Abstract
The present study describes the normal histology of female reproductive organs during the estrous cycle in the Göttingen minipig. For this purpose, sexually mature females were sacrificed at different phases of the cycle (follicular/proliferation, ovulation, and early-, mid-, and late-luteal/secretory phase). Ovaries, uterus, cervix, vagina, and mammary gland tissues were processed for microscopic evaluation. Sexual maturity was assured by selecting females in which at least 1 progesterone peak was measured. Stage-distinguishing features in ovaries were the Graafian follicles (disrupted vs. nondisrupted) and corpora lutea of recent and preceding cycles (size, cell morphology, and structural composition). In the uterus, stage-specific markers were epithelial morphology, secretory activity (using periodic acid–Schiff/hematoxylin staining), and epithelial mitosis and/or apoptosis. The other reproductive organs were not suitable to discriminate between the different phases of the cycle due to a high morphologic variability (mammary gland, and vagina) or absence of clear morphologic differences between the phases (cervix). The increased use of young minipigs (frequently immature/peripubertal) in preclinical testing requires more knowledge on the histologic cyclic changes. With the present morphologic description of the morphologic characteristics of the reproductive tract in recently ovulating minipigs, a guidance for staging the estrous cycle and determination of sexual immaturity is provided.
Keywords
Introduction
Minipigs are increasingly used in preclinical testing, both in general and in reproduction toxicity studies. Due to the similarities to man and suitability for the different study types, the minipig is increasingly becoming a favored nonrodent species (Jørgensen 1998; Svendson 2006; Bode et al. 2010). Background reference data were collected for several years and many of these data sets are reported. Information on spontaneous/background pathology of Göttingen minipigs (McAnulty et al. 2011) is very important for a reliable interpretation of histopathological data from preclinical studies. Besides spontaneous/background pathology, detailed knowledge of the reproductive system of the minipig is a prerequisite for a correct evaluation of reproductive functioning. This information is rather limited for the female minipig, especially information on histopathologic changes of reproductive organs during the normal estrous cycle is lacking in the current literature.
In general, most toxicology laboratories use female minipigs of approximately 4 to 5 months old at the start of the experimental phase. The female minipigs may become sexually mature within the time frame of the study, but most of the females will not reach maturity and a reliable histopathological and functional evaluation of possible test item–related effects on the female reproductive organs is not possible. The age of sexual maturity in female minipigs was considered to be approximately 5 months when they have a body weight of approximately 12 kg (Jørgensen 1998; Creasy 2012). However, a recent retrospective study revealed that although sexual maturity can be reached starting from 5 months of age, only 50% of females of 6.5 months were sexually mature and only at 7.5 months of age (weights around 13 kg) was sexual maturity guaranteed (Tortereau, Howroyd, and Lorentsen 2013).
Knowledge of the estrous cycle of pigs is primarily related to breeding and therefore focused on mating and pregnancy rates and how to manipulate the estrous cycle in order to improve the success rate of the offspring. The average cycle length of the pigs and minipigs are 21 days and starts when hormone levels are high enough for the initiation of the preovulatory luteinizing hormone (LH) surge (i.e., a sudden elevation of LH as a response to rising estrogen levels), which corresponds to the onset of estrus and usually takes 2 days. The LH surge is followed by ovulation and increased progesterone levels, which decrease again at the end of the cycle when no mating occurs and the females do not become pregnant (Howard et al. 1982; Jørgensen 1998; McAnulty et al. 2011; Parvizi et al. 1976). Although in a large number of publications attention is paid to behavioral aspects and hormonal fluctuations of pigs and minipigs at estrus and pregnancy, details on histological characteristics of the reproductive female organs during a normal estrous cycle are lacking.
Such descriptions of the morphological features of the female reproductive organs during the estrous cycle can be found in the literature for numerous species used in preclinical testing. For the rat, a practical histological guide for staging the female reproductive phases has been published (Westwood 2008) and also for the dog the histological characteristics of the female reproductive organs have been described in detail (Rehm, Stanislaus, and Williams 2007). In both animals, the terminology that is used for the different stages is estrus, metestrus, diestrus, anestrus (for dogs only), and proestrus. In pigs and minipigs, the term estrus is primarily used to indicate the period of sexual behavior of the sow (Holtz and Bollen 1999) and is not really correlated to specific histologic features within the female reproductive organs. In nonhuman primates, descriptions of the female reproductive organs during the cycle are in parallel with human literature and terminology used is different from rats and dogs. Different stages are classified based on changes in ovaries (follicular growth in the follicular phase and follicular luteinization after ovulation in the luteal phase) or in the uterus (proliferation phase and secretory phase; Buse et al. 2008).
In the present study, 12 young sexually mature female Göttingen minipigs were used to evaluate the histomorphological characteristics of the reproductive organs. To assure sexual maturity, the minipigs were monitored twice weekly for progesterone concentrations and were sacrificed after at least 1 progesterone peak at different phases of the estrous cycle. A 13th animal with undetectable progesterone concentrations until approximately 7 months of age was evaluated as well. Ovaries, uterus, cervix, vagina, and mammary glands were processed for histopathologic examination. Staging of the estrous cycle was mainly focused on easily recognizable stage-specific morphologic features in the set of reproductive organs evaluated. Since in the present histological evaluation the morphologic characteristics of both ovaries and uterus play an essential role in distinguishing the different phases, the cyclic phases are classified into a follicular/proliferation (F/P) phase and early- (EL), mid- (ML), and late-luteal (LL)/secretion (LS) phases.
Material and Methods
Animals
Naive Göttingen minipigs (in total 13 females; outbred and microbiologically defined) were housed in the facilities of Ellegaard Göttingen Minipigs in Denmark. The animals (except 1) were proven sexually mature by measuring progesterone levels twice weekly starting at the age of 3 to 4 months. Females were considered to be sexually mature when at least 1 progesterone peak was measured. After this peak, necropsy dates were scheduled and, based on the expected second or third peak, the animals were sacrificed at different time points of the estrous cycle (expected cyclic phases: 2/13 females in F/P phase; 2/13 females at/shortly after ovulation; 3/13 females in EL/secretory (ES) phase; 3/13 females in ML/secretory phase; 1/13 females in LL/LS phase; and 1/13 females did not show detectable progesterone levels). At necropsy, the animals were between 5 and 8 months of age.
A controlled environment was maintained in the rooms with approximately 10 air changes per hour, a temperature of 19 ± 3°C, a relative humidity of 60% to 70%, and a 12-hr light/12-hr dark cycle. Animals were group housed in connected pens. Enrichment of the pens consisted of iron chains, rubber bite sticks, chopped straw, and plastic pyramid for climbing. Diet consisted of commercial minipig diet (SDS, Essex, United Kingdom) and was distributed twice daily (total daily amount 110–140 g). Tap water was provided
Progesterone Measurements
Progesterone concentration (ng/ml) was measured in serum, which was sampled twice weekly from the jugular vein, using the Immulite 1000 (Siemens Medical Solution Diagnostic, Inc., in Tarrytown, New York), a sequential competitive immunoassay with an analytical sensitivity of 0.2 ng/ml. For control samples, CON6 Multivalent Control Module was used (human serum based, Siemens Medical Solution Diagnostics). In order to have histologic material of all stages of the estrous cycle, the time of necropsy was individually chosen based on the progesterone curves in which at least 1 progesterone peak was detected.
Necropsy
Minipigs were deeply anesthetized at the different time points of the estrous cycle with a mixture of Zoletile50® Butorphanole, Xylazine, and Ketamine (125-mg tiletamine and 125-mg zolazepam [Virbac laboratories, Carros, France] in 6.25-ml Rompun®Vet [20-mg/ml xylazine; KVP Pharma + Veterinär Produkte GmbH, Kiel, Germany], 1.25-ml Ketaminol® [100-mg/ml ketamine; Intervet, Boxmeer, the Netherlands]), and 2.50-ml Torbugesic® Vet (10-mg/ml butorphanol; [Scanvet Animal Health, Fredensborg, Denmark]) and subsequently euthanized with an overdose of Pentobarbitone (Pentobarbitalnatrium®; pentobarbitalnatrium 30% [Vepidan Aps., Loegstoer, Denmark]).
Necropsy was performed on all minipigs, and ovaries, uterus (including cervix), vagina, and mammary gland area were collected and fixed in 10% buffered formalin (neutral phosphate buffered 4% formaldehyde solution, SSI Diagnostica, Hillerød, Denmark).
Histotechnique
Ovaries, uterus (including cervix), vagina, and mammary gland were trimmed, processed, and embedded in paraffin, cut at a thickness of 2 to 4 μm and stained with hematoxylin/eosin (H&E; Klinipath, Duiven, the Netherlands). Uterus, cervix, and vaginal tissue sections were additionally stained with periodic acid–Schiff/hematoxylin (PAS/hematoxylin; Klinipath, Duiven, the Netherlands). Ovaries were orientated longitudinally and approximately at the center of both ovaries of a female, 1 section was taken for evaluation.
Results
Phases during the Minipig Estrous Cycle
In Figure 1, the course of progesterone levels during a representative estrous cycle is given. The figure was composed of all the progesterone curves available from the females of the present study and drawn as the mean progesterone course during an estrous cycle of 20 to 21 days of a sexually mature female minipig. The peak of the progesterone levels varied significantly among the different females and the different cycles, but the mean of all peaks was approximately 50 ng/ml. (It should be taken into account that progesterone levels were not measured daily; the progesterone data are not provided.) In Figure 1, the estrous cycle is divided into 5 periods representing the different stages (including ovulation). The F/P phase starts after the disappearance of the progesterone peak (day −3, i.e., 3 days before ovulation). Since the exact timing of ovulation (day 0) is difficult to determine, an ovulation period of 2 days is drawn in Figure 1 (from day −1 to day +1). At ovulation (day 0), the progesterone level begins to rise for approximately 5 to 6 days (EL/ES phase), the progesterone concentration stays at a high level for approximately 6 to 7 days (ML/MS phase) and will fall down progressively at the last 3 to 4 days of the cycle (LL/LS phase).
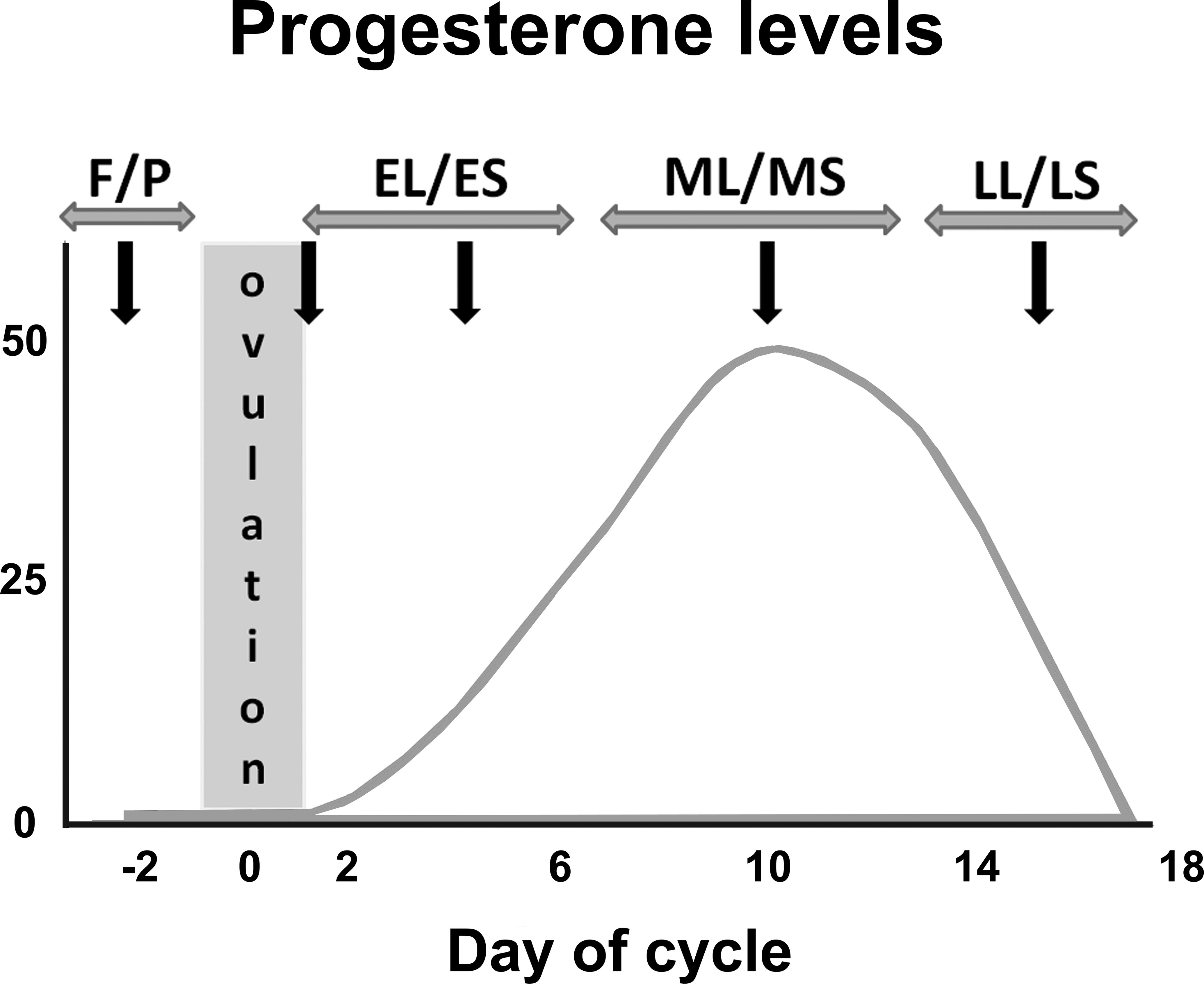
Schematic drawing of a progesterone curve based on measured progesterone levels in serum of female minipigs during an estrous cycle of 20 to 21 days. Progesterone levels (
Morphological Staging of the Minipig Estrous Cycle
A combination of the morphologic characteristics of ovaries and uterus appear to be very useful for staging of the estrous cycle in minipigs. Although other female reproductive organs such as vagina, cervix, and mammary gland can display stage-specific features, changes are less obvious and also much more variable between young sexually mature individuals within the same cyclic stage than the changes in ovaries and uterus. Histopathologic changes, other than cycle related, were not observed in any of the organs examined.
To distinguish between the different stages in the ovaries, it is essential to evaluate tertiary follicles and the preovulatory Graafian follicles together with corpora lutea. Some of these differences between the cyclic phases are already visible at subgross images of the ovaries (Figure 2). A more detailed examination of the morphology and the state of regression or degeneration of the corpora lutea is helpful in distinguishing the stage of the estrous cycle. In addition to follicular size, the disrupted status and the ratio of atretic:nonatretic follicles may be useful distinguishing features. Presence of corpora lutea (mature, regressing, or remnants) is a good marker for the postovulation stage and in most cases it means that a continuous regular estrous cycle will occur in that particular female.
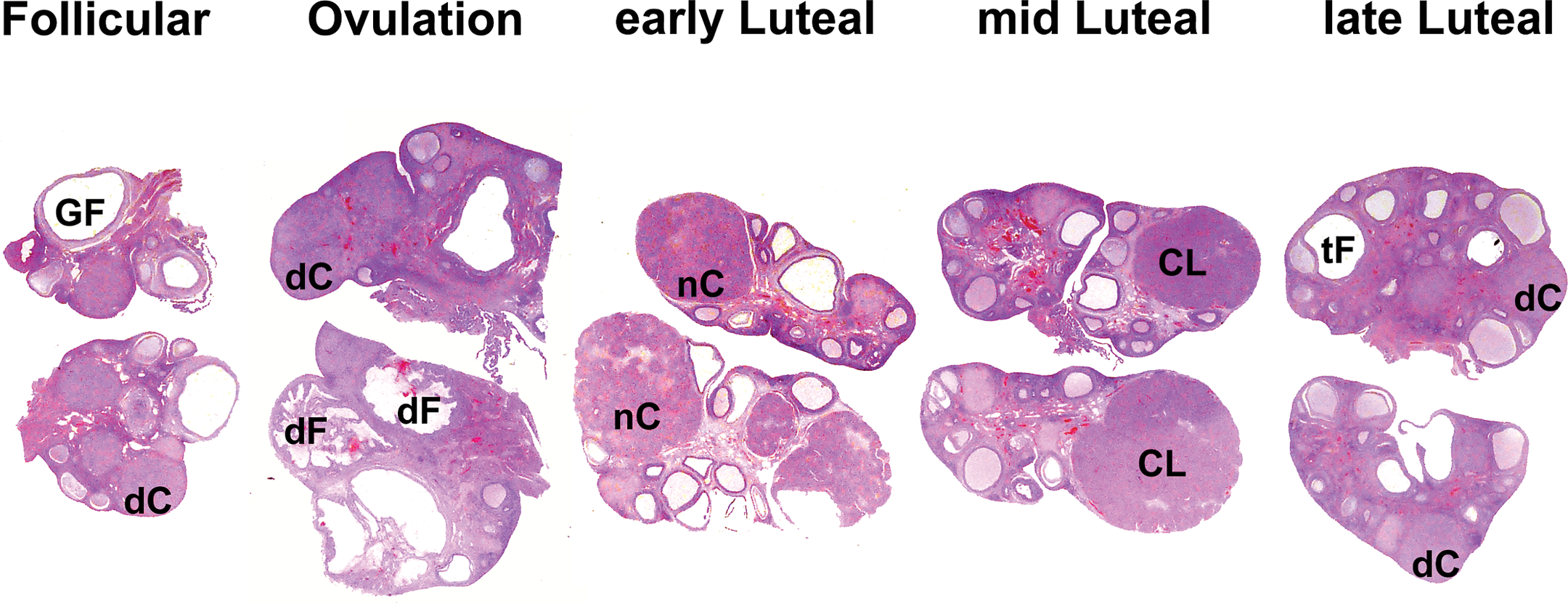
Subgross images of ovaries from the different cyclic phases: follicular phase, phase just after ovulation, early-luteal phase, mid-luteal phase, and late-luteal phase. Some of the phase-specific features are visible at this level such as Graafian follicles (GF), small degenerating corpora lutea (dC), disrupted follicles (dF) with blood, newly formed corpora lutea (nC) of which the lumen is not yet closed, large mature corpora lutea (CL) completely closed, and medium-sized tertiary follicles (tF), possibly developing into Graafian follicle of the next cycle. Magnification approximately 1.7×.
The uterus of the minipig can be divided into 3 functional zones, comparable with the macaque endometrium (van Esch et al. 2008). In Figure 3, the different zones are indicated. Zone I represents the luminal epithelium, zone II is the glandular layer underneath the luminal epithelium (in macaque: zona functionalis), and the more basal glandular zone III is surrounded by the myometrium (in macaque: zona basalis). Since the morphology of the 3 zones may differ during the different stages of the cycle and may be helpful in defining stage-specific morphologic features, the zones have been evaluated separately. In general, the stroma is rather scant and hyaline at all phases of the cycle and no obvious stage-specific stromal changes have been observed.
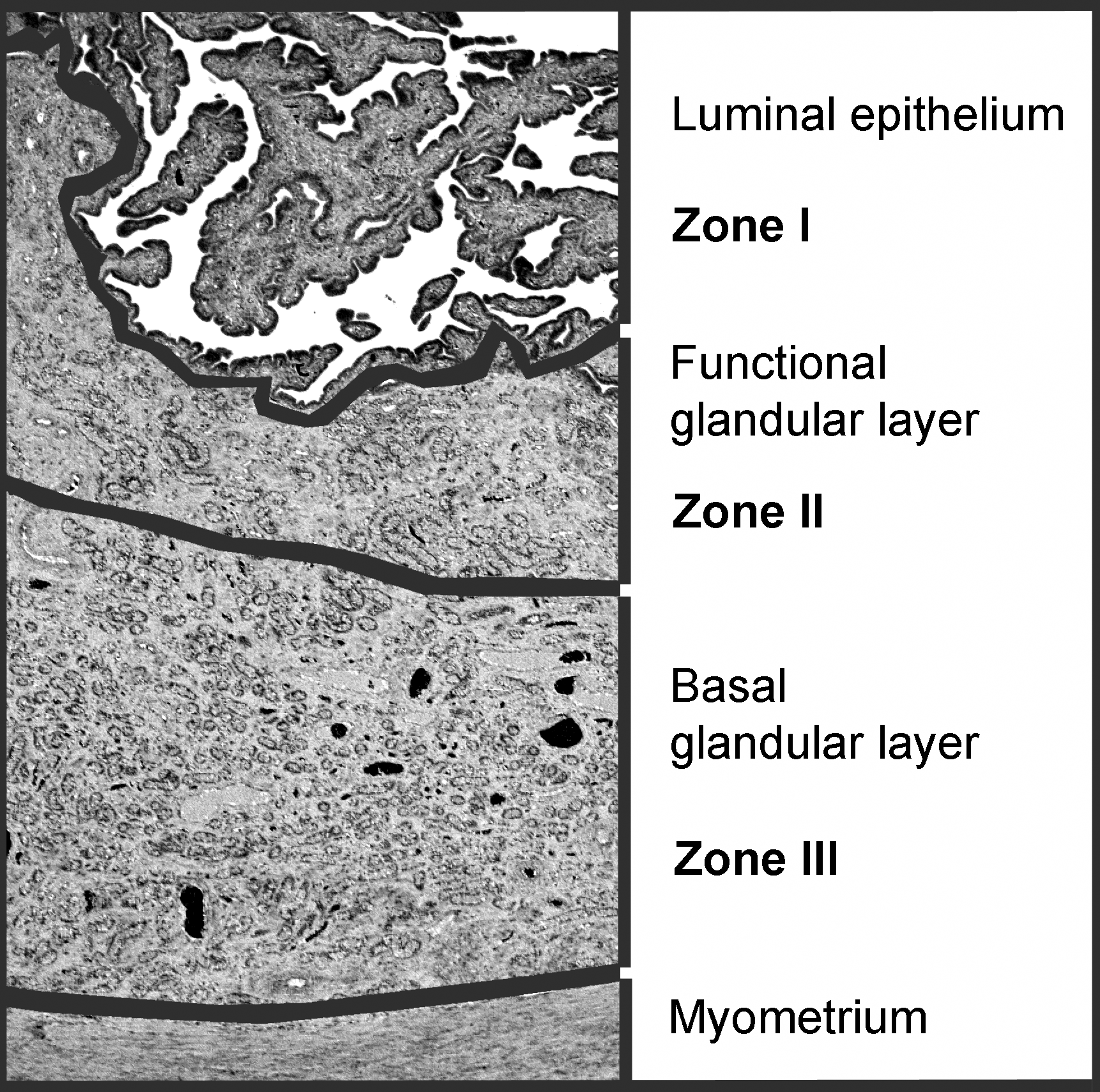
Low power view of the different layers within the uterus of minipig (example of mid-secretory phase). The gray lines separate the different zones within the endometrium (zones I, II, and III) and the endometrium from the myometrium (H&E; OM = original objective lens magnification = 2×). H&E = hematoxylin/eosin.
F/P Phase
Ovaries
In the follicular phase, the large tertiary follicles, Graafian follicles (5–8 mm in diameter), are the most prominent structures in the ovaries (Figure 4A). Macroscopically, they are visible as cyst-like structures; microscopically, they show large fluid-filled cavities that are surrounded by granulosa cells arranged in a layer of 5 to 6 cells. Some of the Graafian follicles are atretic (Figure 4A, inset). Besides large Graafian follicles, medium-sized tertiary follicles are present that are in most cases atretic. Ovaries in the follicular phase also contain corpora lutea of the previous cycle, which can be easily recognized at microscopic evaluation (Figure 4B). They mainly consist of degenerating luteal cells with foamy/vacuolar cytoplasm. The cells are embedded in areas with hyaline degeneration forming broad bands of homogenous material (Figure 4B, inset). In some areas, fibrosis may be observed. Remnants of the corpora lutea from earlier cycles (before the previous one) may also still be present, visible as large hyaline areas with occasionally some remnants of luteal cells (Figure 4C). Atretic follicles (possibly from the former cycle) contain hyaline material as well. In many cases, remnants of granulosa cells and oocytes can still be recognized (Figure 4C, inset).
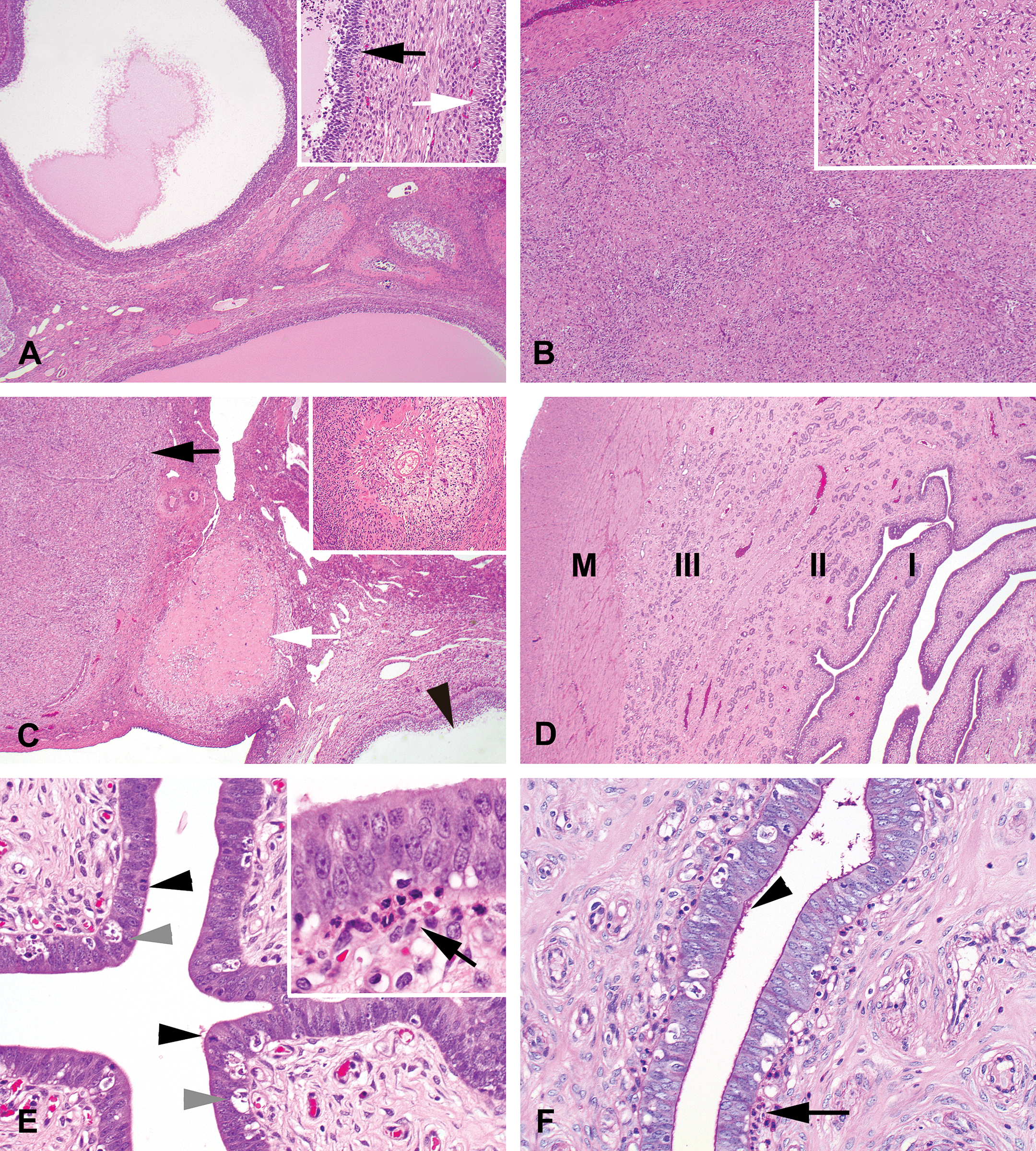
Photomicrographs of ovaries and uterus in the follicular/proliferation phase (OM). (A) Ovary in the follicular stage with fluid-filled large Graafian follicles (hematoxylin/eosin [H&E]; OM = 2×). Inset: section through part of 2 tertiary (Graafian) follicles surrounded by 5 to 6 layers of granulosa cells, 1 follicle shows atresia (black arrow), the other does not (white arrow; H&E; OM = 20×). (B) Corpus luteum from the previous cycle consisting of luteinized cells embedded in eosinophilic material (H&E; OM = 4×). Inset: high magnification of the bands of hyaline material surrounding the enclosed luteal cells (H&E; OM = 20×). (C) Ovary section showing a part of a Graafian follicle (arrowhead), a corpus luteum from the previous cycle (black arrow) and from the cycle preceding the previous cycle (white arrow; H&E; OM = 2×). Inset: Degenerating atretic follicle with hyaline material and remnants of granulosa cells and oocyte (H&E; OM = 10×). (D) Overview of a uterus section in the proliferative stage of the cycle. Zone I (the luminal epithelium) is hardly invaginated (H&E; I, zone I; II, zone II; III, zone III; M = myometrium; OM = 2×). (E) High magnification of the uterus with high luminal epithelium, abundant apoptotic bodies (gray arrowheads), and mitotic figures (black arrowheads; H&E; OM = 20×). Inset: eosinophilic granulocytes underneath the luminal epithelium (black arrow; H&E; OM = 40×). (F) Luminal epithelium of the uterus with subepithelial eosinophilic granulocytes (arrow) and thin layer of PAS-positive material on the luminal epithelial surface (arrowhead, PAS/hematoxylin staining; OM = 20×). PAS = periodic acid–Schiff; OM = original objective lens magnification.
Uterus
In the proliferation phase, the uterus has a relatively small lumen, and invaginations are not, or hardly, visible (Figure 4D). The height of the luminal epithelium is medium-sized (cuboidal) and it contains a marked number of apoptotic and mitotic cells (Figure 4E). Just underneath the luminal epithelium, a layer of eosinophilic granulocytes are present (Figure 4E and F). These granulocytes should not be confused with the larger cells containing many eosinophilic granules that are abundantly present in the stroma of the mid-secretory (MS) uterus (Figure 5G). On the surface of the luminal epithelium, a thin layer of PAS-positive staining can be observed (Figure 4F). Although it is unclear what the content of this PAS-positive material is, the staining seems to be consistently present in the proliferation phase and may therefore be useful as stage-distinguishing feature. The glands in zone II and zone III are similar, although the basal glands have a slightly lower epithelium. Glands in both zones are small and show apoptotic cells (less than in zone I, the luminal epithelium). The only difference between the glands of zones II and III is the presence of some mitotic epithelial cells in zone II.
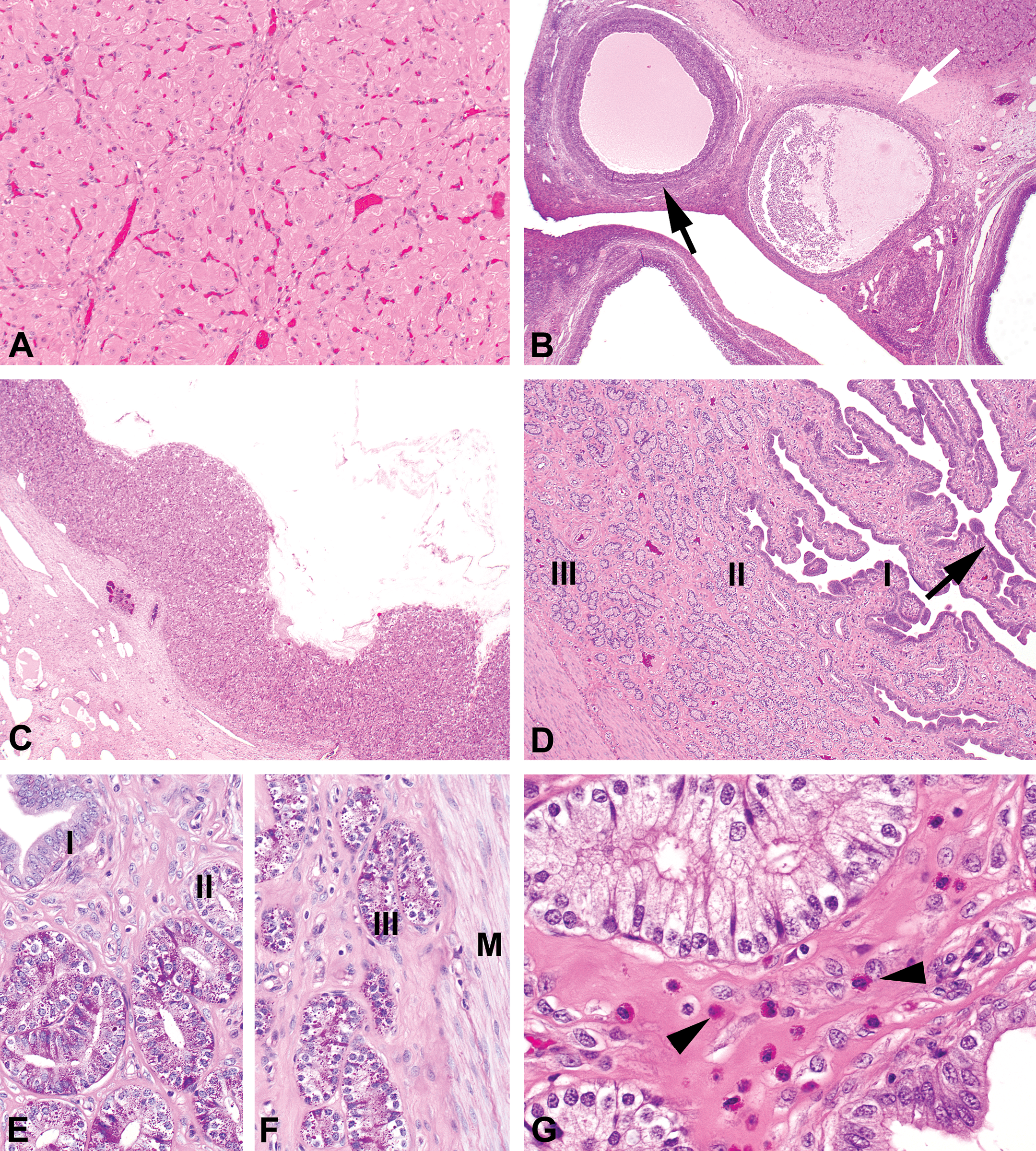
Photomicrograph of ovaries and uterus in the mid-luteal/mid-secretory phase (OM). (A) Large mature corpus luteum in an ovary in mid-luteal phase with luteal cells containing abundant eosinophilic cytoplasm (hematoxylin/eosin [H&E]; OM = 10×). (B) Tertiary follicle in an ovary in mid-luteal phase (black arrow), atretic tertiary follicle (white arrow; H&E; OM = 2×). (C) A luteinized unruptured follicle-like structure in an ovary in mid-luteal phase (H&E; OM = 2×). (D) Uterus in the mid-secretory phase with maximal invagination of the luminal epithelium (zone I, arrow) and the abundant glands in zones II and III (H&E; OM = 2×). (E) Hypertrophic luminal and glandular epithelium with abundant PAS-positive material of an uterus in mid-secretory phase of zones I and II (PAS/hematoxylin staining; OM = 20×) and (F) of zone III (PAS/hematoxylin staining; M = myometrium; OM = 20×). (G) Endometrial lymphocytes in the stroma of an uterus in mid-secretory phase (arrowheads; H&E; OM = 40×). PAS = periodic acid–Schiff; OM = original objective lens magnification.
Ovulation
Ovaries
Ovaries at ovulation, or just thereafter, can easily be recognized by the recently collapsed Graafian follicles. The oocyte has been released and during this process some small vessels are damaged and fresh blood can be seen in the disrupted follicles (Figure 6A). Corpora lutea from the previous cycle are similar to those of the follicular phase, but the fibrotic areas in the center of some of the corpora lutea may be more prominent, indicating the ongoing regression (Figure 6B). These structures end up as the so-called corpora albicans. These corpora albicans entirely consist of hyaline material without any clear remnants of corpora lutea cells and take another cycle to fully develop (see also Figure 4C). Most other tertiary follicles are atretic.
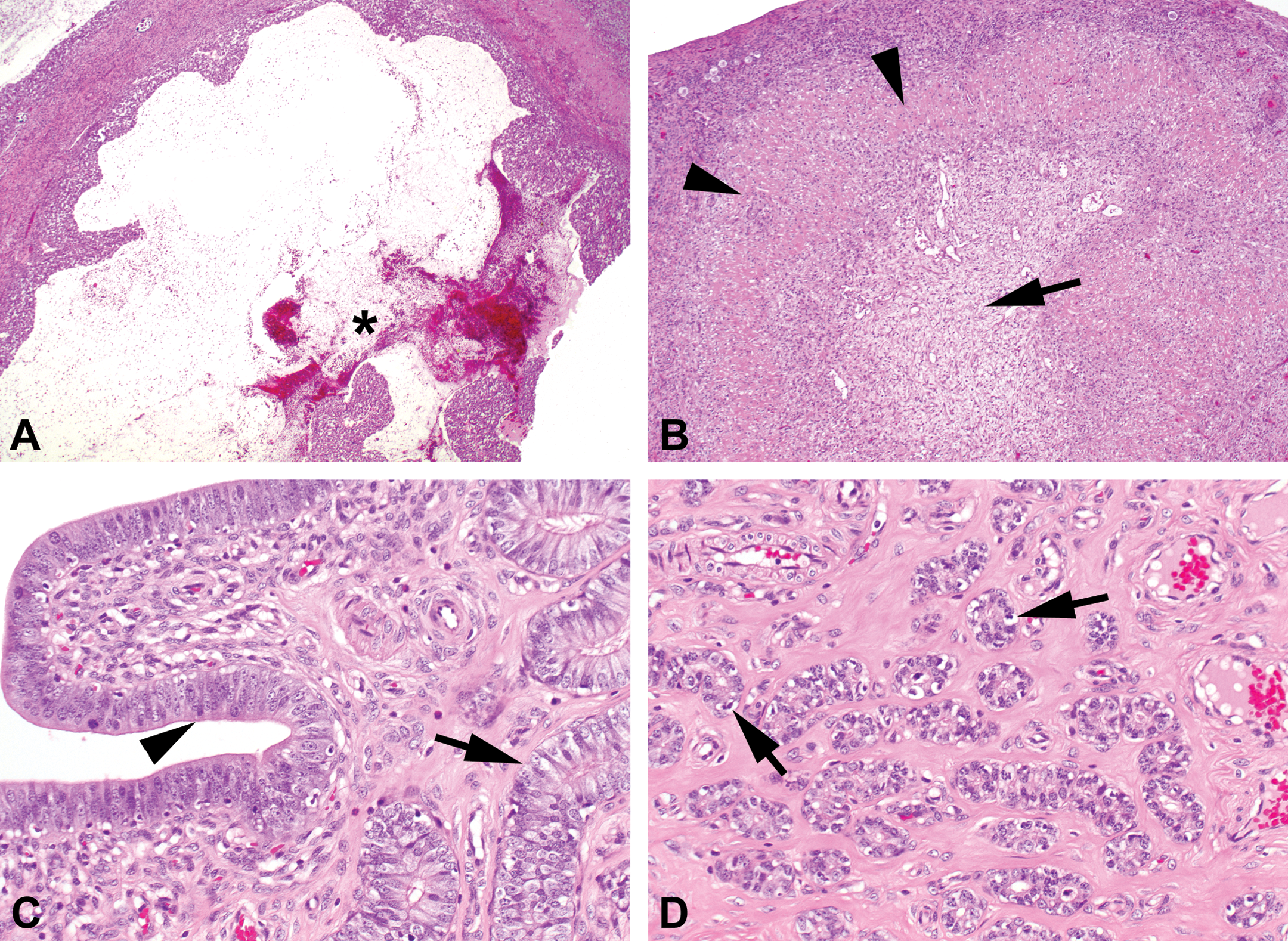
Photomicrograph of ovaries and uterus at or just after ovulation (OM). (A) Disrupted Graafian follicle in an ovary at or just after ovulation which is filled with blood (asterisk; hematoxylin/eosin [H&E]; OM = 2×). (B) Corpus luteum from the previous cycle in an ovary at or just after ovulation with increased hyaline material (arrow heads) and central fibrosis (arrow; H&E; OM = 4×). (C) Uterus at or just after ovulation with luminal epithelium showing pseudostratification of the nuclei (arrowhead) and glandular epithelium of zone II, with only few apoptotic bodies left and increased epithelial height (arrow; H&E; OM = 20×). (D) Uterus at or just after ovulation with glandular epithelium of zone III still containing apoptotic bodies (arrows; H&E; OM = 20×). OM = original objective lens magnification.
Uterus
At ovulation and just after ovulation, the uterus does not differ significantly from the uterus in the proliferation phase. The PAS-positive material is still present on top of the luminal epithelium and apoptotic figures can still be abundant. At a slightly later time point, the apoptotic bodies rapidly disappear from the luminal epithelium and the nuclei become pseudostratified (Figure 6C). Mitotic figures can still be present in epithelium of all layers. Apoptosis in the epithelium is disappearing rapidly in the glands of layer II just after ovulation, but in the glands of layer III apoptosis may still be observed (Figure 6D).
EL/ES Phase
Ovaries
The main feature in the EL phase is the presence of large corpora lutea. Corpora lutea are almost, but not completely, closed and some empty spaces and blood cells can still be found in the center (Figure 7A). Luteinized cells contain abundant eosinophilic cytoplasm and large nuclei with prominent nucleoli (Figure 7A, inset). Corpora lutea from previous cycles are hyaline structures, sometimes with remnants of degenerating luteal cells (Figure 7B). The hyalinization is much more prominent than in the previous phases. Ovaries in the EL phase contain small tertiary follicles.
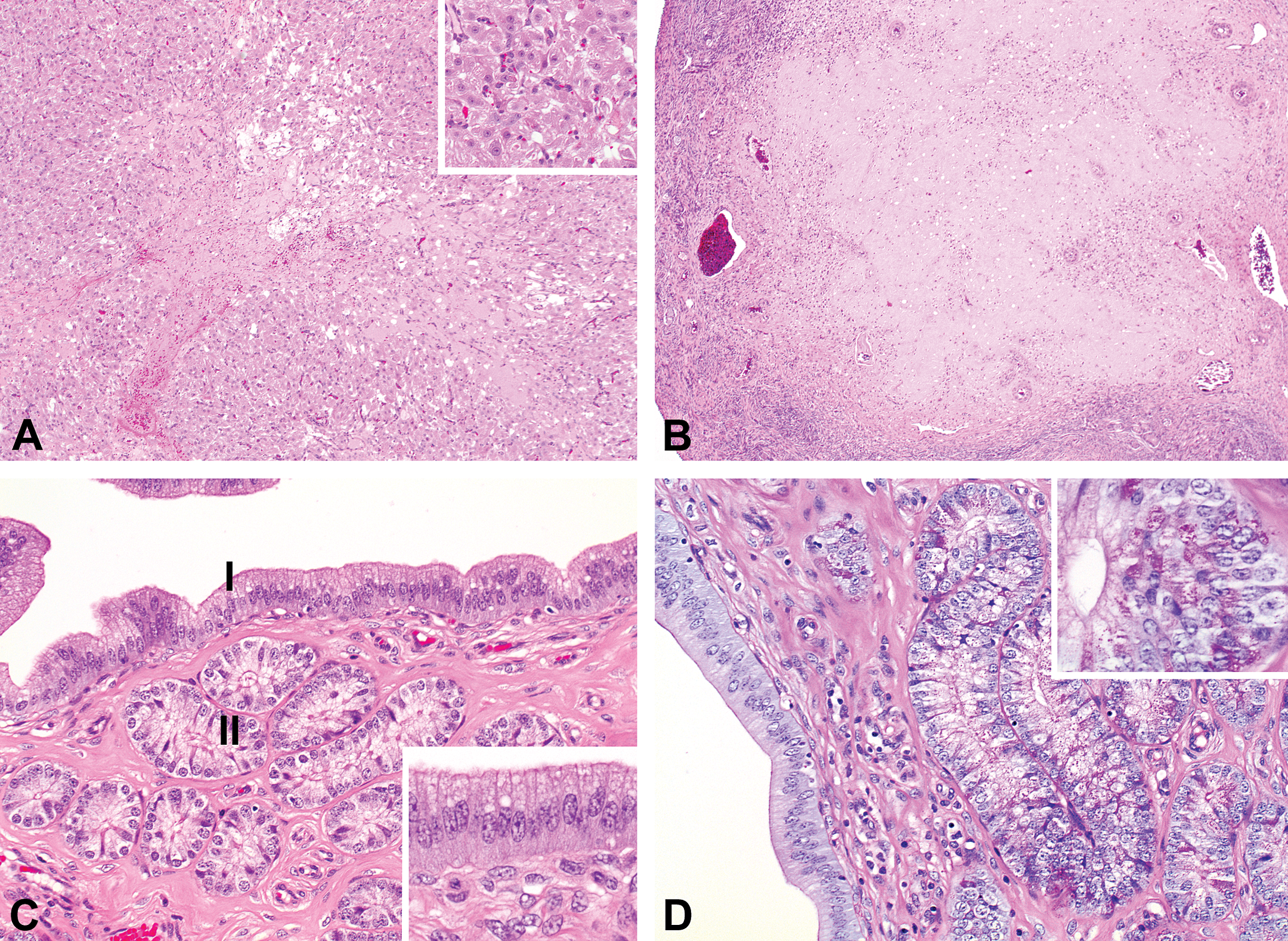
Photomicrograph of ovaries and uterus in the early-luteal/early-secretory phase (OM). (A) Large corpus luteum in an ovary in the early-luteal phase. The central space is not completely closed and some remaining blood is present (hematoxylin/eosin [H&E]; OM = 4×). Inset: luteinized cells with eosinophilic cytoplasm (H&E; OM = 10×). (B) Corpus luteum from the previous cycle in the early-luteal phase containing abundant hyaline material (H&E; OM = 4×). (C) Uterus in the early-secretory phase with hypertrophic epithelium in zones I and II. Nuclei of the luminal epithelium are centrally/basally orientated and cytoplasm is foamy (H&E; OM = 20×). Inset: luminal epithelium (H&E; OM = 40×). (D) In the early-secretory uterus, the PAS-positive material on top of the luminal surface is no longer present (OM = 20×). Inset: small dots of PAS-positive material are present in the cytoplasm of the glandular cells (PAS/hematoxylin staining; OM = 40×). PAS = periodic acid–Schiff; OM = original objective lens magnification.
Uterus
In the ES phase of the uterus (representing the period of increasing progesterone levels shown in Figure 1), the epithelia in the different zones (zones I, II, and III) are clearly distinguishable. Luminal epithelium is maximally invaginated and the epithelial cells are columnar. At a low magnification, it is easy to distinguish the higher epithelial cells of zone II glands from those of zone III. The PAS-positive material on top of the luminal epithelium has disappeared (Figure 7D). In contrast to the period around ovulation, the nuclei are not pseudostratified anymore (Figure 7C), but are arranged centrally/basally within the cells and the cytoplasm contains foamy material particularly at the luminal side (Figure 7C, inset). In all zones, epithelial apoptosis and mitotic figures are rare. Zone II glands are hypertrophic and the glandular cells are higher than the more basally located glands of zone III. In the PAS-stained slides, fine granular dots of PAS-positive material may be visible in both zone II and zone III glands at high magnification (Figure 7D, inset).
ML/MS Phase
Ovaries
At the ML phase (corresponding to the top of the progesterone peak in Figure 1), corpora lutea are fully mature and are filled with luteinized cells containing abundant eosinophilic cytoplasm (Figure 5A). Corpora lutea from the previous cycles are consisting mainly of hyaline material, cells trapped in the hyaline material can barely be recognized as luteinized cells. Small tertiary follicles are present in this phase (Figure 5B). Some structures consist of abundant hyaline material and remnants of granulosa cells and oocytes (Figure 4C, inset) and represent advanced degeneration of follicles. Corpora lutea–like structures with cavities may be present centrally in the ovaries. It is difficult to determine whether these luteinized structures are normal corpora lutea that are still not closed, or nonatretic Graafian follicles of which the oocyte was not released and the follicular cells have been luteinized (Figure 5C). The content and size of these structures are similar to corpora lutea but in contrast to corpora lutea they contain a central well-circumscribed lumen. They are described as luteinized unruptured follicles (LUFs) and are also mentioned later in the Discussion section.
Uterus
In the MS phase in the uterus, the luminal epithelium (zone I) is maximally invaginated and easily distinguishable from the glandular epithelium of the zones II and III (Figure 5D). The zone I epithelial cells are cuboidal to columnar and the nucleus of each cell are located in the center of the cell, giving the epithelium a very regular appearance. In contrast, glandular cells in zones II and III are hypertrophic and secretory material is abundantly present as can easily be observed in the PAS-stained slides (Figure 5E, F). Within the glandular stroma (particularly of zone II), many endometrial lymphocytes can be found (Figure 5G). Endometrial lymphocytes that have been described in the uterus of many species, including humans (van Esch et al. 2008; Manaster et al. 2008; Peel 1989). Other names have also been used for these cells, for example, granulated metrial gland cells in rat (Peel 1989) and uterine natural killer cells in primates (Croy et al. 2003). Neither apoptotic nor mitotic epithelial cells are observed at this stage.
LL/LS Phase
Ovaries
The tertiary follicles in the ovaries of the LL phase are medium to large and do not reach the diameter of the Graafian follicles at this stage (Figure 8A). Most of these follicles are not atretic. The corpora lutea are starting to degenerate in this phase, and the cytoplasm of the luteal cells decreases markedly and loses its eosinophilic character. The degenerating corpora lutea may appear similar to those of the follicular phase, but hyaline layers are less prominent.
Photomicrograph of ovaries and uterus in the late-luteal/late-secretory phase (OM). (A) Medium- to large-sized tertiary follicles and a corpus luteum slightly in regression of an ovary in late-luteal phase (H&E; OM = 2×). (B) Uterus in late-luteal phase with columnar luminal epithelium (left photograph; arrowhead) and exhausted small glands in zone II (glands on the right side of the left photograph) with inspissated material in the glands of zone III (right photograph; arrow; H&E; OM = 20×). OM = original objective lens magnification.
Uterus
In the LL phase in the uterus, when the progesterone peak is declining exponentially as shown in Figure 1, the glands contain flattened epithelium with inspissated eosinophilic material especially in the glands of zone III (Figure 8B). The luminal epithelium is comparable to the epithelium in the proliferation phase. The medium–high luminal epithelial cells in zone I contain a large number of apoptotic cells. Mitotic figures are present in the epithelium of zone I (Figure 8B) but are lacking in zones II and III. A PAS-positive layer on top of the luminal epithelium can be observed in this phase and can be visible until the ES phase.
Cervix, Vagina, and Mammary Gland
As mentioned previously, the other parts of the female reproductive system also changed during the different stages of the cycle, but differences are subtle, especially in HE-stained slides, and the variability is high. Therefore, these organs do not add much to distinguish the different stages of the cycle. The most obvious changes of the vaginal epithelium can be summarized as follows: multilayered epithelium with (1) cuboidal to ovoid cells (before the LH peak at F/P phase and before ovulation corresponds to proestrous stage according to Tortereau, Howroyd, and Lorentsen 2013; Figure 9A), (2) flattened surface cells (just before and after ovulation, described as estrous stage in Tortereau, Howroyd, and Lorentsen 2013; Figure 9B), (3) infiltration of granulocytes (EL/secretory phase) and apoptotic cells (ML/MS phase described as metestrus stage in Tortereau, Howroyd, and Lorentsen 2013; Figure 9C), and (4) possibly with apoptotic cells but without granulocytes (in the luteal/secretory phase, described as diestrous stage in Tortereau, Howroyd, and Lorentsen 2013; Figure 9D). In the young sexually mature females used in the present study, the variation of vaginal epithelium between individuals within the same cycle is rather high and therefore a reliable differentiation of cyclic stages based on vaginal tissue alone is difficult.
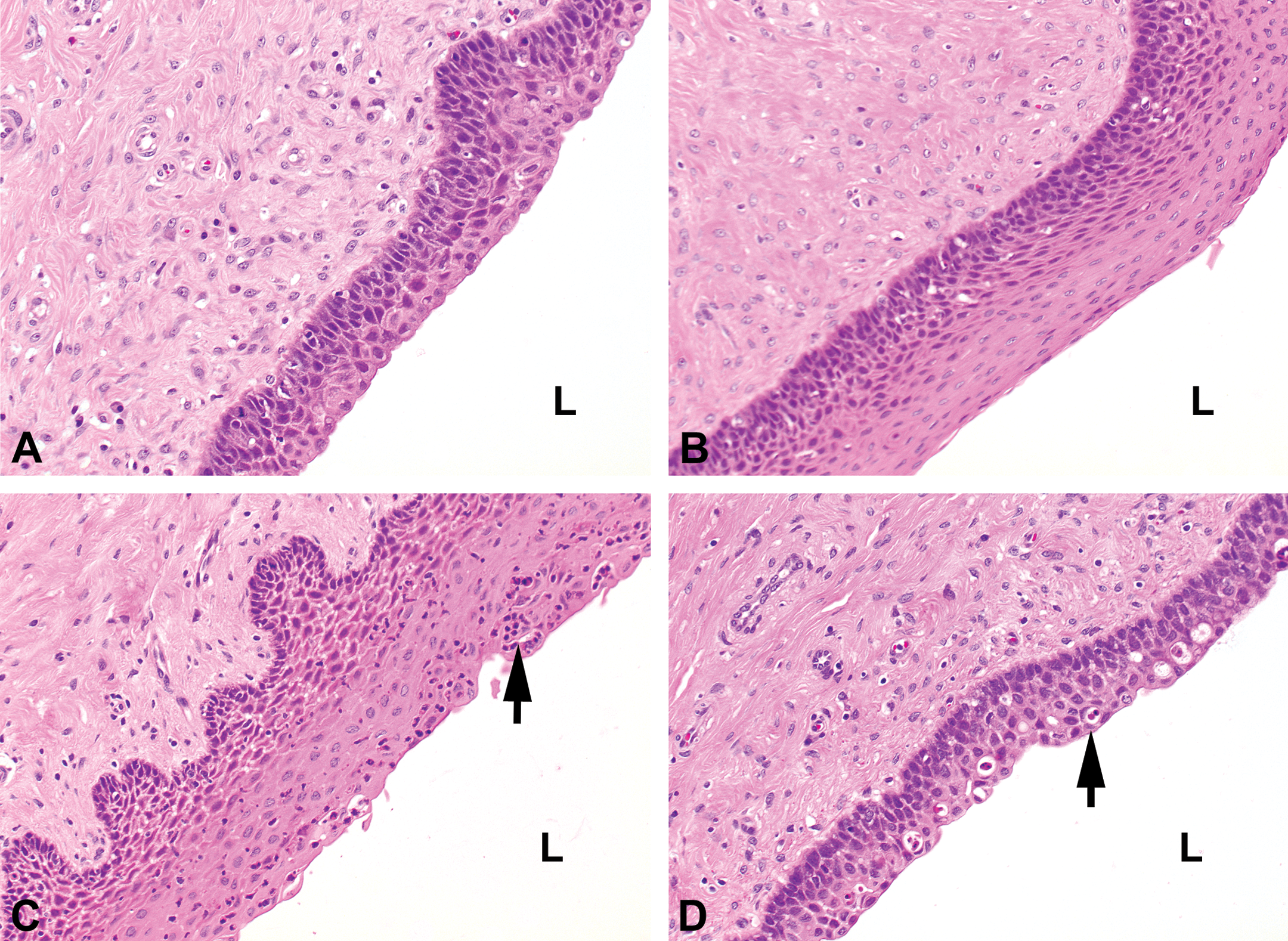
Photomicrograph of vagina in different stages of the cycle (L). (A) Vaginal section in follicular/proliferation phase with cuboidal to ovoid cells at the luminal side (hematoxylin/eosin [H&E]). (B) Vaginal section at or just after ovulation with multiple layers of epithelium and flattened cells at the surface (H&E). (C) Vaginal section at early- to mid-luteal/secretory phase with granulocytic infiltration (arrow; H&E). (D) Vaginal section at late-luteal/secretory phase with apoptotic bodies (arrow; H&E). (Original objective lens magnification 20×: A–D.) L = luminal side.
The cervix of the minipig consists of an endocervix (continuing tissue of the uterine tissue in the direction of the vagina) followed by an ectocervix, which continues into the vaginal tissue. This is comparable with humans and nonhuman primates. The ectocervix is lined by several layers of nonkeratinizing stratified squamous epithelium, which is sometimes hardly distinguishable from the vaginal epithelium (Figure 10D). The endocervix is covered by mucin-secreting, simple columnar epithelium. In the PAS staining, this mucus content is easily recognizable and is also a good marker to distinguish uterine epithelium from endocervix epithelium (Figure 10A). At the transition of endocervix to ectocervix squamous, almost nodular, areas can be found underneath the superficial mucous layer (Figure 10B). At a certain point, the mucous layer covers a continuous layer of squamous epithelium (Figure 10C and D) and the mucous cells in the layer still contain a large amount of PAS-positive material (Figure 10C). Finally, at the vaginal side, the ectocervix consists almost entirely of squamous epithelium (Figure 10D). The transitional area with the subepithelial squamous nodules should not be confused with squamous metaplasia. This latter may be a serious pathologic condition as it is in women (Park and Soslow 2009). The cervix is not changing much morphologically during the cycle, but may be important and sensitive for pathologic changes and should therefore be carefully investigated in toxicity studies (especially with hormonal compounds).
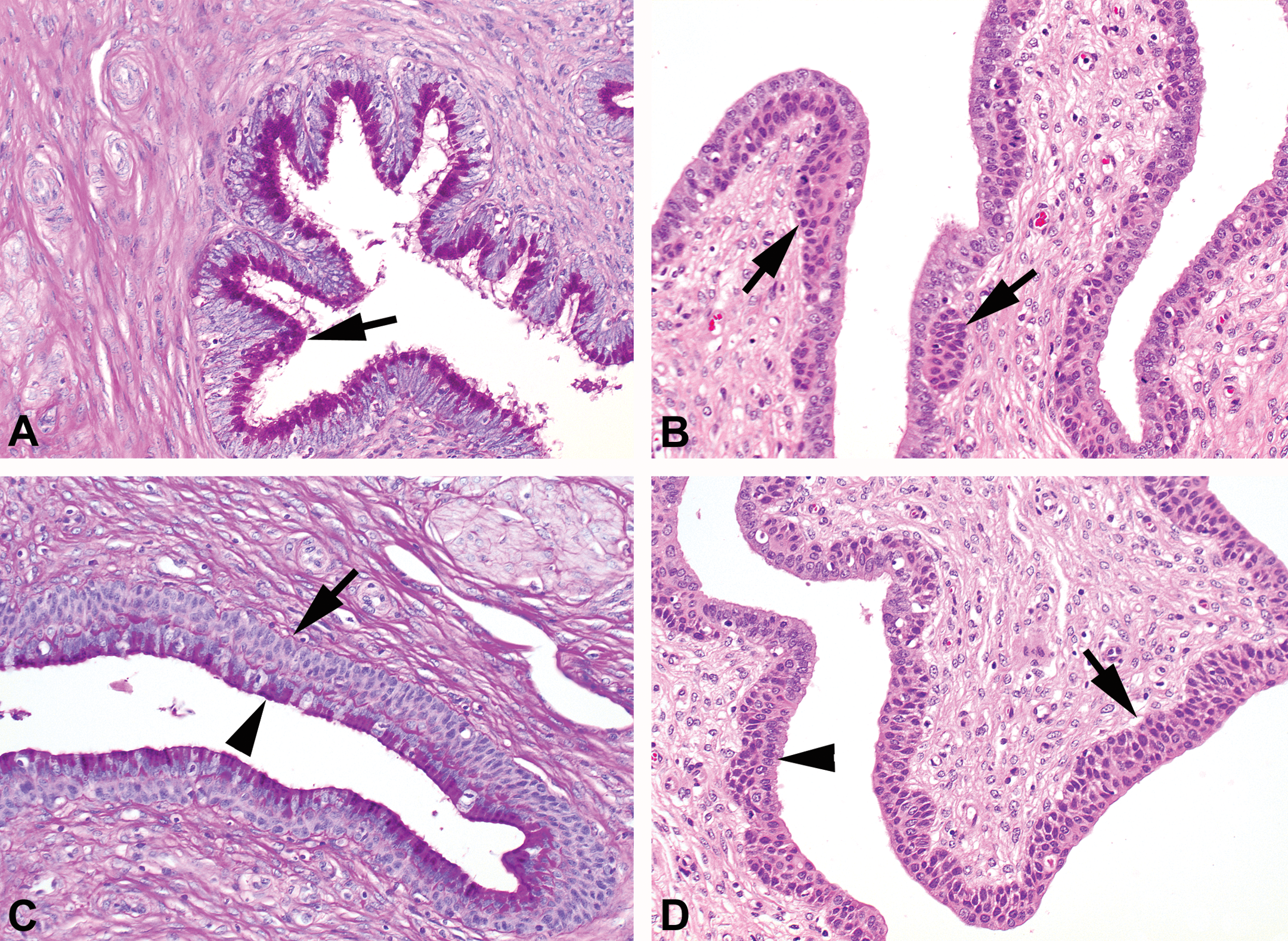
Photomicrograph of cervix in different stages of the cycle. (A) Endocervix with mucified cells producing large amounts of PAS-positive material (arrow; PAS/hematoxylin staining). (B) Transition area between endocervix and ectocervix (more toward endocervix) containing nodular squamous cell areas (arrows) that are covered with mucus-producing cells (hematoxylin/eosin [H&E]). (C) Transition area between endocervix and ectocervix (more toward ectocervix) with a continuous layer of basal squamous cells covered with mucus-producing cells (PAS/hematoxylin staining). (D) Ectocervix containing mainly squamous cells (arrow) but some simple epithelial cells may still be present (arrowhead; H&E; Original objective lens magnification 20×: A–D). PAS = periodic acid–Schiff.
Like vaginal tissue, there are large differences in mammary morphology between individuals within the same cyclic stage in young recently ovulating female pigs and is therefore not suitable for distinguishing the different cyclic stages. It seems logical that mammary gland development and changes within the mammary lobular structure mostly depend on the pregnancy/lactating status and cyclic dependency is not very clear in these young females. Different structures can be observed within the mammary gland of young mature females. The terminal end buds are small and consist mostly of 2 to 3 epithelial layers with some mitotic cells (Figure 11A). They are surrounded by loose periglandular stroma (Figure 11B). Besides terminal end buds, mammary tissue may contain small lobules, sometimes with a small amount of luminal proteinaceous material (Figure 11C). Lobular development with clustered glandular acini and open ductal lumens vary significantly among the individual females and can contain various amounts of secretory product (Figure 11C and D).
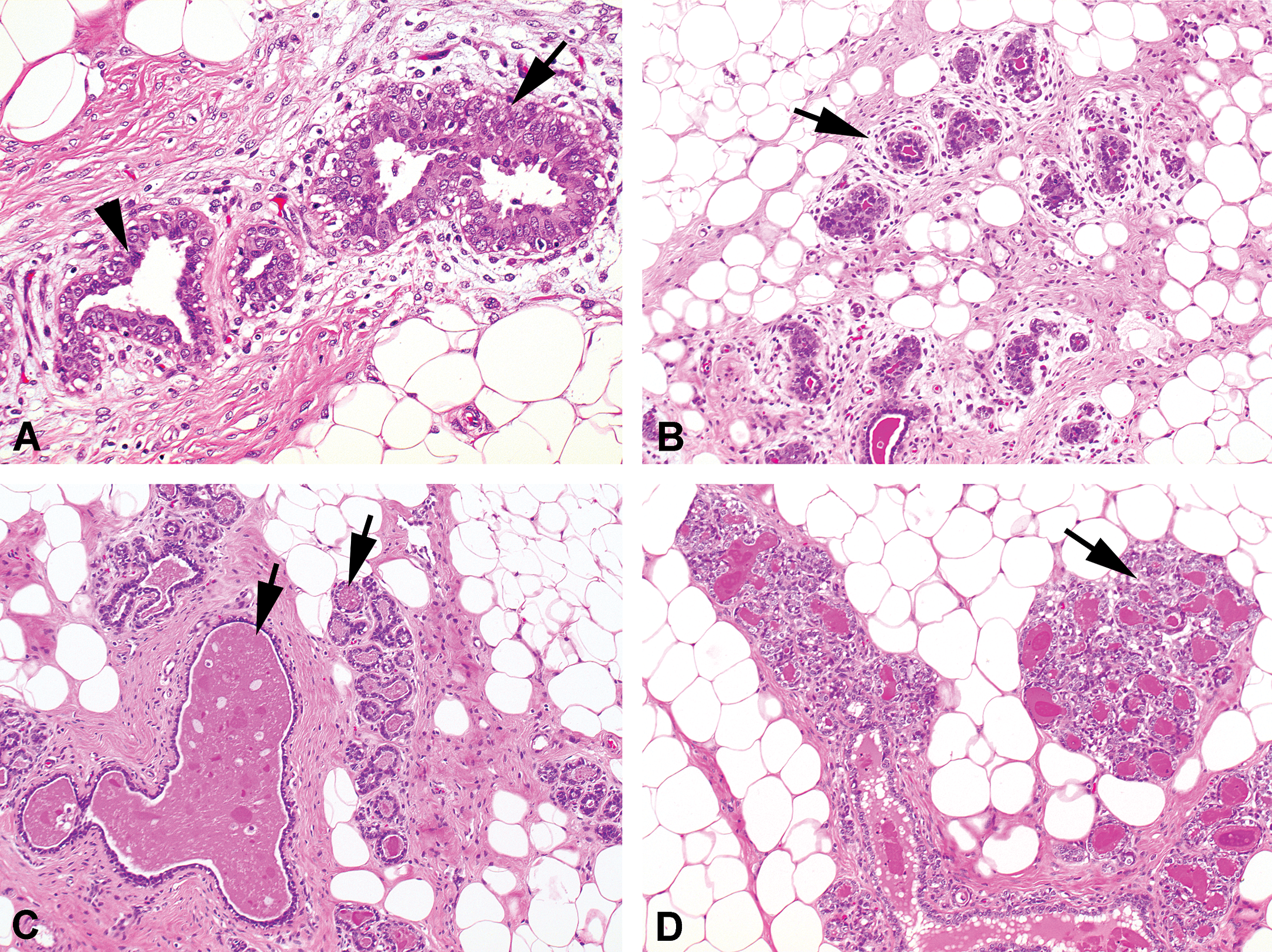
Photomicrograph of different mammary gland structures that can be found within all the different stages of the cycle. (A) Terminal end buds consisting of a few cell layers (arrow) with mitotic figures (arrowhead; hematoxylin/eosin [H&E]). (B) Terminal end buds are mostly surrounded by loose periglandular stroma (arrow; H&E). (C) Development of small lobules and both glandular and ductal secretion (arrows; H&E). (D) More prominent lobular development (arrow) with secretion (H&E; Original objective lens magnification 20×).
Absence Progesterone Peak/Immaturity
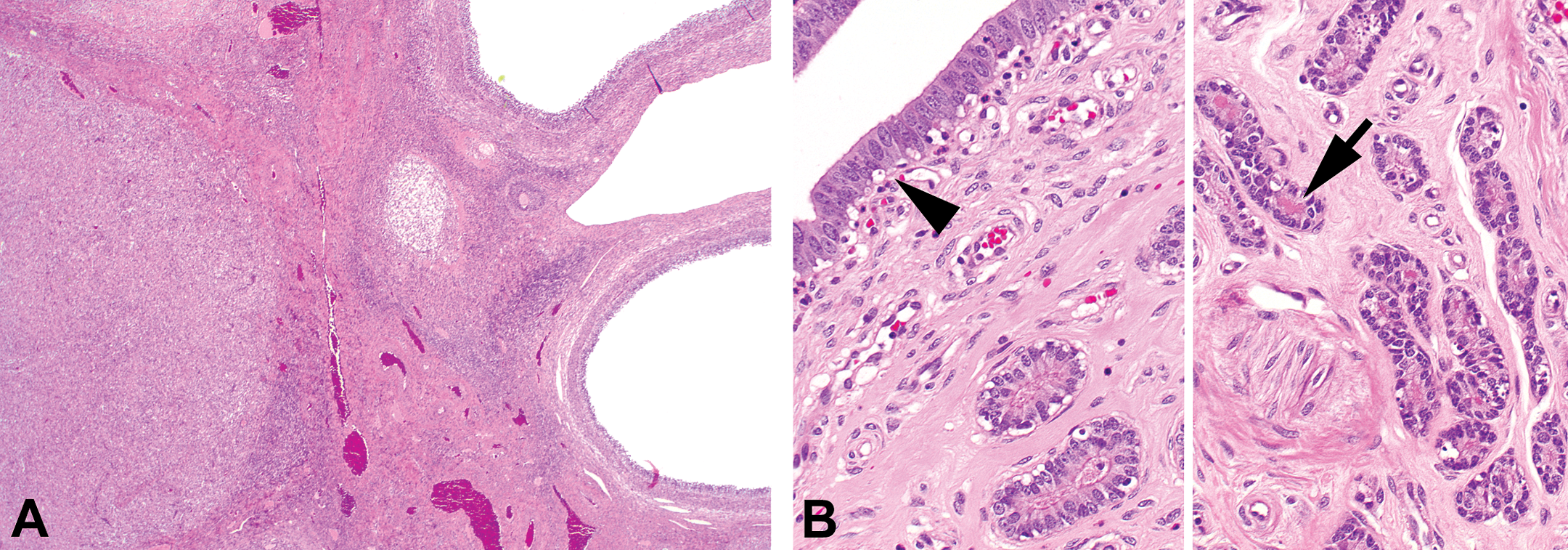
In 1 of the females, a progesterone peak was not detectable until approximately 7 months of age. In the ovaries of this animal, several follicle types were present (including tertiary); however, Graafian follicles were lacking. No mature corpora lutea, degenerating corpora lutea, or remnants of corpora lutea were visible, indicating that no previous ovulation had occurred. The uterus was very small compared to a young just ovulating female and contained a relatively low number of glands with cuboidal to columnar luminal epithelium (Figure 12A and B). The luminal epithelium of zone I showed few mitotic figures indicating ongoing development. Within the few small glands of zones II and III, no mitotic figures were observed. A considerable number of basally orientated cells with clear cytoplasm were evident (Figure 12B, inset). Although these cells look very similar to apoptotic cells, they may well represent the so-called clear cells that are believed to be precursor cells of mucous cells. In cynomolgus monkeys, these “clear cells” are described to be most frequently present in the proliferation phase of the cycle (van Esch et al. 2008). In this particular minipig, the vaginal epithelium was a few layers thick, which is also an indication that the animal was not completely immature and some development has already been started(Tortereau, Howroyd, and Lorentsen 2013). The cervix was morphologically not markedly different from the sexually mature females, although the PAS-staining revealed no or only little PAS-positive material in the cervical mucous layer (both endocervical layer and the transition to the exocervical layer; Figure 12C and D). The mammary gland consisted of terminal end buds and no lobular structures were observed, which also points to the immaturity of this particular female.
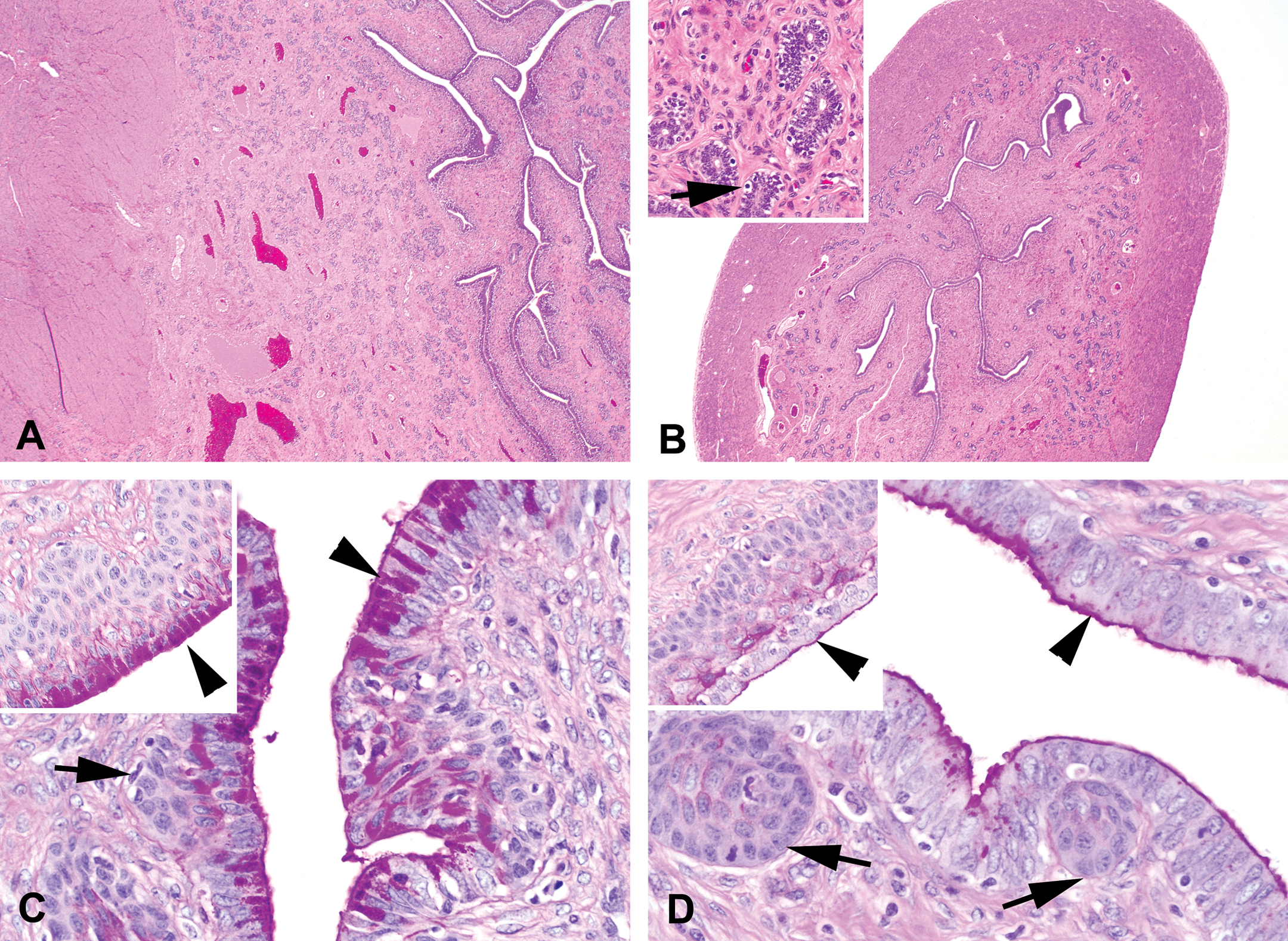
Photomicrograph of uterus and cervix of a mature (A, C) and immature (B, D) female minipig (OM = original objective lens magnification). (A) Uterus of a mature minipig in proliferation phase, which is markedly larger than the uterus of an immature minipig (B; hematoxylin/eosin [H&E]; OM = 2×). Inset: inactive basal glands in zone III with cells containing a large amount of clear cytoplasm (arrow; H&E; OM = 20×). (C) Transitional cervix of a mature female minipig showing squamous nodular areas (arrow) covered with epithelium producing abundant PAS-positive material (arrowhead). Inset: area of cervix at the vaginal side with basally a layer of squamous cells covered by a mucus-producing (PAS-positive material; arrowhead) layer (PAS/hematoxylin staining; OM = 40×). (D) Transitional cervix of an immature female minipig showing squamous nodular areas (arrow) covered with epithelium, similar to the mature females, but lacking the PAS-positive material within the cells (arrowhead). Inset: area of cervix at the vaginal side with basally a layer of squamous cells covered with an epithelial cell layer lacking intracellular PAS-positive material (arrowhead; PAS/Hematoxylin staining; OM = 40×). PAS = periodic acid–Schiff.
Discussion
In the present overview of the reproductive organs of sexually mature female minipigs, some clear distinctive histomorphologic characteristics have been described that can be used to determine the phase of the estrous cycle. These characteristics are summarized in Table 1 and are focused on changes seen in the ovary and uterus. In the other organs (cervix, vagina, and mammary gland), either the variability between animals within the same cycle stage was high or the morphology did not (or barely) change during the different phases of the cycle. In the ovaries, the large Graafian follicles (disrupted or not disrupted) and corpora lutea of both the recent and preceding cycles (size, cell morphology, and structural composition) distinguishes the follicular phase from the ES-, ML-, and LL phases. The presence of corpora lutea of the previous cycle and remnants of corpora lutea of the cycle preceding the previous cycle (corpora albicans) make it possible to draw the conclusion that the females experienced at least 2 estrous cycles. Although this was expected since 2 progesterone peaks were measured in most animals that were scheduled for necropsy, histological confirmation further confirms proper reproductive functioning. In the uterus, epithelial morphology of the different zones (zones I, II, and III; representing the luminal epithelium, functional glandular layer, and basal glandular layer, respectively), secretory activity (which can be visualized by a PAS/Hematoxylin staining), and presence or absence of epithelial mitosis and/or apoptosis are the main features to distinguish the proliferation phase from the ES-, MS-, and LS phases of the cyclic uterus. In the ovaries of the immature minipig, no corpora lutea (or regressing and/or remnants of corpora lutea) could be found. The uterus was extremely small and did not show any signs of glandular activity.
Most distinguishing phase-specific histologic characteristic in ovaries and uterus.

It has been frequently described that the cycle of the minipig is approximately 21 days in length (Bode et al. 2010; Henricks, Guthrie, and Handlin 1972; Howard et al. 1982; Jørgensen 1998; Parvizi et al. 1976). The minipigs in the present study show similar cycle lengths. The cycle consists of a relatively short F/P phase of 2 to 3 days (cf. proestrus), followed by ovulation (also described as estrous period of 2 days) and a very long luteal/secretory phase of 15 to 18 days, which can be subdivided in an ES/MS (cf. metestrus) and LL/LS phase (cf. diestrus). This classification is mainly based on the progesterone concentration curve that increased markedly in the early phase, fell back to submaximal levels in the mid-phase and rapidly decreased at the end of the curve in the late phase. The mean maximum progesterone concentration measured was approximately 50 ng/ml. Both the course of the curve and the progesterone peak levels in the present study were comparable to the curves and maximal progesterone concentrations during minipig estrous cycles published by others (Henricks, Guthrie, and Handlin 1972; Tellmann 2008; Parvizi et al. 1976). This confirms the validity of the progesterone assay used in the present study (Immulite 1000 and human serum control samples).
The cyclic curves of the different hormone concentrations in the blood of minipigs are comparable to humans and other laboratory animals such as rat, mouse, dog, and nonhuman primate. The main difference is a very short F/P phase (or proestrus) with a corresponding short and relatively low rise in estrogen concentration. The luteal phase is much longer and is comparable to the length of the luteal/secretory phase of other species (Buse et al. 2008; Rehm, Stanislaus, and Williams 2007; Staley and Scharfman 2005). In contrast to humans and nonhuman primates, but in parallel with rodents and dogs, minipigs do not have a menstrual phase. Instead, the uterus dynamics of proliferation and degeneration depends on strictly regulated cellular processes like mitosis and apoptosis and not on shedding large parts of uterus tissue as occurs during the menstrual phase of humans and nonhuman primates. In most animals, stromal tissue changes are observed during the cycle, mostly by an edematous response during the proestrus/estrus phase. This stromal tissue change is considered to be a stage-specific feature for several animals (Buse et al. 2008; Rehm, Stanislaus, and Williams 2007), but this response is very limited in the minipig uterus.
From the observed luteinized structures with central lumen in the ovaries of some female minipigs in the ML phase, it is unclear whether they are nonclosed corpora lutea or LUFs. To confirm the presence of LUFs, the oocyte should be present within the structures and to evaluate this possibility further, serial sections will be needed. The presence of LUFs in ovaries is described as a spontaneous lesion in dogs (Sato et al. 2012) and can be induced by hormonal manipulation in rats and monkeys (Lagemaat et al. 2011). However, data on spontaneous occurring incidences of LUFs in the minipig are lacking.
The increasing use of minipigs in preclinical testing programs as a nonrodent alternative for dogs and nonhuman primates increases the need for a detailed description of the female reproductive organs during the normal estrous cycle. In most toxicity studies, young females are used and many of them appear to be sexually immature or peripubertal; only a few of the females within toxicity studies appear to be sexually mature. With the presented morphological descriptions of recently ovulating female minipigs (necropsy after 2 cycles determined by the measurements of 2 progesterone peaks), we attempt to give a guidance for cyclic staging of young mature animals and recognition of sexually immature or peripubertal female animals. As the best practice for cyclic staging in the female Göttingen minipig, we would recommend to examine sections of ovaries and uterus that are routinely fixed in 10% buffered formalin and stained with H&E (both ovaries and uterus) and periodic acid–Schiff/hematoxylin (uterus only). This would facilitate the evaluation of female minipig reproductive function in preclinical toxicity studies.
Footnotes
Acknowledgments
The authors would like to thank Mr. A. Zeltner and Mrs. W. Verhoef for their excellent assistance at the necropsies and histotechnique.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
