Abstract
Benzonatate is a peripheral oral antitussive that dampens the activity of cough stretch receptors. Rodent carcinogenicity studies were performed in Tg.rasH2 mice and Wistar Han rats. Mice were orally gavaged benzonatate at 10, 30, 75, and 100 mg/kg/day for males and 5, 15, and 50 mg/kg/day for females. Rats were gavaged at 10, 30, and 90 mg/kg/day for males and 5, 15, and 50 mg/kg/day for females. Higher doses in males were due to differences in maximum tolerated doses in dose-ranging studies. In both species, benzonatate was not detected in plasma because of rapid ester hydrolysis producing 4-(butylamino) benzoic acid (BBA) and methylated polyethylene glycol polymer. This metabolism was similar in human plasma; therefore, plasma BBA was used to show systemic exposure. Both species had no evidence of a benzonatate-related increase in any neoplasm. A slight increase in nasal cavity exudative inflammation was present in benzonatate-dosed male mice. Retinal atrophy was observed in male rats at ≥30 mg/kg/day, but the incidence was within historical control data range and not related to benzonatate. In conclusion, benzonatate and its 2 major metabolites were not carcinogenic in rodent carcinogenicity studies at BBA exposures of ≥32 and 70 times a 200 mg human benzonatate dose, respectively.
Benzonatate (CAS # 104-31-4; TESSALON®) is a nonnarcotic, peripherally acting, oral prescription antitussive indicated for the symptomatic relief of cough. It was approved in the United States in 1958. The recommended dosage is 100 or 200 mg 3 times a day in adults and children over 10 years of age (Tessalon 2017). Benzonatate consists of a mixture of 15 structural analogues/oligomers that differ in the number of the repeating side chain (OCH2CH2) n , where n = 7 to 50 (Tessalon 2017). Its chemical structure is related to the ester class of local anesthetic agents such as procaine and tetracaine (Figure 1). Benzonatate mechanism of action is through its peripheral anesthetic dampening the activity of pulmonary stretch receptors located in respiratory passages, lungs, and pleura, which mediate the cough reflex (Bucher 1956; Langrehr 1963; Dicpinigaitis et al. 2014). It also has similar activity on aortic baroreceptors, atrial and vagal receptors. Central nervous activity has also been reported (Guffanti 1989). Unlike the narcotics, benzonatate has no inhibitory action in the respiratory center rendering it a better safety profile. Just like the local anesthetics, benzonatate is prone to ester hydrolysis producing the N-butyl substituted metabolite, 4-(butylamino) benzoic acid (BBA), and methylated polyethylene glycol polymer (MPG; Figure 2; Lin, Chen, and Suen 1996).
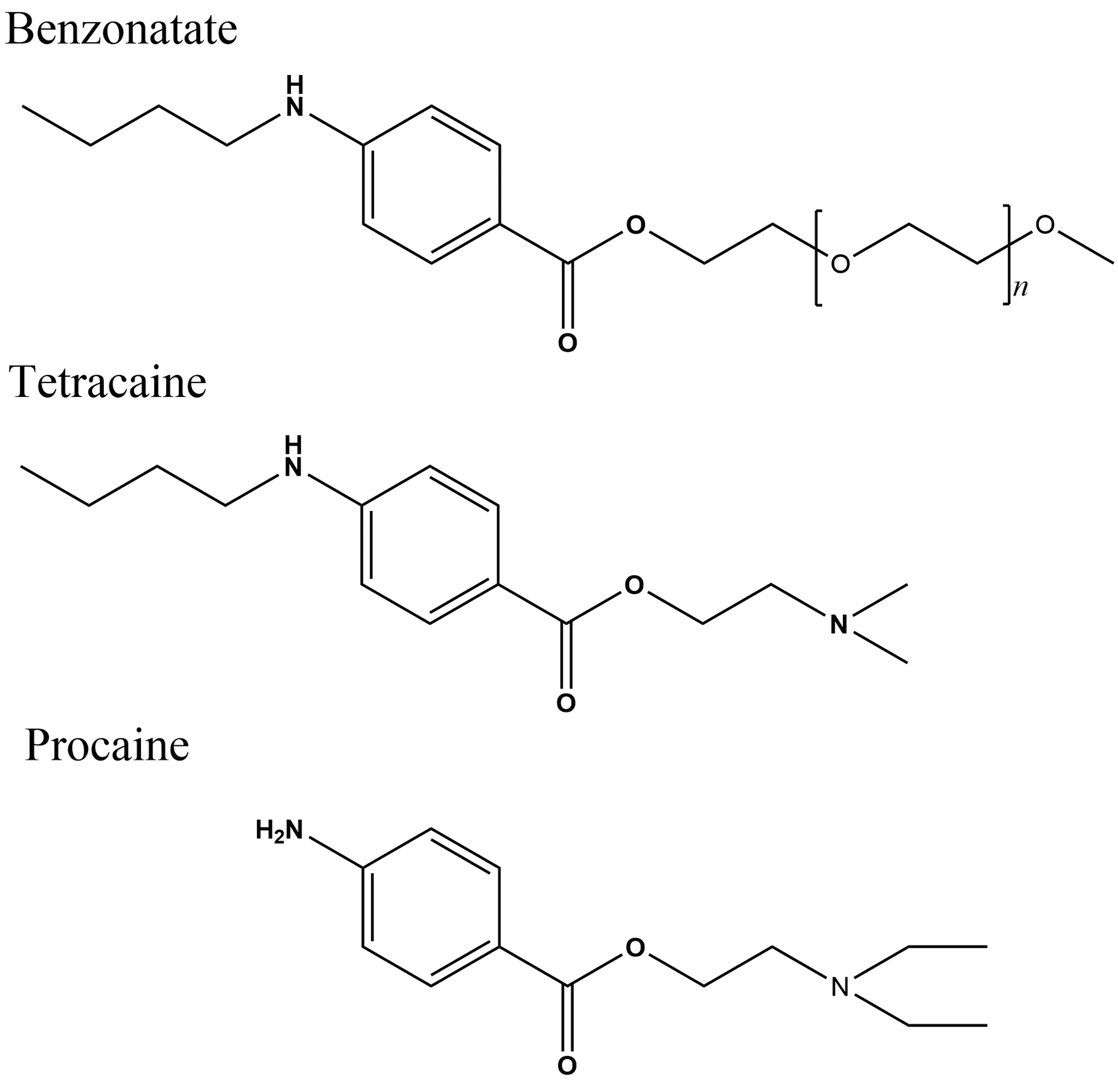
Structures of benzonatate, tetracaine, and procaine.
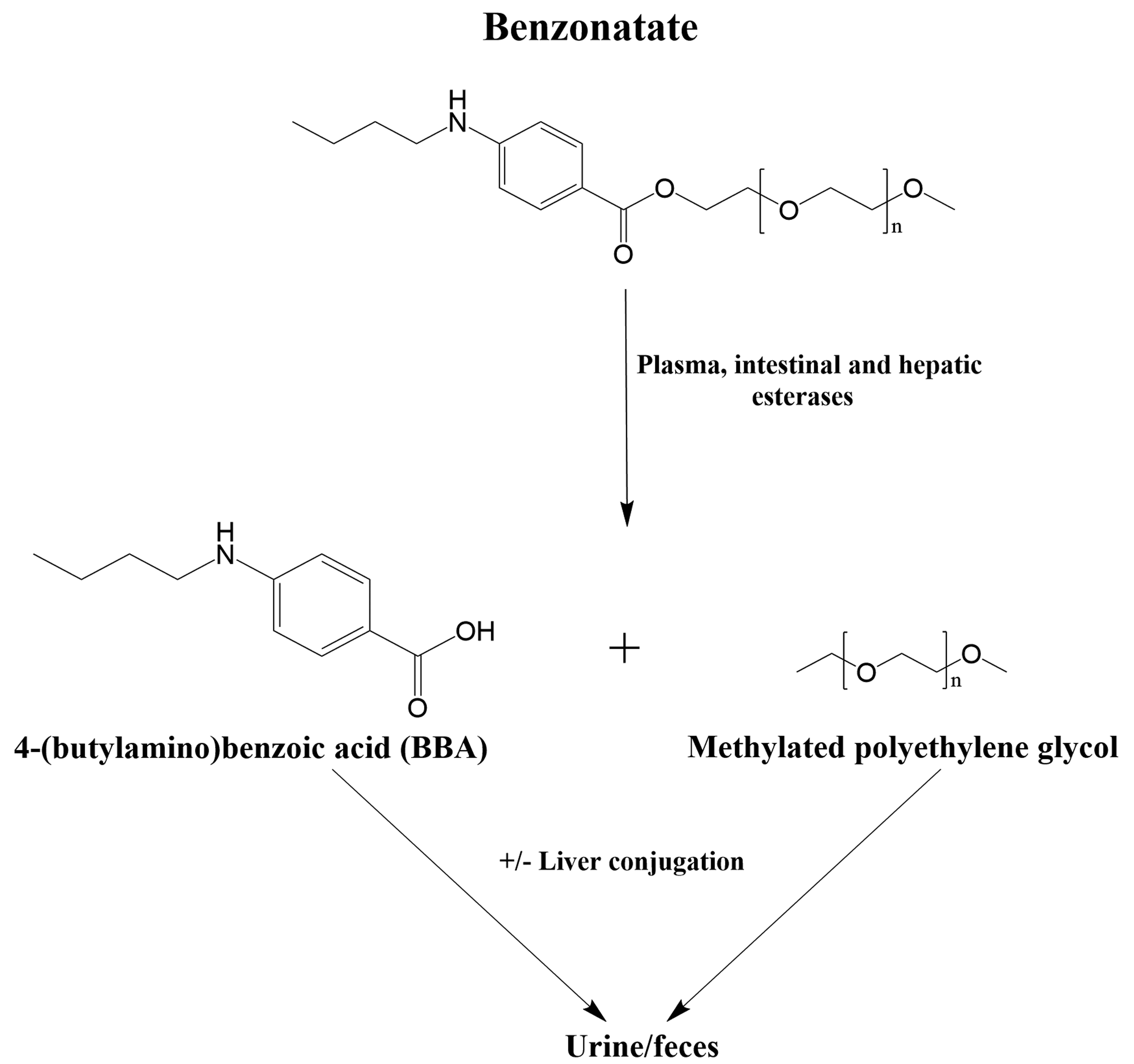
Benzonatate metabolism.
No new nonclinical data have been generated since its approval. To add to the nonclinical safety database, carcinogenicity studies were performed to evaluate the tumorigenic potential of benzonatate following daily oral administration to transgenic Tg.rasH2 mice for 6 months and Wistar Han rats for 24 months (ICH S1B 1997; ICH S1C(R2) 2008). Genotoxicity and reproductive toxicity studies have also been performed. All these data will be used to update the product label.
Material and Method
Test Article, Vehicle, and Positive Control Material
Benzonatate (2, 5, 8, 11, 14, 17, 20, 23, 26-nonaoxaoctacosan-28-yl p-(butyl amino) benzoate; CAS number 104-31-4; purity 99.6%), a light yellow viscous colorless liquid (BASF, Florham Park, NJ), was the test article in both studies. The vehicle for both studies was deionized reverse osmosis water (pH 4–6). Urethane was obtained from Sigma-Aldrich (St. Louis, MO) and was used as a positive control material in the Tg.rasH2 study.
Dosing Formulations
Benzonatate in deionized water dosing formulations were prepared biweekly for both studies. Analysis demonstrated that they were within 94.8% to106% of the target concentrations with 0.05% to 3.82% relative standard deviation. The formulations were stable for at least 16 days under refrigeration (2–8°C).
LC-MS/MS Plasma Assays
Mouse and rat plasma LC-MS/MS assays for benzonatate and BBA (CAS # 4740-24-3) were developed and validated at Celerion Inc. (Lincoln, NE; manuscript in preparation). The method involved protein precipitation extraction and analysis by an HPLC equipped with an AB SCIEX API 4000 triple quadrupole mass detector with an electrospray ionization source. Internal standards were d9-benzonatate and d9-BBA. Sodium EDTA was the anticoagulant and NaF the preservative. Both species’ limits of quantitation (LOD) were 67 and 500 ng/ml for benzonatate and BBA, respectively. Plasma level of the polymer metabolite MPG was not determined.
Six-month Tg.rasH2 Mouse Study
The Tg.rasH2 mouse study was conducted at MilliporeSigma BioReliance® Toxicology Services (formerly BioReliance, Rockville, MD) according to Good Laboratory Practice (GLP) regulations (CFR 2017). The mouse carries the human c-Ha-ras proto oncogene and is an acceptable rodent species for carcinogenicity assay (CHMP Safety Working Party 2005).
Doses for the 6-month study were based on a 28-day dose-range finding study in nontransgenic CByB6F1 wild-type mice (Taconic Farms, Germantown, MD). Mice were dosed by oral gavage at 0, 50, 100, and 350 mg/kg/day with benzonatate-related mortality observed at ≥100 mg/kg/day. The highest dose was reduced to 250 mg/kg/day due to mortality and the study continued. A total of 28 mice died in the study with 22 from the toxicokinetic (TK) dose groups. This included 3 female mice (one Main, 2 TK) that died at the 100 mg/kg/day group. TK blood samples (predose; 10, 20, and 30 min; 1, 2, 4, and 6 hr) were taken at days 1 and 25 postdose from the retro-orbital sinus. Significant clinical findings included hunched posture, decreased motor activity, and rapid shallow breathing with increasing dose. There were no significant changes in body weight, food consumption, and clinical pathology. Exudative inflammatory lesions in the nasal cavity were the primary lesions and observed at ≥100 mg/kg/day. Based on the nasal cavity lesions and mortality, the no-observed-adverse-effect level for the dose-ranging study was 50 mg/kg/day for both sexes. The U.S. Food and Drug Administration’s Executive Carcinogenicity Assessment Committee (eCAC) recommended doses of 0, 10, 30, 75, and 100 mg/kg for males and 0, 5, 15, and 50 mg/kg for females for the 6-month study (Table 1). The high dose of 50 mg/kg/day for females was selected due to mortality at 100 mg/kg/day in the dose-ranging study.
Dosages and Groups for 6-month Transgenic Mouse Study.
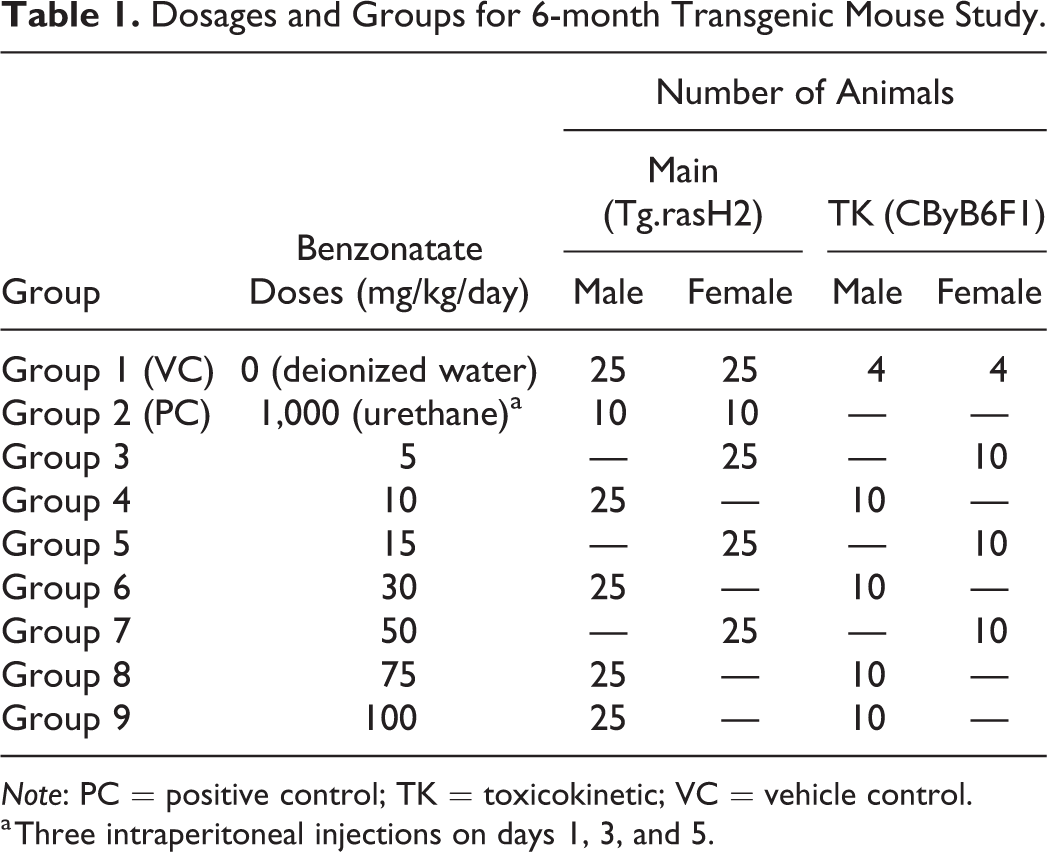
Note: PC = positive control; TK = toxicokinetic; VC = vehicle control.
a Three intraperitoneal injections on days 1, 3, and 5.
The 6-month study Tg.rasH2 (main) and CByB6F1 (TK) mice were housed in an environmentally controlled room with continuous recording of room temperatures of 69°F to 75°F and relative humidity of 30% to 70%, with a 12-hr light/12-hr dark cycle. Mice were provided rodent diet (TEKLAD Global Diet #2018CM, Envigo [formerly Harlan], Madison, WI) and water ad libitum. Animals (7–8 weeks old) were randomized into groups according to body weight (Provantis™ Version 8.2.0.8, Instem, Conshohocken, PA). The vehicle and positive control were deionized reverse osmosis water and urethane, respectively. Benzonatate was formulated in deionized reverse osmosis water. Urethane was formulated in saline and administered intraperitoneally at 1,000 mg/kg/day on days 1, 3, and 5. Mice in the main and TK groups were orally gavaged (10 ml/kg) once daily for 6 months (up to 183 days) and 14 weeks, respectively (Table 1). Clinical observations, body weight, and food consumption were determined. A sparse number of TK blood samples (10, 20, and 60 min postdose) were taken at week 14 to show exposure. All animals of main study were euthanized by CO2 inhalation and necropsied. Macroscopic observations, organ weights, and histopathology were assessed by a board-certified anatomical pathologist and peer reviewed by another board-certified anatomical pathologist. Incidence of lesions was compared to the historical control range established at MilliporeSigma BioReliance.
The incidence of any toxicological effects was calculated separately by sex and dose; numerical data, means, and standard deviations were calculated for each sex and group. Analysis of variance tests were conducted on body weight, food consumption, and organ weight data. If a significant F ratio was obtained (p < .05), the Dunnett’s t test was used for pairwise comparisons using Provantis® (Tables and Statistics Version 8.4.0.1; Instem). The incidence of tumors were analyzed by sex, using Peto’s mortality prevalence method without continuity correction, incorporating the context (incidental, fatal, or mortality independent) in which the tumors were observed (FDA Guidance 2001; Peto, Pike, and Day 1980). The following fixed intervals were used for incidental tumor analyses: day 1 to end of study (up to, but not including, scheduled termination) and scheduled termination. All tumors in the scheduled termination interval were considered incidental for the purpose of statistical analysis. Tumors classified as mortality-independent were analyzed with Peto’s mortality independent method incorporating the day of detection. Each diagnosed tumor type was analyzed separately, and at the discretion of the pathologist, analysis of combined tumor type and/or organs was performed. Any metastases and invasive tumors were considered secondary and not included in the analysis. A one-sided comparison of each benzonatate group with the vehicle control was performed. An exact permutation test was conducted for analyses with low tumor incidence. Findings were evaluated for statistical significance at both the .01 and .05 probability levels. For tumor and mortality data, statistics were conducted using Statistical Analysis System® (SAS Institute Inc., Cary, NC).
Two-Year Wistar Han Rat Study
Rat studies were performed at Covance Laboratories (Madison, WI) according to GLP regulations (CFR 2017). Doses for the 2-year study were based on a 3-month dose-ranging study in Wistar Han rats (Charles River Laboratories, Raleigh, NC). Rats were administered benzonatate by oral gavage at 0, 20, 50, and 90 mg/kg/day. There was no mortality in the 3-month study. Based on feedback from the eCAC, doses of 0, 10, 30, and 90 mg/kg for males and 0, 5, 15, and 50 mg/kg for females were selected due to sex differences in the maximum tolerated dose (significantly lower mean body weight and body weight gain at 90 mg/kg/day in females; Table 2).
Dosages and Groups for 2-year Rat Study.
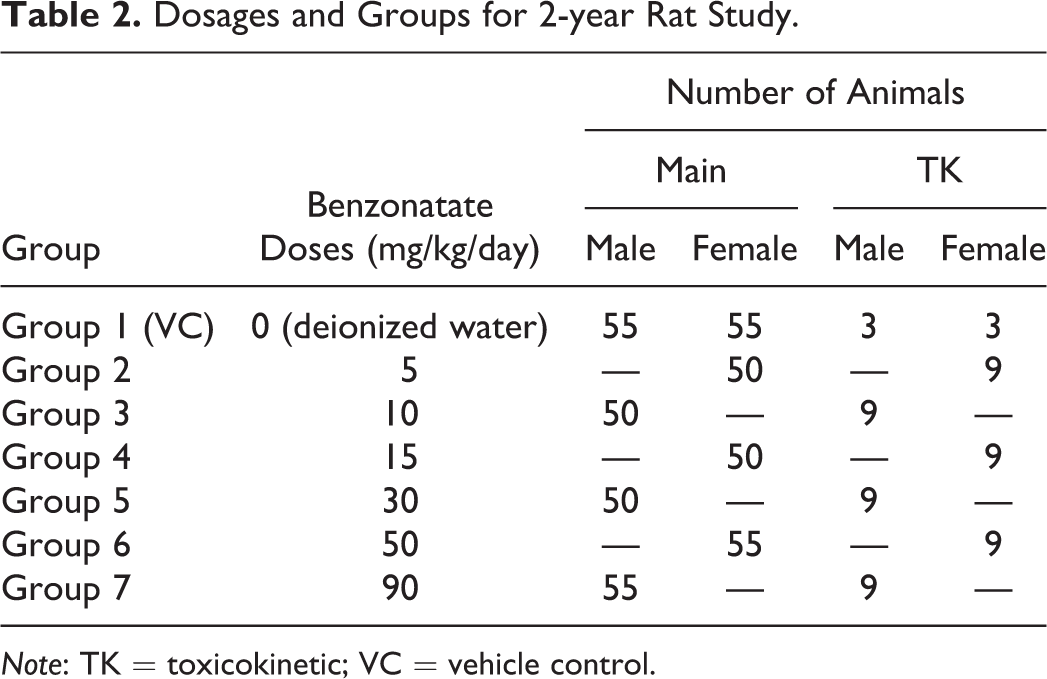
Note: TK = toxicokinetic; VC = vehicle control.
The 2-year study used 6- to 7-week-old Wistar Han rats. Housing, food and randomization, and dose formulation were as described for the mouse study except that a different rodent diet (TEKLAD Global Diet #2016C, Envigo [formerly Harlan]) was used. Rats in the main and TK groups were orally gavaged (5 ml/kg) once daily for 2 years and 48 weeks, respectively (Table 2). Similar measurements were taken as for the mouse study except that complete TK blood sample profiles (predose; 5, 15, and 30 min; 1, 2, 4, 8, and 12 hr) were taken at week 26 (5 min to 12 hr time points), and ophthalmic exams were performed at predose and week 52. All animals of main study were euthanized by intravenous sodium pentobarbital and necropsied. Macroscopic observations, organ weights, and histopathology were assessed by a board-certified pathologist and peer reviewed by another board-certified pathologist. Incidence of lesions was compared to the historical control range established at Covance Laboratories. Statistical analyses were as described for the mouse study.
Results
Six-month Tg.rasH2 Mouse Study
Mortality and survival
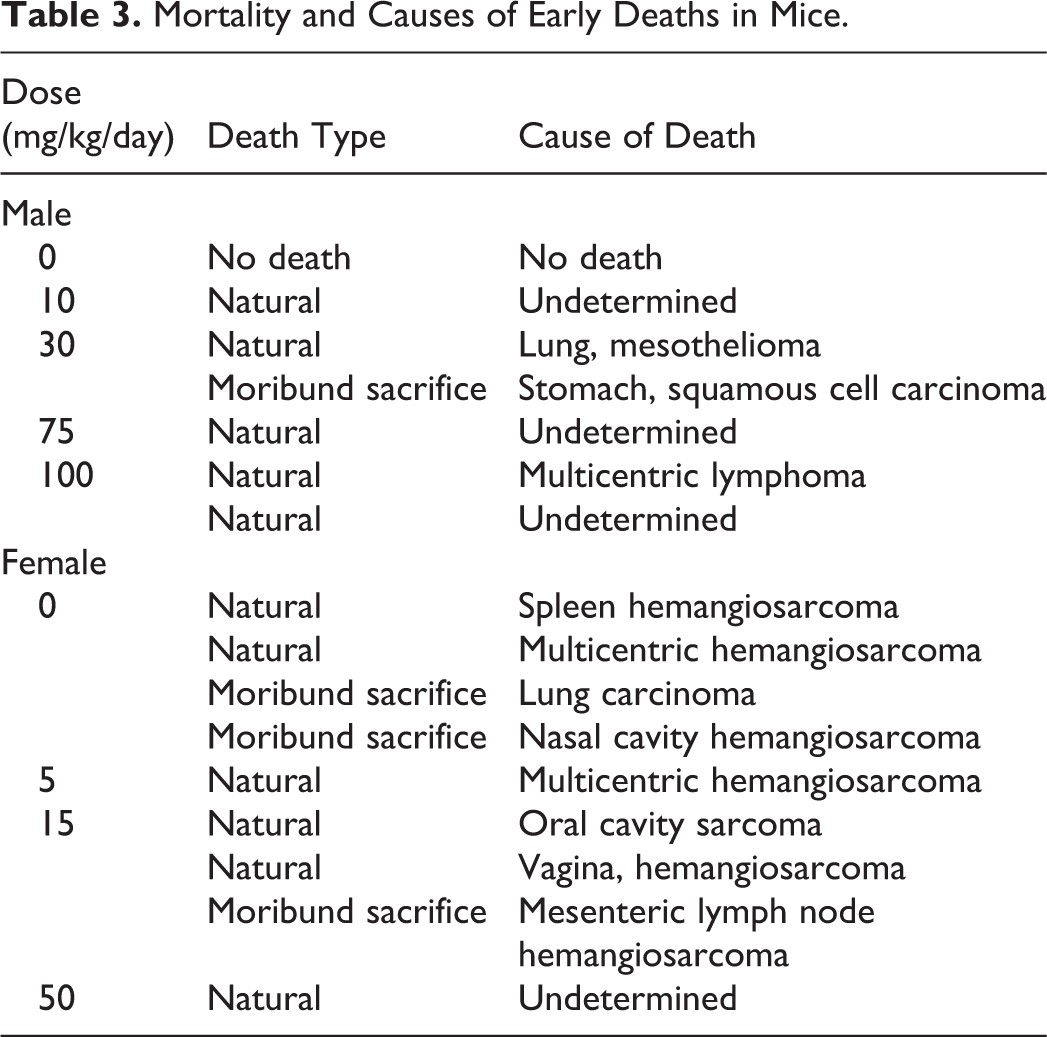
A total of 15 of the 245 animals from the main study dose groups either died or were electively euthanized before the end of the dosing period (Table 3). Mortality in benzonatate-treated groups was not statistically significantly different compared to the vehicle control group. Of the early deaths, 6 were males at doses of 10 (1 mouse), 30 (2), 75 (1), and 100 (2) mg/kg and 9 females at 0 (4), 5 (1), 15 (3), and 50 (1) mg/kg. While all male mice were from benzonatate-treated groups, 4 female mice were from the vehicle group. None of these deaths were considered benzonatate-related due to a lack of a dose response in the mortality and tumor incidences. Survival was above 90% for all benzonatate dose groups at the last day of dosing (Figure 3). The positive control group was euthanized on days 102 and 100 for males and females, respectively, due to the expected early formation of urethane-induced lung nodules (Shah et al. 2012).
Mortality and Causes of Early Deaths in Mice.
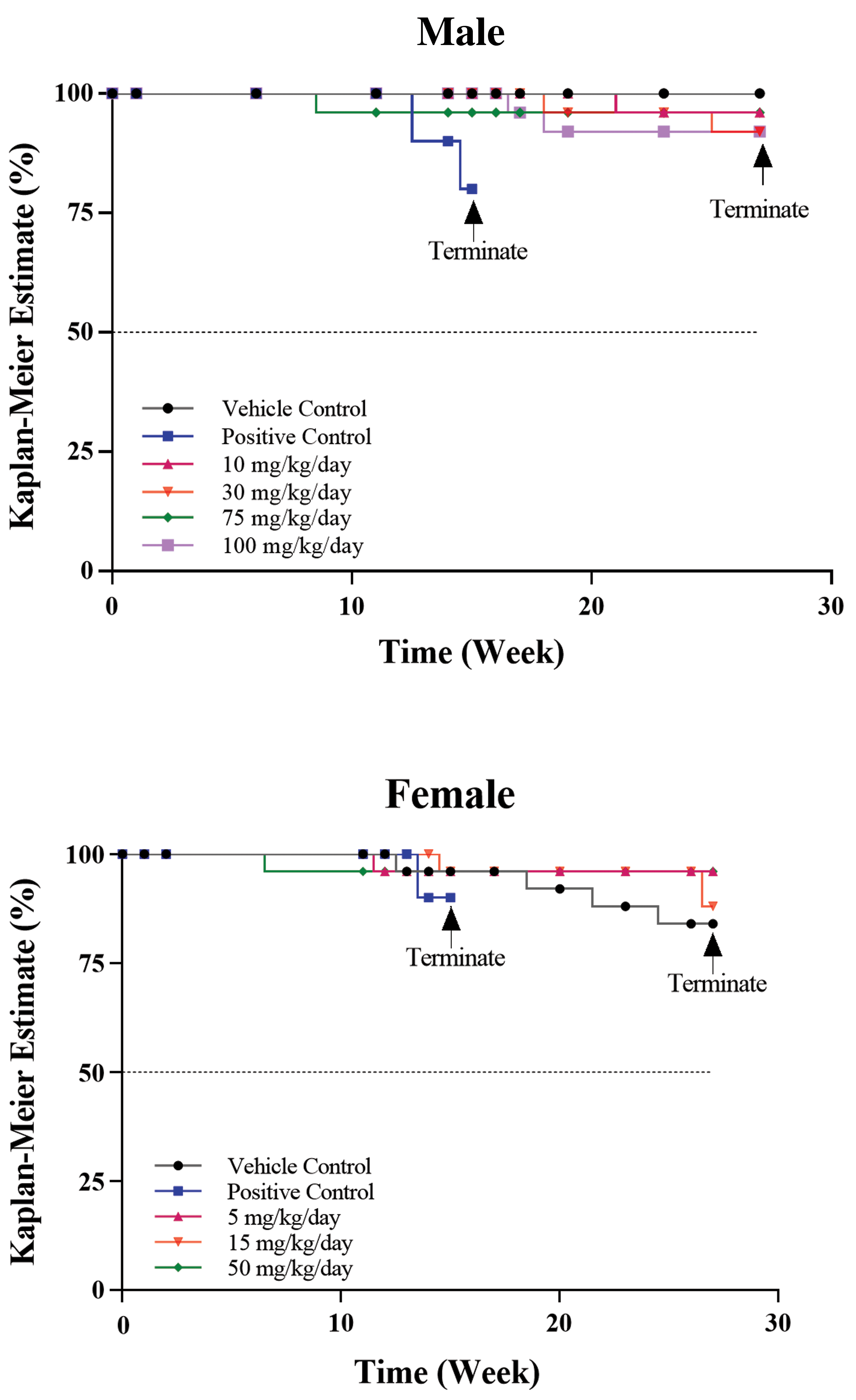
Kaplan–Meier survival plots from mouse study.
Body weight and food consumption
There were no statistically significant differences in the group mean body weights, body weight gains, or food consumption between the control and treated dose groups (Figure 4).
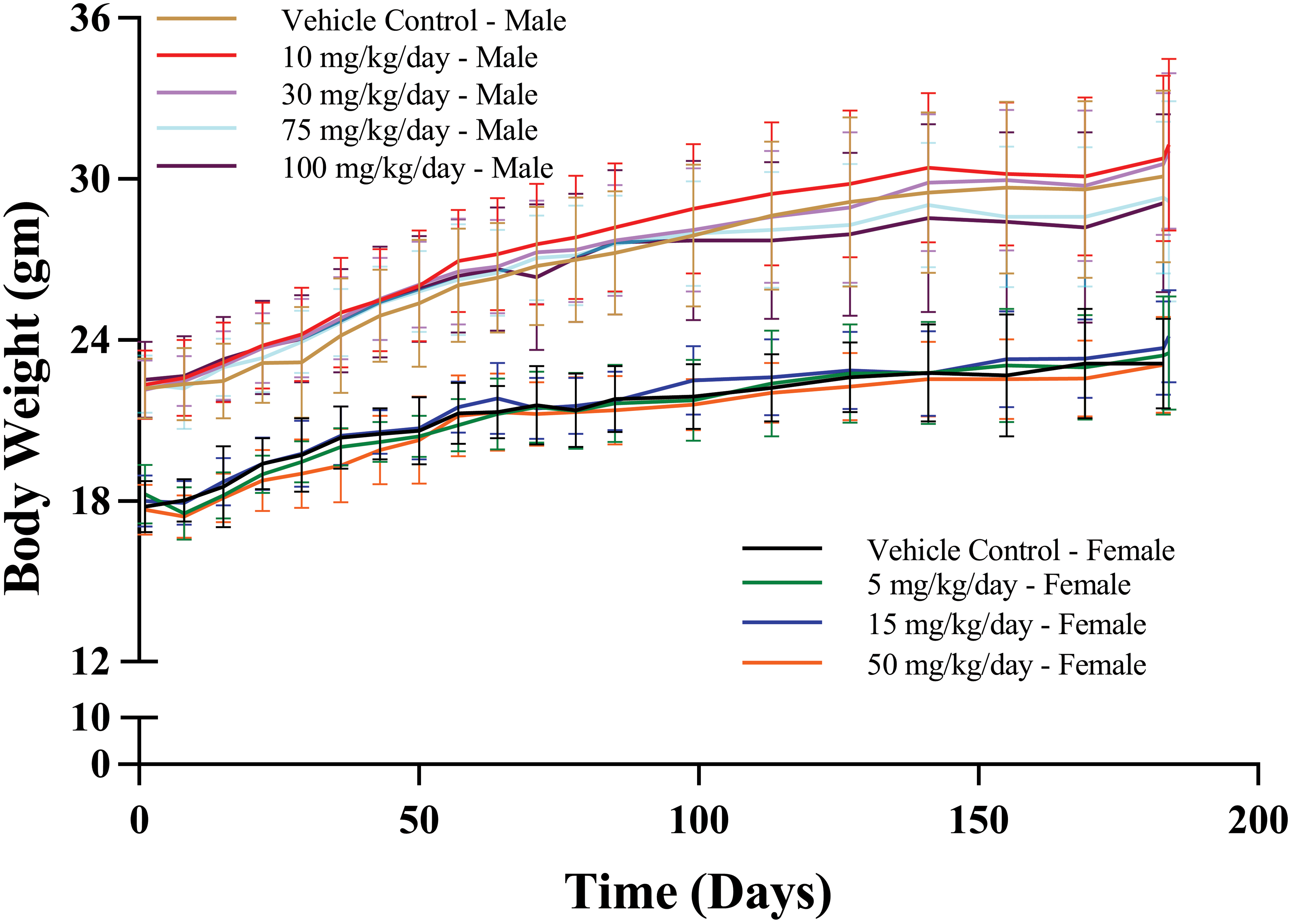
Body weights of mice over time.
Macroscopic findings
The macroscopic findings were consistent with this mouse strain. Benzonatate did not produce any dose-related increase in macroscopic findings. As expected, there was increased incidence of pulmonary tumors in urethane-treated mice.
Microscopic findings
Hyperplastic/neoplastic lesions
Urethane-treated mice had statistically significant increases in the incidence of pulmonary tumors (adenomas, carcinomas; Table 4) and splenic hemangiosarcomas (Table 5) compared to control mice. The overall incidences were within the MilliporeSigma BioReliance historical positive control range of 80–90% (data not shown), thereby validating the study and the transgenic mouse model (Shah et al. 2012). Males administered 30 mg/kg/day benzonatate had statistically significant increased incidences of splenic hemangiosarcomas and combined hemangiosarcomas (Table 5). Even though statistically significant, the incidence was within the historical control range but at the highest end of it. This increase was not considered to be benzonatate-related because (a) the incidence of splenic hemangiosarcomas fell within the historical control range established at MilliporeSigma BioReliance in all dose groups (Paranjpe et al. 2013), (b) it was not dose dependent, and therefore, there was no observed trend for an increase, (c) there were zero splenic hemangiosarcomas in the vehicle control males, which is at the lowest end of the historical control range, and (d) the statistical significance was affected by 2 early deaths (considered incidental to treatment) at this dose compared to no mortality in the vehicle control males. Thus, these neoplasms even though statistically significant were not considered to be biologically and toxicologically significant and therefore not benzonatate related. No significant increases in any other nonvascular and nonpulmonary tumor types were noted in benzonatate-treated mice of both sexes (supplementary mouse pathology report).
Number of Mice with Pulmonary Tumors.
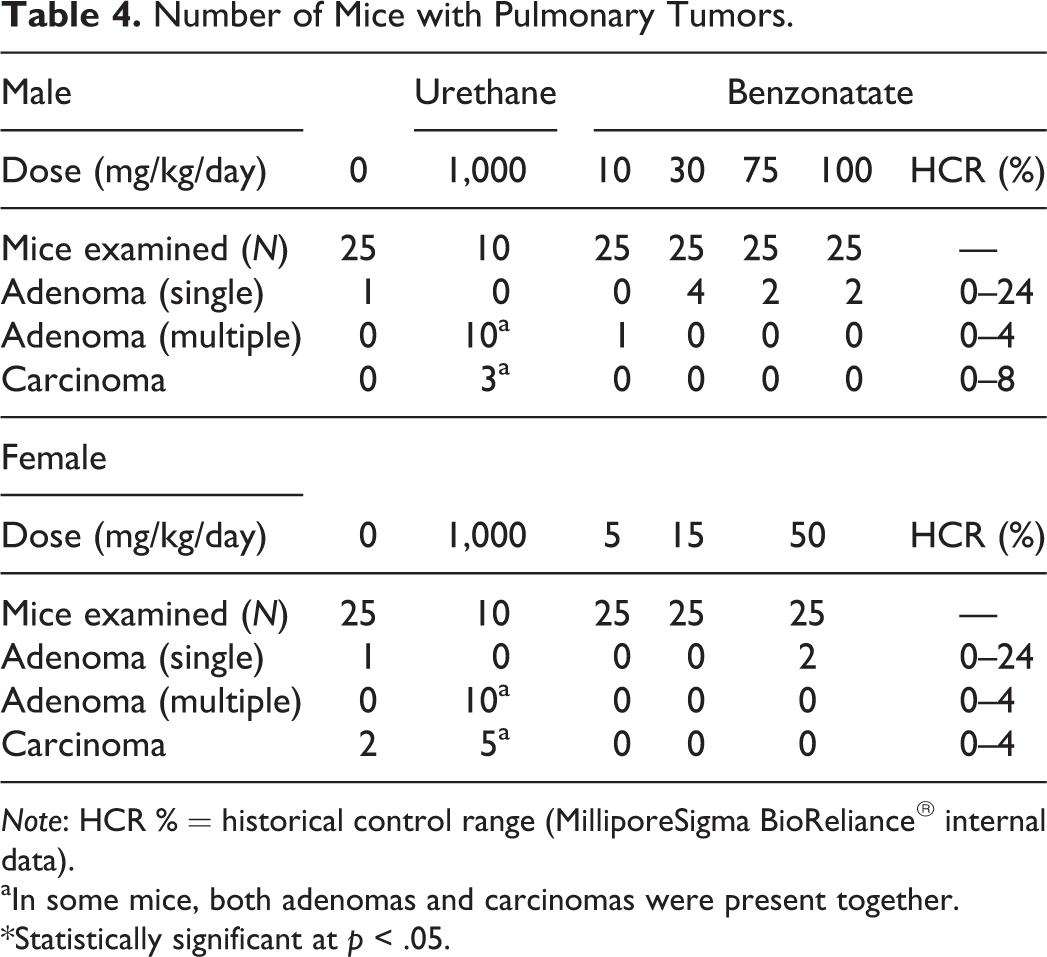
Note: HCR % = historical control range (MilliporeSigma BioReliance® internal data).
aIn some mice, both adenomas and carcinomas were present together. *Statistically significant at p < .05.
Number of Mice with Splenic Hemangiosarcomas.
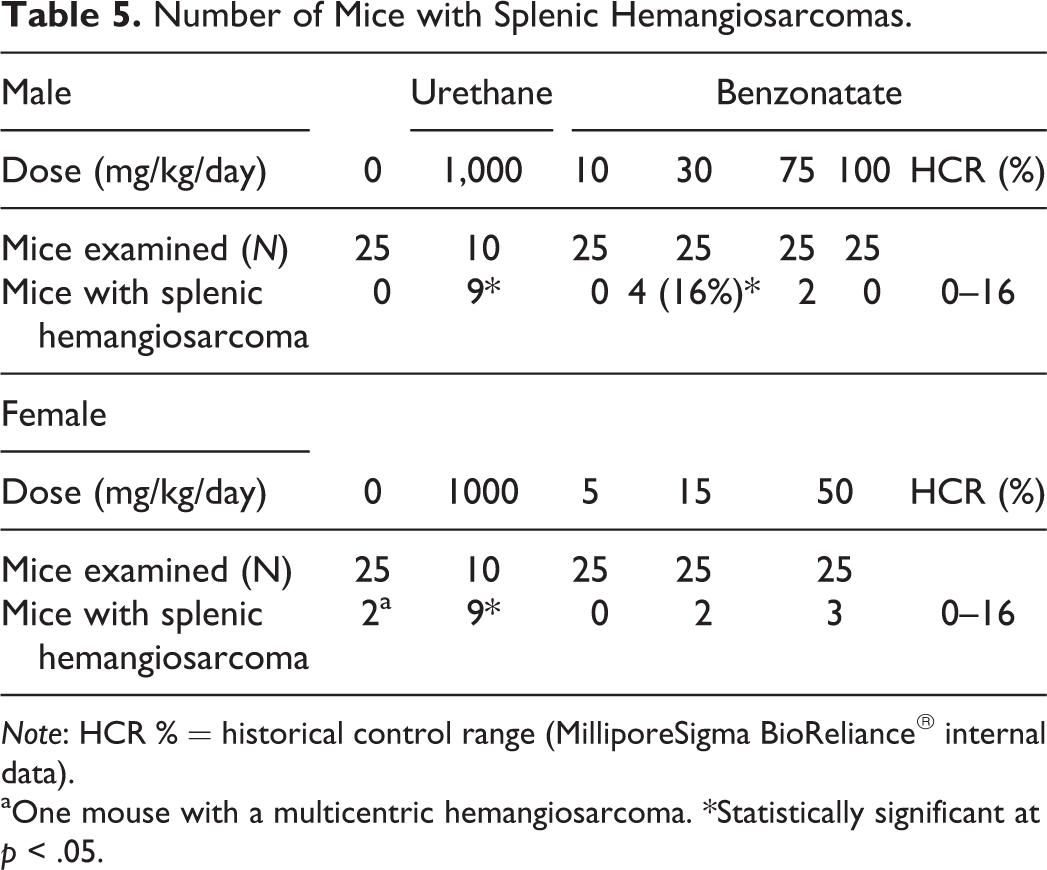
Note: HCR % = historical control range (MilliporeSigma BioReliance® internal data).
aOne mouse with a multicentric hemangiosarcoma. *Statistically significant at p < .05.
Nonneoplastic lesions
The only benzonatate-related nonneoplastic finding was exudative inflammation of the nasal cavity in males. This was characterized by the presence of eosinophilic protein-rich fluid and sparse basophilic mucin within the turbinates with degenerate neutrophils and necrotic debris. The incidence was within the historical control range (Table 6). While nasal inflammation is not a common background lesion, when it occurs, it is often caused by the vehicle alone and/or combination of the vehicle and test article (Paranjpe et al. 2017). In females of this study, the severity and incidence were similar in all groups, including the controls. Based on the 28-day and current study, the nasal lesions were considered to be induced by the deionized water control vehicle with a pH of 4 to 6, gastric reflux, and exacerbation by benzonatate in males but not females. Exudative inflammatory lesions are commonly observed in the nasal cavity of Tg.rasH2 mice as a result of low pH and irritant nature of the vehicle and/or gastric reflux. The pH of the benzonatate dosing formulations however was not determined. There is no evidence of these lesions leading to tumors in the nasal cavity (Paranjpe et al. 2017). Other nonneoplastic lesions were few, sporadic among the groups and not considered preneoplastic or benzonatate related (data not shown).
Number of Mice with Benzonatate-related Nonneoplastic Nasal Cavity Lesions.
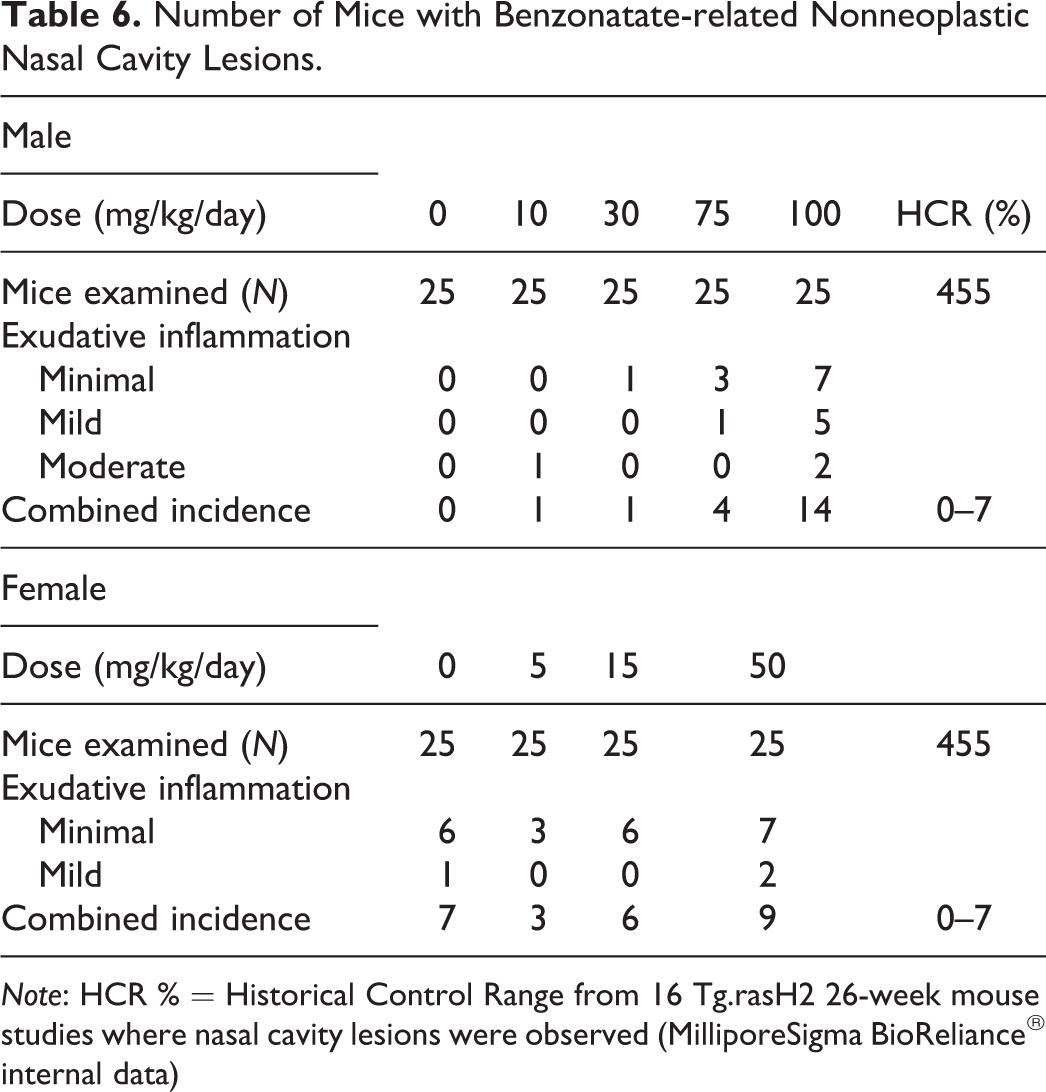
Note: HCR % = Historical Control Range from 16 Tg.rasH2 26-week mouse studies where nasal cavity lesions were observed (MilliporeSigma BioReliance® internal data)
Exposure and safety margins
Benzonatate exposure (AUC0-24) could not be determined in the dose-ranging and carcinogenicity studies, as plasma levels were mostly below the LOD of 67 ng/ml at all doses and time points due to extensive ester hydrolysis. The major metabolite BBA was detected in all treated groups and was therefore used as a surrogate for exposure. In the 28-day dose-ranging study at 100 mg/kg/day for male and 50 mg/kg/day for female, the free unbound plasma BBA exposures and safety margins were at least 60 and 30 times that of a 200 mg human dose, respectively (Table 7). Safety margins would be the same for the carcinogenicity study, as the sparsely sampled (10, 20, and 60 min) mean plasma levels at the 2 doses were similar to those from the dose-ranging studies. Exposure (AUC0-24) to BBA at day 25 of the dose-ranging study was dose proportional from 50 to 10 mg/kg and subproportional from 100 to 250 mg/kg (Table 7).
Plasma BBA Exposure and Safety Margins for Mouse and Rat.
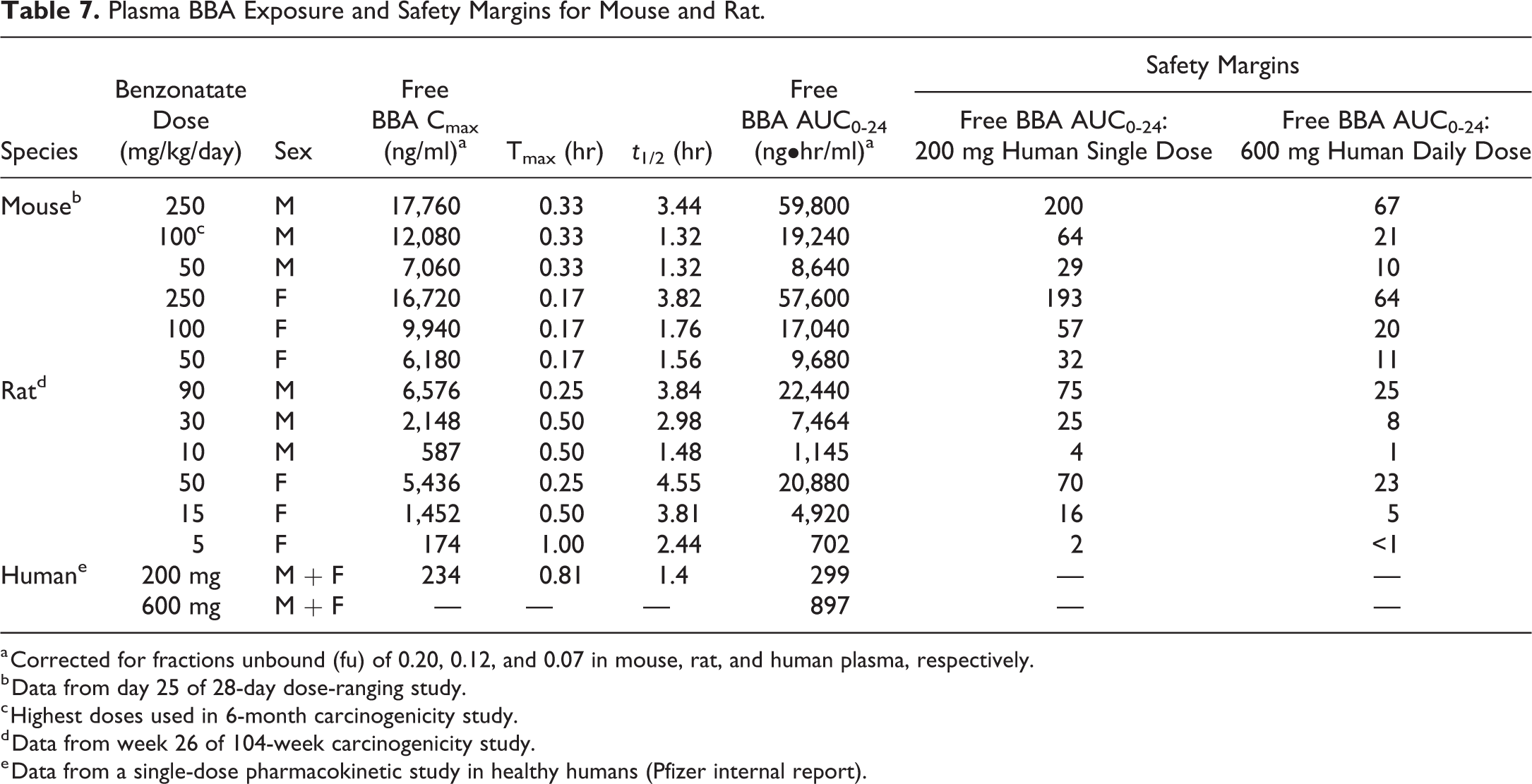
a Corrected for fractions unbound (fu) of 0.20, 0.12, and 0.07 in mouse, rat, and human plasma, respectively.
b Data from day 25 of 28-day dose-ranging study.
c Highest doses used in 6-month carcinogenicity study.
d Data from week 26 of 104-week carcinogenicity study.
e Data from a single-dose pharmacokinetic study in healthy humans (Pfizer internal report).
Two-year Wistar Han Rat Study
Mortality and survival
A total of 202 of the total 420 animals from the main study dose groups either died or were electively euthanized with most deaths occurring near the end of the dosing period. Of the early deaths, 94 were males at doses of 0 (22 rats), 10 (26), 30 (19), and 90 (27) mg/kg and 108 females at 0 (30), 5 (27), 15 (26) and 50 (25) mg/kg (Table 8). Deaths occurred at all doses. Mortality in benzonatate-treated groups was not statistically significantly different compared to the vehicle control group. The causes of early deaths were generally similar across all control and benzonatate-treated groups and were typical for Wistar Han rats in 2-year carcinogenicity studies (Covance historical control database) and not considered benzonatate related. Causes of deaths were either undetermined or spontaneous neoplasias (primarily pituitary, mammary, hematopoietic, and skin) or inflammation. Since survival was above 50% for all dose groups at week 80, the study was considered statistically valid for tumor incidence analysis (Figure 5; FDA Guidance 2001).
Mortality and Causes of Early Deaths in Rats.

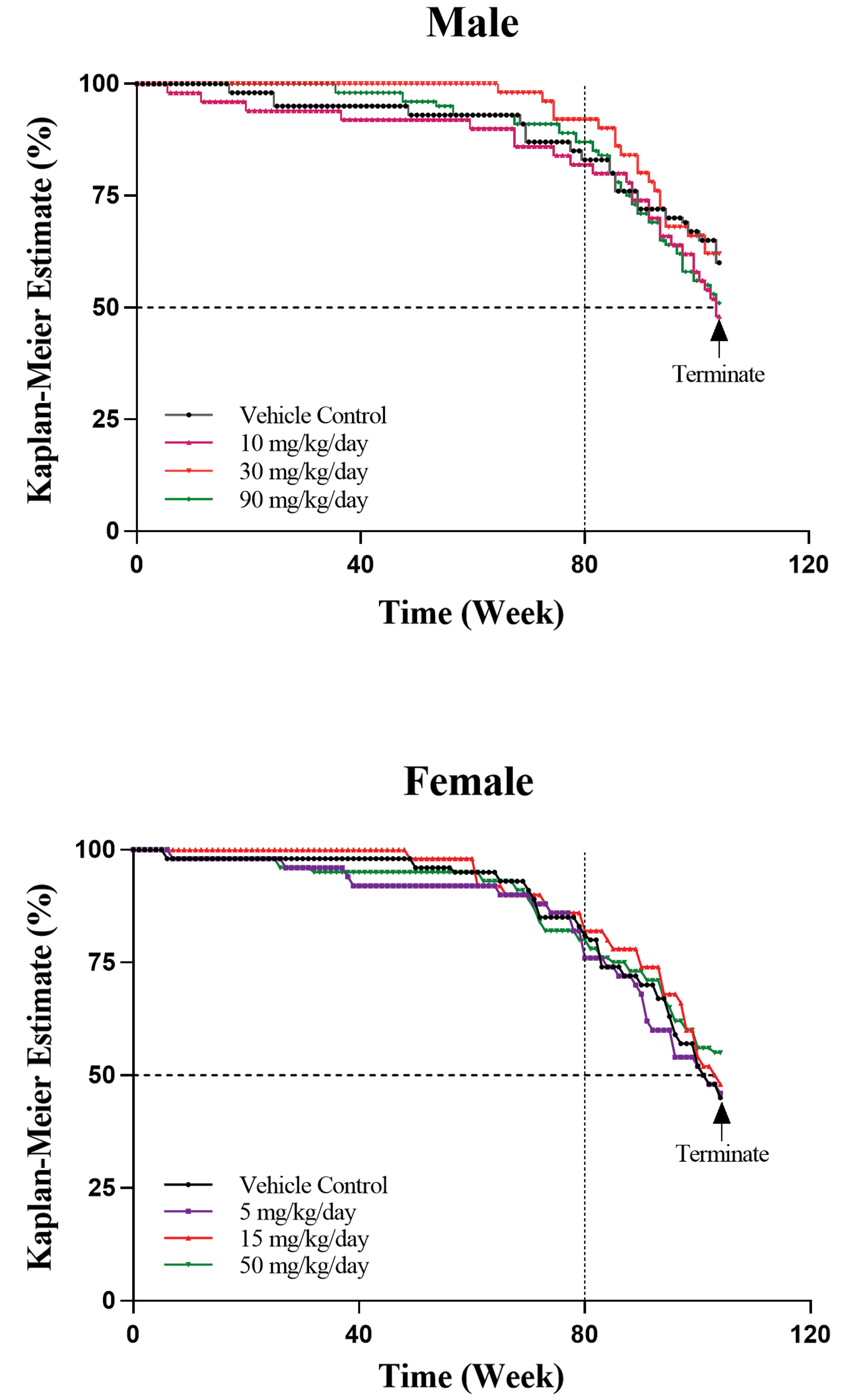
Kaplan–Meier survival plots from rat study.
Body weight and food consumption
There were no statistically significant differences in the group mean body weights, body weight gains, or food consumption between the control and treated dose groups (Figure 6).
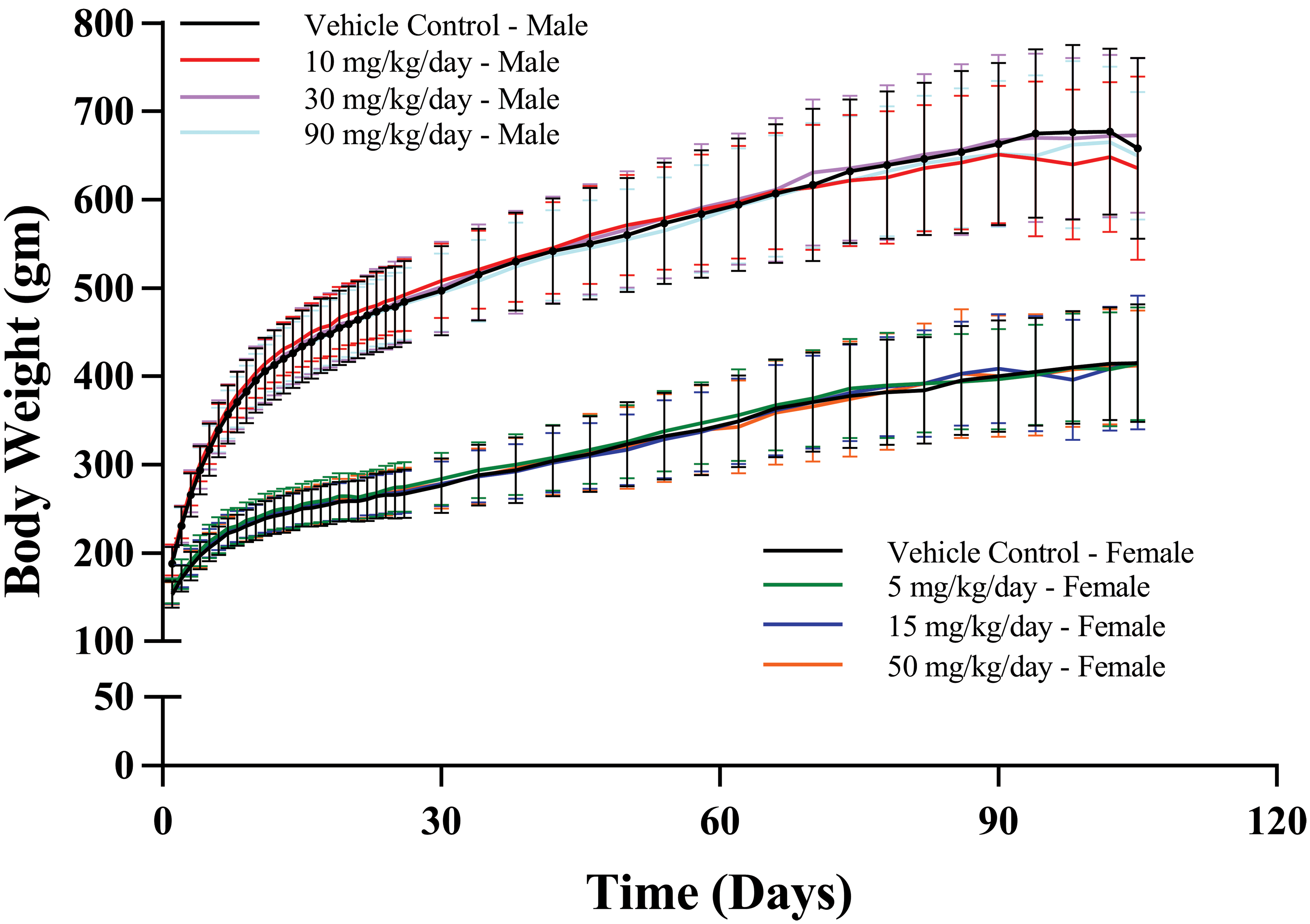
Body weights of rats over time.
Macroscopic findings
No benzonatate-related macroscopic findings were observed. A small number of tissues had an increased incidence of macroscopic findings but with no dose response and no consistent microscopic correlate. Remaining macroscopic findings were background incidental findings and not benzonatate related. There were no significant ophthalmic findings associated with benzonatate administration.
Microscopic findings
Hyperplastic/neoplastic lesions
There were no benzonatate-related hyperplastic or neoplastic findings. Heart endocardial hyperplastic/neoplastic microscopic lesions consisting of endocardial hyperplasia and malignant and benign endocardial schwannoma were observed in one female each at 50 mg/kg/day, and endocardial hyperplasia was observed in one male each at 30 and 90 mg/kg/day (Table 9). Endocardial hyperplasia is a background finding in this rat strain (McInnes 2012a). Historical control incidences were 0.83% and 1.67% for males and females, respectively, from 3 similar Han Wistar rat studies (Covance database). This incidence is similar to that seen in this study. The occurrences here were therefore considered a normal background variation with no clear dose response. The benign and malignant endocardial schwannomas in treated female rats were not statistically significant (p > .05 pairwise and trend) and had 2% and 0% incidences in control males and females, respectively, from similar rat studies (Covance database). The 0% incidence could be due to the small historical control database. No significant increases in any other nonendocardial tumors were noted in benzonatate-treated rats of both sexes (supplementary rat pathology report).
Rat Heart Hyperplastic and Neoplastic Lesions from Scheduled and Unscheduled Necropsies.
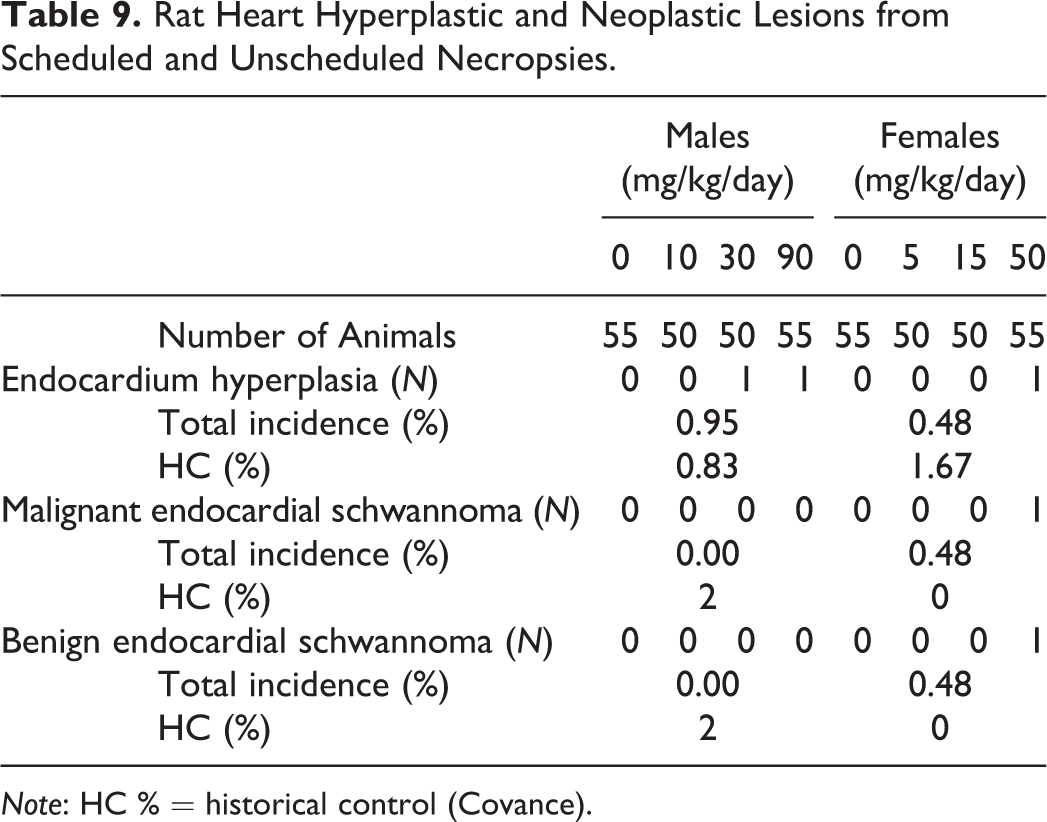
Note: HC % = historical control (Covance).
Nonhyperplastic and nonneoplastic lesions
There were no significant benzonatate-related nonhyperplastic or nonneoplastic findings. There was a minimally increased incidence of retinal atrophy in males administered ≥30 mg/kg/day. This was not considered benzonatate related, since the average severity score of retinal atrophy was similar in all dose groups including control. The incidence of 22.9% was similar to the maximum incidence of 22% obtained in male rats from 3 similar rat studies (Covance database). In addition, retinal atrophy is a common background finding in older rats compounded by varying exposure to light (McInnes 2012b).
Exposure and safety margins
As in the mouse study, benzonatate plasma levels in the carcinogenicity study were mostly below the LOD. The major metabolite BBA was detected in all treated groups. At the 90 mg/kg/day dose for male and 50 mg/kg/day for female, the free unbound plasma BBA exposures and safety margins were at least 70 times that of a 200-mg human dose (Table 7). Exposure (AUC0-24) to BBA at week 26 of the carcinogenicity study was supra-proportional from 10 to 30 mg/kg and 5 to 15 mg/kg/day in male and female, respectively. It was proportional from 30 to 90 mg/kg and 15 to 50 mg/kg/day in male and female, respectively (Table 7).
Discussion
These rodent carcinogenicity studies were performed according to current GLP regulations and International Conference on Harmonization guidelines on carcinogenicity testing, which were not available 60 years ago. They are part of a nonclinical program to update the current product label. The posology of benzonatate is 100 or 200 mg 3 times a day in adults and children over 10 years of age (Tessalon 2017). Benzonatate and its BBA and MPG major metabolites were not tumorigenic at BBA exposures that were at least 30 and 68 times that of a single human 200 mg dose in mouse and rat, respectively (Table 7). In the mouse dose-ranging study, exposure (AUC0-24) was dose proportional from 50 to 100 mg/kg/day and supra-proportional from 100 to 250 mg/kg/day. The supra-proportionality could be due to saturation of benzonatate esterase metabolism and/or renal elimination of BBA. This was supported by the significant increase in t 1/2 between these 2 dose ranges (Table 7). Similarly in the rat, the supra-proportional exposure from 10 to 30 mg/kg in male and 5 to 15 mg/kg in female and increase in t 1/2 could be saturation related (Table 7). No benzonatate-related neoplastic or nonneoplastic lesions were observed. All neoplastic and nonneoplastic lesions noted in rat and mouse studies were considered to be spontaneous.
The metabolism of benzonatate has not been extensively investigated. Human intravenous administration studies of its structural analogue procaine indicated that ester hydrolysis was a major metabolism pathway yielding para-amino benzoic acid (PABA) and diethylaminoethanol (Figure 2; Brodie, Lief, and Poet 1948). A more recent in vitro incubation study of benzonatate in mouse, rat, and human plasma showed similar rapid conversion to the N-substituted PABA, BBA, and the polymeric MPG (Pfizer internal report) with potential conjugation. This metabolic scheme is due to the ubiquitous presence of esterases in plasma, gastrointestinal tract, and liver in major mammalian test species and man (Berry, Wollenberg, and Zhao 2009). Due to the activity of the esterases, benzonatate plasma levels were undetectable at even the first sampling time points of 10 min in mouse and 5 min in rat with rapid corresponding increases in plasma BBA. The 2 carcinogenicity studies therefore mainly tested BBA and MPG. Both polar metabolites would be rapidly eliminated in the urine and feces either unchanged or as conjugates. This is supported by a human study where when 2 g of the structural analogue procaine were infused over 2 hr, ∼80% of the dose was excreted in the urine as PABA and its conjugates, ∼30% as diethylaminoethanol, and ∼2.7% as the parent procaine (Brodie, Lief, and Poet 1948). In addition, when 1 g each of the PABA and diethylaminoethanol metabolites were administered intravenously, both were rapidly conjugated and eliminated in the urine. The lack of any carcinogenic signal for the MPG polymer metabolite ((OCH2CH2)n) is supported by a negative 2-year Fischer 344 rat dietary carcinogenicity on a structural analogue polymer (Polyox N-10; CAS # 25322-68-3) having the general formula H-(OCH2CH2)n-H (Leung et al. 2000). This could be due to the low systemic exposure as the radiolabeled polymer was mainly excreted in the feces (99% of oral dose) with very low amounts (<1%) in the urine. This was confirmed by minimal exposure in the blood (Leung et al. 2000). The 2% dietary dose is equivalent to 1,150 mg/kg/day and is 23 times higher than the lowest 50 mg/kg/day dose of benzonatate in both studies. In addition, benzonatate and its major metabolites BBA and MPG were not mutagenic and did not cause numerical or structural chromosome alterations in an in vivo micronucleus assay (Teo et al. 2018).
Since cough is mostly a short-term indication, benzonatate use and exposure would therefore not be chronic or even lifetime. The rather short human BBA t ½ of 1.4 hr also reduces the extent of exposure. Postmarketing drug surveillance (pharmacovigilance) data from 1997 to 2017 have not shown any reports of cancer from patients prescribed benzonatate (Pfizer internal database). The surveillance data therefore supported results from the current rodent studies.
In summary, the peripherally acting antitussive benzonatate and its major metabolites BBA and MPG were not tumorigenic in mouse and rat carcinogenicity studies at BBA exposures of at least 32 and 70 times that of a 200 mg human dose, respectively.
Supplemental Material
Supplemental Material, DS1_VET_10.1177_0192623318789130 - The Antitussive Benzonatate Is Not Tumorigenic in Rodent Carcinogenicity Studies
Supplemental Material, DS1_VET_10.1177_0192623318789130 for The Antitussive Benzonatate Is Not Tumorigenic in Rodent Carcinogenicity Studies by Steve Teo, Madhav Paranjpe, Marie Mckeon, Peter Mann, Sophie Lee, Richard LaRock, and Tom Brown in Toxicologic Pathology
Supplemental Material
Supplemental Material, DS2_VET_10.1177_0192623318789130 - The Antitussive Benzonatate Is Not Tumorigenic in Rodent Carcinogenicity Studies
Supplemental Material, DS2_VET_10.1177_0192623318789130 for The Antitussive Benzonatate Is Not Tumorigenic in Rodent Carcinogenicity Studies by Steve Teo, Madhav Paranjpe, Marie Mckeon, Peter Mann, Sophie Lee, Richard LaRock, and Tom Brown in Toxicologic Pathology
Footnotes
Authors’ Note
Steve Teo is an employee of Pfizer Inc. Tom Brown was an employee of Pfizer while the studies were conducted. He is now with Clemson University. Madhav Paranjpe, Marie Mckeon, Peter Mann, and Sophie Lee were employed by the contract labs that performed these studies.
Authors Contribution
Authors contributed to conception or design (ST, MP, MM, SL); data acquisition, analysis, or interpretation (MM, SL, ST, PM, TB); drafted the manuscript (ST, MM); and critically revised the manuscript (ST, MP, MM). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The authors declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: These studies were funded by Pfizer Inc.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
