of ovarian follicle development is taken from the International Harmonization of Nomenclature and Diagnostic Criteria (Dixon et al, 2014), although the terminology used in 1 of the publications to which we refer (Tortereau, Howroyd, and Lorentsen 2013) was taken from an older publication on human histology (Wheater, Burkitt, and Daniels 1979).
Study 1—Female Minipigs
Hormonal experiment Study 1 (de Rijk et al. 2014; Peter et al. 2016).
Pilot part of Study 1
Method
Three naive adult female Göttingen minipigs (approximately 10–11 months) supplied by Ellegaard, Denmark, were used. The following measurements (given in chronological order) were performed in the morning between approximately 11:00 and 12:00 a.m.: (1) daily home-cage observations for vulval changes and behavioral signs of heat, (2) daily recording of rectal temperature, (3) cytology of daily vaginal smears, (4) body weight measurements at regular intervals, and (5) collection of blood samples for measurement of the reproductive hormones progesterone and 17β-estradiol twice weekly.
Results
Progesterone analysis was a valuable method, showing a cyclic pattern of release during various cycles of estrous. Observations for signs of heat, cytology of vaginal smears, rectal body temperature, or release of 17β-estradiol were not useful parameters for determination of acquisition of sexual maturity.
Main Part of Study 1
Method
Progesterone levels in the serum were determined twice weekly in 14 naive young female Göttingen minipigs supplied by Ellegaard, Denmark. The animals were 3 to 4 months old at study start and were housed in the same room as adult boars. The females were necropsied at different time points in the estrous cycle based on the previous progesterone peak or, in the 1 animal that did not start to cycle, at 7 months of age. The ovaries, uterus, cervix, and vagina were examined histologically.
Results
The first progesterone peak was at 3.7 to 4.2 months of age (8.4–12 kg body weight) in 7 of the 13 animals that reached maturity, while in the other 6 animals, it occurred at 6.1 to 6.5 months of age (12.1–16 kg body weight). Histologically, all animals that had experienced a progesterone peak had corpora lutea or disrupted Graafian follicles. In contrast, the animal that had not started to cycle at 7 months of age had only small follicles and no degenerating corpora lutea or remnants of corpora lutea that were visible, indicating that no previous ovulation had occurred. The uterus of this animal was very small and contained very few endometrial glands with cuboidal to columnar luminal epithelium. The cervical epithelium had much less periodic acid-Schiff positive mucous material than in mature animals, the vaginal epithelium was only a few cells thick, and the mammary gland had terminal end buds but no lobular structures. These appearances were, however, less useful than those of the ovaries and uterus in distinguishing immature from mature animals.
Study 2
Post mortem evaluation (Tortereau, Howroyd, and Lorentsen 2013).
Method
This work correlated the following observations with age at necropsy: body weight, ovary, and uterus weights (absolute and relative to body weight) and histological features of the ovaries, uterus, vagina and mammary glands, thymus, and distal femoral epiphyseal growth plate. The subjects were 39 female Göttingen minipigs supplied by Ellegaard, Denmark, aged 5.8 to 20.7 months at necropsy that were controls in 10 nonreproductive general toxicology studies on pharmaceuticals. Boars were not present in the same room.
Results
The most useful indications of maturity were the presence of at least 1 corpus luteum in the histological sections of ovaries (by definition) and the weight and development of the uterus. Judging by the presence of at least 1 corpus luteum in the section of the ovaries, only 50% of the females were mature by 6.5 months of age, while all were mature by 7.7 months of age (13.5 kg body weight). The ovaries of mature females also had follicles, some of which were vesicular, tertiary, or Graafian.
Females that were considered completely immature had primordial and primary follicles. As they approached maturity, secondary, vesicular, and tertiary follicles were also present. Absolute ovarian weight was not a useful guide to the attainment of maturity, however. The uterus increased in size and development of the endometrial glands during maturation, and there was a clear difference in absolute and relative uterine weight between animals that had no corpora lutea (absolute weight up to approximately 50 g) and those which had started to cycle (absolute weight generally above 100 g). The histological maturation of the vagina and mammary gland was also described. Although the vaginal epithelium of completely immature animals was very thin (only 1 cell layer), the number of cell layers in partially mature animals was not always less than that in mature animals in the metestrous or diestrous phases. Mammary glands of mature animals had alveolar development and secretion, which were absent in completely immature or partially mature animals. All of the animals had open epiphyseal growth plates and none had thymic involution.
Study 3—Male Minipigs
Histological assessment of testes and epididymides (Evans et al. 2012).
Method
Testicular and epididymal histology was assessed at 12 to 24 weeks (2.7–4.4 months of age) in tissue removed at castration from Yucatan minipigs supplied by Sinclair BioResources, Columbia, MO. There were 4 or more animals in each age-group. Maturity was defined in this study as the presence in the testes of identifiable stages of the seminiferous epithelium with elongated spermatids. If no spermatids were present, or none had developed beyond the round stage, the animal was classed as immature.
Results
At 2.7 months of age, no spermatids or only round spermatids were present in the seminiferous epithelium of the testis. At 3.1 months of age, 3 of the 4 animals had spermatids that were sometimes elongate. A few epididymal sperms were present in 1 of the 4 animals. At 3.6 to 4 months of age, some testicular tubules were mature, but others lacked elongated spermatids. However, sperm had started to appear in the epididymis. At 4.4 months of age, almost all seminiferous tubules were mature, and there were plentiful epididymal sperm.
Study 4
Histological assessment of testes and epididymides (Taberner et al. 2015).
Method
Testicular and epididymal histology was assessed at 5, 6, 7, or 8 weeks (1–2 months) of age in 24 Göttingen minipigs (6 animals at each age), supplied by Marshall BioResources, North Rose, NY, that had not been in contact with mature boars. Postpubertal animals were defined in this study as having a majority cell type presence of elongated spermatids in the seminiferous tubules/tubule lumen (i.e., >60% tubules displaying a full spermatogenic cycle, including elongating spermatids). In fact, an average of 77.8% tubules examined displayed elongated spermatids as the most developed cell type. In conjunction with this, postpubertal animals had spermatozoa in the seminiferous tubule lumen (an average of 12% of tubules had spermatozoa) and cauda epididymis (an average of about 40% tubules in the cauda epididymis had spermatozoa). In peripubertal animals, >50% tubules contained 2 generations each of spermatogonia and primary/secondary spermatocytes, in addition to the presence of round or elongated spermatocytes, with a few spermatozoa in the seminiferous tubule lumen but none in the cauda epididymis. The least mature category, prepubertal, was defined as having >70% of seminiferous tubules, presenting spermatogonia and primary/secondary spermatocytes as the most advanced cell types.
Results
At 1 month of age, no testicular tubules were mature, but at 2 months of age, all animals were mature (postpubertal; Figure 1). The authors found a shift in the relative numbers of the different cell types with age: the percentage of primary/secondary spermatocytes declined as round and then elongated spermatids became more numerous.
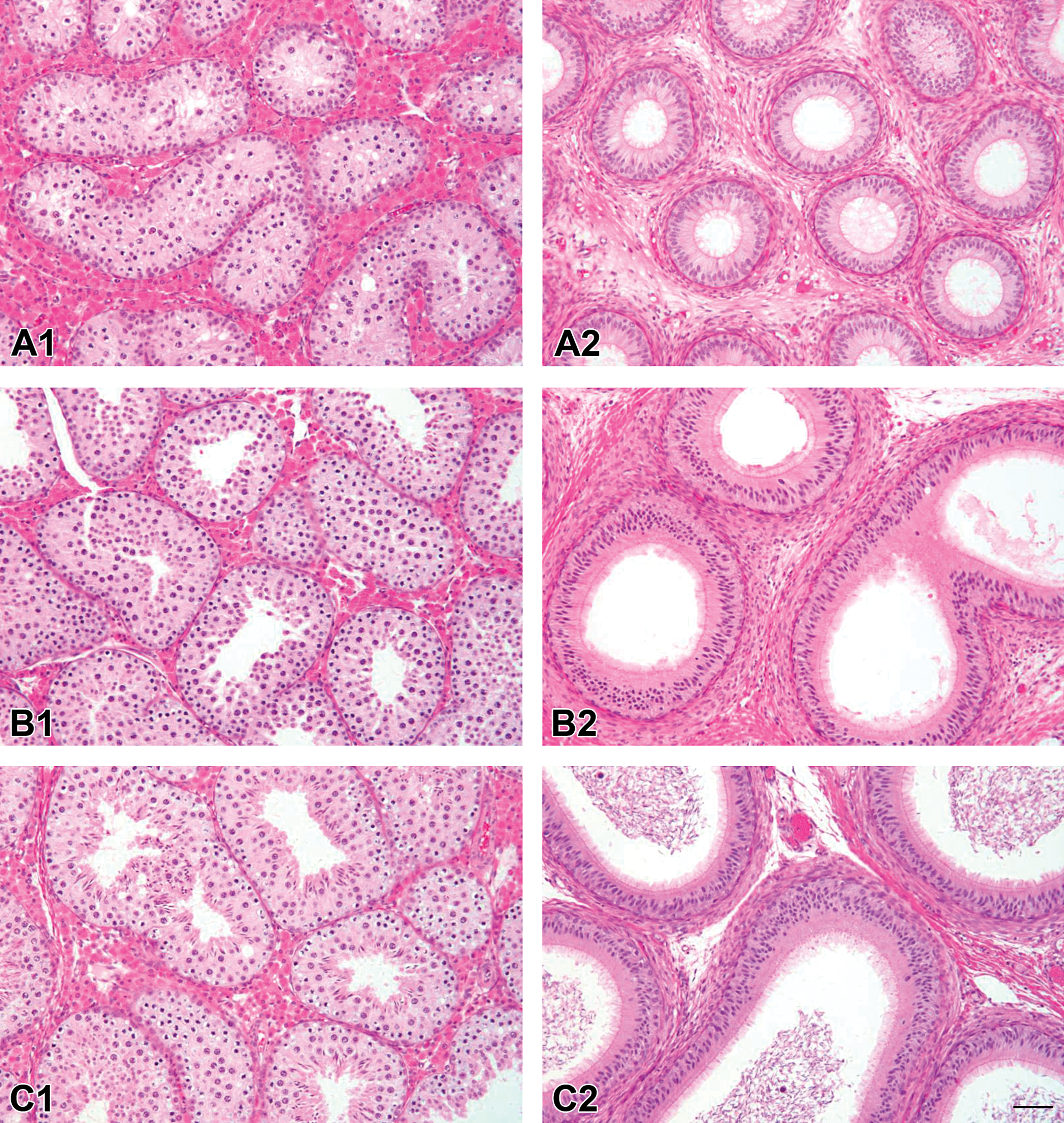
Representative histological images of testis and epididymal tissue from pre-, peri-, and postpubertal minipigs (
Discussion
There is no single parameter that can be used to define whether an animal is mature, in the sense of being able to mate, producing sufficient viable gametes and being able to maintain pregnancy and give birth; instead, a spectrum of parameters must be assessed. The various parameters do not all transition between immaturity and maturity in exact parallel. Even considering only 1 parameter, an animal transitions gradually between immaturity and maturity. The assessment of some parameters is subjective, so different observers may reach different conclusions about whether maturity has been reached, even if they are assessing the same parameter. There is also considerable interanimal variation (some intrinsic and some due to environmental, nutritional, or other external differences) in when maturation occurs. Each of the studies reviewed here used relatively few animals. For all of these reasons, it is not surprising that different experiments on the age of maturity can yield different results. More work is required in both sexes to confirm the findings; to examine the importance of environmental, nutritional, and genetic factors; and to correlate behavioral, hormonal, anatomic, and physiological changes leading to the acquisition of maturity. Studies on other breeds of minipig (female Yucatan, micro-Yucatan, Hanford, or Sinclair breeds) would also be valuable.
The later onset of maturity in the female minipigs evaluated postmortem compared with those in which progesterone was evaluated may have been due to the absence of boars in the same rooms in the former cohort. Unpublished data in Göttingen minipigs supplied by Ellegaard, Denmark, have shown successful mating rates (over 80%) for female minipigs at the ages of 6 months, and minipigs at an average age of 5.6 months have been successfully mated and given birth without complications. These ages are lower than would be expected from the anatomic results of Tortereau, Howroyd, and Lorentsen (2013), in females that were not in the presence of a boar, but are more consistent with the hormonal results of Peter et al. (2016), in females that were housed in the same room as adult boars. These results should be confirmed and put into the context of environmental factors such as the age when a boar was introduced, and equivalent data on the breeding performance of young male minipigs should be obtained. In female minipigs, external or behavioral signs of heat, cytology of vaginal smears, rectal body temperature, or release of 17β-estradiol are not useful indicators of the acquisition of sexual maturity (Peter et al. 2016).
The age of acquisition of mature histological features in the male differed between 2 months for Göttingen minipigs supplied by Marshall BioResources and 4.4 months for Yucatan minipigs supplied by Sinclair BioResources. This marked difference, which is very important in terms of selection of animals for studies and interpretation of experimental results, may be linked with how maturity was defined in the 2 studies but alternatively could be due to intrinsic interstrain differences or to husbandry differences. The authors of this review suggest that it may be useful for a panel of pathologists to reexamine the sections reported by Evans et al. (2012) and Taberner et al. (2015) simultaneously in order to ensure that consistent histological criteria have been applied. It would also be useful in future work to correlate the histological appearances of the maturing testis and epididymides with those of the prostate and seminal vesicles.
In summary, in female Göttingen minipigs, the first progesterone peak together with either an active corpus luteum or recently ovulated follicle, indicative of sexual maturity, were seen at 3.7 to 4.2 or 6.1 to 6.5 months of age. These animals were in the presence of a boar. In female Göttingen minipigs in toxicology studies, which were not in the presence of a boar, at least 1 corpus luteum in the ovaries was present in only 50% of the females by 6.5 months of age, while all were mature by 7.7 months of age. Histological maturity in the male Yucatan minipig is reported to be attained at about 4.4 months old but in male Göttingen minipigs at about 2 months old.
Footnotes
Author Contributions
Authors contributed to conception or design (PH, ED, BP), data acquisition, analysis, or interpretation (PH, ED, BP); drafting the manuscript (PH); and critically revising the manuscript (ED, BP). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
