Abstract
It is sometimes difficult to assess the relevance of tumors that occur in treated animals in short-term studies. This report is intended to establish a general profile of tumor occurrence in young Han Wistar rats. Data were collected and evaluated from 29 rat carcinogenicity studies and from a few 2-, 4-, 13-, and 26-week studies conducted between 1995 and 2009 at Huntingdon Life Sciences, UK. The route of administration was dietary, oral gavage, or inhalation, and the analysis was confined to sporadic deaths (decedents) in carcinogenicity studies. In Han Wistar rats, the most common and earliest occurring tumor was malignant lymphoma in both sexes, the earliest being seen in the 16th and 26th week in males and females, respectively. The incidence of malignant lymphoma was slightly higher in males than in females. The second most common type of tumor was brain tumors in males and mammary tumors in females. Compared with Sprague-Dawley rats, where the most common early tumor was pituitary tumor in females, the most common early tumor in Han Wistar rats was malignant lymphoma in both sexes. These early tumor profiles are consistent with the lifetime tumor occurrence in these strains.
Introduction
Lifetime spontaneous tumor profiles and incidence rates of laboratory rodents are easily accessible, but the data on tumor occurrence in the first year of life are comparatively rare. Information in Sprague-Dawley rats has been reviewed in the past (Son and Gopinath 2004). Environmental factors influencing the incidence of spontaneous tumors in rodents have been reviewed by several authors, although most of them are lifespan data (Everett 1984; Hardisty 1985; Maita et al. 1988; Ettlin, Stirnimann, and Prentice 1994; Gopinath 1994). Occurrence of occasional tumors in short-term studies has always caused concern, especially when they fall among treated groups. It is very useful to have data on incidence of spontaneous tumor in animals of lower age groups comparable to short-term studies. Han Wistar rats are a widely used strain of laboratory rats in toxicity studies, and tumor data in this strain are not easily accessible for the younger age group. It was considered useful to establish tumor profiles of Han Wistar rats from 2-, 4-, 13-, 26-, and 52-week studies.
Materials and Methods
Animals
Male and female Wistar rats supplied by three different animal suppliers (Harlan UK Ltd, Bicester, UK; Charles River Limited, Kent, UK; and Biological Research Labs Ltd, Basel, Switzerland) were used. For the carcinogenicity and short-term studies, 1 to 4 male or female rats, aged 5 to 6 weeks old, were randomly selected and were single-sex housed in stainless steel wire cages or suspended solid-bottomed polycarbonate cages with sawdust or wood-chip bedding. Animal room temperature and relative humidity were generally maintained at 19–23°C and 40–70%, respectively, with a 12-hour light/dark cycle and 15 filtered air changes per hour. All rats had free access to tap water in bottles with sipper tubes and diet (most of them were SDS Special Diets, Witham, UK; and a few of them were UAR VRF 1 or RM1 [E] SQC, SDS). Drinking water and diet were routinely subjected to chemical analysis to monitor for contaminants that might influence the studies. Food hoppers and water bottles were changed at daily to twice-weekly intervals. Studies commenced after an acclimatization period of 14 days, during which animal health was monitored by a veterinary officer. All studies were conducted using protocols approved by the Huntingdon Life Sciences Animal Care and Use Committee.
Histopathology
Comprehensive necropsy was performed on all decedent animals from carcinogenicity and short-term studies and all terminal kill animals from short-term studies. Tissues were fixed in 10% neutral buffered formalin (except eyes, which were fixed in Davidson’s fluid; and testes and epididymides, which were initially fixed in Bouin’s solution and then transferred to 70% industrial methylated spirit). Samples of macroscopically abnormal tissues (including all tissue masses) were routinely fixed, along with adjacent normal tissues where appropriate. All tissues were embedded in paraffin wax, and 4–5 μm sections were stained with hematoxylin and eosin. A full list of tissues was examined for all decedent animals. After initial examination by a study pathologist, slides and results were peer-reviewed by a second pathologist.
Study Design
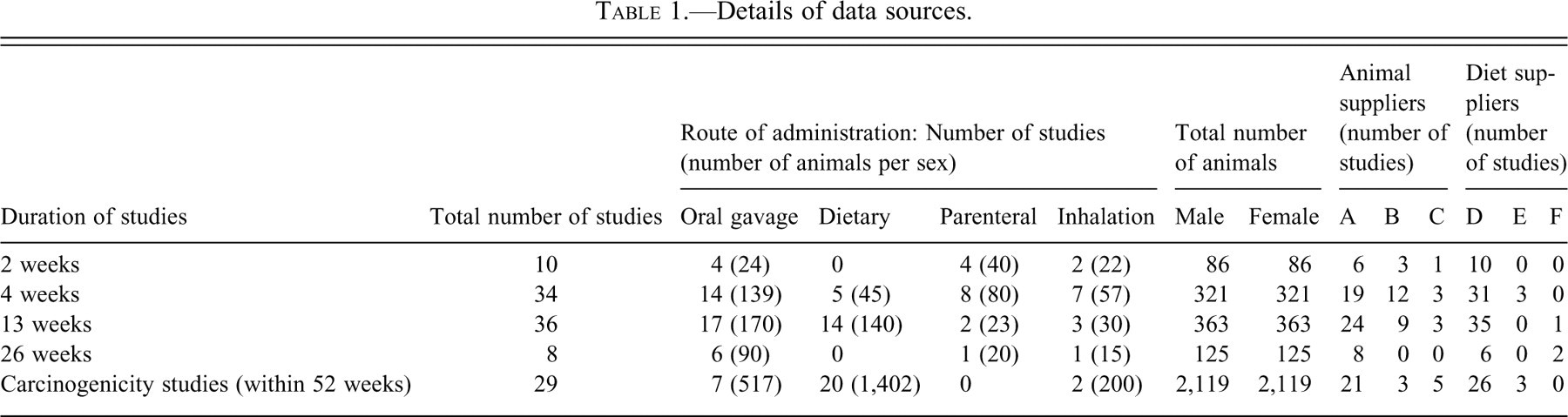
The details of the sources of data are presented in Table 1 . This report is based on 88 short-term studies, including 2-, 4-, 13-, and 26-week studies and 29 carcinogenicity studies conducted at Huntingdon Life Sciences, UK, using Han Wistar, during the period 1995–2009. Information from carcinogenicity studies was gathered from control groups (a total of 2,119 male and 2,119 female rats). Control groups varied in size. For short-term studies, most comprised 10–15 animals per sex, and there were at least 50 male and 50 female animals for carcinogenicity studies. For short-term studies, data from all decedent and terminal kill animals were collected. For carcinogenicity studies, data were collected from all control animal deaths occurring within the first 52 weeks. Mortality profiles from the carcinogenicity studies were compared with the previous report on Sprague-Dawley rats (Son and Gopinath 2004).
Details of data sources.
Results
Short-Term Studies
There were no incidences of any tumors in any of the 2-, 4-, 13-, or 26-week studies reviewed for control animals.
Carcinogenicity Studies
Data for mortality profiles and tumors from all carcinogenicity study decedents in control groups before week 52 are presented in Tables 2, 3, 4, and 5. The most common factor contributory to death was the tumors in both sexes (76% in males; 54% in females), and the skin wounds were the second most common contributing factor to deaths (14% in males; 11% in females). Most animals were killed for humane welfare reasons (86% in males; 96% in females), and a few of them were found dead in their cage (14% in males; 4% in females). In males, 29 sporadic deaths occurred before week 52, and of these, 22 rats died from tumors and 7 rats from non-neoplastic conditions. In the 28 female decedents, 15 deaths were associated with neoplastic and 13 with non-neoplastic lesions. There was a wide spectrum of tumor types. The most frequently occurring tumor was malignant lymphoma in both sexes. This tumor incidence increased progressively with age, with a higher incidence in males. The earliest identified malignant lymphoma was in a male rat during the 16th week of study. Renal tubular carcinoma was also found in a male during week 17 of study. In males, three brain tumors (astrocytoma, malignant ependymoma, and mixed glioma) were seen between 41 and 52 weeks of study. Other tumors, seen in occasional males dying after week 26, include fibrosarcoma of the skin, pituitary adenoma, renal mesenchymal tumor, leiomyosarcoma of the cecum, malignant Schwannoma in the thorax and a hepatocellular carcinoma.
Mortality profiles of decedents in Han Wistar carcinogenicity studies (up to 52 weeks).

Comparison of mortality profiles between Sprague-Dawley rats (up to 50 weeks) and Han Wistar rats (up to 52 weeks).
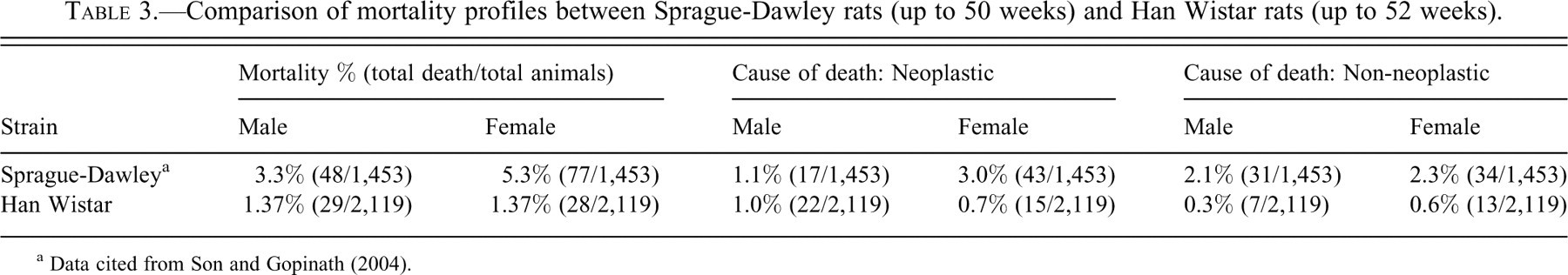
a Data cited from Son and Gopinath (2004).
Tumor incidence profiles of decedents from the control groups in Han Wistar carcinogenicity studies expressed by time of death—Males.

Tumor incidence profiles of decedents from the control groups in Han Wistar carcinogenicity studies expressed by time of death—Females.
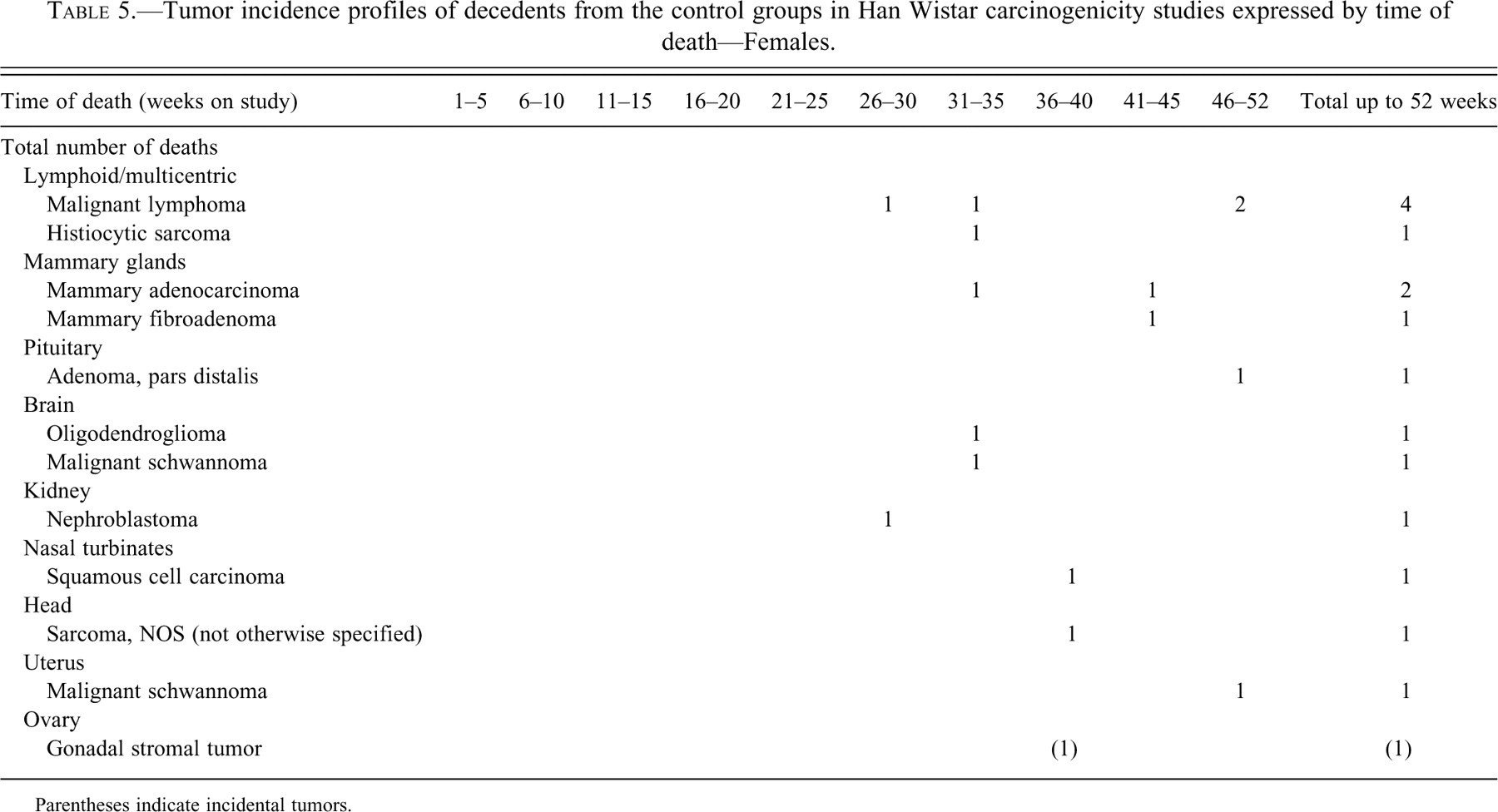
Parentheses indicate incidental tumors.
In females, several hematopoietic tumors were seen, with malignant lymphoma being the most common, the earliest being seen in the 26th week of study. Also nephroblastoma of the kidney was seen in a female during the 27th week of study. The mammary gland showed the second highest tumor incidence. Tumors seen in occasional animals, mainly from week 31 onward, were oligodendroglioma and malignant schwannoma of the brain (intracranial), squamous cell carcinoma of the nasal turbinates, undifferentiated sarcoma in the head, pituitary adenoma, and malignant schwannoma of the uterus. One gonadal stromal tumor was identified in the ovary as an incidental tumor in the 40th week of study.
Discussion
It is valuable to have background data of early-occurring tumors. Often it is problematic when interpreting tumors in short-term toxicology studies as to whether they are treatment-related. Although they are rare in incidence, using weight of evidence basis, they must be considered carefully. Comparison with background data is very helpful to determine their relevance to treatment.
Previously we have reported early-occurring tumor data for Sprague-Dawley rats and CD-1 mice. Han Wistar rats are a widely used strain for preclinical studies, and the paucity of available information prompted us to collect data from the large pool of studies undertaken at Huntingdon Life Sciences.
The use of Han Wistar rats has some benefits compared with other strains, namely, smaller body size which enables a reduction in the amount of test material required; relatively stable and lower tumor incidence; and longer survival rates, which improve study integrity. Recently Hooks et al. (2008) reported that Wistar rats have a well-defined tumor profile with relatively low incidences of tumors, with only minor differences over the period of the survey or between the routes of administration (oral versus dietary). Thus, this strain continues to be suitable for use in carcinogenicity studies, particularly in view of the regulatory requirement to reach 2 years with an adequate number of survivors.
The incidence of spontaneous tumors can be influenced by various factors, including animal supplier, type of caging, laboratory practice, husbandry, and diet. As the majority of both the animals and diets were each sourced from one, although different, suppliers, and there were no noticeable differences of tumor incidences between these two suppliers and other suppliers, it is considered unlikely that any possible factors were influenced by this. Furthermore, most of these variable factors are controlled and standardized in our laboratory, where several carcinogenicity studies are conducted each year.
Another important factor is time-dependent progression of tumor occurrence (Gopinath 1994; Rao et al. 1990a, 1990b). A review of the historical control data generated from carcinogenicity studies over the time interval covered by this report has shown little variability (Son and Gopinath 2004). Hooks et al. (2008) have indicated that the time period over which studies were conducted did not greatly influence tumor occurrence in their survey, which reviewed studies conducted between 1994–1999 and 2000–2006 with Han Wistar rats. Therefore, the data in this report, which surveyed studies during approximately a 14-year period in the same laboratory, were not thought to be greatly affected by time trends for tumor incidences.
The tumors in this report were classified either as incidental or fatal tumors from the decedent animals. Incidental tumors are found either at terminal kills or in decedents that were found incidentally and did not cause the death of the animals. Fatal tumors are those that were considered causes of death. It is sometimes difficult to determine whether tumors are incidental or fatal in nature. Some intercurrent deaths from skin or mammary tumors were classified as fatal, as the size and condition of the tumors were factors in the decision to euthanatize the animal for humane reasons (Son and Gopinath 2004). Although our previous report on early-occurring tumors in Sprague-Dawley rats did not include short-term studies, this current report comprises control data not only from carcinogenicity studies but also from short-term studies. Incidences from the short-term studies are few, and there were no tumors in control animals from the shorter term toxicity studies employed in this survey. In carcinogenicity studies, the majority of tumors were classified as fatal, with only the single gonadal stromal tumor in the ovary being considered incidental. This means that early tumor occurrence in shorter term toxicity studies is extremely rare, and most that do occur are fatal tumors.
In control Han Wistar rats, the most common and earliest occurring tumor was malignant lymphoma in both sexes, the earliest being seen in the 16th and 26th weeks on study in males and females, respectively. This tumor showed a greater incidence in males, even at a young age, during the first 52 weeks of study, which is consistent with the lifetime incidence of this tumor in this strain of rat. The second most common tumor was tumors of brain in males and mammary tumors in females at this age group.
When compared with the early tumor occurrence in the Sprague-Dawley rat, pituitary tumor was the most common tumor in female Sprague-Dawley rats (Son and Gopinath 2004), whereas the most common tumor in female Han Wistar rats was malignant lymphoma. These early tumor profiles are consistent with the lifetime tumor profile in these strains. Interestingly, while no malignant schwannoma was reported in young Sprague-Dawley rats (Son and Gopinath 2004), three malignant schwannomas were seen (one in the thorax [male], one in the brain [female], and one in the uterus [female]) in this present survey.
It is hoped that the data presented here will be useful when interpreting tumor occurrence on the rare occasions they are encountered in shorter term toxicity studies, as there is little information on tumor incidences in young animals. It is not often suitable or useful to use data from 2-year studies when evaluating tumor incidences in young animals. This report provides a valuable comparison of early tumor occurrence between Sprague-Dawley and Han Wistar rats.
Footnotes
Acknowledgments
The authors would like to thank Dr. Gopinath for his scientific advice and Ms. Amie Bowen for her assistance in collection of some of the data.
