Abstract
A continuing education course entitled “What You Always Wanted to Know About Immunotoxicology in Pharmaceutical Development…But Were Afraid to Ask” was offered at the Society of Toxicologic Pathology (STP) 36th annual symposium in Montreal. This article summarizes some key points made during the presentation dedicated to immunophenotyping. It describes how clusters of differentiation (CDs) are well-defined antigens used to characterize cell subsets, and how lymphocyte subsets in humans and different rodent and nonrodent species can be defined by detection of various combinations of CDs. It provides an overview of immunophenotyping study design considerations and applications to safety assessment.
Keywords
Immunophenotyping: General Concept and Definitions
General Concept
Immunophenotyping is the process of identifying and characterizing cell subsets on the basis of different types of markers or antigens expressed on the cell surface, in the cytoplasm, or in the nucleus. This term refers to a technique or a test that utilizes antibodies, which specifically recognize different antigens. The antibodies are usually labeled with fluorochromes, isotopes, or enzymes for detection purposes. Rarely can a type of cell be identified by a unique protein strictly associated with this cell type. It is usually a combination of several markers that is required to phenotype cells. Commonly, a panel of antibodies can be used to simultaneously distinguish several types or subsets of cells in 1 assay. Immunophenotyping encompasses different approaches that fit different types of specimen and involve specific methodologies. Immune cells can be analyzed as cell suspensions from peripheral blood or from lymphoid organs and examined by flow cytometry. Tissue resident cells can be stained in situ and analyzed by either immunohistochemistry (IHC) or immunofluorescence (IF).
Clusters of Differentiation (CDs)
Immunophenotyping heavily relies on a library of antigens and their expression patterns. Most of the cell surface molecules are identified by CD numbers. The CD nomenclature was established by the International Workshop and Conference on Human Leukocyte Differentiation Antigens (HLDAs) run by the Human Cell Differentiation Molecules (HCDM) organization (ww.hcdm.org). It has been universally adopted by the scientific community and is officially approved and sanctioned by the World Health Organization (Bernard and Boumsell 1984). The purpose of the CD nomenclature is to provide consistency and uniformity when referring to identical molecules and the antibodies binding to the same antigen (Engel et al. 2015). Each antigen is matched to a unique CD number. As of 2017, CD markers range from CD1 to CD371. The CD nomenclature consists of “CD” followed by a number. There are some exceptions: a lower case “w” preceding the number designation (e.g., CDw149) stands for “workshop” and indicates a tentative designation. Uppercase letters following the CD number imply spliced variants of the extracellular domains of the cell surface proteins (e.g., CD45RA). A lowercase letter following the number designates a group of proteins with a certain association. For example, CD11a, CD11b, and CD11c share a common chain. CD66a-f all belong to the same gene family CD66. CD60a-c are the CD60 molecules with different modification of the same carbohydrate sequence. As a result, CD1 to CD371 covers over 400 molecules. Immunophenotyping uses the CD nomenclature to phenotype leukocyte subsets based on anti-CD antibody binding features. Cells are defined by the presence “+” or absence “−” of the antigen or the binding of the antibody. The intensity of the antibody staining is also depicted as high/mid/low or bright/mid/dim to characterize the intensity of staining that represents the expression levels of the antigens. Of note, while more than 400 antigens have been identified with a unique CD nomenclature, only a small group of them are routinely used for immunophenotyping in the context of toxicology studies.
As stated above, the CD nomenclature was established and is managed by HCDM for molecules expressed on human cells. There is no organization overseeing animal CDs. However, for nonclinical research and immunopharmacology and immunotoxicology studies, several animal species including rodents, nonhuman primates, dogs, pigs, and guinea pigs are routinely used. Researchers have studied panels of antibodies from HLDA workshops for cross-reactivity with such animal species. If a molecule is highly conserved across species and shares high homology with its human orthologue, there is a better chance that cross-reactive antibodies will be identified. Because cross-reactive antibodies are not always available, it is not uncommon to raise antibodies against preclinical species immune markers for immunophenotyping purposes. Nonhuman primates are often used for pharmacological and toxicological assessments of protein therapeutics. A list of antibodies recognizing nonhuman primate leukocytes were tested and published as public resources, for example, www.nhpreagents.org. Many vendors also test their antibody products and provide species cross-reactivity information. For rodents, a wide spectrum of antibodies is available for both mice and rats (Ward et al. 2006). Rodents are widely used as research models, and most of the antibodies were raised specifically against rodent antigens. Small-molecule therapeutic candidates are often evaluated in dog toxicology studies. Many reagents have been established to conduct immunophenotyping in that species (Lebrec et al. 2012). The pig is also now a species of choice as a preclinical model (Bode et al. 2010). Panels of antihuman CD antibodies were tested for reacting with the pig orthologues as were panels of antihuman CD antibodies tested for reacting with Guinea pig orthologues (Haverson et al. 2001; Lasco et al. 2007). In spite of the efforts that different groups devoted to characterize CD molecules on nonhuman species, there is still a shortage of reagents to detect cell surface proteins matching that of human and rodents.
Antibody cross-reactivity is important to define CD proteins in nonclinical species. However, the study of the cellular distribution of the antigens is also critical to interpret human relevance of immunophenotyping data from nonclinical species. Antigens have been categorized into 3 groups based on corresponding antibody binding characteristics and cell distribution information. First, there are CDs for which both antibody reactivity and cellular distribution are consistent with human antigens, for example, CD21, or complement receptor 2, a protein expressed on both human and dog B cells (Byrne et al. 2000; Platt et al. 2013). Secondly, there are CDs for which an antibody binds to the same antigen but expression in various cell subsets is inconsistent between species. For instance, CD4 is highly expressed on canine neutrophils but not on mouse neutrophils (Moore et al. 1992). For a third group of CDs, the antibody staining appears extremely dim or negative on nonhuman leukocytes in comparison to human leukocytes. These considerations need to be taken into account when making interspecies comparisons and for markers that don’t entirely agree between species, specific characterization of the immunophenotype for the species of interest needs to be done.
Method
There are 2 major methods for immunophenotyping studies. Flow cytometry has become the method of choice for leukocytes that are in suspension in whole blood, in isolated peripheral blood mononuclear cells (PBMCs), or from dissociated lymphoid tissues. For tissue resident cells, IHC and IF are commonly used.
Flow Cytometry
Flow cytometry is the most powerful technique of quantitative single-cell analysis for cell size, granularity, and extracellular or intracellular fluorescent dye-labeled antibodies (Maecker, McCoy, and Nussenblatt 2012). Flow cytometers are the essential instruments for this analysis. Nowadays, the state-of-the-art flow cytometers are capable of analyzing up to 16 parameters concomitantly. This number keeps increasing with the discovery of new dyes. The single-cell suspension labeled with multiple fluorescent tagged antibodies is aspirated into a flow cell and passes through a focused laser beam 1 cell at a time. The fluorochrome of each labeled antibody bound onto the cell is excited by the laser light and emits a light of a different wavelength. The cell also scatters light depending on the size and internal structures. Lights with desired wavelength pass through optical filters and are captured by photodetectors. The resulting electrical pulses are translated into digital data for final analysis (Adan et al., 2017).
Flow cytometric data can be presented with various plot formats including dot plot, pseudo-color dot plot, contour plot, density plot, zebra plot, histogram, and cumulative distribution function plot. Each plot presents different visual representation of the data. In a pseudo-color dot plot, each dot represents a single cell (Figure 1). Data analysis requires selection of certain cell populations with gating strategies. For instance, lymphocytes form a cluster based on forward scatter and side scatter outputs because they have similar size and complexity/granularity. When gating on lymphocytes, each fluorochrome-labeled antibody can further separate cells into distinct subsets, for example, CD3+ T cells and CD20+ B cells. Each subset can be further defined through subsequent gating approaches. CD3+ T cells are categorized into CD4+ T cells and CD8+ T cells based on the staining profile of CD4- and CD8-specific antibodies. Sometimes, the antibody presents a “smear” type of staining, and the separation of cell subsets is more difficult and likely relies on additional staining data. The nature of the spread of the staining profile of a population indicates how the antigen expression is heterogeneous within a cell subset.
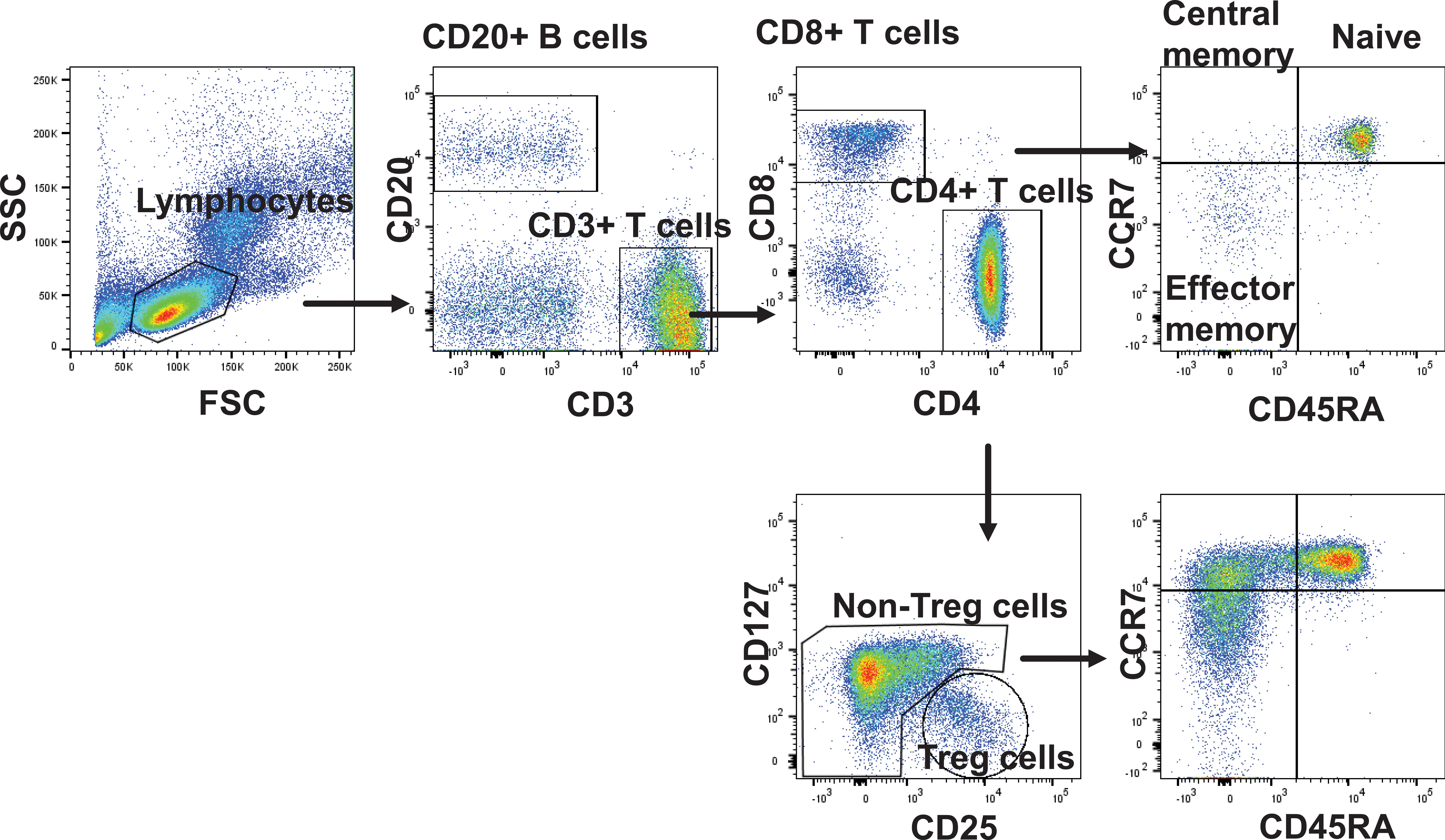
T-cell immunophenotyping of human peripheral blood mononuclear cell by flow cytometry. Flow cytometry data are presented with pseudo-color dot plots. Lymphocytes are first gated with forward scatter versus side scatter. T cells and B cells are gated based on the antibody staining against CD3 and CD20, respectively. From CD3+ T cells, CD8+ and CD4+ T cells are further separated. For CD8+ T cells, naive, central memory, and effector memory cells are divided based on different combination of CD45RA and CCR7 staining. For CD4+ T cells, regulatory T (Treg) cells are defined as CD25+ CD127 low. Naive, central memory, and effector memory cells are subgrouped from non-Treg cells. CD = clusters of differentiation.
The heterogeneity of cells is also reflected by the variety of antigens expressed by cells. Mass cytometer (also known as cytometry by time-of-flight) is a next-generation flow cytometry in which antibodies are labeled with stable isotopes of heavy metal ion instead of fluorochromes. Mass cytometry requires minimal signal compensation between different metal tags and enables measurement of multiplex parameters (up to 40) on single cells. This technique is primarily restricted to basic research due to its low throughput and high-content nature (Bendall et al. 2012).
In the context of clinical immunology studies, the Human Immune Phenotyping Consortium (HIPC) was formed by the Federation of Clinical Immunology Societies to promote standardization of flow cytometry immunophenotyping analysis. To achieve this, the HIPC immunophenotyping panel was developed to phenotype major immune cell subsets in PBMCs (Maecker, McCoy, and Nussenblatt 2012). The use of lyophilized reagent cocktails and automated analysis was tested for standardization (Finak et al. 2016). Although immunopharmacology and immunotoxicology phenotyping studies remain highly nonstandardized, there are good practices that can be recommended to ensure data quality and reproducibility including reagent selection, sample preparation, assay protocol, instrument setup, and data analysis. The design of staining panels and the selection of antibodies are essential to satisfy specific aims of each study. Fluorochromes that emit lights with largely overlapping wavelength should be avoided in the panel. A proper titration of each antibody should be done in advance to decide the optimal concentration for staining. Sample handling, cell preparation, and staining procedure should follow a standard protocol to reduce cell death and the change of antigen expression (Maecker, McCoy, and Nussenblatt 2012). For example, T-cell activation status may change with prolonged incubation at or above room temperature. Instrument setting and data analysis by different operator may result in inconsistent results. Automated operation and gating may be solutions to promote standardization especially for large data sets.
IHC and IF
IHC and IF can be applied to in situ imaging antigens on cells in a pretreated tissue section. Both techniques use the principle of antibody- and antigen-specific recognition but differ by the detection method (Figure 2). IHC utilizes antibodies conjugated to an enzyme, such as a peroxidase, which can catalyze a chemical reaction resulting in color change. Alternatively, IF visualizes antigens with fluorochrome-linked antibodies. Both IHC and IF can directly detect antigens with labeled primary antibodies or indirectly with the help of labeled secondary antibodies that recognize the Fc portion of the primary antibody.
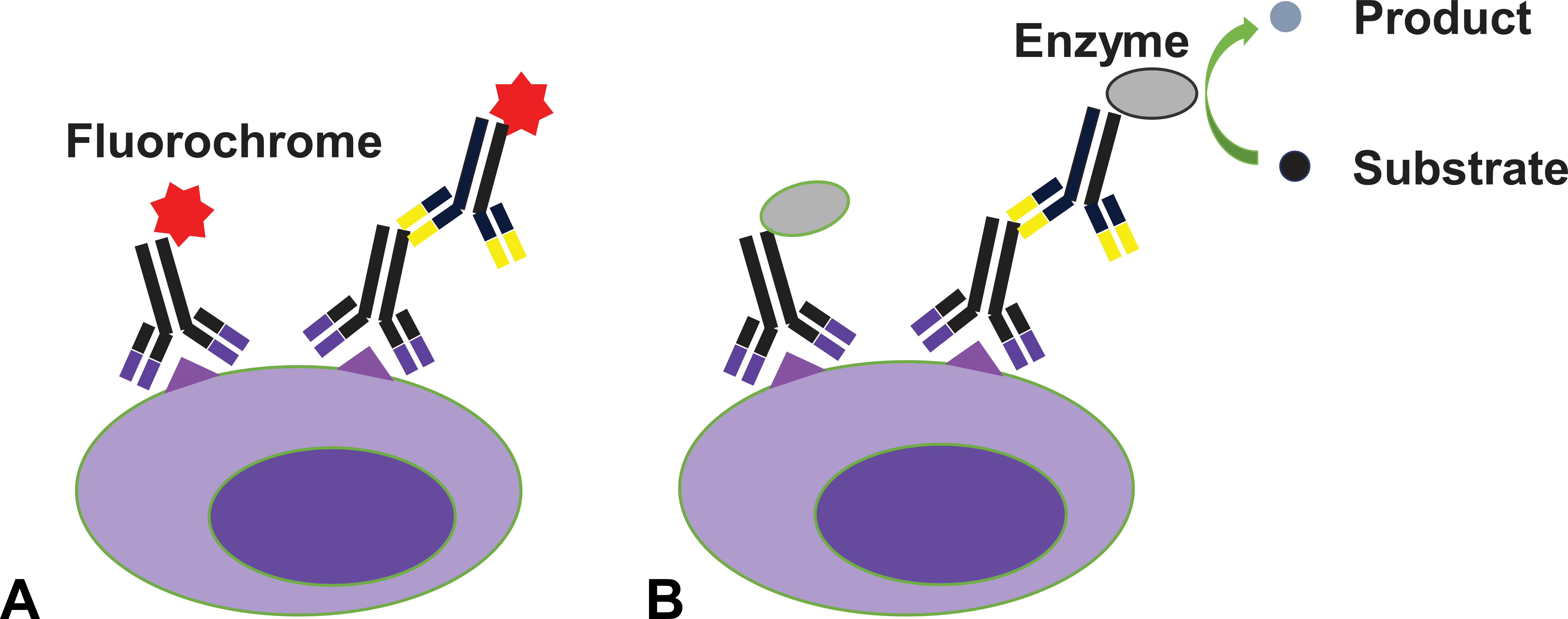
Illustration of imaging antigens in immunohistochemistry (IHC) and immunofluorescence (IF). (A) IF uses a fluorochrome-labeled primary antibody to directly bind cellular antigen or uses a fluorochrome-labeled secondary antibody to detect antigen-bound nonlabeled primary antibody. The antigen is imaged with fluorescent color. (B) For IHC, enzyme-linked primary or secondary antibodies are used to image cellular antigen, then the enzymatic reaction will transform a substrate into colored products to complete antigen imaging.
IHC and IF require fixation to preserve tissue morphology and retain the antigenicity of the target molecules. Microtome sectioning is performed on formalin-fixed paraffin-embedded (FFPE) tissues, and cryostat sectioning is performed on optimal cutting temperature compound embedded frozen tissues followed by fixation before staining. An antigen retrieval step is often added to reverse changes produced during fixation for FFPE tissues. IHC/IF assays are not standardized assays and require careful examination and interpretation by qualified personnel (Lin and Chen 2014).
Immunophenotyping in Various Species
Basic immunophenotyping of peripheral blood cells or isolated PBMCs generally consists of enumerating T cells (including T helper and cytotoxic T lymphocytes), B cells, and natural killer (NK) cells. Specific antigens of choice are used for the identification of T, B, and NK cells in humans and in various species frequently used in toxicology studies have been identified (Baker et al. 2008; Morris and Komocsar 1997; Gibson et al. 2004; Rubic-Schneider et al. 2016; Maecker, McCoy, and Nussenblatt 2012) and are listed in Table 1. T cells specifically express CD3 and comprise CD4+ T cells and CD8+ T cells in human, nonhuman primate, rat, dog, and mini pig. It should be emphasized that in the mini pig CD8 can be expressed at relatively low levels on CD4+ T cells. CD3 is a coreceptor for T-cell receptors (TCRs) and is critical for TCR-mediated T-cell activation. Both CD8 and CD4 are coreceptors to interact with major histocompatibility complex (MHC) class I and class II molecules, respectively. CD20, a signature B cell differentiation antigen with unknown function, is a marker for B cells in human and nonhuman primates, while CD45RA, an isoform of tyrosine phosphatase CD45, is used to define rat B cells. CD21 (complement receptor type 2) and surface immunoglobulins (Sallusto et al. 1999) are used to label dog and mini pig B lymphocytes respectively. In human, NK cells can be subdivided into different populations based on the expression of CD56 (neural cell adhesion molecule) and CD16 (Fc γ receptor). CD56+ NK cells constitute the majority of NK cells in peripheral blood and secondary lymphoid organs (Poli et al. 2009). In nonhuman primates, CD56 is also expressed on monocytes and as a consequence CD16 is a more appropriate marker for NK cells (Carter et al. 1999). CD159a (NKG2A, an inhibitory receptor) is also an NK cell marker that can be used in nonhuman primates (Mavilio et al. 2005). In rodents, CD161a (NK1.1) is expressed on both immature and mature NK cells and serves as a reliable marker for strains expressing the isoform (Huntington, Vosshenrich, and Di Santo 2007). Because most of the NK cell markers can be expressed on CD8+ T cells, a gating strategy selecting CD3 negative lymphocytes is important to distinguish NK cells from T cells.
Standard Immunophenotyping Panels of Human and Nonclinical Species.
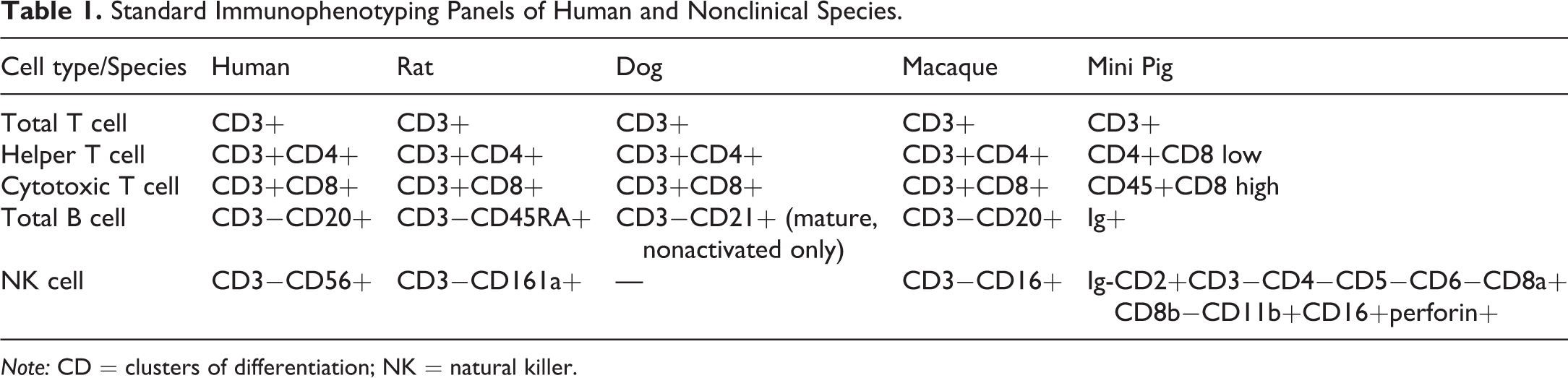
Note: CD = clusters of differentiation; NK = natural killer.
Across animal species and as described in humans, lymphocytes can be further divided into subsets on the basis of functionality, differentiation, or activation status using distinct combinations of surface markers. This is illustrated in Table 2 for T lymphocytes. Markers defining such subsets for T lymphocytes largely overlap across animal species with some exceptions (Table 2).
T-cell Immunophenotyping Panels of Human and Nonclinical Species.
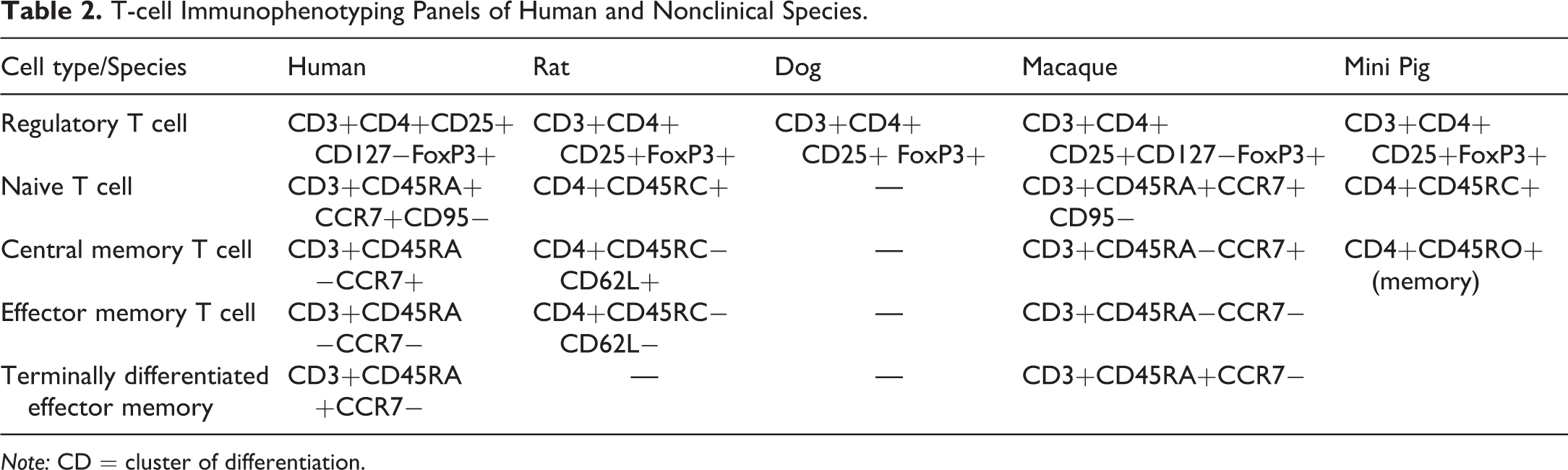
Note: CD = cluster of differentiation.
Regulatory T cells (Treg) are commonly identified by surface expression of CD25 (interleukin [IL]-2R α) and intracellular expression of the Foxp3 transcription factor in human and nonclinical species. Treg is highly dependent on cytokine IL-2 for development, maintenance, and function and Foxp3 for function (Plitas and Rudensky 2016). In addition, Treg cells express low level of CD127 (IL-7R α) and are defined as CD3+/CD4+/CD25+/CD127low/Foxp3+ in human and cynomolgus macaques (Liu et al. 2006; Seddiki et al. 2006; Clark, Narayanan, and Fort 2012). CD4+ and CD8+ T cells are separated into naive and various memory subsets with additional cellular markers. In humans and nonhuman primates, the expression of CD95 differentiates memory T cells from a naive population, and the differential expression of CD45RA and chemokine receptor CCR7 further defines central memory, effector memory, and terminally differentiated effector memory cells (Farber, Yudanin, and Restifo 2014; Romero et al. 2007; Lugli et al. 2013; Sallusto et al. 1999). CD95 is involved in activation-induced apoptosis, and activated T cells are susceptible to CD95/CD95L-mediated apoptosis (Brunner et al. 1995). CCR7 is the lymph node-homing C-C motif chemokine receptor 7. Naive T cells uniformly express CCR7, which reflects their predominant location in lymphoid tissue, whereas CD45RA− CCR7+ central memory T cells are primarily residents of lymphoid tissues, and CD45RA−CCR7− effector memory cells travel and reside in multiple peripheral tissues. In the rat and the mini pig, the identification of naive versus memory T cells rely on other CD45 isoforms CD45RC and CD45RO. CD62L (L-selectin) is commonly used in rodents for identifying T memory cell subsets. CD62L acts as a homing receptor for lymphocytes to enter secondary lymphoid tissues via high endothelial venules. The exact function of isoforms of CD45 (CD45RA, CD45RC, and CD45RO) is not well-defined. The memory T-cell subsets are best characterized in human and mouse and to a lesser extent in nonhuman primates. The lack of cross-reactive antibodies makes it difficult to define them in other species.
Immunophenotyping in Nonhuman Primates—Study Design Considerations
In preclinical study of large molecules, the nonhuman primate may often be the only relevant species to assess the nonclinical safety and pharmacology end points, given the poor cross-reactivity of drug candidates in rodents. Flow cytometry immunophenotyping is frequently included as an adjunct procedure to standard toxicity testing and is designed as a minimally invasive test from peripheral blood. In order to proper design the immunophenotyping end points in immunotoxicological studies, several factors should be considered.
Gender and Age
In human, peripheral blood leukocyte subset counts may differ between genders and change with aging (Rudy et al. 2002; Yan et al. 2010). Females have higher total CD4+ T cells and CD4+ memory T cells counts and lower CD16+ NK cell counts than males. Both CD4+ memory T cell counts and CD8+ T memory cell counts are increased with age. In addition, greater heterogeneity in different immune cell subsets was displayed in older healthy human populations than younger individuals (Kaczorowski et al. 2017). Gender- and age-associated change of lymphocyte subsets was also observed in cynomolgus monkeys. A retrospective interlaboratory comparison of flow cytometry immunophenotyping end points in nonhuman primates was conducted to identify factors associated with average lymphocyte counts and the within-animal cell count variability in naive cynomolgus monkeys (Krejsa et al. 2013). In this study, flow cytometry data from a total of 17,277 samples were collected for counts of total T cells, CD4+ T cells, CD8+ T cells, B cells, NK cells, and monocytes from peripheral blood. For the analysis of each cell type, 936 to 3,000 samples were available. A small but statistically significant difference was found in the average cell counts of total T cells, CD4+ T cells, CD8+ T cells, B cells, and NK cells, with higher mean counts in males than in females for all cell types except NK cells. Within-animal variability analysis revealed that B cells were associated with greater variability in males than in females. Typically, animals of both genders are recruited into nonclinical studies. Male and female immunophenotype data are analyzed separately similar to the analysis of standard hematology data.
The association of age and lymphocyte counts is debatable. Trends in mean counts among age-groups were not statistically significant in the abovementioned study (Krejsa et al. 2013). In another study with broader range of age, CD4 CD8 double-positive T cells were increased, and memory cells in both CD4+ and CD8+ T cell groups were elevated with age (Lee et al. 2003). Both thymic maturation and increased viral exposure could explain the age-related differences in immune cell count. Normally, animals included into studies are restricted within an age range, and the abovementioned items are unlikely to be impactful.
Commercial Origin of Animals
Statistically significant differences in average cell count for total T cells, CD8+ T cells, CD4+ T cells, and B cells were observed between cynomolgus monkeys of different commercial origin identified as corresponding to “island monkeys” (Mauritius, Indonesia) and Mainland monkeys (China) or directly referred to countries (Caldwell, Marshall, and Fishel 2016; Krejsa et al. 2013). For example, male monkeys from Mainland China exhibited higher cell counts with 1,960 to 4,270 T cells per microliter of blood, while male monkeys from Mauritius had total T-cell counts varying from 1,060 to 3,240 cells per microliter of blood. NK cell and monocyte counts were trending toward higher numbers in animals from Mauritius without reaching statistically meaningful significance. Similar patterns were observed with females from different origins. Cynomolgus monkeys from Mauritius have previously been reported to have higher red blood cell count and lower leukocyte count than cynomolgus of Asian origin (Choi et al. 2016; Drevon-Gaillot et al. 2006). Among island monkeys, there was no significant difference of any immune cell counts between Mauritian and Indonesian monkeys. Of note, origin doesn’t impact interanimal variability (Krejsa et al. 2013).
Power Analysis
The power to detect a relative change in cell counts of each peripheral blood immune subsets is a function of inter- and intra-animal variability. For each cell type, calculations can be performed to determine group sizes required in order to detect with confidence a certain effect (e.g., ∼50% change). It was, for instance, estimated that in order to detect with statistical confidence a 50% change in total CD3+ T cell count in adult cynomolgus monkeys by comparing pretreatment with posttreatment counts within a single given group of animals, 6 to 8 animals were necessary. A greater number of animals would be necessary if similar comparisons were conducted between different groups of animals (Krejsa et al. 2013).
Interpretation and Reporting
When reporting immunophenotyping data in tabular and/or graphical representations, individual animal and group mean along with SD values are useful. Absolute counts as number of cells per microliter of blood should be reported whenever possible. The relative percentage of lymphocyte subsets can be reported only when absolute counts cannot be determined (e.g., from analysis of lymph node or spleen samples). If blood samples are taken from individual animals prior to and post of treatment over a specific time period, the percentage of change over the baseline value should preferably be determined using the second (closest to start of study) of 2 predose samples. The first predose sample may be used to understand intra-animal longitudinal variability and contribute to overall interpretation. The mean fluorescence intensity of a relevant cell surface marker may also be reported as intensity of expression of cell surface markers and are often indicators for activation status of immune cells. Statistical analysis (Finak et al. 2016) may contribute to interpretation of immunophenotyping data.
Applications of Immunophenotyping to Safety Assessment
Immunophenotyping has been recognized for many years as an important component of immunotoxicity testing (Luster et al. 1992. Subsequently, this methodology has been cited by various regulatory guidance documents as an important tool that should complement standard toxicology study end points (e.g., hematology data, anatomic pathology data) and immune function tests such as the T-cell-dependent antibody response on a case-by-case basis (ICH 2005). In drug development, immunophenotyping analysis is often a standard end point during the preclinical evaluation of therapeutic drug candidates that could potentially affect the immune system.
It may be used during early target liability assessment to understand target distribution. The target expression may be altered under disease conditions, and expression may therefore be examined in both normal human tissue/cells and diseased human tissue/cells. Target expression and distribution should also be investigated in toxicology species in order to identify pharmacologically relevant species. A number of pharmacodynamics (PDs) end points rely on immunophenotyping assays, in particular for immunomodulatory therapeutics. Leukocyte depletion, activation, change of downstream signaling pathways and receptor occupancy (RO) are all common PD biomarkers (Liang et al. 2016). The comparison of target expression profile and PD biomarkers between animals and human is often critical to support the selection of pharmacologically relevant species for toxicology assessment and also to correctly understand the relevance of nonclinical toxicity findings to humans. Anatomically and functionally, all vertebrates share highly similar immune systems, including innate and adaptive immunity components (Holsapple, West, and Landreth 2003). However, there are subtle differences in cellular receptors and surface markers distribution, kinetics, and intensity of expression across species. These differences can result in dissimilar responses to therapeutics and misinterpretation or poor extrapolation from nonclinical findings to humans. A well-known example is the severe adverse events observed with anti-CD28 superagonist monoclonal antibody Theralizumab (TGN-1412) in a human trial in the United Kingdom (Suntharalingam et al. 2006). The disconnect between the severe toxicity in healthy volunteers and findings from nonclinical animal studies was partly due to species difference in CD28 expression on CD4 effector memory T cells (Eastwood et al. 2010). This instance emphasized the importance of comparing immunophenotypes of relevant subsets in animals and humans. Immunophenotyping assays can be used to monitor translational biomarkers of immunotoxicity. For instance, in the preclinical setting, NK cell counts have been found to be good indicators of IL-15 blocking antibody activity, as the survival of NK cells is highly dependent on IL-15 in animals (Kennedy et al. 2000). Administering cynomolgus monkeys with an anti-IL-15 antibody caused a profound reduction in circulating NK cell counts, an effect that did not translate to humans likely as the consequence of species-specific NK homeostasis requirements (Lebrec et al. 2013).
Conclusions
Immunophenotyping is a widely used methodology in both preclinical and clinical settings, and the technology associated with it is rapidly evolving toward greater automatization and increased content. Risk assessment requires a good understanding of the translational aspect of findings in preclinical species, and investigators should leverage this methodology with a good understanding of reagent specificity and cross-reactivity as well as relevance of CDs of interest across species.
Footnotes
Author Contribution
All authors (XW, HL) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
