Abstract
Endometrial carcinoma is the most common gynecologic malignancy is the United States and accounts for 6% of all cancers in women. The disease is classified as type I or type II based on clinicopathologic and molecular features. It is a multifactorial disease with a number of risk factors, including environmental exposures. How environmental exposures, such as flame retardants, may affect the incidence of endometrial cancer is a topic of current and ongoing interest. Tetrabromobisphenol A (TBBPA) is a widely used brominated flame retardant found in a variety of household products. A recent 2-year National Toxicology Program carcinogenicity study found that exposure to TBBPA was associated with a marked increase in the development of uterine tumors, specifically uterine carcinomas, in Wistar Han rats. Molecularly, TBBPA-induced uterine carcinomas in Wistar Han rats were characterized by a marked increase in
Introduction
Endometrial cancer (EC) is one of the leading causes of cancer morbidity and mortality in women in the United States, with an estimated 49,560 new cases and 8,190 deaths in 2013, and even higher estimates for 2014 (American Cancer Society [ACS] 2013, 2014). EC in women is classified into 2 subtypes, type I and type II, based on clinicopathologic and molecular features. These 2 subtypes of EC have unique dualistic phenotypic and molecular features that correlate relatively well with the biologic behavior of these neoplasms (Horn et al. 2007; Lax 2004; Liu 2007; Llobet et al. 2009; Matias-Guiu et al. 2001; Ryan et al. 2005). Type I ECs account for 80 to 90% of cases and generally have a more favorable prognosis, while type II ECs, which represent approximately 10 to 20% of cases, are associated with a poor prognosis. While the features of the two EC subtypes may overlap to some extent, subtyping is of particular utility when determining clinical outcome, prognosis, or therapeutic options for these tumors.
Based on clinicopathologic features, type I tumors occur predominately in postmenopausal women, although they are also less frequently reported in premenopausal and perimenopausal women. Type I tumors are more often associated with nulliparity, are estrogen receptor-alpha (ERα) and progesterone receptor (PR) positive, and are slowly progressive with a lower risk of myometrial invasion (Mills and Longacre 2011). Alternatively, type II ECs occur in postmenopausal women, are associated with multiparity, and are high grade with an aggressive clinical course. Morphologically, type I tumors have endometrioid differentiation, characterized by irregularly shaped, variably sized glandular structures forming a merged, cribiform pattern or papillary proliferations, and associated with a desmoplastic stroma (Horn et al. 2007). Type I ECs are typically low grade and arise within a background of atypical hyperplasia, but high-grade type I tumors do occur that behave more aggressively. Type II ECs (nonendometrioid) are much more poorly differentiated, characterized by tubulocystic, papillary, or solid forms composed of small, cuboidal to “hobnail-” shaped cells with pleomorphic nuclei, frequent mitoses, and often bizarre nuclear forms. Necrosis may be common. Type II ECs are subdivided into a “clear cell” subtype based on cytoplasmic glycogen, or a “serous” subtype based on eosinophilic cytoplasm with apically located nuclei and focal cytoplasmic clearing. Features of both phenotypes may be present in the same tumor. Type II ECs arise within an estrogen-deficient, atrophic uterine epithelium and are associated with a precursor lesion of endometrial intraepithelial carcinoma. They are highly aggressive and infiltrative tumors, often spreading along the peritoneal cavity, and harbor a poor prognosis.
Aside from clinical and morphologic features, ECs in women can also be subclassified based on molecular phenotype (Horn et al. 2007; Lax 2004; Liu 2007; Llobet et al. 2009; Matias-Guiu et al. 2001; Ryan et al. 2005). Type I EC is associated with a higher incidence of phosphatase and tensin homolog (
While all type II ECs are considered high grade, an accepted and widely used histologic grading system has been developed for type I ECs to distinguish more aggressive, high-grade type I tumors from low-grade type I tumors. High-grade type I ECs have overlapping morphologic and molecular features of type I and type II tumors, including increased local invasion, loss of ERα and PR expression, increased KI67 protein expression, high rates of
Clinicopathologic and molecular features of human EC subtypes.
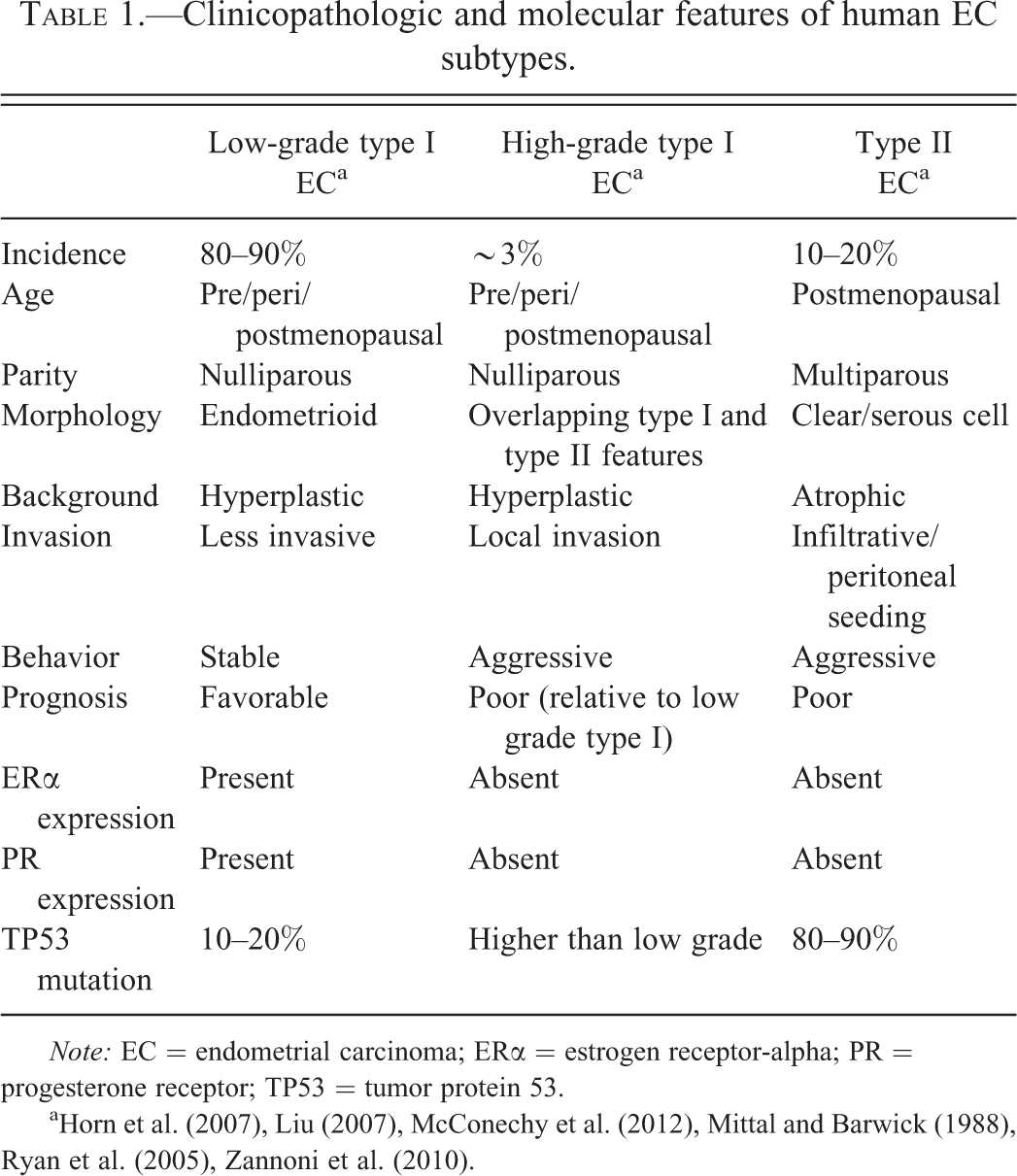
a Horn et al. (2007), Liu (2007), McConechy et al. (2012), Mittal and Barwick (1988), Ryan et al. (2005), Zannoni et al. (2010).
A major risk factor for EC in women is hormone dysregulation. Many of the contributing factors to the development of EC influence estrogen levels, and increased estrogen is associated with EC development. Hormone (estrogen) therapy in menopausal women is associated with increased risk (Beral, Bull, and Reeves 2005), whereas the use of oral contraceptives and multiparity are protective (Modan et al. 2001). Early menarche and late menopause are risk factors, and nulliparity is a risk factor, since pregnancy shifts the balance of hormones from estrogen to progesterone (Emons et al. 2000). Other risk factors include family history, diabetes, obesity, tamoxifen therapy, and ovarian pathologies such as granulosa cell tumors and polycystic ovarian syndrome, which are associated with unabated estrogen secretion (Emons et al. 2000). Aside from family history, the majority, if not all of these risk factors, are associated with hormone imbalance. It is becoming evident that an increasing number of compounds that people are exposed to in their environment, whether they are environmental pollutants or occupational compounds, have the ability to influence hormone production and function.
Tetrabromobisphenol A (TBBPA), a compound structurally related to bisphenol A and formed by bromination of bisphenol A, is a widely used flame retardant in electrical equipment, plastics, paper, furniture, and carpeting. It is a high-volume production chemical in the United States and the most widely used brominated flame retardant in the world (Schauer et al. 2006). It has been identified in fish, shellfish, and drinking water. Like bisphenol A, TBBPA has been shown to have endocrine disrupting activity
Materials and Methods
Animals, Histology, and Immunohistochemistry
TBBPA-induced uterine carcinomas were obtained from Wistar Han [Crl:WI(Han)] rats in the 2-year NTP bioassay. The care of animals on this study was according to National Institutes of Health (NIH) procedures as described in the “The U.S. Public Health Service Policy on Humane Care and Use of Laboratory Animals,” available from the Office of Laboratory Animal Welfare, National Institutes of Health, Department of Health and Human Services, RKLI, Suite 360, MSC 7982, 6705 Rockledge Drive, Bethesda, MD 20892-7982 or online at http://grants.nih.gov/grants/olaw/olaw.htm#pol. The use of animals was approved by the National Institute of Environmental Health Sciences (NIEHS) animal care and use committee. Forty-one formalin-fixed, paraffin-embedded (FFPE) uterine carcinomas from TBBPA-exposed animals were used in this study (10 low dose, 15 mid dose, and 16 high dose). A total of 10 FFPE spontaneous carcinomas were available from control animals from various NTP studies. Due to the low incidence of spontaneously occurring uterine carcinomas in the current TBBPA carcinogenicity bioassay (2 spontaneous tumors in corn-oil vehicle gavage control group), additional FFPE spontaneous tumors were obtained from other available Wistar Han rat carcinogenicity bioassay studies: one from polybrominated diethyl ether mixture (DE-71-NTP TR 589, 2014d; corn-oil vehicle gavage), 2 from green tea extract (GTE-NTP TR 585, 2014c; deionized water vehicle), 2 from metal working fluids (MWF-NTP TR 586, 2014b; whole body inhalation), and 3 from antimony trioxide (AT-NTP TR 590, 2014a; whole body inhalation). Five FFPE normal uteri from corn-oil vehicle gavage control females in the concurrent TBBPA study were used as normal uterus controls. Four board-certified anatomic veterinary pathologists reviewed the histology of all uterine carcinomas using the standardized system of nomenclature and diagnostic criteria (SSNDC) guide for female reproductive proliferative lesions in rats to morphologically characterize the endometrial adenocarcinomas as well as differentiated or poorly differentiated tumors (Table 2). If a tumor had areas that were well differentiated and poorly differentiated, then the tumor was classified as poorly differentiated. Tumors available for molecular biology included those >0.5 cm at the time of necropsy. These tumors were sectioned in half, and one half was flash frozen in liquid nitrogen for molecular analysis and the other half fixed in 10% neutral buffered formalin (NBF) for histology and immunohistochemistry (IHC). Frozen tissue was available from 6 uterine carcinomas from TBBPA-exposed animals, and 9 vehicle control animals from the abovementioned studies (1 from TBBPA, 2 from GTE, 3 from AT, 2 from DE71, and 1 from MWF). The abovementioned frozen tissues and paired FFPE tissues were used for quantitative polymerase chain reaction (
SSNDC classification of endometrial adenocarcinomas.

DNA Isolation, Polymerase Chain Reaction (PCR) Amplification, and Autosequencing
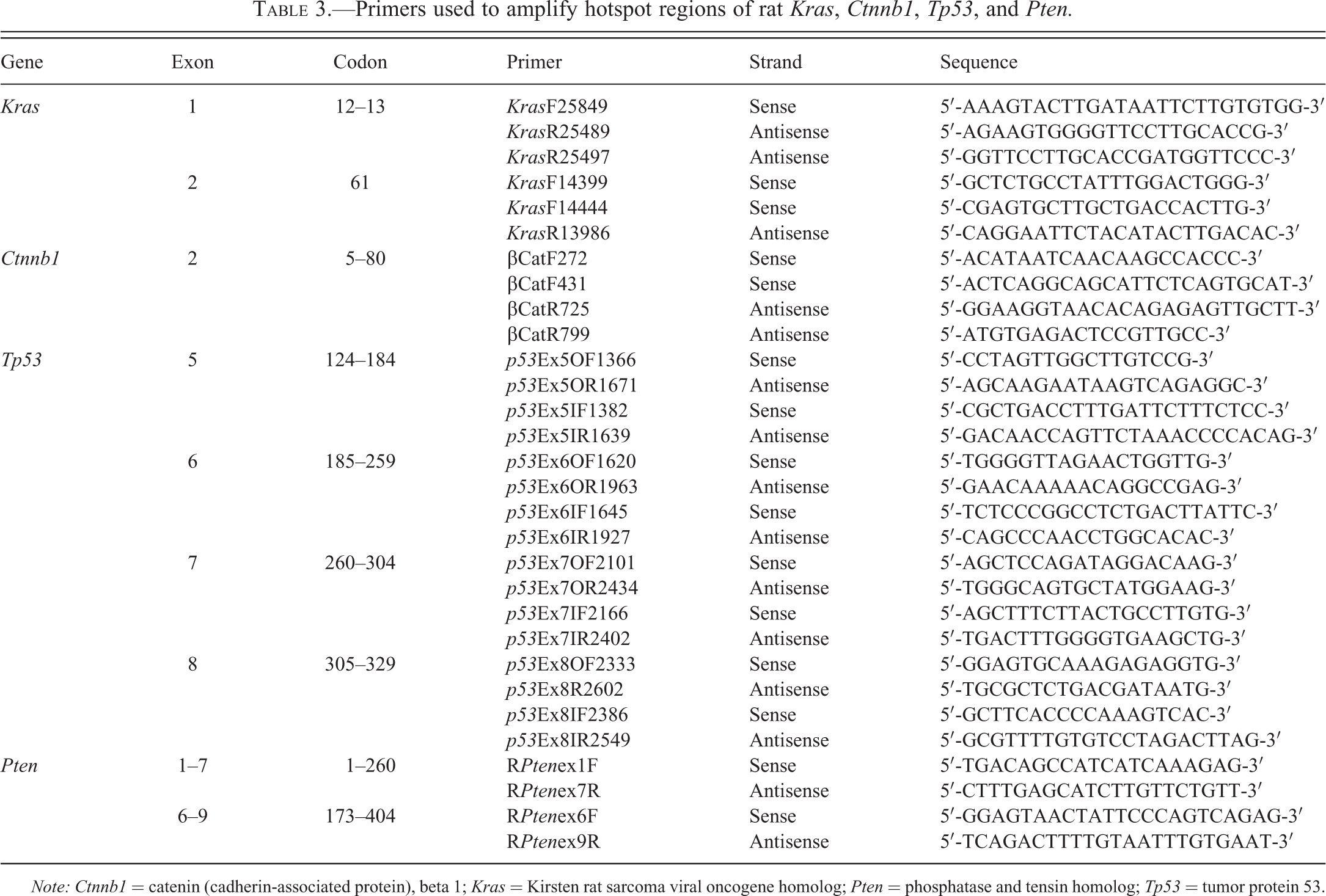
DNA was isolated and extracted from 22 FFPE TBBPA-induced uterine carcinomas and 10 FFPE spontaneous uterine carcinomas from rat control animals with DNeasy Tissue Kit (Qiagen, Valencia, CA). Amplification reactions were carried out by semi-nested PCR using the designed primer sets (Table 3) for
Primers used to amplify hotspot regions of rat
qPCR
RNA was extracted and purified from frozen samples using the Invitrogen TRIzol Kit (Invitrogen, Carlsbad, CA). Fold increases and decreases in gene expression were determined by quantification of cDNA from target samples relative to a calibrator sample of controls. The
Statistical Analysis
Incidences of neoplasms and nonneoplastic lesions were tested using one-sided poly-3 tests (Bailer and Portier 1988; Portier and Bailer 1989; Piegorsch and Bailer 1997). This method modifies the Cochran–Armitage linear trend test and Fisher’s exact test to take length of survival into account, by more closely approximating the total number of animal-years at risk. The poly-3 trend test assesses the significance of the linear trend of incidences with dose, while the pairwise poly-3 tests assesses the significance of pairwise differences in incidences of each dose group compared to the control group. Differences are considered statistically significant if the one-sided
Results
Exposure to TBBPA was associated with a treatment-related increase in uterine carcinomas in Wistar Han rats. The results of the 2-year NTP chronic bioassay indicated that TBBPA exposure resulted in a statistically significant (
Incidence of epithelial uterine neoplasms in TBBPA-exposed Wistar Han rats in the 2-year NTP bioassay.
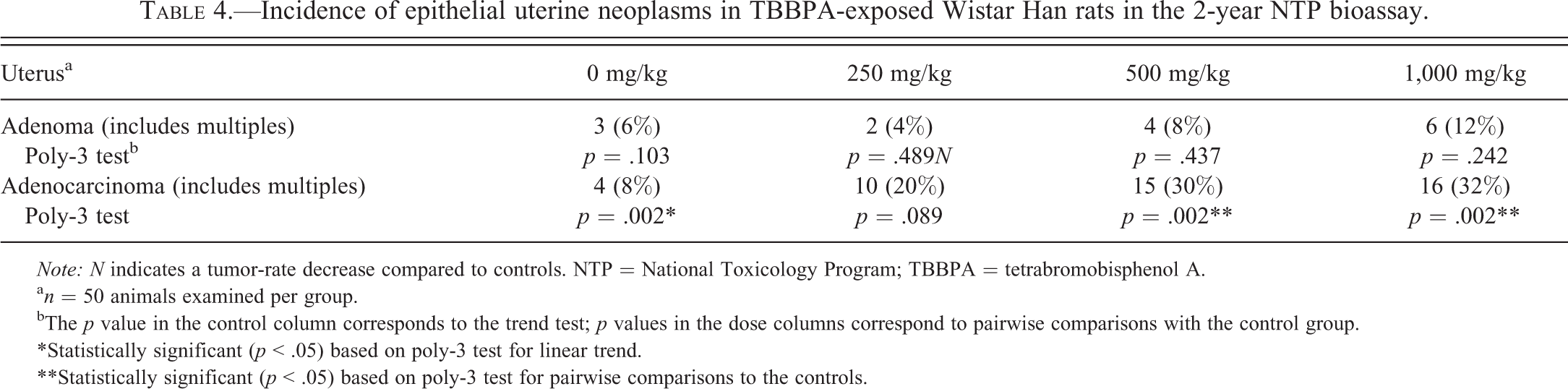
a
bThe
*Statistically significant (
**Statistically significant (
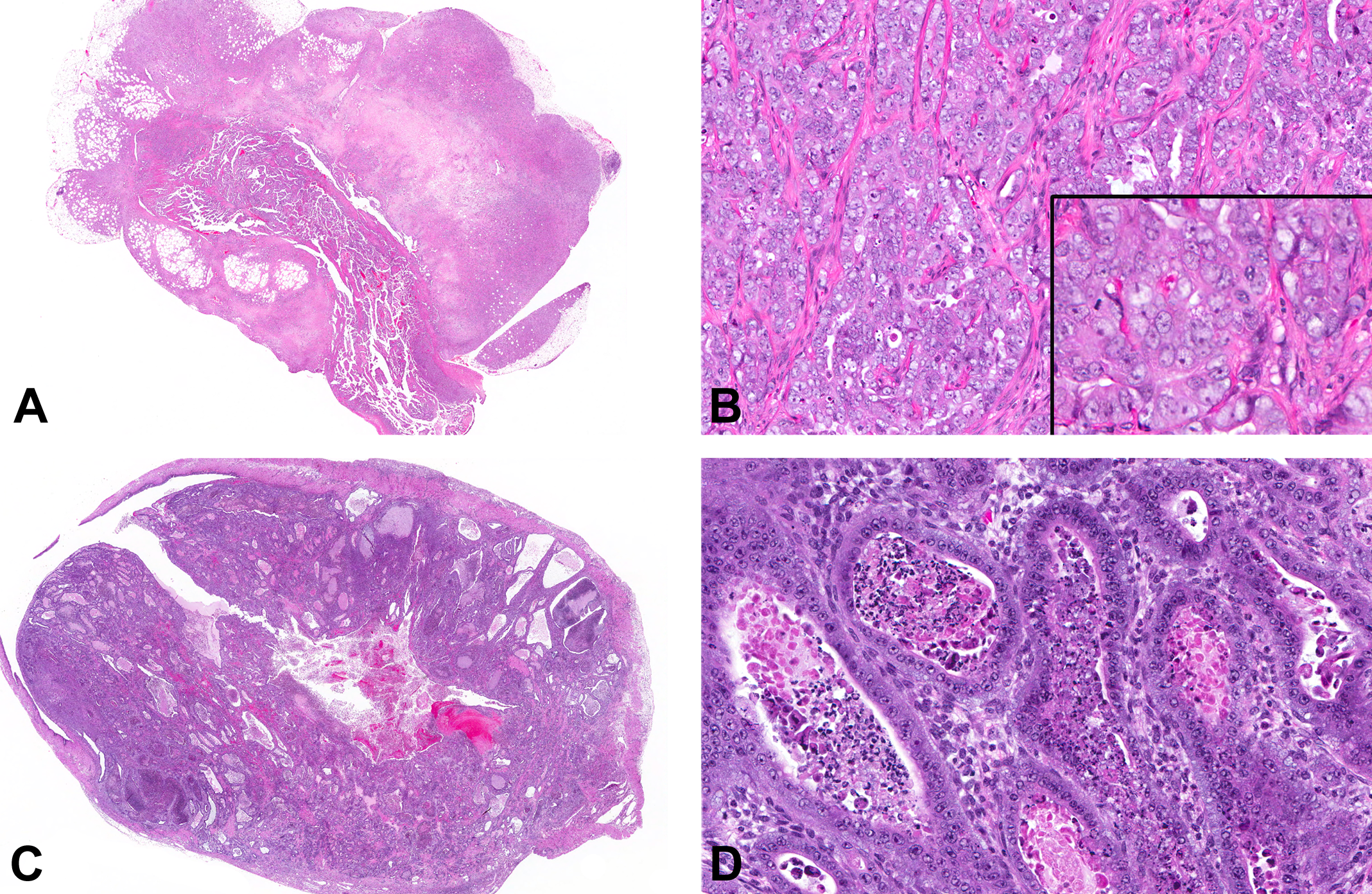
Uterine carcinomas from tetrabromobisphenol A (TBBPA)-exposed Wistar Han rats. (A) Poorly differentiated uterine carcinoma characterized by marked invasion through the uterine wall into the peri-uterine tissues, accompanied by large zones of necrosis. (B) Poorly differentiated uterine carcinomas were characterized by solid clusters, cords, and nests of poorly differentiated epithelial cells with marked pleomorphism and cellular atypia, including large vesicular nuclei, multiple nucleoli, marked anisocytosis and anisokaryosis (inset), and mitotic figures. (C) Well-differentiated carcinomas were fairly well circumscribed with stromal invasion relatively limited to the superficial and mid-endometrium rather than replacing large areas of the deep stroma and a glandular phenotype. (D) Glands were generally lined by one or more layers of fairly well-differentiated cuboidal to columnar epithelial cells with minimal to mild pleomorphism and few mitotic figures.
Morphologic and immunohistochemical features of uterine carcinomas from control and TBBPA-exposed WH rats.
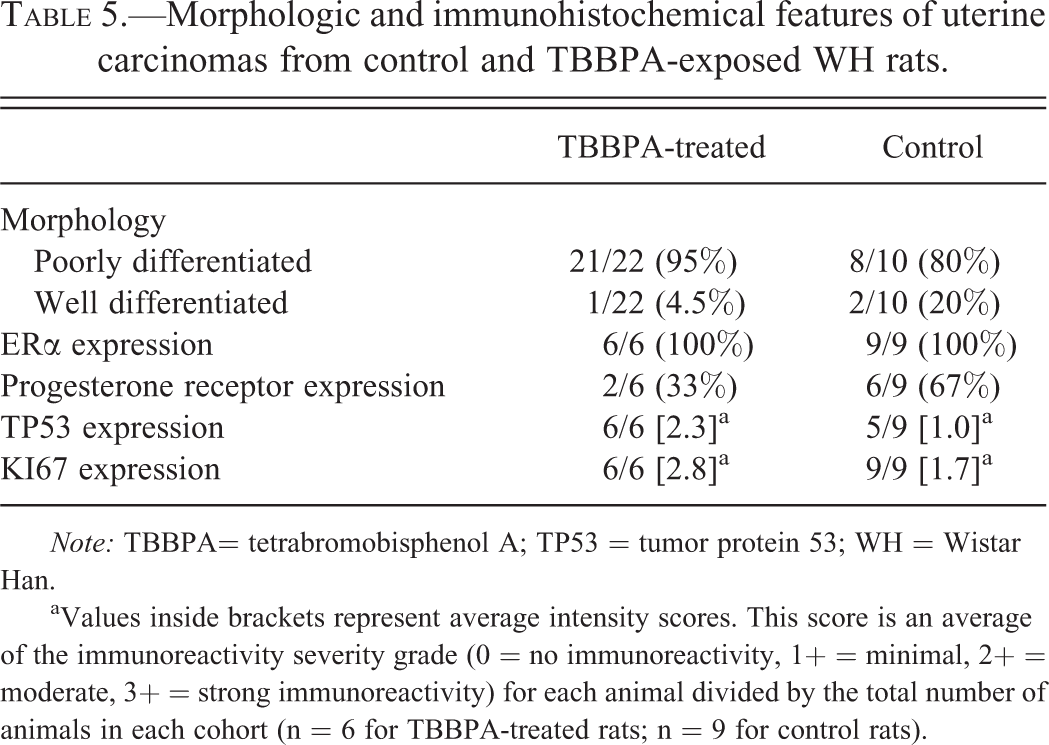
aValues inside brackets represent average intensity scores. This score is an average of the immunoreactivity severity grade (0 = no immunoreactivity, 1+ = minimal, 2+ = moderate, 3+ = strong immunoreactivity) for each animal divided by the total number of animals in each cohort (n = 6 for TBBPA-treated rats; n = 9 for control rats).
Uterine carcinomas in TBBPA-exposed Wistar Han rats show a tendency toward loss of PR expression and increased TP53 protein expression compared to spontaneous uterine carcinomas. Since criteria for classifying human type I and II endometrial carcinoma relies partly on hormone (ERα/PR) expression, proliferation, and
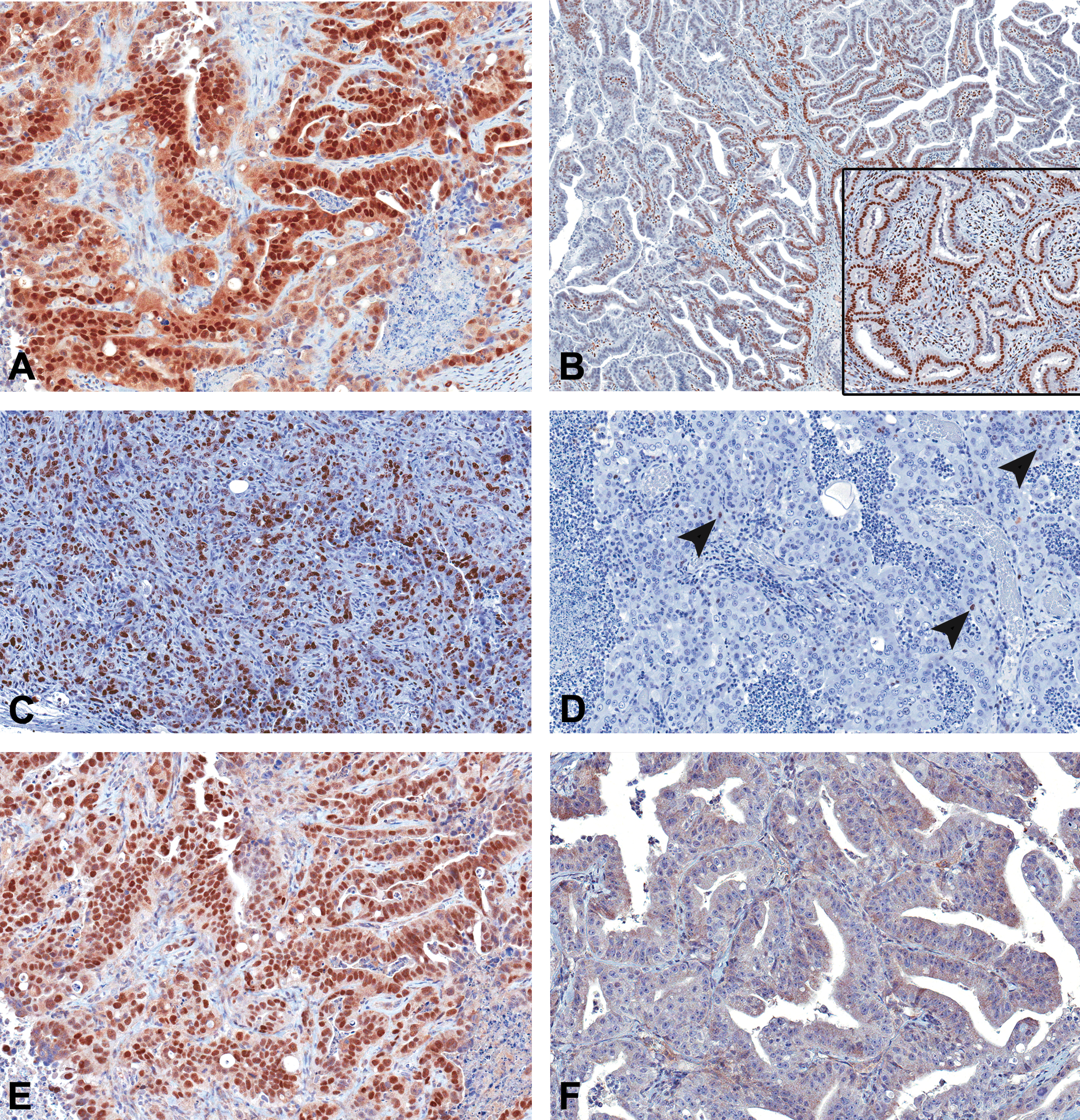
Immunohistochemical features of uterine carcinomas. (A) All spontaneous and tetrabromobisphenol A (TBBPA)-induced uterine adenocarcinomas had diffuse nuclear immunoreactivity to estrogen receptor-alpha (ERα). (B) Two uterine carcinomas from TBBPA-exposed animals had focal nuclear immunoreactivity to progesterone receptor (PR) antibody, and 1 spontaneous tumor showed diffuse nuclear immunoreactivity (inset). (C) Poorly differentiated uterine carcinomas showed diffuse KI67 expression, whereas well-differentiated tumors (D) had single cell (arrowheads) to no expression of KI67. (E) Uterine carcinomas from all TBBPA-exposed animals showed strong nuclear immunoreactivity to antibodies to
Uterine carcinomas in TBBPA-exposed Wistar Han rats had increased incidence of
Mutation frequency of

Morphologic and molecular features in uterine carcinomas from humans and TBBPA-exposed WH rats.
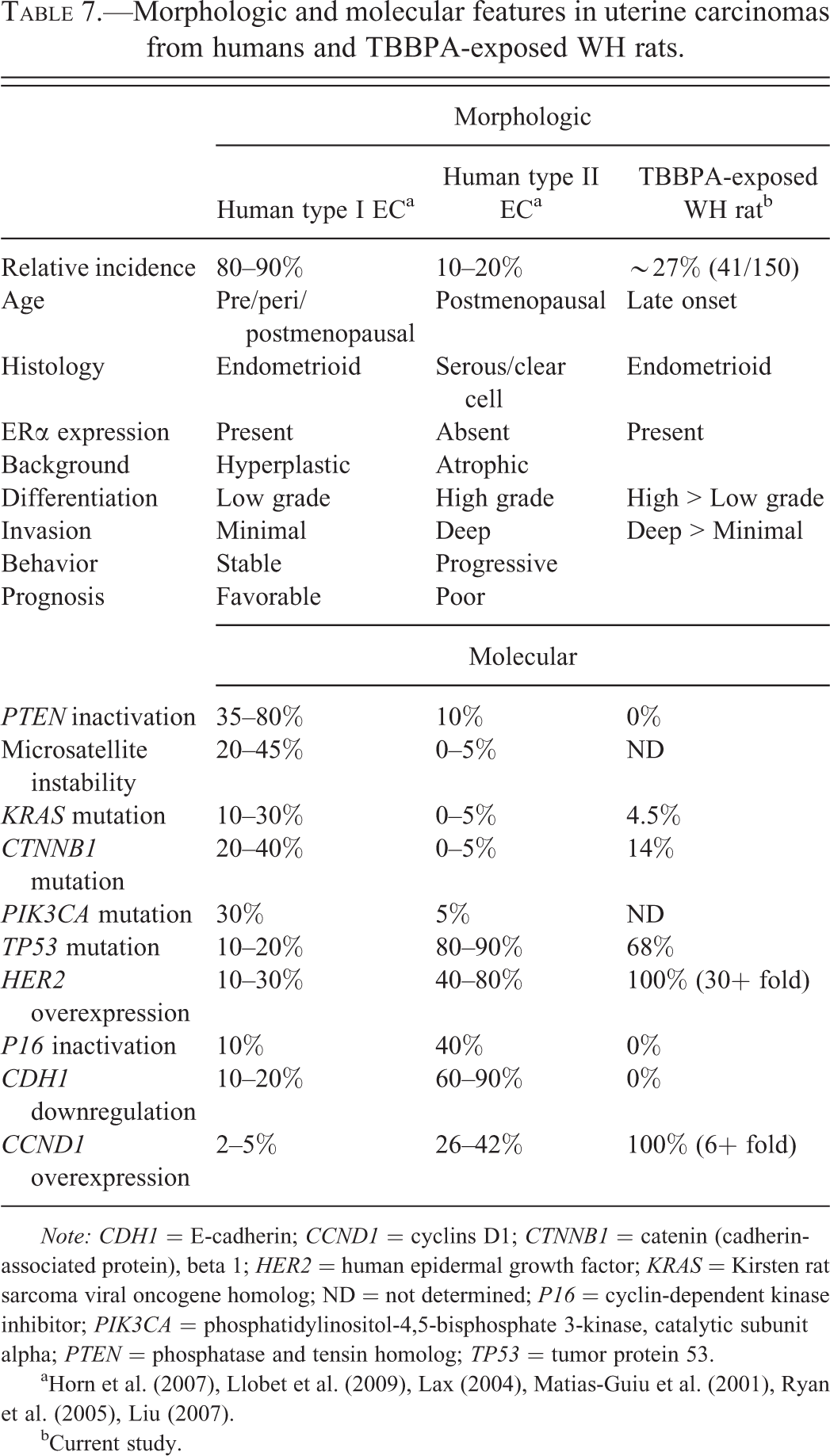
a Horn et al. (2007), Llobet et al. (2009), Lax (2004), Matias-Guiu et al. (2001), Ryan et al. (2005), Liu (2007).
bCurrent study.
Uterine carcinomas from TBBPA-exposed rats had increased
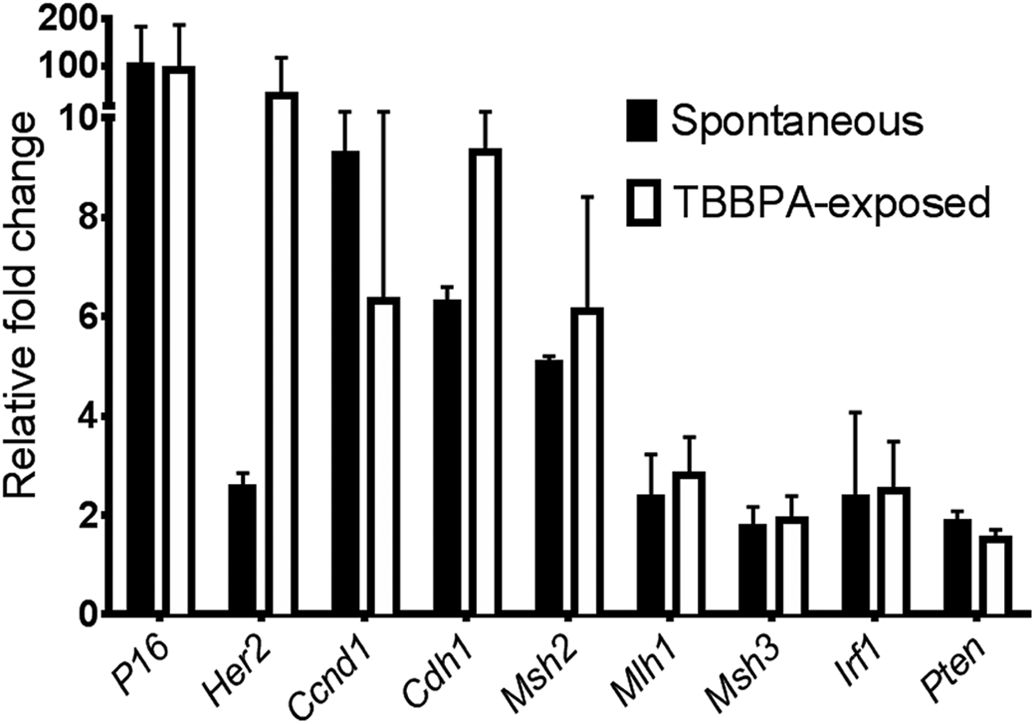
Real-time quantitative PCR (qPCR) gene expression of mediators involved in human endometrial carcinogenesis. Uterine carcinomas in tetrabromobisphenol A (TBBPA)-exposed animals averaged a 30-fold overexpression of human epidermal growth factor 2 (
Discussion
Long-term exposure of Wistar Han rats to TBBPA resulted in treatment-related increased frequencies in uterine carcinomas. The majority of uterine carcinomas in both TBBPA-exposed animals and spontaneous tumors were considered poorly differentiated. Uterine carcinomas in this study varied from a relatively more well-differentiated glandular and papillary form to poorly differentiated, infiltrative clusters, ribbons, or solid sheets of anaplastic glandular epithelial cells. A majority of poorly differentiated carcinomas also expressed high levels of KI67, in contrast to well-differentiated tumors. In women, KI67 expression is a prognostic factor correlated with higher grade, more aggressive ECs and a poor prognosis (Lax 2004; Oreskovic et al. 2004; Salvesen et al. 1999; Zannoni et al. 2010). In addition to similar morphologic features, for the most part alterations in gene expression and mutation spectra relevant for human EC were similar in Wistar Han rat uterine carcinomas regardless of TBBPA exposure. Uterine carcinomas in both groups had nuclear expression of ERα, increased expression of
The primary molecular differences observed in uterine carcinomas from TBBPA-exposed animals compared to spontaneous tumors included a marked increase in
Spontaneous uterine carcinomas and carcinomas from TBBPA-exposed rats both shared morphologic and molecular features with type I and II EC in women. Features common to both spontaneous and TBBPA-exposed tumors that were shared with type I EC tumors in women included late onset, endometrioid differentiation, and ERα hormone receptor expression. Features shared between rat uterine carcinomas and human type II EC include poor differentiation and deep invasion, high mitotic rate and KI67 index, increased cellular pleomorphism and atypia,
By far, the most striking differences between uterine carcinomas in TBBPA-exposed animals and controls were the marked increase in
Other features of type I and II EC in women include
The results of this study suggest that uterine carcinomas in TBBPA-exposed Wistar Han rats most closely resemble high-grade type I EC in women on the molecular level, which are typified in humans by conservation of ERα expression, a high rate of
In summary, uterine carcinomas were observed with a higher frequency in Wistar Han rats exposed to TBBPA, have higher rates of proliferation, an increased rate of
Footnotes
Acknowledgments
The authors would like to thank the CMPB Necropsy Core Laboratory personnel, Histology, Immunohistochemistry, and Sequencing Core Laboratories for their technical expertise; and Ms. Beth Mahler and Ms. Eli Ney (NIEHS) for images and figures.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Institutes of Environmental Health Sciences (NIEHS), National Institutes of Health (NIH), and the Division of the National Toxicology Program (DNTP).
Author Contribution
Authors substantially contributed to conception or design (AP, MH), contributed to acquisition (JH, TO, H-HH, SB, T-VT, AP, TM, SP, MH), contributed to analysis (JH, TO, H-HH, SB, T-VT, AP, TM, JD, SP, SEMH), contributed to interpretation (JH, TO, AP, SP, SE, MH), and drafted the manuscript (JH, TO, AP, SP, MH). All authors critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Authors’ Note
Janice B. Harvey and Tanasa S. Osborne contributed equally to this work. This article may be the work product of an employee or group of employees of the National Institute of Environmental Health Sciences (NIEHS), National Institutes of Health (NIH); however, the statements, opinions, or conclusions contained therein do not necessarily represent the statements, opinions, or conclusions of the NIEHS, NIH, or the U.S. government.
