Abstract
Regulatory T cells (Tregs) are a rare subset of lymphocytes that inhibit the activation and effector functions of T cells and are important regulators of immune responses. Although Tregs are well characterized in humans and rodents, little is known about their immunophenotyping (IP) profile in cynomolgus macaques (Macaca fascicularis), which is an important species for pharmacological and toxicological evaluation of potential immune modulators because of their similar physiologic, genetic, and metabolic response patterns to humans. The authors have developed an immunophenotyping panel using a high-throughput 96-well microtiter plate–based assay to detect circulating Tregs (CD3+CD4+CD25hiFoxP3+) and have determined the normal range for the number of Tregs in naive healthy cynomolgus macaques to be 56.4 to 179.7 cells/µL (mean ± SEM = 113.6 ± 5.1 cells/µL; n = 25). Furthermore, the authors compared the resulting FoxP3+ Treg profiles with a CD127lo cell-surface panel (CD3+CD4+CD25hi CD127lo) and found a close correlation between the absolute numbers of CD3+CD4+CD25hiFoxP3+ and CD3+CD4+CD25hiCD127lo cells (mean ± SD = 120 ± 8.0 cells/µL). Quantification of circulating Tregs in cynomolgus macaques in this high-throughput assay may help to identify drug candidates that affect this rare, but critical, immunoregulatory cell population.
The impact on immune status and function in preclinical studies in animals is an important component of safety assessment of immunomodulatory agents. Although the benefits of immunomodulatory therapeutics have dramatically improved the lives of many patients, unexpected side effects have occurred resulting in adverse responses including increased risk of infection due to immunosuppression and cytokine release syndrome (Hansel et al. 2010; Piccotti et al. 2009). These toxicities underscore the necessity to develop assays that detect the immunotoxic potential of pharmaceuticals in a species predictive of human responses (National Research Council 1992). Nonhuman primates (NHP), such as rhesus and cynomolgus macaques, have been used for preclinical disease modeling and drug safety assessment because of genetic homology and physiologic and metabolic similarities to humans. Rhesus macaques have been the NHP of choice for allograft and tumor rejection studies (Haanstra et al. 2007), and cynomolgus macaques have historically been the preferred NHP for toxicological assessment, including immunotoxicity studies (Green and Black 2000).
Regulatory T cells (Tregs) are a rare subset of lymphocytes that inhibit the activation and effector functions of T cells and are key players in transplantation biology (Porter et al. 2007), allergies (Trzonkowski et al. 2009), and autoimmune disorders (Feuerer et al. 2007; Sakaguchi 2004; Sakaguchi et al. 1995). Increasingly, Tregs are being investigated as possible therapeutic targets because of their critical mechanism of maintaining self-tolerance and regulating the immune response (Hori, Takahashi, et al. 2003; Jung and Seoh 2009; Sakaguchi 2004). Therefore, screening for potential changes in circulating Treg numbers is crucial during the profiling of prospective therapeutic agents, which may influence the number or function of this immunosuppressive lymphocyte subset. Tregs are identified by intracellular expression of the forkhead box P3 transcription factor (FoxP3). FoxP3 expression is directly related to the immunosuppressive function of these cells (Baumgarth and Roederer 2000; Fox et al. 2008; Hori and Sakaguchi 2004; Pillai and Karandikar 2008; Rudensky et al. 2006). Furthermore, it has been shown that human Tregs express lower levels of the IL-7 receptor alpha chain (CD127) (Liu et al. 2006; Seddiki et al. 2006) and that the expression of CD127 inversely correlates with high expression of the IL-2 receptor (CD25). CD127 expression is a useful marker for Treg immunophenotyping (IP) panel development because of its expression on the cell surface, and as such, it requires fewer experimental manipulations compared with intracellular FoxP3 staining. Tregs have been previously enumerated in rhesus and cynomolgus macaques and baboons, but differing assay methods have resulted in variable staining results and ranges for Tregs in these species (Law et al. 2009; Porter et al. 2007). To our knowledge, there is a lack of information on the range of absolute numbers of circulating Tregs in healthy, naive cynomolgus macaques, as has been previously performed for other lymphocyte subsets (Baker et al. 2008; Bleavins et al. 1993).
Therefore, we developed a standardized, high-throughput IP panel that identifies CD3+CD4+CD25+FoxP3+ Tregs and used this panel to establish a reference range of Treg cell counts in naive cynomolgus macaques. Next, we compared CD127 surface expression on circulating cynomolgus CD4+CD25+ T cells to that of FoxP3 expression from the same blood sample, which was divided into two portions for differential analysis of these markers. Our data show that either FoxP3 or CD127 expression may be used to identify circulating Tregs in cynomolgus macaques.
Materials and Methods
Antibodies and FoxP3 Kits
We compared two commercially available FoxP3 buffer kits from eBioscience (cat No. 00-5523) and BD Biosciences (cat No. 560133, Franklin Lakes, NJ). The FoxP3 IP panel consisted of anti–nonhuman primate CD3 PerCP (clone SP34-2, BD Biosciences, catalog No. 552851), antihuman CD4 FITC (clone L200, BD Biosciences, catalog No. 550628), and antihuman CD25 APC (clone 4E3, Miltenyi Biotec, catalog No. 130-0925), and antihuman FoxP3 PE (clone 206D, BioLegend, catalog No. 320108). In the CD127 IP panel, FoxP3 was replaced with antihuman CD127 PE (clone HIL-7R-M21, BD Biosciences, catalog No. 557938).
Blood Sources
Whole blood was collected in EDTA from five experimentally naive male cynomolgus macaques, ranging in age from 4 to 9.5 years (colony source A: Washington National Primate Research Center, Seattle, WA) or 20 naive female cynomolgus macaques ranging in age from 3 to 5 years (colony source B: Shin Nippon Biomedical Laboratories USA Study Center, Everett, WA). Colony A origin samples were each collected over four time points, drawn 2 weeks apart and used to estimate our assay variability and determine the FoxP3+ Treg cell population fluctuation over time. Blood from animals of colony B origin was sampled at a single time point for split analysis of the FoxP3 IP panel versus CD127 IP panel. All animals in both colonies were purpose bred. The geographic origin of animals in colony A was varied. Animals in colony B originated from Cambodian-derived stock. Animals at both colonies were maintained in compliance with approved standard operating protocols for animal care and experimental procedures and in accordance with the Guide for the Care and Use of Laboratory Animals (Institute of Laboratory Animal Research et al, 1996) and in accordance with all federal policies and guidelines governing the use of vertebrate animals, including approval by the Institutional Animal Care and Use Committees.
FoxP3 IP Panel Optimization for Detection of Circulating Tregs in Cynomolgus Macaques
Starting with 100 µL of whole blood, we collected at least 400,000 live events with a minimum of 30,000 events in the lymphocyte gate and 10,000 events in the CD3+CD4+gate. By collecting at least 10,000 events in the CD3+CD4+ gate, we obtained ≥500 events in the FoxP3+CD25+ gate or the CD127loCD25+ gate (see Fig. 1 for gating schemes).
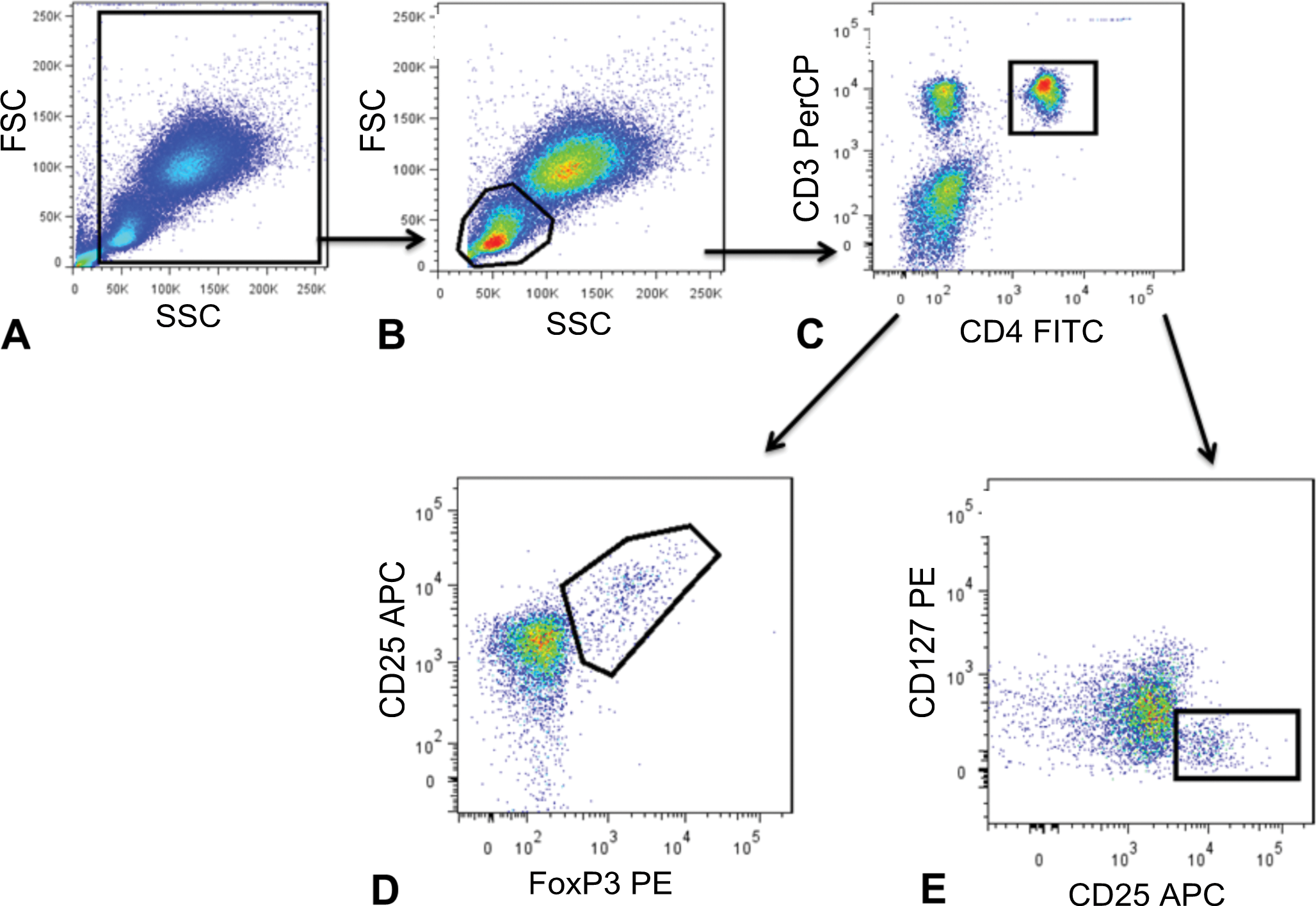
Gating scheme for divided sample analysis using either the FoxP3 IP or CD127 panel. Tregs were identified as CD25+ FoxP3+ subset of CD3+ CD4+ T cells from cynomolgus macaque whole blood. A hierarchical gating strategy was used: (A) forward versus side scatter gate to eliminate debris, (B) lymphocyte gate based on forward versus side scatter, (C) CD4+ CD3+ gate, and (D) CD25+ FoxP3+ gate. Gating scheme for CD127lo IP panel: A hierarchical gating strategy was used as above. Tregs were identified as CD127lo CD25+ subset of CD3+ CD4+ T cells as indicated in (E) CD25+ CD127lo gate. At least 10,000 events in the CD4+CD3+ gate were acquired for each panel.
Staining Protocols
IP was performed by adding 100 µL whole blood/well to a 96-well round-bottom plate (Falcon cat No. 353077). Samples were incubated with 100 µL of antibodies directed at cell-surface proteins at the appropriate dilution for 30 min, on ice, protected from light. The plate was then centrifuged at 250 × g for 5 min, and supernatants were removed. Red blood cells were lysed by adding 180 µL 1X Pharm Lyse (BD Biosciences) per well for 10 min. Plates were then centrifuged as described above, supernatants were removed, and cells were washed twice with 180 µL/well wash buffer (1× phosphate-buffered saline, 2% fetal bovine serum, 0.1% sodium azide, 22 µm filtered). For intracellular staining of FoxP3 expression, cells were resuspended in 180 µL cold fixative buffer per well and kept overnight at 4°C in the dark. Plates were centrifuged at 280 × g for 3 min, and fixative was removed from the well. Cells were washed twice with 180 µL/well of permeabilization buffer (PB) at room temperature (RT). Twenty microliters of PB and 10 µL anti-FoxP3 antibody were added to cell pellets and incubated for 60 min at RT, protected from light. Cells were washed once with 180 µL PB, centrifuged at 250 × g for 5 min, and resuspended in 180 µL wash buffer. Plates were analyzed on an LSRII cytometer (BD Biosciences), and a hierarchical gating scheme was used (Fig. 1) to determine cellular subsets. Lymphocytes were gated by side scatter channel/forward scatter channel characteristics, and CD3+CD4+ cells were gated within this population. The FoxP3 and CD127 gates were set using FMO control wells. The data were analyzed with FlowJo software version 7.6.1 (Treestar, Ashland, OR). The absolute numbers of FoxP3+ CD25hi CD4+ T cells and CD127lo CD25hi CD4+ T cells were determined by multiplying the percentage of these cells in the lymphocyte gate by the number of circulating lymphocytes per microliter blood, as determined by an Advia 120 Hematology Analyzer (Siemens, Deerfield, IL).
Statistical Methods
The statistical significance of mean cell counts, determined by either method of assay (within split samples from the same macaque or from different groups of macaques) was done by paired or unpaired t tests, respectively. Intraassay and intraindividual variation were estimated using the coefficient of variation, pooling results within and between experiments accordingly. All p values lower than .05 were considered significant. GraphPad Prism version 5.01 (GraphPad Software, La Jolla, CA) was used for all analyses.
Results
Development of FoxP3 IP Panel
We compared intracellular FoxP3 staining results using two different fixative/permeabilization buffer kits (eBiosciences, San Diego, CA, and BD Biosciences, San Jose, CA, respectively) and found eBiosciences provided clear staining with our panel of antibodies (data not shown). Use of the eBiosciences FoxP3 buffer kit with antihuman FoxP3 clone 206D labeled with phycoerythrin (PE) allowed for the detection of a distinctive population of CD25hi FoxP3+ CD4+ T cells in whole blood (WB; Fig. 1). Furthermore, we were able to collect adequate numbers of FoxP3+ CD4+ T cells from 100 µL of WB using a 96-well microtiter plate–based staining method (see the Materials and Methods section). Using this small volume of WB, we were consistently able to collect 10,000 CD3+ CD4+ events and at least 500 CD3+CD4+ CD25hi FoxP3+ events.
Qualification of FoxP3 IP Panel for Quantification of Circulating Tregs in Cynomolgus Macaques
Flow cytometric analysis of blood samples from five male cynomolgus macaques (colony A) at four different time points reliably showed that 6.53 ± 1.14% of CD4+ T cells and 2.03 ± 0.54% of total lymphocytes were FoxP3+ Tregs. Furthermore, the circulating numbers of FoxP3+ Tregs in these animals ranged from 56.4 to 175.5 cells per microliter of blood (mean ± SD =110.4 ± 32.8 cells/µL). The intraassay coefficient of variation was 2.8% for triplicate samples for each animal (Fig. 2 , and data not shown). The intraindividual coefficient of variation for each animal was 29.8% (Fig. 3 ). In addition, flow cytometric analysis of WB samples from 20 female cynomolgus macaques (colony B) did not differ from those of colony A (mean ± SD = 116.98 ± 32.31 cells/µL, p = .53 unpaired t test). Therefore, no significant sex-related differences, within these age ranges, were detected between the two colonies. Based on this, we combined data from the two different animal colonies and determined the reference ranges for Tregs in whole peripheral blood of naive cynomolgus macaques to be 113.6 ± 32.32 cells/µL (n = 25), with a mean of 2.03% of total lymphocytes for CD3+CD4+CD25+ FoxP3+ cells (±0.12 SEM). Human ranges for CD3+CD4+CD25+ FoxP3+ have been reported from 0.44 to 2.8% of total lymphocytes depending on the method of fixation and antibody combination used (Law et al. 2009), underscoring the benefit of using a standardized method for Treg detection.
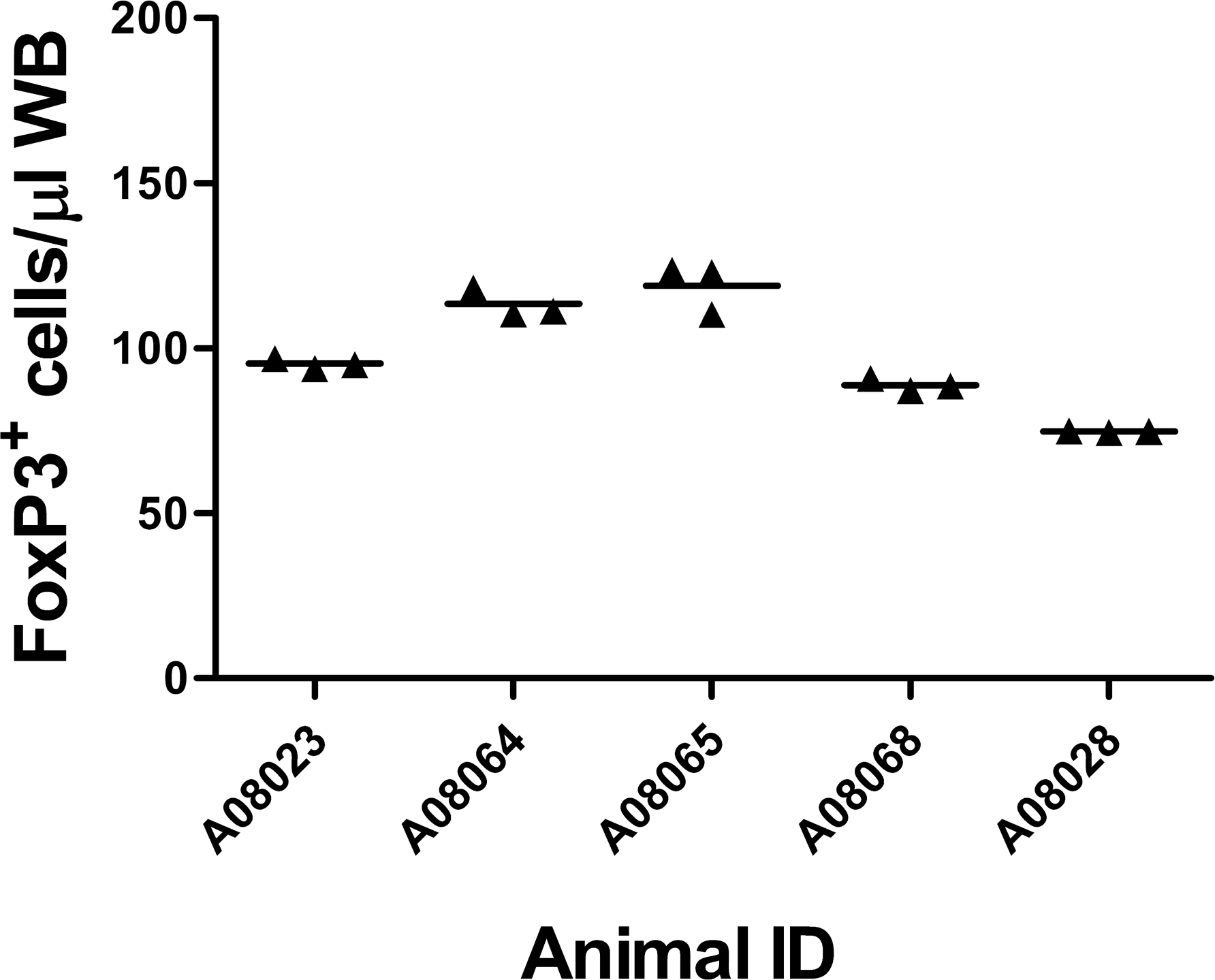
Intra-assay variation of the number of circulating FoxP3+ regulatory T cells per microliter of blood in cynomolgus macaques. Individual animal numbers are shown on the x-axis; symbols indicate the results from triplicate samples for each animal using the FoxP3 immunophenotyping panel. Data shown are from week 3 blood draw. Data from all time points gave an intraassay coefficient of variation of 2.8%.
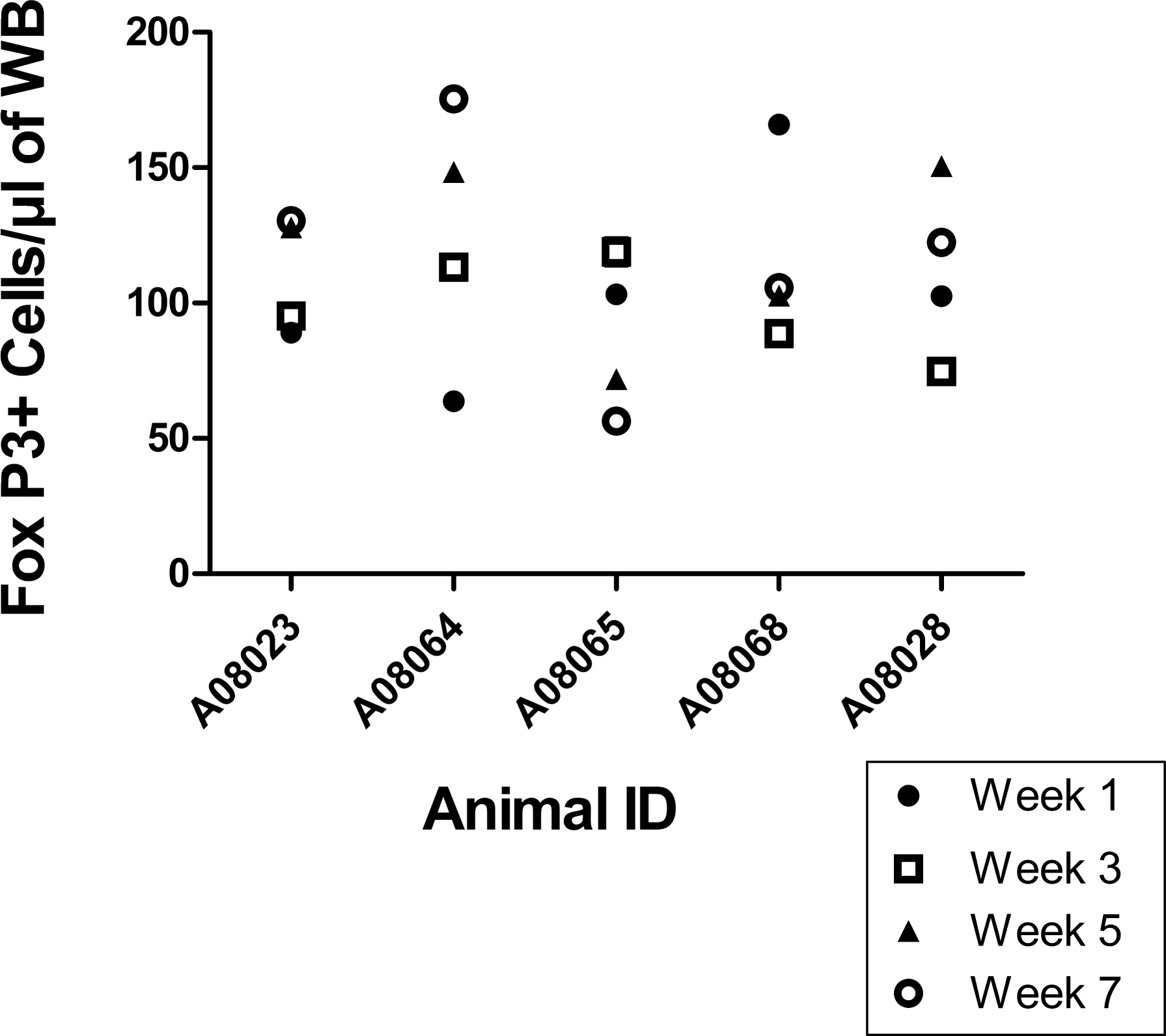
Normal variation in the numbers of circulating FoxP3+ regulatory T cells (Tregs) in cynomolgus macaques over time. Blood samples were taken every two weeks from 5 cynomolgus macaques, for a total of 4 collection time points. The number of circulating Tregs was determined using the FoxP3 IP panel. Symbols indicate results from week 1 (closed circle), week 3 (open square), week 5 (closed triangle), and week 7 (open circle). Data from all animals gave an intra animal coefficient of variation of 29.8%.
Comparison of FoxP3 IP Panel and CD127 IP Panel for Identification of Circulating Cynomolgus Tregs
Next, we compared the absolute cell counts of cynomolgus Tregs identified from our FoxP3 staining panel to a cell surface IP panel using CD25+ CD127lo expression as a marker for Tregs (CD3+CD4+CD25+CD127lo). We determined that the latter could be used to identify Tregs, in lieu of the potentially problematic cellular fixation step required for FoxP3 staining. The process of cellular fixation can result in conformational changes of cell-surface protein epitopes, thus limiting IP panel development options. While it would have been optimal to stain for both FoxP3 and CD127 in the same panel, we were constrained by the limited number of fluorochromes that were commercially available for the CD127 and FoxP3 clones that were cross-reactive with cynomolgus macaques and resulted in unambiguous staining. The most sensitive detection of FoxP3 and CD127 expression was achieved only when using PE-labeled antibodies; therefore, we could not use both the FoxP3 and CD127 antibodies simultaneously in the same panel. In lieu of this, blood samples from each of 20 cynomolgus macaques were split between two of the IP panels (FoxP3 and CD127), and the absolute number of FoxP3+ CD25+ CD4+ T cells and CD127lo CD25+ CD4+ T cells were directly compared for each animal. No statistically significant difference was detected between the absolute numbers of FoxP3+ Tregs and CD127lo Tregs (mean ± SEM 120 ± 8 cells/µL for FoxP3+ Tregs and 120 ± 7.2 cells/µL for CD127lo Tregs; p = .57, paired t test), and linear regression analysis gave r 2 = .68 (Fig. 4 ).
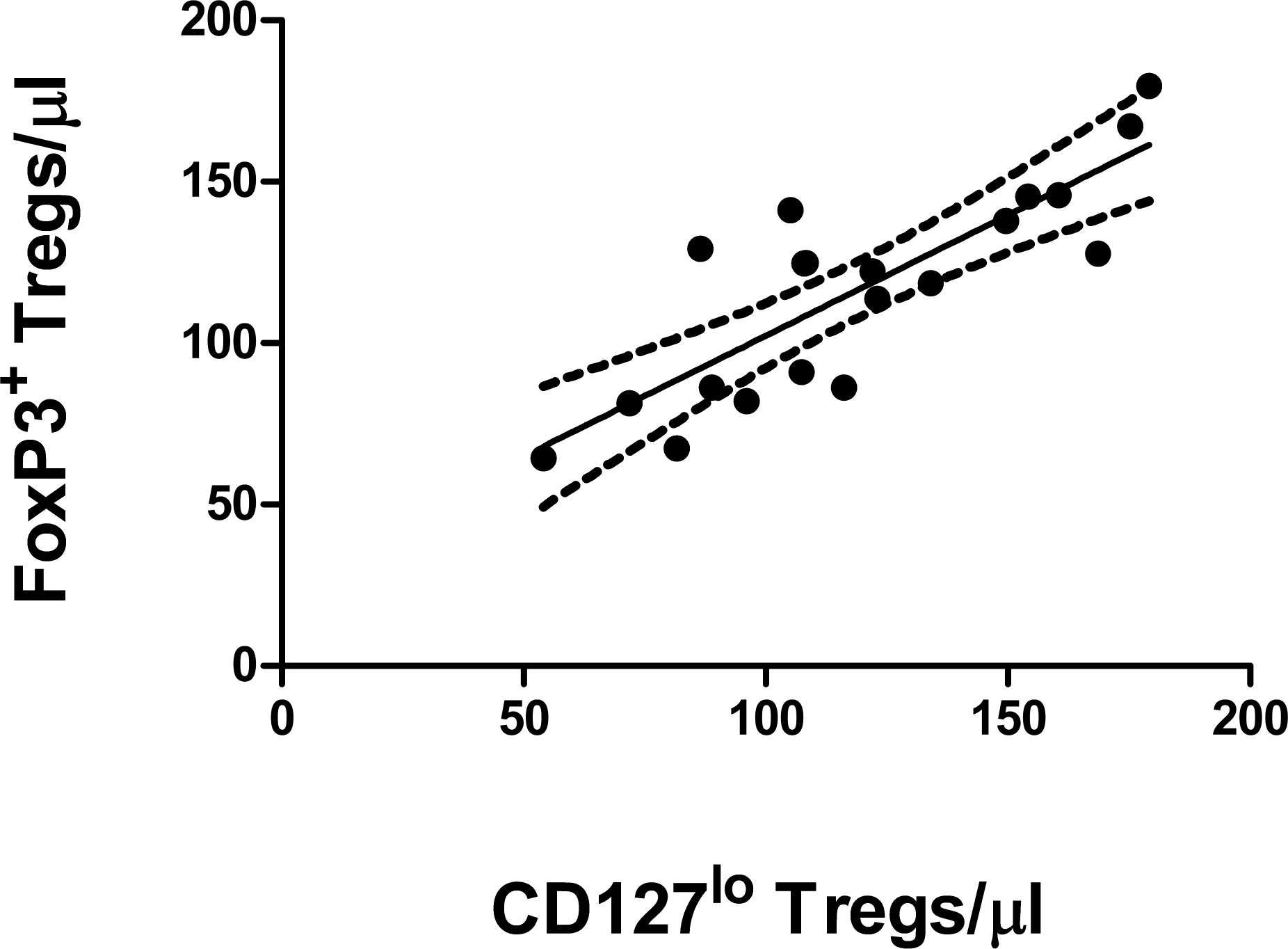
Direct comparison of the numbers of regulatory T cells (Tregs) as determined using either the FoxP3 immunophenotyping (IP) panel or the CD127lo IP panel using linear regression analysis. Blood samples from 20 cynomolgus macaques were divided into equal halves and processed using either the FoxP3 IP panel or the CD127lo IP panel. Each symbol represents the number of FoxP3+ CD25+ CD4+ T cells and CD127lo CD25+ CD4+ T cells for each animal. All samples were analyzed on the same day. The solid line indicates the best-fit line; dotted lines indicate the 95% confidence intervals. r 2 = .684, p < .001.
Discussion
For the purposes of this study, we focused on the “natural” Treg population, thymically derived FoxP3+ T cells, as opposed to adaptive or induced Tregs, which are effector T cells that acquire FoxP3 expression after exposure to anti-inflammatory mediators such as interleukin-10 or transforming growth factor–β (Bluestone and Abbas 2003). As the development of adaptive Tregs in the periphery requires the presence of antigenic stimulation (e.g., vaccination or infection), the circulating Tregs that we enumerated from naive cynomolgus monkeys were part of the natural Treg population. The FoxP3 transcription factor plays a critical role in Treg cell differentiation and function (Fontenot et al. 2003; Gavin et al. 2006; Hori, Nomura, et al. 2003; Hori and Sakaguchi 2004; Rudensky et al. 2006). It is known that Tregs constitutively express the IL-2R alpha chain (CD25), and our IP panels use this marker in our gating strategy to further delineate Tregs in addition to FoxP3 internal staining. Differences in staining reagents and protocols resulted in variable Treg percentages and absolute counts being reported in humans, thus underscoring the need for a standardized method to detect Tregs (Fox et al. 2008; Law et al. 2009; Pillai and Karandikar 2008). The FoxP3 IP panel used in this study identified Tregs from 100 µL cynomolgus WB using 96-well microtiter plate–based methodology. Our data indicate that the absolute counts of circulating Tregs in healthy naive cynomolgus macaques range from 56 to 180 cells per microliter of blood (n = 25) and that this range was consistent in two separate animal colonies. Furthermore, we did not detect any sex-related differences in either the proportion or absolute counts of circulating Tregs in cynomolgus macaques. When the absolute numbers of circulating Tregs in individual animals over an 8-week period were determined, the intra-animal variation was calculated to be almost 30%. In humans, it has been shown that intraindividual variation in circulating T-cell subsets, B-cell subsets, and natural killer cells is inversely correlated with the size of the lymphocyte subpopulation (Sekiguchi et al. 2011). Thus, the relatively large intra-animal variation in circulating Tregs in cynomolgus macaques is likely to be reflective of the small numbers of these cells compared with the total T-cell population, which has a reference range of 1.6 to 7.2 × 103 cells/µL blood (Baker et al. 2008).
Our results suggest a possible cell-surface IP panel (CD127lo) that may be substituted for intracellular FoxP3 staining to obtain Treg cell counts with fewer experimental manipulations. With cynomolgus macaque lymphocytes, antibody clones 4E3, HIL-7R-M21, and 206D gave the best staining for CD25, CD127, and FoxP3, respectively. The clone 206D worked best when labeled with PE; use of this clone with other fluorochromes (e.g., Pacific Blue) could not delineate a distinct FoxP3+ cell population in our hands (data not shown). In a similar way, the relatively low expression of CD25 and CD127 on the surface of T cells necessitated the use of antibodies labeled with either PE or APC/AF647 to obtain the clearest staining. These constraints resulted in our inability to look directly at FoxP3 expression on CD127lo CD25+ CD4+ T cells to confirm their status as Tregs. However, as low CD127 expression on CD25+ CD4+ T cells is associated with immunosuppressive function in humans (Liu et al. 2006; Seddiki et al. 2006), it is likely that the CD127lo CD25+ CD4+ T cells in cynomolgus macaques have the same function. As new reagents with greater sensitivity and selectivity become available, possible future experiments may include staining for CD 127 and FoxP3 expression in the same panel.
In conclusion, Tregs are responsible for maintaining self-tolerance, and autoimmunity can result from a deficiency in functional Tregs in both mice and humans (Brunkow et al. 2001; Fontenot et al. 2003; Kekalainen et al. 2007; Kim et al. 2007; Sakaguchi 2004; van der Vliet and Nieuwenhuis 2007). Because of the importance of the immunosuppressive function of Tregs, these cells are potential targets for therapeutic intervention for cancer and autoimmune disease (Fort and Narayanan 2010). The ability to monitor circulating Tregs and knowledge of the normal numbers of circulating Tregs in cynomolgus macaques will be key for understanding any test article–related effects seen in preclinical safety studies.
Footnotes
Acknowledgments
The authors would like to thank Drs. Nianyu Li, Marc Gavin, and Nancy Everds for their useful discussions and technical expertise and Duc Tran for technical assistance.
S.M.C., P.K.N., and M.M.F. are employees of Amgen Inc.
