Abstract
Macrophages and myofibroblasts are important in fibrogenesis. The cellular characteristics in pancreatic fibrosis remain to be investigated. Pancreatic fibrosis was induced in F344 rats by a single intravenous injection of dibutyltin dichloride. Histopathologically, the induced pancreatic fibrosis was divided into 3 grades (1+, 2+, and 3+), based on collagen deposition. Immunohistochemically, CD68-expressing M1 macrophages increased with grade and CD163-expressing M2 macrophages also increased later than M1 macrophage appearance. Double immunofluorescence showed that there were macrophages coexpressing CD68 and CD163, suggesting a possible shift from M1 to M2 types; similarly, increased major histocompatibility complex class II- and CD204-expressing macrophages were polarized toward M1 and M2 types, respectively. These findings indicated the participation of M1- and M2-polarized macrophages. Mesenchymal cells staining positive for vimentin, desmin, and α-smooth muscle actin (α-SMA) increased with grade. There were mesenchymal cells coexpressing vimentin/α-SMA, desmin/α-SMA, and glial fibrillary acidic protein (GFAP)/α-SMA; Thy-1-expressing immature mesenchymal cells also increased in fibrotic lesions. Because α-SMA expression is a reliable marker for myofibroblasts, α-SMA-expressing pancreatic myofibroblasts might be originated from GFAP-expressing pancreatic stellate cells or Thy-1-expressing immature mesenchymal cells; the myofibroblasts could simultaneously express cytoskeletal proteins such as vimentin and desmin. The present findings would provide useful information for analyses based on features of macrophages and myofibroblasts in chemically induced pancreatic fibrosis.
Introduction
Fibrosis is a repair mechanism after tissue injury, but progressive fibrosis due to repeated injuries and chronic stimuli may cause functional insufficiency of organs. Studies on experimentally induced hepatic and renal fibrosis indicate that macrophages and myofibroblasts play important roles mutually in the pathogenesis; factors produced by macrophages reactive to injury could induce the development of myofibroblasts capable of producing collagens. 1 –5 The comprehensive mechanisms of progressive fibrosis remain to be fully investigated.
Macrophages show heterogeneous functions. M1/M2 polarization has been investigated for functional properties of macrophages appearing in pathological settings. 6 –8 M1 macrophages are regarded as classical type with phagocytic activity and cytotoxicity by producing inflammatory cytokines. On the other hand, M2 macrophages are related to tissue repair and fibrogenesis by producing anti-inflammatory and fibrogenic factors. Immunohistochemically, M1 and M2 macrophages express CD68 and CD163 antigens, respectively. 9,10 Along with CD68 and CD163, antibodies against antigens such as major histocompatibility complex (MHC) class II, CD204, and ionized calcium-binding adapter molecule 1 (Iba-1) are also used for the analysis of macrophage functions. 3,11 Major histocompatibility complex (MHC) class II is expressed in activated dendritic cells and macrophages with antigen-presenting ability. 12,13 CD204 is a class A scavenger receptor for lipid metabolism and degradation of reactive oxygen species (ROS). 14 Ionized calcium-binding adapter molecule 1 (IBA-1) is involved in cell migration and phagocytic activity of macrophages. 15,16
Myofibroblasts are mesenchymal cells with an intermediate nature between fibroblasts and smooth muscle cells. 17 Immunohistochemically, myofibroblasts express vimentin, desmin, and α-smooth muscle actin (α-SMA) in various proportions during the development. 18 –20 Myofibroblasts are derived from preexisting fibroblasts; in addition, in hepatic fibrosis, hepatic stellate cells (HSCs) expressing glial fibrillary acidic protein (GFAP) are considered as the origin of the hepatic fibroblasts. 21,22
Focal necrosis/injury in the pancreas leads to reparative fibrosis. The chronic type may give rise to serious conditions such as emaciation and secondary diabetes. 23 Dibutyltin dichloride (DBTC), which has been used primarily as stabilizers for polyvinylchloride plastics, is used for induction of pancreatitis in experimental animals; DBTC-induced pancreatitis in rats resembles the human form of this disease. 24 We investigated cellular characteristics of macrophages and myofibroblasts appearing in DBTC-induced rat pancreatic lesions because the pathogenesis based on these cells has not been sufficiently studied.
Materials and Methods
Animals and Experimental Procedures
Twenty 7-week-old male F344 rats, purchased from Charles River Japan (Hino, Shiga, Japan), were used. Rats were maintained in a room at 21°C ± 3°C with a 12-hour light–dark cycle, fed a standard diet for rats (DC-8; CLEA Japan, Tokyo, Japan), and supplied with tap water ad libitum. After a 1-week acclimatization, the rats were randomly divided into 2 groups: a vehicle (ethanol:glycerol = 2:3) group (n = 4) and DBTC treatment group (4 or 8 mg/mL/kg body weight [BW]; n = 8 in each dose). 25 The vehicle and DBTC solution were injected intravenously once via tail vein to rats. Following injection, 4 rats were euthanized at 1 and 3 days for 8 mg/mL/kg and at 7 and 15 days for 4 mg/mL/kg by exsanguination under deep isoflurane anesthesia; 4 rats in the control were killed on day 1. The animal experiments were conducted under the institutional guidelines approved by the ethical committee of Osaka Prefecture University for the Care and Use of Experimental Animals (No. 23-24).
Histopathology and Immunohistochemistry
Pancreas tissues were collected and immediately fixed in 10% neutral-buffered formalin (NBF) or periodate–lysine–paraformaldehyde (PLP) solution processed by the PLP-AMeX (acetone, methyl benzoate, and xylene) method. 26 Three or 4 tissue sections including duodenal and splenic poles were examined in the pancreas of the individual animal. Fixed tissues were dehydrated and embedded in paraffin and sectioned at 3 to 4 µm in thickness. The deparaffinized sections were stained with hematoxylin and eosin (HE) for histopathological examination. A part of pancreatic tissue was also taken as snap-frozen samples.
Tissue sections fixed in 10% NBF were deparaffinized and used in immunohistochemistry for CK19, vimentin, α-SMA, GFAP, and Thy-1 staining. Tissue sections fixed in PLP were deparaffinized and used in immunohistochemistry for CD163, CD68, Iba-1, CD204, MHC class II, and LC3B staining. The sections for CK19, CD68, and CD163 were incubated with 10 μg/mL proteinase K in 50 mM Tris–HCl buffer (pH 7.5) at room temperature for 20 minutes. The sections for LC3B, MHC class II, CD204, Iba-1, vimentin, GFAP, α-SMA, and Thy-1 were boiled in citrate buffer in the microwave for 20 minutes. After treatment with phosphate-buffered saline (PBS) containing 3% H2O2/0.065% NaN3 for 10 minutes, the sections were incubated with 5% skimmed milk for 30 minutes, followed by 1-hour incubation with primary antibodies. Details of antibodies are listed in Table 1. The sections were incubated with horseradish peroxidase–conjugated secondary antibody (Histofine simple stain MAX PO; Nichirei, Inc, Tokyo, Japan), which was applied for 30 minutes. Then they were incubated with 3, 3′-diaminobenzidine (Nichirei, Inc) for 5 minutes. The sections were counterstained with HE for 1 minute.
Primary Antibodies Used for Immunohistochemistry and Immunofluorescence.
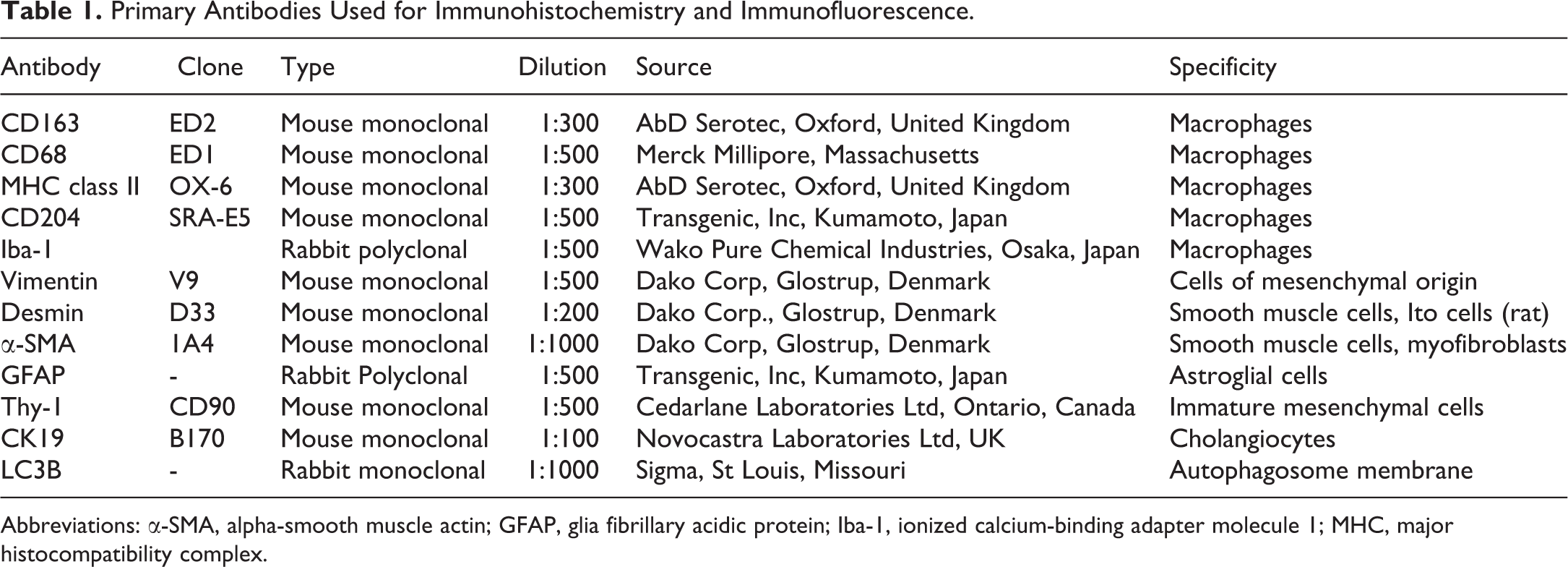
Abbreviations: α-SMA, alpha-smooth muscle actin; GFAP, glia fibrillary acidic protein; Iba-1, ionized calcium-binding adapter molecule 1; MHC, major histocompatibility complex.
Fresh frozen pancreases were sectioned at 10 µm thickness and used for immunohistochemical staining for desmin. After fixation in cold acetone:methanol (1:1) for 10 minutes at 4°C, the sections were incubated with 5% skimmed milk for 15 minutes followed by reaction with the antidesmin antibody 60 minutes at room temperature. The procedure after reacting with the primary antibody was the same as described above. For negative controls, tissue sections were treated with mouse or rabbit nonimmunized serum instead of the primary antibody. Cells expressing each antigen in immunohistochemistry were evaluated semiquantitatively as mentioned in footnotes of Tables 2 and 3.
Semiquantitative Evaluation of Immunoreactivity of Macrophage Markers in DBTC-Induced Pancreatic Fibrosis.a

Abbreviations: Iba-1, ionized calcium-binding adapter molecule 1; MHC, major histocompatibility complex.
a The intensity of immunoreactions (immuno-positive cells) are shown as follows: −: negative;
Semiquantitative Evaluation of Immunoreactivity of Myofibroblast Markers in DBTC-Induced Pancreatic Fibrosis.a

Abbreviations: α-SMA, α-smooth muscle actin; GFAP, glial fibrillary acidic protein; NE, not examined.
a The intensity of immunoreactions (immunopositive cells) is shown as follows; −; negative,
Double Immunofluorescence Staining
Fresh frozen pancreas sections at 10 µm thickness were used for double immunofluorescence to more fully characterize macrophages (using with CD163, CD68, MHC class II, and CD204 antibodies) and myofibroblasts (using with vimentin, desmin, GFAP, and α-SMA antibodies). Briefly, after fixation in cold acetone:methanol (1:1) for 10 minutes at 4°C, the sections were incubated with 10% normal goat serum for 30 minutes at room temperature followed by reaction with the appropriate primary antibody overnight at 4°C. After rinsing with PBS, the sections were incubated for 45 minutes at room temperature with a secondary goat anti-mouse IgG conjugated with Alexa 488 or 568 (Invitrogen, Carlsbad, California) for CD68, CD163, MHC class II, and CD204; goat anti-mouse IgG2a-conjugated with Alexa 488 or 568 (Invitrogen) for α-SMA; goat anti- mouse IgG1 conjugated with Alexa 488 or 568 for vimentin and desmin; and goat anti-rabbit IgG-conjugated with Alexa 488 (Invitrogen) for GFAP. For CD68, CD163, MHC class II, and CD204, the sections were incubated for 45 minutes at room temperature, with the second primary antibody labeled with fluorescent dye-conjugated secondary antibody: Alexa 488-labeled CD163 (AbD Serotec, Oxford, United Kingdom) for CD163/CD204 combination; Alexa 488-labeled MHC class II (AbD Serotec) for CD163/MHC class II combination; and Alexa 488-labeled CD68 (AbD Serotec) for CD68/CD163 combination. The sections were visualized with Vectashield mounting medium containing 4′, 6-diamidino-2-phenylindole (Vector Laboratories, Inc, Burlingame, California) for nuclear staining and analyzed by a virtual slide scanner (VS-120; Olympus, Tokyo, Japan). Cells expressing antigen in immunofluorescence were counted, at a microscope magnification of ×10, in 3 different, randomly selected, areas.
Reverse Transcriptase Polymerase Chain Reaction
Freshly dissected pancreatic samples were immediately immersed in RNAlater (Qiagen GmbH, Hilden, Germany) overnight at 4°C and stored at −80°C until use. Total RNA was extracted from pancreatic tissues by using an SV total RNA isolation system (Promega, Madison, Wisconsin), according to the manufacturer’s instructions. Two and half micrograms of total extracted RNA was reverse-transcribed with Superscript VILO reverse transcriptase (RT; Life Technologies, Carlsbad, California). Real-time polymerase chain reaction (PCR) was performed using TaqMan gene expression assays (Life Technologies) in a PikoReal Real-Time 96 PCR System (Thermo Scientific, Waltham, Massachusetts). The TaqMan probes specific for cytokines used were as follows (Assay IDs): monocyte chemoattractant protein 1 (MCP-1), Rn00580555_m1; interleukin 1β (IL-1β), Rn00580432_m1; transforming growth factor β1 (TGF-β1), Rn00572010_m1; IL-4, Rn01456866_m1; IL-6, Rn01410330_m1; IL-10, Rn00563409_m1; and 18S ribosomal RNA, Hs99999901_s1. The messenger RNA (mRNA) expression was normalized against the expression of 18S ribosomal RNA as the internal control. Three animals per group were used for the RT-PCR cytokine analysis. The data were analyzed using the comparative Ct method (ΔΔCt method).
Statistical Evaluation
Data obtained in the RT-PCR analyses were expressed as mean ± standard deviation. Statistical analysis was performed using Dunnett test. Significance was considered at P < .05.
Results
Histopathology of Pancreas
No lesions were seen in control pancreas (Figure 1A, E). Regardless of time course or dosage (4 or 8 mg/mL/kg BW), pancreatic lesions varied from animal to animal irrespective of the dose administered. Therefore, pancreatic lesions were individually evaluated histopathologically as follows: rats with the grade of mild fibrosis (1+; n = 5) showed slight atrophy of acinar cells and interstitial edema with a few inflammatory cells and slight deposition of collagens between lobules (Figure 1B, F); in the grade of moderate fibrosis (2+; n = 6), atrophy of acinar cells with decreased zymogen granules and inflammatory cell infiltration was more prominent between the acinar lobules and spindle-shaped mesenchymal cells (myofibroblasts as mentioned below), and marked deposition of collagens was observed which partly destroyed the pancreatic structure (Figure 1C, G); in the grade of marked fibrosis (3+; n = 5), a large number of inflammatory cells were seen locally or diffusely, and pancreatic tissues were replaced by fibrotic areas, almost completely destroying the normal lobular structures in the organ (Figure 1D, H).
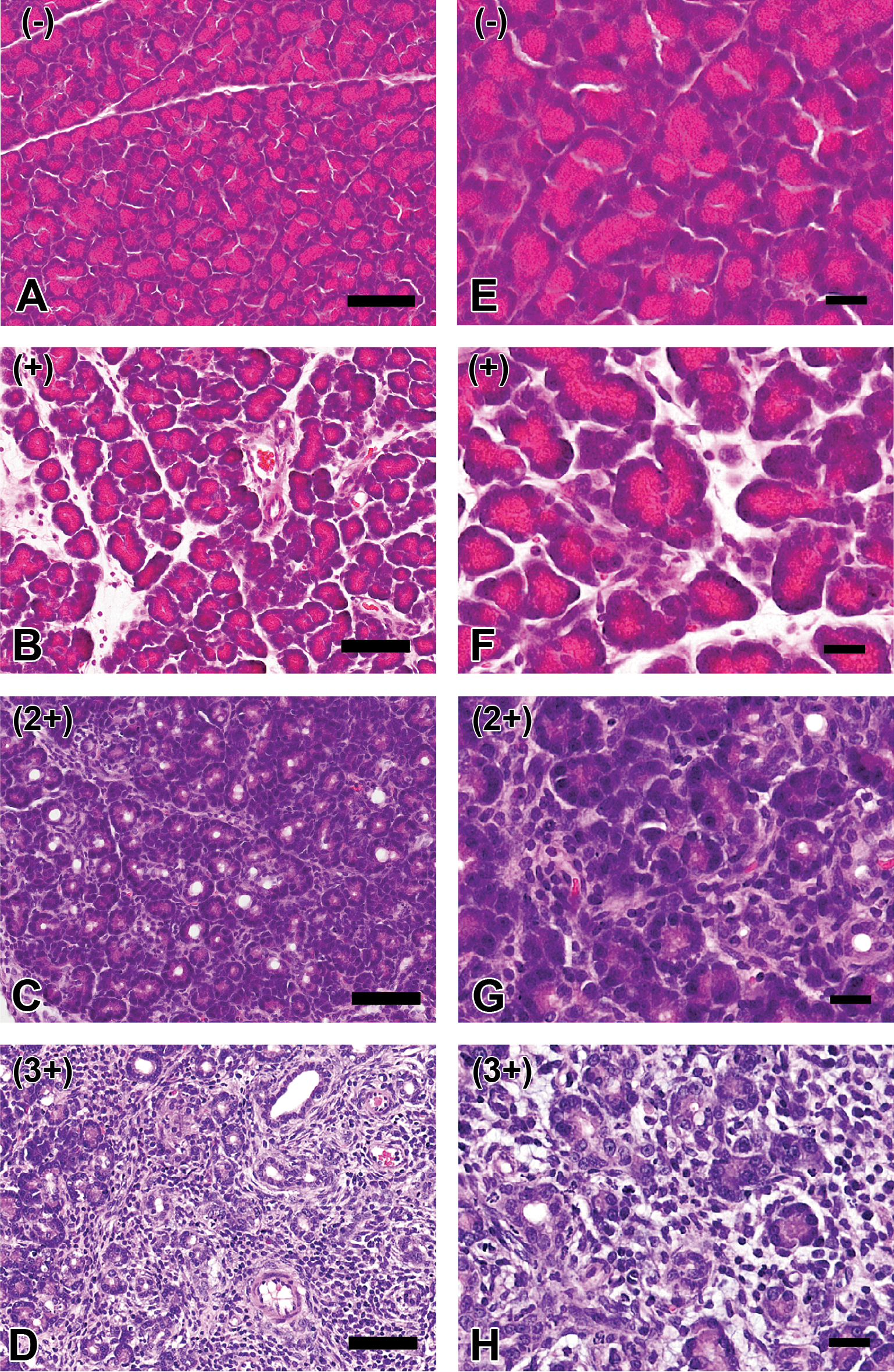
Histopathology in control pancreas (A and E) and dibutyltin dichloride–induced pancreatic fibrosis of grade 1+ (B and F), 2+ (C and G), and 3+ (D and H). A and E, No lesions were seen in control pancreas. B and F, In grade 1+ lesion, slight atrophy of acinar cells and interstitial edema are seen with a small number of inflammatory cells. C and G, In grade 2+ lesion, interstitial fibrosis and atrophy of acinar cells with decreased zymogen granules are more prominent, accompanied with inflammatory cell infiltration. D and H, In grade 3+, marked deposition of collagens and a large number of inflammatory cells are observed, almost destroying lobular structures. Bars A to D = 50 µm and E to H = 20 μm.
Immunohistochemical staining for CK19, a marker for the pancreatic duct epithelium, showed that there were some pancreatic ducts reacting to CK19 in controls (Figure 2A); ductal epithelial cells reacting to CK19 markedly increased with the grade of fibrosis; the appearance of CK19-positive cells was most prominent in the marked fibrosis (3+), and the positive ductal epithelium in the pancreatic fibrosis lesion showed small tubular, indicating hyperplasia of pancreatic ducts (Figure 2B).
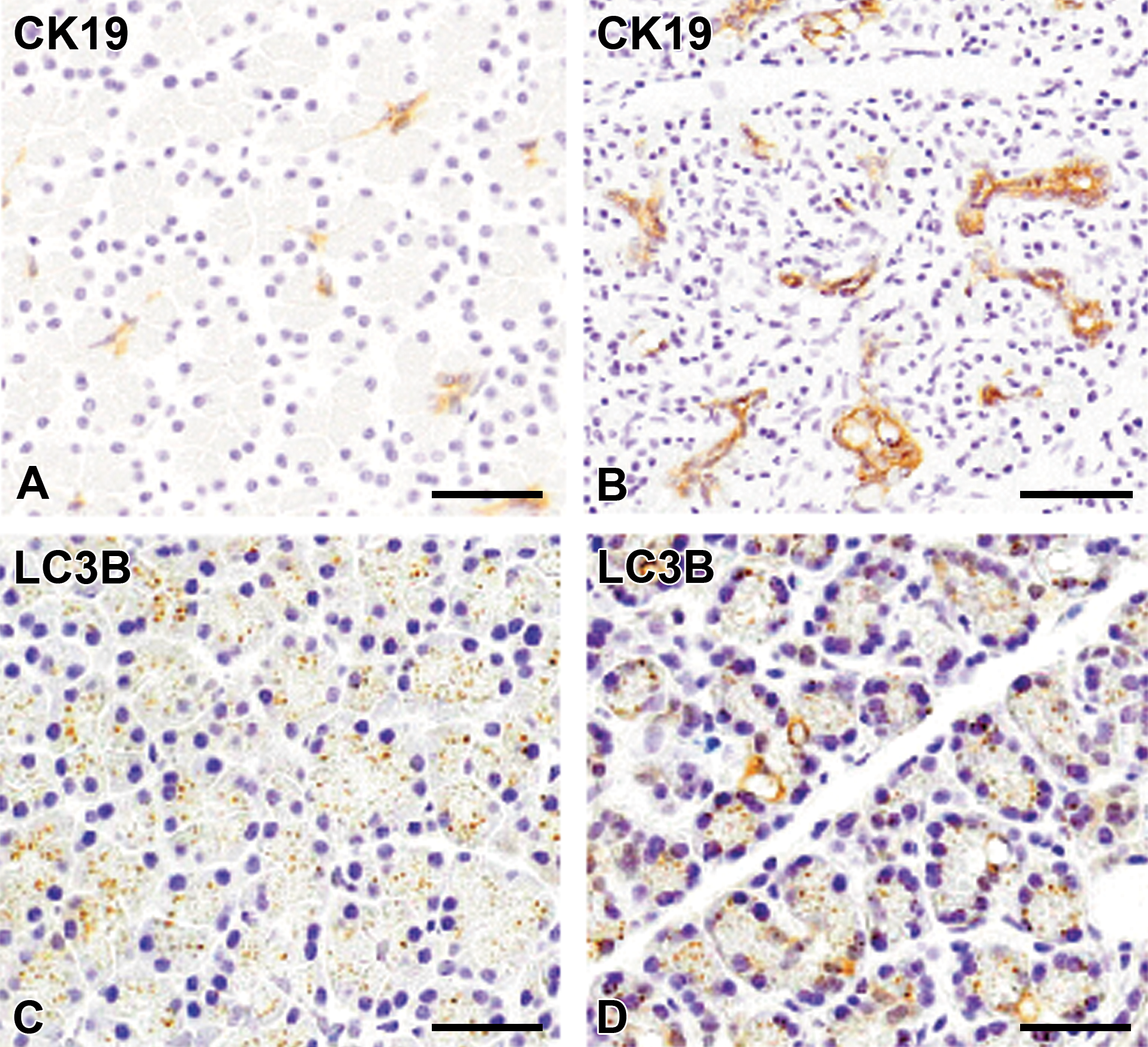
Immunohistochemistry with CK19 antibody for pancreatic duct epithelial cells and LC3B antibody for autophagosomes in control pancreas (A and C) and pancreatic fibrosis lesion (B and D). A and B, There are some ductal epithelial cells including centroacinar cells expressing CK19 in control pancreas (A), whereas increased pancreatic ductal epithelial cells positive to CK19 are seen in grade 3+ lesion (B), forming ducts and tubules (hyperplasia). C and D, LC3B-reacting fine granules are seen in the cytoplasm of acinar cells, whereas there are irregularly vacuolated structures reacting to LC3B in cytoplasm of some acinar cells (arrows) indicative abnormal formation of autophagosomes. Counterstained with hematoxylin. Bar = 50 μm.
Interestingly, fine granules reacting to LC3B, a marker for autophagosomes, were seen in the cytoplasm of acinar cells of control pancreas (Figure 2C), whereas abnormal vacuolar structures reacting to LC3B often appeared in the cytoplasm of some acinar cells in the moderate fibrosis (2+) (Figure 2D). LC3B-positive granules disappeared in the cytoplasm of trapped acinar cells in the marked fibrosis (3+). These findings indicated that abnormality in autophagosomes occurred with progressive fibrosis.
M1/M2 Macrophages
A few CD68-expressing and CD163-expressing cells, indicative of resident macrophages, 9 were sporadically seen in control pancreas (Figure 3A, C). With an increasing grade of pancreatic fibrosis (1+, 2+, and 3+), CD68-expressing M1 macrophages and CD163-expressing M2 macrophages gradually increased (Table 2). Interestingly, CD68-expressing M1 macrophages began to increase from the grade 1+, whereas CD163-expressing M2 macrophages clearly increased from the grade 2+, and in the grade 3+, the appearance was greater in number than CD68 M1 macrophages. These findings indicated that M2 macrophages appeared later than did M1 macrophages. Macrophages reacting to CD68 (Figure 3B) and CD163 (Figure 3D) in pancreatic fibrosis showed a round or spindle-shaped configuration, morphologically.
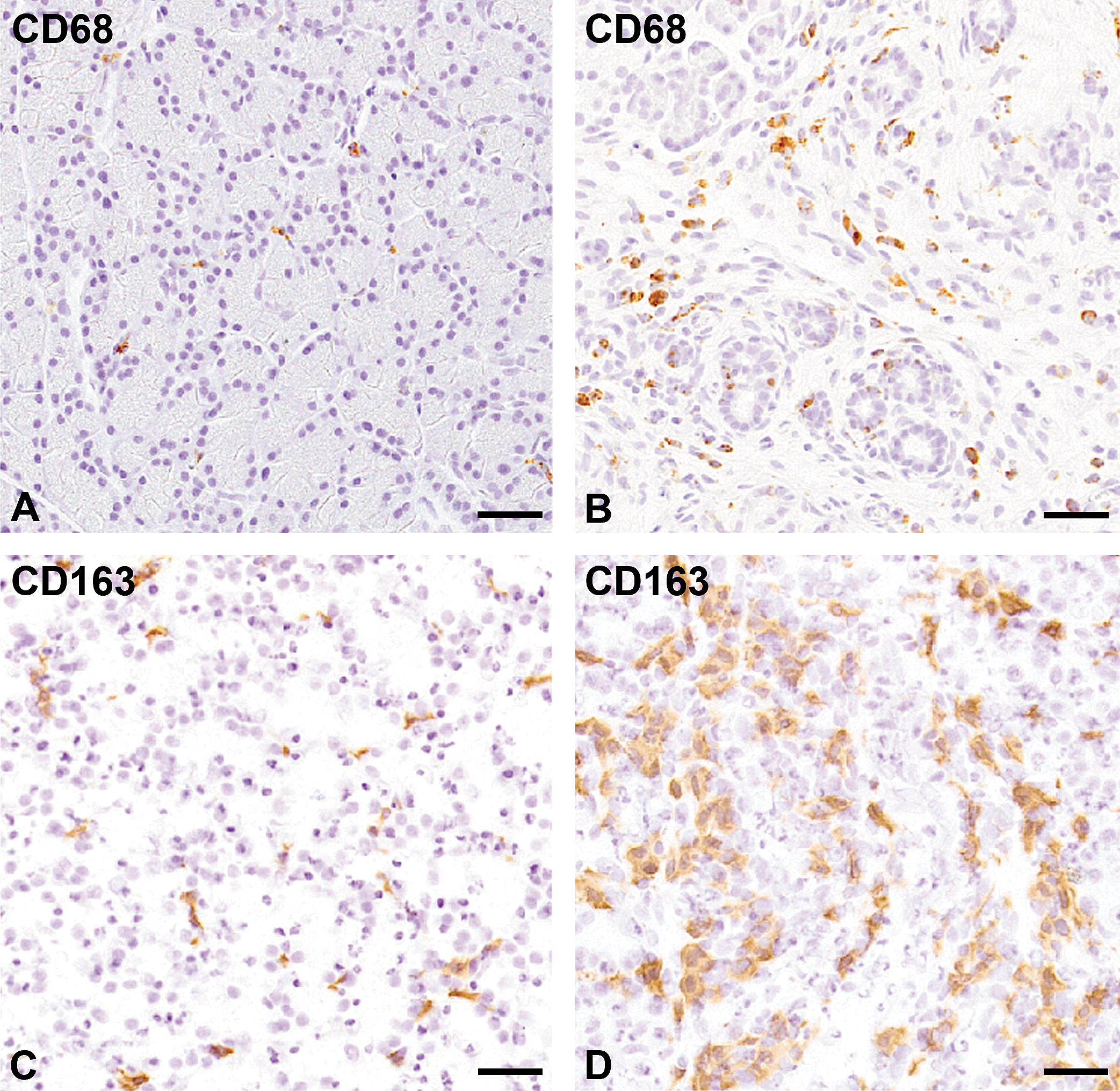
Immunohistochemistry with CD68 for M1 macrophages and CD163 for M2 macrophages in control pancreas (A and C, respectively) and in dibutyltin dichloride-induced grade 3+ pancreatic fibrosis (B and D, respectively). A few cells can be seen reacting to CD68 in the control tissue (A); on the contrary, increased numbers of CD68-positive macrophages are present in the grade 2+ lesion (B). Macrophages reacting to CD163 are seen sporadically in the control tissue (C) whereas there are many macrophages reacting to CD163 in the grade 3+ lesion (D). Counterstained with hematoxylin. Bar = 50 μm.
Macrophages Reacting to CD204, MHC Class II, and Iba-1
Although MHC class II-expressing macrophages were not seen in control pancreas or in the pancreatic fibrosis of grade 1+ and 2+, the positive cells quickly increased in pancreatic fibrosis grade 3+ (Table 2; Figure 4A, B). Although CD204-expressing macrophages were small in number in each of the grades (Table 2), the positive cells slightly increased in the grade 2+ and 3+ (Figure 4C, D).On the contrary, although Iba-1-positive cells were sporadically seen in control pancreas, the number increased with the increasing grades (1+, 2+, and 3+; Table 2; Figure 4E, F). Macrophages expressing MHC class II, CD204, and Iba-1 in pancreatic fibrosis showed round or spindle-shaped morphology (Figure 4B, D, F). In addition to CD68- and CD163-expressing macrophages, it was likely that macrophages expressing to antigens such as MHC class II, CD204, and Iba-1 could contribute to pancreatic fibrosis.
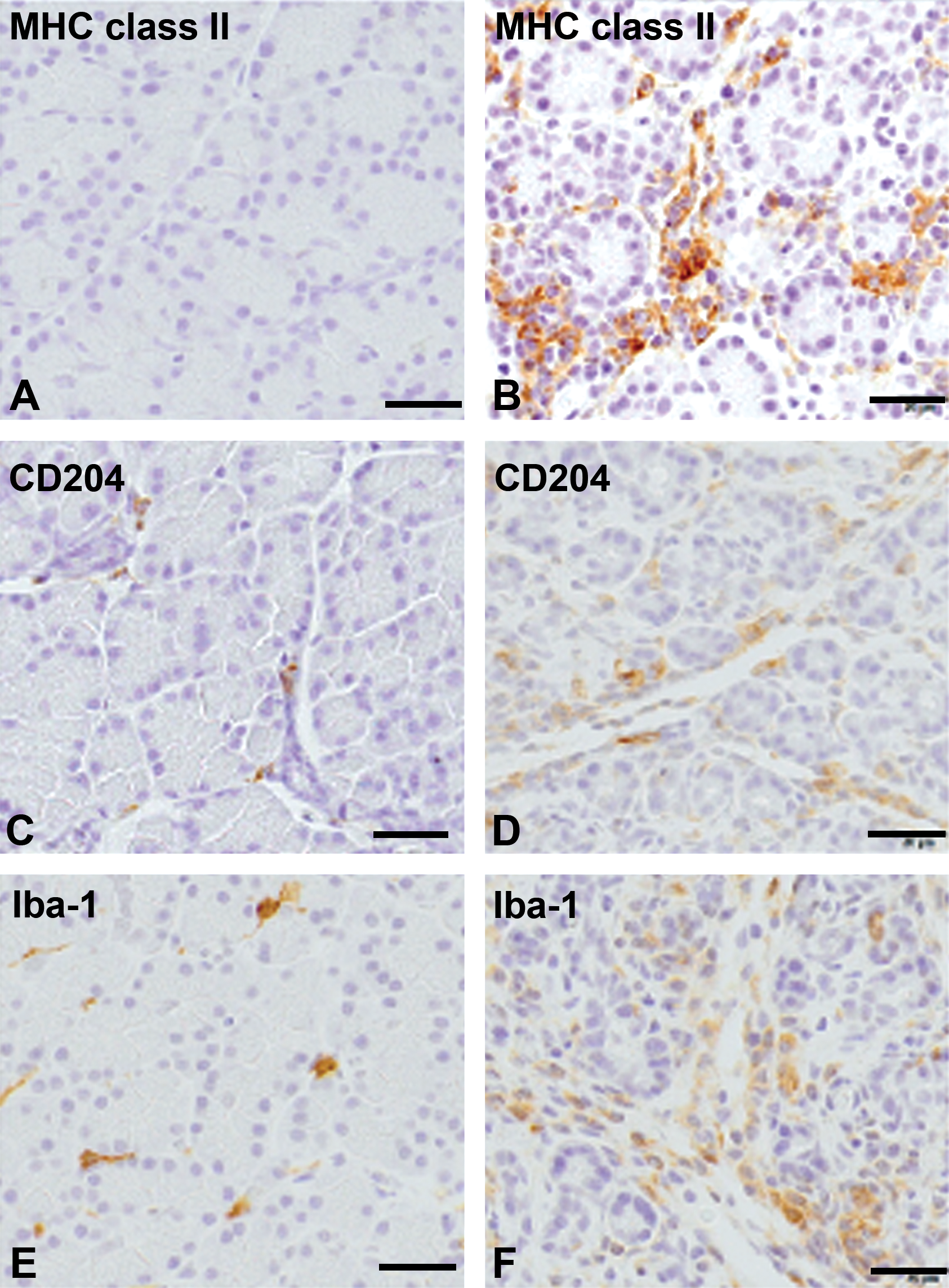
Immunohistochemistry with major histocompatibility complex (MHC) class II, CD204, and ionized calcium-binding adapter molecule 1 (Iba-1) for macrophages in control pancreas (A, C, and E, respectively) and dibutyltin dichloride–induced pancreatic fibrosis of grade 3+ (B, C, and F, respectively). Macrophages reacting to MHC class II, CD204, and Iba-1 are rarely (for MHC class II) or sporadically (for CD204 and Iba-1) seen in controls, whereas macrophages reacting to these antibodies are increased in the fibrotic pancreas. Counterstained with hematoxylin and eosin. Bar = 20 μm.
Double Immunofluorescence Staining for Macrophages
Double immunofluorescence staining was conducted by using samples of the pancreatic fibrosis grade 2+. Macrophages coexpressing CD68 (for M1) and CD163 (for M2) accounted for 44%, whereas those expressing solely CD68 and CD163 were 47% and 9%, respectively (Figure 5). These findings indicated that there were macrophages expressing simultaneously CD68 and CD163 in the pancreatic fibrosis.
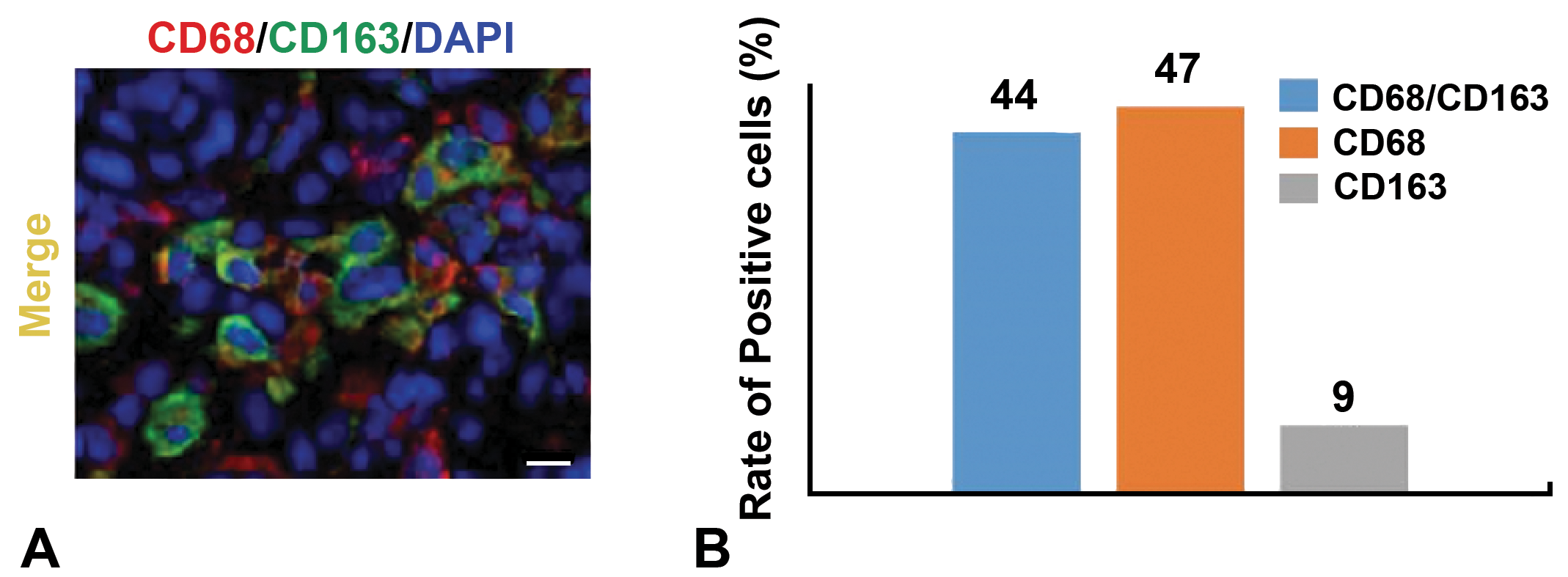
Double immunofluorescence by combination with CD68 antibody for M1 macrophages and CD163 antibody for M2 macrophages using dibutyltin dichloride–induced pancreatic fibrosis of grade 2+. A, Double immunofluorescence staining. Bar = 10 μm. B, The percent of positive cells was 44% for CD68/CD163, 47% for CD68, and 9% for CD163.
In double immunofluorescence staining, macrophages expressing MHC class II in CD68-positive M1 macrophages and CD163-positive M2 macrophages were 34% and 3%, respectively (Figure 6A, C). Macrophages expressing CD204 in CD68-positive M1 macrophages and CD163-positive M2 macrophages accounted for 23% and 89%, respectively (Figure 6B, C). These findings indicated that MHC class II- and CD204-expressing macrophages tended to be polarized toward M1 and M2 macrophages, respectively.
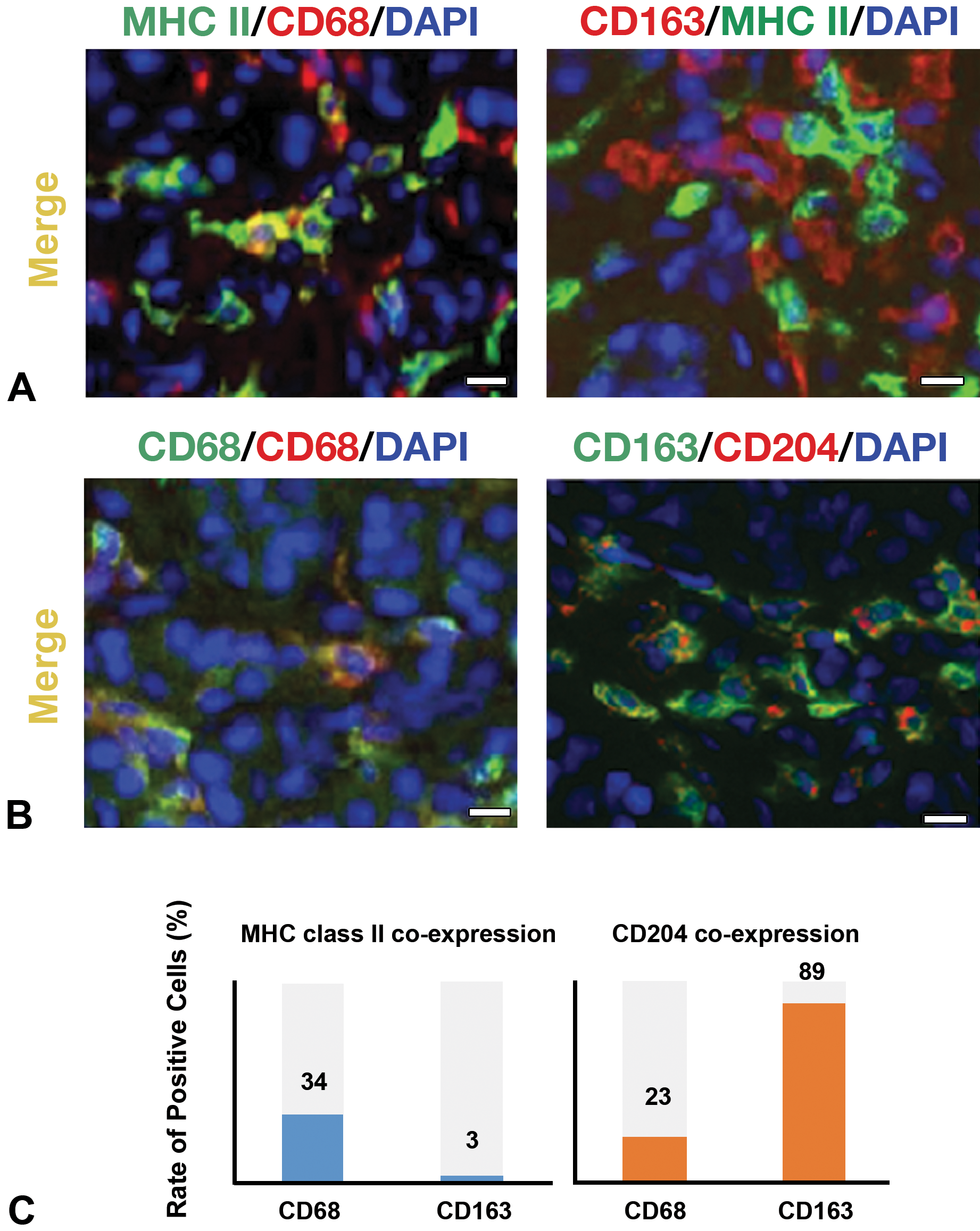
Double immunofluorescence by combination with major histocompatibility complex (MHC) class II/CD68 and MHC class II/CD163 (A), as well as CD204/CD68 and CD204/CD163 (B) in dibutyltin dichloride–induced pancreatic fibrosis of grade 2+. A, MHC class expression in CD68 M1 and CD163 M2 macrophages. B, CD204 expression in CD68 M1 and CD163 M2 macrophages. Bar = 10 μm. C, Rate of positive cells is 34% for MHC class II/CD68, 3% for MHC class II/CD163, 23% for CD204/CD78 and 89% for CD204/CD163.
M1- and M2-Related Macrophage Factors
Inflammatory cytokines (such as IL-1β, IL-6, and MCP-1 for M1 macrophages) and anti-inflammatory cytokines (such as IL-4, IL-10, and TGF-β1 for M2 macrophages) were analyzed.
Although there were no marked changes in each cytokine item, mRNAs, but mRNAs of IL-1β, IL-6, MCP-1, IL-4, and IL-10 showed a tendency to increase in the grade 2+ and/or 3+ (Figure 7). Transforming growth factor-β1 did not show any significant change in the pancreatic fibrosis (Figure 7).
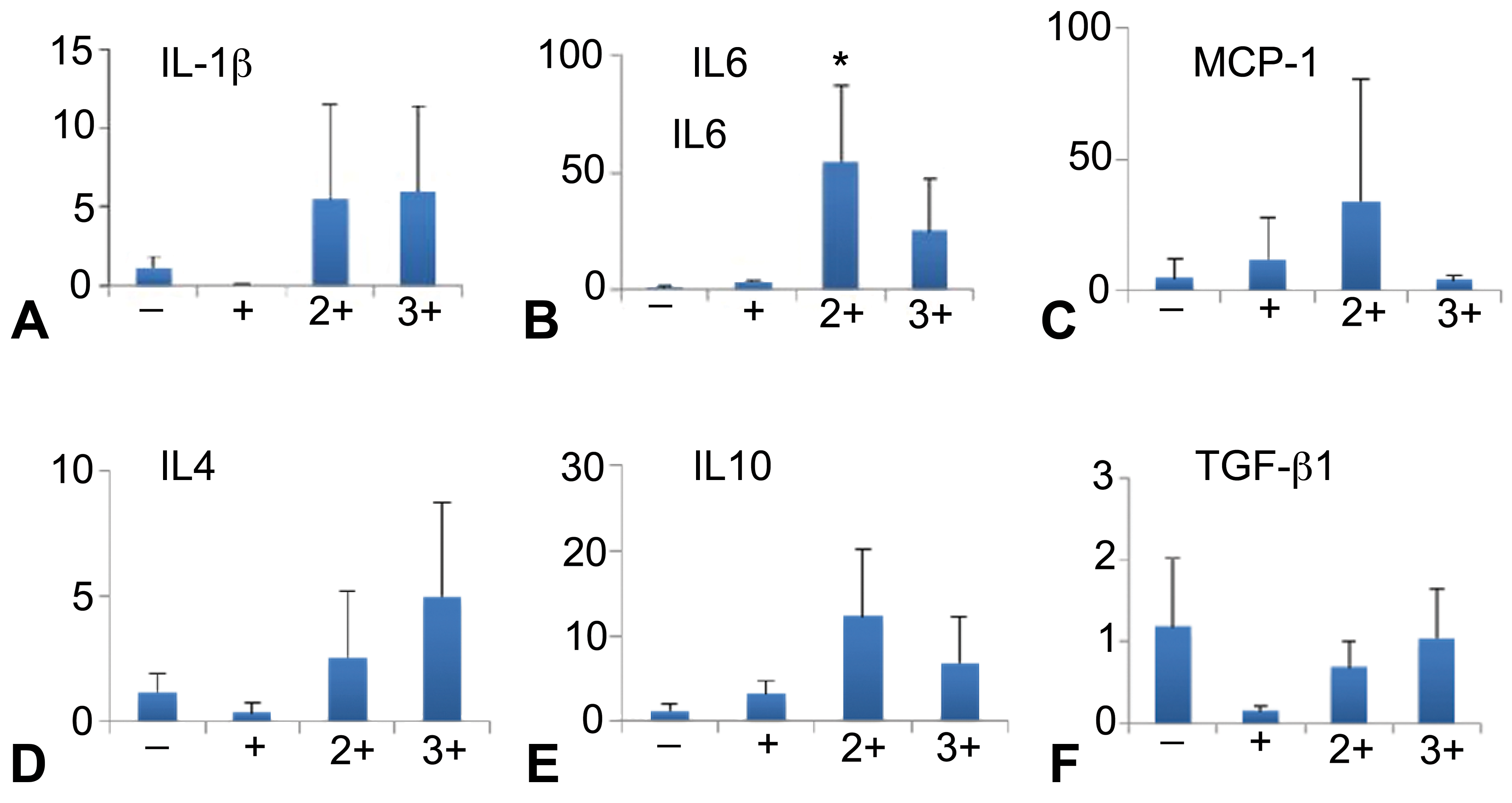
Messenger RNA expression of M1 macrophage-related inflammatory factors (interleukin 1β [IL-1β] [A], IL-6 [B], monocyte chemoattractant protein 1 [MCP-1] [C]), M2 macrophage-related anti-inflammatory factors (IL-4 [D], IL-10 [E], and transforming growth factor β1 [TGF-β1] [F]). −, normal; 1+, 2+, and 3+, grade of pancreatic fibrosis. n = 3. *P < .05 by one-way analysis of variance and Dunnett.
Myofibroblast Characteristics
Immunohistochemical analyses were made for myofibroblasts in rat DBTC-induced pancreatic fibrosis (Table 3). Spindle-shaped mesenchymal cells expressing vimentin were found sporadically around the acinar lobules in control pancreas, and the positive cell number increased with the increasing grade of pancreatic fibrosis (2+ and 3+; Figure 8A). Desmin-expressing mesenchymal cells were similar to the distribution of vimentin-expressing cells; round to spindle-shaped mesenchymal cells expressing desmin were seen in the grade 2+ and 3+ of pancreatic fibrosis (Figure 8B). Mesenchymal cells expressing α-SMA were not seen in control pancreas (except vascular smooth muscle cells) and the grade 1+; on the contrary, α-SMA-expressing mesenchymal cells increased with increasing grade (2+ and 3+; Figure 8C). These mesenchymal cells expressing vimentin, desmin, or α-SMA in the fibrotic lesions were regarded as myofibroblasts.
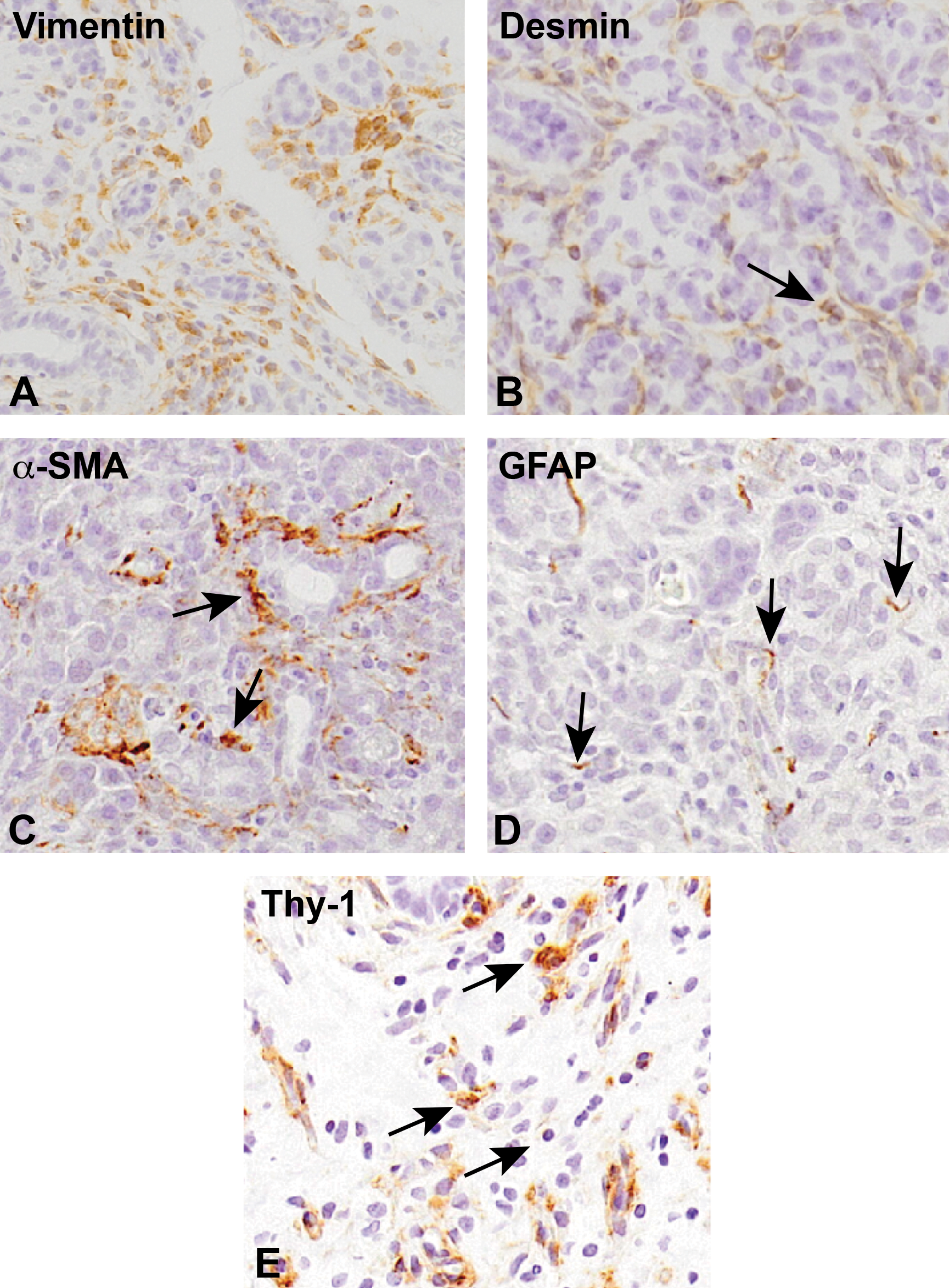
Immunohistochemistry for vimentin (A), desmin (B), α-smooth muscle actin (α-SMA; C), glial fibrillary acidic protein (GFAP; D), and Thy-1 (E) in pancreatic fibrosis with grades if 1+ (D), 2+ (C), and 3+ (A, B, and E). Counterstained with hematoxylin. Arrows indicate immuno-positive cells.
Myofibroblasts Reacting to GFAP and Thy-1
Glial fibrillary acidic protein–expressing mesenchymal cells around the acinar lobules seen in normal pancreas were considered as pancreatic stellate cells (PSCs). 23,27 Glial fibrillary acidic protein (GFAP)–expressing mesenchymal cells increased in the pancreatic fibrosis, particularly of the grade 2+ and 3+ (Table 3); the GFAP-positive cells in the pancreatic fibrosis looked less in number than vimentin-, desmin-, and α-SMA-positive cells (Figure 8D). Thy-1-positive mesenchymal cells were rarely seen in control pancreas; however, the positive cells increased with the grades (1+, 2+, and 3+; Table 3); the Thy-1-positive cells were mainly present around blood vessels as perivascular aggregates (Figure 8E).
Double Immunofluorescence Staining for Myofibroblasts
The expression of α-SMA is regarded as the reliable marker of myofibrobalsts. 27 By using samples of the grade 2+, mesenchymal cells expressing vimentin, desmin, and α-SMA were analyzed by double immunofluorescence. The percentage of mesenchymal cells coexpressing vimentin/α-SMA, desmin/α-SMA, and GFAP/α-SMA was 65%, 50%, and 60%, respectively (Figure 9).
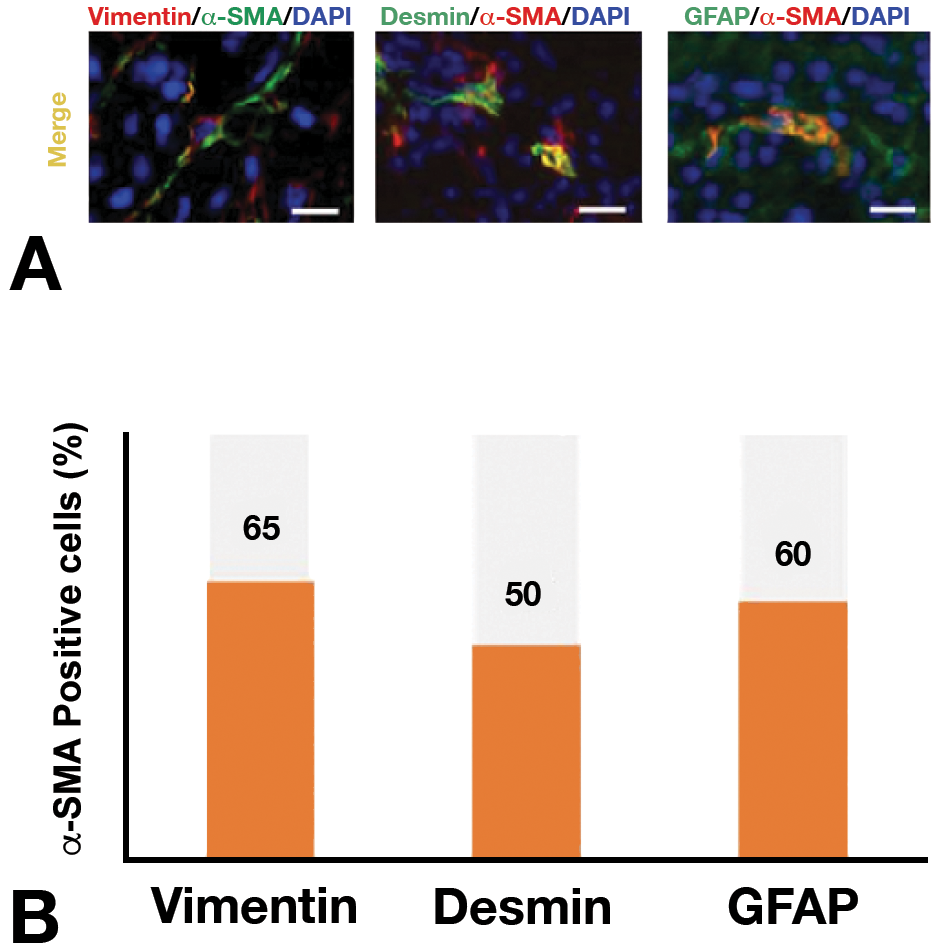
Double immunofluorescence (A) with vimentin/ α-SMA antibodies, desmin/α-SMA antibodies, and GFAP/α-SMA antibodies using lesions with grade 2+ pancreatic fibrosis. Bar = 10 μm. B, The percent of positive cells is 65% for vimentin/α-SMA, 50% for desmin/α-SMA, and 60% for GFAP/α-SMA. Abbreviations: GFAP, glia fibrillary acidic protein; α-SMA, α-smooth muscle actin.
Discussion
Histopathology of DBTC-Induced Pancreatic Lesion
Dibutyltin dichloride (DBTC) is used as a stabilizer for the production of polyvinyl chloride plastic products. 28 As the main cytotoxic effect, the chemical causes epithelial necrosis on the biliopancreatic duct, presumably leading to obstruction of the duct and subsequent inflammation in the surrounding; in addition, DBTC effects cause direct injury of pancreatic acinar cells which may involve mitochondrial damage, autophagy dysfunction, and cell necrosis. 24 The present DBTC-induced rat pancreatic lesions were characterized by interstitial edema, inflammatory cell reaction, and fibrosis, being accompanied with atrophy of acinar cells. Unexpectedly, the rats injected with a single injection of DBTC did not show any dose-/time-dependent change. Presumably, the degree of obstructed pancreatic ducts varied individually between rats, resulting in the occurrence of different lesions from animal to animal. Therefore, we evaluated the pancreatic lesions (1+, 2+, and 3+) based on histopathological changes, mainly of collagen deposition/expansion, as mentioned in the “Results” section. The DBTC-induced pancreatitis and subsequent fibrosis in rats resemble the human counterpart. 24
Interestingly, along with histopathological findings as reported previously, 24 we showed the ductal hyperplasia, demonstrable with CK19 immunohistochemistry, in pancreatic fibrosis, indicative of regeneration of ducts after injury by DBTC. Ductal proliferation has been reported in spontaneous pancreatic fibrosis cases of dogs and cats. 23 Furthermore, the immunohistochemistry with LC3B antibody could demonstrate the presence of autophagosomes in the cytoplasm of acinar cells; the autophagosome is a spherical structure with double-layer membranes. 29 In the cytoplasm of acinar cells, fine granular autophagosomes reacting to LC3B were seen in controls, whereas autophagosomes staining positively to LC3B developed vacuolated or irregular structures in the grade 2+ pancreatic damage and then disappeared in the grade 3+ pancreatic damage. It is considered that autophagy could promote cell survival by the elimination of damaged organelles and proteins aggregates, as well as by facilitating cellular homeostasis. 29 The abnormal structure of LC3B-positive autophagosomes might be related to toxic effects of DBTC on the pancreatic acinar cells. Pancreatic ductal proliferation and abnormal autophagosome formation might be used as hallmarks of experimentally induced pancreatic lesions.
CD68-Expressing M1 Macrophages and CD163-Expressing M2 Macrophages
Macrophages show heterogeneous functions based on microenvironmental conditions. Macrophages appearing in pathological settings are generally characterized by M1/M2 polarization. 6 –8 In DBTC-induced rat pancreatic fibrosis, CD68-expressing M1 macrophages began to increase from the grade 1+ and then CD163-expressing M2 macrophages clearly increased from the grade 2+; seemingly, M1 macrophages appeared earlier than M2 macrophages. Generally, M1 macrophages appear first when tissue is damaged, by producing inflammatory factors capable of promoting further tissue damage as well as phagocytic activity to remove damaged tissues. 30,31 After that, M2 macrophages appear by releasing anti-inflammatory and fibrotic factors, leading to tissue repair/reparative fibrosis. 31,32 The appearance of M1/M2 macrophages in the present pancreatic fibrosis was generally corresponding to the concept of macrophage polarization.
Interestingly, the double immunofluorescence staining using the grade 2+pancreatic fibrosis samples showed that 44% of macrophages coexpressed with CD68/CD163, suggesting a possible shift from M1 to M2 macrophages, functionally, as mentioned in chemically induced rat hepatic fibrosis. 11 It has been considered that CD68 M1 macrophages might be derived from blood monocytes as inflammatory cells at early stages. 3 It is speculated that blood monocyte-derived CD68 M1 macrophages might have changed into CD163 M2 macrophages during the development of pancreatic fibrosis.
Macrophages Expressing MHC Class II, CD204, and Iba-1
Along with CD63 and CD163 expressions, rat macrophages could express MHC class II, CD204, and Iba-1. 12,13,33 Functionally, activated antigen-presenting macrophages including dendritic cells can express MHC class II, of which expression may be related to the complicated immune response; CD204 is a scavenger receptor antigen for class A which may be associated with ROS degradation 14 ; Iba-1 is considered to be a protein involved in macrophage scaffolds attributable to cell migration and phagocytosis. 15,16 In the present DBTC-induced pancreatic fibrosis, MHC class II-, CD204-, and Iba-1-expressing macrophages increased with the fibrosis grade, although the degree of appearance was different between these antigen-expressing macrophages.
Interestingly, in the double immunofluorescence staining using the fibrosis grade 2+ samples, 34% of CD68-positive M1 macrophages simultaneously expressed MHC class II and 89% of CD163-positive M2 macrophages coexpressed CD204. In short, there was a tendency to polarize MHC class II- and CD204-expressing macrophages into M1 and M2 macrophages, respectively. Similar macrophage polarization for MHC class II- and CD204-expressing macrophages was reported in experimentally induced rat hepatic fibrosis. 11 Based on M1/M2 polarization, MHC class II-expressing macrophages could have functions to promote inflammation/tissue damage, whereas CD204-expressing macrophages might be related functionally to anti-inflammation and reparative fibrosis. Recently, MHC class II expression might be induced by the damage-associated molecular patterns produced by injured hepatocytes at very early stages. 34 Immunohistochemical analysis using antibodies to CD68, CD163, MHC class II, CD204, and Iba-1 would be useful to find out the pathological roles of macrophages.
M1 macrophage-related factors (such as IL-6, IL-1β, and MCP-1) and the M2 macrophage-related factors (such as IL-4 and IL-10) tended to increase with the increasing grade of pancreatic fibrosis. The M1 macrophage-related factors are involved in the activation of inflammatory cells and further induction/migration of macrophages. 35 –39 On the other hand, IL-4 could induce M2 macrophages, 35,40 and IL-10 has a wide variety of biological activity including anti-inflammation and immune functions. 41 There was no apparent increase in TGF-β1, the greatest fibrogenic factor which has been considered to be produced mainly by M2 macrophages. 35 The discrepancy between TGF-β1 expression at mRNA level and histopathological pancreatic fibrosis was considered to be a matter of sampling; as mentioned above, DBTC-induced rat pancreatic fibrosis lesions might vary not only from animal to animal but also from portion to portion in 3 or 4 portions examined in an individual animal.
Characteristics of Myofibroblasts
Myofibroblasts are formed by factors produced by macrophages and then become the main cell-producing collagens leading to fibrosis. During the development, the myofibroblasts can express vimentin, desmin, and α-SMA, of which the expressions are analyzed by immunohistochemistry with antibodies to these cytoskeletal proteins. 42 Myofibroblasts expressing vimentin, desmin, and α-SMA have been described in experimentally induced hepatic and renal fibrosis in rats. 5,20,42 Similarly, gradually increased numbers of myofibroblasts reacting to vimentin, desmin, and α-SMA were observed in the present DBCT-induced rat pancreatic fibrosis. Mesenchymal cells with immunophenotypical characteristics similar to myofibroblasts seen in the present experimentally induced rat pancreatic fibrosis were reported in spontaneous pancreatic fibrosis in dogs and cats. 23 On the basis of these findings, myofibroblasts should contribute to the development of pancreatic fibrosis.
Properties of GFAP-Expressing Myofibroblasts
Myofibroblasts reacting to GFAP are reported to appear in hepatic fibrosis. Because HSCs are identified by GFAP immunoexpression, it has been considered that myofibroblasts in hepatic fibrosis are derived from HSCs. 27 Pancreatic stellate cells are also known to express GFAP. 23,43,44 Glial fibrillary acidic protein-expressing PSCs were sporadically seen around the acinar lobules in control pancreas, and in the present DBTC-induced pancreatic fibrotic lesion, there were mesenchymal cells immunopositive for GFAP, showing increased number according to the grade (1+, 2+, and 3+) of fibrosis.
The expression of α-SMA is the most important characteristic of myofibroblasts. 3,45 The double immunofluorescence with a combination of vimentin/α-SMA and desmin/α-SMA indicated that 65% of vimentin-positive cells and 50% of desmin-positive cells expressed α-SMA. In addition, the double immunofluorescence staining of GFAP/α-SMA indicated that 60% of GFAP-positive cells coexpressed α-SMA. Increased number of GFAP-expressing cells was observed in spontaneous pancreatic fibrosis in dogs and cats. 23 Collectively, myofibroblasts might be originated partly from GFAP-expressing PSCs, showing vimentin, desmin, and α-SMA in various degrees.
Thy-1-Positive Mesenchymal Cells
Thy-1 is expressed in immature mesenchymal cells such as pericytes. 45 Thy-1-expressing mesenchymal cells were seen in the present DBTC-induced rat pancreatic fibrosis lesions, showing the increased number with the grade. Similarly, myofibroblasts with vimentin, desmin, and α-SMA expression were also increased with the grade. In addition to GFAP-positive PSCs, immature mesenchymal cells expressing Thy-1 were considered to be a possible source of myofibroblasts. 46 –49 Thy-1-positive immature mesenchymal cells including pericytes have been considered as a possible derivation of cutaneous wound fibrosis and renal fibrosis in rats. 45,50
In conclusion, the present study showed the cellular characteristics of macrophages and myofibroblasts in rat DBTC-induced pancreatic fibrosis. Macrophages could be analyzed based on the concept of CD68-expressing M1 and CD163-expressing M2 macrophage polarization. In addition, MHC class II- and CD204-expressing macrophages were polarized toward M1 and M2 macrophages, respectively. Myofibroblasts appeared in the fibrotic lesions, showing a variety of cytoskeletons such as vimentin, desmin, and α-SMA. Furthermore, the myofibroblasts expressed GFAP (a marker for PSCs) and Thy-1 (a marker for immature mesenchymal cells), suggesting the possible derivation from PSCs and immature mesenchymal cells. The present immunohistochemical findings would be useful for the analyses of inflammatory and reactive cells participating in experimentally induced pancreatic lesions.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no real, perceived or potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported partly by JSPS KAKENHI [grant numbers 26292152 and 19H03130 to J.Y.] and by the Platform Project for Supporting Drug Discovery and Life Science Research [Basis for Supporting Innovative Drug Discovery and Life Science Research (BINDS)] from AMED [grant numbers 3P18am 0101123 to J.Y.].
