Abstract
Biotherapeutics are pharmaceutical products derived from or synthesized by biological systems. Such molecules carry the potential for immunogenicity which may lead to adverse immune responses. The cynomolgus macaque (Macaca fascicularis) is the species of choice in nonclinical safety assessment of biotherapeutics. The main aim of this study was to confirm whether mononuclear cell infiltrates at specific locations represent a generic effect of biotherapeutics, and therefore the result of their immunogenicity. Following a review of microscopic findings in studies conducted over a 10-year period at one test facility, 15% of biotherapeutics were reported to have such findings. The most commonly affected site was the choroid plexus and less frequently the meninges and ciliary body. The reporting of such findings as test article–related becomes more subjective as the severity and incidence decreases. To assess the accuracy of such associations, a mathematical approach was employed to determine the probability of obtaining the observed results by chance. There was good agreement between this approach and the original findings. In addition to an increased number and size of mononuclear cell infiltrates in the brain, biotherapeutic administration was strongly associated with the presence of plasma cells and eosinophils.
Biotherapeutics are pharmaceutical products derived from or synthesized by biological systems. For many years, an increasingly large proportion of drugs being developed to treat a wide range of human diseases fall into this category. Due to the nature of biological molecules, typically recombinant proteins or immunoglobulin, they can be engineered to be highly specific to both the host and the intended immunological or physiological function. Such molecules are potentially immunogenic and antidrug antibodies (ADAs) are typically measured in both nonclinical and clinical toxicology studies to monitor this. These have the potential to interfere with drug clearance and target binding, compromising drug efficacy and may also lead to adverse infusion reactions such as anaphylaxis (type I hypersensitivity). However, such reactions are rare despite ADAs being produced in response to most biotherapeutics (Salazar-Fontana et al. 2017; Song et al. 2016).
Nonhuman primates (NHPs) are the most commonly used species in nonclinical toxicology studies using biotherapeutics intended for use in humans, mainly due to their close phylogenic relationship to humans. Of the available species, the cynomolgus macaque (Macaca fascicularis) has become the most widely used species and the majority of these are purpose bred for laboratory use. As biotherapeutics are intended for use in humans, the potential for these to contain epitopes that are distinct from the cynomolgus macaque version of the molecule means that immune responses are more likely in this species and thus likely to overestimate the risk to humans. This is potentially exacerbated by the high doses administered in such studies intended to provide a safety margin for human risk assessment. Reactions to biotherapeutics in nonclinical studies may result in immediate or delayed adverse effects such as anaphylaxis, circulatory effects, immune complex (IC)–mediated hypersensitivity reactions resulting in vasculitis, glomerulonephritis, and coagulopathies (Everds and Tarrant 2013; Frazier et al. 2015; Krishna and Nadler 2016; Kronenberg et al. 2017; Mease et al. 2017).
The main aim of this study is to present and discuss some of the histopathological lesions considered to be the result of immune responses to biotherapeutics within cynomolgus macaques from studies carried out at Charles River Laboratories, Edinburgh, by reviewing all available pathology reports within a 10-year period. Specifically, lesions of vascular inflammation, glomerulonephritis, and mononuclear cell infiltration in the brain were searched for, based on known predilection sites for IC deposition (Roiko et al. 2014). Some studies were reported to have notable mononuclear cell infiltrates, particularly in the choroid plexus. However, because the presence of low-grade mononuclear infiltrates is known to occur here and in many other sites in NHPs spontaneously (Chamanza et al. 2010; Sato et al. 2012; Butt et al. 2014), this meta-analysis was complimented by reevaluation of the brains from available studies with such findings and a selection of studies without such findings. This was to remove operator variation, to confirm the presence/absence of certain cell types within the infiltrates, and to compare the choroid plexus with the meninges. With this large robust data set, additional information relating to dose and study duration was also assessed. The ciliary body of the eye is also a known site of mononuclear cell infiltration (Sinha, Cartwright, and Johnson 2006) and an increased incidence was suspected in some studies, so the eyes were also reevaluated from several studies.
Materials and Methods
Animals
The animals were purpose bred for laboratory use and came from accredited suppliers (Bioculture, Mauritius; Harlan, UK; Guanxi Grandforest Primate company, China; Belgrave Services, Vietnam) from three geographical regions: Mauritius (more than 90% of the animals), China, and Vietnam. Rhesus macaques (Macaca mulatta) were not included in the analysis due to the potential for species-specific differences and the low number of studies available. No potential immune-mediated effects were reported in studies using this species.
Ages were between 2 and 6.5 years. They were housed in groups, typically of 2 to 3 (but up to 6) animals of the same sex and dose group in custom-designed Home Office approved primate cages (Chapter 14, section 21, UK Animals [Scientific Procedures] Act of 1986). The temperature and humidity were automatically controlled at 21°C ± 4°C and 55% ± 10%, respectively, with a minimum of 15 air changes per hour. An automatic light cycle of 07.00 a.m. to 19.00 p.m. (12-hr cycle) was maintained. Each individual gang pen had drinking water facilities and depending on cage type was fitted with food hoppers or scattered on pen floor, and animals were fed a commercial primate diet (Mazuri diet, Special Diet Service Ltd., Witham, Essex, England). Twice weekly fruit supplements were offered to all animals.
All studies were conducted in accordance with the UK Animals (Scientific Procedures) Act 1986, which conforms to the European Convention for the Protection of Vertebrate Animals Used for Experimental and Other Scientific Purposes (Strasbourg, Council of Europe).
Pathological Evaluation
Animals were euthanized by intravenous injection with sodium pentobarbitone and exsanguinated via femoral veins. Tissues were preserved in 10% neutral-buffered formalin, embedded in paraffin wax, sectioned to a 4 to 5 μm thickness, and stained with hematoxylin and eosin. They were examined histopathologically and the findings entered directly onto a computerized database (PLACES 2000 or Provantis 8, Instem, Stone, Staffordshire, UK).
Clinical Examination and Laboratory Evaluation
A thorough clinical examination that included physical examination, ophthalmology, and electrocardiography was carried out on each of the animals by a veterinary surgeon or trained technical staff.
All animals on the study were tested and confirmed free of the following viral diseases using serology during quarantine: simian immunodeficiency virus, cercopithecine herpes B virus (except those from Mauritius, which are certified free from this disease), simian retroviruses type D, rabies, simian T-cell leukemia virus, and filoviruses. Tuberculin tests, other bacteriology screening tests for Shigella, Yersinia, and Salmonella, and parasitology screening (including malaria smears) were carried out on arrival at the test facility.
Study Design
Pathology reports for all studies on the Charles River Laboratories Edinburgh electronic data base over a 10-year period (2007–2016) were reviewed for findings considered to potentially indicate an immune response to the test article, including mononuclear cell infiltration at certain sites, glomerulonephritis, and vascular/perivascular inflammation. Various details of such studies were compiled and tabulated to determine potential trends and risk factors. Of the 15 studies with effects, slides were available for 11 studies. The brain and eye slides were reassessed by a single pathologist specifically searching for mononuclear cell foci. Foci were counted for each of the two compartments (meninges or choroid plexus), and the presence of plasma cells, eosinophils, and neutrophils and any particularly large foci were recorded. A further eight studies without any reported immune-mediated effects were similarly reassessed. These were selected on the basis of the test article being a monoclonal antibody, the highest dose being 50 mg/kg or higher, a duration of dosing of 4 weeks or more, conducted within a time frame (2007 to 2013) and slides being available for review.
The standard trimming procedure for the brain at the test facility prior to 2012 was three sections—the frontal pole, the caudal thalamus, and the mid cerebellum (corresponding to sections 1, 4, and 5B, Pardo et al. 2012). Additional sections were added to the standard trimming procedure in 2012 corresponding to those described by Pardo et al. (2012). For consistency across all studies, only sections corresponding to the original three were reassessed from these studies.
Statistics
In order to define objectively how many foci per animal should be considered an increase, the distribution within a large cohort of control animals (99 males and 91 females from all 19 studies reevaluated) was utilized to determine the lowest number of foci/animal that resulted in less than 5% of controls being considered immune-responsive. This was applied to foci in the meninges and choroid plexus separately. In the case of meninges, this was 3 foci per animal, whereas in the choroid plexus, 1 focus per animal was used due to the low incidence of foci in control choroid plexi (only 7/190 with 1 focus). In addition, the presence of additional cells such as plasma cells or polymorphonuclear cells or foci judged to be large was also used to define an immune-responsive animal. Each study group in each study (sexes combined) was then analyzed using a Bernoulli trial to determine the probability of observing the proportion of animals considered to be immune responsive, based on the above criteria, under the assumption that animals were selected at random from the same population as the 190 controls. Thus, the proportion of control animals defined as immune responsive, based on the above criteria, was used as the probability of any treated animal being considered immune responsive, that is, the probability of success (p) in the Bernoulli trial. Using the appropriate p for each criteria in a Bernoulli trial, the probability p of obtaining the observed result of k successes in a group of n animals was calculated for each dose group from each study, using the formula P(k) = nCk pk q1−k (where q = 1 − p; Papoulis 1984). The level of P below which an increase was considered to be significant was set at .05 for a study with one dose group and was adjusted down to correct for multiple comparisons depending upon the number of dose groups in a study according to the Bonferroni method, that is, .025 for 2 dose groups and .0125 for 4 dose groups (Chen, Feng, and Xiaolian 2017).
Results
Review of Toxicological Studies in Cynomolgus Macaques for Immune Reactions to Test Articles
All studies with cynomolgus macaques listed on the Charles River Laboratories Edinburgh electronic database with available pathology reports issued within a 10-year period (a total of 160), testing 120 different test articles, were reviewed. A total of 15 studies/12 test articles were reported to have one or more microscopic findings suggestive of an immune response to the test article. These findings were inflammatory (primarily lymphoid) cell infiltration in the choroid plexus (Figure 1) and/or meninges (Figure 2; 11 test articles), ciliary body (Figure 3; 3 test articles), vasculitis (3 test articles), and glomerulonephritis (Figure 4; 2 test articles; Table 1).
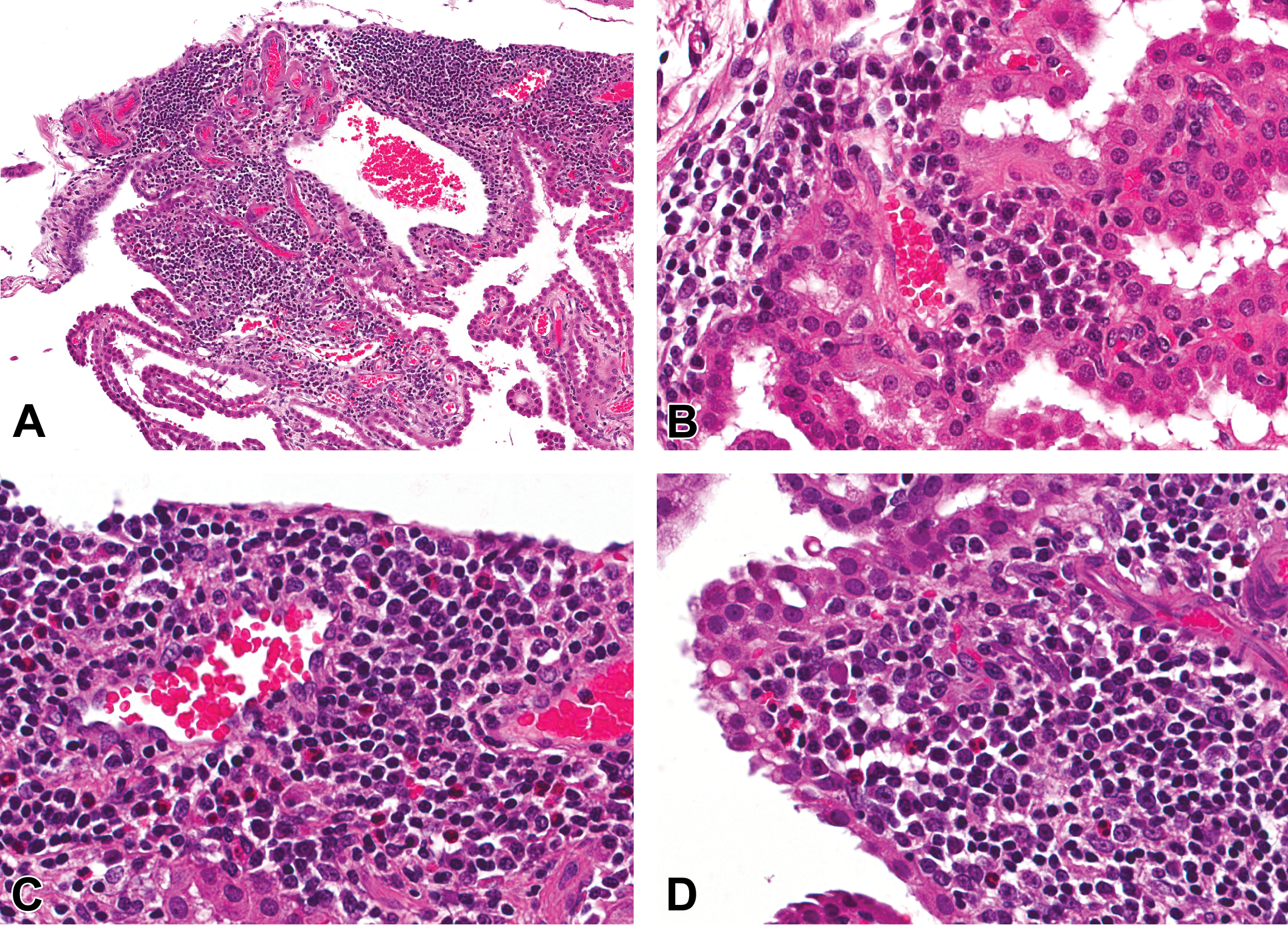
Mononuclear cell infiltration in the choroid plexus of cynomolgus monkeys given biotherapeutics. Extensive mononuclear cell infiltration in the choroid plexus (A). High-power views of choroid plexus infiltrates including plasma cells (B–D) and eosinophils (C and D; hematoxylin and eosin).
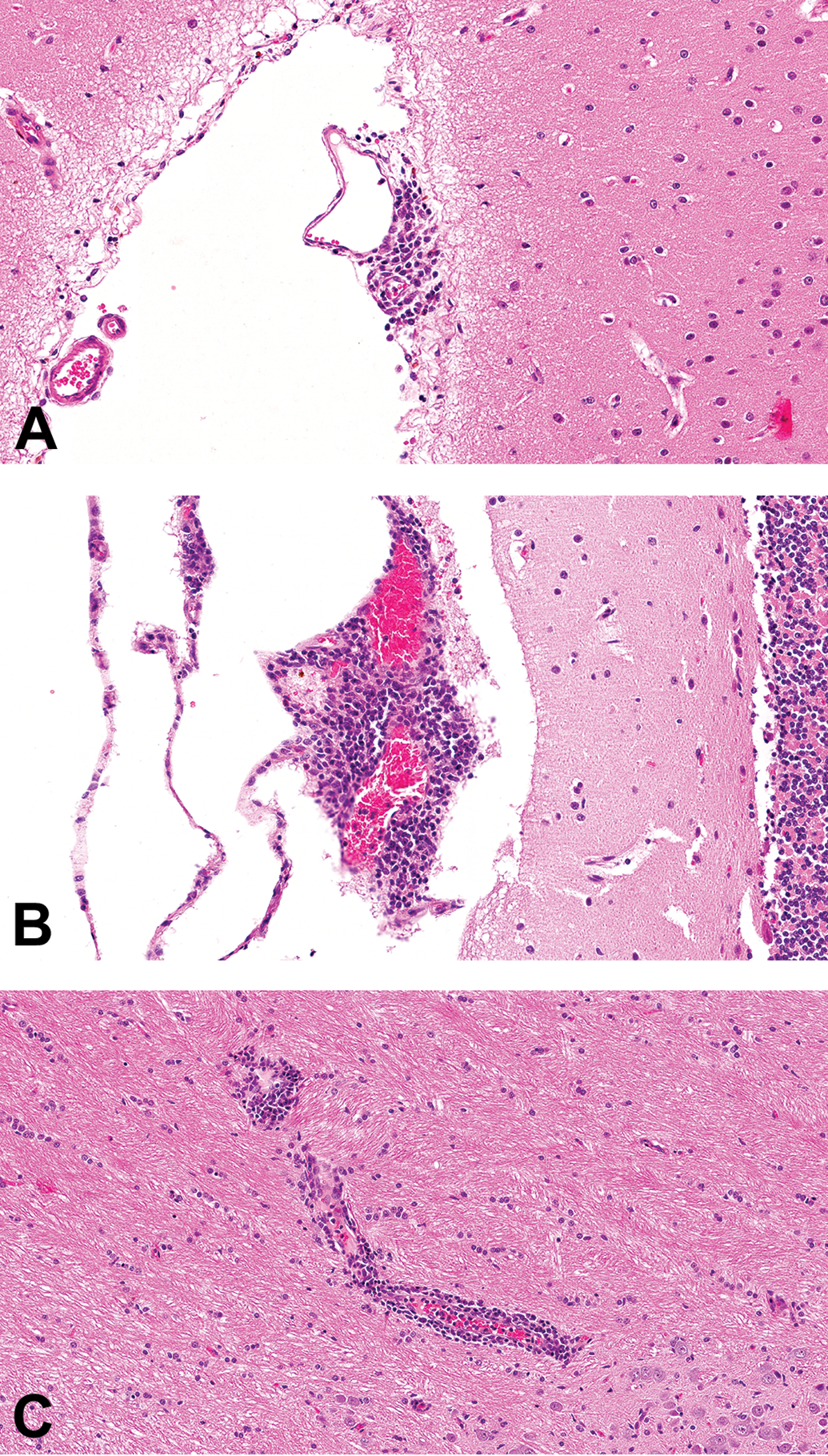
Mononuclear cell infiltration in the meninges of the brain in cynomolgus monkeys given biotherapeutics (hematoxylin and eosin). A small perivascular focus in the meninges, typical of those that occur spontaneously (A). A larger meningeal focus, associated with animals given biotherapeutics, as in this case (B). A focus surrounded by brain parenchyma but associated with a blood vessel therefore unlikely to have fully crossed the blood-brain barrier (C).
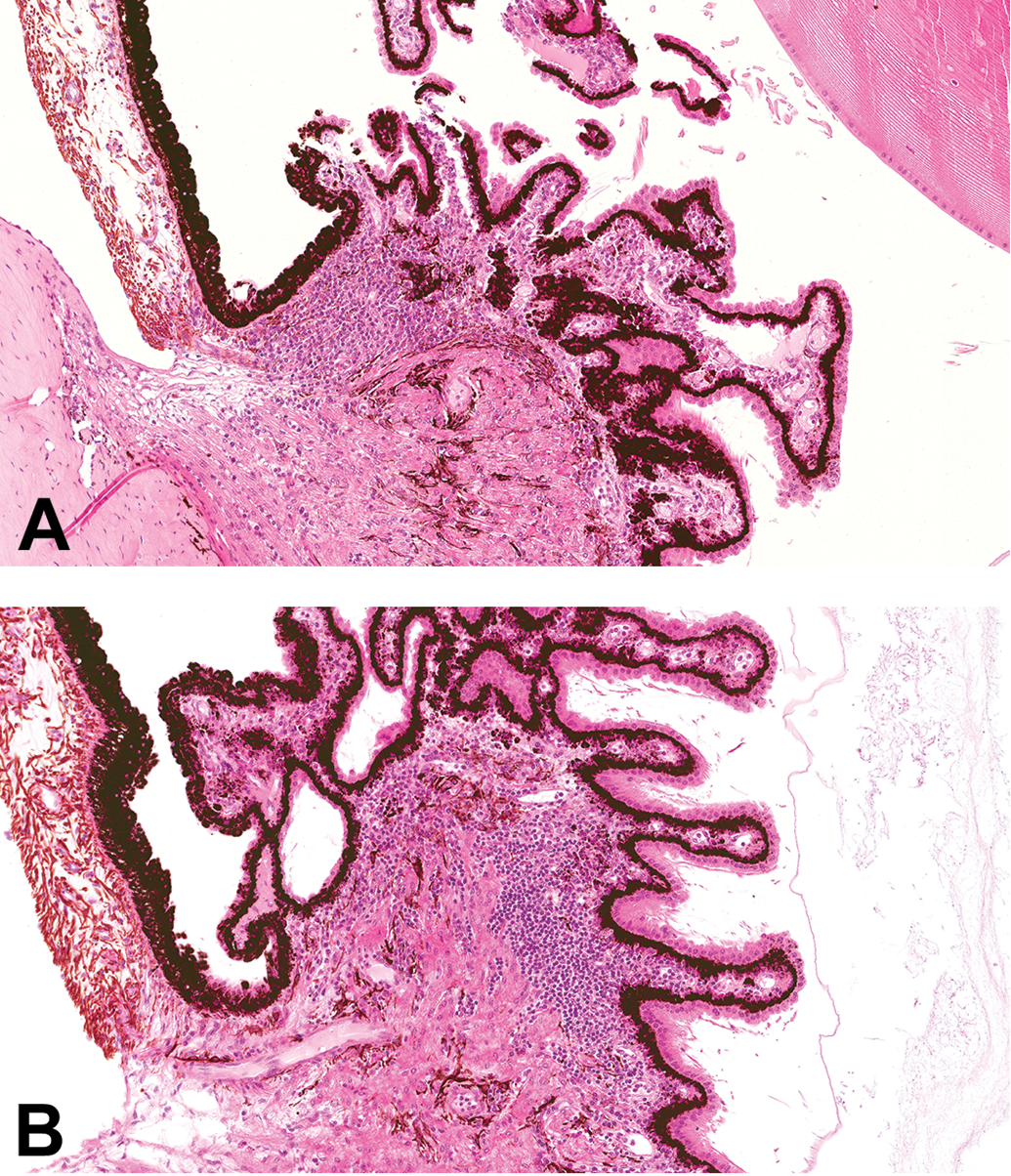
Lymphocytic infiltration in the ciliary body of the eye of cynomolgus monkeys (A and B; hematoxylin and eosin).
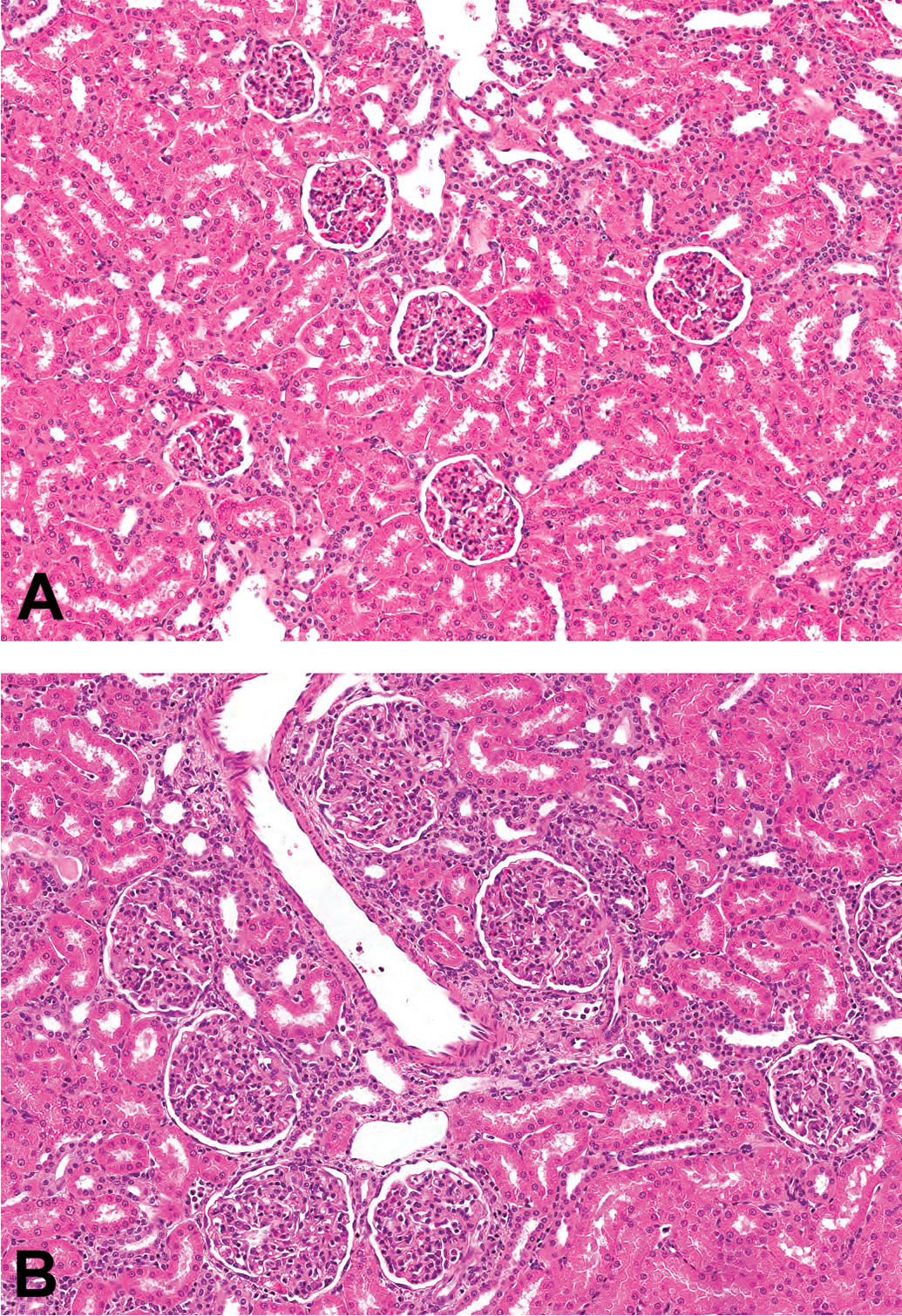
Glomerulonephritis. Glomeruli in a control cynomolgus monkey kidney (A). Enlarged glomeruli with increased cellularity and thickened basement membrane in biotherapeutic-associated glomerulonephritis (B; hematoxylin and eosin).
Studies/Test Articles with Reported Findings Consistent with Immune Mediated Responses to Test Articles.
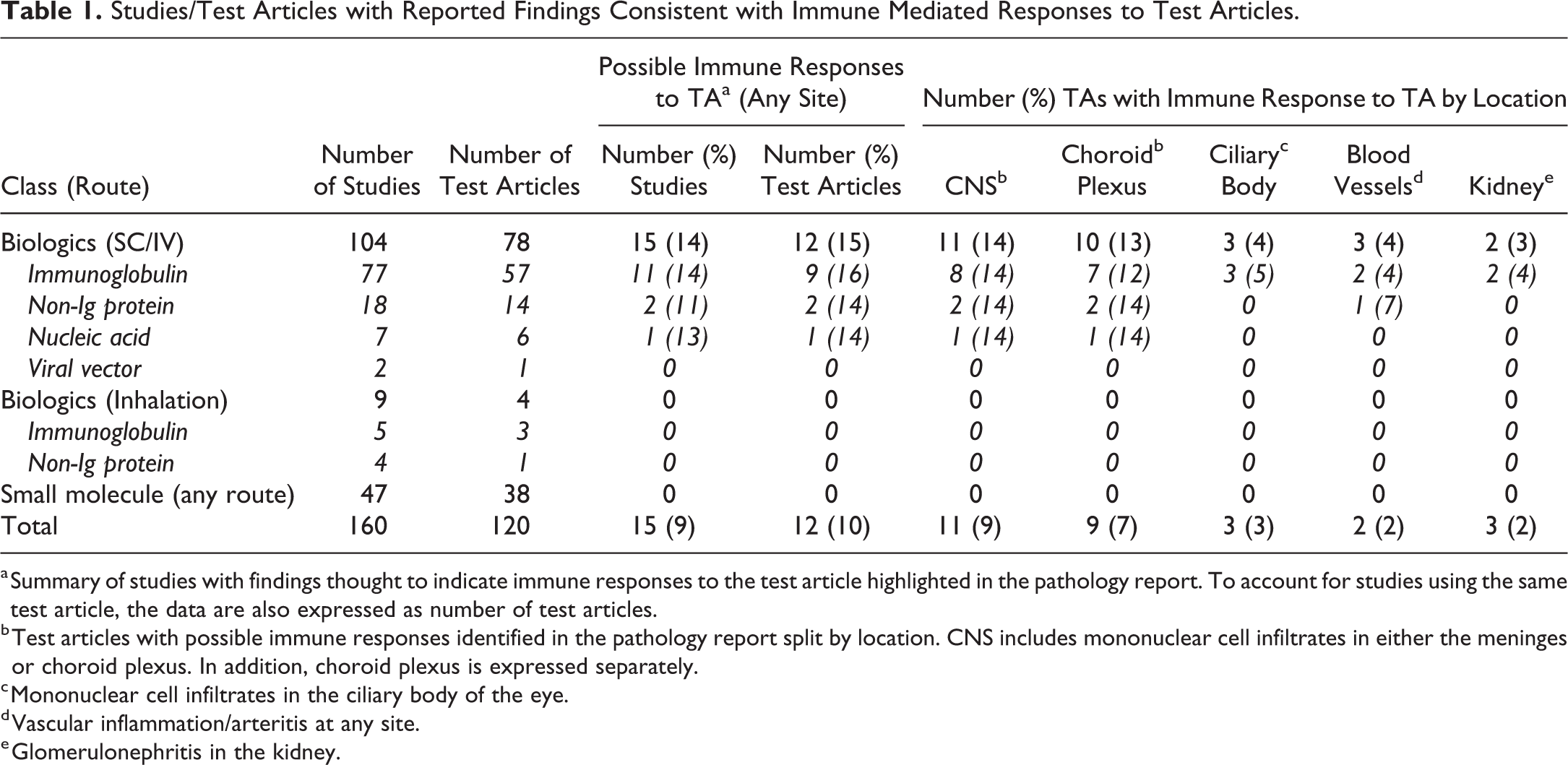
a Summary of studies with findings thought to indicate immune responses to the test article highlighted in the pathology report. To account for studies using the same test article, the data are also expressed as number of test articles.
b Test articles with possible immune responses identified in the pathology report split by location. CNS includes mononuclear cell infiltrates in either the meninges or choroid plexus. In addition, choroid plexus is expressed separately.
c Mononuclear cell infiltrates in the ciliary body of the eye.
d Vascular inflammation/arteritis at any site.
e Glomerulonephritis in the kidney.
All studies with such findings tested an immunoglobulin, a nonimmunoglobulin protein, or nucleic acid biotherapeutic administered by the subcutaneous or intravenous route. There were no examples in studies testing small molecules by any route (47 studies/38 test articles) or biotherapeutics administered by inhalation or intranasal routes. However, it was noted that the latter routes almost always yielded local immune reactions ranging from peribronchial/perivascular lymphoid infiltration to mixed cell bronchiolo-alveolar or alveolar inflammation.
There was a positive relationship between the duration of dosing and the risk of observing an immune-mediated finding. Of studies in which biologicals were administered via the intravenous or subcutaneous route, immune responses were reported in 11.5% (3/37) of studies with a single/double dose or less than 4 weeks duration, 12.2% (5/41) of studies of 4 to 8 weeks, 21.7% (5/23) of studies of 9 to 13 weeks, and 33.3% (5/15) of studies of 26 weeks or more.
Quantitative Assessment of Selected Immune Responses within a Subset of Studies
The brain and eye sections, for studies in which they were still available, were reassessed by a single pathologist specifically searching for mononuclear cell foci. This was to confirm the effect apparent from the meta-analysis of pathology reports, to remove operator variation, allow a more direct comparison between different studies for the effect of dose and duration of exposure, and potentially increase the sensitivity for numerical increases in more subtle foci. The number of distinct foci were recorded for each animal and any foci containing plasma cells and/or eosinophils and any unusually large foci were noted. A sample of studies without any reported immune mediated effects were similarly reassessed. The purpose of this was to increase the cohort of controls used to determine the background level of findings and test whether there was a low-level immunogenic effect being underreported, due to the small group sizes and variation in operator sensitivity.
Background Levels of Mononuclear Cell Infiltrates within Control Animal Brains
It is established that mononuclear cell infiltrates occasionally occur within the meninges and choroid plexus of the brain and the uveal structures of the eye (Figures 1 –3). These have been reported in the literature (Chamanza et al. 2010; Sato et al. 2012; Butt et al. 2014) and recorded by the authors in previous studies. To test whether test article administration results in a quantifiable increase in the number or character of such foci, controls and treated animals were reassessed in a blinded fashion. When all the control data were compiled for the brain, there were no mononuclear cell foci at any site in 145/190 (76%) of animals. Of the 41 control animals with mononuclear cell foci in the meninges of the brain, only five had four or more foci, a large focus, or foci with additional cells such as plasma cells or polymorphonuclear cells. This forms the basis for a threshold for the analysis of test article-treated animals described later. The proportion of control animals with choroid plexus lesions was low, only 7/190 (3.6%) with a single focus. Unusual meningeal lesions within controls were one animal with a focus associated with vasculitis and associated macrophages and one neutrophil, one large focus containing plasma cells, two other animals with large foci, and one animal with a regular-sized focus with a single plasma cell.
Treatment-related Increases in Mononuclear Cell Infiltrates in the Brain
All groups (males and females combined) from 11 studies (representing 8 test articles) with available slides and pathology report findings indicative of possible treatment-related immune-mediated effects were reevaluated, specifically to assess the number and character of mononuclear foci in the brain. The findings of the review are compared with the original diagnoses in the pathology reports (Table 2). The review uses the statistical approach described in the Method section whereas the pathology report is the judgment of the study pathologist. The choroid plexus was the tissue most frequently affected, and the review identified additional cell types in studies where this was not recorded by the study pathologist. The meninges and eyes were less frequently affected (3/8 studies each). The review confirmed the study pathologist’s findings in most cases and identified an additional study with a test article–related effect in one extra study for both the meninges and eyes. Instances where the review failed to conclude a test article effect was due to some slides containing foci that were not examined in the reevaluation, in order to examine the same number of slides per animal for each study.
Studies with Test Article–related Effects on Mononuclear Cell Infiltrates—Results of Reevaluation versus Original Findings.
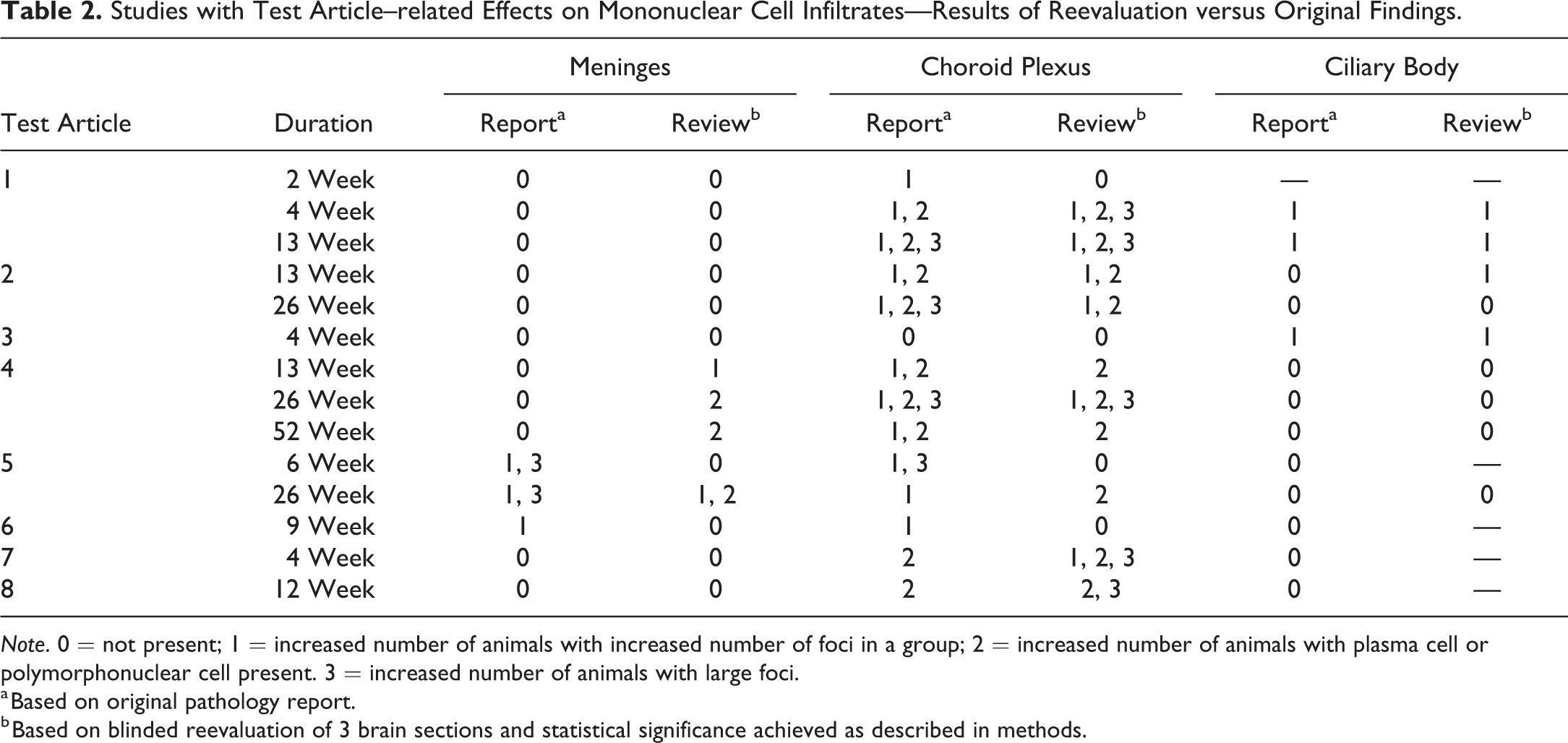
Note. 0 = not present; 1 = increased number of animals with increased number of foci in a group; 2 = increased number of animals with plasma cell or polymorphonuclear cell present. 3 = increased number of animals with large foci.
a Based on original pathology report.
b Based on blinded reevaluation of 3 brain sections and statistical significance achieved as described in methods.
At the 5% significance level, no control group yielded a significant result. All but two test articles resulted in statistically significant increases in at least one treated group. The exceptions all had single animals that had large or high numbers of mononuclear cell foci that stood out as sufficiently unusual for the study pathologist to mention in the report (Table 2). However, the presence in only one animal was insufficient to yield a statistically significant effect in the reevaluation.
Cell Types within Mononuclear Foci in the Meninges and Choroid Plexus
All foci were composed primarily or entirely of lymphocytes. It was observed during the primary read of some studies with test article–related increases in the number or severity of mononuclear cell foci, that many foci contained a proportion of plasma cells and occasionally eosinophils, particularly those in the choroid plexus (Figure 1b–d). During the reevaluation of these studies, the presence of these cell types was recorded (Tables 3 and 4). Of the animals with foci, a small minority had plasma cells within meningeal foci, but the proportion was higher in treated animals (12.7%) than controls (4.9%). In contrast, most treated animals with choroid plexus foci had plasma cells (67%) compared with 0% of controls. Eosinophils were only observed in treated animals, with a greater proportion in the choroid plexus (18.2%) than meninges (6.3%). Another contrast between the meninges and the choroid plexus was a strong association between the presence of eosinophils and plasma cells in the choroid plexus (9/10 animals with plasma cells also had eosinophils). Also, neutrophils were observed in meningeal foci in a small proportion of both control and treated animals; however, none were recorded in the choroid plexus foci.
Number of Animals with Foci Containing Cells in Addition to Lymphocytes.

Percentage of Animals with Foci, with Foci Containing Cells in Addition to Lymphocytes.
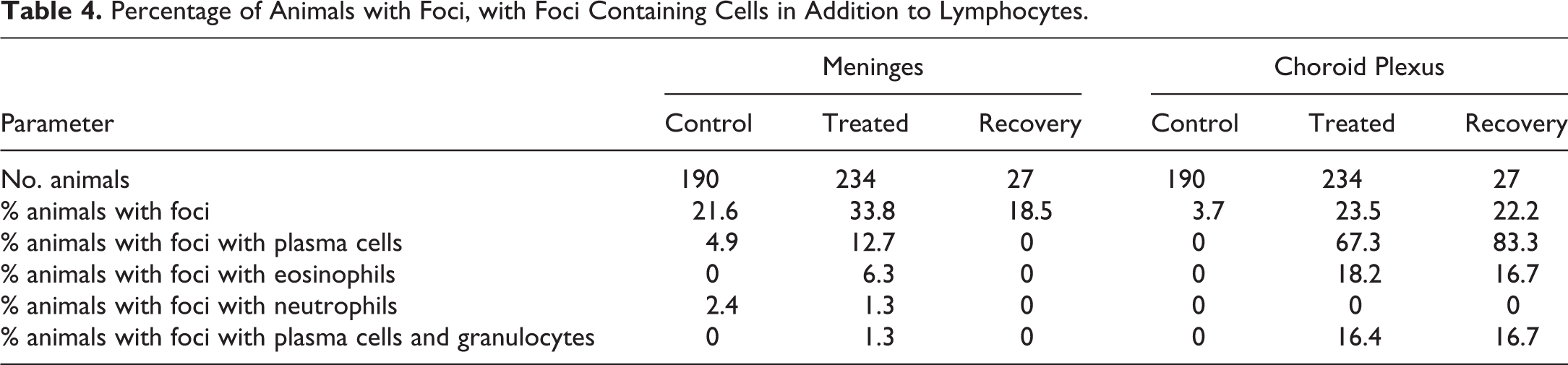
In recovery animals, a lower proportion of animals had meningeal foci suggesting partial resolution. In the choroid plexus however, all parameters were similar between terminal and recovery animals. There were two studies with recovery animals with increases in meningeal foci (one with an 8 week and the other with a 13-week recovery period), whereas there were four studies with recovery animals with increases in choroid plexus foci (three with an 8 week and one with a 13-week recovery period).
Test Article–related Increases in Mononuclear Cell Infiltrates in the Ciliary Body of the Eye
Applying a similar approach to that used for mononuclear foci in the brain, a threshold for the number of ciliary bodies within a section containing infiltrates was established using control animals. Only 4/90 controls had 2 or more foci. Data were analyzed using Bernoulli trials, as described for the brain.
Nine studies, testing five test articles were evaluated. At the 5% significance level, no control group yielded a significant result. Three test articles had statistically significant increases in at least one treated group (Table 2).
Discussion
The primary aim of this study was to demonstrate the association between administration of biotherapeutics to the NHP species most commonly used in safety assessment and mononuclear cell infiltrates in the brain and eye. This is to provide study and peer reviewing pathologists, toxicologists, and regulators, with evidence to assist in decision-making with respect to whether a trend observed in a particular study is likely to be real, often a problem with the small group sizes used in toxicology safety assessment in NHPs, and to provide reassurance that this occurs in a significant proportion of test articles, with classification as a biotherapeutic being the only common factor, rather than the intended pharmacological target. The absence of any observed adverse effects in these studies related to the findings also provides reassurance that these are not predictive of risk to humans, especially considering that the molecules are likely to be more “foreign” to the simian immune system than to the intended human recipients of these molecules.
Typical of immune-mediated effects, these findings were present in only certain individuals within a study with little evidence of a dosage relationship. Of note was the observation that findings often persisted after a recovery period.
In order to be consistent within the cohort of studies selected, only three brain sections were assessed in the reexamination of sections. More recently, the additional sections taken with the Pardo method (Pardo et al. 2012) have increased the amount of brain tissues evaluated, in particular the amount of choroid plexus, and therefore the likelihood of sampling a section containing mononuclear cell infiltrates. In addition, the number of toxicology studies using biotherapeutics has increased in recent years. The authors have been aware of observing control animals with large foci, sometimes containing plasma cells or eosinophils in the choroid plexus. Although the incidence over recent studies has not been calculated, it is highly likely that it exceeds the incidence reported in the studies reexamined for the present article, examining only three sections.
The presence of eosinophils and plasma cells has been proposed to be an indicator of an induced lesion. However, the authors have observed rare foci with such cells in control animals with (perceived) greater frequency recently, as discussed above. It is possible that the presence of such cells and the size of a focus are both related to the local inflammatory stimulus, so the association with biotherapeutic administration is secondary to increasing this stimulus. The presence of multinucleated giant cells was not reported in the reviewed studies or observed in the reevaluated tissues.
The precise immunological mechanism resulting in the increased incidence of mononuclear cell foci at these sites has not been established. Sites known to be predisposed to immune complex deposition include the choroid plexus and ciliary body, and the presence of ADAs is commonly demonstrated in response to biotherapeutic administration. Immune complex deposition in the choroid plexus has previously been demonstrated by immunohistochemistry following biotherapeutic administration (Rojko et al. 2014). As the choroid plexus is a site of entry for immune cells into the CNS (Ghersi-Egea et al. 2018; Kunis et al. 2013; Meeker et al. 2012; Strazielle et al. 2016), it is also plausible that general immune-stimulation resulting from biotherapeutic administration increases the rate of activated lymphocyte trafficking through sites of active immunosurveillance (Young et al. 2011) and/or the responsiveness of infiltrating lymphocytes to local immunostimulatory signals.
Despite the choroid plexus being a point of entry into CNS for leukocytes in inflammatory conditions (Demeestere, Libert, and Vandenbroucke 2015; Kipnis 2016), there was no evidence that the lesions observed in these studies were associated with inflammation within the CNS and were therefore not considered adverse.
The data presented here provide evidence to support the view that a significant proportion of biotherapeutics result in cellular infiltrations of lymphocytes, plasma cells, and eosinophils at specific sites where such cells are infrequently observed in controls. Within a specific study, there may be uncertainty regarding whether the presence of such findings can be attributed to a test article within a specific animal or dose group, but this study provides reassurance that this represents a generic class effect rather than a specific test article effect.
Footnotes
Acknowledgment
The authors would like to thank Marion Thom for her advice and reassurance with respect to the mathematical aspects of this article.
Author Contributions
Authors contributed to conception or design (SN, MCza, AB, MCar), data acquisition, analysis, or interpretation (SN, MCza, WH, MS, AB, MCar); drafting the manuscript (SN); and critically revising the manuscript (MCza, WH, MS, AB, MCar). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
