Abstract
The liver is one of the vital organs in mammals, playing roles such as nutritional metabolism and detoxification. Acute liver failure or fulminant hepatic failure is a drastic, unpredictable syndrome in which the severe deterioration of liver function rapidly occurs; such conditions may be typically followed by hepatic encephalopathy, coagulopathy, and, in some cases, progressive multiorgan failure (Bernal et al. 2010; Wlodzimirow et al. 2012). It has been reported that several chemical substances including acetaminophen, thioacetamide (TAA), and carbon tetra chloride (CCl4) can cause hepatic disorders, and these chemicals have been used to develop liver lesions as animal models (Lee et al. 2008).
Macrophages are immune cells that are widely distributed throughout tissues in the body; out of them, hepatic macrophages including Kuppfer cells and interstitial dendritic cells in the Glissons’s sheath play a crucial role in regulating liver homeostasis and hepatic injury (Varol, Mildner, and Jung 2015). Following the exposure of toxicants, hepatic macrophages can show hypersensitivity, so that abundant release of mediators including pro-inflammatory and anti-inflammatory factors occur, which can foster acute liver injury or tissue repair (injury resolution/reparative fibrosis; Varol, Mildner, and Jung 2015; Ju and Tacke 2016; Ide et al. 2003). Hepatic macrophages in normal condition are divided into three types—exudate macrophages, resident macrophages, and antigen-presenting macrophages according to their morphology and immunophenotypes (Takahashi, Naito, and Takeya 1996; Hume 2008). The massive infiltration of macrophages is also a hallmark of acute and chronic liver injury (Antoniades et al. 2012; Liaskou et al. 2013). Hepatic macrophages in pathologic condition can change microenvironments, and the microenvironments may influence functions of hepatic macrophages. Therefore, hepatic lesions/hepatotoxicity may be evaluated based on the concept of M1/M2-macrophage polarization; M1-macrophages express CD68 antigen that shows high phagocytosis and cytotoxicity, whereas M2-macrophages express CD163 antigen that may play a role in tissue repair and reparative fibrosis (Duffield et al. 2005; Martinez et al. 2008). The properties of hepatic macrophages and M1/M2-polarization in
The aims of the present study were to investigate the immunophenotypic characteristics of hepatic macrophages and also to focus on the functions of macrophages that take part in
Material and Method
Animal Model
Twenty four, six-week-old F344 male rats (110–120 g BW) were obtained from Charles River, Yokohama, Japan, and used in this experiment after one-week acclimatization. Rats were housed in an animal room maintaining at 20–22°C temperature with 12-h light–dark cycle. The animals were fed a standard diet (DC-8; CLEA Japan, Tokyo, Japan) and tap water ad libitum. Rats were injected intraperitoneally with
Histopathology and Immunohistochemistry Staining
Liver tissues were collected from the left lobes of liver sections and fixed in 10% neutral-buffered formalin (NBF) or periodate-lysine-paraformaldehyde (PLP) solution which was further processed by PLP-Amex (acetone, methyl-benzoate, and xylene) method as described (Golbar et al. 2011). NBF-fixed tissues were dehydrated, embedded in paraffin, and then cut at 4 μm thickness. Then, sections were stained with hematoxylin and eosin for histopathological examinations.
PLP-fixed tissue sections were used for immunohistochemical analyses with primary antibodies listed in Table 1. After pretreatments, Histostainer (Histofine, Nichirei Bioscience Inc., Tokyo, Japan) was used for tissue staining. Briefly, the tissue sections were incubated with 5% skimmed milk in PBS for 10 min, followed by 1 hr incubation with primary antibodies. Then, the sections were treated with 3% H2O2 in PBS for 15 min and were incubated for 30 min with horseradish peroxidase-conjugated secondary antibody (Histofine simple stain MAX PO; Nichirei Inc., Tokyo, Japan). Positive reactions were observed with 3,3’-diaminobenzidine (DAB; Nichirei Inc., Tokyo, Japan) for 10 min and the slides were counterstained lightly with hematoxylin. As negative controls, tissue sections were treated with mouse nonimmunized serum instead of the primary antibody.
Primary Antibodies Used for Immunohistochemistry.
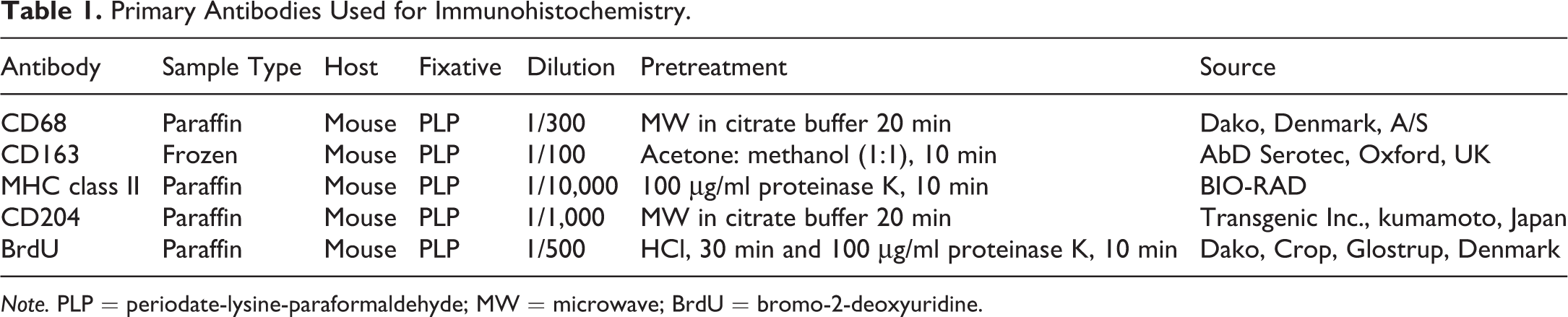
Note. PLP = periodate-lysine-paraformaldehyde; MW = microwave; BrdU = bromo-2-deoxyuridine.
Terminal Deoxyribonucleotide Transferase (TdT)–mediated Deoxyuridine Triphosphate Nick End Labeling (TUNEL)
A standard in situ TUNEL method (Appop TagR Peroxidase In situ Apoptosis Detection Kit, Millipore, Bedford) was used for the examination of apoptotic cells according to the manufacturer’s instructions. The formalin-fixed tissue sections were dewaxed, washed with PBS, and then treated with proteinase K (100 μg/ml) for 15 min at room temperature. Afterward, tissue sections were treated with 3% H2O2 for 5 min to inactivate endogenous peroxidases. Sections were incubated with TdT enzyme for 60 min at 37°C and the reaction was stopped by applying stop buffer for 10 min in room temperature. Then, sections were treated with horseradish peroxidase-conjugated anti-digoxigenin for 30 min at room temperature. The sections were then visualized with DAB. Negative control sections were incubated with distilled water instead of TdT enzyme.
Double Immunofluorescence Staining
To find out the CD68 M1- and CD163 M2-macrophage polarization, co-expression of cell specific antigens was investigated by using double immunofluorescence with combination of CD163/MHC class II, CD163/CD204, CD68/MHC class II, CD68/CD204, and CD163/CD68. Fresh frozen liver tissue sections (10-μm thick) from control and
Real-time Reverse Transcriptase Polymerase Chain Reaction (RT-PCR)
Liver samples were immersed in RNA laterR (Qiagen GmbH, Hilden, Germany) and stored at −80°C until use. Total RNA from the liver was extracted by using SV total RNA isolation system kitR (Promega, Madison, WI) according to manufacturer’s instruction. The concentration of RNA was determined by using a nanodrop 1000™ spectrophotometer (Thermo Scientific, Wilmington, DE) and 2.5 μg of total RNA was reverse-transcribed to cDNA by using a superscript VILO reverse transcriptase (Invitrogen). Real-time PCR was conducted by using TaqMan gene expression assays (Life Technologies, Carlsbad, CA) in a PikoReal Real-time PCR System (Thermo Scientific). Probes used are shown in Table 2. The mRNA expression was normalized against that of 18 s rRNA as an internal control. The data were calculated using the comparative Ct method (ΔΔCt method).
Real-time RT-PCR Probes.
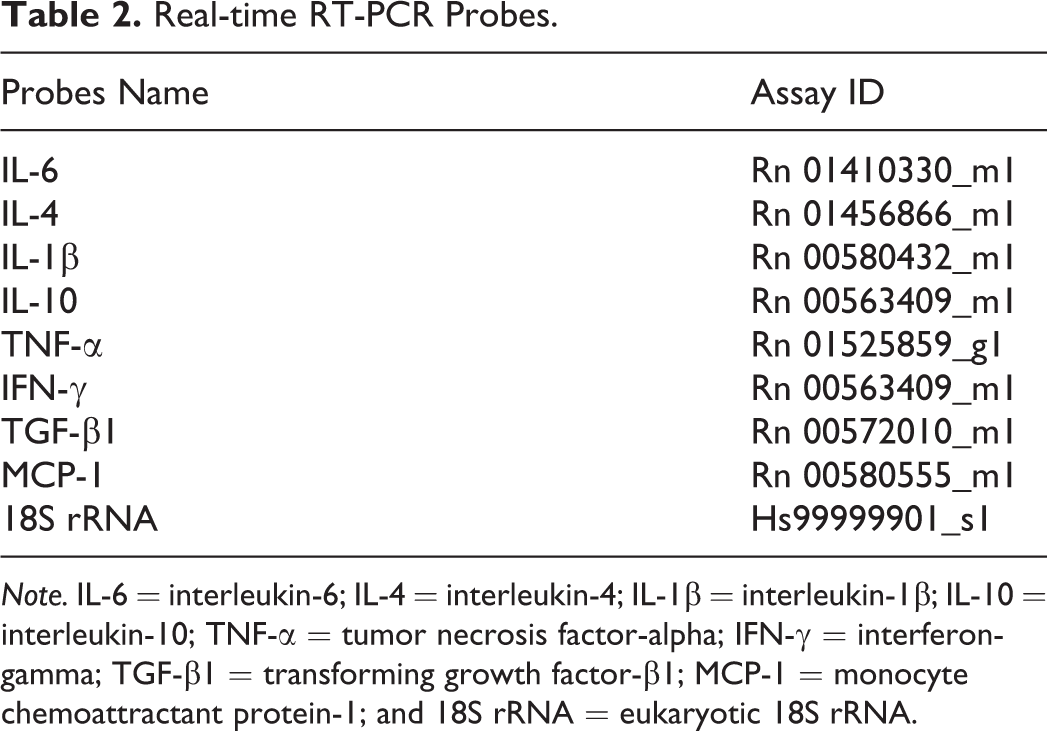
Note. IL-6 = interleukin-6; IL-4 = interleukin-4; IL-1β = interleukin-1β; IL-10 = interleukin-10; TNF-α = tumor necrosis factor-alpha; IFN-γ = interferon-gamma; TGF-β1 = transforming growth factor-β1; MCP-1 = monocyte chemoattractant protein-1; and 18S rRNA = eukaryotic 18S rRNA.
Data Analyses
Cells expressing CD68, CD163, MHC class II, and CD204 were counted per 10× field, from randomly selected five areas in the perivenular (PV) portion and periportal (PP) portion including Glisson’s sheath. BrdU and TUNEL-positive cells were counted per 40× field and were compared with the total number of hepatocytes. Quantitative data were shown as mean ± standard deviation, and statistical analysis was performed using Dunnett’s multiple comparison tests. Significance was considered at p < .05.
Results
d -GalN-induced Hepatic Lesions and BrdU Immunohistochemistry
The serum levels of AST and ALT were significantly increased on PSI days 1 and 2 with a peak on PSI day 1, in contrast to those of the control groups where serum samples were collected on PSI day 1. These levels returned quickly to control levels on PSI day 3 onward (Figure 1A and B).
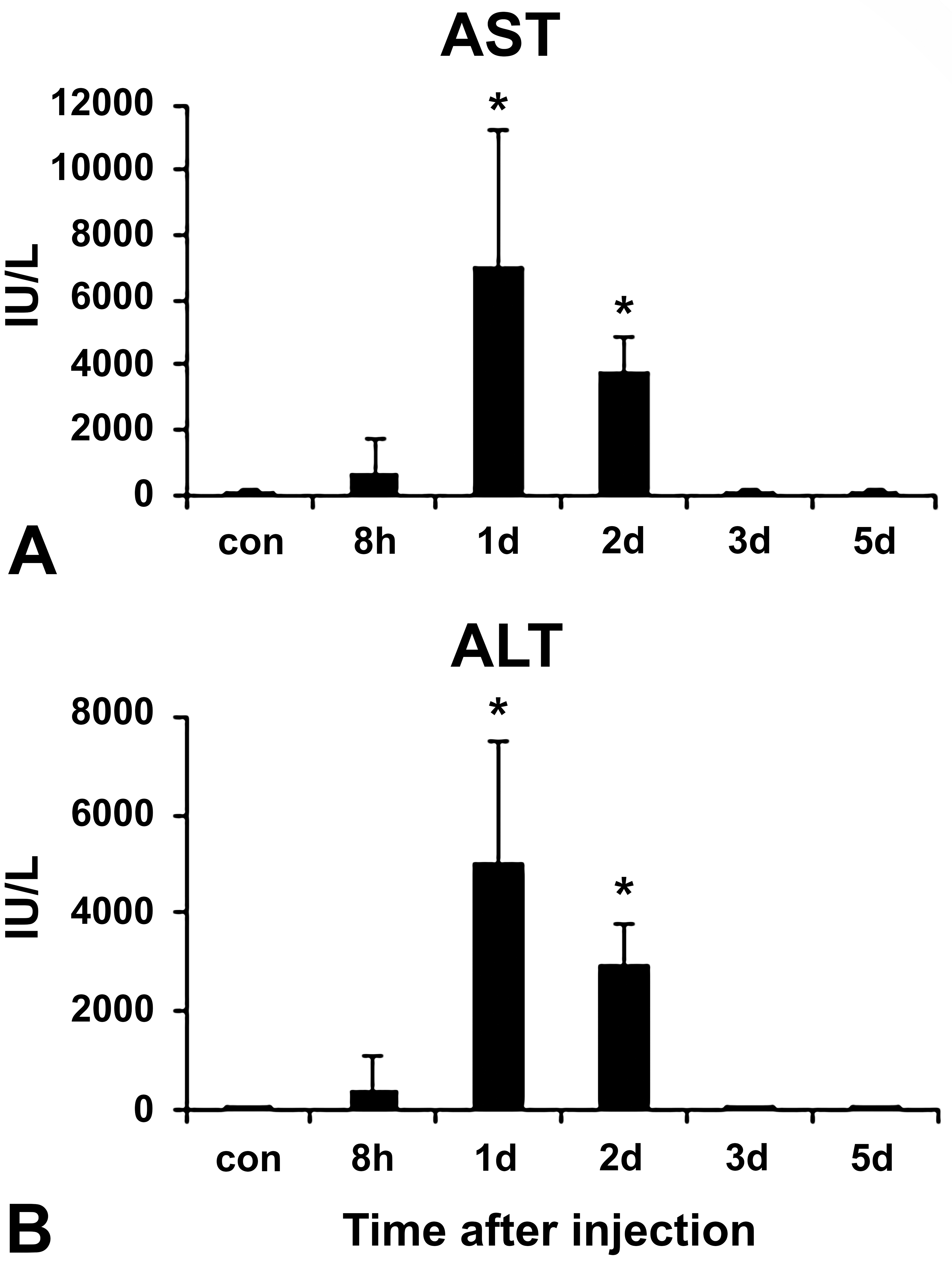
Biochemical analyses on the serum levels of aspartate transaminase (AST) (A) and alanine transaminase (ALT) (B) in control and
Histological lesions were shown in Table 3. Hepatic lesions induced by
Time-dependent Histological Changes in the Liver after

Note. PV = perivenular; PP = periportal.
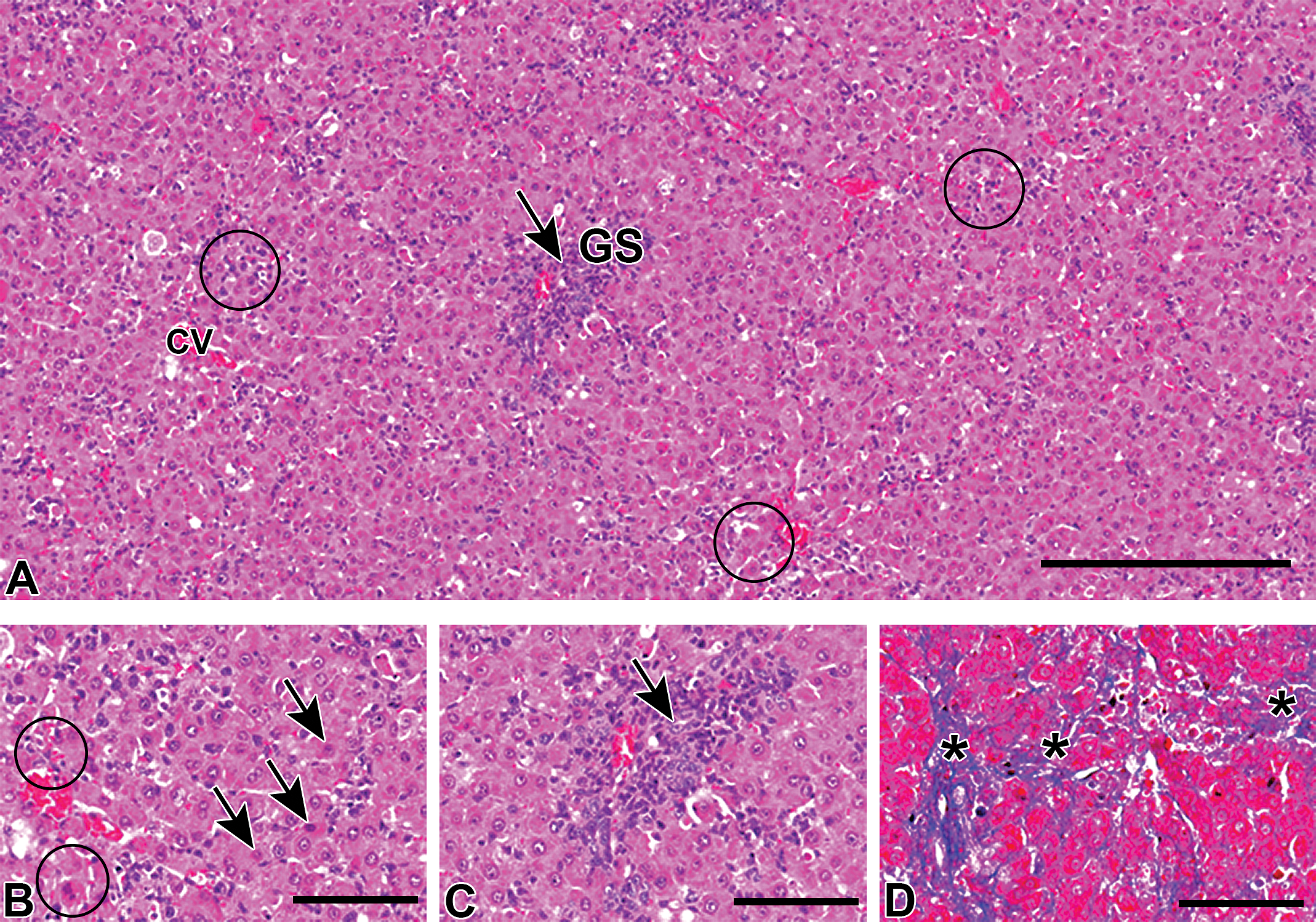
Histopathological changes of livers in
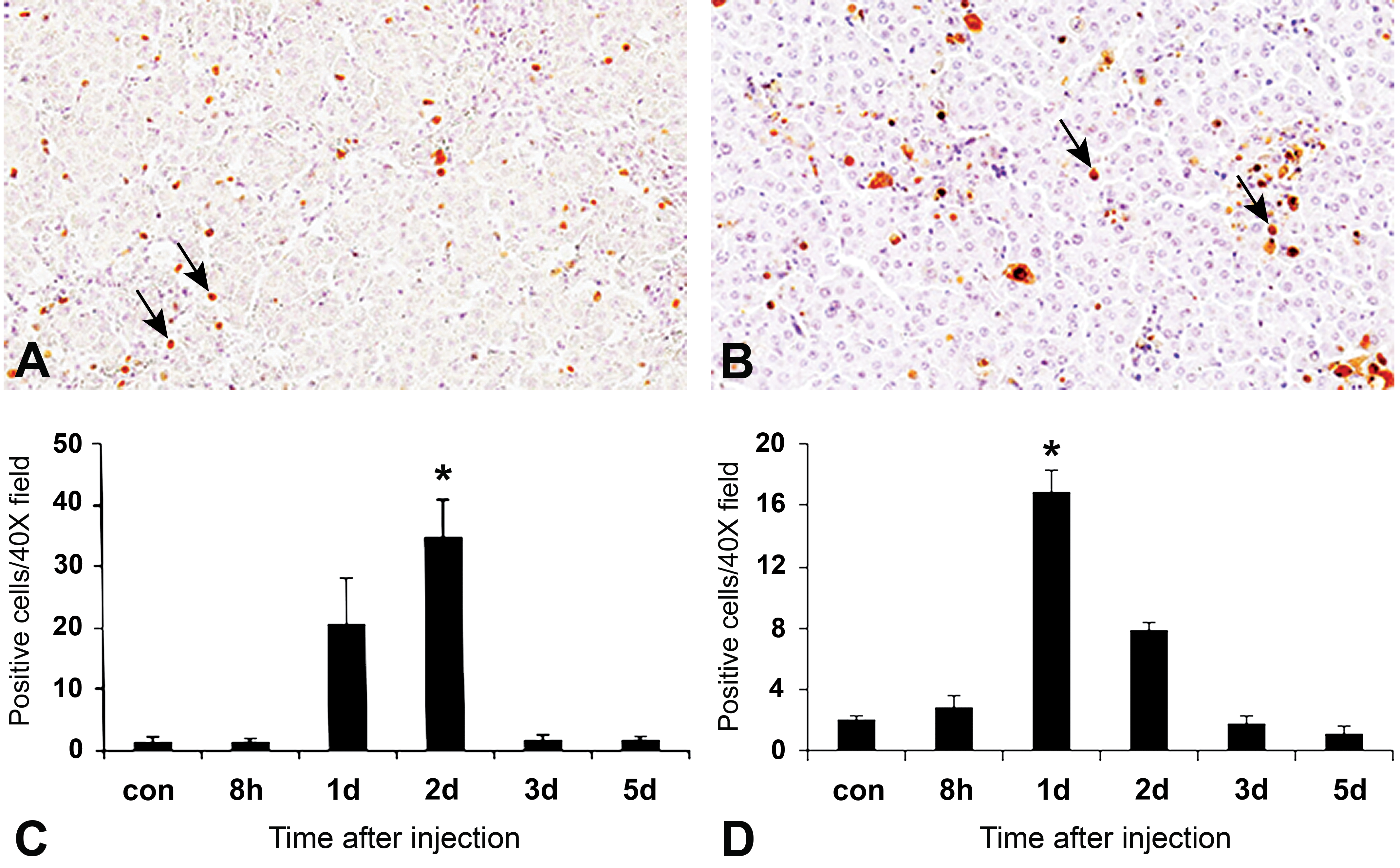
Proliferating marker bromo-2-deoxyuridine (BrdU) (A and C) for proliferating cells, as well as terminal deoxyribonucleotide transferase–mediated deoxyuridine triphosphate nick end labeling (TUNEL) method for apoptosis (B and D) in
TUNEL Method for Apoptosis
There were a few TUNEL-positive cells in control livers. TUNEL-positive cells, indicative of apoptotic cells and hepatocyte necrosis, were seen in the injured areas (Figure 3B), and a significantly increased number of positive cells were seen on PSI day 1 compared to that of controls (Figure 3D), indicating the involvement of apoptosis in the
Immunophenotypes of Hepatic Macrophages
For immunophenotypical analyses, we used four different antibodies against CD68 (for M1-macrophages), CD163 (for M2-macrophages), MHC class II, and CD204. The kinetics of macrophages positive for these antibodies is shown in Figure 4A–D.
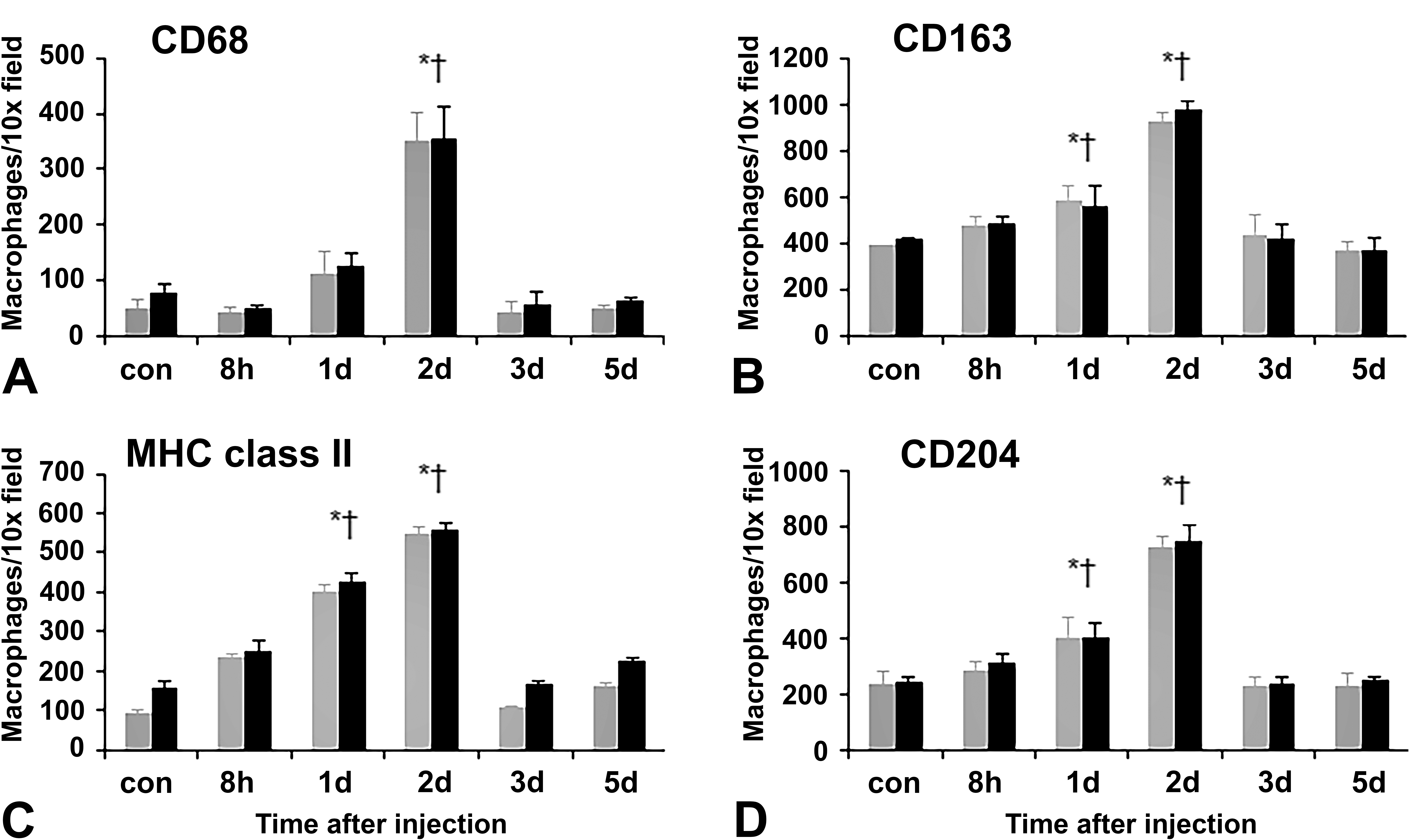
Kinetics of macrophages reacting to CD68 (A), CD163 (B), MHC class ΙΙ (C), and CD204 (D) in PV and PP areas including Glisson’s sheath of livers of control and
CD68 and CD163 Immunostaining
The number of CD68-positive M1-macrophages in the PV and PP areas was gradually increased, showing a significant increase on PSI day 2, and then, returned to the control level on PSI day 3 onward (Figure 4A). CD163-positive M2-macrophages were seen along the sinusoids in control livers, indicative of Kupffer cells (Golbar et al. 2012); the number of cells reacting to CD163 in the PV and PP areas was significantly increased on PSI days 1 and 2 with a peak on PSI day 2, and the positive cells returned to control levels on PSI day 3 onward (Figure 4B). The presence of both CD68- and CD163-positive cells was frequently seen in the injured areas of PV and PP areas (Figure 5B and D; Online Supplemental Figure 1B and D). Morphologically, CD68-reacting cells showed round to spindle configuration (Figure 5B, inset), whereas CD163-positive cells were large round to oval in shape (Figure 5D, inset). The numbers of CD163 cells were consistently greater than that of CD68 cells (Figure 4A and B).
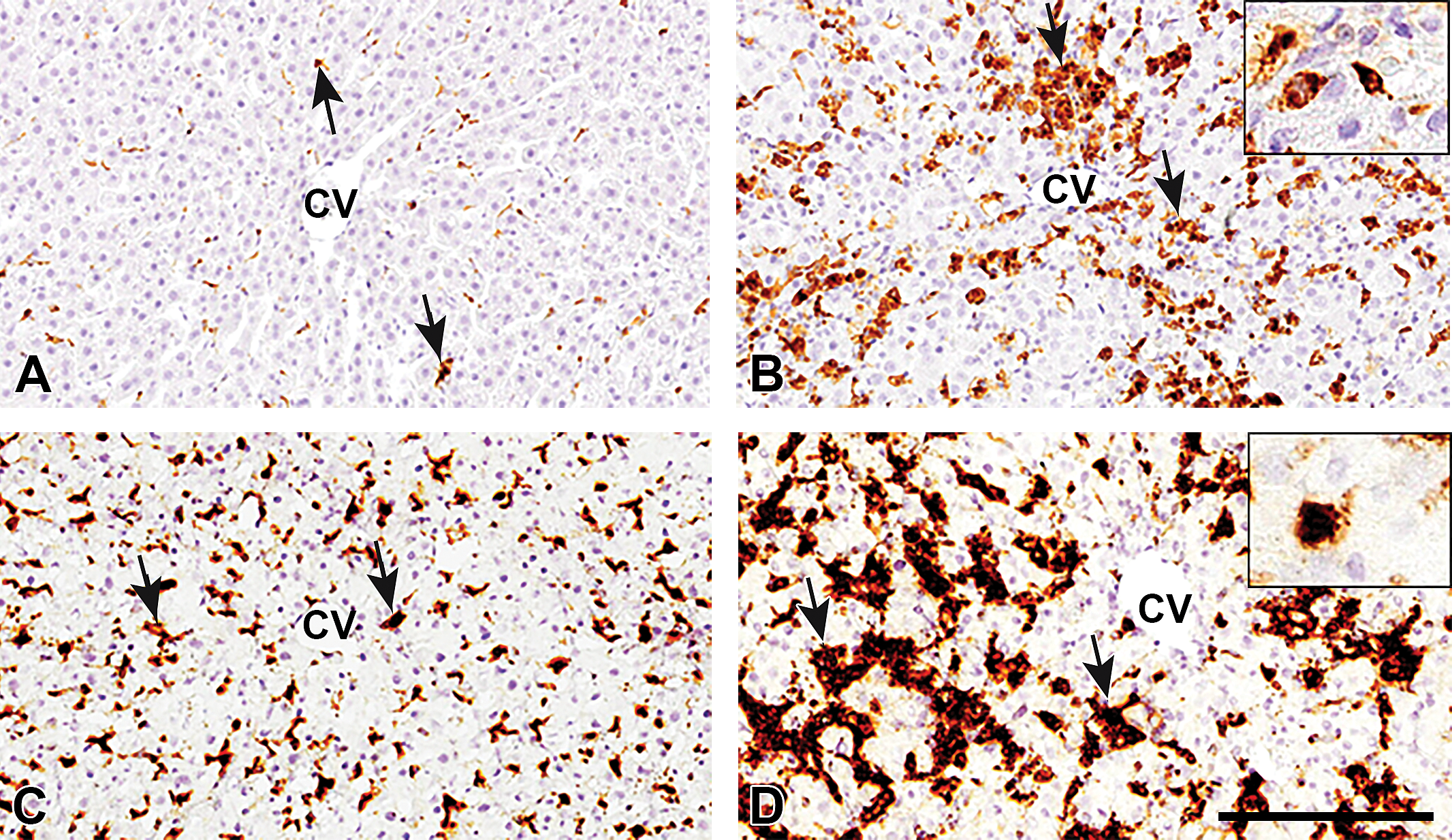
Immunoreactivity in the perivenular (PV) areas of livers of control (A, C) and
MHC Class II and CD204 Immunostaining
In control livers, a small number of MHC class II–positive cells was seen in the PV and PP areas. In
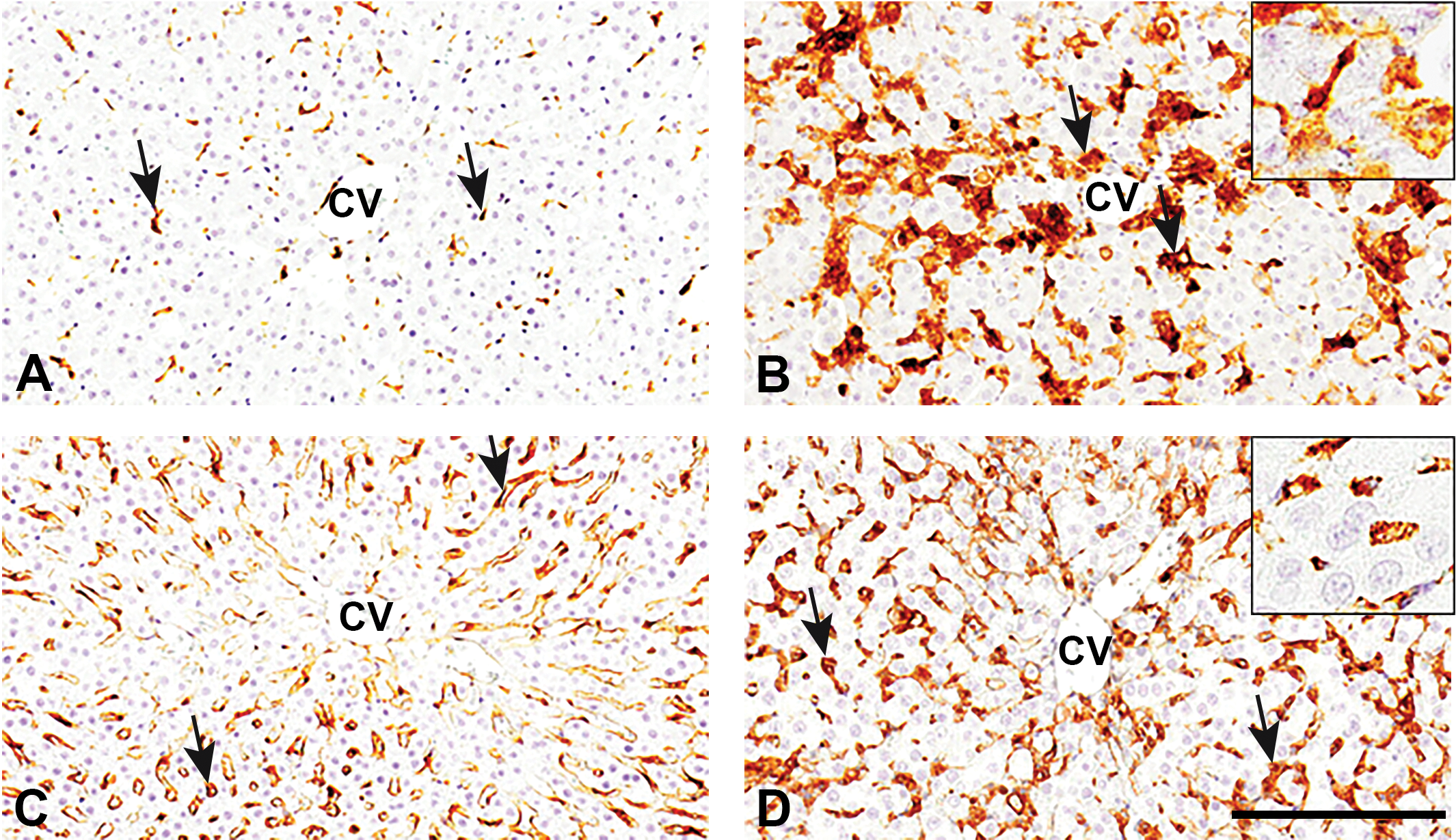
Immunoreactivity in perivenular area of livers of control (A, C) and
Cells reacting for CD204 were frequently seen in control livers. The number of positive cells in
Double Immunofluorescence Staining for CD68 M1-macrophages or for CD163 M2-macrophages
Double immunofluorescence staining with combination of CD163/CD68 (Figure 7A and B), CD163/MHC class II (Figure 8A and B), CD68/MHC class II (Figure 8C and D), CD163/CD204 (Figure 9A and B), and CD68/CD204 (Figure 9C and D) was conducted using samples obtained in control and PSI day 2. In control livers, 63% of CD163-positive Kupffer cells reacted simultaneously to CD68 (Figure 7A; Online Supplemental Figure 3); the percentage of double-positive cells for CD163/CD68 on PSI day 2 was 82% (Online Supplemental Figure 3). Similarly, a small number of MHC class II–positive cells in control livers co-expressed CD163 (9%) and CD68 (7%; Figure 8A and C; Online Supplemental Figure 4A and B); the percentages of double-positive cells for CD163/MHC class II and CD68/MHC class II on PSI day 2 were 65% and 75%, respectively (Figure 8B and D; Online Supplemental Figure 4A and B). These findings indicated an increase in double-positive cells for CD163/CD68 and CD163/MHC class II or CD68/MHC class II after
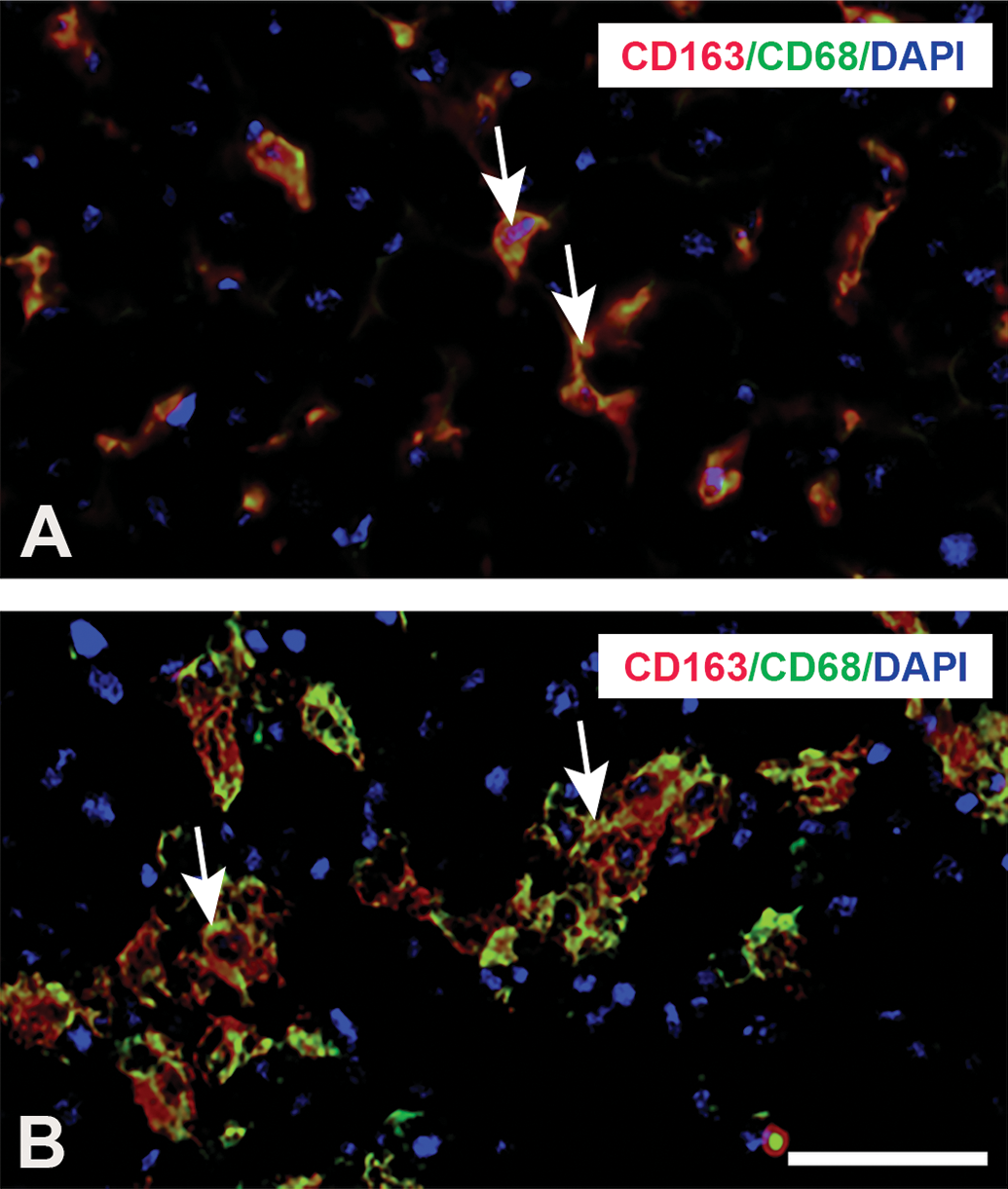
Double immunofluorescence staining for CD163/CD68 in livers of control (A) and
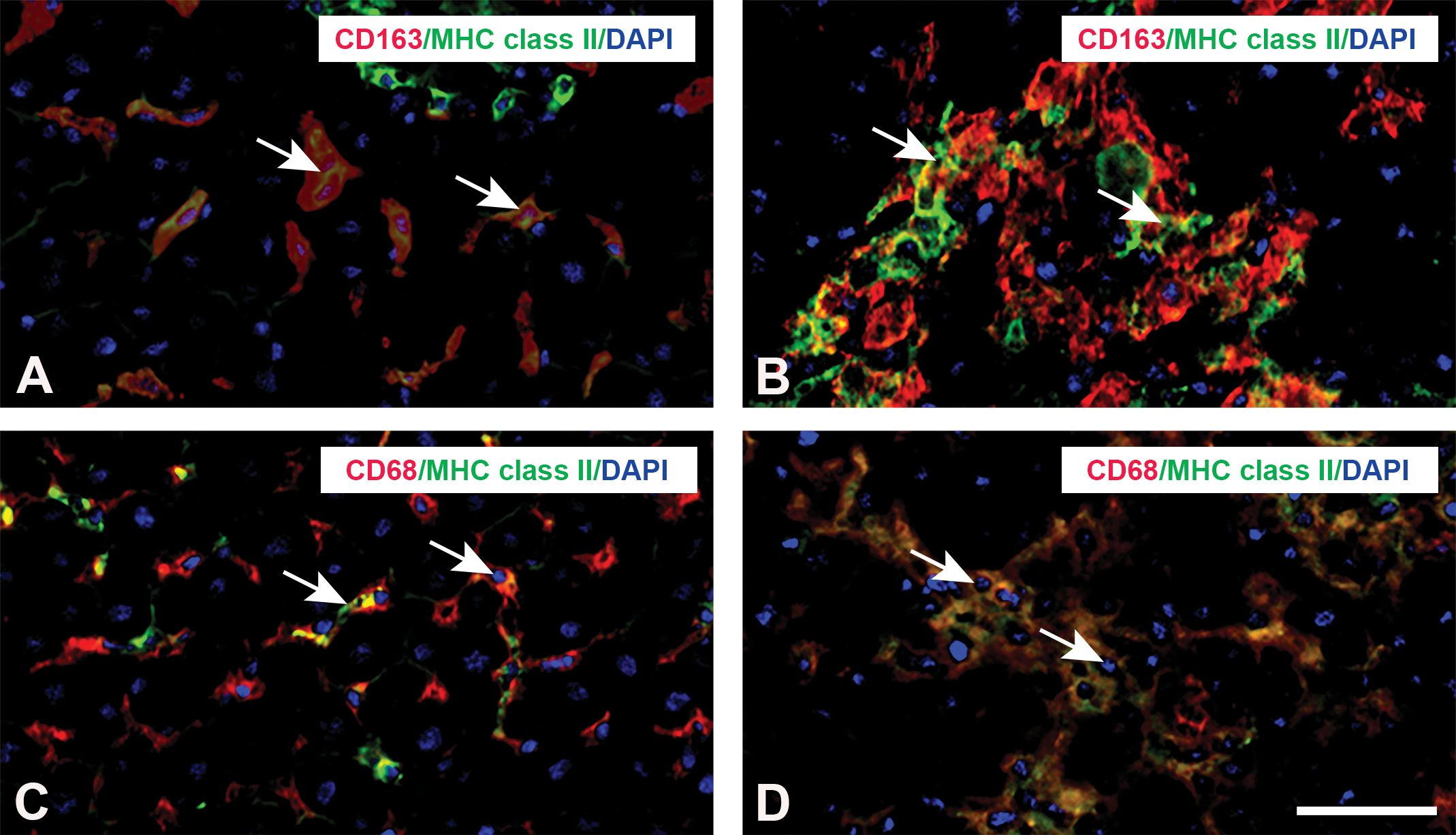
Double immunofluorescence staining for CD163/MHC class ΙΙ (A and B), CD68/MHC class ΙΙ (C and D) in livers of control, and
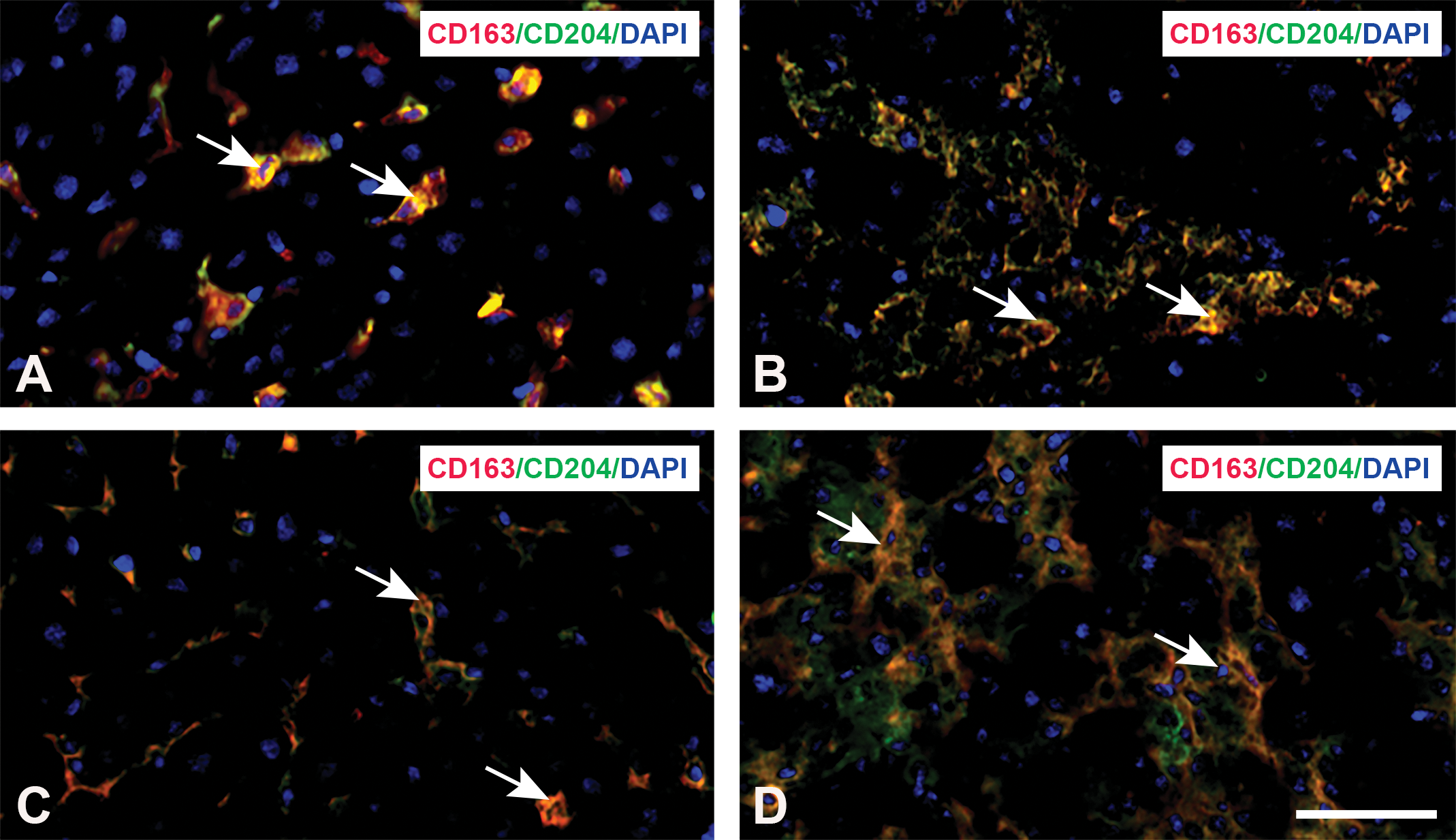
Double immunofluorescence staining for CD163/CD204 (A and B) and CD68/CD204 (C and D) in livers of control and
mRNA Expressions of Inflammatory (M1-macrophages)/Anti-inflammatory (M2-macrophages) Cytokines
M1-macrophage-related cytokines (tumor necrosis factor [TNF]-α, interleukin [IL]-6, and interferon-γ) were significantly increased on PSI days 1 and 2 (Figure 10A–C) compared to those of controls. However, IL-1β was decreased significantly at PSI 8 hr and on days 3 and 5 (Figure 10D).
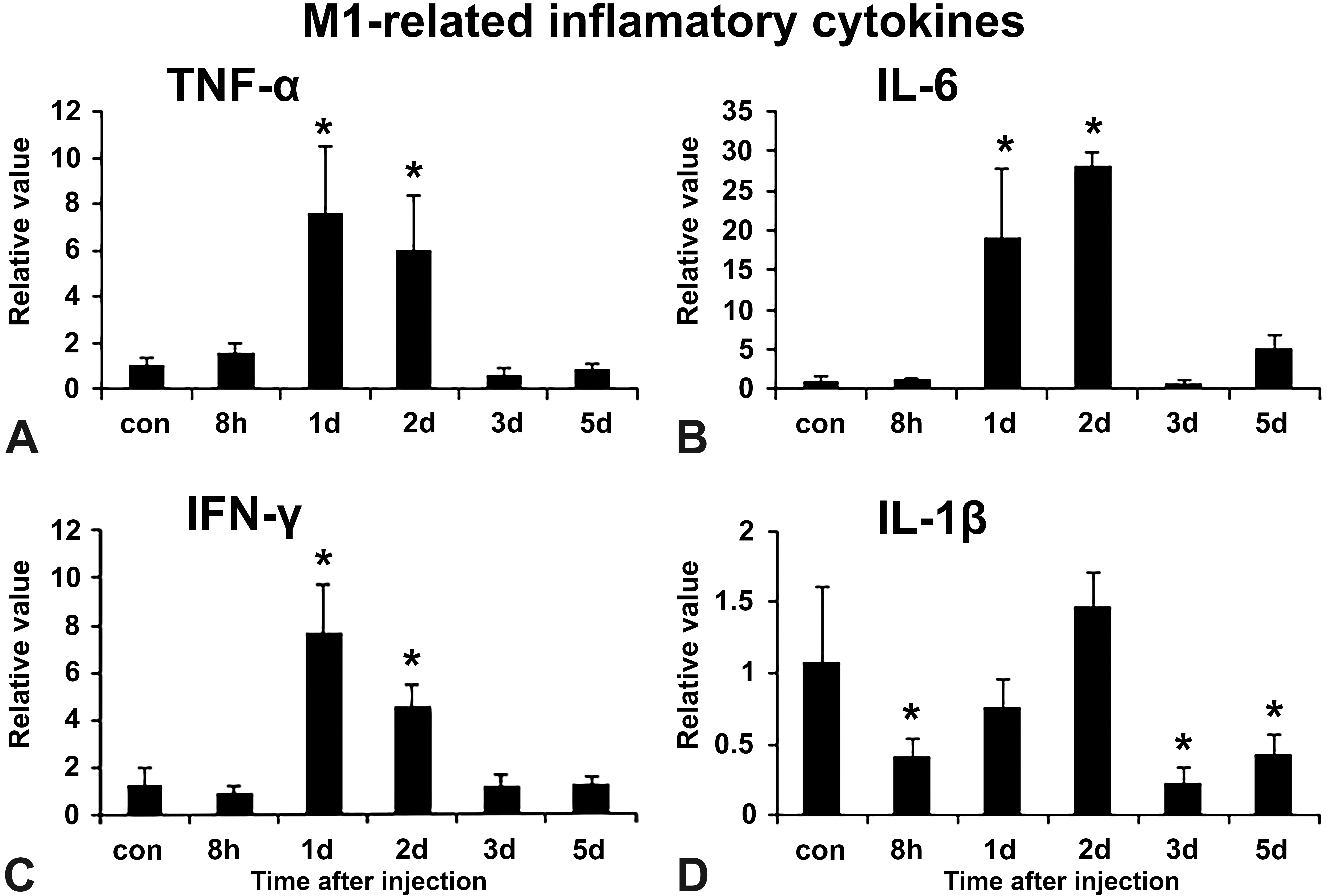
mRNA expression of M1-macrophage-related inflammatory cytokines such as tumor necrosis factor-α (A), interleukin (IL)-6 (B), INF-γ (C), and IL-1β (D) in control and
M2-macrophage-related cytokines such as transforming growth factor (TGF)-β1, monocyte chemoattractant protein (MCP)-1, and IL-4 were significantly increased on PSI days 1 and 2 (Figure 11A, C, and D), and IL-10 expression showed a peak on PSI day 2 with a significant change (Figure 11B).
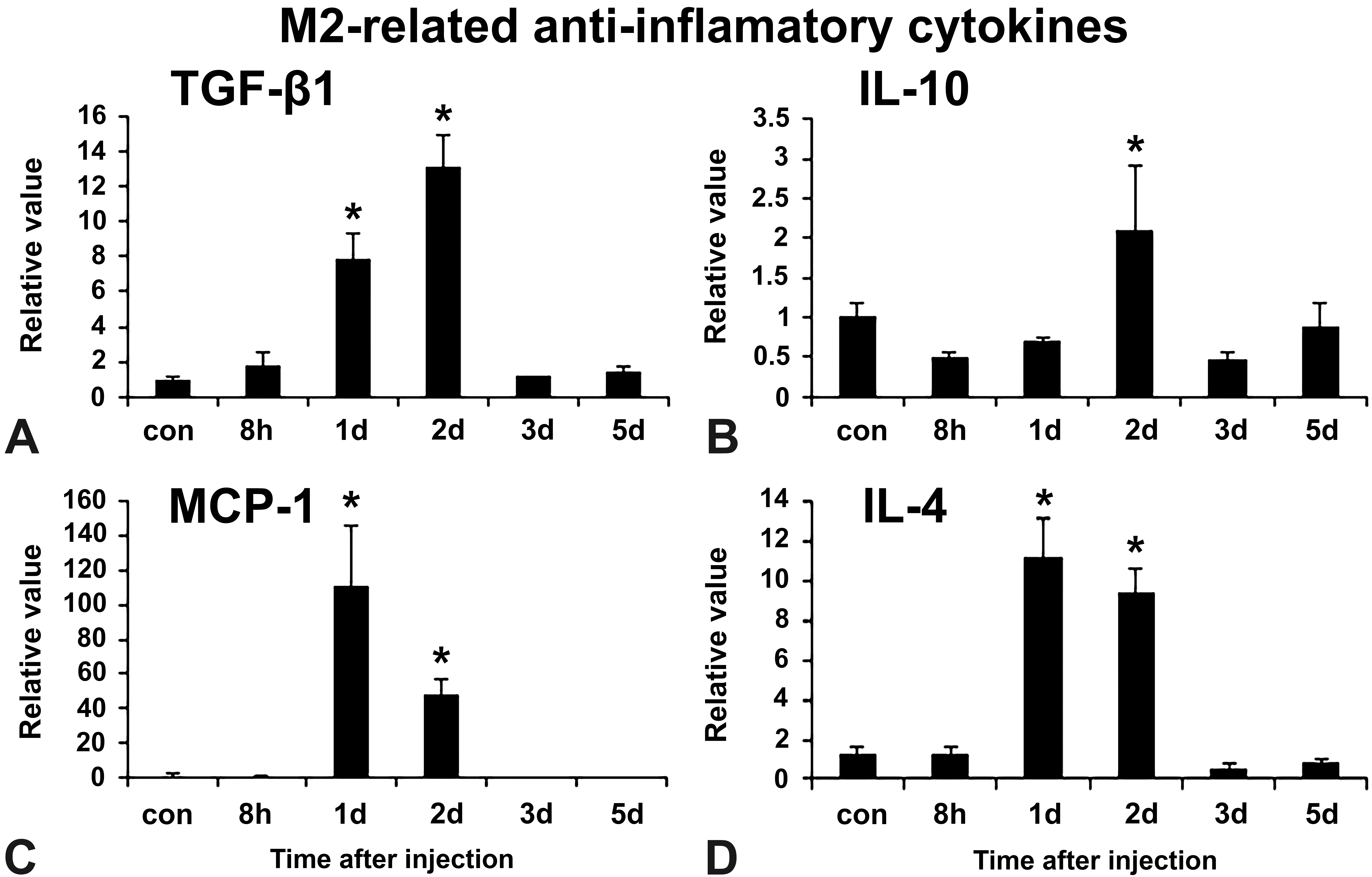
mRNA expression of M2-macrophage-related anti-inflammatory cytokines such as transforming growth factor-β1 (A), interleukin (IL)-10 (B), monocyte chemoattractant protein (MCP)-1 (C), and IL-4 (D) in control and
Discussion
Interestingly, the dose (800 mg/kg BW) used in this study developed hepatic lesions on PSI days 1 and 2; then, the lesions quickly recovered on PSI day 3 onward, regardless of multifocal necrosis. The lesion development was reflected by significantly increased levels of serum AST and ALT on PSI days 1 and 2. Interestingly, along with multifocal necrosis and inflammatory cell reactions, reparative fibrosis (characterized by the appearance of mesenchymal fibroblasts and collagen deposition) occurred simultaneously on PSI days 1 and 2. In addition, TUNEL-positive cells (indicative of single-cell death) on PSI day 1 and regenerating hepatocytes (demonstrable with BrdU staining) were seen on PSI day 2, with a statistically significant change. In TAA-induced hepatic lesions seen in the PV areas that were reported previously in our laboratory, the reparative fibrosis was followed by coagulation necrosis on subsequent days with still increased values of AST and ALT (Ide et al. 2003).
Generally, M1-macrophages are regarded as promoter of injury by producing cytotoxic/pro-inflammatory factors such as INF-γ, TNF-α, and IL-1β (Martinez 2011; Pello et al. 2011), whereas M2-macrophages may be related to tissue repair/reparative fibrosis by producing IL-4, IL-10, MCP-1, and TGF-β1 (Sica and Mantovani 2012; Njoku et al. 2009). In response to cytokines, macrophages can express specialized and polarized functional properties (Gordon and Taylor 2005). These M1- and M2-macrophages could modify hepatic lesions induced by hepatotoxicans (Wijesundera et al. 2014). It is interesting to evaluate the hepatotoxicity based on M1- and M2-macrophage functions, along with direct influences on hepatocytes by hepatotoxicans.
Usually, in immunohistochemical analyses, M1-macrophages are identified by the expression of CD68, whereas M2-macrophages by CD163. In the present
Additionally, in double immunofluorescence on PSI day 2, approximately 82% of hepatic macrophages expressed simultaneously with CD163/CD68, in contrast to controls (63%); the increased percentage of CD163/CD68-double-positive macrophages might indicate that CD163-expressing Kupffer cells became CD68-expressing M1-macrophages after
As mentioned above, CD163 M2-macrophages appeared mainly on days 1 and 2, and CD163-expressing Kupffer cells might become CD68 M1-macrophages on PSI day 2. Therefore, M1-macrophage-related factors (TNF-α, IL-6, and INF-γ) and M2-macrophage-related factors such as TGF-β1, IL-10, MCP-1, and IL-4 were increased significantly on PSI days 1 and 2; the simultaneous co-expressions of these factors of M1- and M2-macrophages might be reflected by histopathological findings such as degeneration/multifocal necrosis accompanied with reparative fibrosis on PSI days 1 and 2.
An interesting finding of the present study was a significant reduction of IL-1β mRNA level at PSI 8 hr and on PSI days 3 and 5 after
In double immunofluorescence analyses, it was reported in TAA-induced hepatic lesions of the PV areas that the majority of MHC class II–expressing macrophages became CD68-expressing M1 macrophages, whereas CD204-expressing macrophages are regarded as CD163-expressing M2-macrophages; MHC class II- and CD204-expressing macrophages are considered to be polarized toward M1- and M2-macrophages, respectively (Wijesundera et al. 2014). In the present double immunofluorescence staining in
CD204 is a scavenger receptor for lipid metabolism, and its expression may also be related to phagocytosis (Tomokiyo et al. 2002). Approximately 95% of CD204-expressing macrophages co-expressed with CD163 or CD68 on PSI day 2, in contrast to controls (85% for CD68 and 84% for CD163); these findings indicated that there was no clear differentiation in CD163/CD204 and CD68/CD204-double-positive cells between control livers and
In conclusion, the present study showed that the
Supplemental Material
Supplemental Material, DS1_TPX10.1177_0192623318801574 - M1/M2-macrophage Polarization-based Hepatotoxicity in d -galactosamine-induced Acute Liver Injury in Rats
Supplemental Material, DS1_TPX10.1177_0192623318801574 for M1/M2-macrophage Polarization-based Hepatotoxicity in
Supplemental Material
Supplemental Material, DS2_TPX10.1177_0192623318801574 - M1/M2-macrophage Polarization-based Hepatotoxicity in d -galactosamine-induced Acute Liver Injury in Rats
Supplemental Material, DS2_TPX10.1177_0192623318801574 for M1/M2-macrophage Polarization-based Hepatotoxicity in
Supplemental Material
Supplemental Material, DS3_TPX10.1177_0192623318801574 - M1/M2-macrophage Polarization-based Hepatotoxicity in d -galactosamine-induced Acute Liver Injury in Rats
Supplemental Material, DS3_TPX10.1177_0192623318801574 for M1/M2-macrophage Polarization-based Hepatotoxicity in
Supplemental Material
Supplemental Material, DS4_TPX10.1177_0192623318801574 - M1/M2-macrophage Polarization-based Hepatotoxicity in d -galactosamine-induced Acute Liver Injury in Rats
Supplemental Material, DS4_TPX10.1177_0192623318801574 for M1/M2-macrophage Polarization-based Hepatotoxicity in
Footnotes
Author Contributions
Authors contributed to conception or design (NR, MP, MK, MRK, TI, MKuw, JY); data acquisition, analysis, or interpretation (NR, MP, MK, MRK, TI, MKuw, JY); drafting the manuscript (NR); and critically revising the manuscript (NR, MP, MK, MRK, TI, MKuw, JY). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported partly by JSPS KAKENHI Grant Numbers 26292152 (to Yamate), by the Platform Project for Supporting Drug Discovery and Life Science Research (Basis for Supporting Innovative Drug Discovery and Life Science Research [BINDS]) from AMED and Grand Numbers 3P18 am 0101123 (to Yamate) and by The Otsuka Toshimi Scholarship Foundation (Number 16-S79, 17-S42 to N. Rahman).
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
