Abstract
In preclinical studies, it is important to know whether the animals used are sexually mature or not. Precise data have not yet been published, however, about the histological features of the female reproductive organs during the peripubertal period or about the age of acquisition of sexual maturity in the minipig. The histological characteristics of the genital organs of female control minipigs from toxicology studies were described and, based on the presence of ovarian corpora lutea, used to assess the age at which maturity was reached. Only 50% of females can be considered mature at about 6.5 months old (a body weight of 11.8 kg), and 100% were not mature until about 7.5 months old (13.1 kg), although it is said that females reach sexual maturity at the age of approximately 5 months, by the time the body weight is about 10 to 12 kg. The uterine weights of mature females were higher than 94.4 g, whereas the maximum weight reached in the immature females was 55.2 g. In contrast, the differences between immature and mature ovarian weights were not significant. The histological appearance of the mature vagina in the various stages of the estrous cycle is also described.
Introduction
Göttingen minipigs are purpose-bred miniature pigs used in biomedical research. They can be a valuable alternative to the use of dogs or monkeys, as their anatomy and physiology are similar to those of humans in many aspects (Pond and Houpt 1978; Barth, Pfeuffer, and Scholtissek 1990; Bollen and Ellegaard 1996; Svendsen 2006; Squier et al. 2008; Bode et al. 2010; van der Laan et al. 2010). Precise data have not yet been published, however, about the histological features of the female reproductive organs during the peripubertal period in this strain or about the age of acquisition of sexual maturity, although it is said that females reach maturity at the age of approximately 5 months, by the time the body weight is about 10 to 12 kg (Jorgensen 1998; Creasy 2012). In preclinical studies, it is important to know whether the animals used are sexually mature or not, as this can affect responses to the drugs tested, and because histological features of the immature reproductive organs can mimic atrophic effects of compounds under study. The age of puberty in other laboratory species is much better studied than in the minipig; female mice reach puberty at about 1 month, rats at about 2 months, rabbits between 3 and 4 months (breed dependent), dogs between 7 and 10 months, and cynomolgus macaques between 2.5 and 3 years (Kilborn, Trudel, and Uhthoff 2002; Jorgensen 1998; Whitney and Wickings 1987). We defined immature females as those with ovaries that lacked any corpora lutea. In the absence of a corpus luteum (CL), there is no evidence that ovulation has occurred and that the animal would be fertile (Noakes 2009).
Materials and Method
Animals and Necropsy and Histology Procedures
Histological sections of ovaries, uterus, vagina, and mammary glands were reexamined from 39 female control animals from 9 toxicology studies on pharmaceuticals (between 2 and 8 animals from each study) conducted at Ricerca Biosciences S.A.S., France, between 2005 and 2010. All the animals were supplied by Ellegaard Göttingen Minipigs, Dalmose, Denmark. The dates of birth and necropsy of each animal were known. Their ages at necropsy ranged from about 5.5 to 21 months. All animals were housed individually or in pairs, in pens, without any contact with boars, at 22 ± 3°C, humidity 35 to 70%, with at least 10 air changes per hour, and with a 12-hr artificial light/dark cycle. They were fed a pelleted complete commercial diet (SMP MOD (E) SQC, Special Diet Services), 300 g diet/animal/day, and a daily piece of fruit, with filtered water ad libitum. The animals were dosed by the dermal, subcutaneous, or intravenous routes, depending on the study, with water or saline in 5 of the studies and with various other vehicles in the other studies. The studies were performed in compliance with European regulations governing the housing and use of animals and following the principles stated in the Guide for the Care and Use of Laboratory Animals (National Research Council 1996).
A complete necropsy was performed. The weights of the ovaries (weighed together, without the bursae) and uterus were taken from the study records. Weights were analyzed as absolute and as relative values (gram of tissue per 100 g of body weight). Tissues (representative parts or entire) were fixed with 10% formalin, trimmed, paraffin-embedded, sectioned at approximately 5 μm, and stained with hematoxylin and eosin (H&E). Longitudinal sections were made through the ovaries, without adjusting the plane to deliberately include corpora lutea or any other grossly visible normal structures. Both horns of the uterus were sectioned transversely, near their junction with the body, as well as a longitudinal section taken through the cervix. The vagina was sectioned transversely in the middle of its length. Mammary tissue was taken from the left inguinal region.
Histological Methods and Criteria
The presence of ovarian primordial (PD) follicles (having a primary [PY] oocyte, i.e., an oocyte arrested in prophase, surrounded by a simple squamous epithelium of flattened granulosa cells), PY follicles (PY oocyte surrounded by a simple epithelium of cuboidal granulosa cells), secondary follicles (oocyte surrounded by a zona pellucida and multiple layers of granulosa cells), tertiary or Graafian follicles (possessing an antrum and a theca), corpora lutea, and corpora albicans (CA) was recorded for the ovaries of each animal (Priedkalns and Leiser 2006; Banks 1993). Atretic follicles (AF) varied from cystic structures containing nuclear debris to smaller hyaline structures, sometimes containing oocytic remnants. Uterine gland development was classified as minimal, moderate, or marked, and their morphology was recorded (branched, coiled, or containing secretion). The appearance of the vaginal epithelium was recorded.
Mammary gland development was classified as little development or moderate or marked development, depending on the presence and the development of alveoli, cellular differentiation, the size of the ducts, and the presence of alveolar or ductal secretory product.
All the females in this study, including those that were sexually mature, had a thymus that was not involuted and a tibial growth plate that was still open, so sexual maturity is reached before thymic and skeletal maturity. We use the word “maturity” to indicate sexual maturity.
Statistics
To determine whether ages, body weights, and organ weights were statistically significantly different between immature and mature minipigs, homogeneity tests were used, after having checked the normality of distribution by a boxplot (a Fisher and Student’s test if normal distribution, or a Welch test or Kruskal–Wallis test in case of nonnormality), using the R software (Ihaka and Gentleman 1996). Determination of correlation between the proportion of mature females and either ages or body or organ weights was done using Pearson tests in the case of normal distribution, or otherwise a Spearman’s rank correlation test. In all of the box plots presented, the lowest horizontal line is the minimal value, the second line (the bottom of the box) represents the first quartile (25% of the values are below this value), the third line is the median value (50% of the values are below this value), the fourth line (the top of the box) is the third quartile (75% of the values are below this value), and the highest line is the maximal value.
Results
Histological Characteristics of Mature and Immature Female Minipigs
Ovaries
Of the 39 females included in this study, 11 were immature (i.e., lacking corpora lutea in the histological sections examined). Immature females could be described as either completely immature or partially mature, based on the degree of development of the genital organs and mammary gland. In all the immature females, PD, PY, and secondary follicles were present, in contrast to tertiary follicles that were present in only 60% of immature females (Figures 1 and 2), consistent with partial maturity. Several large follicles with thecal layers but no visible oocytes were sometimes present (Figure 2); these are presumably normal tertiary follicles, although the possibility that they are pathological cystic structures cannot be excluded. AF were present in the ovaries of immature females with tertiary follicles (Figure 3). Mature females (with 1 or more corpora lutea, Figure 4) all had PD, PY, secondary, and tertiary follicles and 90% also had CA (Figure 5). Among the animals with CA were the 2 youngest mature females, aged 174 and 188 days at necropsy, which must, therefore, have first ovulated at least 1 cycle before necropsy.
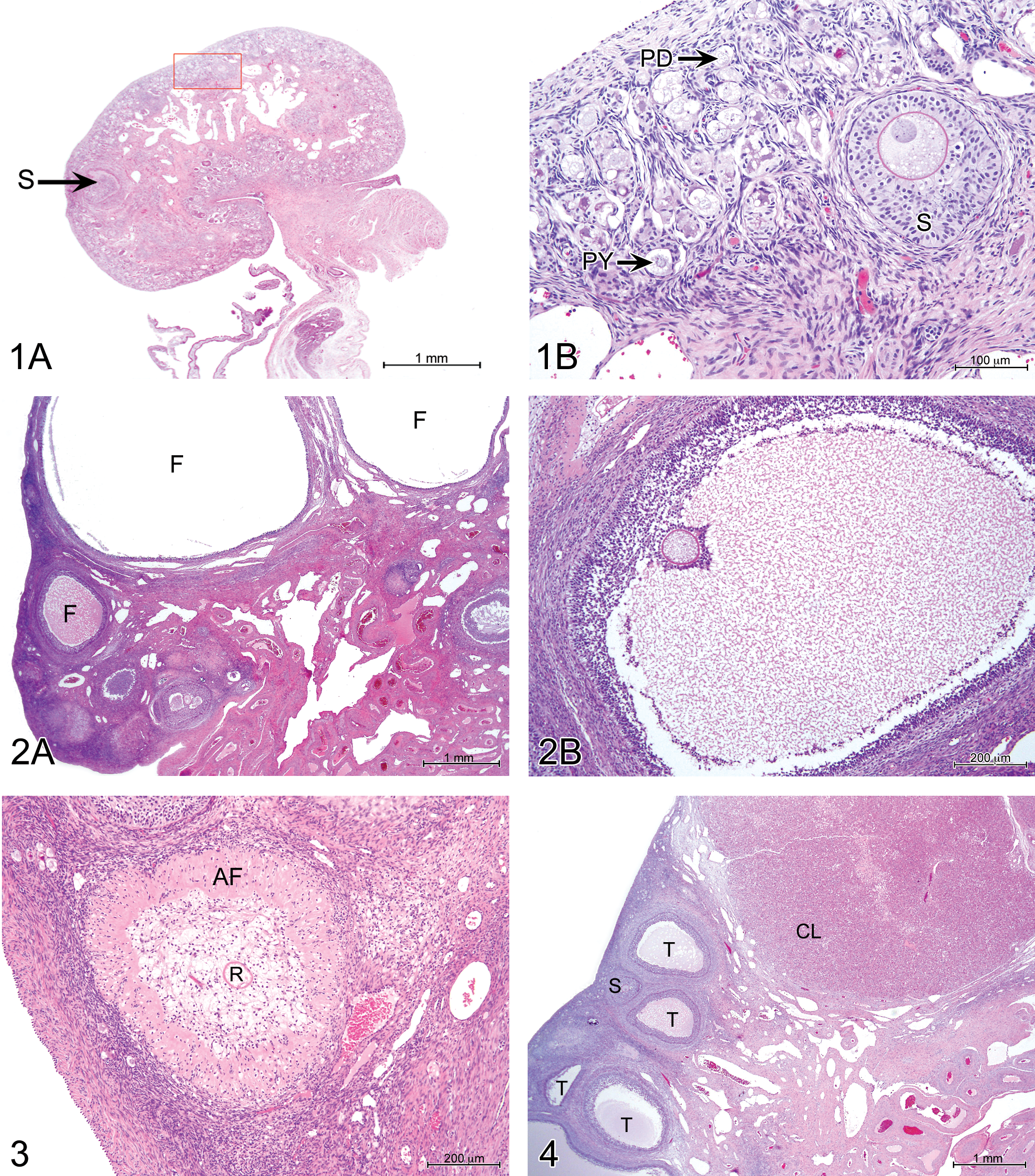
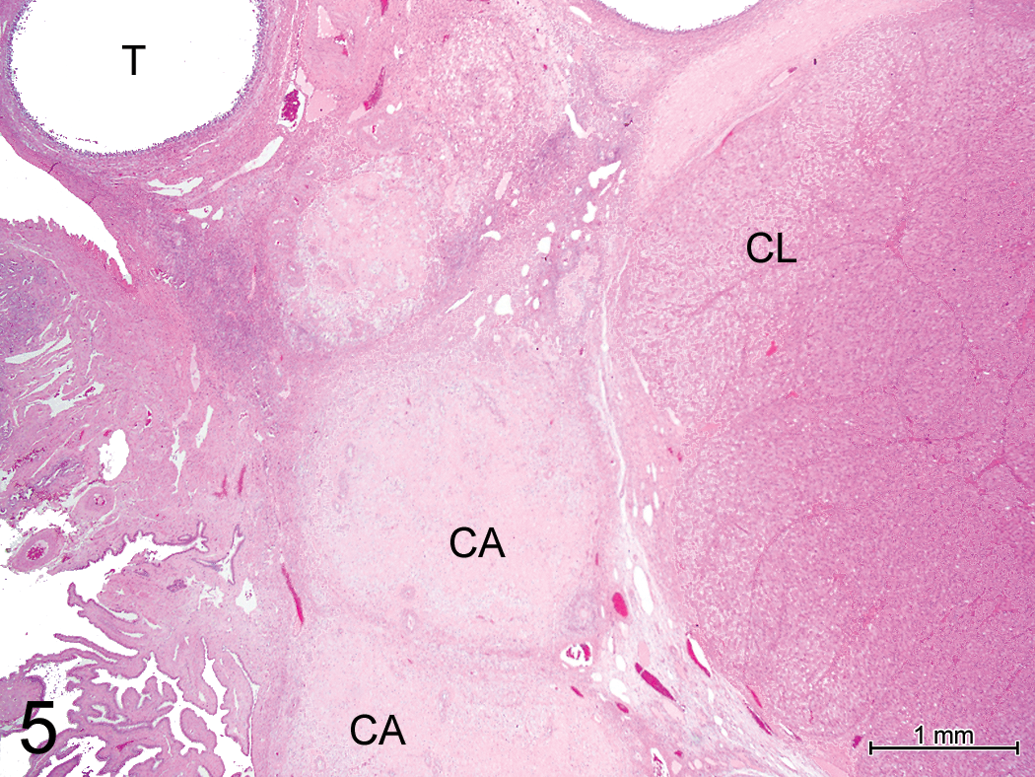
Ovary, fully mature pig, 433 days (14½ months) old at necropsy. Hematoxylin and eosin (H&E). 2× objective corpora albicans (CA), corpus luteum (CL), and tertiary follicle (T).
Uterus
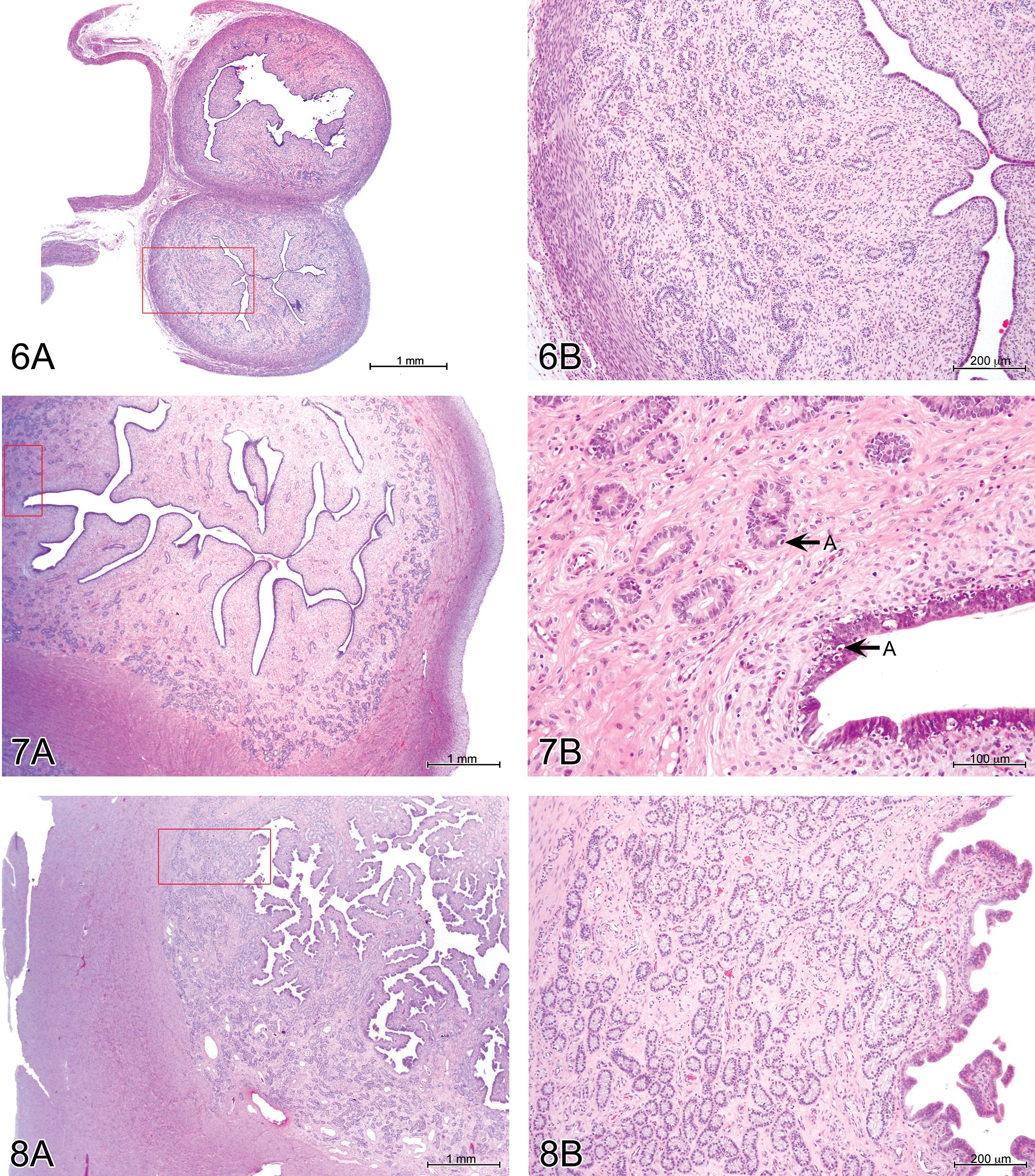
Immature females (Figures 6 and 7) consistently had few endometrial glands, which were slightly to moderately developed and mostly simple and tubular. No secretion was observed. The uterine lumen was typically narrow. There were frequent apoptotic bodies among glandular and superficial epithelial cells in partially mature animals. Mature females (Figure 8) had moderately to markedly developed endometrial glands, often branched and coiled, with a mucinous secretion in 50% of the cases. The uterine lumen was moderately to well developed, and the surface endometrium was papillary. Epithelial apoptotic bodies were not frequent. Vascular congestion was present in 22 of the 28 cases.
Vagina
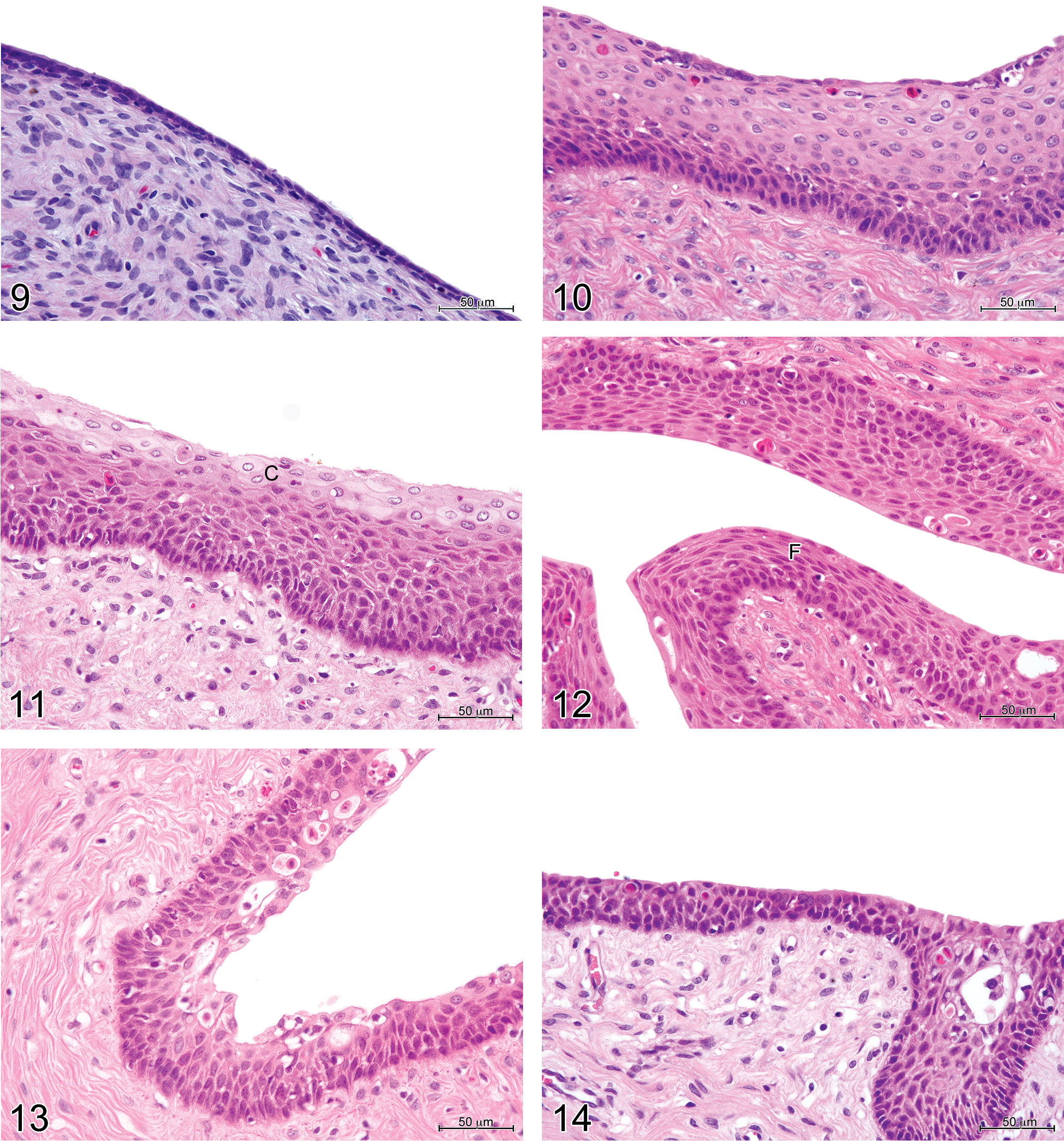
Vaginal histology in immature and mature minipigs was consistent with the appearances described for domestic pigs by Wilson (1926). Bal and Getty (1972) have also described changes with maturity in the vaginal histology of the domestic pig. Immature females had regular and uniform vaginal epithelium, varying in height between females from 1 to 4 cell layers in some cases to 18 layers (Figures 9 and 10), sometimes with polymorphonuclear neutrophil infiltration. The stage of the estrous cycle of the mature females was defined according to the appearance of the vaginal epithelium. The proestrous phase was determined by the presence of a stratum germinativum (i.e., 1 layer of basal cells), up to 10 intermediate layers, and a superficial layer of cuboidal to ovoid cells with pale cytoplasm (Figure 11). The estrous phase was characterized by the presence of a stratum germinativum, intermediate layers, and up to 6 layers of superficial, flattened cells (Figure 12). No cornification was present. During the metestrous phase (diestrus I), there were 4 to 8 layers of cuboidal to ovoid cells, with polymorphonuclear cells infiltrating both the epithelium and the lamina propria, and epithelial vacuoles containing cellular debris (Figure 13). In the diestrous phase (diestrus II), there were between 5 and 8 layers of cells, morphologically similar to those seen during metestrus, but with no, or minimal, polymorphonuclear cell infiltration (Figure 14). Of the 28 mature females, 10 were in the diestrous phase of the cycle, 14 in the proestrous/estrous phase, and 4 in metestrus.
Mammary Gland
Immature females had little mammary gland development with no secretion. Ducts were few and small. Alveoli, where present, had epithelial cells that were either quiescent (small and single layered) or showed early development (with larger, multilayered cells; Figures 15 and 16). In all mature females, mammary glandular tissue was moderately to markedly developed, with numerous alveoli (Figure 17). Hyperplastic glandular cells were visible in about one-third of the cases with markedly developed mammary glands, and alveoli and canals were filled with eosinophilic homogeneous secretory material in many cases.
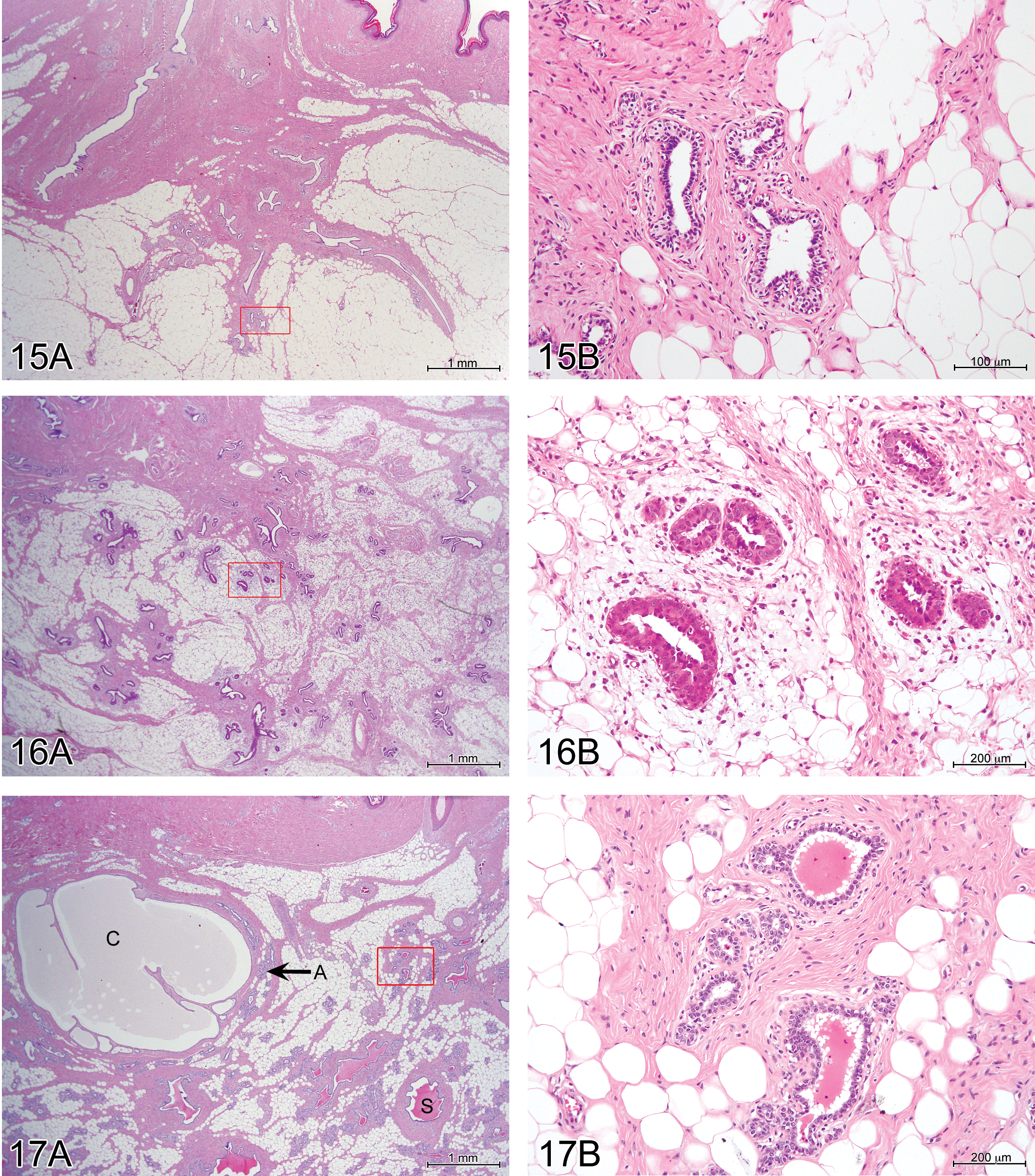
The ovarian and uterine appearances and degree of development of mammary tissue did not vary with the phase of the estrous cycle.
Age of Female Minipigs at Maturity
Figure 18 shows the relationship between age and sexual maturity. The ages at necropsy of the immature females ranged from 164 to 229 days, whereas for mature females, ages ranged from 174 to 433 days. The first females reached maturity at the end of the fifth month of life, 50% of females were mature at about 6.5 months, and all were mature at about 7.5 months. It is, therefore, impossible to predict whether a female is mature from its age alone over a period of 2 months. A Pearson’s correlation test showed, however, that correlation between age and maturity was high (0.95) and significant (p < .001).
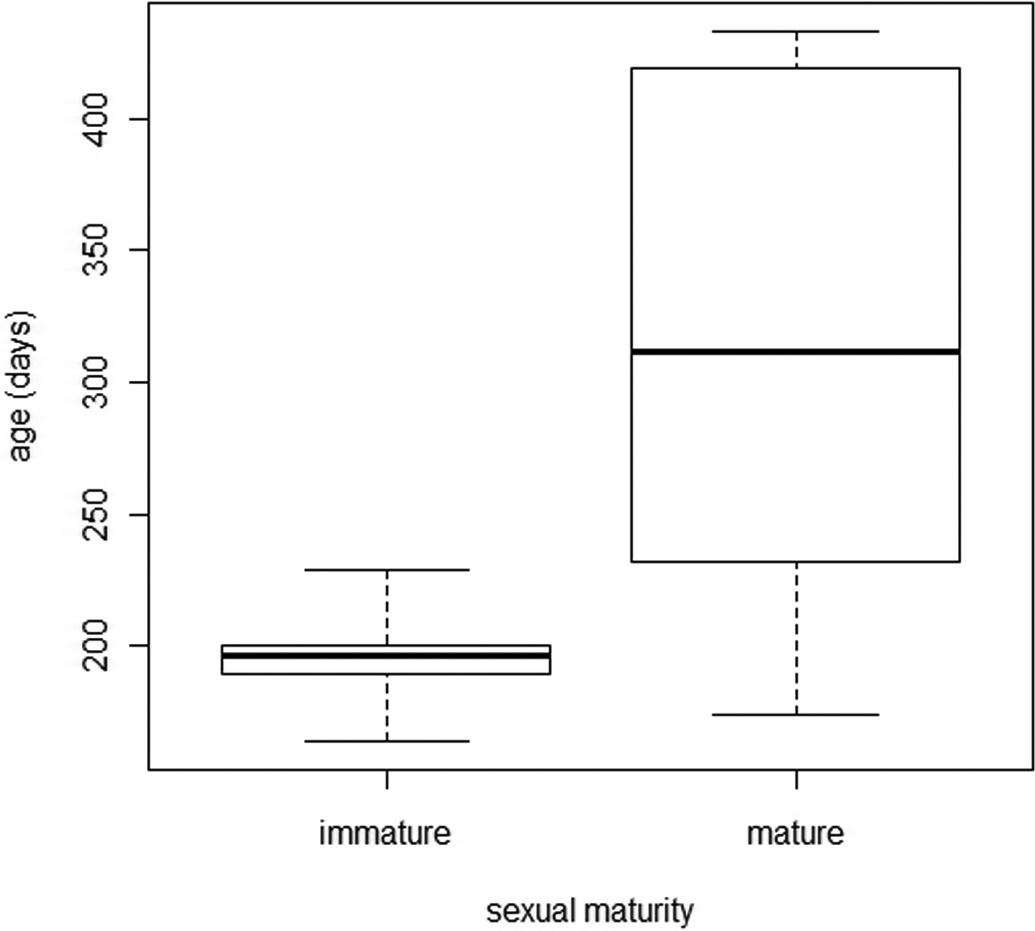
Ages at necropsy of immature and mature female minipigs.
Weights
Figure 19 shows the relationship between body weight and sexual maturity. The difference between immature and mature mean body weights was significant (11.41 vs. 14.8 kg, p < .001). The percentage of mature females and total body weight were significantly correlated (p < .001 and r = .981, Pearson’s test). Thus, by weighing the animals, it was possible to predict the probability of them being mature. Fifty percent of females were mature at a body weight of 11.8 kg. No immature females had a body weight of more than 13.1 kg and all mature females had a body weight of more than 11.1 kg.
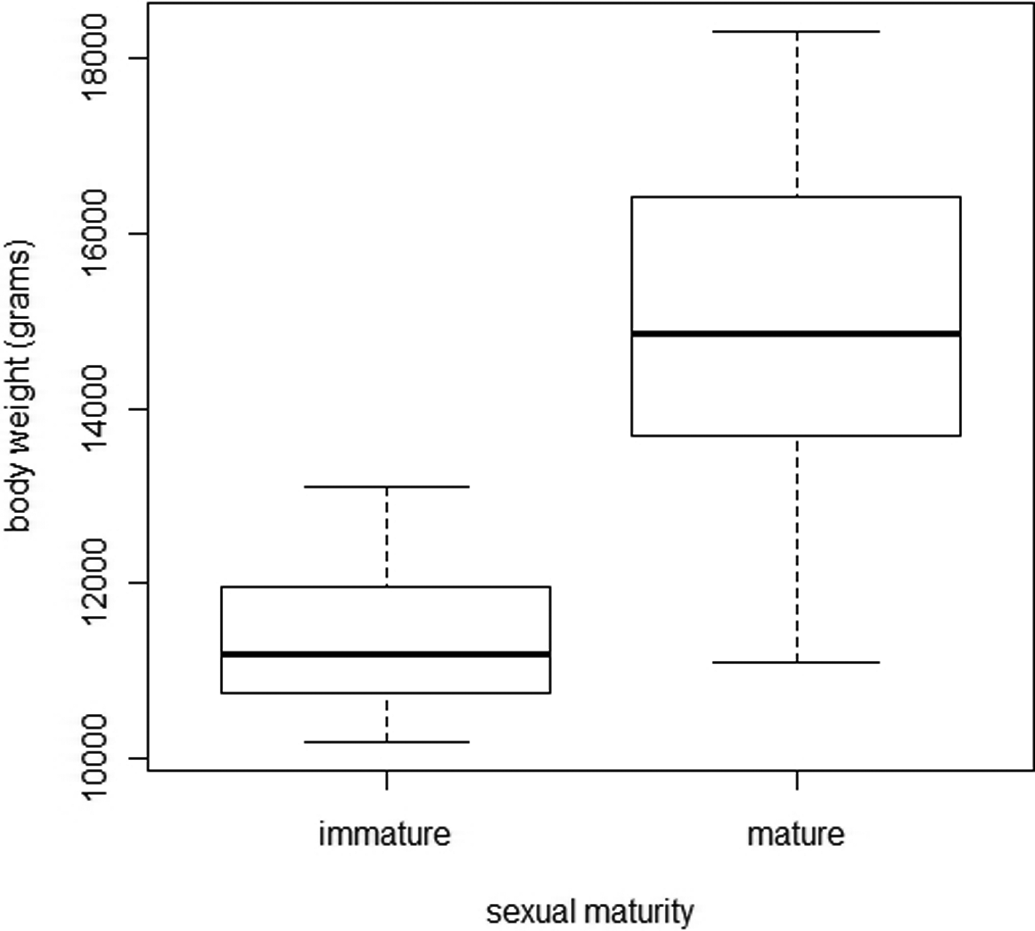
Comparison of body weight in immature and mature female minipigs.
Figure 20 shows the relationship between uterine weight and sexual maturity. Immature females had significantly lower mean uterine weights than mature females (19.4 g vs. 157.8 g, p < .001: bilateral Welch test). All the values of uterine weights of mature females were higher than 94.4 g, whereas the maximum reached in the immature females was 55.2 g. This difference was not due solely to higher body weight in mature females, as the relative uterus weights were also higher (data not shown). In mature females, uterus weights were not significantly different according to the phase of estrous cycle (Kruskal–Wallis test, with p < .001).
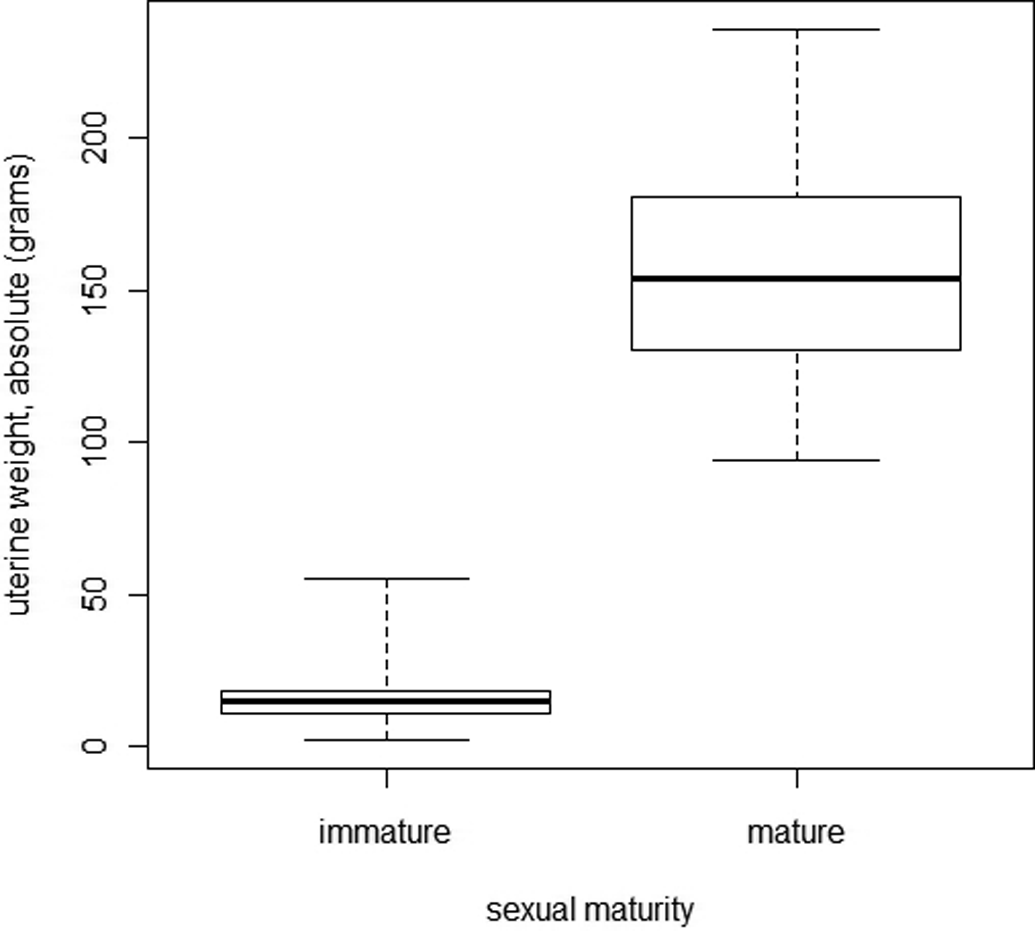
Comparison of uterine weight between immature and mature female minipigs.
Figure 21 shows the relationship between ovarian weight and sexual maturity. The distribution of ovarian weights was not normal, and the bilateral Welch test showed that the difference between immature and mature minipigs was not statistically significant (p > .001).
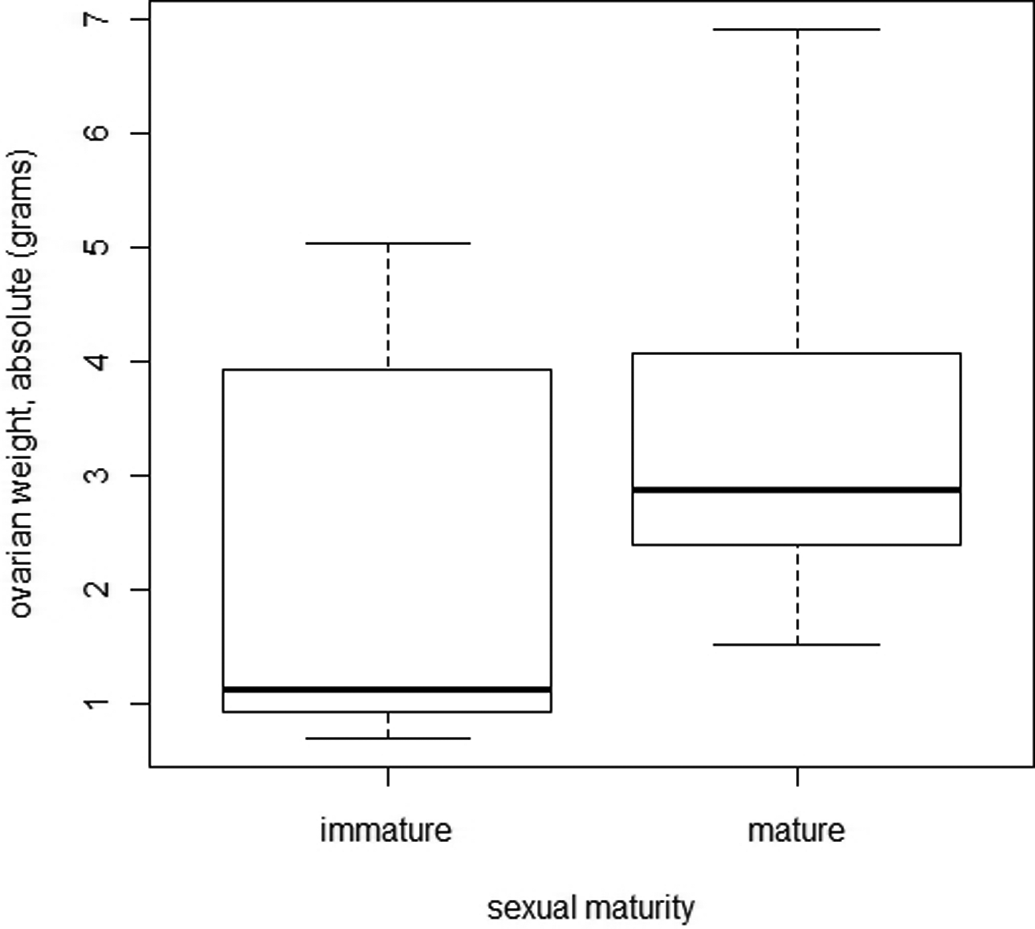
Comparison of ovarian weight between immature and mature female minipigs.
Discussion
Göttingen minipigs are commonly used in toxicologic studies, and the pathologist must have a precise knowledge of the biologic features of the strain to make an accurate interpretation of lesions. The data presented here contribute to the knowledge on the onset of puberty and normal histology of peripubertal female minipigs. Our study compares the histological characteristics of the immature and mature genital organs, and aims at determining if age and body, uterine and ovarian weights correlate with the acquisition of sexual maturity.
Histology is helpful in determining the sexual maturity of females that have been necropsied, as maturation of follicles, ovulation and the development of corpora lutea and then CA indicate the transition from immaturity to sexual maturity. The histological observation of a CL was used to confirm maturity in this study. Histological findings must be interpreted cautiously, however, because in 1-year-old cynomolgus macaques, groups of luteal cells representing small luteal bodies can occur, although ovulation has not yet started (Buse, Zoller, and van Esch 2008). In the minipigs in our study, however, the corpora lutea were all well developed, and it is likely that they did derive from follicles that had ovulated. The presence of adult serum levels of follicle-stimulating hormone is an alternative indication of maturity (as proposed for cynomolgus macaques by Buse, Zoller, and van Esch 2008), but hormone levels were not measured in our study. The presence of estrous appearances (redness and swelling of the vulva) and behavior (tendency to mount or to be mounted) were also not assessed in our study, although it is possible that these could start, especially in an irregular fashion, before the first ovulation.
The possibility cannot be excluded that a CL was present in an ovary but was not included in the histological section, although since porcine corpora lutea are relatively large in relation to the size of the ovary, this is unlikely. The plane of section was not adjusted to include grossly visible normal structures such as corpora lutea, in the interest of using a standardized procedure throughout the study. It could be argued, however, that the number of grossly visible corpora lutea should be recorded at necropsy, and that when they are present, representative corpora lutea should be included in sections.
We found that the histological features of other reproductive organs correlate very well with ovarian findings. Uterus, vagina, and mammary gland are often slightly developed in immature females, whereas in mature females, they are moderately to markedly developed, with increased number and size of glands in the uterus and mammary gland, sometimes associated with secretion. The vaginal epithelium showed no evidence of estrous cycling in immature animals, while mature females demonstrated features characteristic of the various estrous cycle stages that are reported in other species. In the partially mature animals, the histological appearance of the vaginal epithelium resembled that of the mature metestrous or diestrous phases. More work is, however, required, to correlate the histological appearance of the vagina with estrous behavior in the mature minipig, as differentiation of proestrus from estrus was not always clear on histological criteria.
The transition from sexual immaturity to maturity usually happens later than at the age of 4 to 5 months that is generally quoted. Only 50% of females can be considered mature at 196 days (6.5 months), and 100% were not mature until 230 days (7.5 months). Female minipigs can be mated and become pregnant at 5.5 months (unpublished observations), however, which is in line with our post mortem observations that females just under 6 months old can be histologically mature. Body weight provides an indication of the probability of maturity. This noninvasive method would be helpful in assessing maturity in minipigs in toxicology studies. We also show that if the uterus weighs more than 94 g, a female can be considered mature (regardless of stage of the estrous cycle). On the other hand, ovarian weight is useless in this context, as the differences between immature and mature ovarian weights were not significant.
The histological appearance of the mature vagina allows one to determine the phase of the sexual cycle, using features that are consistent with data from other species used in toxicology studies. However, a distinction between the different phases is not as obvious as in rodents, rabbits, dogs, or monkeys.
All the females included in this study were from the same supplier and were bred, housed, and fed in the same way. Because housing, management, and diet during the prepubertal period influence sexual maturity and reproductive performance (Mavrogenis and Robinson 1976; Klindt, Yen, and Christenson 2001), variations from our results could occur in females that are not bred and kept in the same conditions. The absence of contact with boars in these minipigs could have slowed the acquisition of sexual maturity. The route of dosing did not seem to influence the acquisition of maturity, and the vehicles used were not expected to affect this.
There is a need for further work on the confirmation of sexual maturity in the female minipig, especially correlating our results with other data such as hormonal analysis, imaging, vaginal cytologic examination (Rodgers et al. 1993), and behavioral observations.
In summary, we describe the histological features of the female reproductive organs during the peripubertal period in the Göttingen minipig. Age, body weight, and uterine weight correlate well with whether the animal is histologically sexually mature. Only 50% of females can be considered mature at 196 days (about 6.5 months) of age or a body weight of 11.8 kg. Between about 5.5 and 7.5 months of age and about 11 and 13 kg bodyweight, the animals could be either immature or mature.
Footnotes
The authors were employed or otherwise financially supported by either Ricerca Biosciences S.A.S. or Ellegaard Gottingen Minipigs during the preparation and submission of this article.
