Abstract
Though rare due to measures and practices to control the risk, infections can occur in research and toxicology studies, especially in nonhuman primates (NHPs) exposed to xenobiotics, particularly immunomodulatory drugs. With such xenobiotics, immunocompromised or immunosuppressed animals will not be able to mount a protective response to infection by an opportunistic pathogen (bacteria, virus, parasite, or fungus) that might otherwise be nonpathogenic and remain clinically asymptomatic in immunocompetent animals. The respiratory tract is one of the most commonly affected systems in clinic, but also in toxicology studies. Pulmonary inflammation will be the main finding associated with opportunistic infections and may cause overt clinical disease with even early sacrifice or death, and may compromise or complicate the pathology evaluation. It is important to properly differentiate the various features of infection, to be aware of the range of possible opportunistic pathogens and how they may impact the interpretation of pathology findings. This review will present the most common bacterial, viral, parasitic, and fungal infections observed in the respiratory tract in NHPs during research and/or toxicology studies.
Keywords
Introduction
Modulation of the immune system by low molecular weight (LMW) drugs or by biologics drugs (eg, recombinant protein, monoclonal antibody [mAb], and derived antibody like bispecific, fragment antigen binding [Fab], single chain fragment variable [scFv], or single-domain antibody) has been rapidly developing in the last 3 decades, due to the increasing delineation of the immune system and its players. 1 Chronic inflammatory, autoimmune, and allergic diseases, and transplant rejection, are diseases driven by a dysregulated immune system (eg, reactive to self-antigens [autoimmunity] or to isoantigen [alloimmunity]). 2,3 The aim of an effective therapeutic for these immune-mediated diseases is to suppress or modify the immune system to dampen the inflammation.
Compared to LMW drugs, toxicity of biologics is usually related to exaggerated pharmacology and is more predictable. 4 This is particularly true with immunomodulatory drugs. If LMW immunomodulatory drugs tend to be associated with pharmacology and nonpharmacology related toxicities (ie, hepatotoxicity or nephrotoxicity), biologic immunomodulatory drugs typically display toxicities primarily related to on-target exaggerated pharmacology (eg, immunosuppression or immune activation), including on-target binding in nonintended tissue/organs, but also in some cases, as a result of off-target binding or immunogenicity. 4,5 Since many chronic inflammatory diseases require prolonged treatment, immunosuppressive drugs are often linked to increased risk of infection and/or cancer in treated patients due to compromised immune function affecting innate (myeloid and monocyte lineage) and/or acquired (lymphoid lineage) immunity. Viral, bacterial, or fungal infections, which do not cause disease in immunocompetent healthy host (human or preclinical species) but become pathogenic in immunocompromised host, are defined as opportunistic infection. The opportunistic infection profiles will depend on the duration of treatment and which part of the immune system is targeted, that is, innate and/or acquired versus humoral and/or cell-mediated immunity; but also on the species used to assess the preclinical safety of the drug. 6
Rodents should show a lower risk of infection since they are generally in specific pathogen free (SPF) controlled areas (barrier maintenance), however SPF conditions are difficult to maintain. Most of the agents causally related to a disease are removed and constantly monitored via the use of sentinel animals. Spontaneous infections should rarely be encountered in rodent colonies maintained under SPF conditions. A compromise of the SPF barrier is more likely to be responsible for the identification of pathogens in a study, rather than the impairment of the immune system. There are, however, a few opportunistic pathogens which can still cause disease when the immune system is inactivated by immunomodulatory drugs or in study models (eg, severe combined immunodeficiency disorder). A survey showed that several infectious agents are still present in facilities, despite measures to eliminate them. 7,8
Dogs are non-SPF, but have low levels of opportunistic infections and are vaccinated for the most common canine pathogens. For beagle dogs used in toxicological studies, the control of infectious agents is obtained via established vaccination protocols against the most common diseases (eg, canine distemper, parvovirosis, and leptopirosis), as well as treatment for parasites. The presence of opportunistic pathogens is therefore limited; however, dogs are natural carriers of some potential opportunist pathogens, as are nonhuman primates (NHPs), but to a lesser extent. 9
Nonhuman primates are not SPF and carry several opportunistic infections depending on the origin of the animals and transit conditions. Elimination of infectious diseases from NHP colonies has been in progress for a long time, resulting in higher quality of animals with reduced burdens of infectious agents. 9 However, complete removal of all agents from NHP colonies is not possible due to husbandry practices and ethical reasons. Caesarean section and isolation of NHP infants to reduce pathogen transfer is not practical or ethical. Nonhuman primate breeding colonies are large corrals with outside access, so exposure to infectious agents present in wild animals and the outside environment is difficult to avoid completely. 10
Nonhuman primates are often the nonrodent and the only toxicity species of choice to assess the potential toxicity associated with biologic drugs because it is the most pharmacologically relevant species with respect to human target/protein and pharmacology. Various endemic opportunistic pathogens (virus, bacteria, parasite, or fungus) described in NHPs may affect toxicity studies. 11 In immunocompetent animals, these pathogens remain clinically unapparent, while causing increased morbidity or mortality in immunocompromised NHPs. 12
For chronic inflammatory, autoimmune, and allergic diseases in the respiratory system, such as asthma, idiopathic pulmonary fibrosis, and chronic obstructive pulmonary disease (COPD), an expanding field of research is aiming to dampen the inflammatory response or to inhibit the effector immune cells, carrying however an inherited risk of immunosuppression. 13 -16 Inhaled administration is often preferred over systemic administration, not only for direct delivery of the drug into the lung but also as a site of high absorption for systemic treatment. These treatments also require long-term administration, thus the possibility of infection and/or cancer remain a substantial risk.
Inhaled corticosteroids are an example of an inhaled drug for treatment of chronic inflammatory diseases (mainly asthma but also COPD) that has been in use for over 70 years. 17 There is evidence that this treatment is not free of risk for infections in the lungs, especially in the patients with chronic lung diseases. 17 -19 However, the authors of this publication are unaware of any preclinical evidences of such infections via inhalation of corticosteroids.
Biologic immunomodulatory drugs could result in opportunistic infection with pulmonary lesions, which may complicate interpretation of and/or be mistaken for toxicologic findings attributable to the expected pharmacology of the product. It is important to properly differentiate the various features of infection. It is equally important to be aware of the range of possible opportunistic pathogens and how they may impact the interpretation of pathology findings. As no literature on inhaled biologic immunomodulatory drugs is available, this review will focus on parenteral administration of biologic immunomodulators and also inhaled LMW immunomodulators to bridge the gap between biologics and inhalation. In addition, the high dose of biologics (100-150 mg/kg), often systemically administered to animals in toxicity studies, will saturate the target in the lung and induce immune suppression even if it was not delivered directly to the lung by inhalation. Infectious respiratory tract pathology following administration of immunomodulatory drugs in the NHP and other preclinical species will be discussed and translated to clinical experience.
Immunomodulatory Drugs and Respiratory Tract Pathology Related to Infections in NHPs
Respiratory tract pathology related to infection can be difficult to differentiate from direct drug toxicity. Pulmonary inflammation, broncho-alveolar or interstitial, are the most common lung findings observed in toxicology studies conducted in NHPs and is often secondary to infection following dosing with immunomodulatory agents. 11,20,21 Inflammation is generally characterized by inflammatory cell infiltration in the alveolar lumen and/or septa associated with respiratory bronchial epithelium degeneration/regeneration, pneumocyte type II hyperplasia, and/or material accumulation in the alveolar lumen. Inflammation may be diffuse or focal around blood vessels with potential involvement of the vascular wall. The pattern of inflammation will depend on the pathogen involved in the infection. The most frequent respiratory pathogens are briefly discussed below, but further information can be found in literature. 22 -25
Cytomegaloviruses
Cytomegaloviruses (CMV) are members of the betaherpesvirinae family, characterized by slow cytolytic activity and enlargement of the nucleus (karyomegaly).
26
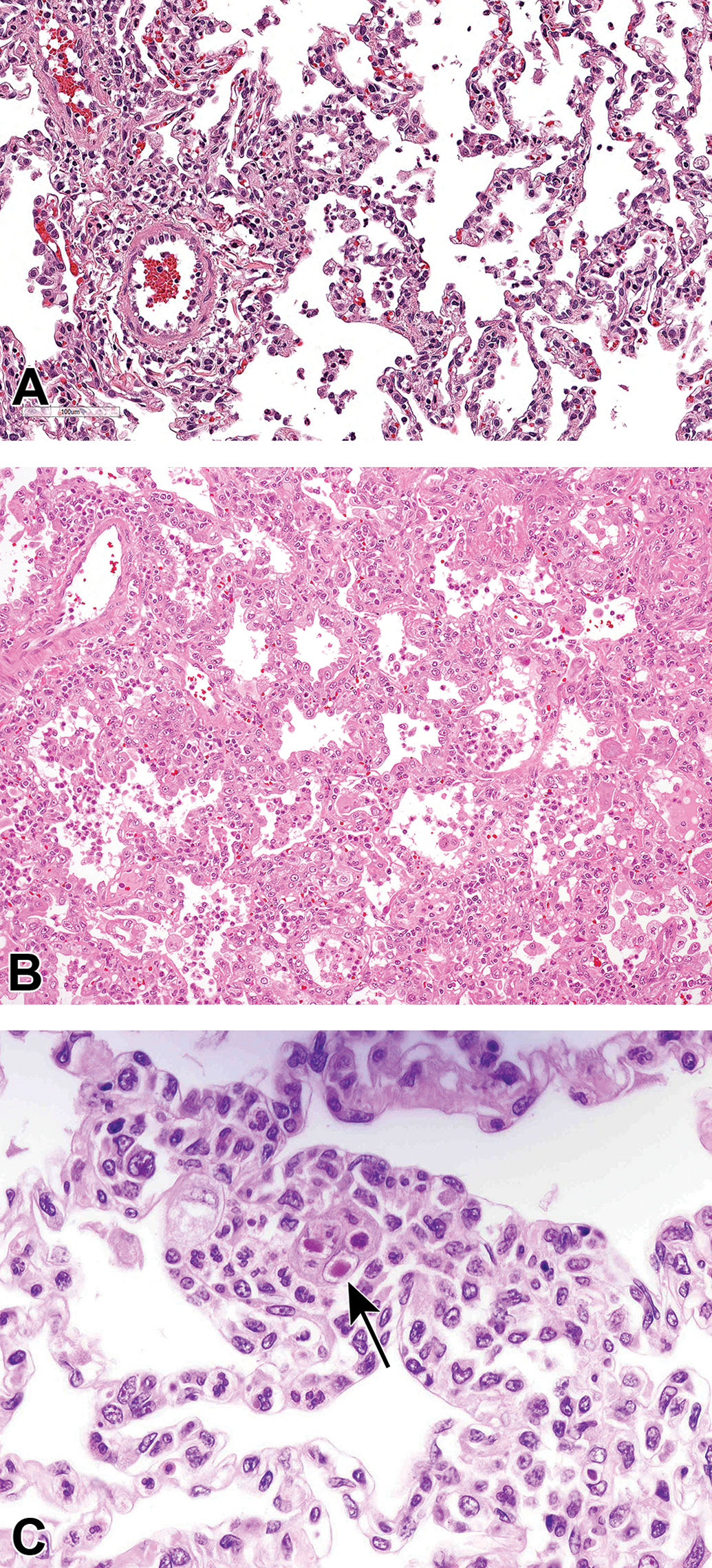
Lung inflammation observed in a cynomolgus monkey treated with a biologic immunosuppressive drug. Slight interstitial inflammation (A), severe exudative interstitial pneumonia (B), and Cytomegalovirus (CMV) intranuclear inclusion body in alveolar interstitium (arrow; C). Hematoxylin & eosin staining.
Other latent herpesviruses can be reactivated and cause disease in immunosuppressed subjects; they usually belong to the family of Alphaherpesvirinae such as the Simian varicella virus (
Adenoviruses
Adenoviruses are a family of nonenveloped, DNA viruses with wide diffusion and wide species range of infection, with high species specificity for the host infection. 34 Adenoviral infection is a common opportunistic infection causing either respiratory or GI infection. 35 Its prevalence is quite variable across NHP colonies but is often above 60%. 36 Infection is asymptomatic in most cases except in immunosuppressed NHPs in which latent infection will develop and cause adenoviral pneumonia or enteritis. 35,37 Adenoviral pneumonia consists of necrosis of the bronchiolar and alveolar epithelium, large basophilic intranuclear inclusions, and exudate in alveolar spaces composed of necrotic cellular debris, fibrin, alveolar macrophages, and neutrophils. 38 In the GI tract, epithelial cell necrosis will cause villous atrophy and clinical signs of diarrhea. Infection can spread in the pancreas, liver, or kidney causing chronic active pancreatitis, necrotizing hepatitis, or tubulointerstitial nephritis, respectively. In a 13-week intravenous toxicity study in rhesus monkeys, pulmonary and renal inflammatory infiltrates and reactive lymph nodes in 2 animals treated with a mid-dose of a biologic immunomodulatory drug were associated with the presence of adenovirus and/or cryptosporidium (see Figure 2) in 1 of these 2 animals, as well as in 5 other animals whom only had lymphoid increased cellularity (Flandre et al, paper in review).
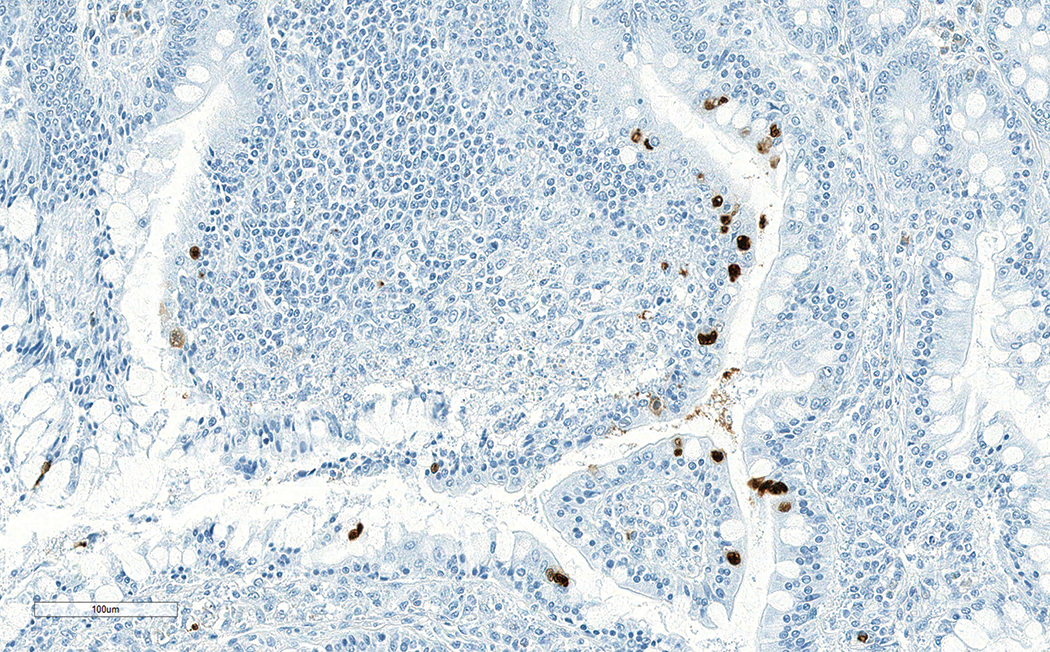
Detection of adenovirus in enterocytes of the colon mucosa observed in cynomolgus monkey treated with a biologic immunosuppressive drug. Immunohistochemistry using a mouse antiadenovirus antibody (MAB8052 Millipore).
Measles
Measles (rubeola) is a RNA virus of the genus Morbillivirus whose natural host is the human; nevertheless, it has broad capability to infect other primates.
34
This is rare in laboratory NHPs as they are usually vaccinated against this agent, yet more difficult as monovalent human vaccine is no longer available; however, it could cause significant outbreaks with severe consequence in case of transmission from infected humans to unvaccinated or immunocompromised NHPs.
39,40
Acute measles infection induces severe immunosuppression (myeloid cells, B- and T-cells) that will often cause secondary infection by opportunistic pathogens like CMV, adenovirus, C
Simian Virus 40
Simian virus 40 (SV40, also called cynomolgus polyoma virus or CPV in cynomolgus monkey, 43 a DNA virus of the family Polyomaviridae, is a common latent infection with prevalence of more than 45% in cynomolgus monkeys and 75% in rhesus monkeys. 44 Simian virus 40 is not associated with disease in healthy animals and is only observed in immunosuppressed NHPs. Infection by SV40 affects mainly the brain, with lesions similar to progressive multifocal leukoencephalopathy, but also the kidneys and lungs. 45 Pulmonary infection displays a proliferative interstitial pneumonitis with inclusions in hypertrophied type II pneumocytes. 46 In a kidney transplantation model in NHPs with administration of rituximab, sirolimus, and thymoglobulin, graft rejection was noted due to SV40 infection causing polyomavirus nephropathy and inclusion in the lungs and other organs. 47
Parainfluenza Viruses
Parainfluenza viruses (PIV), especially PIV-1 also called Sendai virus, have been associated with fatal pneumonia. 48,49 Parainfluenza viruses affect the upper and lower respiratory tract causing serous or purulent nasal exudate and acute interstitial pneumonia.
Tuberculosis
Tuberculosis is one of the most common respiratory infections associated with some biologic immunomodulatory drug classes in the clinics, due to reactivation of latent infections. In preclinical toxicity studies, tuberculosis is normally not observed as animals are prescreened in quarantine by intradermal tuberculin skin test; nevertheless, natural tuberculosis in NHPs can still occur in macaques imported from Asia as the skin test can fail to detect latent infections.
50
Animal models of tuberculosis are used to screen or benchmark immunomodulatory drugs for potential risk of tuberculosis. A NHP model of tuberculosis was used for anti-tumor necrosis factor α (anti-TNFα) biologic drug to assess the immune modulation following TNF blocking during primary or latent infection by
Bordetella bronchiseptica
Pneumocystis spp
Cryptosporidium parvum
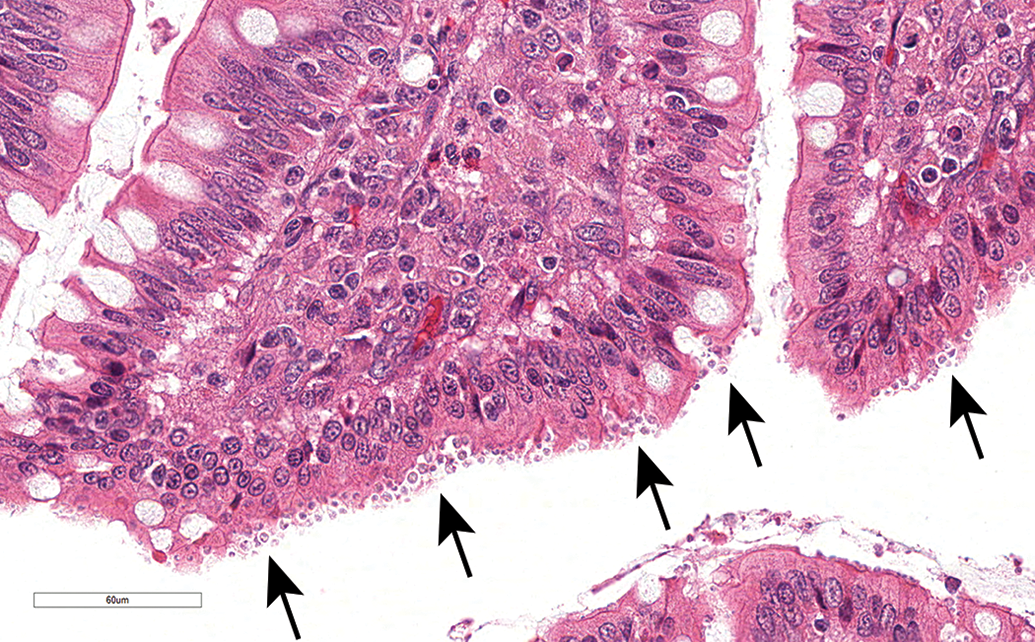
Cryptosporidium organisms focally infecting the enterocytes of the intestinal mucosa (arrows) observed in cynomolgus monkey treated with a biologic immunosuppressive drug. Hematoxylin & eosin staining.
Candida albicans
A few more pathogens causing respiratory disease (
Other pathogens (ie,
Clinical Translation
Infection associated with biologic immunosuppressive drugs given to patients in the clinic are extensively characterized and described in the literature and Health Authorities submission package. For an overview on some biologic immunosuppressive drugs discussed further below, please refer to Table 2. Infection in patients were mostly seen in the (by order of most to lower incidence) respiratory tract, skin, urinary tract, gastrointestinal tract, and nervous system. All biologic immunomodulatory drugs have an increased risk of developing a particular profile of infections: viral (CMV, Epstein-Barr virus [EBV], herpes simplex virus [HSV], hepatitis-B virus [HBV], and human polyomavirus 2 [JCV]), fungal (Candidiosis, Aspergillosis, Pneumocytosis, Histoplasmosis), or bacterial (tuberculosis, legionellosis, staphylococcal, or streptococcal).
Overview of Mode of Action of Biologics; Target Organ Prediction in Preclinical Species and Correlation With Immunosuppression in the Clinics.

Abbreviations: anti-TNFα, anti-tumor necrosis factor α; GI, gastrointestinal tract; IL, interleukin.
Anti-TNFα drugs (infliximab, adalimumab, golimumab, certolizumab pegol, and etanercept) despite having different pharmacological modes of action, nevertheless have a common action in neutralizing soluble TNFα, negatively impacting multiple inflammatory cell responses and pathways. The pharmacological outcome of anti-TNFα treatment (via effect on innate phagocytic immunity) may include reduced neutrophil and eosinophil chemoattraction; reduced macrophage chemokine production; downregulation of adhesion molecules important for trafficking cells to inflammatory sites (eg, leukocyte adhesion molecule, endothelial leukocyte adhesion molecule-1, and vascular cell adhesion molecule-1); reduced promotion of vasodilation and its contribution to inflammation via inhibition of release of eicosanoids and platelet-activating factors, as well as negatively impacting oxidative and nitrosative stress in inflammatory cells. 67 All of which ultimately lead to a reduced ability to mount an inflammatory response and predisposition to opportunistic infection of the respiratory tract in animals and humans. They have been associated in clinics with increased susceptibility to tuberculosis or other opportunistic infections, bacterial and fungal mostly, but also viruses like HBV. 68 Infection (histoplasmosis) or signs of infection (increased lung weight with mononuclear cell infiltration) related to anti-TNFα drugs golimumab (Simponi) or etanercept (Enbrel) have been reported in NHP toxicity studies. 69,70 Infliximab and its biosimilars are interesting examples of anti-TNFα drugs because human and chimpanzees are the only species whose TNFα binds cA2. Thus, chimpanzees are the only preclinical species that might provide safety information relevant to humans for that biologic’s target. However, as an endangered species, chimpanzees may not be necropsied, so study outcomes are limited to clinical observations. Chimpanzees showed no clinical observations consistent with the presence of opportunistic infections, albeit in a study limited to 5 animals. 71 Some of the nonclinical toxicology studies supporting infliximab submission package were also conducted in mice with a surrogate anti-mouse TNFα mAb. 71,72
Conversely, there are immunomodulatory compounds where immunosuppressive effects resulted in increased infections in humans only, despite showing pharmacological effects and morphological changes consistent with immunosuppression in preclinical species exposed to the same compounds. Immunosuppression was not manifested in the latter by increased susceptibility to opportunistic infections. Thus, the presence or absence of opportunistic infections in preclinical species is not necessarily predictable to what might occur in humans.
Ustekinumab (marketed as Stelara), a fully humanized mAb, exerts its pharmacological effect by neutralizing interleukin-12 (IL-12)- and IL-23 (Th17-) mediated cellular immune responses.
73
The cynomolgus monkey was chosen since the mAb neutralizes the monkey and human cytokines to a similar extent. In the
In future development of biologics that exert their pharmacologic effects via the same or similar pathways, it will be worthwhile to recall that past experience showing opportunistic infections have not been typical for compounds with this mechanism of action. And when such opportunistic infections might occur in a study, other causes for immunosuppression (eg, stress or concurrently viral infections like measles virus or simian retrovirus type D) or colony factors might be considered and not automatically be assumed to be associated with immunomodulatory pharmacologic activities of the test article.
Natalizumab, a recombinant, humanized mAb, acts as a selective adhesion molecule inhibitor. 75 Acting on adaptive cellular immunity, it specifically inhibits T cell transition into the central nervous and GI. It blocks the T cell α4 integrin and inhibits both α4β1 interaction with VLA-4 expressed on brain endothelial cells as well as α4β7 interaction with MadCAM-1, expressed on gut endothelial cells. It can also be immunosuppressive via inhibition of the interaction of α4integrin-expressing leucocytes with ligands like osteopontin and epitopes of fibronectin (ie, important for immune cell activation and survival). Several repeat dose toxicity studies in cynomolgus monkeys, including a 26-week study followed by a 17-week dose-free period, were performed. Findings consistent with immunosuppression were commonly observed across the studies and they included dose-related, significantly increased white blood cell counts extending into recovery periods and severe increases in spleen weights. Despite this, no opportunistic infections were reported in any organ systems. 75
Abatacept, a recombinant fusion protein consisting of the extracellular domain of human CTLA-4 and the hinge CH2-CH3 regions of the Fc domain of human IgG1, has been modified to prevent complement fixation and antibody-dependent cell cytotoxicity. 76 It blocks adaptive cellular immune response through inhibition of various costimulatory signals required for T cell and inflammatory effector cell (macrophages, B cells) activation. It was shown to cause adverse effects of increased severity in juvenile versus adult rats. In addition to morphologic findings in the spleen and lymph nodes consistent with immunomodulation, thyroiditis and pancreatic islet cell inflammation were observed and considered an outcome of autoimmunity. Again, there was clear evidence of immunomodulation, where secondary opportunistic infections were not an outcome. However, autoimmunity, a safety concern in humans, was predicted in the preclinical species. 76
Thus, when testing immunomodulatory compounds, the occurrence of opportunistic infections, respiratory or otherwise, is not necessarily as given in preclinical species despite the appearance of histopathologic or hematologic findings consistent with immunomodulation. Understanding the pharmacologic activity of the test article and the mechanism by which immunomodulation occurs could be instrumental in determining if opportunistic infection is a consequence of such pharmacologic activity or might be attributed to other factors (eg, stress, colony factors, NHP species).
In summary when reviewing available data in literature or briefing book, infections were more frequently observed in human than in preclinical studies. In general, preclinical species are only 30% to 80% predictive of toxicities observed in clinic, even though the animals are dosed at multiples of human exposure.
77,78
Multiple factors can explain the lack of sensibility/translatability of our preclinical species. One explanation is the power of clinical studies compared to preclinical studies with cohort of thousands of patients compared to the few treated animals (up to 12 NHPs by dose), which are in controlled environments compared to patients. The multifactorial disease status of patients associated with multiple concomitant medications, especially immunosuppressive drugs, will also impact clinical safety.
79
One way to try and predict infection risk outcome following administration of a biologic immunomodulatory drugs is to look at gene-associated human immunodeficiencies, which are linked to specific infection types. Human adaptive immune deficiencies are frequently associated with viral infections; as well as, bacterial infections with antibody deficiency, mycobacteria, and fungi infections with cellular deficiency and all type of pathogens with combined antibody and cellular deficiency. In contrast, innate immunity deficiencies (phagocyte or complement defect) are frequently associated with bacterial infections, while viral infection is a rare event. Mycobacteria and fungi are also frequent infectious agents in human with phagocyte deficiency. For review see studies by Bonilla and Geha
80
and Picard et al.
81
Knowing the infection related to the pathway could help to determine infection type (viral [CMV, adenovirus], bacterial, fungal [Cryptosporidium,
How to Diagnose
Firstly, in case of known potential infections, collecting at necropsy additional tissue samples (eg, kidney, lungs, GI, heart, mesenteric lymph node, spleen, and any tissues with clinical signs), ideally snap frozen organs, could help in definitively diagnosing such infections via H&E examination and/or special investigation.
Without special investigations, diagnosis of infections in NHPs is often based on a weight of the evidence approach: in-life data, clinical pathology, gross pathology, and histopathology findings. One should be careful when reviewing histopathology tables as the incidence among treatment groups may not be dose-related but may in fact be linked to the immune status of individual animals and pathogen burden. A careful examination of other tissues is also important as inclusion bodies might be found in distant tissues with little inflammation or only one focus of inflammatory cell infiltrate.
Special investigations are the only way to diagnose the pathogen involved.
82
For this purpose, specific sampling is normally needed. But if not performed, the left over blood samples or formalin-fixed, paraffin-embedded tissue might be used. With tissues, molecular localization techniques including immunohistochemistry (eg, CMV, see Figure 4), in-situ hybridization, or electron microscopy (eg, Adenovirus) are key for identifying viral infections, while special staining like Gram’s, methenamine silver, or toluidine blue staining will be useful to identify bacteria (
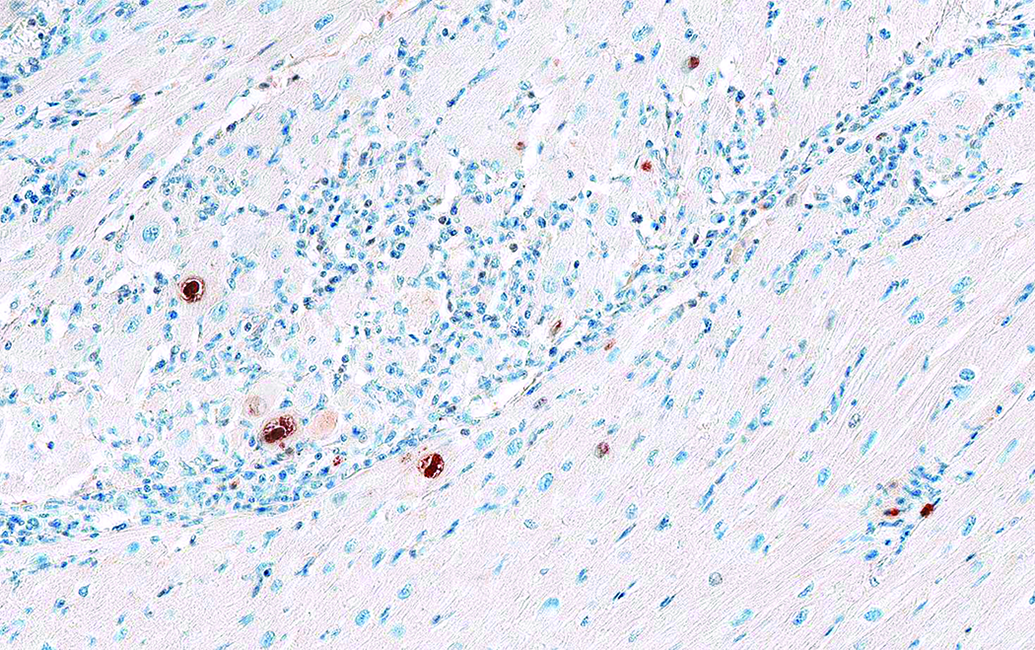
Detection of Cytomegalovirus (CMV) in heart of a cynomolgus monkey treated with a biologic immunosuppressive drug. Immunohistochemistry using a rabbit anti-RhCMV IE1 (University of California, Davis).
Conclusion
Respiratory infection following systemic administration of biologic immunomodulatory drugs is infrequent in preclinical studies. Globally, the types of infections seen in preclinical studies are underpredictive of the outcome in the clinics. In preclinical studies, low numbers of young, healthy animals (SPF for rodent and prescreened animals for nonrodent) housed in a regulated environment are used to assess potential drug toxicity. In the clinical situation we face a large range in patient age and health status, in an uncontrolled environment with an unknown pathogen burden prior to and during treatment; interference of other medications (multidrug therapy is the standard) and longer duration of treatment.
Diagnosis of infection in NHPs is often based on a weight-of-evidence approach (ie, in-life data, clinical, and histopathology). Precise identification of an infectious agent is not always pursued as it will require special investigation. However, pathologist should bear in mind that inflammatory cell infiltration or inflammation observed in the lungs following administration of a biologic immunomodulatory drug could be an indirect pharmacological consequence resulting in opportunistic infection, rather than direct toxicity of the drug. Understanding the pharmacologic activity of the test article and the mechanism by which immunomodulation occurs could also help in determining if opportunistic infection is a consequence of pharmacologic activity or might be attributed to other factors.
The main issues associated with infection in preclinical species are morbidity with premature sacrifice and/or mortality that could compromise interpretation of the toxicity study. To limit such complications, good practices for testing biologic immunomodulatory drugs might include extensive serology of NHPs to understand the level of background opportunistic pathogen agents (if any); study planning/design (eg, limit blood sampling volume/time point, housing with restricted passage and enhancement of hygienic measures, isolation of infected animals, planning for blood/organ sampling in infected animals); temperature monitoring (by chip); and additional organ sampling for diagnostic purposes.
To conclude, not only will proper interpretation of microscopic findings be key, but knowing the target pathway and gene-associated human immunodeficiencies will greatly help to predict potential infection types.
Footnotes
Acknowledgments
The authors thank Franck Brennan and Keith Mansfield (Novartis Institutes for Biomedical Research) for expert review of the manuscript.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.

