Abstract
In the literature, experimental data on sexual maturation of female Göttingen minipigs are lacking. This may impede a reliable evaluation of reproductive functioning, particularly in the young (immature) sow used in toxicity studies. To find suitable method(s) to detect ovulation during in-life, a pilot study was performed with 3 adult sows (approximately 10–11 months), followed by a study with 14 immature females (approximately 3–4 months). From the tested parameters, progesterone analysis was the most reliable predictor. First progesterone peaks were observed in 13 sows at 3.7–4.2 or 5.5–6.5 months with a cycle length of 17–22 days. One sow did not show progesterone release until necropsy at 7 months of age. Histopathology of the reproductive organs confirmed sexual maturity for all sows, except the one without progesterone peak. In conclusion, the age range of sexual maturity of female Göttingen minipigs (3.7–6.5 months) is much wider than previously thought, and in-life progesterone analysis is a useful tool to determine sexual maturity of individual animals.
Keywords
The emergence of biologicals poses new challenges to preclinical safety testing in animals. To obtain valuable information on pharmacological and toxicological effects of a drug candidate for subsequent clinical development in man, the commonly used species (mice, rats, rabbits, and dogs) may not always be appropriate. The minipig could be a good alternative as its anatomy and physiology are very similar to those of humans in many aspects (Jørgensen 1998; Bode et al. 2010; McAnulty et al. 2012). In regulatory toxicology studies, female Göttingen minipigs are typically 4–5 months of age at the start of experimental studies. At this age, they are expected to be sexually mature based on breeding experience (Jørgensen 1998; Bode et al. 2010; Creasy 2012). However, experimental data on the onset of sexual maturation in female Göttingen minipigs are lacking in the literature and are required for a reliable evaluation of reproductive functioning. To elucidate the timing of sexual maturation in female Göttingen minipigs, 2 separate studies were performed. The objective of the initial study was to find useful method(s) to detect ovulation in sexually mature female minipigs. Subsequently, a more extensive study was performed to correlate in-life observations with data from morphologic examination of the female reproductive organs and to define the age when sexual maturity is reached.
Female minipigs from Ellegaard Göttingen Minipigs A/S (outbred and microbiologically defined) were used. They were group housed in connected pens in the same room as adult boars, resulting in olfactory, auditory, and (limited) contact stimuli from the males. For the pilot study, 3 naive adult female Göttingen minipigs (approximately 10–11 months) were used. In the main study, a total of 14 naive, immature female Göttingen minipigs (approximately 3–4 months), provided in 2 cohorts, were used: one group was born during the period of August 30 to September 13, 2013, and the other group during the period of November 16–23, 2013. Further details on housing conditions were described previously (De Rijk et al. 2014). The study protocol was reviewed and agreed upon by the Laboratory Animal Welfare Officer and the Ethical Committee of WIL Research as required by the Dutch Act on Animal Experimentation (February 1997). The following measurements (given in chronological order) were performed in the morning between approximately 11:00 a.m. and 12:00 noon. Pilot study involved (1) daily home cage observations for signs of heat including increased restlessness accompanied by characteristic grunts, frequent urination, ear straightening, tendency to “rub,” tendency to “mount” cage mates, immobility following firm pressure on the back, and characteristic changes in the vulva; (2) daily recording of rectal temperature; and (3) cytology of daily vaginal smears as described for rodents previously (Cora, Kooistra, and Travlos 2015). Pilot and main study involved (4) recording of body weights at regular intervals; (5) collection of blood samples for hormone measurements twice weekly (serum was stored at ≤−75°C until hormone analyses). Concentrations of progesterone and/or 17β-estradiol (pilot study only) were determined using a sequential- and a solid-phase competitive immunoassay, respectively, for use on the Immulite 1000 (Siemens Medical Solution Diagnostics B.V., Den Haag, The Netherlands). After necropsy at different time points in the estrous cycle based on the previous progesterone peak, tissues from the reproductive tract (including the ovaries, uterus, cervix, and vagina) and mammary glands were collected and processed for histopathology as described earlier by De Rijk et al. (2014). Both studies were descriptive, and no statistical analyses were performed.
In Figure 1, representative data (collected over 29 days) from one mature sow of the pilot study are shown. Results of vaginal cytology and rectal temperature did not reveal a clear pattern, neither did observations for estrus-like behavior and appearance. Progesterone analysis revealed a distinct peak from day 8 to 26. For 17β-estradiol, a rise in concentration was seen prior to the rise in progesterone, but also elevated concentrations were detected during the period of increased progesterone release. The age range of sexual maturity determined in 14 sows by the onset of the first progesterone peak (correlated to body weight) is depicted in Figure 2. The first peak was seen in 7/14 sows at the age of 3.7–4.2 months (8.4–12 kg) and in 6/14 sows at the age of 6.1–6.5 months (12.1–16 kg). In 1/14 sows, no progesterone peak was detected until necropsy at the age of 7 months. After the first progesterone peak, hormone analysis was continued until completion of at least 1 entire cycle of release. A schematic drawing of such progesterone curve is given by De Rijk et al. (2014). All animals (except the one without progesterone release) showed regular cycles of 17–22 days. To confirm that the first progesterone peak was directly correlated to the onset of sexual maturity, morphologic examination of the reproductive organs (in particular the ovaries) was needed. In Figure 3, ovaries of females that were sacrificed at different stages of the estrous cycle are shown. Mature minipig ovaries have large Graafian follicles and all stages of corpora lutea and degenerating stages of corpora lutea. The order of development and regression follows the alphabetic order in Figure 3 from “a” to “i.” Large Graafian follicles are found in the follicular phase of the cycle (Figure 3a), and after the surge of the luteinizing hormone (LH) the oocytes are released from these follicles at ovulation. The recently ruptured follicles collapse and contain blood as a consequence of the rupture (Figure 3b). In the luteal phase, the small follicles develop into a Graafian follicle for the next cycle, and the collapsed follicle develops into a corpus luteum with very active progesterone-producing luteinized cells (Figure 3c). At the end of the luteal phase, the corpora lutea start to regress, and in the next follicular phase the corpus luteum consists of small luteinized cells surrounded by spindle-shaped fibroblasts (Figure 3d). This process of regression with increasing fibrous tissue and fewer and smaller degenerating luteinized cells continues (Figure 3e and f), and in the follicular phase of the third (or greater) cycle, the majority of the corpus luteum consists of a mass of hyaline material (Figure 3g). The regressing corpus luteum becomes smaller and during ovulation of the third cycle (or greater) almost all luteinized cells have disappeared (Figure 3h), finally ending in the third (or greater) luteal phase in a small hyaline structure (corpus albicans; Figure 3i). In contrast, the ovaries of the immature minipig (the one without a progesterone peak) had only small follicles and no remnants of corpora lutea. The other reproductive organs were evaluated as well, and except for the small, inactive uterus of the sexually immature female minipig, all other females showed a uterus morphology that is characteristic of sexual mature animals as described in detail in De Rijk et al. (2014). Morphological evaluation of vagina and mammary gland appeared not to be useful for determination of sexual maturity.

Results of one adult female minipig (10-11 months of age) were collected during a period of 29 days. On the horizontal axis the day of study and stage of estrus as determined by vaginal cytology are depicted (P = proestrus, E = estrus, M = metestrus, D = diestrus). Different signs of heat are indicated by coloured bars. Observations for this particular sow included restlessness ( ), ear straightening (
), ear straightening ( ), rubbing (
), rubbing ( ), mounting (
), mounting ( ), immobility after pressure on the back (
), immobility after pressure on the back ( ), redness/swelling of the vulva (
), redness/swelling of the vulva ( ). Furthermore results from rectal body temperature measurement (
). Furthermore results from rectal body temperature measurement ( ), 17β-estradiol analysis (
), 17β-estradiol analysis ( ) and progesterone analysis (
) and progesterone analysis ( ) are given. On the vertical axis, rectal body temperature (in °C), progesterone (in ng/ml) and 17β-estradiol (in pg/ml) are shown.
) are given. On the vertical axis, rectal body temperature (in °C), progesterone (in ng/ml) and 17β-estradiol (in pg/ml) are shown.
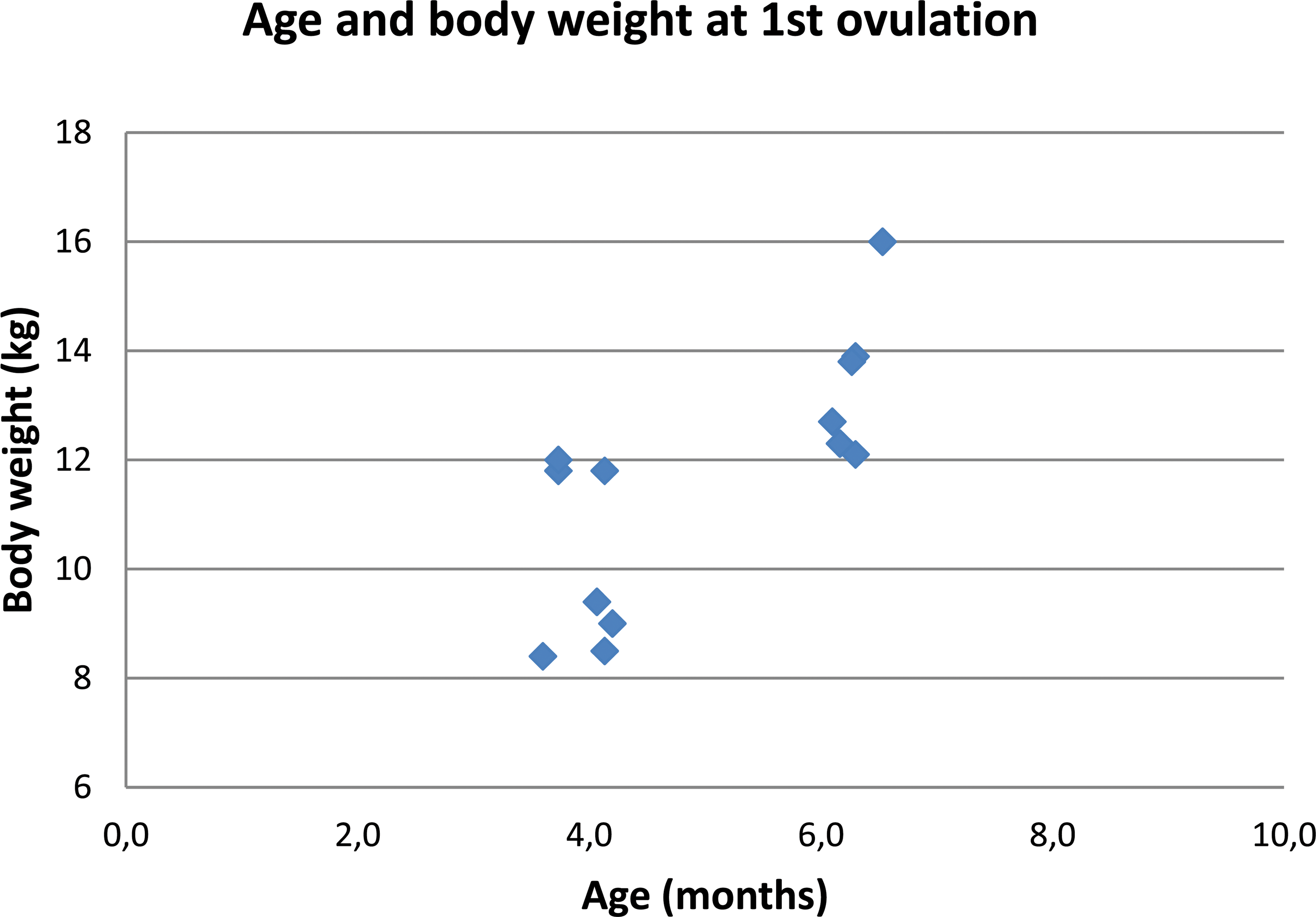
Age and body weight at first onset of progesterone release. For each of the 13 sows with cyclic progesterone release, the age and body weight at start of the first progesterone peak are indicated.
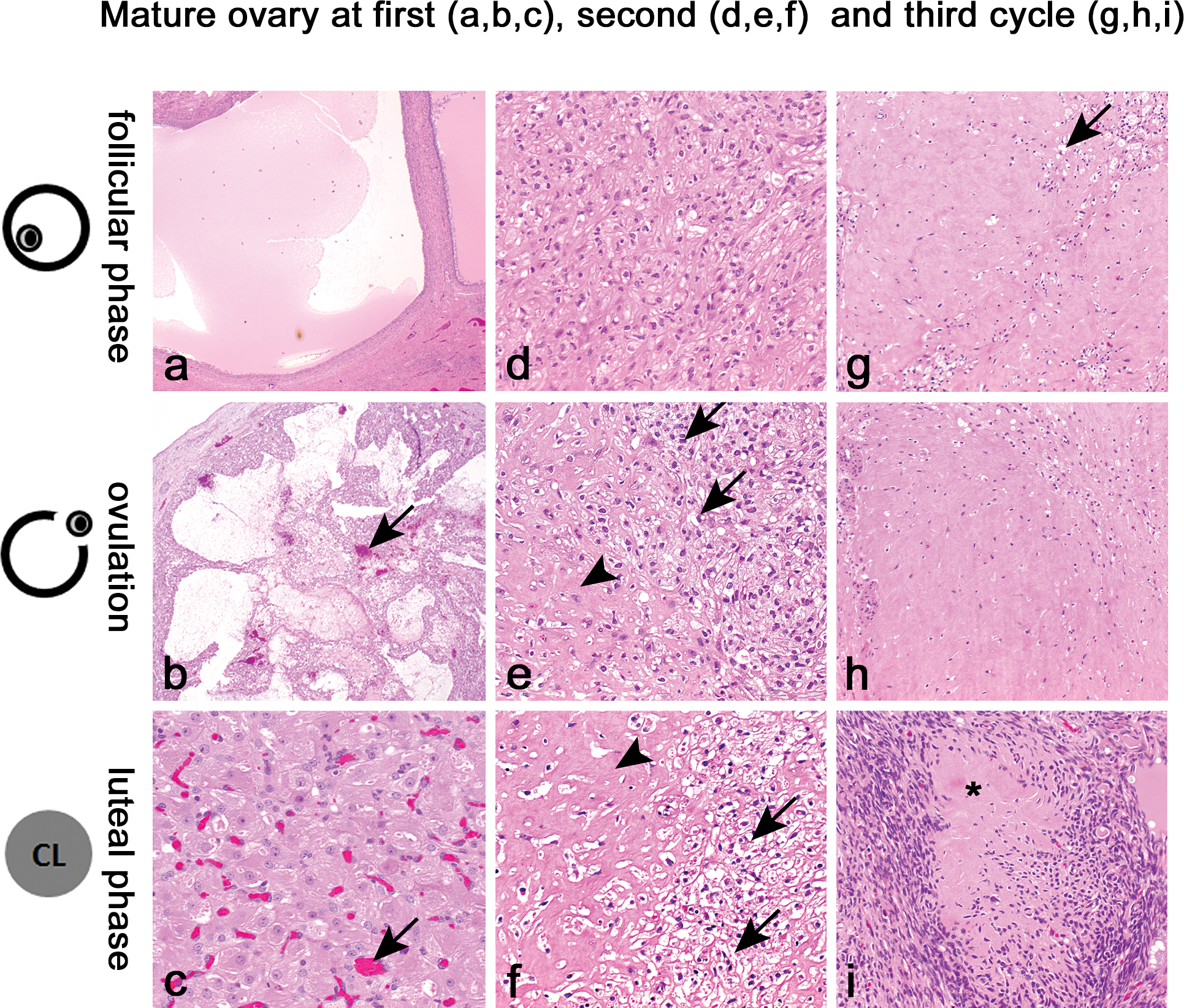
Morphologic characteristics of the ovaries of sexually mature female Göttingen minipig. Ovary in the first (a, b, and c), second (d, e, and f), and third (g, h, and i) cycle. Ovary in the follicular phase (a, d, and g), at ovulation (b, e, and h), and in the luteal phase (c, f, and i). The subsequent events of the changes from large Graafian follicle to corpus luteum and the regression of the corpus luteum are as follows: (a) Graafian follicle; (b) Ruptured follicle containing blood (arrows); (c) Mature corpus luteum, well vascularized (arrows) and containing large active cells with abundant cytoplasm; (d) Early regression of the corpus luteum with slight fibrosis; (e and f) Continuing of the regression with more and more hyaline material (arrowheads) and small degenerating luteinized cells (arrows); (g and h) Late stages of regression of corpora lutea, some luteinized cells may still be present (arrow), but the corpora lutea consist mainly of hyaline material; and (i) The final regression stage of the corpus luteum consisting of a small, complete hyaline structure (asterisk) which is also called a corpus albicans. H&E-staining.
In summary, observations for signs of heat were not useful in detecting estrus, most likely due to practical limitations in the daily routine at the animal facility. Animals cannot be observed continuously, and estrus-like behavior is more likely to occur when the animals feel undisturbed. Vaginal cytology did not provide meaningful data due to the morphologic variability of the vaginal epithelium during the different estrous stages as was described previously for the young females of the main study (De Rijk et al. 2014). This was also true for daily recording of rectal body temperature, a method described for humans to predict the time of ovulation (Pallone and Bergus 2009). As physical activity can also cause small rises in temperature, basal temperature should be taken. This, however, is not possible in a normal laboratory setting. Another potentially useful parameter for detecting ovulation is analysis of reproductive hormone(s) in serum. For animal welfare reasons, it was decided to limit blood collection to twice weekly. Consequently, 17β-estradiol and progesterone were analyzed and not LH and follicle-stimulating hormone (FSH), which need much more frequent sampling due to their relatively short presence in blood (2 days) during the cycle. In the current study, rises in 17β-estradiol were seen both prior to the rise in progesterone (i.e., coinciding with onset of estrus) and coinciding with the progesterone peak. This is in line with previous pig studies (Henricks, Guthrie, and Handlin 1972). As such, progesterone, which is released only once during the cycle, appeared to be a more useful predictor of estrus. The obtained progesterone data indicated regular cycles of approximately 17–22 days. This is in line with previously published data (Howard et al. 1982). The cycle consists of 3 phases: follicular/proliferation phase with low progesterone levels, ovulation with progesterone levels that start to increase, and luteal/secretory phase with high progesterone levels that drop sharply at the end (see De Rijk et al. 2014). Maximum values of progesterone ranged from 21 to 85 ng/ml, which is comparable to previously published data (Henricks, Guthrie, and Handlin 1972; Parvizi et al. 1976; Tellmann 2008). The ultimate proof of sexual maturity would be offspring, but an indication for ovulation is also accepted as strong evidence. Since in the present study cycles of progesterone release were very regular from the first onset, the next peak(s) could be predicted based on the former peak(s), and necropsy could be performed at different stages of the estrous cycle. Histopathological examination of the reproductive organs confirmed sexual maturity for all sows (except one) by the presence of corpora lutea (or remnants of corpora lutea from previous cycles) in the ovaries, and ovary morphology correlated directly with the number of estrous cycles (1, 2, or 3) predicted previously by progesterone analysis. Although uterus morphology can be used for determination of sexual maturity and staging of the cycle, it cannot be used to detect earlier cycles. The morphology of other reproductive organs (mammary gland, cervix, and vagina) is highly variable in these young females and therefore not suitable for sexual maturity or for staging (De Rijk et al. 2014). In the present study, 13 of the 14 investigated females reached sexual maturity within 7 months of age, with 7 females between 3.7 and 4.2 months (8.4–12 kg), and another 6 females between 6.1 and 6.5 months (12.1–16 kg). This range was much wider than anticipated but was in line with the results of a recent retrospective study (Tortereau, Howroyd, and Lorentsen 2013). Whether the trend toward a bimodal age distribution is a real biological effect or a chance finding caused by the relatively low number of females used in the present study needs further investigation with inclusion of many more animals and a proper statistical analysis. Both a cohort difference and a genetic cause can be excluded since animals from the 2 cohorts and sister pairs could be found in both the 3.7–4.5 months and the 6.1–6.5 months group.
In conclusion, the age range at which female Göttingen minipigs reach sexual maturity (3.7–6.5 months) is much wider than previously thought, and a small percentage may even not be sexually mature at 7 months of age. In-life progesterone analysis in combination with histological staging of the estrous cycle are reliable tools for the evaluation of female reproductive function. Sexual maturation should be taken into consideration when designing toxicity studies using young sows and interpreting data hereof.
Footnotes
Acknowledgments
The authors would like to thank H. Lorentsen, DVM, for coordinating this project within Ellegaard Göttingen Minipigs A/S, and the staff of WIL Research Europe B.V. and Ellegaard Göttingen Minipigs A/S for their excellent assistance during this study.
Authors’ Contribution
Authors contributed to conception or design (BP and ED); data acquisition, analysis, or interpretation (BP, ED, AZ, and HE); and drafting the manuscript (BP and AD). All authors critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
