Abstract
To investigate pathogenesis of post–bile duct (BD) injury fibrosis, interlobular BD epithelial injury was induced in male F344 rats by a single IP injection of α-naphthylisothiocyanate (75 mg/kg body weight) and rats were observed for 12 days. On days 1 to 2, cholangiocytes were injured and desquamated. On days 3 to 5, the affected BD began to regenerate, showing positive staining for CK19 and vimentin. On days 5 to 9, fibrotic areas gradually developed around regenerating BD in Glisson’s sheath. These consisted of cells positive for vimentin, desmin, and α-SMA; vimentin- and desmin-positive cells were increased in early stage (days 1–3), whereas α-SMA-positive cells appeared in mid (days 4–7) and late stages (days 8–12), although there were cells coexpressing these cytoskeletons. On day 12, BD regeneration almost completed, with reduced fibrosis. Macrophages positive for ED2 (CD163) increased transiently in early stage, whereas those reacting to ED1 (CD68), OX6 (MHC II), and SRA-E5 (CD204) showed a consistent increase throughout the experiment. Interestingly, OX6-positive cells were limited to Glisson’s sheath, whereas SRA-E5-positive cells were seen exclusively along sinusoids of hepatic lobules. MCP-1 mRNA increased significantly in early stage. This study shows that macrophages exhibiting different immunophenotypes and distributions participate in post-BD injury fibrosis associated with myofibroblasts expressing various mesenchymal cytoskeletons.
Introduction
Biliary fibrosis is a pathological process observed in cholangiopathies such as primary sclerosing cholangitis, bile duct stenosis/obstruction, and drug-induced bile duct damage, resulting in hepatic insufficiency and cirrhosis (Lazaridis, Strazzabosco, and LaRusso 2004). The abnormal interplay between injured cholangiocytes, mesenchymal cells (hepatic stellate cells [HSCs], portal fibroblasts, and vascular smooth muscle cells), and inflammatory cells (neutrophils, macrophages, and lymphocytes) has been implicated in cholangiopathy-related biliary fibrosis (Lazaridis, Strazzabosco, and LaRusso 2004; Strazzabosco, Fabris, and Spirli 2005). Perhaps because of these complicated interactions, the pathogenesis of biliary fibrosis remains poorly understood.
It has been recently reported that macrophages and myofibroblasts are two major cell components with crucial roles in hepatic and renal fibrogenesis (Mo et al. 1999; Schuppan et al. 1999; Ide et al. 2001). Macrophages secrete soluble factors responsible for the induction of myofibroblasts capable of producing extracellular matrices (Wynn 2008). Therefore, fibrotic lesions may be abrogated by modulation of macrophage functions. Macrophages appearing in hepatic lesions show heterogeneous functions, demonstrable with immunohistochemical methods (Ide et al. 2005; Mori et al. 2009). Myofibroblasts have been regarded as contractile mesenchymal cells expressing various cytoskeletons such as vimentin, desmin, and α-smooth muscle actin (α-SMA) (Yamate et al. 2005; Hinz et al. 2007); the origin and properties have not yet been clarified in biliary fibrosis. In the present study, we investigated the immunophenotypes of macrophages and myofibroblasts in relation to biliary fibrosis in alpha-naphthylisothiocyanate (ANIT)–induced cholestatic rat liver lesions. ANIT is a well-characterized hepatotoxicant known to cause cholestatic liver injury (Kossor et al. 1993). Because the expression of colony stimulating factor-1 (CSF-1), monocyte chemoattractant protein-1 (MCP-1), and transforming growth factor-β1 (TGF-β1) are correlated with the extent of hepatic injury (Kim et al. 1998; Qi et al. 1999; Zhang et al. 2004; Bataller and Brenner 2005; Guo et al. 2006; Pinzani et al. 2008), we examined the expression patterns of these factors. The present study demonstrates that macrophages and myofibroblasts, each showing heterogeneous immunophenotypes, participate in post–bile duct injury fibrosis, and that their appearance correlates with the cytokines examined, in particular MCP-1.
Materials and Methods
Animals and ANIT-Induced Hepatic Lesions
Five-week-old, male F344 rats (100 to 120 g body weight) were purchased from Charles River Japan (CRJ, Hino, Shiga, Japan). These animals were housed in an animal room at a controlled temperature of 21 ± 3°C and with a 12-hour light-dark cycle; they were provided a standard diet for rats (MF, Oriental Yeast Co. Ltd., Tokyo, Japan) and tap water ad libitum. After a 1-week acclimatization period, they were divided into control (3 rats) and ANIT injection groups (21 rats). The animals in the ANIT group were given a single intraperitoneal (IP) injection of ANIT at a dose of 75 mg/kg body weight dissolved in olive oil (Calvo et al. 2001). Three rats were euthanized by exsanguination under ether anesthesia on each of postinjection (PI) days 1, 2, 3, 5, 7, 9, and 12. Control rats received an equal volume of olive oil and were euthanized immediately after the injection (PI day 0). One hour before euthanasia, all the rats received IP injection of 5-bromo-2′-deoxyuridine (BrdU: Sigma-Aldrich Co., St. Louis, MO, USA) at the dose of 50 mg/kg body weight.
At necropsy, blood was collected from the abdominal artery, and serum samples were tested for aspartate transaminase (AST), alanine transaminase (ALT), alkaline phosphatase (ALP), and total bilirubin (T. Bil) with a Clinical Analyzer 7170 (Hitachi, Tokyo, Japan).
The animal experiments were conducted under the institutional guidelines approved by the ethical committee of Osaka Prefecture University for animal care.
Histopathology and Immunohistochemistry
Tissues from the left external lobe of the livers were fixed in 10% neutral buffered formalin, Zamboni’s fixative (0.21% picric acid and 2% paraformaldehyde in 130 mM phosphate buffer, pH 7.4), and periodate-lysine-paraformaldehyde (PLP) solutions (Ide et al. 2003). These tissues were dehydrated and embedded in paraffin. Deparaffinized sections, cut at 4 µm in thickness, were stained with hematoxylin and eosin (HE) for histopathology. Zamboni’s solution– or PLP-fixed sections were used for the immunohistochemistry with mouse monoclonal antibodies against macrophages (ED1 (for CD68), ED2 (for CD163), OX6 (for major histocompatibility complex (MHC) class II), SRA-E5 (for CD204)), mesenchymal cytoskeletal proteins (vimentin, desmin, and α-SMA), bile duct epithelia (CK19), and incorporated BrdU for proliferating cholangiocytes (Table 1 ). After pretreatment shown in Table 1, tissue sections were treated with 3% H2O2 in phosphate buffered saline (PBS) to quench endogenous peroxidase, and then with 5% skimmed milk in PBS to inhibit nonspecific reactions. The sections were incubated with each primary antibody overnight at 4°C, followed by reaction with the secondary antibody (Histofine simplestain MAX-PO, Nichirei, Tokyo, Japan). Positive reactions were visualized with 3,3′-diaminobenzidine (DAB substrate kit, Vector Laboratories, Burlingame, CA, USA). Nonimmunized mouse IgG served as a negative control. Sections were counterstained lightly with hematoxylin.
Primary antibodies used for the immunohistochemistry and immunofluorescence staining.
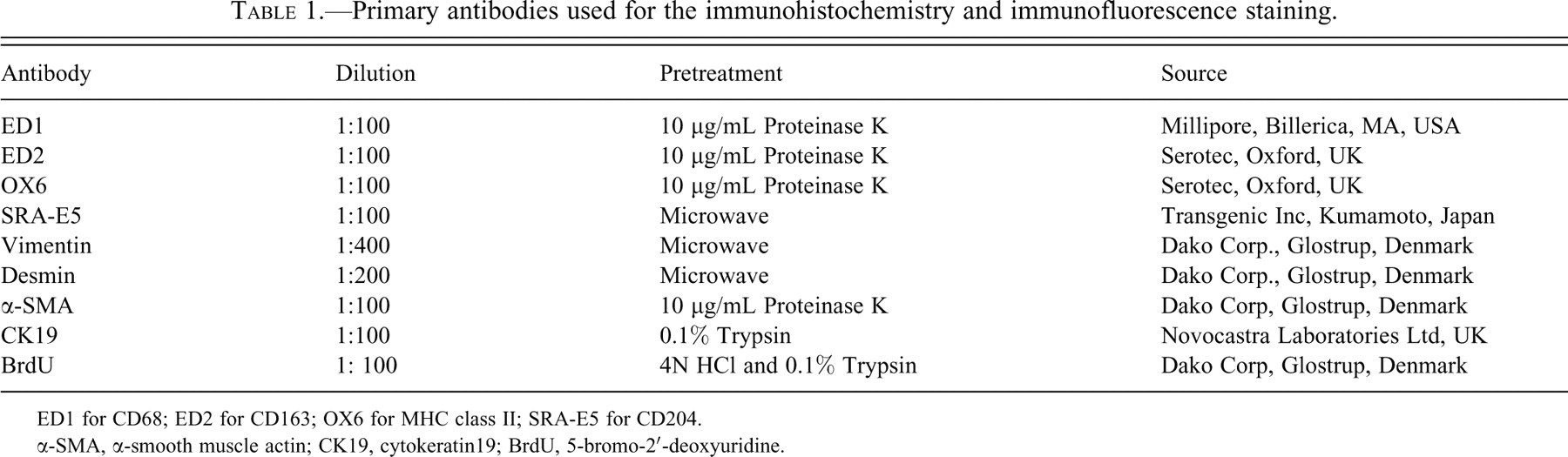
ED1 for CD68; ED2 for CD163; OX6 for MHC class II; SRA-E5 for CD204.
α-SMA, α-smooth muscle actin; CK19, cytokeratin19; BrdU, 5-bromo-2′-deoxyuridine.
Cells showing a distinct immunopositive reaction for ED1, ED2, OX6, or SRA-E5 were counted in 5 randomly selected areas (0.2 mm2) of each rat at the periportal area (including Glisson’s sheath) of the hepatic lobule at a magnification of ×400 using ImageJ software (http://rsbweb.nih.gov/ij/). Cells positive for vimentin, desmin, and α-SMA in the periportal area including the Glisson’s sheath was evaluated semiquantitatively using grades as shown in Table 2 .
Semiquantitative analyses of mesenchymal cells and cholangiocytes in the Glisson’s sheath.
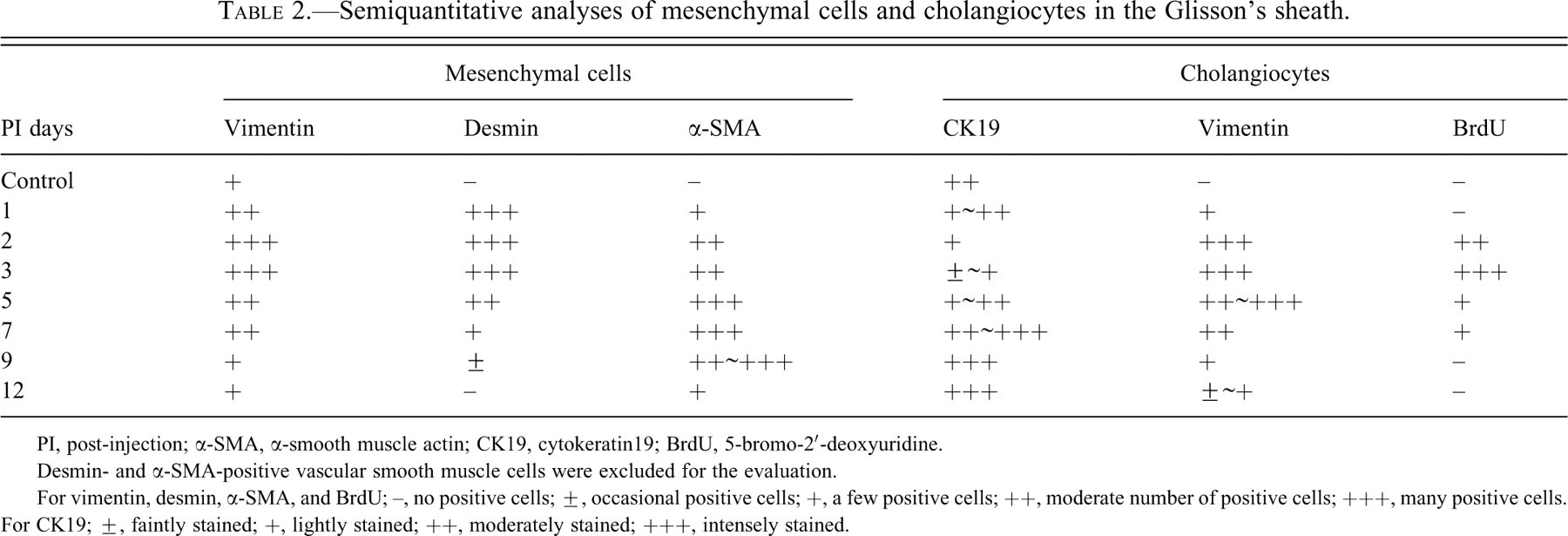
PI, post-injection; α-SMA, α-smooth muscle actin; CK19, cytokeratin19; BrdU, 5-bromo-2′-deoxyuridine.
Desmin- and α-SMA-positive vascular smooth muscle cells were excluded for the evaluation.
For vimentin, desmin, α-SMA, and BrdU; –, no positive cells; ±, occasional positive cells; +, a few positive cells; ++, moderate number of positive cells; +++, many positive cells. For CK19; ±, faintly stained; +, lightly stained; ++, moderately stained; +++, intensely stained.
Immunofluorescence Staining
Double immunofluorescence labeling was carried out using ANIT-induced fresh frozen liver sections obtained on PI day 3. The combinations in the dual immunofluorescence labeling were ED1/OX6, ED2/OX6, desmin/vimentin, α-SMA/vimentin, desmin/α-SMA, CK19/vimentin, or CK19/α-SMA. Briefly, after fixation in cold acetone/methanol (1:1) for 10 min, the sections were incubated with 10% normal goat serum for 30 min. The sections were reacted with the primary antibody overnight at 4°C, and then with the secondary antibody conjugated with Alexa 488 (Invitrogen, Carlsbad, CA, USA) for 1 hr at room temperature. The sections were incubated with the second primary antibody followed by Cy3 conjugated secondary antibody (Jackson Immunoresearch Laboratories, Inc., Baltimore, PA, USA). Alexa fluor 488 labeled OX6 (AbD Serotec, Oxford, UK) and Hilyte fluor 555 (Dojindo Molecular Technologies, Inc., Tokyo, Japan) labeled anti-vimentin antibodies were used for ED1/OX6 and ED2/OX6, as well as CK19/vimentin combinations, respectively. The sections were covered with Vectashield mounting medium containing 4′6 diamidino-2-phenylindole (DAPI) (Vector Laboratories Inc., Burlingame, CA, USA) for nuclear staining and analyzed by a laser scanning confocal microscope system (C1si, Nikon, Japan). Nonimmunized mouse IgG was used as a negative control, and PBS was used as washing solution between steps.
Reverse Transcriptase Polymerase Chain Reaction (RT-PCR)
Liver samples were immersed in RNAlater® (Qiagen GmbH, D-40724 Hilden, Germany) and stored at –80°C. The extraction of RNA was carried out by using an SV total RNA isolation system kit® (Promega Corporation, Madison, WI, USA). After the isolation, concentration of RNA was measured on a nanodrop1000™ spectrophotometer (Thermo Scientific, Wilmington, DE, USA) and then reverse transcribed to cDNA with the superscript II transcriptase system® (Invitrogen, Carlsbad, CA, USA) (Ide et al. 2003) and amplified, in duplicate, with the SYBR® Green Real-Time PCR Master Mix (Toyobo Co. Ltd, Osaka, Japan) using the Linegene system (Bioflux, Tokyo, Japan) for each of the specific primers of rat MCP-1, TGF-β1, CSF-1, and ribosomal protein S18 (Rps18; an internal control gene). The following conditions were used for the amplification: for MCP-1, after 1 min of denaturation at 95°C, 40 cycles of 15 sec of denaturation at 95°C, 15 sec of annealing at 62°C, and 20 sec of extension at 72°C; for TGF-β1, after 1 min of denaturation at 95°C, 40 cycles of 15 sec of denaturation at 95°C, 15 sec of annealing at 64°C, and 20 sec of extension at 72°C; for CSF-1, after 1 min of denaturation at 95°C, 40 cycles of 15 sec of denaturation at 95°C, 15 sec of annealing at 60°C, and 20 sec of extension at 72°C. The oligonucleotide sequences used for PCR are shown in Table 3 . The polymerase chain reaction (PCR) products were electrophoresed in 2% agarose gel, and DNA was stained with ethidium bromide on the gel.
Oligonucleotide sequences of primers used in the real-time PCR.

MCP-1, monocyte chemoattractant protein-1; TGF-β1, transforming growth factor-β1; CSF-1, colony stimulating factor-1; Rps18, ribosomal protein s18.
Statistical Analyses
Obtained data represented mean ± standard deviation (SD), and statistical analysis was performed using the Tukey’s test or Student’s t-test. Significance was accepted at p < .05.
Results
ANIT-Induced Hepatic Lesions
The body weight was decreased slightly on days 1 and 2, in contrast to that on day 0, indicating possible influence of the ANIT injection (Fig. 1 ). Histopathologically, no significant changes were seen in the livers of control rats (Fig. 2A). In ANIT-injected rats, lesions were seen mainly in the Glisson’s sheath and in the adjacent areas. On PI day 1, desquamation of necrotic cholangiocytes of the interlobular bile ducts in the Glisson’s sheath was seen, accompanied by edema and infiltration of a small number of neutrophils; sinusoidal dilation containing a few inflammatory cells and degenerating hepatocytes with eosinophilic cytoplasm were observed in the vicinity of the Glisson’s sheath (Fig. 2B). On PI day 2, in addition to a few neutrophils, mononuclear cells and spindle-shaped cells began to be seen in the affected Glisson’s sheath, having more increased edematous change (Fig. 2C); these mononuclear cells and spindle-shaped cells were regarded as macrophages and myofibroblasts, respectively, as mentioned below. On PI day 3, epithelial cells with hyperchromatic nuclei began to surround the damaged interlobular bile ducts, indicating regeneration of the ducts; neutrophils almost disappeared, although mononuclear cells were still detectable (Fig. 2D). On PI days 5, 7, and 9, the severity of lesions compared with those seen on PI days 1–3 decreased gradually, along with decreased numbers of mononuclear cells, but the spindle-shaped cells became more prominent in the affected lesions, particularly on PI days 5 (Fig. 2E) and 7 (Fig. 2F). By PI day 12, the interlobular ducts were relatively normal and inflammatory changes, including cellular infiltrates and edema, had disappeared, although the fibrotic lesion was still present.
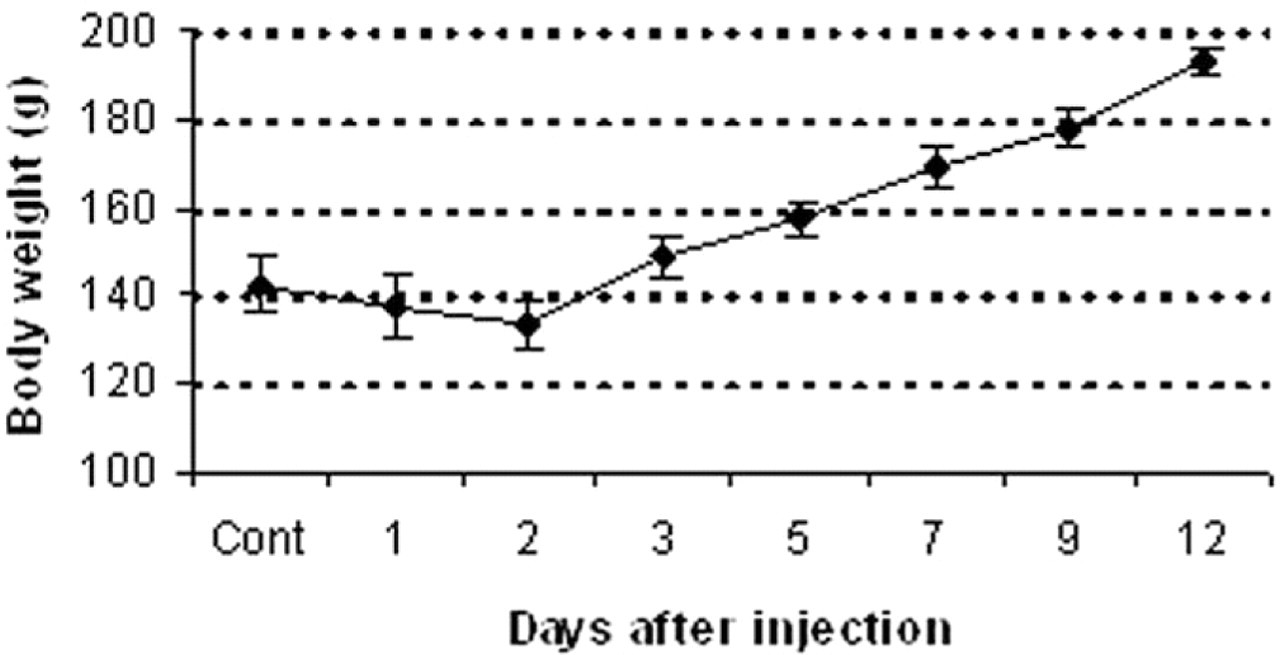
Body weight changes in rats on days 0 (control), 1, 2, 3, 5, 7, 9, and 12 following a single injection of α-naphthylisothiocyanate (ANIT). Cont: control.
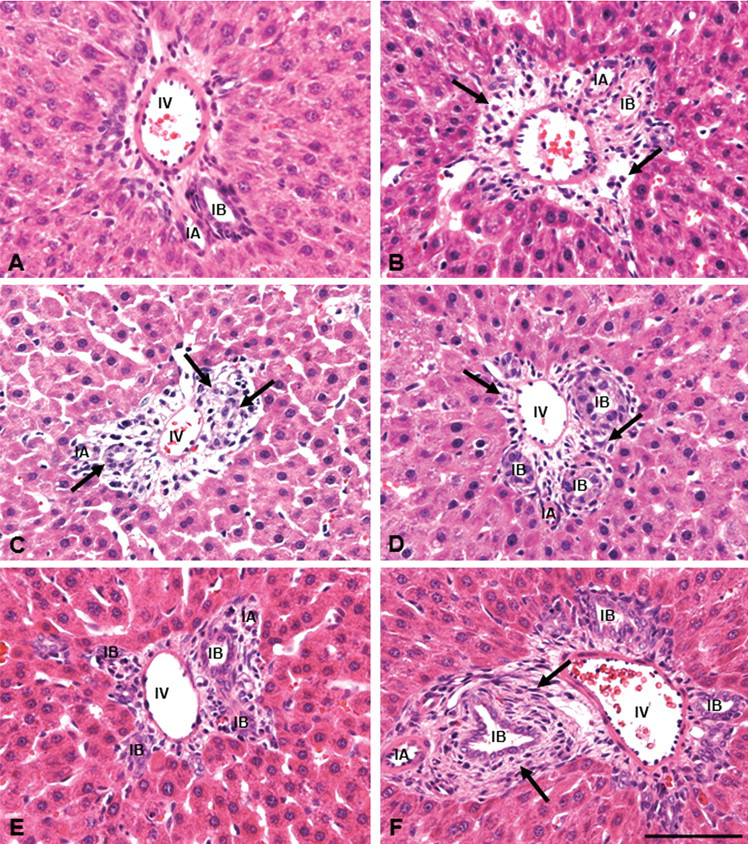
Histopathology in control (A) and ANIT-injected (B–F) rats. The Glisson’s sheath in control consists of interlobular arteriole, interlobular vein, and interlobular bile ducts, as well as fibroblastic cells (A). In ANIT-injected rats, desquamation of bile duct epithelia, edema, and infiltration of a small number of neutrophils (arrows) are seen in the affected Glisson’s sheath on postinjection (PI) day 1 (B); on PI day 2, regenerating cholangiocytes in interlobular bile ducts (arrows), edema, and infiltration of mononuclear cells are observed (C); almost regenerated bile ducts, less edema, and infiltrating mononuclear cells (arrows) are seen on PI day 3 (D). In the affected Glisson’s sheath, on PI day 5 a greater number of mononuclear cells are present (E); and on PI day 7 (F), spindle-shaped fibroblastic cells (arrows) are developed around the bile ducts, indicating biliary fibrosis. IA, interlobular arteriole; IB, interlobular bile duct; IV, interlobular vein. HE. Bar: A–F, 50 µm.
The values of AST on PI days 1 and 2, ALT on PI days 1–3, and ALP on PI days 2 and 3 showed a significant increase (Figs. 3A-C), indicating transient hepatic damage due to the action of ANIT. The T. Bil levels were significantly increased on PI day 1, peaked on PI day 2, and then gradually decreased on PI days 3 to 12; however, values did not return to control levels until day 12 (Fig. 3D), indicative of persistent cholestasis in ANIT-induced hepatic lesions.
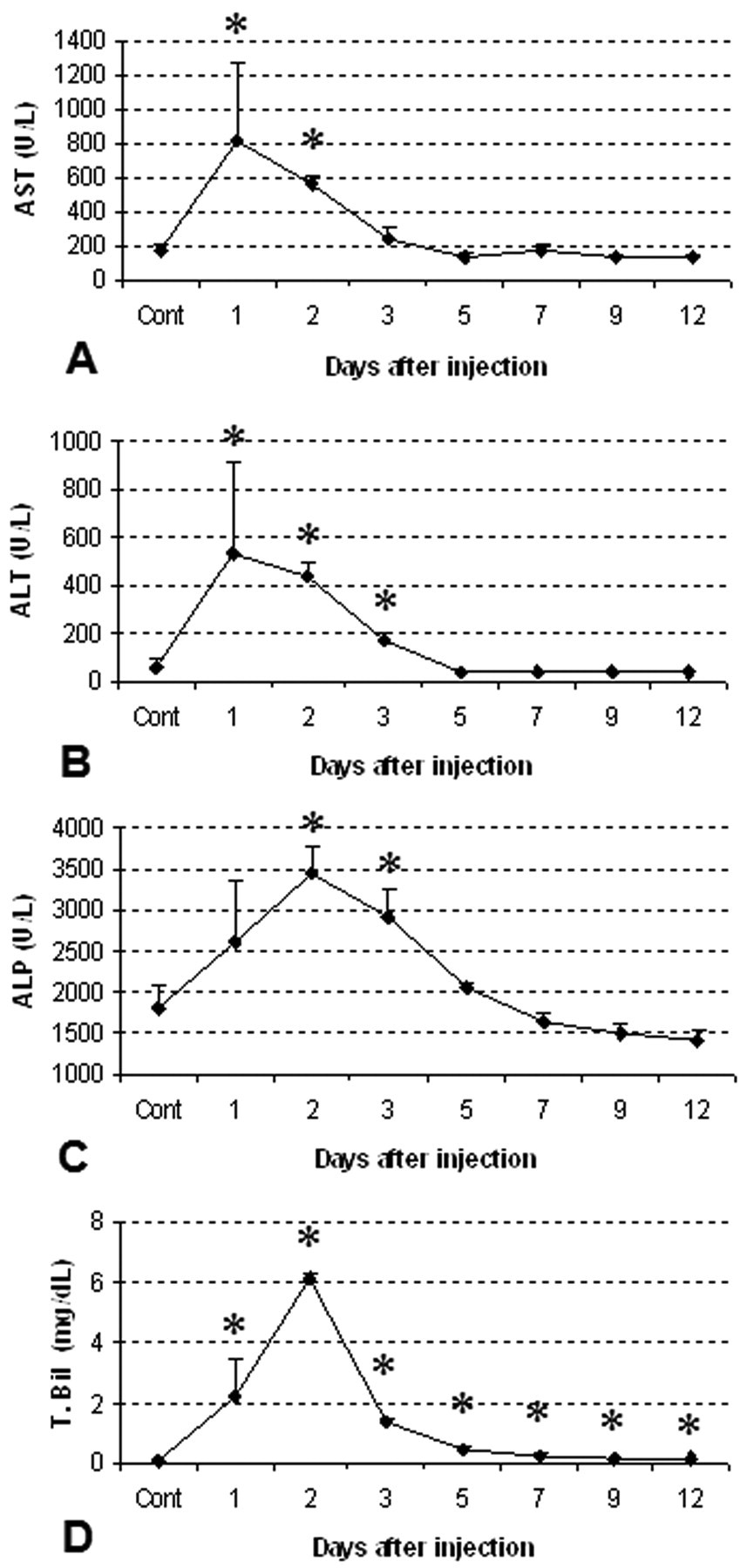
Blood biochemical analyses in control and in ANIT-injected rats (PI days 1–12). Aspartate transaminase (AST) (A), alanine transaminase (ALT) (B), alkaline phosphatase (ALP) (C), and total bilirubin (T. Bil) (D). Cont: control. Student’s t-test. *, p < .05, significantly different from controls.
Immunohistochemistry for Macrophages
The kinetics of macrophages reacting to ED1, ED2, SRA-E5, and OX6 is shown in Figure 4 . In the liver of controls, some ED1-positive cells were seen in the hepatic lobules and Glisson’s sheath (Fig. 5A). Although a small number of ED2-positive cells were also observed in the Glisson’s sheath, the positive cells were present mainly along the sinusoids of the hepatic lobule, indicative of Kupffer cells (Fig. 5C). SRA-E5-positive cells were present exclusively along the sinusoids, like ED2-positive Kupffer cells, but no SRA-E5-reacting cells were seen in the Glisson’s sheath (Fig. 5E). On the other hand, OX6-positive cells were rarely seen in the hepatic lobules of the controls; the positive cells with spindle-like or dendritic appearances were present only in the Glisson’s sheath (Fig. 5G).
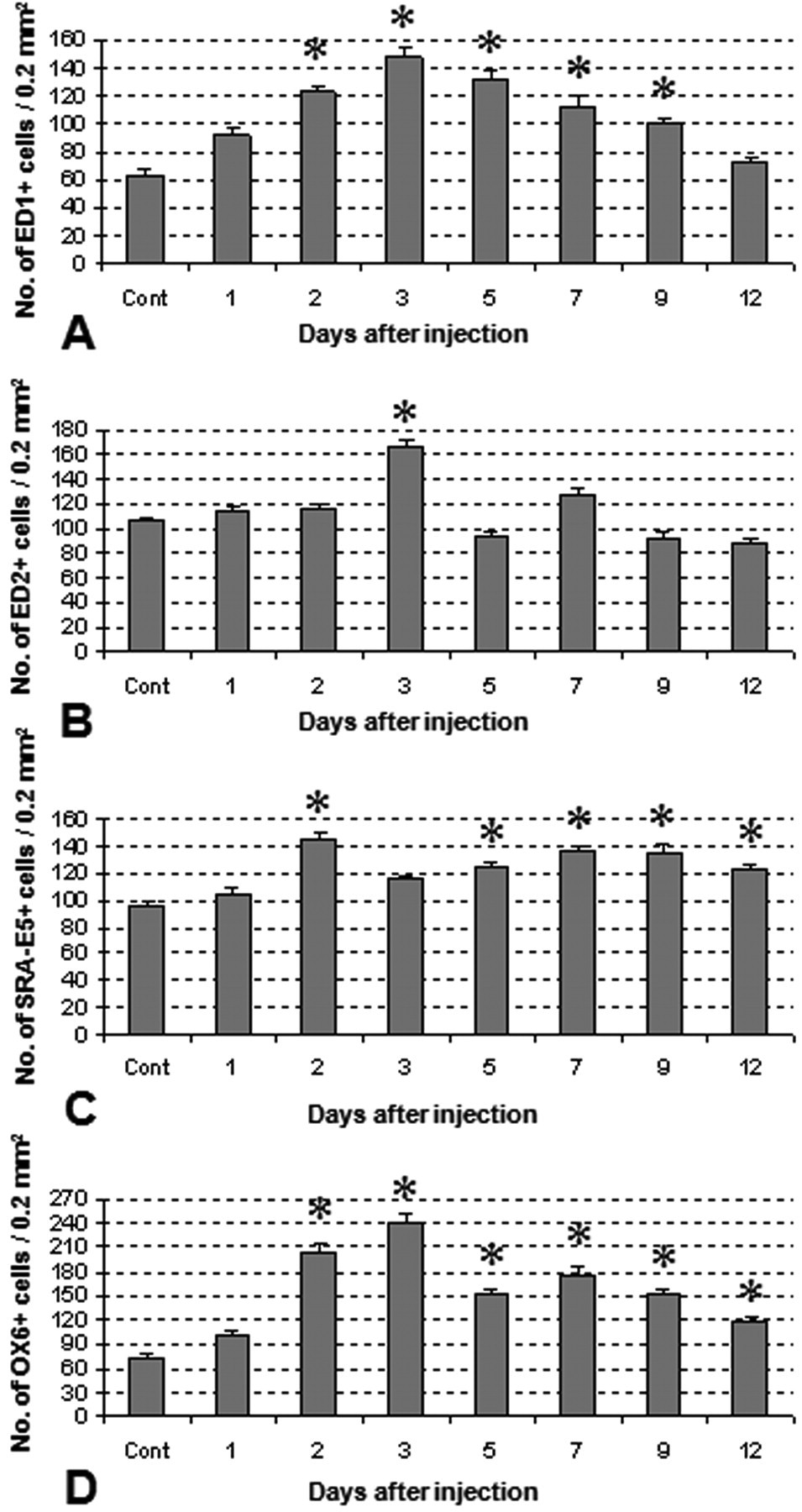
The kinetics of macrophages in the Glisson’s sheath and hepatic lobule around the Glisson’s sheath in livers of control and ANIT-injected (PI days 1–12) rats. The number of macrophages positive for ED1 (for CD68) significantly increases on PI days 2–9 (A), and that for ED2 (for CD163) increases transiently on PI day 3 (B). The macrophage number for SRA-E5 (for CD204) (C) and OX6 (for MHC class II) (D) shows a consistent increase on PI days 2–12 (except on PI day 3 for SRA-E5). Tukey’s test; *, p < .05, significantly different from controls.
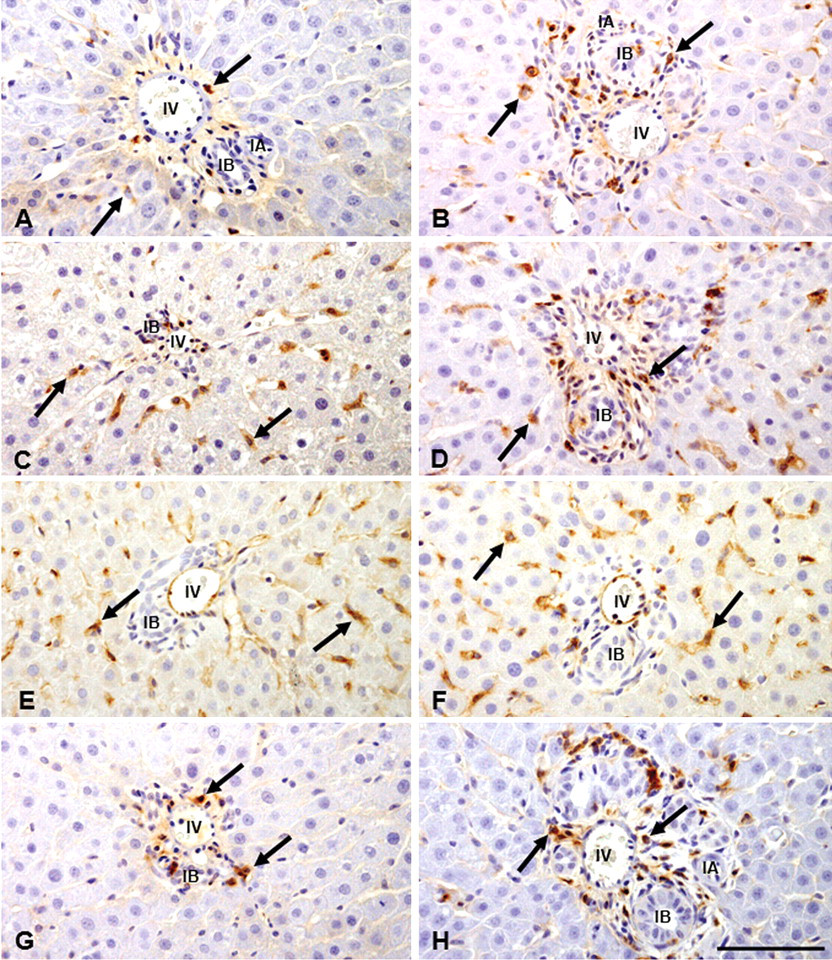
Distribution of macrophages in the Glisson’s sheath and the surrounding hepatic lobule of control (A, C, E, G) and ANIT-induced liver lesions on PI day 3 (B, D, F, H). As compared with controls (A, C, E, G), ED1(B)-, ED2(D)-, SRA-E5(F)-, and OX6(H)-positive macrophages show increased numbers in the affected areas. Interestingly, ED2-positive cells in control are along the sinusoids (C), indicating Kupffer cells; increased numbers of ED1(B)- and ED2(D)-positive cells are seen in the affected Glisson’s sheath; SRA-E5-positive cells are seen mainly along the sinusoids (F) of hepatic lobule around the Glisson’s sheath, whereas the appearance of OX6-positive cells are limited to the Glisson’s sheath (H). Immunohistochemistry, counterstained with hematoxylin. Arrows indicate positive cells. IA, interlobular arteriole; IB, interlobular bile duct. Bar: A-H, 50 µm.
The number of ED1-positive cells began to increase significantly as early as PI day 2, reaching a maximum on PI day 3, and thereafter maintaining significantly increased numbers up to PI day 9 (Fig. 4A); these ED1-positive cells were round or spindle-shaped in appearance (Fig. 5B). Increased numbers of ED2-positive cells were present only on PI day 3, and the positive cells were round to oval in shape, having abundant cytoplasm (Fig. 5D). The number of SRA-E5-positive cells was significantly increased on PI days 2 to 12, except on day 3, suggesting a consistent increment in cell numbers (Fig. 4C); the positive cells were more prominent in the sinusoids in the vicinity of the Glisson’s sheath, and they exhibited round to fusiform shapes (Fig. 5F). The OX6-positive cell number showed a consistent, significant increase on PI days 2 to 12 (Fig. 4D); the positive cells were large round or stellate in shape, and distributed mainly in the Glisson’s sheath (Fig. 5H).
The double immunofluorescence labeling on PI day 3 for OX6 and ED1 (Fig. 6A–C) or ED2 (Fig. 6D–F) revealed that 56% and 26% of the OX6-positive macrophages in the Glisson’s sheath were reactive for ED1 and ED2, respectively.
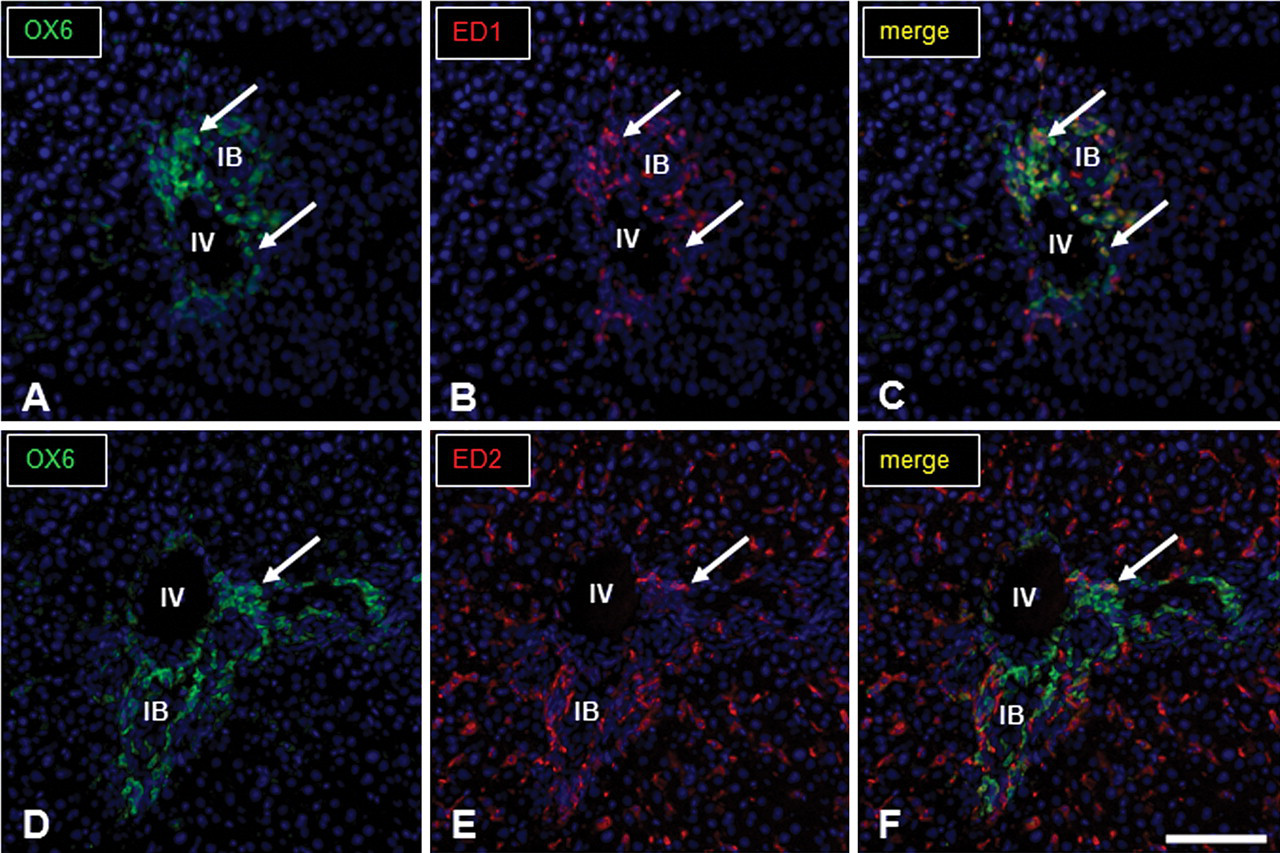
Immunofluorescence staining for OX6 and ED1 or OX6 and ED2 in the Glisson’s sheath of ANIT-treated livers on PI day 3. Photomicrographs show macrophages positive for OX6 (green, A and D; arrows) and ED1 (red, B; arrows) or ED2 (red, E; arrows) and the digital merges (yellow, C for OX6 and ED1 or F for OX6 and ED2; arrows). The number of macrophages coexpressing OX6/ED1 (C) is greater than those of OX6/ED2 (F). No signal was observed in negative control sections. IV, interlobular vein; IB, interlobular bile duct. Immunofluorescence, DAPI for nuclear staining. Bar: A-F, 100 µm.
Immunohistochemistry for Myofibroblasts Using Cytoskeletal Markers Such as Vimentin, α-SMA, and Desmin
In the Glisson’s sheath of control livers, vascular smooth muscle cells reacted weakly for desmin and strongly for α-SMA. However, no mesenchymal cells positive for desmin or α-SMA were apparent. Vimentin-positive cells were sporadically seen in the Glisson’s sheath and along sinusoids in the vicinity of Glisson’s sheath; the latter were regarded as HSCs.
In ANIT-injected rats, vimentin-positive cells (Fig. 7A) began to increase on PI day 1 and peaked on PI days 2 and 3 (Table 2). Desmin-positive cells showed the highest number as early as PI day 1 and the increased number persisted until PI day 3 (Table 2). In contrast, α-SMA-positive cells gradually increased from PI days 1 to 9, and slightly decreased onward (Table 2). Desmin- and α-SMA-positive cells were located around the interlobular bile ducts (Fig. 7B, C), whereas vimentin reactivity was seen not only in spindle cells in the Glisson’s sheath but also a few HSCs present along the sinusoids. These spindle cells in the Glisson’s sheath showed co-expression of vimentin, desmin, and α-SMA in the double immunofluorescence labeling on PI day 3 (Fig. 8A–I). Interestingly, regenerating cholangiocytes were also reactive for vimentin.
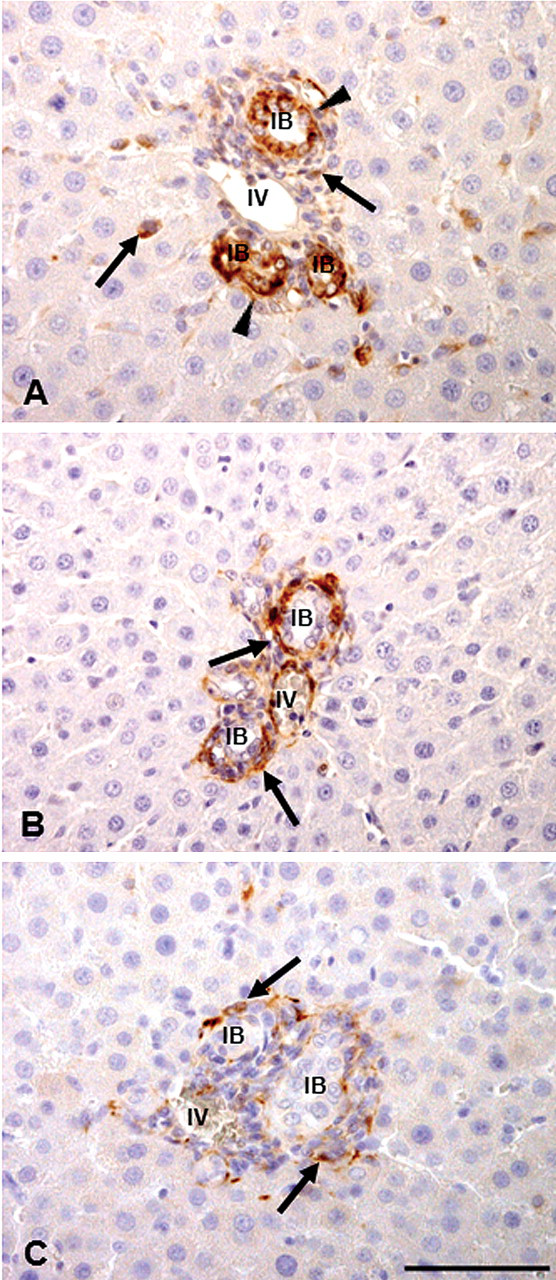
Mesenchymal cells reacting to vimentin (A), desmin (B), and α-smooth muscle actin (α-SMA) (C) in the Glisson’s sheath of ANIT-induced liver lesions on PI day 3. Vimentin-positive mesenchymal cells in the hepatic lobule and affected Glisson’s sheath are seen (A, arrows); desmin– (B, arrows) and α-SMA– (C, arrows) positive mesenchymal cells are present mainly around regenerating bile ducts in the affected Glisson’s sheath. Note that regenerating cholangiocytes
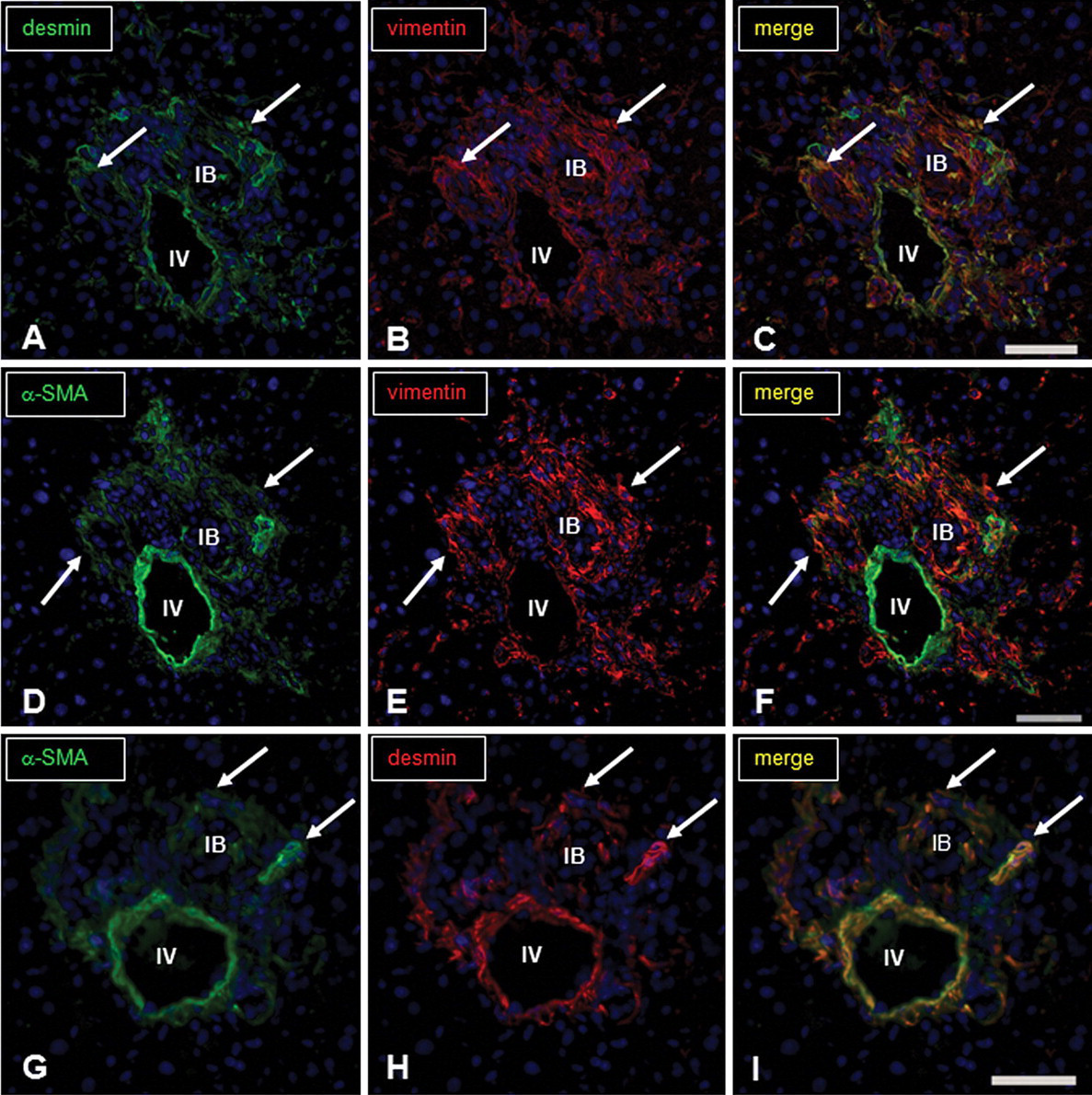
Immunofluorescence staining for vimentin-, desmin-, and α-SMA-positive myofibroblasts in the Glisson’s sheath of ANIT-treated livers on PI day 3. There are myofibroblasts showing immunofluorescence labeling for desmin (A, green; arrows) and vimentin (B, red; arrows), and coexpressing desmin and vimentin (C, digital merge, yellow; arrows); those for α-SMA (D, green; arrows) and vimentin (E, red; arrows), and coexpressing α-SMA and vimentin (F, digital merge, yellow; arrows); those for α-SMA (G, green; arrows) and desmin (H, red; arrows), and coexpressing α-SMA and desmin (I, digital merge, yellow; arrows). No signal was observed in negative control sections. Immunofluorescence, DAPI for nuclear stain. IV, interlobular vein; IB, interlobular bile duct. Bar: A–I, 50 µm.
Immunohistochemistry for Cholangiocytes
Anti-CK 19 antibody is useful for the detection of cholangiocytes. The CK19 reactivity of injured cholangiocytes on PI days 1 to 3 was decreased or lost (Fig. 9A), and on PI days 5 to 12, it gradually increased with reconstitution of bile ducts (Fig. 9B). In addition, vimentin immunoreactivity was seen in regenerating cholangiocytes (Fig. 7A), as mentioned above. To confirm the findings, double immunofluorescence staining for CK 19 and vimentin was conducted using liver samples on PI days 2 and 3, and coexpression of these markers was confirmed in regenerating cholangiocytes (Fig. 10A–C). Vimentin expression was seen at the basal surface of the cholangiocytes. Double immunofluorescence staining for CK19 and α-SMA revealed no colocalization (Fig. 10D–F).
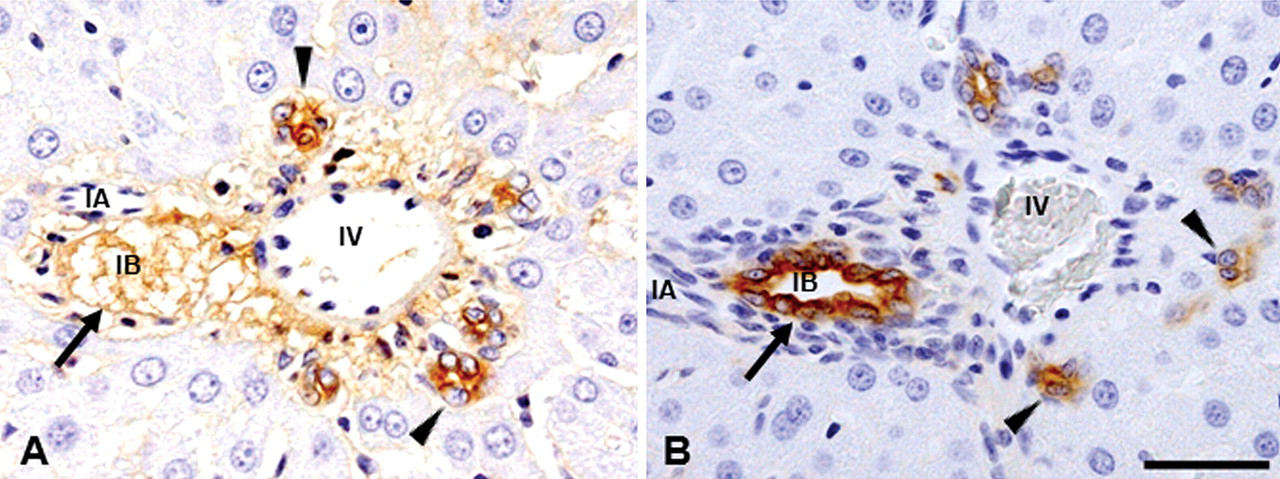
Immunohistochemistry for cholangiocytes in the Glisson’s sheath of ANIT-induced livers. Loss of bile duct epithelia with edematous change is shown (arrow) on PI day 1, although cholangiocytes in some bile ducts are still reactive for CK19 (arrowheads) (A). On the other hand, intense immunoreactivity for CK19 is seen in completely regenerated bile duct epithelia on PI day 12 (B, arrow); interestingly, aggregations of CK19-positive cells without forming clear ducts are present within and around the affected Glisson’s sheath (arrowheads). No signal was observed in negative control sections. Immunohistochemistry, counterstained with hematoxylin. IA, interlobular arteriole; IV, interlobular vein; IB, interlobular bile duct. Bar: A–B, 50 µm.
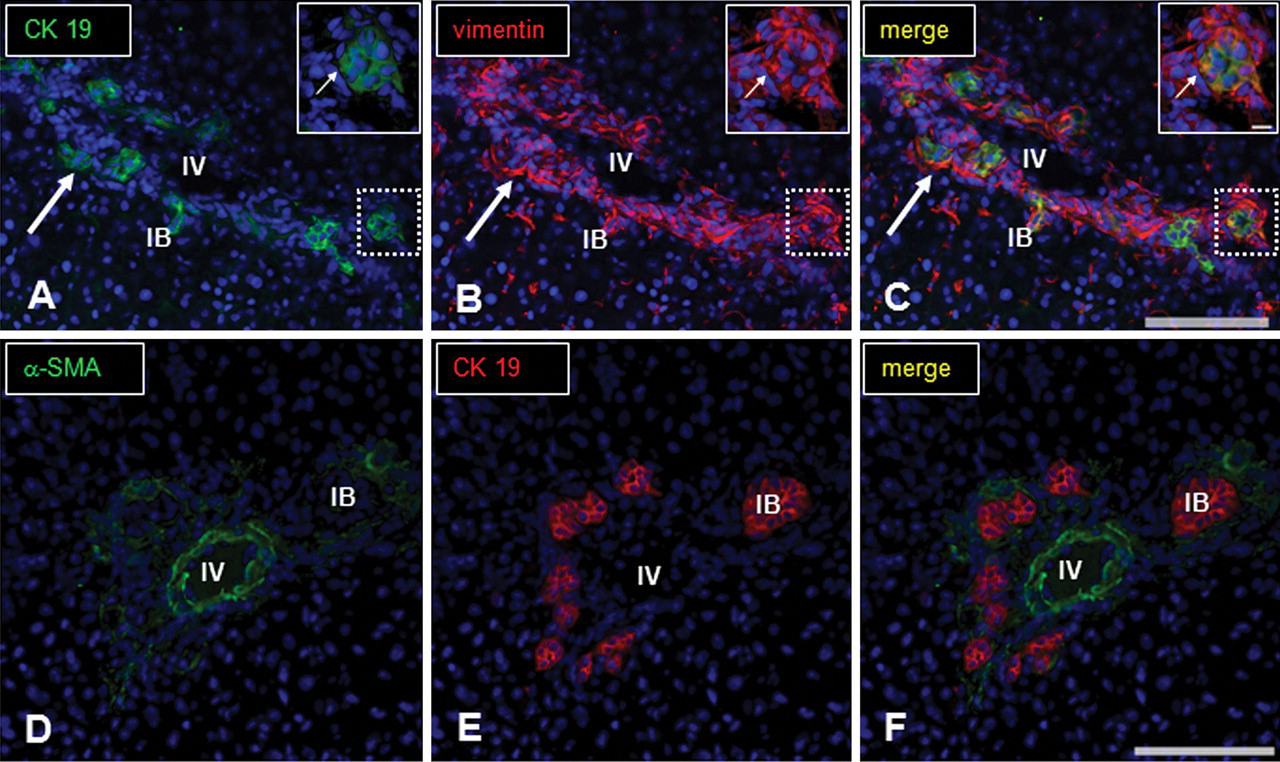
Immunofluorescence staining for CK19, vimentin, and α-SMA in the Glissons’s sheath of ANIT-treated livers on PI day 3. There are regenerating cholangiocytes positive for CK19 (A, green; arrow) and vimentin (B, red; arrow), and coexpressing CK19 and vimentin (C, digital merge, yellow; arrow); insets in “B” for vimentin and “C” for coexpression indicate a dotted rectangle (inset) in “A” for CK19. Although the regenerating cholangiocytes give a positive reaction to CK19 (E, red; arrows), the cells do not react to α-SMA (D, green for α-SMA; F, digital merge for CK19 and α-SMA). No signal was observed in negative control sections. Immunofluorescence, DAPI for nuclear stain. IV, interlobular vein; IB, interlobular bile duct. Bar: A–F, 100 µm.
The BrdU- immunohistochemistry demonstrated that injured cholangiocytes started to proliferate on PI day 2 and continued until PI day 7 (Fig. 11 ) with a peak on PI day 3 (Table 2).
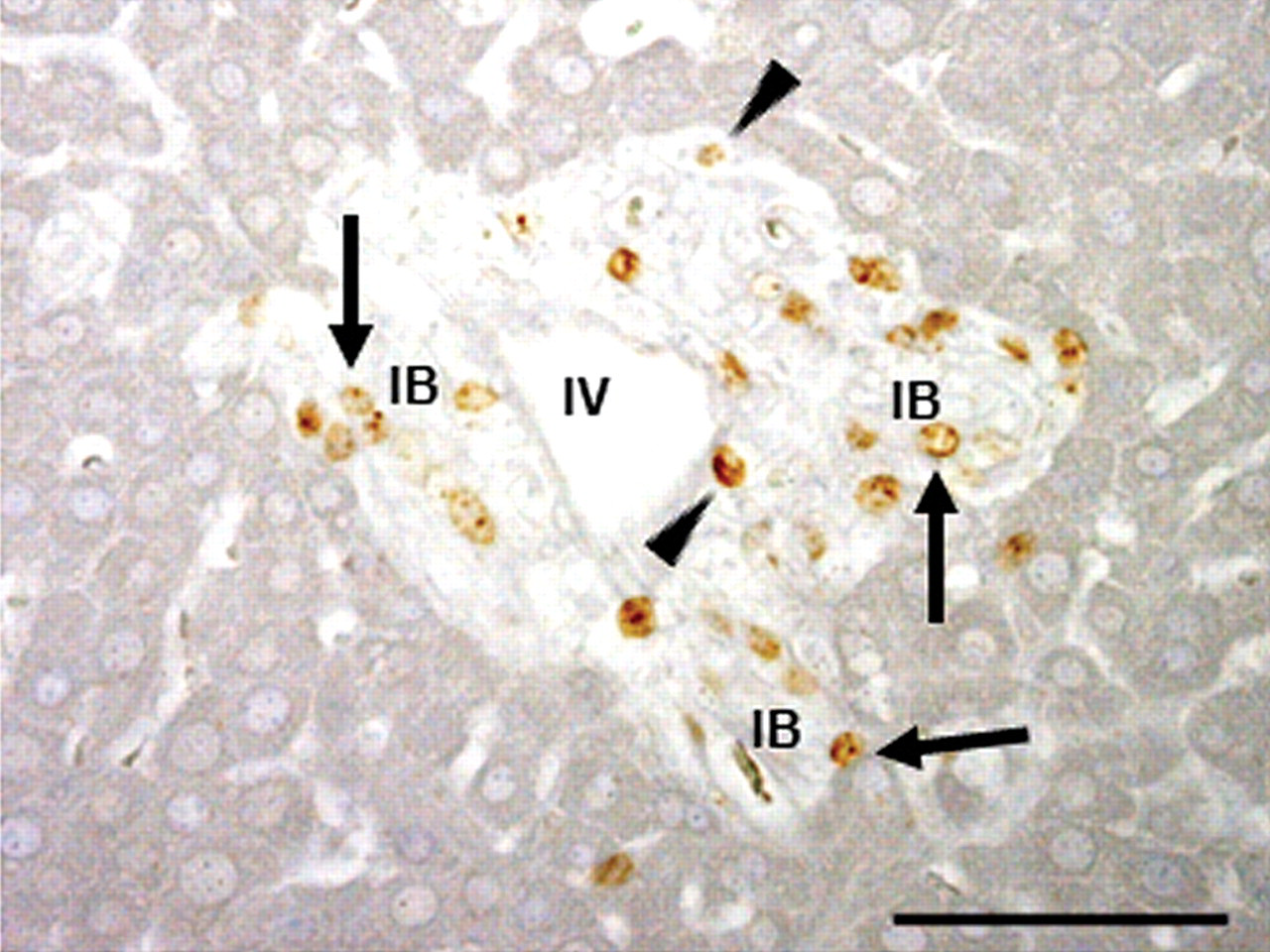
Proliferating cells in the affected Glisson’s sheath on PI day 2 using the BrdU immunostaining. Regenerating cholangiocytes show a positive reaction for BrdU (S-phase in cell cycle) (arrows). Some mesenchymal cells also react to BrdU (arrowheads), indicating proliferating myofibroblasts. No signal was observed in negative control sections. Immunohistochemistry, counterstained with hematoxylin. IV, interlobular vein; IB, interlobular bile duct. Bar: 50 µm.
Expression of mRNAs
Consistent with the increased number of macrophages, MCP-1 mRNA was increased significantly on PI days 1 and 2; on subsequent days (PI days 3 to 12), MCP-1 expression returned to control levels (Fig. 12A). The expression of TGF-β1 mRNA was significantly increased only on PI day 9 (Fig. 12B). CSF-1 mRNA did not show any significant change.
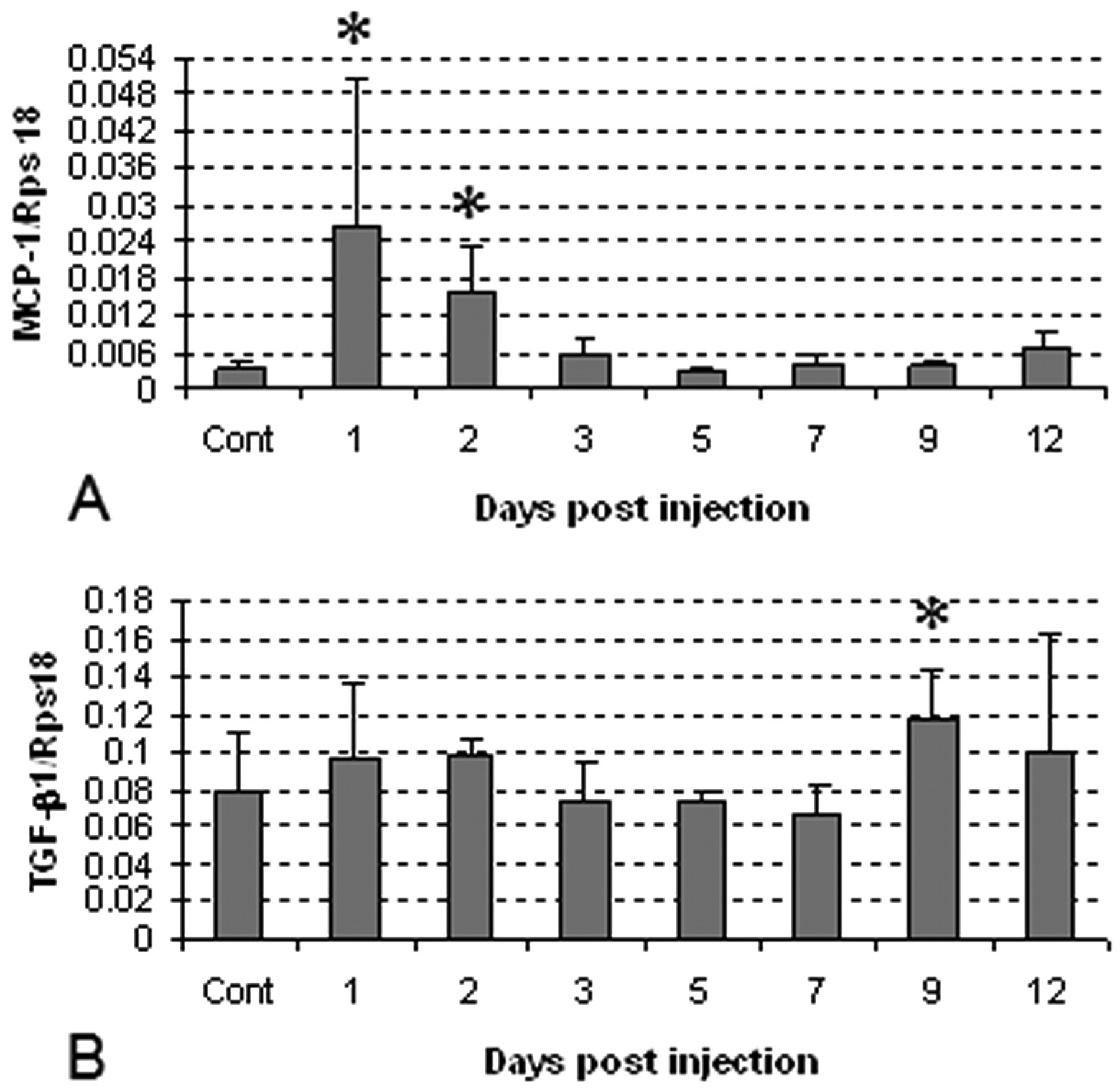
mRNA expressions of macrophage chemoattractant protein-1 (MCP-1) (A) and transforming growth factor-β1 (TGF-β1) (B) in control and ANIT-injected rats on PI days 1–12 by the RT-PCR methods. Cont: control. Student’s t-test; *, p < .05, significantly different from controls.
Discussion
ANIT-Induced Hepatic Lesions
ANIT-induced hepatic lesions were characterized by bile duct epithelial cell necrosis/desquamation and infiltration of inflammatory cells in the Glisson’s sheath, as well as degeneration of hepatocytes in the vicinity of the Glisson’s sheath. These histopathological changes are consistent with those reported previously (Kossor et al. 1993). Additionally, hepatic injury due to ANIT injection was indicated by markedly increased levels of AST, ALT, ALP, and T. Bil at early stage. The mechanism behind ANIT-induced liver injury has not yet been fully characterized. However, it has been suggested by others that, after direct injury of cholangiocytes by ANIT secreted in bile, inflammatory cells in the Glisson’s sheath produce oxidative factors (oxidants), resulting in hepatocyte injury (Dietrich et al. 2001; Kodali et al. 2006).
In the present study, cholangiocytes began to regenerate on PI days 2 and 3, demonstrable by the BrdU immunohistochemistry. Following regeneration, CK19-positive cholangiocytes were noted around the injured bile ducts, indicating remodeling of the bile duct. Although vimentin is generally specific for mesenchymal cells (Azumi and Battifora 1987; Sappino, Schürch, and Gabbiani 1990), the double immunofluorescence staining showed that CK19-positive regenerating cholangiocytes also reacted to vimentin. Interestingly, vimentin expression was characteristically seen at the basal cytoplasm adjacent to the basement membrane. Vimentin expression in regenerating cholangiocytes indicated reconstitution of cholangiocytes in the bile duct system (Milani et al. 1989). In the present ANIT-induced lesions, it is interesting to note that biliary fibrosis began to develop on PI day 2 and, on subsequent days, the fibrosis advanced gradually with increased numbers of myofibroblasts. The finding that T. Bil level did not return to control levels until PI day 12 is consistent with cholestasis due to the biliary fibrosis observed.
Macrophages
Some investigators have suggested that neutrophils play important roles in the pathogenesis of ANIT-induced hepatic lesions (Dietrich et al. 2001). In the present study, neutrophils were temporarily seen only on day 1. In contrast, macrophages appeared as the major cell component from day 2 onward. To our knowledge, this is the first report of macrophage appearance using different macrophage-specific antibodies in ANIT-induced hepatic lesions.
Macrophages can be divided into three categories: blood monocyte-derived exudative macrophages, resident macrophages (Kupffer cells), and antigen-presenting dendritic cells. They differ in ontogeny, morphology, immunophenotype, tissue distribution, and function; the immunophenotypes and functions may be changed depending on microenvironmental conditions (Takahashi, Naito, and Takeya 1996; Yamate et al. 2000). In this study, we used four different antibodies (ED1, ED2, OX6, and SRA-E5) to detect the appearance and distribution of macrophages.
ED1 antibody recognizes CD68, which is located on lysosomes, especially phagosomes of blood monocyte-derived infiltrating macrophages and resident macrophages. Expression implies enhanced phagocytosis (Damoiseaux et al. 1994; Suda et al. 1998). In the present study, the number of ED1-positive cells increased as early as PI day 2 and the level was maintained until PI day 9. ED1-positive cells are likely to have phagocytic activity for cell debris after ANIT-induced tissue injury. ED2 labels resident macrophages (Kupffer cells) in normal livers (Golbar et al. 2010). Recently, it has been reported that CD163, the antigen recognized by ED2, functions as a scavenger receptor for hemoglobin-haptoglobin complexes, that the presence of CD163 in lesions reflects the production of inflammatory mediators such as IL-6 and TNF-α (Polfliet et al. 2006). The ED2-positive cell number was transiently increased on PI day 3, which could be attributable to the production of such inflammatory factors during ANIT-induced lesion development.
The SRA-E5 antibody was generated by immunizing scavenger receptor knockout mice with recombinant human type I scavenger receptor protein (CD204) (Tomokiyo et al. 2002). CD204 expression is related to lipid metabolism in macrophages via the scavenger receptor (Greaves, Gough, and Gordon 1998). The SRA-E5-positive cell number showed a consistent increase on PI days 2 to 12. The consistent increase in numbers of SRA-E5-positive cells suggests that abnormal metabolism of lipid may be occurring in the affected liver due to ANIT injection.
Interestingly, compared with SRA-E5-positive cells, which were observed exclusively along sinusoids, the distribution of OX6-positive macrophages was limited to the affected Glisson’s sheath, suggesting differences in the properties of SRA-E5- and OX6-positive cells. The increased number of OX6-positive cells was consistently seen on PI days 2 to 12, in general agreement with the kinetics of SRA-E5- and ED1-positive cells (Fig. 4A, C, D). The MHC class II molecule recognized by OX6 is expressed in activated macrophages and dendritic cells (Zhang et al. 1993; Yamashiro et al. 1994; Ide et al. 2005; Zhao et al. 2006). Dendritic cells are major antigen-presenting cells, consisting of the interstitial dendritic cells, interdigitating follicular cells of the spleen and lymph nodes, and Langerhan’s cells of the epidermis (Gao, Mondal, and Lawrence 2007; Zimmerli and Hauser 2007). Interstitial dendritic cells are widely distributed in connective tissues, including the Glisson’s sheath (Takahashi, Naito, and Takeya 1996). Intriguingly, the present double immunofluorescence labeling demonstrated that 56% of the OX6-positive cells also reacted to ED1, suggesting that macrophages, which phagocytose cell debris, might also express MHC class II in ANIT-induced lesions. MHC class II expression is related to subsequent induction of CD4+ T cells (Perrigoue et al. 2009). Although the reason the OX6-positive cells showed a consistent increase remains to be investigated, the positive cells could potentially play vital roles in regeneration of injured cholangiocytes or development of biliary fibrosis in ANIT-induced rat liver lesions.
MCP-1 is a member of the C-C chemokine family and acts as a monocyte/macrophage chemotactic agent in injured tissues (Schecter et al. 1997; Marra et al. 1998; Sakai et al. 2006). In the present study, MCP-1 mRNA was increased significantly on PI days 1 and 2 in ANIT-injected rat livers, suggesting that a transient increase of MCP-1 at early stage might mediate macrophage infiltration into injured areas. Besides chemotaxis, MCP-1 has important roles in fibrogenesis (Gharaee-Kermani, Denholm, and Phan 1996; Tsuruta et al. 2004; Sakai et al. 2006). The role of MCP-1 in fibrogenesis is evident by the in vitro finding that human peripheral CD14-positive monocytes showed enhanced TGF-β1 mRNA expression when MCP-1 was added (Sakai et al. 2006). TGF-β1 is a well-known fibrogenic factor produced mainly by macrophages in injured livers (Zhang et al. 2004; Guo et al. 2006). It has also been reported that MCP-1-added to a rat macrophage cell line in vitro enhanced expression of CD68 (ED1) and CD163 (ED2) molecules (Mori et al. 2009). Therefore, the increased mRNA level of MCP-1 at early stage could have enhanced macrophage immunophenotypes and mediated subsequent biliary fibrosis, although increased TGF-β1 mRNA was seen only on day 9, which might be related to the late stage of biliary fibrosis.
Mesenchymal Cells in Glisson’s Sheath
Fibrogenesis is evoked by the development of myofibroblasts capable of producing collagens (Abdel-Aziz et al. 1991). The myofibroblasts are mesenchymal cells expressing vimentin; in addition to vimentin, they can express various cytoskeletal proteins (desmin and α-SMA) under fibrogenic factor stimuli (Yamate et al. 2005). After injury of cholangiocytes, vimentin- and desmin-positive cells were increased on days 1 to 3 in the Glisson’s sheath; thereafter, expression was gradually decreased, whereas α-SMA-positive cells appeared instead, showing the greatest expression on days 5 to 9. Although the double immunofluorescence labeling revealed that there were myofibroblasts coexpressing vimentin/desmin, desmin/α-SMA, or vimentin/α-SMA, it was shown that immunophenotypes of myofibroblasts were changed depending on stages of the lesions in the Glisson’s sheath; vimentin and desmin expression were enhanced at the early stage, whereas α-SMA expression was seen at the mid and late stages. Particularly, the appearance of α-SMA-expressing cells appeared to be related to the development of progressing biliary fibrosis. Myofibroblasts expressing α-SMA have been considered to be most active for collagen production (Wynn 2008). The derivation of myofibroblasts in biliary fibrosis has not been clarified. The vimentin-positive cells coexpressing desmin or α-SMA were located around the regenerating bile ducts at the early stage and, subsequently, α-SMA-positive cells expanded throughout the Glisson’s sheath at the late stage. The preexisting mesenchymal cells around the bile ducts might differentiate into myofibroblasts leading to the biliary fibrosis. After hepatocyte injury, HSCs can differentiate into myofibroblasts (Li and Friedman 1999; Friedman 2003). HSCs present in the vicinity of the Glisson’s sheath might represent another possible precursor of the myofibroblasts in biliary fibrosis.
In conclusion, the present study demonstrates, for the first time, that macrophages positive for ED1, ED2, SRA-E5, and OX6 appear in ANIT-induced hepatic lesions, showing different distribution and kinetics. Based on the immunophenotypes, these macrophages might have functions such as phagocytic activity, inflammatory factor production, lipid metabolism, and antigen-presentation. In addition, the participation of myofibroblasts variously expressing mesenchymal cytoskeletal markers was confirmed in the injured Glisson’s sheath; the cells expressed vimentin and desmin at the early stage, and α-SMA at the mid and late stages. Collectively, it is likely that interaction between macrophages and myofibroblasts is important in the progressive biliary fibrosis after cholangiocyte injury due to ANIT injection. The mechanisms of cholangiopathies are very complicated. ANIT-induced hepatic lesions and subsequent biliary fibrosis would be beneficial to investigate the molecular mechanisms. Additionally, the immunohistochemical analyses used in the present study might be useful when hepatotoxicant-induced hepatic lesions are evaluated pathologically.
