Abstract
Nitinol stents are widely used for the treatment of peripheral arterial diseases in lower extremity arteries and have shown different clinical outcomes depending on implanted arterial segments. We aimed to compare histopathological responses to nitinol stents in femoral artery (FA) with those in femoropopliteal artery (FPA), which is markedly bended during knee flexion. A single nitinol stent was implanted in FA and FPA of 21 domestic swine. The stented vessels were angiographically assessed and then harvested for histopathology at 1 and 3 months after implantation. Angiographic late lumen loss was significantly greater in FPA than in FA at 3 months. Neointimal area decreased in FA and increased in FPA from 1 to 3 months. Compared with FA, peri-strut area of FPA showed more pronounced hemorrhage and fibrin deposition at 1 month and angiogenesis and inflammation at 1 and 3 months. Injury to internal elastic lamina or media was minimal in both FA and FPA at both time points. In conclusion, vascular responses to nitinol stents were different between FA and FPA with respect to time course of neointimal formation and progress of healing, suggesting that repetitive interaction between stent and vessel wall during dynamic vessel motion affected vascular responses.
Introduction
Nitinol (nickel titanium) self-expanding stents are widely implanted in lower extremity arteries for the treatment of peripheral arterial diseases in clinical settings, since nitinol has excellent biocompatibility and shape memory effects, which is required to resist arterial deformation by posture changes (Maleckis et al. 2018). Clinical outcome after nitinol stent implantation varies depending on implanted arterial segments (Aboyans et al. 2018; Gerhard-Herman et al. 2017; Malyar, Reinecke, and Freisinger 2015). In particular, long-term vessel patency rate after stent implantation is usually low in an arterial segment including popliteal arteries, due to repetitive knee flexion stress caused, at least partially, by the mechanical stress on arterial segments owing to posture changes (Chang et al. 2011; Kroger, Santosa, and Goyen 2004; Rastan et al. 2015). However, it has not been investigated by histopathology if clinical differences between arterial segments implanted with nitinol stents show meaningful differences in vascular response after stenting. Moreover, in previously reported animal studies, implantation of nitinol stents has been limited to iliofemoral and femoral arteries (FAs) (Hou et al. 2016; Radeleff et al. 2009; Saguner et al. 2012; Torii et al. 2018; Zhao et al. 2009), and histopathological analysis of implanted femoropopliteal arteries (FPAs) is lacking despite clinical use of nitinol stents in FPA.
To develop novel nitinol stents improving clinical outcomes, better understanding is needed, namely, with respect to differences in vascular responses among anatomically distinct arterial regions. The aim of this study was to investigate histopathological differences in the chronic vascular response to nitinol stents implanted in FA versus FPA. Considering that the pig is the most common animal model for stents in lower extremity arteries due to anatomical similarity to human (Hou et al. 2016; Radeleff et al. 2009; Torii et al. 2018; Zhao et al. 2009), we assessed vascular responses 1 and 3 months after implantation of bare nitinol stents of the same design in porcine FA and FPA.
Materials and Methods
Animal Experimental Procedures
To compare vascular responses in FA and FPA, the data of 21 domestic swine from 3 studies in 2 test facilities (Terumo Corporation, Japan; Shin Nippon Biomedical Laboratories, Ltd., Japan) were retrospectively analyzed (Table 1). A total of 21 domestic swine (crossbred of Landrace and Yorkshire, 3–5 months old) were obtained from San-Esu Breeding (Chiba, Japan). All animals were orally administered with aspirin (81 mg) and clopidogrel (75 mg) daily from a minimum 3 days prior to the procedure through to the day of euthanasia. After sedation with intramuscular medetomidine (0.04–0.06 mg/kg) and midazolam (0.2–0.3 mg/kg), the animals were intubated and connected to mechanical ventilation. Anesthesia was maintained with 2–4% sevoflurane by inhalation throughout the procedure. Anticoagulation during catheterization was achieved with intravenous heparin (300 units/kg) to maintain the activated clotting time at ≥250 sec, as typically performed in clinical practice. At stent implantation, the guiding sheath was advanced over the 0.035 inch (i.e., 0.9 mm in diameter) guidewire to the target arteries. A single nitinol stent was implanted in the target arterial segment per animal (Figure 1). The stent design and diameter (6 mm) were the same, but the stent length was different (40 or 60 mm) among the original studies (Table 1). The 6-mm-diameter stent was indicated for arteries with diameter of 4 to 5 mm. Although FPA is distal to FA and is smaller than FA, we achieved equivalent baseline lumen diameter (4–5 mm) in both FA and FPA through selection of animal size. Animals of different body weight range were used between FA and FPA (Table 1). Angiographic images were acquired at baseline (i.e., before stenting on 0 day), immediately after stenting, and at termination (i.e., the day of euthanasia) by C-arm angiography machines with Infinix Celeve-i INFX-8000V (Toshiba Medical Systems, Tochigi, Japan). After stent implantation, the animals were recovered and then returned to routine care. At 1 month (27–28 days) or 3 months (84–90 days) after stent implantation, all animals received follow-up angiography and were euthanized. Animal preparation at euthanasia was performed in the same fashion as that at stent implantation. All procedures involving animals were approved by the Institutional Animal Care and Use Committees, where the studies were conducted.
Methods Difference in Each Arterial Segment and Time Point.
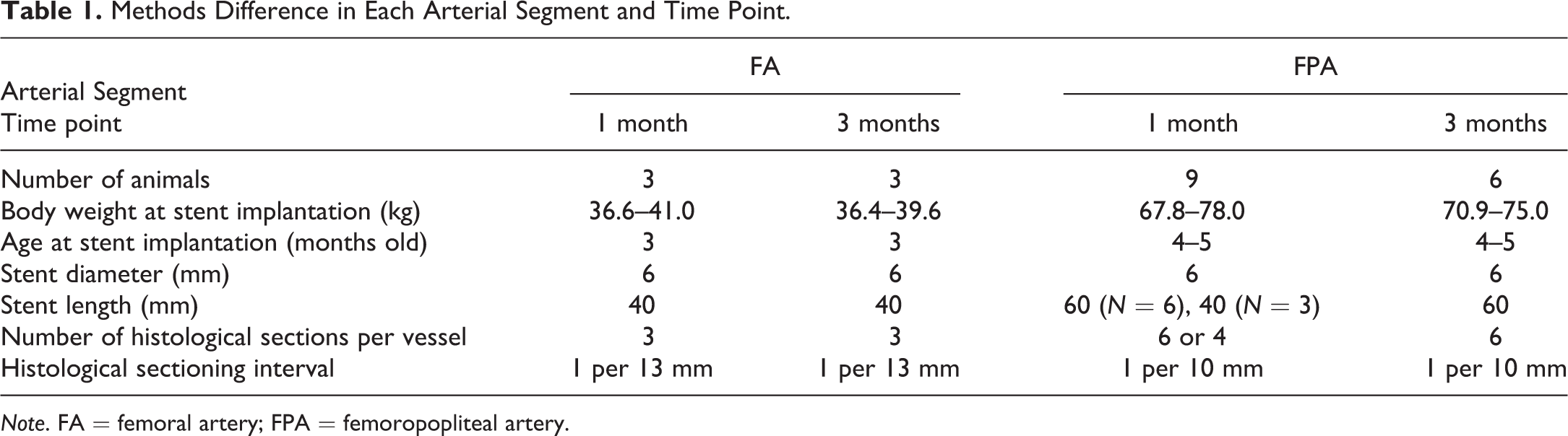
Note. FA = femoral artery; FPA = femoropopliteal artery.
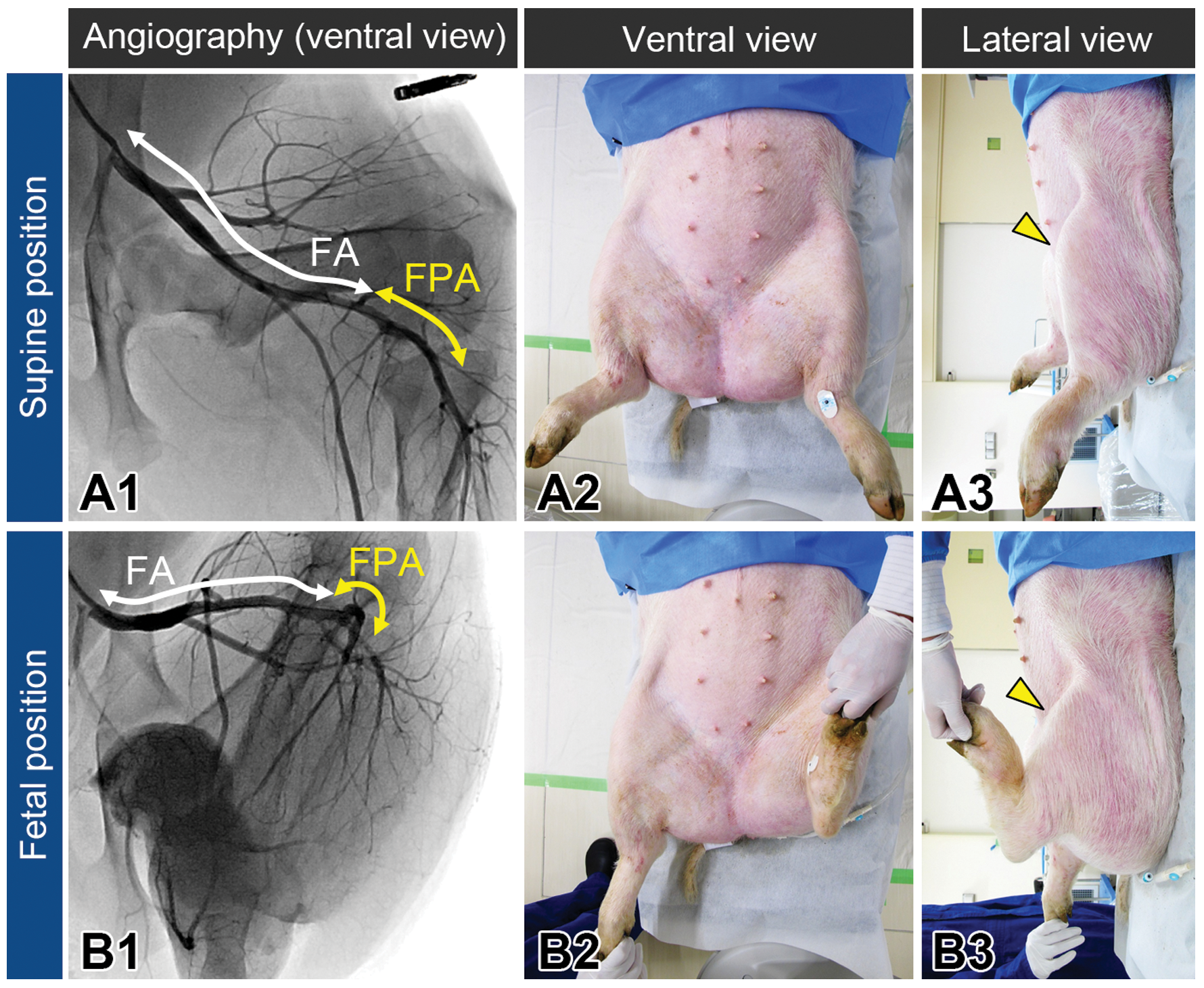
Angiographic images of lower extremity arteries and the corresponding appearance of animal postures (ventral and lateral views) in supine (A) and fetal (B) positions. Knee positions are indicated by yellow arrowheads (A3, B3). FA = femoral artery; FPA = femoropopliteal artery.
Quantitative Vascular Analysis by Angiography
The lumen diameters of stented arteries were measured with a CAAS 2000 (Pie Medical Imaging, Maastricht, the Netherlands)-based quantitative vascular analysis software using an automated edge detection system at prestenting (i.e., baseline) and terminally (i.e., the day of euthanasia). Nominal stent-to-artery ratio was calculated as labeled stent diameter (i.e., 6 mm)/baseline lumen diameter (Zhao et al. 2009). Late lumen loss was calculated as baseline lumen diameter—terminal lumen diameter.
Tissue Harvest and Histological Processing
After euthanasia, the lower limbs were perfusion-fixed at 100 mmHg with 10% neutral-buffered formalin, followed by harvesting the stented vessels. The stented vessels were embedded in Spurr resin, affixed to plastic slides, cut by diamond blade saw, and ground and micropolished to optical finish with Exakt Cutting Grinding System (Exakt Apparatebau, Norderstedt, Germany). The sections were obtained at equally spaced 3 to 6 levels per vessel (1 section per 10–13 mm in length) from proximal to distal (Table 1) and stained with hematoxylin and eosin and elastin trichrome (Masson’s trichrome with Van Gieson modification for elastin staining).
Quantitative Histomorphometric Analysis and Histopathology
Quantitative histomorphometric analysis was performed on the elastin trichrome–stained sections from each stented artery. For each histological section, parameters of lumen area, internal elastic lamina (IEL, Figure 2) bounded area, and external elastic lamina (EEL, Figure 2) bounded area were directly measured using standard light microscopy and computer-assisted image measurement systems (Image-Pro Plus, Media Cybernetics, Silver Springs, MD). From the above parameters, the following parameters were calculated as follows: neointimal area as IEL-bounded area − lumen area, medial area as EEL-bounded area − IEL-bounded area, and percent area stenosis as neointimal area/IEL-bounded area × 100. Vessel wall injury was scored in strut-by-strut manner as follows: 0 = IEL intact, 1 = IEL lacerated, 2 = media lacerated, and 3 = EEL lacerated (Schwartz et al. 1992). The average vessel wall injury score for each section was calculated by dividing the sum of scores by the total number of struts. Other histopathological changes were scored in section-by-section manner as follows: 0 = not present or normal, 1 = minimal or focal, 2 = mild or multifocal, 3 = moderate or regionally diffuse, and 4 = severe or markedly diffuse. The histomorphometric and histopathological values for each vessel were calculated by dividing the sum of values by the total number of sections.
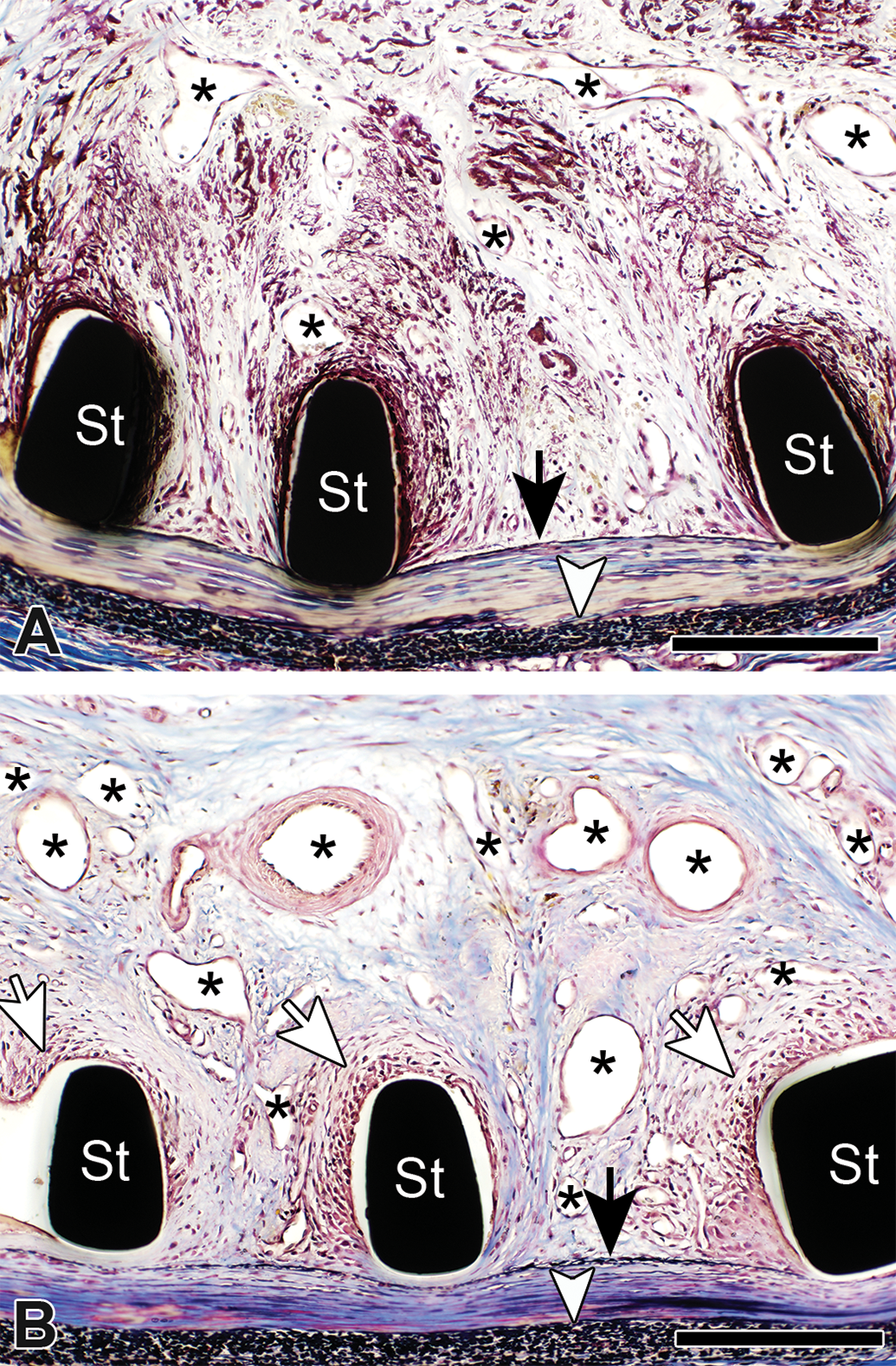
Representative histopathological images of femoropopliteal arteries (FPAs) at 1 (A) and 3 months (B). Despite intact internal elastic lamina (black arrow), peri-strut area showed marked hemorrhage at 1 month and remarkable angiogenesis (asterisks) with moderate inflammation surrounding stent struts (white arrows) at 3 months in FPA. Elastin trichrome staining. Scale bars represent 200 μm. St = stent strut; white arrowhead = external elastic lamina.
Statistical Analysis
Data are presented as mean ± standard deviation. Continuous data (quantitative vessel analysis and histomorphometric data) were compared by Student’s t-test following confirmation of the homogeneity of variance tested using the F-test (p > .05). Ordinal data (histopathological scores) were compared by Wilcoxon rank-sum tests. All statistical analyses were performed with GraphPad Prism 7 (GraphPad Software, San Diego, CA). A p value < .05 was considered to indicate statistical significance.
Results
All stents were successfully implanted in the targeted arterial segments. Although baseline lumen diameter was slightly larger and nominal stent-to-artery ratio was slightly lower in FA than those in FPA, these differences were not statistically significant, indicating equivalent stent implantation settings at 0 day (Table 2).
Quantitative Vessel Analysis of Angiography.
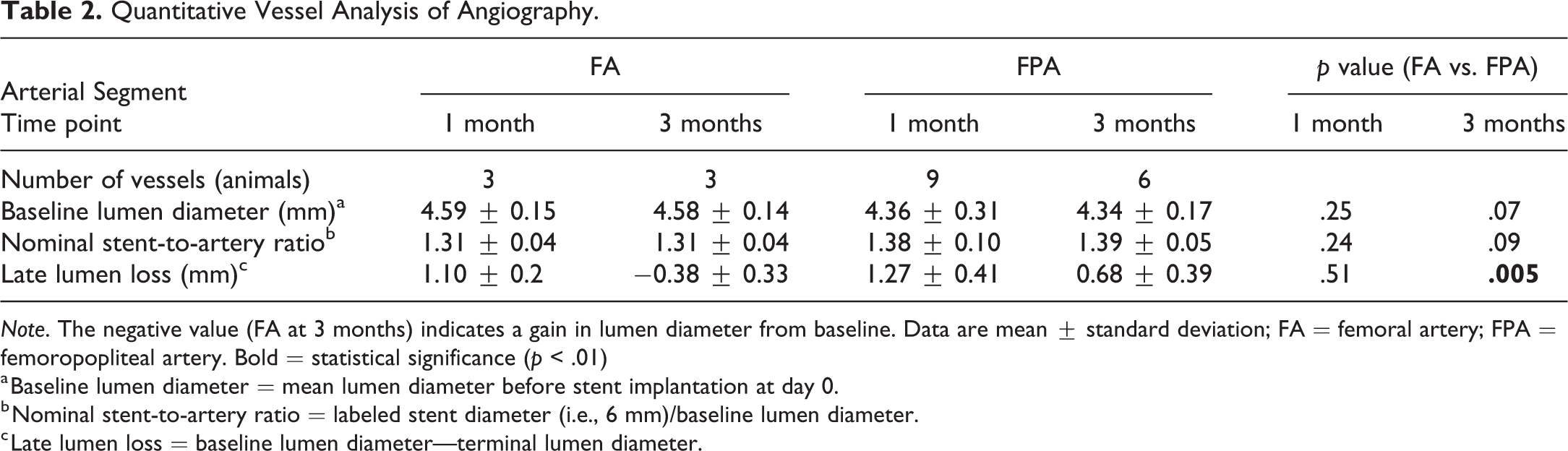
Note. The negative value (FA at 3 months) indicates a gain in lumen diameter from baseline. Data are mean ± standard deviation; FA = femoral artery; FPA = femoropopliteal artery. Bold = statistical significance (p < .01)
a Baseline lumen diameter = mean lumen diameter before stent implantation at day 0.
b Nominal stent-to-artery ratio = labeled stent diameter (i.e., 6 mm)/baseline lumen diameter.
c Late lumen loss = baseline lumen diameter—terminal lumen diameter.
All animals survived until scheduled necropsy without any abnormalities caused by the implanted stents, as determined by daily clinical observation. Terminal angiography indicated all the stented vessels were patent, but there was significantly larger late lumen loss at 3 months in FPA (Figure 3, Table 2). Negative value of late lumen loss in FA at 3 months (−0.38 mm) meant stent expansion after stenting canceled lumen loss by neointimal formation.
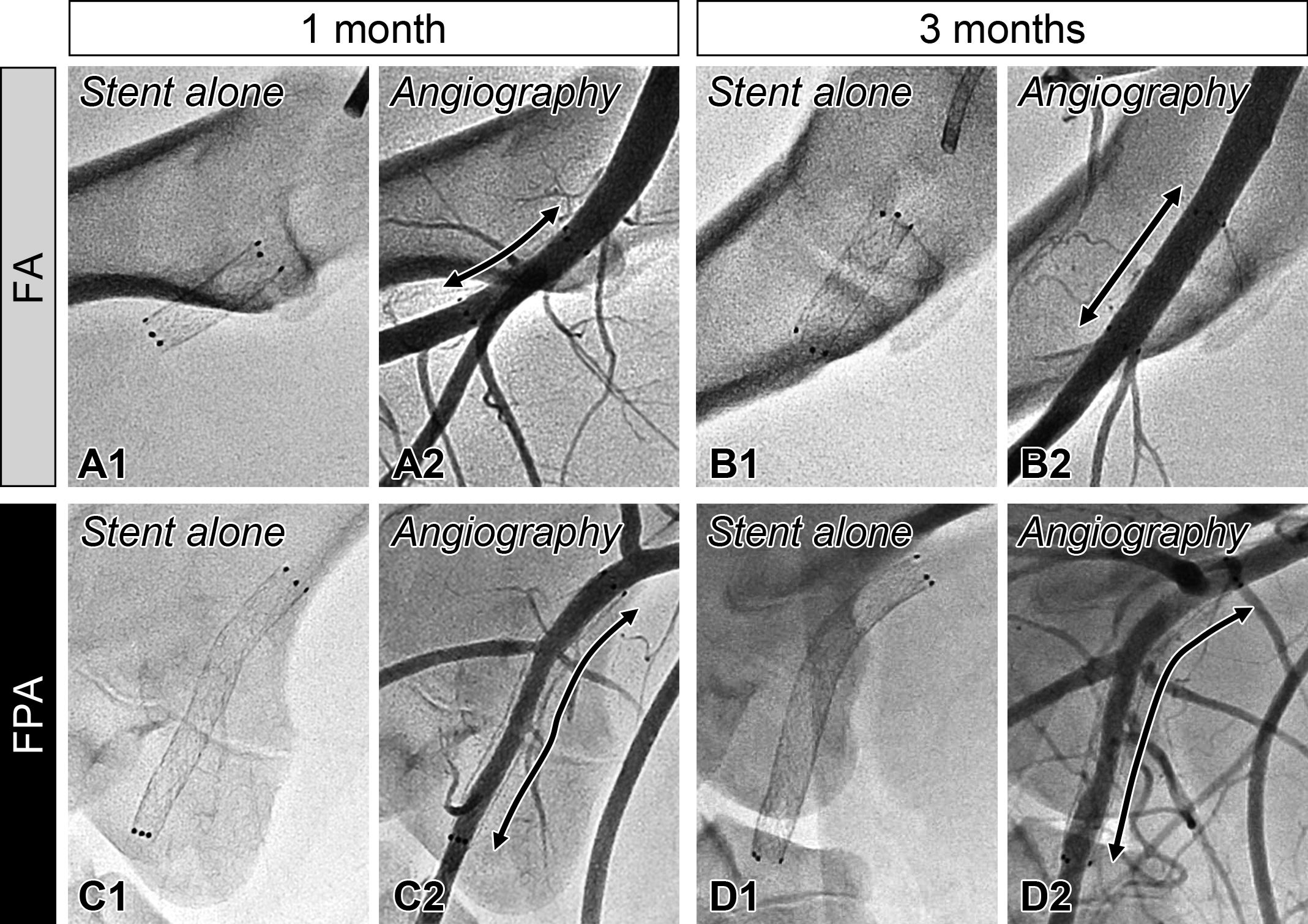
Representative angiographic images of stented femoral (FA) and femoropopliteal arteries (FPAs) at 1 and 3 months. In each arterial segment at each time point, left panels are fluoroscopic images of stented arteries (A1, B1, C1, and D1). Right panels are angiographic images with injected contrast media (i.e., blood flow) in the corresponding stented arteries, which are indicated by double-headed arrows. No significant gap was observed between stent outer line and contrast media in FA at 1 (A2) and 3 months (B2), whereas gaps were apparently observed in FPA at both 1 (C2) and 3 months (D2), indicating existence of tissues between stent struts and arterial lumen in FPA. Note that angiographic images are projections of three-dimensional vessel structures onto a two-dimensional plane, resulting in shorter appearance of stent especially in FA than actual length (A1 and B1).
Histopathological and histomorphometric analyses demonstrated the following morphological difference between FA and FPA (Figures 4 and 5). At 1 month, distribution of neointima in FPA appeared eccentric, whereas that in FA, neointima was generally concentric, despite equivalent neointimal area between FPA and FA (7.9 mm2 vs. 7.4 mm2, p = .64, Figures 4A, C, and 5A). At 3 months, neointima in FPA also appeared eccentric (Figure 4D), and neointimal area was significantly larger in FPA than in FA (12.3 mm2 vs. 4.9 mm2, p = .003, Figure 5A).
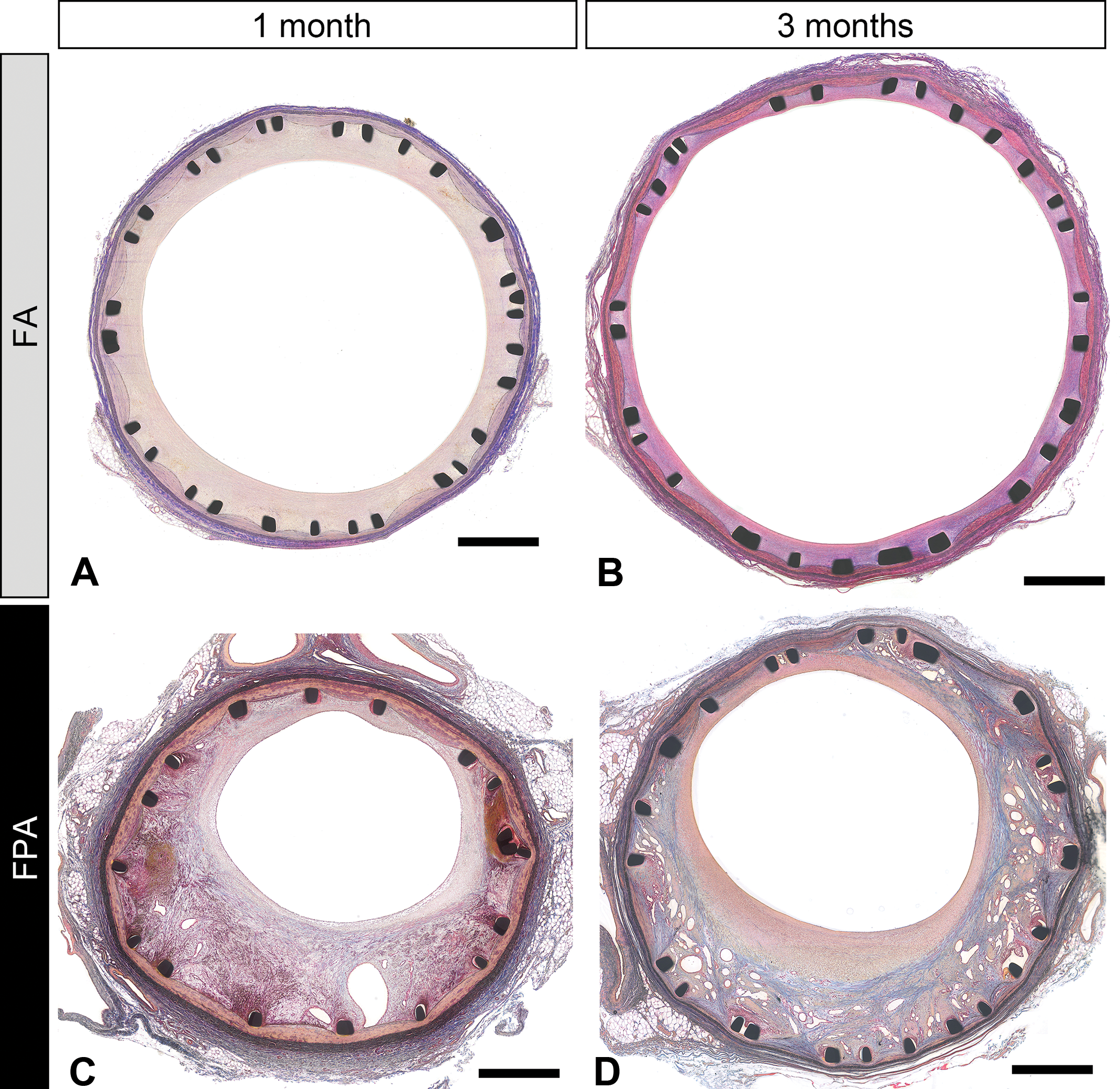
Representative histopathological cross-sectional images with stents in femoral (FA; panels A and B) and femoropopliteal arteries (FPAs; panels C and D) at 1 and 3 months. Black squares compressed onto media are stent struts. In FA, stent struts were covered with matured neointima composed of predominantly smooth muscle cells at 1 month (A) and the neointima regressed at 3 months (B). In FPA at 1 month, whereas surface neointima facing arterial lumen appeared matured like FA, hemorrhage and fibrin deposition was marked in peri-strut area. In FPA at 3 months, neointima appeared two layers; inner neointima facing arterial lumen was matured, while outer neointima accompanied remarkable peri-strut angiogenesis. Elastin trichrome staining. Scale bars represent 1 mm.
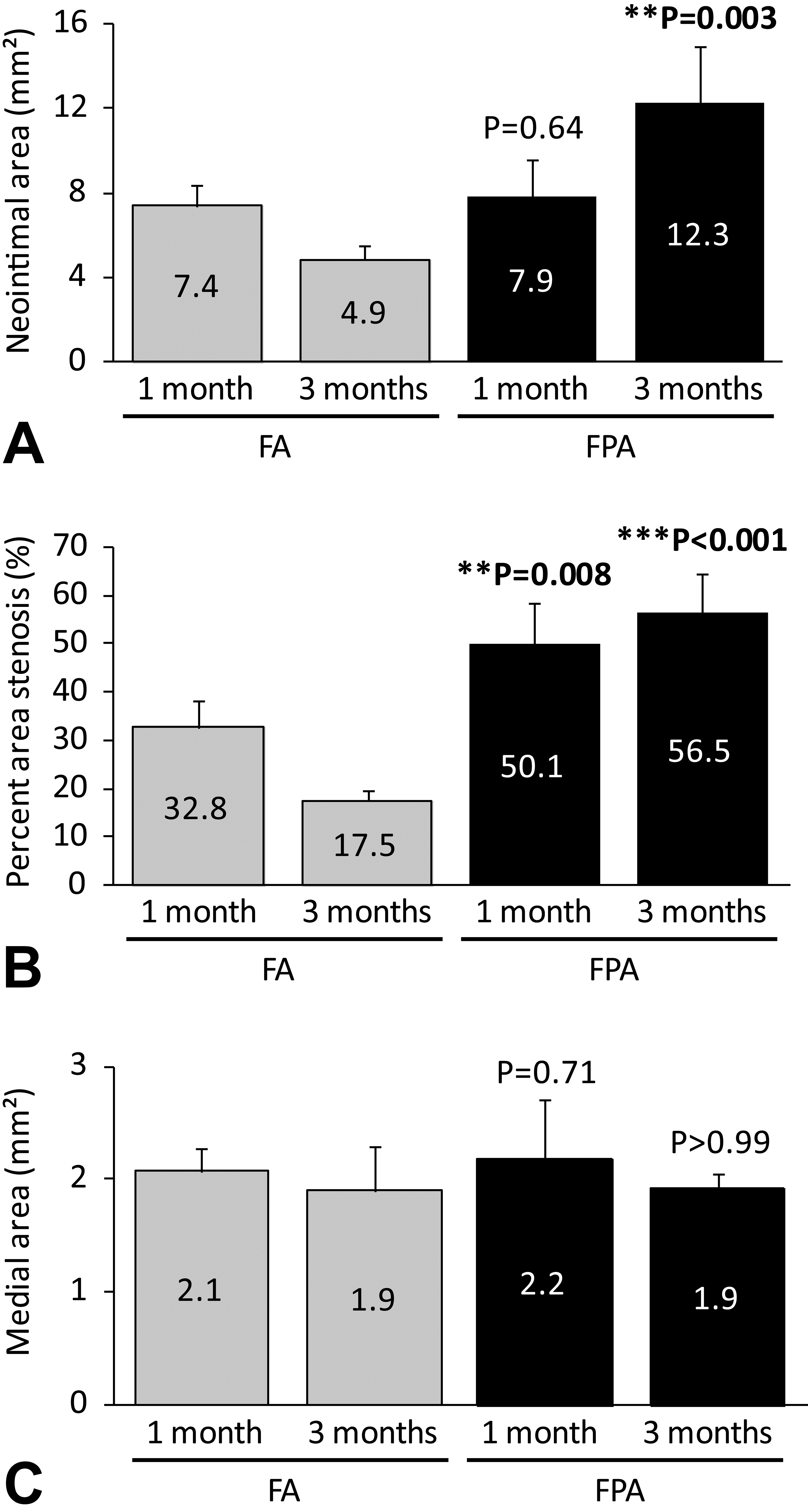
Neointimal area (A), percent area stenosis (B), and medial area (C) in femoral (FA) and femoropopliteal arteries (FPAs) at 1 and 3 months. Neointimal area in FPA was significantly greater than that in FA at 3 months, while no significant differences were found between FA and FPA at 1 month. Percent area stenosis in FPA was significantly greater than that in FA at both 1 and 3 months. Medial areas were equivalent between FA and FPA at both 1 and 3 months. Data are mean ± standard deviation.
Distinct histopathological changes were found in peri-strut area between arterial locations. Compared with FA, peri-strut area of FPA showed more pronounced hemorrhage and fibrin deposition at 1 month (Figures 6A, B and 7A, C1, and C2). Peri-strut area of FPA also showed hemosiderin deposition, inflammation, and more remarkable angiogenesis at 1 and 3 months (Figures 6C, D and 7). Vessel wall injury was overall minimal in all groups, and there were statistically significant differences between FA and FPA (0.00 ± 0.00 vs. 0.18 ± 0.16 at 1 month, 0.07 ± 0.08 vs. 0.48 ± 0.17 at 3 months, respectively, Figure 8). Equivalent medial area between FA and FPA also supported that outward injury (notable compression or laceration of media) by stent struts was minimal (Figure 5C). As indicated in Figure 2, marked hemorrhage and remarkable angiogenesis were observed in the peri-strut area at 1 and 3 months, respectively, with neointimal thickening, but the local IEL was almost intact (i.e., minimal vessel wall injury).
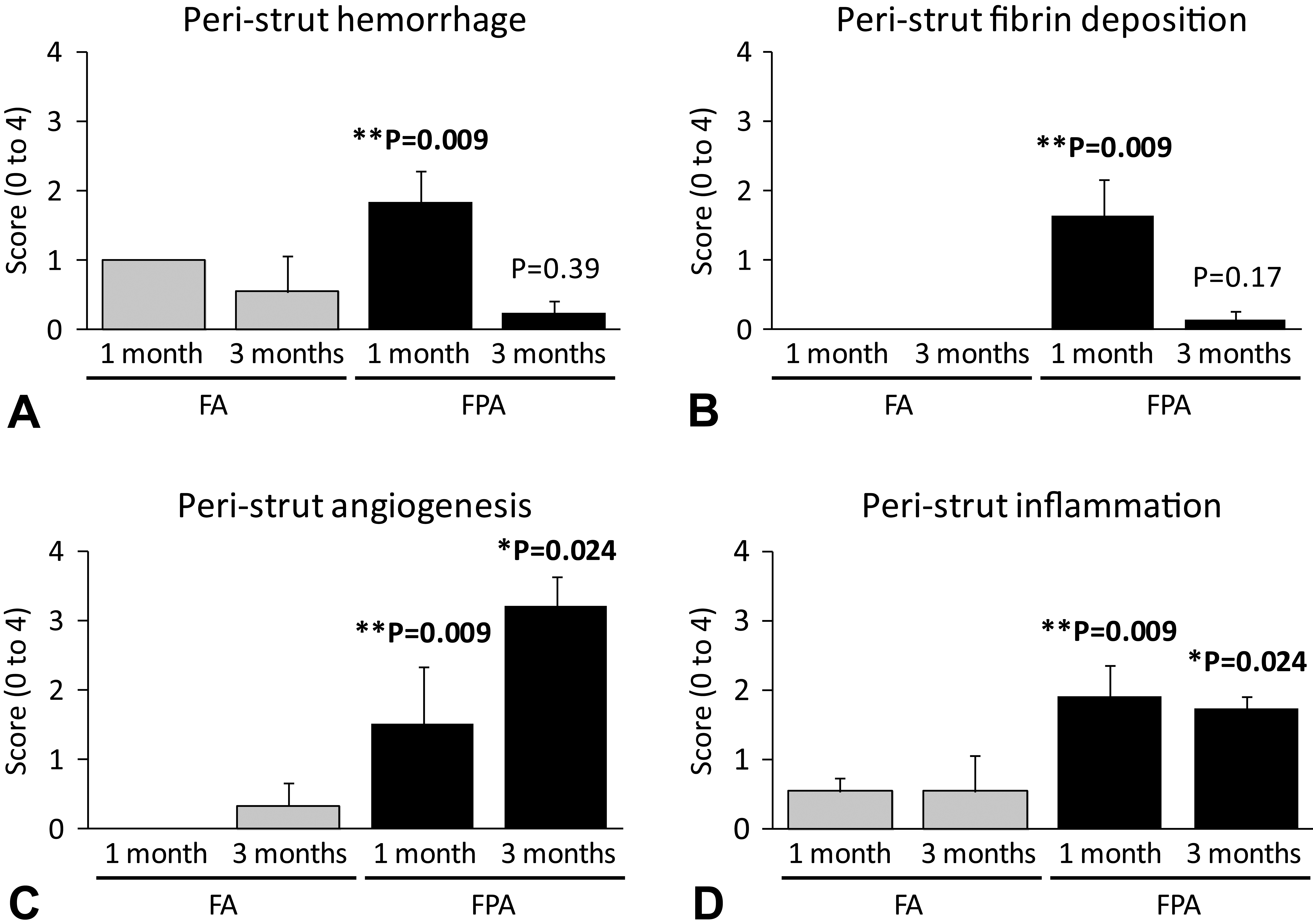
Semiquantitative assessment of peri-strut hemorrhage (A), fibrin deposition (B), angiogenesis (C), and inflammation (D) in femoral (FA) and femoropopliteal arteries (FPAs) at 1 and 3 months. Compared with FA, peri-strut area of FPA showed more pronounced hemorrhage and fibrin deposition at 1 month and angiogenesis and inflammation at 1 and 3 months. Data are mean ± standard deviation.
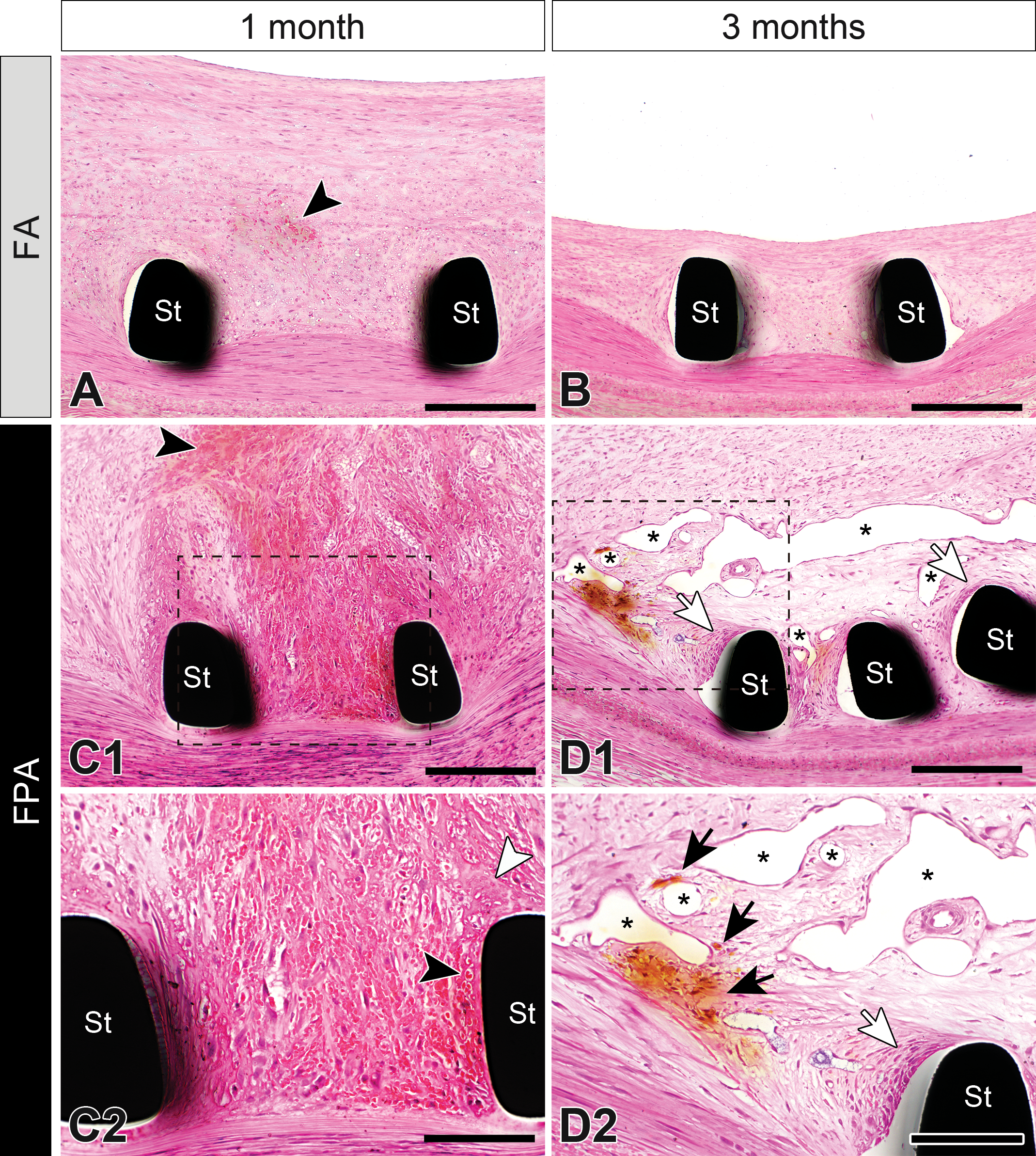
Representative histopathological images of peri-strut areas in femoral (FA) and femoropopliteal arteries (FPAs) at 1 and 3 months. Panels C2 and D2 are magnified images of dotted-boxed area in panels C1 and D1, respectively. In FA at 1 month, neointima was composed of smooth muscle cells and extracellular matrix with slight hemorrhage (black arrowhead, A). In FA at 3 months, neointima was apparently regressed without hemorrhage or other changes (B). In FPA at 1 month, neointima was characterized by marked peri-strut hemorrhage (black arrowhead) and fibrin deposition (white arrowhead; C2). In FPA at 3 months, angiogenesis (asterisks) was remarkable in peri-strut area (D1 and D2) with occasional hemosiderin deposition (black arrows) and inflammation (white arrow). Hematoxylin and eosin staining. Scale bars represent 200 μm (A, B, C1, and D1) and 100 μm (C2 and D2). St = stent strut.
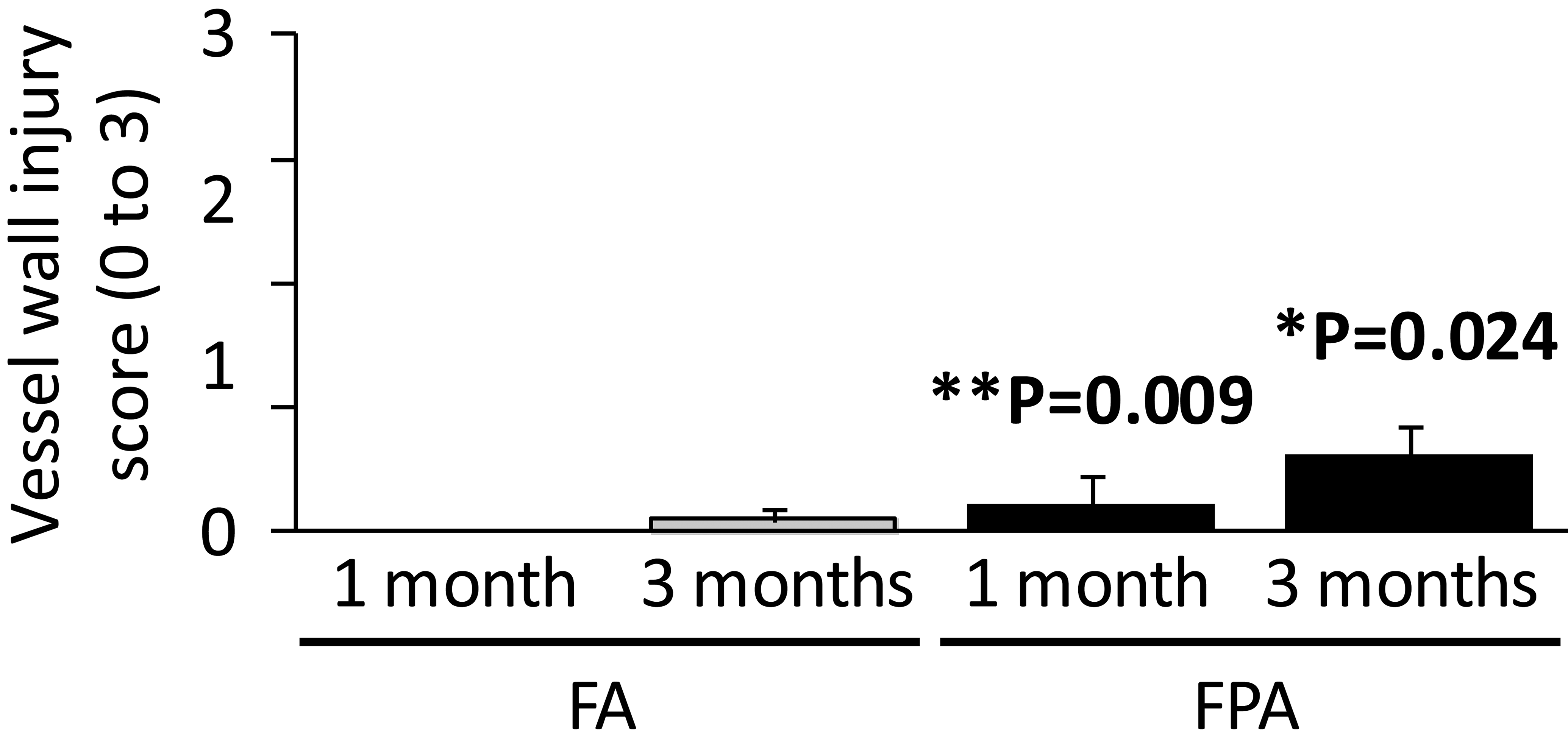
Semiquantitative assessment of vessel wall injury in femoral (FA) and femoropopliteal arteries (FPA) at 1 and 3 months. Vessel wall injury was scored in strut-by-strut manner as follows: 0 = internal elastic lamina (IEL) intact, 1 = IEL lacerated, 2 = media lacerated, 3 = external elastic lamina lacerated.
Discussion
By using a porcine model including a popliteal segment, the FPA, subjected to repetitive stress during knee flexion, we demonstrated in the current study that chronic vascular responses to the FA and the FPA were different. Neointima formation regressed from 1 to 3 months in FA, whereas it progressed in FPA, resulting in significantly larger neointimal area in FPA at 3 months. The neointima including interstrut and peri-strut areas was mainly composed of smooth muscle cells in FA at both 1 and 3 months. By contrast, the peri-strut area in FPA was characterized by more pronounced hemorrhage, fibrin deposition, angiogenesis, and inflammation at 1 month and showed more pronounced angiogenesis and persistent inflammation at 3 months.
Vascular responses to stents have long been investigated in porcine coronary arteries, although the number of citations addressing vascular responses in lower extremity arteries is limited. One of the critical predictors of neointimal thickening is vessel wall injury caused by the stent. Vessel wall injury was graded histopathologically based on the depth of mural penetration; laceration reaching IEL, media, and adventitia was scored as 1, 2, and 3, respectively. These criteria have been widely used in the stent literature. Greater vessel wall injury, particularly score 2 to 3, has been reported to yield thicker neointima formation in porcine coronary arteries (Kornowski et al. 1998; Schwartz et al. 1992). Similarly, high oversizing (nominal stent-to-artery ratio: >1.4) of nitinol stents induced larger neointimal area in conjunction with greater vessel wall injury (score > 1) in porcine iliofemoral arteries (Saguner et al. 2012; Zhao et al. 2009). In the current study, we made efforts to avoid high oversizing implantation, and mean nominal stent to artery ratios were actually less than 1.4 in both FA and FPA (Table 2), resulting in minimal vessel wall injury (score < 0.5 on average) in both FA and FPA. Therefore, neointimal thickening in FPA in the current study was not predominantly caused by vessel wall injury, an established predictor of neointimal thickness.
Peri-strut inflammation has also been shown to play an accelerating role in neointimal thickening (Kornowski et al. 1998). Neointima thickness correlated with the presence of greater inflammation in porcine coronary arteries. Additionally, greater inflammation was reported to be induced by deeper arterial laceration (i.e., greater vessel wall injury score). Lastly, neointimal thickening was observed to accompany inflammation even in the absence of vessel wall injury. Kornowski et al. proposed that peri-strut inflammation was induced independently by vessel wall injury and by foreign body reaction to stents, concluding that both factors lead to neointimal thickening. In the current study, vessel wall injury was minimal as described above, but greater peri-strut inflammation was found in FPA than FA at both 1 and 3 months. Because the material of the implanted stent was identical in FA and FPA, it is unlikely that the inflammation was augmented in FPA as a foreign body reaction to the stent or indirectly via injury. This suggested another mechanism of inflammation and neointimal thickening specific to FPA, independent from mural laceration or foreign body response to the stent.
Chronological histopathological study documented healing of peri-strut area after bare metal stent implantation in porcine coronary arteries as follows (Virmani et al. 2003). At 1 to 3 days, the peri-strut area predominantly consisted of platelet/fibrin deposition and scattered neutrophils within adherent luminal thrombi. By 7 days, mural thrombi extending between stent struts contained smooth muscle cells and inflammatory cells. By 28 days, the neointima contained a larger number of smooth muscle cells and extracellular matrix, and fibrin was usually absent. This 28-day histopathological profile in coronary arteries was similar to the 1-month observations in FA in the current study. In addition, neointimal area peaked at 28 days and regressed at 3 months and later time points in coronary arteries (Virmani et al. 2003), which was in line with the time course of neointimal formation in FA in the current study. FPA showed a different histopathological profile and a different time course. Hemorrhage, fibrin, and angiogenesis were specific to peri-strut areas in FPA at 1 month. The unique findings in FPA suggested leakage of erythrocytes and plasma from abundant immature vessels in peri-strut area. It is plausible that immature vessels were injured and disrupted by repetitive interaction between stent struts and early neointima, which could be caused by notable bending stress during knee flexion. The repetitive interaction in FPA might have resulted in the enlargement of neointimal area from 1 to 3 months and also caused persistent inflammation at both 1 and 3 months. This mechanism may explain poor clinical outcome of nitinol stents in popliteal arterial segments. A clinical study supported the hypothesis that inflammatory responses to stents are different depending on the anatomical location; stent implantation in FPA was associated with a more extensive inflammatory response (i.e., higher serum C-reactive protein, serum amyloid A, and fibrinogen levels) than that in iliac and carotid arteries, independent of lesion morphology and interventional factors (Schillinger et al. 2002).
In conclusion, the current study demonstrated that vascular responses to the same-design bare nitinol stents were different between FA and FPA. Histopathological changes and time course of neointimal morphology in FA were similar to those previously described in porcine coronary arteries. On the other hand, FPA in the current study showed clearly different histopathological features that included continued enlargement of neointimal area from 1 to 3 months, greater hemorrhage and fibrin deposition at 1 month, and angiogenesis and inflammation at 1 and 3 months in peri-strut area. These FPA-specific changes were suggested to be caused by repetitive interaction between stent struts and early neointima, which was caused by notable bending stress during knee flexion. These results suggest that for the development of novel nitinol stents, it may be worth considering implanting in high biomechanical stress porcine FPA as a model for high-risk (but clinically relevant) preclinical assessment.
Footnotes
Acknowledgments
The authors would like to thank Mr. Shinichiro Kira and Dr. Taizo Iwasaki of Terumo Corporation for catheterization procedures, and Mr. Shinji Utsunomiya and Mr. Koichiro Nojiri of Shin Nippon Biomedical Laboratories, Ltd., for conducting in-life phase of the animal studies.
Author Contributions
Authors contributed to conception or design (AS); data acquisition, analysis, or interpretation (AS, JS, SR, TM, AT, HH, KN, MT); drafting the manuscript (AS); and critically revising the manuscript (JS, SR, TM, AT, HH, KN, MT). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded in part by Strategic Promotion of Innovative Research and Development (S-innovation) from Japan Science and Technology Agency, by Japan Agency for Medical Research and Development (grant number 15he1302048g0001), and by Terumo Corporation, Tokyo, Japan.
