Abstract
To investigate the significance of the appearance of hepatic macrophages and expression of inflammatory factors in normal and macrophage-depleted livers, hepatic macrophages were depleted with liposome (Lipo)-encapsulated clodronate (CLD; 50 mg/kg, i.v.) followed by lipopolysaccharide (LPS) administration (0.1 mg/kg, i.p.) in F344 rats (CLD + LPS). Vehicle control rats (Lipo + LPS) received empty-Lipo before LPS. The low dose of LPS did not result in microscopic changes in the liver in either treatment group but did modulate M1 and M2 macrophage activity in Lipo + LPS rats without altering repopulating hepatic macrophages in CLD + LPS rats. LPS treatment in Lipo + LPS rats dramatically increased the M1 (IL-1β, IL-6, TNF-α, and MCP-1) but not M2 macrophage-related factors (IL-4 and CSF-1) compared to CLD + LPS rats. In the CLD + LPS rats, the M2 macrophage-related factors IL-4 and CSF-1 were elevated. In conclusion, low-dose LPS activated hepatic macrophages in rat livers without causing liver injury or stimulating repopulating hepatic macrophages. These data suggest that LPS may alter the liver microenvironment by modulating M1 or M2 macrophage-related inflammatory mediators and macrophage-based hepatotoxicity.
Hepatic macrophages play a vital role in inflammation and immune responses in the liver. Although CD163+ Kupffer cells are the predominant macrophage in the liver, there are other important hepatic macrophages that are identified by the immunophenotypic characteristics (Golbar et al. 2012). For example, CD68 is a glycoprotein on lysosomal membranes, particularly on the phagosomes of macrophages, and its expression implies enhanced phagocytosis (Zhu et al. 2012). MHC class II is expressed on mature dendritic cells and activated macrophages (Ide et al. 2005), being related to the activation of T cells and subsequent induction of other macrophages (Perrigoue et al. 2009). Galectin-3 (Gal-3) is a member of the galectin family of mammalian proteins and its expression on the activated macrophage surface may promote fibrogenesis (Dong and Hughes 1997). Ionized calcium binding adaptor molecule-1 (Iba-1) has actin cross-linking activity and may act in membrane ruffling and phagocytosis of activated macrophages (Sasaki et al. 2001). CD204 expression is related to lipid metabolism in macrophages via scavenger receptors (Greaves, Gough, and Gordon 1998). Therefore, hepatic macrophages may show heterogeneous functions depending on microenvironments. They actively participate both in the maintenance of liver homeostasis and in the development of liver pathology.
Hepatic macrophages respond to a variety of classical biological factors including bacterial toxins. Lipopolysaccharide (LPS), a potent inducer of inflammation, is the major structural component of the gram-negative bacterial cell wall. LPS elicits a wide variety of pathophysiological effects in liver. Large doses of LPS produce liver injury characterized by sinusoidal neutrophilia and midzonal hepatocellular necrosis (Yee et al. 2000). While LPS itself is not hepatotoxic at low concentrations, it stimulates an inflammatory response that may lead to liver pathology (Ganey and Roth 2001). In mammals, usually a large number of gram-negative bacteria colonize in the intestine, and thus, the liver is continuously exposed to some amounts of LPS via the translocation from the intestinal lumen into portal venous blood (Ganey and Roth 2001). Kupffer cells are located in hepatic sinusoids and are the first macrophages exposed to gastrointestinal-derived bacteria and bacterial products such as LPS. Kupffer cells phagocytize and remove most of the LPS from the circulation (Martinex and Gordon 2014). LPS stimulation of Kupffer cells is via toll-like receptors and leads to the production of inflammatory mediators and contributes to the pathogenesis of liver injury (Vodovotz et al. 2001). Therefore, the precise roles of hepatic macrophages in liver homeostasis and pathology initiated by LPS should be investigated in detail.
To better understand the definite roles of divergent macrophage populations in liver, we used selective depletion of macrophages with a single intravenous injection of liposome-encapsulated dichloromethylene diphosphonate clodronate (CLD), a bisphosphonate molecule. CLD causes selective killing of macrophages by intracellular release of the calcium-binding drug from the liposomes upon phagocytosis (Van Rooijen, Sanders, and Van den Berg 1996). The present study was undertaken to clarify the pathophysiological effect of low dose of LPS on hepatic macrophage populations and liver-specific regulatory inflammatory factors in normal and hepatic macrophage-depleted rat livers by CLD.
Material and Method
Animals and Experimental Procedures
Forty-eight 5-week-old male F344 rats purchased from Charles River Japan (Hino, Shiga, Japan) were used. Rats were maintained in a room at 21°C ± 3°C with a 12-hr light–dark cycle, fed a standard diet (DC-8, CLEA Japan, Tokyo, Japan), and supplied with tap water ad libitum. After a one-week acclimatization, the rats were randomly divided into two groups: a CLD-treated macrophage-depleted group (CLD + LPS; 24 rats) and a vehicle liposome (Lipo)-treated control group (Lipo + LPS; 24 rats). A liposome-encapsulated CLD (5 mg/ml suspension; Foundation Clodronate Liposomes, Amsterdam, the Netherlands) at the dose of 50 mg/kg body weight (Pervin et al. 2016) or Lipo was injected intravenously once via tail vein in CLD + LPS and Lipo + LPS rats, respectively. Twenty-four hours after CLD or Lipo administration, LPS was injected intraperitoneally at the dose of 0.1 mg/kg body weight (Escherichia coli 055: B5, Sigma-Aldrich, St Louis, MO) in 20 rats from each group. Four rats in each group were euthanized by exsanguination under deep isoflurane anesthesia after LPS injection (postinjection [PI]) at 6, 12, 24, 48, and 72 hr. The remaining four rats in each group were injected with sterile 0.9% NaCl instead of LPS and sacrificed immediately after injection (Lipo + LPS or CLD + LPS at 0 hr). At necropsy, blood samples were collected from the abdominal aorta and separated sera were subjected to biochemical assays for aspartate transaminase (AST), alanine transaminase (ALT), alkaline phosphatase (ALP), and γ-glutamyl transferase (γ-GTP) by SRL Inc. (Tokyo, Japan).
The animal experiments were conducted under the institutional guidelines approved by the ethical committee of Osaka Prefecture University for the Care and Use of Experimental Animals.
Histopathology and Immunohistochemistry
Liver tissues from the left lateral lobe were collected and immediately fixed in 10% neutral-buffered formalin (NBF) and periodate-lysine-paraformaldehyde (PLP) solution processed by the PLP-acetone, methyl benzoate, and xylene method (Pervin et al. 2016). NBF-fixed tissues were dehydrated and embedded in paraffin and sectioned at 3 to 4 µm in thickness. The deparaffinized sections were stained with hematoxylin and eosin (H&E) for histopathological examination.
Tissue sections fixed in PLP were deparaffinized and used in immunohistochemistry for CD163, CD68, Gal-3, Iba-1, CD204, and MHC class II staining. After pretreatment, tissue sections were stained by the Histostainer (Histofine, Nichirei Bioscience Inc., Tokyo, Japan). Briefly, sections were incubated with 5% skimmed milk for 10 min, followed by 1 hr incubation with primary antibodies. Details of antibodies are listed in Table 1. After treatment with 3% H2O2 for 15 min, horseradish peroxidase–conjugated secondary antibody (Histofine simple stain MAX PO®; Nichirei Inc., Tokyo, Japan) was applied for 30 min. Then, they were incubated with 3, 3′-diaminobenzidine (Nichirei Inc., Tokyo, Japan) for 5 min. Sections were counterstained with hematoxylin for 1 min. For negative controls, tissue sections were treated with mouse or rabbit nonimmunized serum instead of the primary antibody. Cells expressing CD163, CD68, MHC class II, Iba-1, Gal-3, and CD204 were counted per 40× field in five randomly selected areas in the perivenular (PV) and periportal (PP) areas including the Glisson’s sheath (GS) of the liver. The GS areas were comprised of interlobular connective tissues and portal triad.
Primary Antibodies Used for Immunohistochemistry and Immunofluorescence.

Double Immunofluorescence Staining
Fresh frozen liver sections from the left lateral lobe of Lipo + LPS and CLD + LPS rats collected at PI 0, 6, 24, and 48 hr were used in double immunofluorescence using CD163 in combination with CD68, Gal-3, MHC class II, and CD204. Briefly, after fixation in cold acetone: methanol (1:1) for 10 min at 4°C, the sections were incubated with 10% normal goat serum for 30 min at room temperature followed by reaction with the primary antibody overnight at 4°C. For the CD163/Gal-3 combination, both primary antibodies were used together for overnight at 4°C. After rinsing with phosphate-buffered saline, the sections were incubated for 45 min at room temperature with the secondary antibody goat anti-mouse IgG-conjugated with Alexa 568 (Invitrogen, Carlsbad, CA) for CD163 and CD204 and goat anti-rabbit IgG-conjugated with Alexa 488 (Invitrogen, Carlsbad, CA) for Gal-3. Then, the sections were incubated with the second primary antibody labeled with fluorescent dye-conjugated secondary antibody: Alexa 488-labeled CD163 (AbD Serotec, Oxford, UK) for CD163/CD204, Alexa 488-labeled MHC class II (AbD Serotec, Oxford, UK) for CD163/MHC class II, and Alexa 488-labeled CD68 (AbD Serotec, Oxford, UK) for CD68/CD163. The sections were visualized with Vectashield™ mounting medium containing 4′, 6-diamidino-2-phenylindole (Vector Laboratories Inc., Burlingame, CA) for nuclear staining and analyzed by a virtual slide scanner (VS-120, Olympus, Tokyo, Japan).
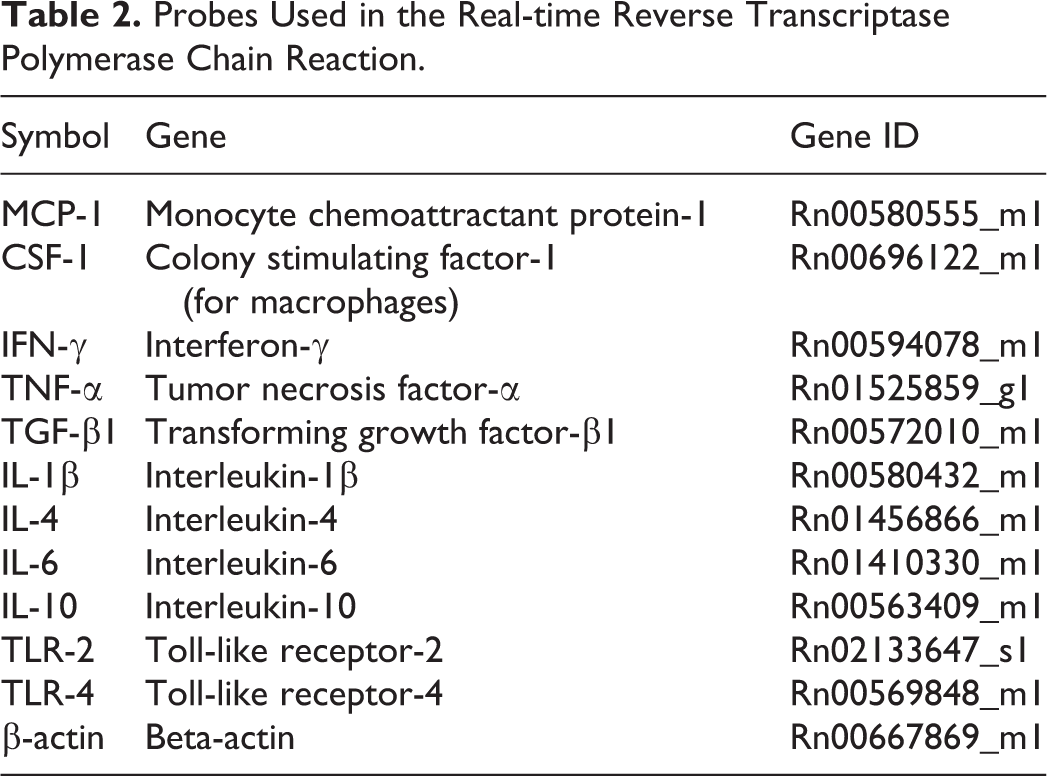
Reverse Transcriptase Polymerase Chain Reaction
Liver samples from the left median lobe were immediately immersed in RNAlater® (Qiagen GmbH, Hilden, Germany) overnight at 4°C and stored at −80°C until use. Total RNA was extracted from liver tissues by using an SV total RNA isolation system (Promega, Madison, WI) according to the manufacturer’s instructions. Two and half microgram of total extracted RNA was reverse transcribed with Superscript VILO reverse transcriptase (Life Technologies, Carlsbad, CA). Real-time PCR was performed using TaqMan gene expression assays (Life Technologies, Carlsbad, CA) in a PikoReal Real-Time 96 PCR System (Thermo Scientific, Franklin, MA). The TaqMan probes used are listed in Table 2. The messenger RNA (mRNA) expression was normalized against the expression of β-actin mRNA as the internal control. The data were analyzed using the comparative Ct method (ΔΔ Ct method).
Probes Used in the Real-time Reverse Transcriptase Polymerase Chain Reaction.
Statistical Evaluation
Data obtained were expressed as mean ± standard deviation. Statistical analysis was performed using Tukey–Kramer test. Significance was considered at p < .05.
Results
Histomorphology of Liver and Serum Biochemistry
In H&E-stained sections, the liver was unremarkable histologically at all time points after low-dose LPS treatment in both Lipo + LPS and CLD + LPS rats (Figure 1A–F). In Lipo + LPS rats, administration of low-dose LPS caused transient, mild elevations (less than 2-fold) in ALT activity but did not alter the activity of ALP or γ-GTP (Figure 2A–D). In CLD + LPS rats, administration of low dose of LPS did not change AST and ALT levels in comparison with 0 hr controls (Figure 2A and B); the values of ALP and γ-GTP were significantly increased at PI 72 hr and 24 to 72 hr, respectively (Figure 2C and D). In addition, the values of AST, ALT, ALP, and γ-GTP were significantly higher at all observation points including 0 hr in CLD + LPS rats as compared with those in Lipo + LPS rats (Figure 2A–D), which might be due to the depletion of hepatic macrophages as reported previously (Pervin et al. 2016).
(A)–(F) Hematoxylin and eosin staining of liver in liposome-treated control (Lipo + LPS) and hepatic macrophage-depleted (CLD + LPS) rats. No histological change of hepatic architecture is seen in both groups. CV = central vein; LPS = lipopolysaccharide; CLD = clodronate. Bar = 100 µm.
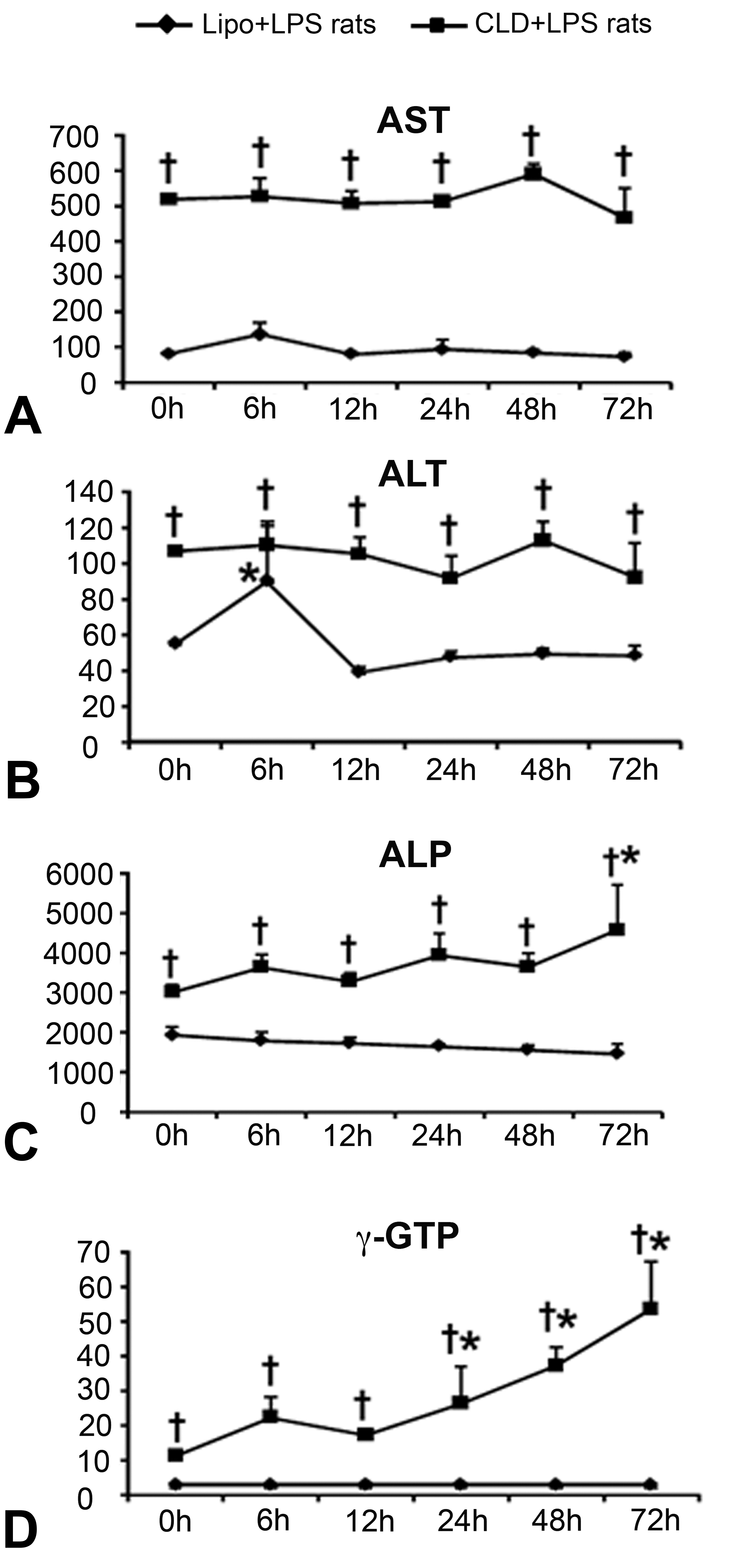
(A)–(D) Blood biochemical analyses in Lipo + LPS and CLD + LPS rats. Aspartate transaminase (A), alanine transaminase (B), alkaline phosphatase (C), and γ-glutamyl transferase (D). Tukey’s test; *p < .05, significantly different from 0 hr in Lipo + LPS and CLD + LPS rats; † p < .05, significantly different between Lipo + LPS and CLD + LPS rats at respective examination points. LPS = lipopolysaccharide; CLD = clodronate.
Kinetics of Hepatic Macrophages in Rat Livers
The kinetics of hepatic macrophages expressing CD163, CD68, Gal-3, Iba-1, CD204, and MHC class II in the PV and PP areas is shown in Figures 3A–F and 4A–F, respectively, and representative findings of CD163+ Kupffer cells are presented in Figure 5A–H for the PV areas and Figure 5I–P for the PP including the GS areas.
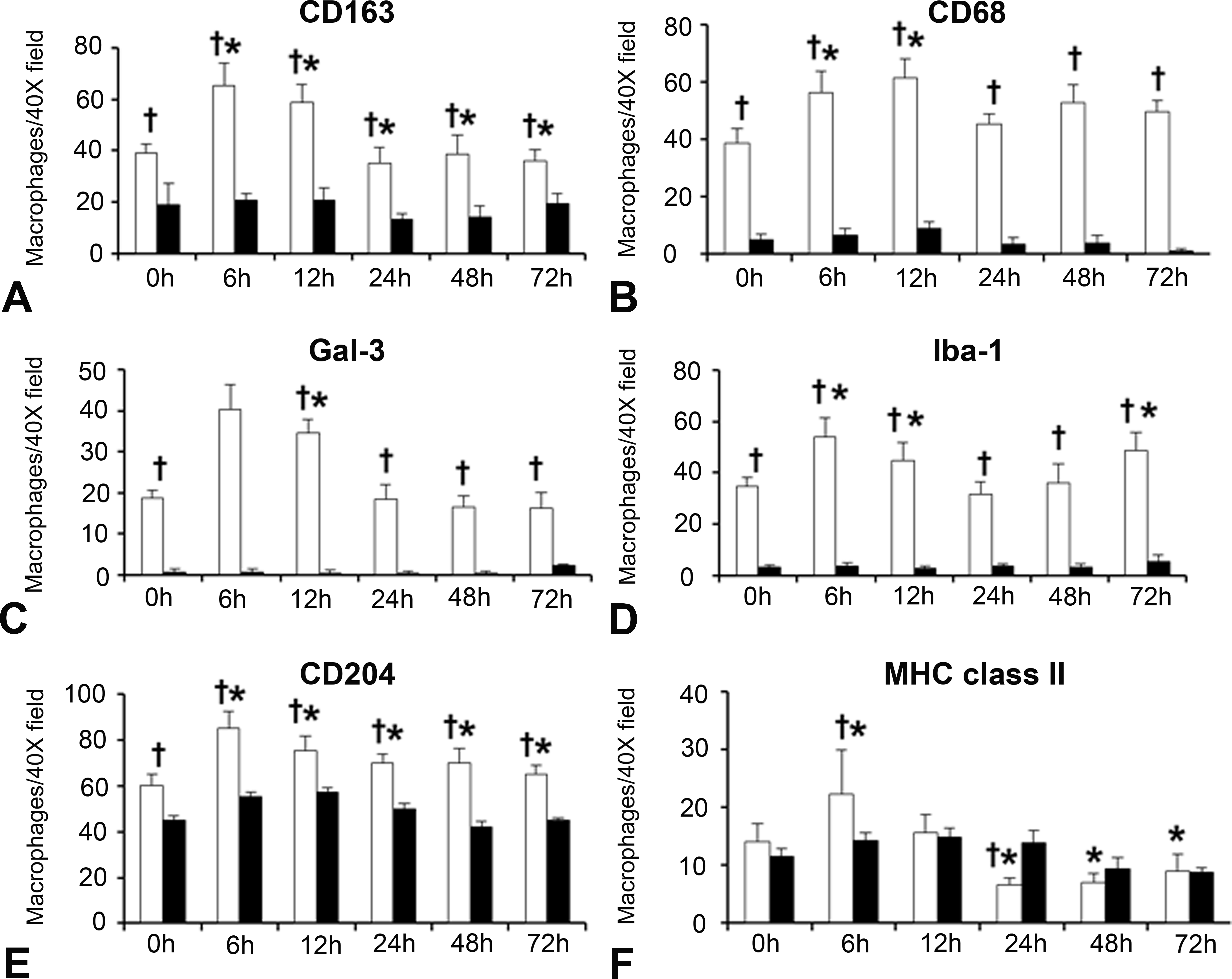
The kinetics of macrophages expressing CD163 (A), CD68 (B), Gal-3 (C), Iba-1 (D), CD204 (E), and MHC class II (F) in the perivenular area of the liver in Lipo + LPS (□) and CLD + LPS (▪) rats. Tukey’s test; *p < .05, significantly different from 0 hr in Lipo + LPS and CLD + LPS rats; † p < .05, significantly different between Lipo + LPS and CLD + LPS rats at respective examination points. LPS = lipopolysaccharide; CLD = clodronate.
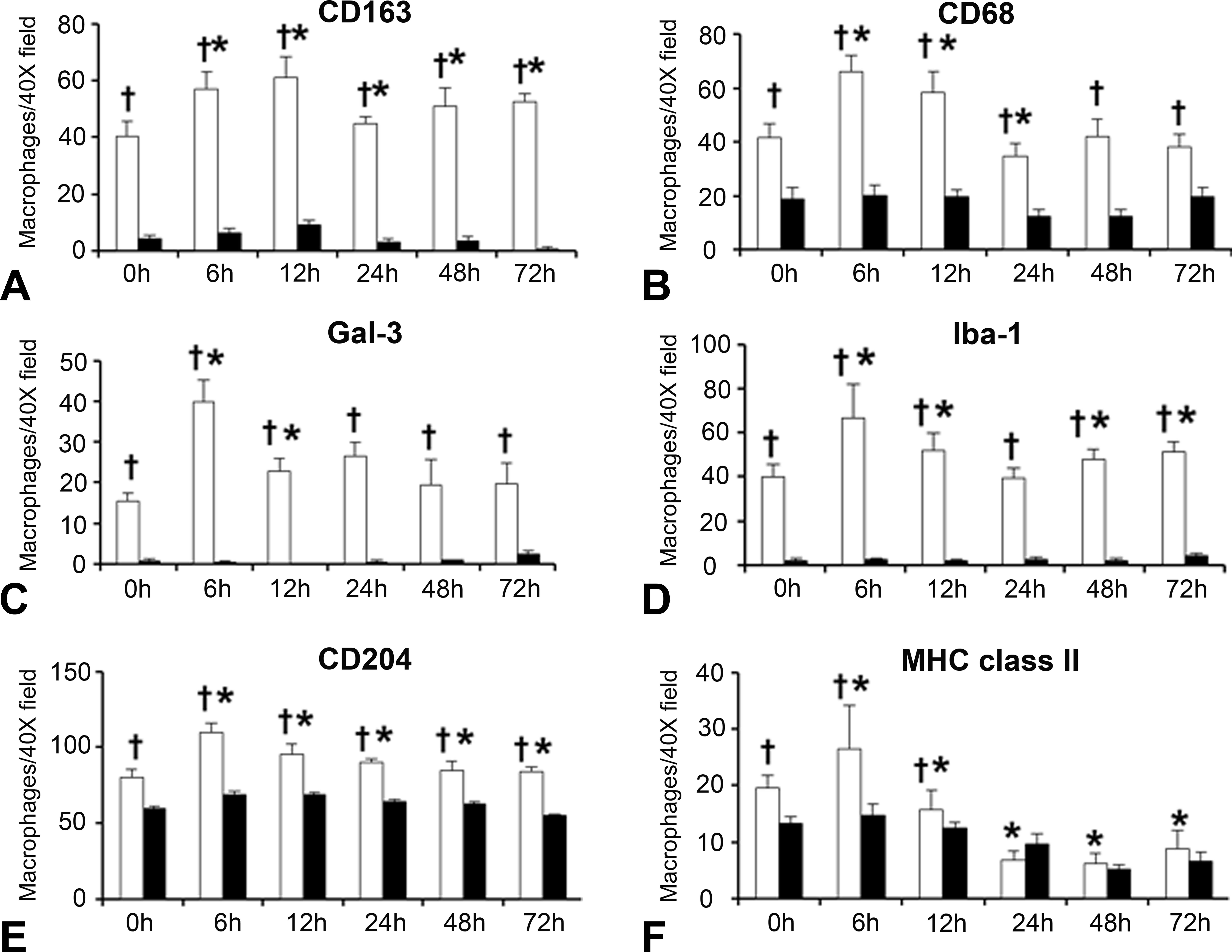
The kinetics of macrophages expressing CD163 (A), CD68 (B), Gal-3 (C), Iba-1 (D), CD204 (E), and MHC class II (F) in the periportal area of the liver in Lipo + LPS (□) and CLD + LPS (▪) rats. Tukey’s test; *p < .05, significantly different from 0 hr in Lipo + LPS and CLD + LPS rats; † p < .05, significantly different between Lipo + LPS and CLD + LPS rats at respective examination points. LPS = lipopolysaccharide; CLD = clodronate.
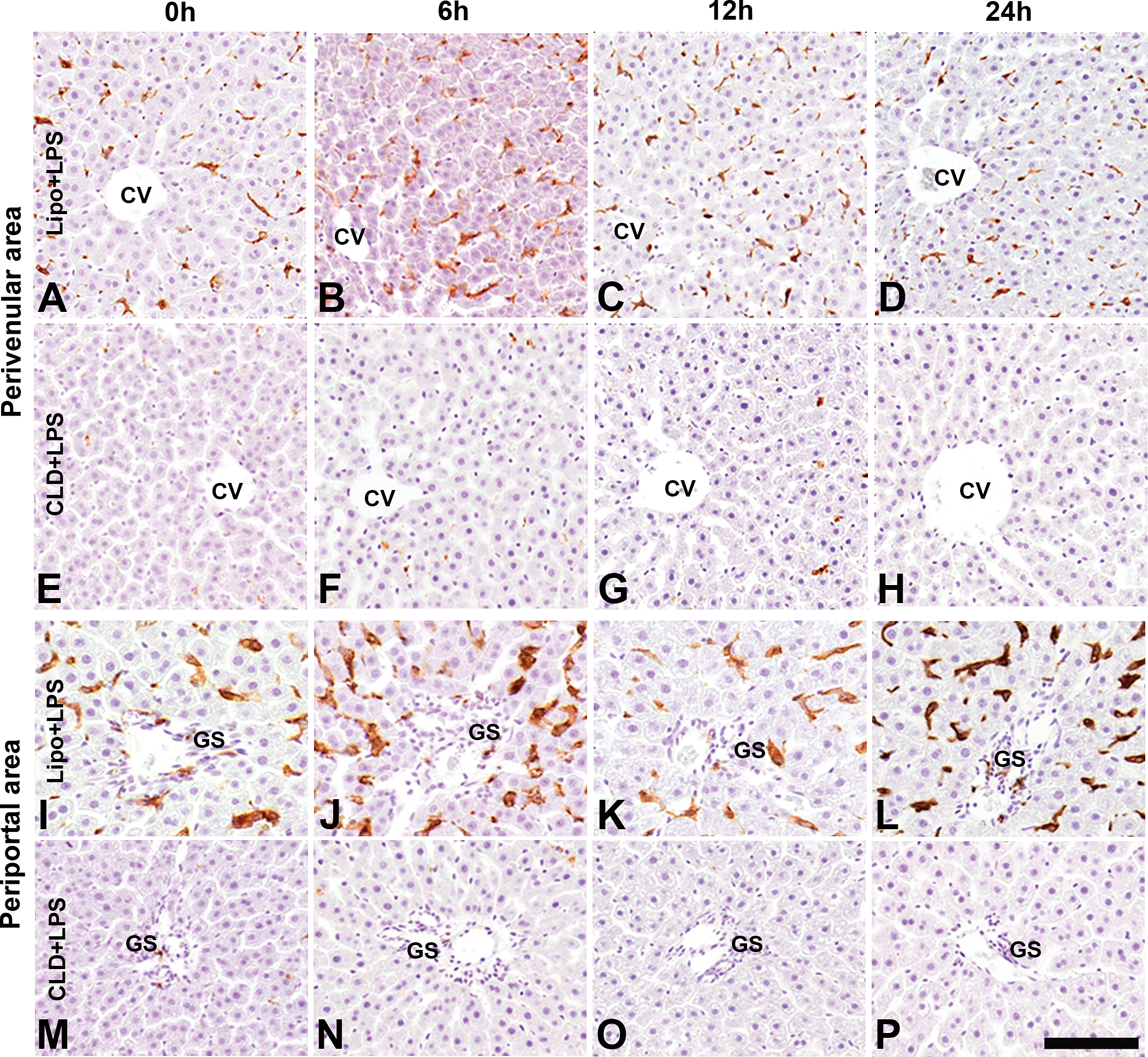
CD163+ Kupffer cells (arrows) seen along the sinusoid in the perivenular (A–H) and periportal (I–P) areas of liver parenchyma in Lipo + LPS and CLD + LPS rats. Kupffer cells are rarely seen in the perivenular and periportal areas of CLD + LPS rats. CV = central vein; GS = Glisson’s sheath; LPS = lipopolysaccharide; CLD = clodronate. Bar = 100 µm.
CD163+ Cells
CD163 antigen is generally expressed on the cell surface of resident macrophages (Kupffer cells) located mainly along sinusoids in the PV and PP areas (Golbar et al. 2012). In Lipo + LPS but not CLD + LPS rats, CD163+ Kupffer cells were significantly increased at PI 6 to 72 hr as compared with those of 0 hr controls (Figures 3A and 4A) both in PV and PP areas (Figures 3 –5). CD163+ Kupffer cells were significantly lower in CLD + LPS rats at all examination points as compared with those of Lipo + LPS rats (Figures 3A and 4A). These findings indicated that LPS is a potential activator for Kupffer cells in untreated rats.
CD68+, Iba-1+, Gal-3+, and CD204+ Cells
Exposure of LPS resulted in a significant increase in CD68+, Gal-3+, Iba-1+, and CD204+ hepatic macrophages both in PV and PP areas of Lipo + LPS rats (Figures 3B–E and 4B–E). Particularly, the numbers of CD68+ macrophages were markedly increased at PI 6 and 12 hr in PV and PP areas. However, the number of CD68+ macrophages was significantly decreased in PP areas at PI 24 hr and then returned to normal level as seen in 0 hr controls (Figures 3B and 4B). Gal-3+ macrophages were significantly increased at PI 6 to 12 hr in PV areas and at PI 6 hr in PP areas (Figures 3C and 4C). The macrophages expressing Iba-1 showed a significant increase at PI 6, 12, and 72 hr after LPS administration in PV areas, which remained increased on all observation points in PP areas (Figures 3D and 4D). The number of CD204+ macrophages showed kinetics similar to that of CD163+ macrophages. CD204+ macrophages were significantly increased throughout the observation periods as compared with those of 0 hr controls both in PV and PP areas (Figures 3E and 4E).
Hepatic macrophages reacting to CD68, Iba-1, Gal-3, and CD204 were not changed in CLD + LPS rats after LPS treatment in contrast to those of 0 hr controls (Figures 3B–E and 4B–E). CD68+, Gal-3+, Iba-1+, and CD204+ hepatic macrophages were significantly lower in CLD + LPS rats at all examination points as compared with those of Lipo + LPS rats (Figures 3B–E and 4B–E), indicating that LPS is a more potent macrophage activator in normal rat livers than in macrophage-depleted livers.
MHC Class II+ Cells
In the liver parenchyma, the numbers of MHC class II+ cells were lower in comparison with other hepatic macrophages expressing CD163, CD68, Iba-1, Gal-3, and CD204. MHC class II expressing cells were mainly located in the GS and, therefore, regarded as antigen presenting dendritic cells (Golbar et al. 2012). In Lipo + LPS rats, LPS treatment significantly increased MHC class II+ macrophage number initially at PI 6 hr in PV areas and at PI 6 to 12 hr in PP areas as compared with those of 0 hr controls. Interestingly, thereafter, the number of MHC class II+ macrophages was significantly decreased at PI 24 to 72 hr in PV and PP areas (Figures 3F and 4F). As seen in the other hepatic macrophages, no significant changes in MHC class II+ cells were observed in CLD + LPS rats (Figures 3F and 4F). In addition, between these groups, MHC class II+ cells were significantly lower in CLD + LPS rats at PI 6 hr as compared with those of Lipo + LPS rats. A significant increase in MHC class II+ cells was observed in PV areas of CLD + LPS rats at PI 24 hr as compared to Lipo + LPS rats (Figures 3F and 4F).
Immunophenotypes of CD163+ Kupffer Cells
To investigate the functional properties of Kupffer cells, double immunofluorescence for CD163+ cells with antibody against CD68, MHC class II, Gal-3, or CD204 was conducted in the liver of Lipo + LPS and CLD + LPS rats at PI 0, 6, 24, and 48 hr.
In Lipo + LPS rats, approximately 99% of CD68+ cells were also CD163+ at all examination points, whereas 70% were CD68+CD163+ in CLD + LPS rats (Figure 6A, C, and E). In Lipo + LPS rats, 80% to 90% of MHC class II+ cells showed a positive reaction to CD163; in CLD + LPS rats, the percentage of MHC class II+CD163+ cells were decreased (40–60%) at different examination points (Figure 6B, D, and F). In Lipo + LPS rats, 82 to 87% of Gal-3+ cells were also positive for CD163; the CD163+Gal-3+cell number was decreased (2–12%) in CLD + LPS rats at different examination points (Figure 6G). Almost all CD204+ cells (97%) coexpressed CD163 in livers of both groups (Figure 6H). The number of macrophages expressing CD68, Gal-3, MHC class II, and CD204 was smaller than that of CD163+ cells, indicating that CD163+ cells do not always express antigens of CD68, Gal-3, MHC class II, or CD204. However, the coexpression ratios of CD163+ Kupffer cells with other macrophage markers were lower in CLD + LPS rats in comparison with those in Lipo + LPS rats, supporting the previous conclusion that LPS treatment may increase Kupffer cell activity in normal rat livers.
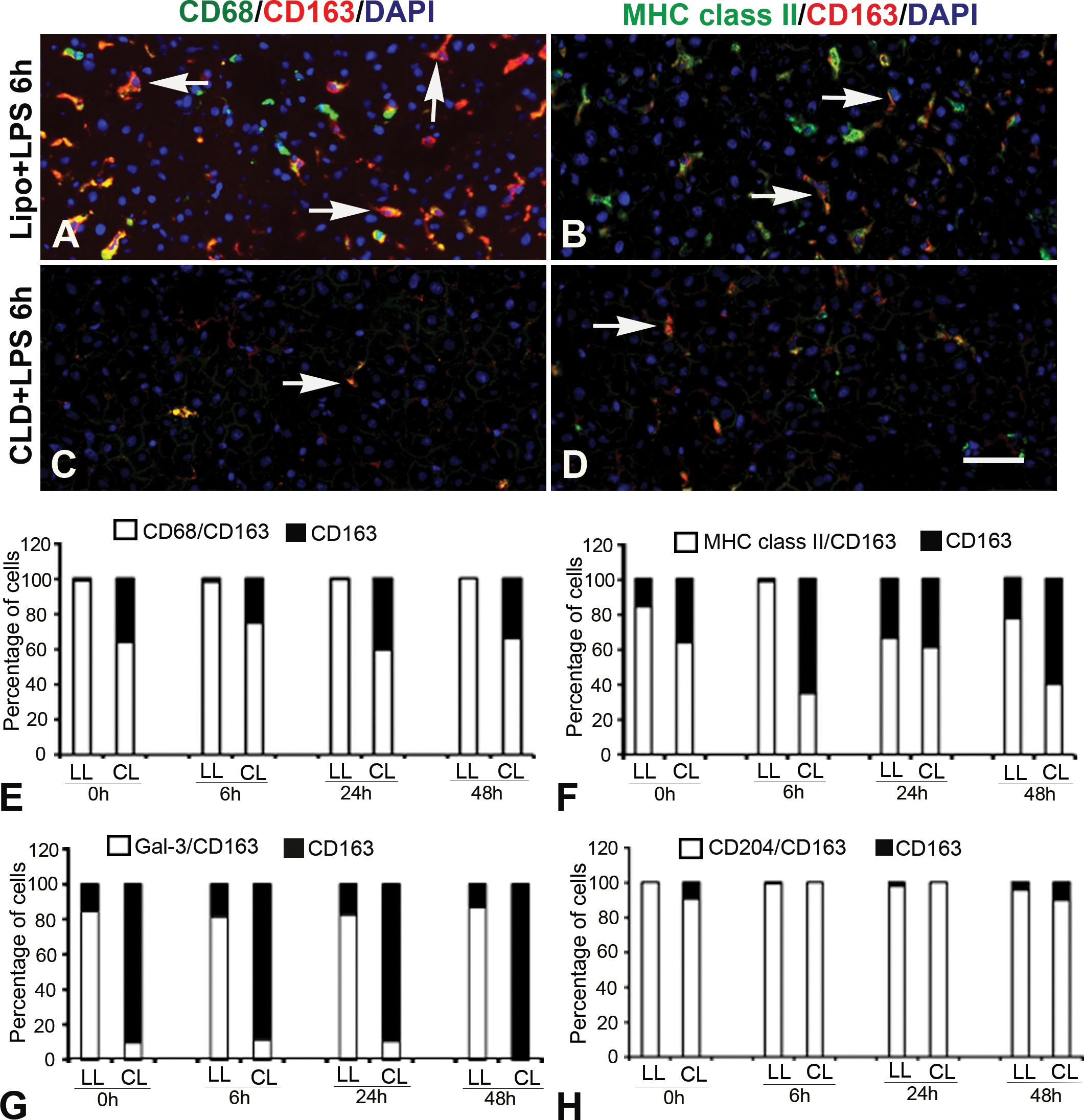
Double immunofluorescence for CD68/CD163 and MHC class II/CD163 in the livers of Lipo + LPS (A and B) and CLD + LPS (C and D) rats. Yellow color indicates double positive reaction (arrows). Blue is nuclei stained with 4′, 6-diamidino-2-phenylindole. E–H: Graph represents the percentage (%) of double positive macrophages for CD68/CD163 (E), MHC class II/CD163 (F), Gal-3/CD163 (G), CD204/CD163 (H). LL = Lipo + LPS rats; CL = CLD + LPS rats; LPS = lipopolysaccharide; CLD = clodronate. Bar = 50 µm.
Expressions of M1/M2 Macrophage-related Factors
At an mRNA level, the expression of M1 macrophage-related factors such as MCP-1, IL-1β, IL-6, TNF-α, and TLR-2 was increased as early as PI 6 hr in Lipo + LPS rats as compared with those of 0 hr controls, with a statistical significance (Figure 7A–E). On the contrary, these factors were not changed in CLD + LPS rats (Figure 7A–E). Additionally, in comparison between these groups, the mRNAs of IL-1β, IL-6, TNF-α, and TLR-2 were significantly decreased at PI 6 hr in CLD + LPS rats as compared with those of Lipo + LPS rats (Figure 7A–E). Unlike other M1 macrophage-related factors, IFN-γ-expression was significantly increased in CLD + LPS rats at PI 6 hr as compared with those of 0 hr controls, and no change was seen in Lipo + LPS rats. In between these two groups, a significantly higher expression in IFN-γ mRNA was seen in CLD + LPS rats at PI 6 hr as compared with that of Lipo + LPS rats (Figure 7F). TLR-4 mRNA was not changed in Lipo + LPS or CLD + LPS rats as compared with those of 0 hr controls; however, the expression was significantly decreased at all observation points including 0 hr in CLD + LPS rats as compared with those in Lipo + LPS rats (Figure 7G).
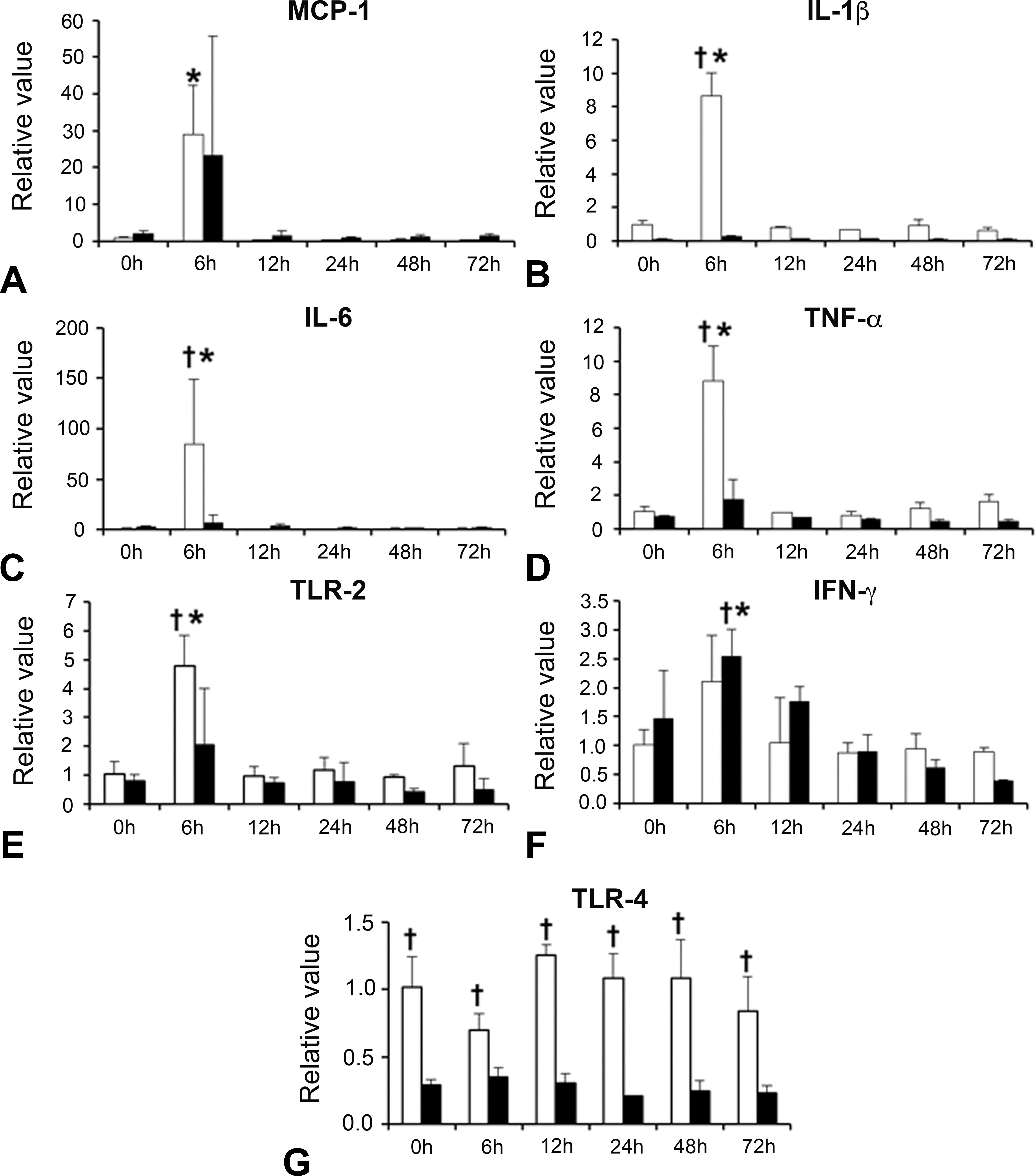
(A)–(G): mRNA expressions for M1 macrophage-related factors in Lipo + LPS (□) and CLD + LPS (▪) rats. M1 macrophage-related factors such as MCP-1 (A), IL-1β (B), IL-6 (C), TNF-α (D), TLR-2 (E), IFN-γ (F), and TLR-4 (G) are significantly increased in Lipo + LPS rats at postinjection 6 hr, showing significant difference from CLD + LPS rats. Expression levels are normalized to β-actin mRNA level. Tukey’s test; *p < .05, significantly different from 0 hr in Lipo + LPS and CLD + LPS rats; † p < .05, significantly different between Lipo + LPS and CLD + LPS rats at respective examination points. LPS = lipopolysaccharide; CLD = clodronate.
Expression of M2 macrophage-related factors such as IL-4 (Figure 8A) and CSF-1 (Figure 8B) did not show any significant change in Lipo + LPS rats after LPS treatment. Interestingly, in CLD + LPS rats, IL-4 mRNA was significantly elevated at PI 6 hr (Figure 8A); CSF-1 mRNA was also increased initially at PI 6 hr but significantly decreased later at PI 24 to 72 hr (Figure 8B). In comparison between these groups, IL-4 and CSF-1 mRNAs were significantly increased in CLD + LPS rats in contrast to those in Lipo + LPS rats at PI 6 hr (Figure 8A and B). IL-10 is a potent M2 macrophage-related factor and showed mRNA expressions similar to M1 macrophage-related factors; IL-10 mRNA was significantly increased at PI 6 hr in Lipo + LPS rats, which was reduced under macrophage depletion conditions in CLD + LPS rats (Figure 8C). There was no significant change in TGF-β1 mRNA expression after LPS treatment in Lipo + LPS or CLD + LPS rats; however, at PI 12 hr, a statistically significant higher expression was seen in TGF-β1 in Lipo + LPS rats as compared with that in CLD + LPS rats (Figure 8D).
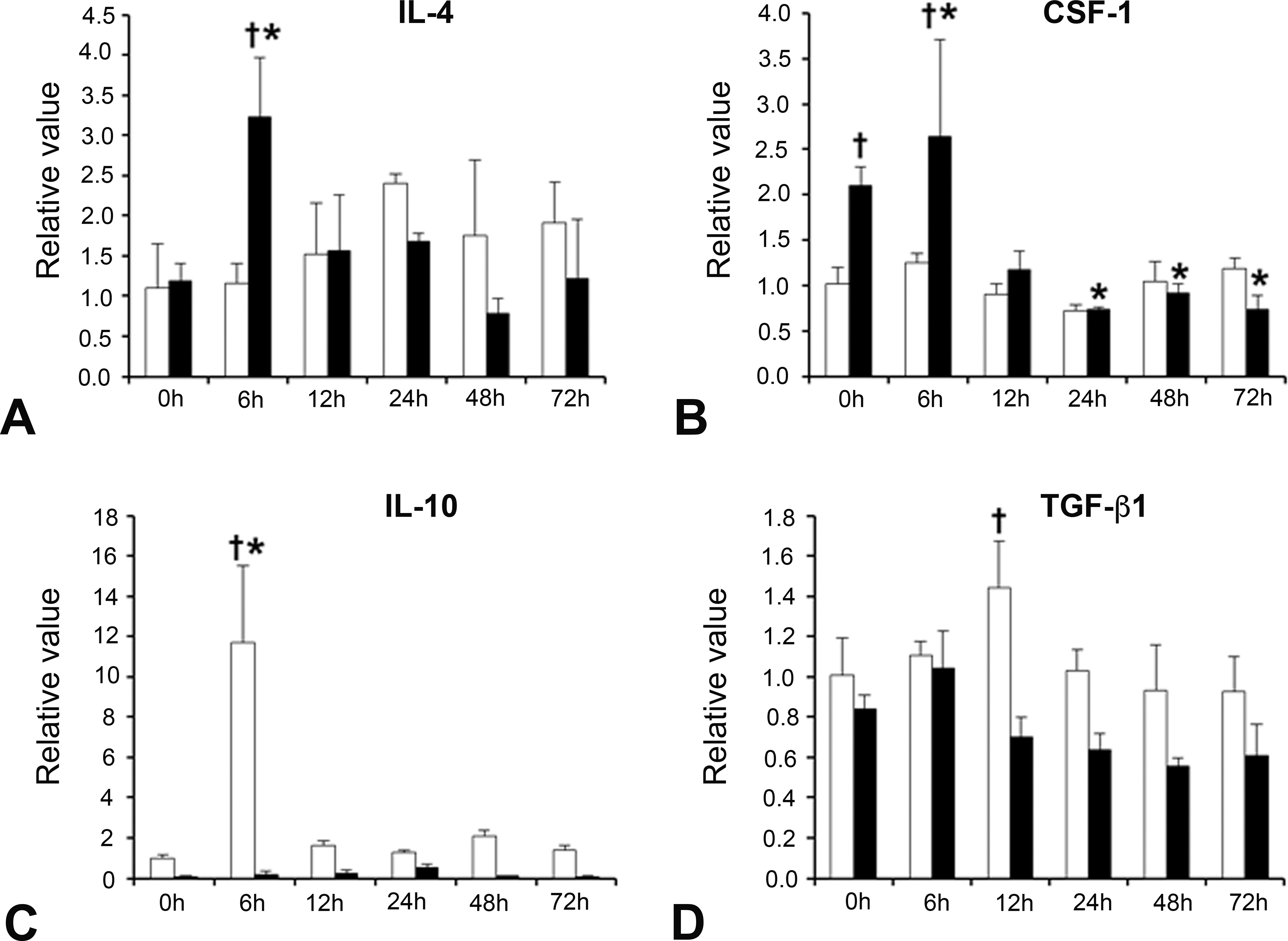
mRNA expressions for M2 macrophage-related factors such as IL-4 (A), CSF-1 (B), IL-10 (C), and TGF-β1 (D) in Lipo + LPS (□) and CLD + LPS (▪) rats. The expression of mRNA for IL-4 and CSF-1 is significantly increased in CLD + LPS rats at postinjection 6 hr, showing significant difference from Lipo + LPS rats. Expression levels are normalized to β-actin mRNA level. Tukey’s test; *p < .05, significantly different from 0 hr in Lipo + LPS and CLD + LPS rats; † p < .05, significantly different between Lipo + LPS and CLD + LPS rats at respective examination points. LPS = lipopolysaccharide; CLD = clodronate.
Discussion
Effects of LPS on Liver Histomorphology and Serum Biochemistry
Hepatic macrophages play important roles in liver homeostasis as well as liver pathology through phagocytosis, cytokine/growth factor production, and antigen presentation (Ju and Tacke 2016). At high doses in rats and in endotoxemia in humans, LPS leads to liver injury. Hepatic macrophages, especially Kupffer cells, rapidly act to remove LPS from circulation. In patients with liver failure, Kupffer cell dysfunction results in the failure of effective LPS clearance resulting in severe endotoxemia. In humans, recurrent endotoxemia may contribute to the untoward effects of environmental-borne hepatotoxicants (Liu et al. 2000; Anders et al. 2016). In the present study, the low dose of LPS (0.1 mg/kg body weight) did not result in significant liver pathology microscopically in either liposome-treated control rats (Lipo + LPS rats) or CLD-treated hepatic macrophage-depleted rats (CLD + LPS rats). Low-dose LPS treatment also only resulted in mild (<2×) and transient (only at 6 hr PI) changes in ALT activity while not affecting ALP, AST, and γGTP activities. These findings indicated that the LPS dose (0.1 mg/kg body weight) did not develop any liver lesion. On the contrary, the increased value of ALP at PI 72 hr and γ-GTP at 24 to 72 hr in CLD + LPS rats indicated that CLD pretreatment might enhance susceptibility of rat livers to hepatocellular and hepatobiliary injury because of hepatic macrophage depletion. In addition, hepatic enzymes such as AST, ALT, ALP, and γ-GTP were significantly increased in CLD-treated macrophage-depleted (CLD + LPS) rats including 0 hr before LPS injection; these increased values of hepatic enzymes might be also related to depletion of Kupffer cells because Kupffer cells have roles in liver homeostasis via clearance of such enzymes (Radi et al. 2011; Pervin et al. 2016).
Effects of LPS on Hepatic Macrophages with Different Immunophenotypes
Macrophages are heterogeneous cell populations, belonging to the mononuclear phagocyte system. Hepatic macrophages display different immunophenotypes and may exert distinct functions in hepatotoxicity (Takahashi, Naito, and Takeya 1996; Yamate et al. 2000). Based on the type 1 or type 2 helper T-cell polarization concept, macrophages appearing in pathological settings are recently grouped as classically activated macrophages (M1) and alternatively activated macrophages (M2; Martinez and Gordon 2014; Yamate, Izawa, and Kuwamura 2016). CD68, Iba-1, and MHC class II-expressing macrophages are categorized as M1 macrophages, whereas M2 macrophages express CD163, Gal-3, and CD204 (Wijesundera et al. 2014; Yamate, Izawa, and Kuwamura 2016). The appearance of different hepatic macrophages in hepatic lesions may reflect their varied roles in hepatotoxicity (Yamate et al. 2000; Ju and Tacke 2016). In this study, CLD pretreatment significantly reduced the macrophage numbers detectable by various macrophages markers (CD163, CD68, Gal-3, Iba-1, CD204, and MHC class II) throughout the observation periods in CLD + LPS rats as compared with those in Lipo + LPS rats, indicating that CLD effectively depleted both M1 and M2 macrophages.
Low-dose LPS administration resulted in significant increases in both M1 macrophages (expressing CD68, Iba-1, and MHC class II) and M2 macrophages (expressing CD163, Gal-3, and CD204) in Lipo + LPS rats. However, LPS-mediated activation of hepatic macrophages varied with their immunophenotypical characteristics. M2 macrophages express CD163 mainly on the cell surface of resident macrophages, which are regarded as Kupffer cells, and also coexpress high levels of scavenger receptor molecule type A (CD204; Gordon 2003). In this study, the numbers of CD163+ and CD204+ macrophages were significantly increased at PI 6 to 72 hr both in PV and PP areas following LPS injection in Lipo + LPS rats, which might be related to the production of proinflammatory factors and phagocytosis (Polfliet et al. 2006). M1 macrophages expressing CD68, MHC class II, and Iba-1 were activated immediately after LPS injection in Lipo + LPS rats. It is well known that M1 macrophages function mainly in early inflammatory reactions with tissue destruction (Martinez et al. 2008). There was no difference in the increase of hepatic macrophages between the PV and PP areas following low dose of LPS injection in Lipo + LPS rats; however, the macrophages in PP area remained increased during observation period in contrast to those in PV area. Because macrophages present in PP areas are closer to intralobular blood vessels in the GS, they may be quickly activated in response to LPS.
The hepatic macrophage populations were not changed in CLD-treated macrophage-depleted (CLD + LPS) rats after LPS treatment, indicating that LPS acts as a potent macrophage activator in the presence of hepatic macrophages in rat livers, possibly via TLR-dependent activation pathway (Schwabe, Seki, and Brenner 2006; Seki et al. 2007). This is supported by the increased expression of TLR-2 mRNA in Lipo + LPS rats and not CLD + LPS rats. These findings suggest that TLRs work as a receptor for LPS and that Kupffer cells are the most sensitive to LPS via TLRs (Seki et al. 2007).
Phenotypic Properties of CD163+ Kupffer Cells
Additionally, we analyzed phenotypic properties of Kupffer cells because macrophage polarizations are the major determinant of macrophage functions (Patel et al. 2012). In the present study, CD163+ Kupffer cells located along the sinusoids reacted simultaneously to CD68, Gal-3, MHC class II, or CD204 in livers of all rats; however, the expression levels of such antigens (CD68, Gal-3, Iba-1, MHC class II, or CD204) in Kupffer cells were much lower in CLD + LPS rats in comparison with those in Lipo + LPS rats after LPS treatment. Therefore, LPS treatment enhanced Kupffer cell immunoexpressions in normal rat liver in Lipo + LPS rats, resulting in presentations of various functions in Kupffer cells. However, LPS treatment did not activate repopulating Kupffer cells in livers of CLD + LPS rats. These findings indicate that repopulating hepatic macrophages after CLD treatment may be functionally less active.
Effects of LPS on Regulatory Inflammatory Factors
Kupffer cells are an important initiator of inflammatory responses to liver injury by producing M1 macrophage-related proinflammatory factors (Soehnlein and Lindbom 2010; Laskin et al. 2011; Ju and Tacke 2016). In the present study, after LPS injection at low dose, proinflammatory M1 macrophage-related factors (IL-1β, IL-6, TNF-α, and MCP1) were early increased in Lipo + LPS rats, indicating the pivotal role of Kupffer cells in the production of proinflammatory cytokines (Mori et al. 2009; Laskin et al. 2011; Zizzo et al. 2012). Increased levels of TLR-2 may promote a proinflammatory signaling pathway (Schwabe, Seki, and Brenner 2006). In addition, after low-dose LPS injection, M1 macrophage-related factors were decreased in CLD + LPS rats in comparison with those in Lipo + LPS rats, indicating that hepatic macrophages are a major source of these proinflammatory factors. Therefore, macrophage-depleted CLD + LPS rats may be more to development of hepatic lesions in the concomitant exposure of hepatotoxin. Recently, it was reported that hepatic macrophage depletion enhanced the thioacetamide-induced liver injury in rats with the prolonged hepatocyte coagulation necrosis (Golbar et al. 2016). The action of M1 macrophages is regulated by M2 macrophages, which are primarily involved in downregulation of inflammation during recovery from hepatic pathology (Martinez et al. 2008; Laskin et al. 2011). M2 macrophages, which can release anti-inflammatory cytokines (IL-4 and IL-10), contribute to the resolution of inflammation by phagocytizing cell debris and producing growth factors (TGF-β1) important in tissue remodeling (Martinez 2011; Novak and Koh 2013). In this study, M2 macrophage-related factors (IL-4 and CSF-1) were significantly increased in CLD + LPS rats and did not show significant changes in Lipo + LPS rats after LPS treatment. These findings indicate that liver-constituting cells other than Kupffer cells also play an important role in providing anti-inflammatory milieu during liver homeostasis.
In conclusion, low-dose LPS activated M1 and M2 macrophage populations in the rat liver without producing overt pathology. LPS treatment did not alter the repopulation of hepatic macrophages in macrophage-depleted rats but did modulate the production of inflammatory mediators that may enhance or suppress liver injury. Therefore, the present findings would provide the baseline information on macrophage-related microenvironments for evaluating liver injury, particularly in hepatic macrophage-based hepatotoxicity.
Footnotes
Author Contribution
Authors contributed to conception or design (MP, MRK, MKur, TI, MKuw, JY); data acquisition, analysis, or interpretation (MP, MRK, MKur, TI, MKuw, JY); drafting the manuscript (MP); and critically revising the manuscript (MP, MRK, MKur, TI, MKuw, JY). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported partly by the JSPS KAKENHI Grant Number 26292152 (to Yamate), by the Platform Project for Supporting Drug Discovery and Life Science Research (Basis for Supporting Innovative Drug Discovery and Life Science Research [BINDS]) from AMED under Grant Number JP18am0101123, and by the Ichiro Kanehara Foundation for the Promotion of Medical Science and Medical Care (The 31st International Student Grant to Karim).
