Abstract
Despite the use of rabbits in biomedical research, including regulatory toxicology and cardiovascular studies, little data exist on heart findings in this species. This study was designed to document myocardial findings in female rabbits and the impact of study-related procedures typical for vaccine toxicology studies. One hundred and forty 6- to 8-month-old female New Zealand White rabbits were divided equally into 2 groups, high and low study procedure groups (group 1 and group 2, respectively). All animals received intramuscular (IM) injections of sterile saline every 2 weeks for 5 times and were necropsied 2 days after the final IM injection. Clinical chemistry, hematology, and urinalysis were evaluated. Blood for stress biomarkers (norepinephrine, epinephrine, cortisol, and corticosterone), C-reactive protein, cardiac troponin I, and creatine kinase were collected at time 0 (just before dose administration) and then at 4, 24, and 48 hr after dose administration in group 1 only. Hearts were assessed histologically. Focal to multifocal minimal inflammatory cell infiltrates were common (∼80%), particularly in the left ventricle and interventricular septum, and were similar to the types of infiltrates identified in other laboratory animal species. Additionally, study-related procedures elevated serum stress biomarkers and exacerbated the frequency and severity of myocardial inflammatory cell infiltrates.
Keywords
In order to assess human risk from animal toxicity studies, it is essential to have an understanding of the range of morphological findings that occur spontaneously or are associated with study procedures within the species under examination. Rabbits have been used extensively in biomedical research, most frequently as animal models of disease, but also in nonclinical safety studies, particularly for developmental toxicity or local irritation studies. Rabbits have been a typical species for regulatory toxicology studies for vaccines (World Health Organization 2013). Despite the use of rabbits in various nonclinical studies, including as models of cardiovascular disease and physiology (Fan et al. 2015; Kang et al. 2016; Lang, Koren, and Odening 2016), there is a paucity of literature on background microscopic heart findings in rabbits. Inflammatory cell findings in rabbit hearts are well recognized within the comparative pathology community. The earliest publication on inflammatory heart lesions in otherwise healthy rabbits was in 1924 (Miller 1924), which described multifocal myocardial infiltrates of lymphocytes, macrophages, or a combination of both cell types with or without fibrosis, heterophils, plasma cells, mast cells, or degenerating cardiomyocytes (loss of striations). The locations of these findings, from greatest to least, were papillary muscle, interventricular septum, left ventricular free wall, right ventricular free wall, and atria. The lesions were described as being most common within the myocardium but were occasionally evident subendocardially or subepicardially. While the lesions were less frequent in younger rabbits and occurred in approximately 60% of the 34 rabbits evaluated, the ages were not specified. Myocardial inflammatory cell infiltrates have been reported in the rabbit heart as an incidental finding (Bradley 2012; Van Vleet and Ferrans 1986) and within Pfizer’s internal historical database.
In an effort to characterize the nature and range of microscopic findings in rabbit hearts, Pfizer designed a large study in female New Zealand White (NZW) rabbits patterned after standard vaccine studies but with either more or fewer procedures than in a typical study. Inflammatory findings were the most common diagnosis in the rabbit heart, and this article details these inflammatory findings and demonstrates that increases in the number of routine study procedures in rabbits can increase the distribution and severity of these heart findings.
Materials and Methods
Animals and Housing
One hundred and forty (140) 6- to 8-month-old female NZW rabbits (Covance Research Products, Denver, PA) were housed single in stainless steel/plastic cages (Allentown Euro Rack, Allentown, Allentown, NJ) at the Pfizer, Groton, CT animal facility. Only female rabbits were evaluated in order to optimize the power of the study in the face of limitations in study size capacity at the Pfizer, Groton, CT facility. Rabbits were fed a standard diet of pelleted food (Certified Rabbit Diet 5322; PMI Feeds, Richmond, IN) and municipal drinking water, further purified by reverse osmosis, was provided ad libidum. Rabbits were randomly divided into 2 groups of 70 animals each. Animals in group 1 were housed in rooms separate from animals assigned to group 2. This study was conducted in an Association for Assessment and Accreditation of Laboratory Animal Care–accredited institution and in accordance with the current guidelines for animal welfare (National Research Council Guide for the Care and Use of Laboratory Animals, 2011).
Study Design to Evaluate the Effect of Handling and Procedural Stress in Rabbits
This study was carried out under an approved Pfizer Institutional Animal Care and Use Protocol and designed to mimic a 5-dose vaccine toxicology study. All animals received intramuscular (IM) injections of sterile saline (1 ml per dosing day administered as bilateral 0.5 ml injections into each quadriceps muscle) on 5 occasions (once every 2 weeks). Animals were necropsied on day 59, 2 days following the last injection.
All rabbits were subjected to the same types of standard procedures performed in a typical repeat-dose toxicology study; however, the number/frequency of procedures conducted was different between animals in groups 1 and 2 (Table 1). Animals in group 1 (increased procedures) were subjected to the typical procedures performed in a standard repeat-dose toxicology study, except that additional blood samples were taken (i.e., 21 blood collections in this study vs. ∼6 in a standard study). Animals in group 2 (minimal procedures) had few procedures and only 2 blood collections in order to minimize the restraint and handling (less than in a standard study) of the animals.
Number of Scheduled Procedures per Group.a
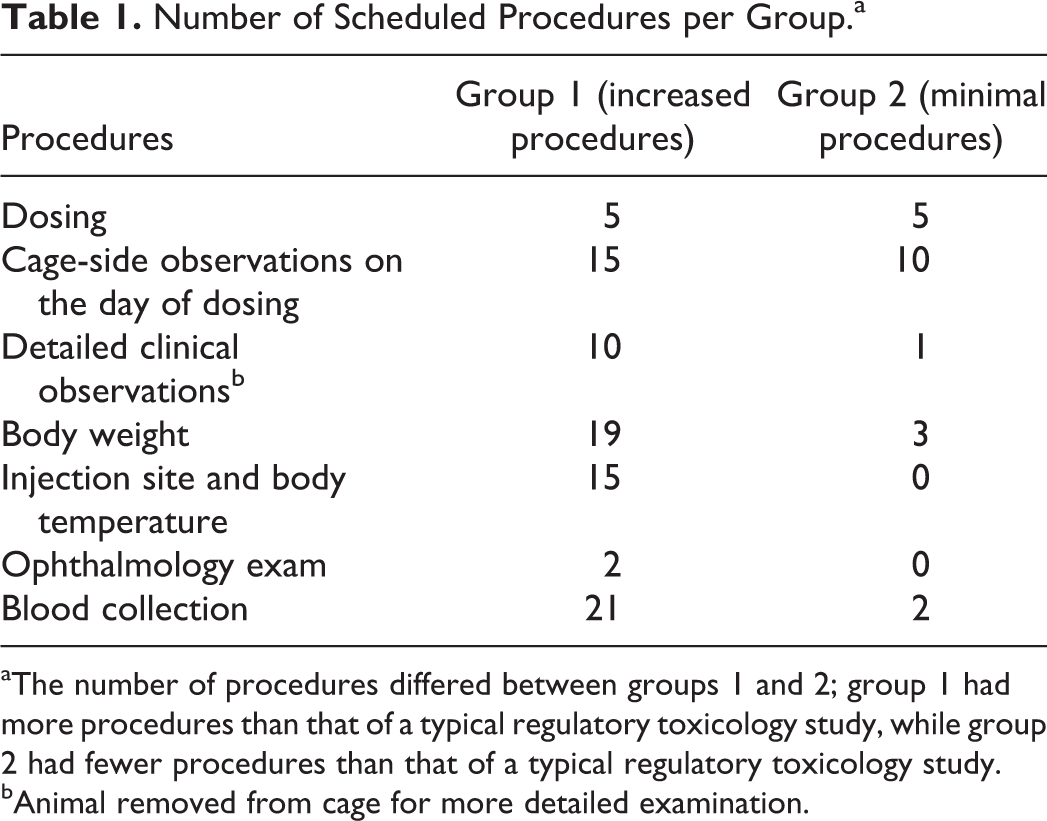
aThe number of procedures differed between groups 1 and 2; group 1 had more procedures than that of a typical regulatory toxicology study, while group 2 had fewer procedures than that of a typical regulatory toxicology study.
bAnimal removed from cage for more detailed examination.
Blood and Urine Collection
Blood was collected primarily from the auricular artery from rabbits restrained in metal restrainers (rabbit restrainer cage, model no. RR1; Draper Metal Fabrication Co., Holbrook, MA). Blood was collected from all animals for coagulation, hematology, and clinical chemistry analysis prior to the start of the study in sodium citrate anticoagulant tubes, ethylenediaminetetraacetic acid anticoagulant tubes, and serum separator tubes, respectively (total of 8.5 ml blood collected); blood for clinical chemistry was also collected at necropsy. Serum C-reactive protein (CRP), creatine kinase (CK), cardiac troponin I (cTnI), and stress-associated serum biomarkers (epinephrine, norepinephrine, cortisol, and corticosterone) were obtained prior to the start of the study and at necropsy in both groups. In addition, 1 ml of blood was collected from group 1 animals for cTnI, CK, CRP, epinephrine, norepinephrine, cortisol, and corticosterone prior to IM administration of saline (time 0) and at 4, 24, and 48 hr after administration on each of the 5 dosing days (with the exception of the 48-hr time point on day 57 which coincided with necropsy; a total of 20 ml blood/rabbit over 8 weeks). Serum for corticosterone, cortisol, epinephrine, and norepinephrine and urine for epinephrine and norepinephrine were protected from light and stored at −70°C until analyzed. Urine was collected by cystocentesis at necropsy.
Serum Clinical Chemistry, cTnI, CRP, Creatine Kinase, and Stress Biomarkers
Routine clinical chemistry parameters were evaluated using a Siemens Advia 1800 clinical chemistry analyzer (Siemens Corp., Tarrytown, NY). CRP was evaluated using the Life Diagnostic rabbit-specific solid phase sandwich immunoassay (Life Diagnostics, Inc., West Chester, PA). Hematology was evaluated using a Siemens Advia 120 analyzer (Siemens Corp.). Fibrinogen, activated partial thromboplastin time, and prothrombin time were evaluated on the Diagnostic Stago STA Compact coagulation analyzer (Stago, Parsippany, NJ). cTnI was measured using the human TnI-Ultra® assay on the Siemens Advia Centaur immunoanalyzer (Siemens Corp.). An internal assay validation was performed as well as a preliminary evaluation of cTnI responses in normal minimally restrained rabbits. As part of the internal validation, the limit of quantitation was determined by running (in duplicate) daily for 10 days a series of rabbit serum samples close to the limit of blank and calculating the mean, standard deviation, and percentage coefficient of variation. The limit of quantitation (the lowest concentration of cTnI within the acceptable precision limit of 20%) was 0.03 ng/ml. When normal rabbits were assessed with this assay, many had results <0.03 ng/ml but some had cTnI results up to 0.05 ng/ml and thus a threshold for background finding of 0.06 ng/ml was established. Preliminary studies also included collection of serum from a rabbit administered 3 mg/kg isoproterenol (peak mean concentration of cTnI of 133.5 ng/ml at 6 hr postdosing) as positive a control (data not shown). Epinephrine and norepinephrine were quantitated using an ultrahigh-pressure liquid chromatography–tandem mass spectrometry (LC-MS/MS) assay. Samples were prepared for analysis by the addition of an internal standard followed by solid phase extraction. Epinephrine and norepinephrine were separated from the other sample components by gradient elution with reverse phase chromatography and then detected by monitoring unique fragment ions in the mass spectrometer. Calibration curves for epinephrine and norepinephrine were constructed from known standards by plotting the ratio of the analyte peak area to internal standard peak area versus concentration. The concentration in the samples were calculated by interpolation using 1/x 2 weighted linear regression analysis from the calibration curve for each analyte. Urine measurements were normalized to urinary creatinine. Methods deployed were developed in-house specifically for rabbit samples and are similar to those published in literature (Bicker et al. 2013). Assay performance was determined by quality control standards throughout the individual runs.
Serum corticosterone and cortisol were measured using an LC-MS/MS assay. Samples were prepared for analysis by the addition of an internal standard followed by supported liquid extraction. Prior to detection, the corticosterone and cortisol were separated from the other sample components by reverse phase chromatography coupled with gradient elution. The analytes and internal standard were detected by monitoring unique fragment ions. Calibration curves for corticosterone and cortisol were constructed from known standards by plotting the ratio of the analyte peak height to internal standard peak height versus concentration. The concentration in the samples was calculated by interpolation using 1/x 2 weighted linear regression analysis from the calibration curve for each analyte. Assay performance was determined by quality control standards throughout the individual runs. Methods deployed were developed in-house specifically for rabbit samples and are similar to those published in literature (Guo et al. 2006).
Necropsy and Histology
Animals were euthanized by intravenous administration of barbiturate anesthesia (Nembutal® [sodium pentobarbital]; Oak Pharmaceutical, Lake Forest, IL) followed by exsanguination on day 59, 2 days after the last IM saline injection. All animals were fasted overnight prior to necropsy. The necropsy included macroscopic examination of the external surface of the body and the thoracic and abdominal cavities and their contents. Hearts were excised, gently flushed with 10% neutral buffered formalin (StatLab Medical Products, McKinney, TX) to remove the blood, and immersion fixed in 10% neutral buffered formalin for at least 48 hr. After fixation, the hearts were sectioned generating 2 longitudinal sections. In order to fit in cassettes, these longitudinal samples were then cut in cross section to generate 4 sections, 2 with the apical region of the heart and 2 with the base of the heart (Figure 1). All samples were processed with paraffin, and 5 µm sections were stained with hematoxylin and eosin (H&E). In addition to the routine H&E staining, special stains (periodic acid Schiff [PAS], Grocott’s methenamine silver [GMS], Gram, Giemsa, Masson’s trichrome, and/or phosphotungstic acid hematoxylin [PTAH]) were utilized on sections of heart from selected animals to further clarify and/or characterize findings.
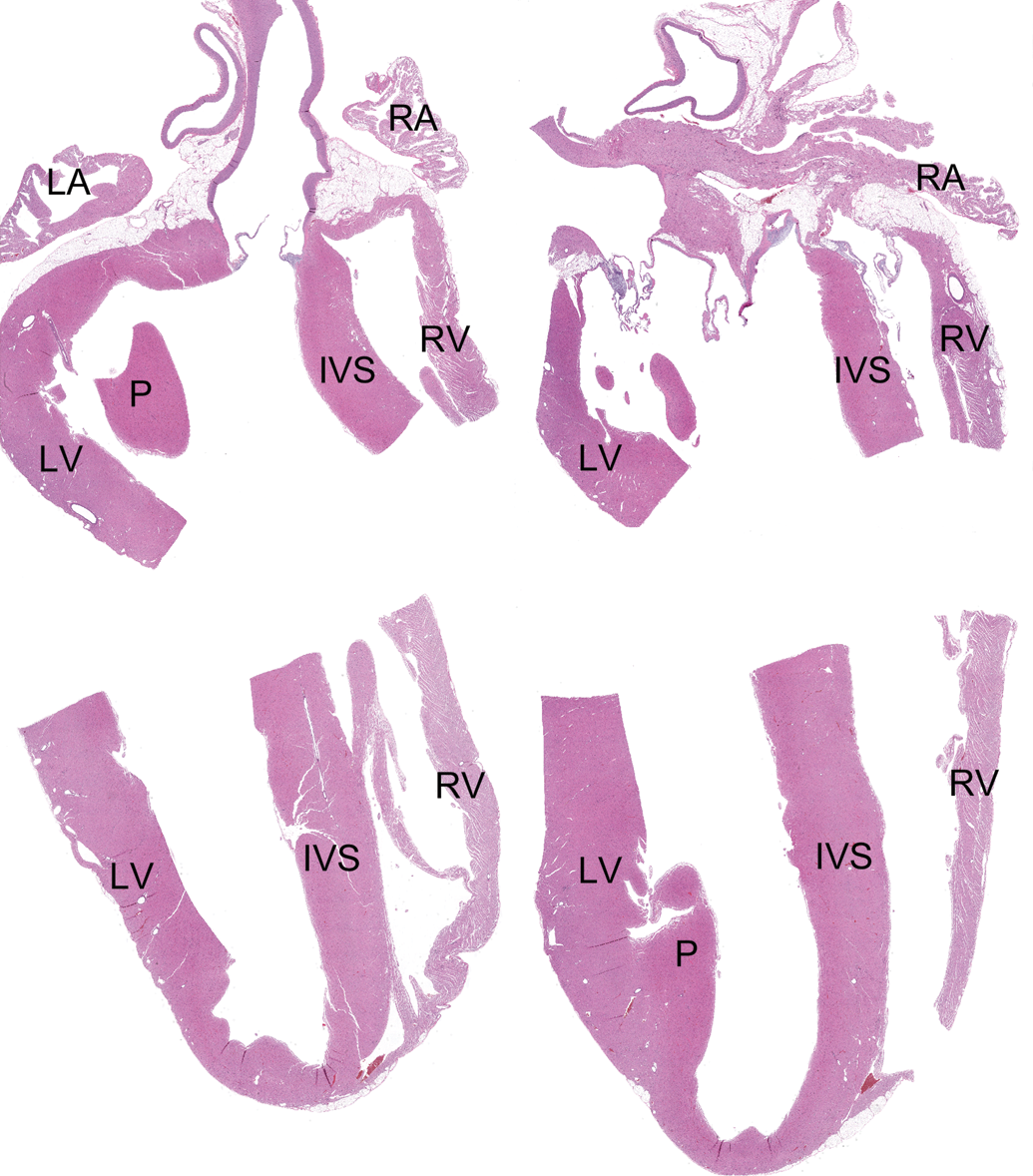
Histological sections of the rabbit heart. Formalin-fixed hearts were incised to generate two longitudinal sections. These samples were then bisected to fit in cassettes for processing, generating four hematoxylin and eosin slides; two slides included the atria (right atrium [RA] and left atrium [LA]) and an upper part of the ventricles and two slides contained the ventricles, including the right ventricle (RV), left ventricle (LV), papillary muscle (P), and the interventricular septum (IVS). Hematoxylin and eosin. Original Objective 0.4×.
Histopathologic Evaluation
Microscopic findings were graded on a scale of 1–5 as minimal, mild, moderate, marked, or severe, respectively. Each region of the heart was evaluated and in order to understand the distribution of findings within the heart, these findings were recorded separately for the left ventricle, right ventricle, interventricular septum, right atrium, and left atrium. All findings were recorded, regardless of the size or extent. Following completion of the tissue evaluation by the study pathologist, an independent peer-review evaluation of all heart sections was performed by a second pathologist.
Results
In life
All rabbits survived to scheduled euthanasia and were in good health. There were no differences in body weight, food consumption, or procedure-related clinical effects.
Necropsy
Complete necropsies with gross evaluation of the external surfaces, thoracic, and abdominal cavities and contents were performed. Abnormal color at the injection site was observed at necropsy in 5 animals in group 1 and 13 animals in group 2. No findings were identified grossly in the heart or in any other tissues.
Inflammatory and Other Findings in the Rabbit Heart
A range of inflammatory findings were evident in the heart microscopically. Inflammatory findings were identified in 80% (56 out of 70) of the rabbits in the limited handling group (group 2) and 86% (60 out of 70) in the increased handling group (group 1). Very rarely, there was individual cardiomyocyte necrosis unassociated with inflammatory cells (Figure 2A). More commonly, however, the necrotic cardiomyocytes were associated with macrophages (Figure 2B) and occasionally with low numbers of heterophils (Figure 2C; termed mixed inflammatory cell infiltrates) and rarely with interstitial edema/basophilic interstitial fibrillar matrix. Aggregates of macrophages without evidence of necrotic cardiomyocytes were also evident (Figure 2D), in which the necrotic cardiomyocyte(s) was either no longer present or obscured by inflammatory cells. Small lymphocytic infiltrates with or without plasma cells were rarely evident (Figure 2E); these may be comparable to the lymphocytic infiltrates common in many organs unassociated with tissue damage and will not be the subject of further discussion here (Drevon-Gaillot et al. 2006; Helke et al. 2016; Sato, Doi, Wako, et al. 2012). Occasionally fibrosis was present (Figure 2F). These findings were minimal to moderate in severity grade, focal to multifocal in distribution, and located within the myocardium. While minimal severity grades were found in both groups at comparable (similar) incidences (60 of the 70 and 56 of the 70 animals, respectively; Table 2), mild and moderate inflammatory findings were only identified in group 1. Individual cardiomyocyte necrosis unassociated with inflammatory cells was present in two of the group 1 hearts and none of the group 2 hearts. Fibrosis was evident in a single group 2 rabbit (with minimal inflammatory cell infiltrates) and a single group 1 rabbit (unassociated with inflammatory cell infiltrates). The foci were usually comprised of macrophages and occasionally heterophils and/or individual necrotic cardiomyocytes. These findings were widely distributed but were most commonly localized to the left ventricular free wall, less commonly in the interventricular septum and the right ventricular free wall, and least frequently in the atria (Table 3). Many of the minimal foci recorded in this study had such low numbers of inflammatory cells that they would most likely fall beneath the routine recording thresholds by most pathologists because of their small size and/or incidence across all study groups. Compared to the Pfizer historical database (of the 1,254 rabbits where an approximately 13% incidence of inflammatory cell foci were recorded in the heart), much higher incidences were recorded in this study. Possible reasons for such large differences in the recorded incidences include the judicious application of thresholds by individual pathologists during evaluation of regulatory toxicology studies, the high sensitivity for recording heart findings in this study regardless of extent, and the increase in surface area evaluated in this investigation (approximately 200% more).
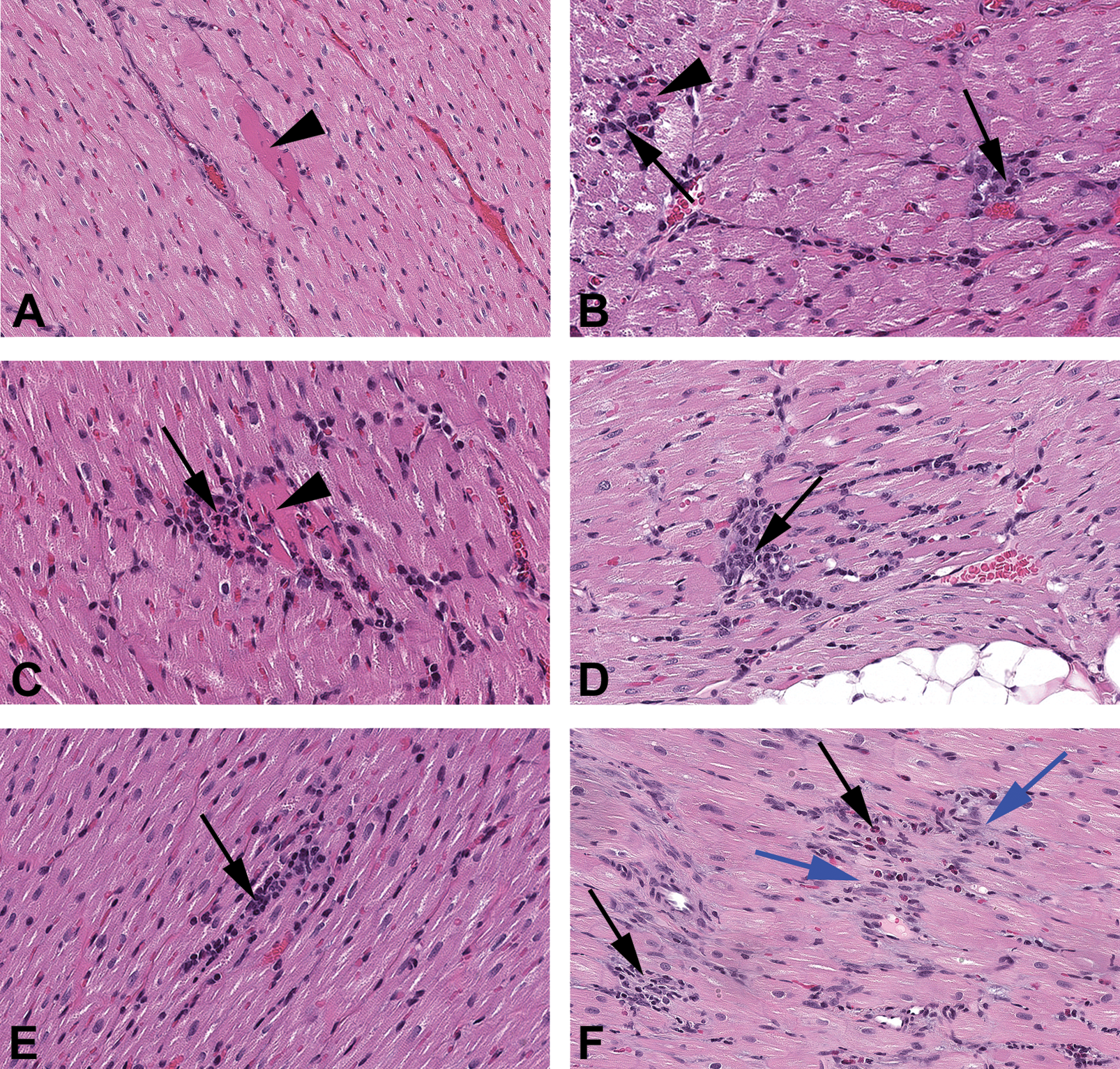
Photomicrographs of the heart from minimally handled rabbits (group 2), showing the progression of lesions. (A) Focal cardiomyocyte necrosis with no apparent inflammation (arrowhead). (B) Focal cardiomyocyte necrosis (arrowhead) with few inflammatory cells adjacent to focal small macrophage aggregate (black arrow). (C) Focal cardiomyocyte necrosis (arrowhead) associated with a slightly greater number of inflammatory cells (black arrow). (D) Inflammatory cell infiltrate (black arrow) with macrophages and heterophils without cardiomyocyte necrosis within the lesion. (E) Chronic inflammatory cell infiltrate comprised of lymphocytes (black arrow). (F) Myocardial fibrosis (blue arrows) associated with low numbers of mixed inflammatory cell infiltrates (black arrows). Hematoxylin and eosin. (A–E) Original objective 20×.
Group Incidences and Severities of the Inflammatory Microscopic Heart Findings.a
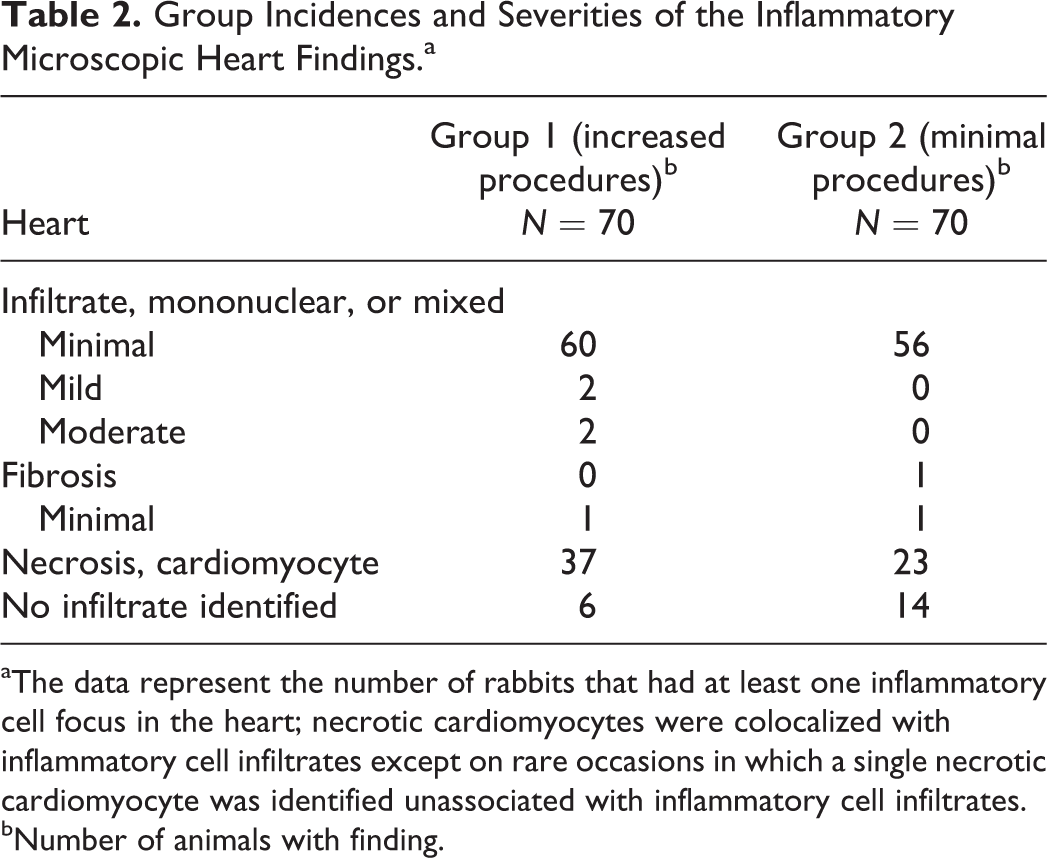
aThe data represent the number of rabbits that had at least one inflammatory cell focus in the heart; necrotic cardiomyocytes were colocalized with inflammatory cell infiltrates except on rare occasions in which a single necrotic cardiomyocyte was identified unassociated with inflammatory cell infiltrates.
bNumber of animals with finding.
Incidence and Severity of Inflammatory Findings According to Location in the Various Heart Subregions.a

Note: LV = left ventricle; RV = right ventricle; IVS = interventricular septum; LA = left atrium; RA = right atrium.
aFindings were recorded once for each region of the heart (i.e., a single score for each finding based on multiple slides); however, one animal might have two types of infiltrates recorded for the same region of heart (mixed cell or mononuclear cell). Cardiomyocyte necrosis was associated with inflammatory cell infiltrates in all but two instances.
In addition to these findings, aortic mineralization was evident in 9% of the rabbits (ranging from minimal to moderate and focal to multifocal) and was similarly distributed between the two groups. Mineralization of the pulmonary artery was identified in a single animal, but as this vessel was infrequently present in the heart sections, the true incidence of mineralization at this site is uncertain.
Influence of Procedures on Myocardial Inflammatory Findings
While the incidence of inflammatory findings was similar between groups 1 and 2, the distribution of foci within the heart was increased in group 1 as compared to group 2. Specifically, more subregions (i.e., left ventricle, right ventricle, left atrium, right atrium, and interventricular septum) in individual hearts were identified as having at least one inflammatory focus (Table 3). The areas of the heart that demonstrated the greatest increase in inflammatory findings were the left ventricle and interventricular septum (Table 3). In addition, mild and moderate inflammatory findings were present only in group 1 (4 of the 70 hearts) and, similar to most of the minimal findings, were comprised of macrophages with scattered heterophils and necrotic cardiomyocytes (Figures 3 and 4); these were differentiated from minimal findings in that they were more extensive and/or more widely distributed. Mild and moderate findings were localized to the interventricular septum and left ventricle/papillary muscle (Table 3; Figure 3). These more severe inflammatory infiltrates were sometimes organized in a linear pattern, suggesting a response along the length of individual cardiomyocytes (Figures 3C and 4D). Mild and moderate lesions were not attributable to infectious disease (negative PAS, GMS, Gram, and Geimsa histochemical stains) or vascular fibrin thrombi (negative PTAH staining in vasculature; data not shown).
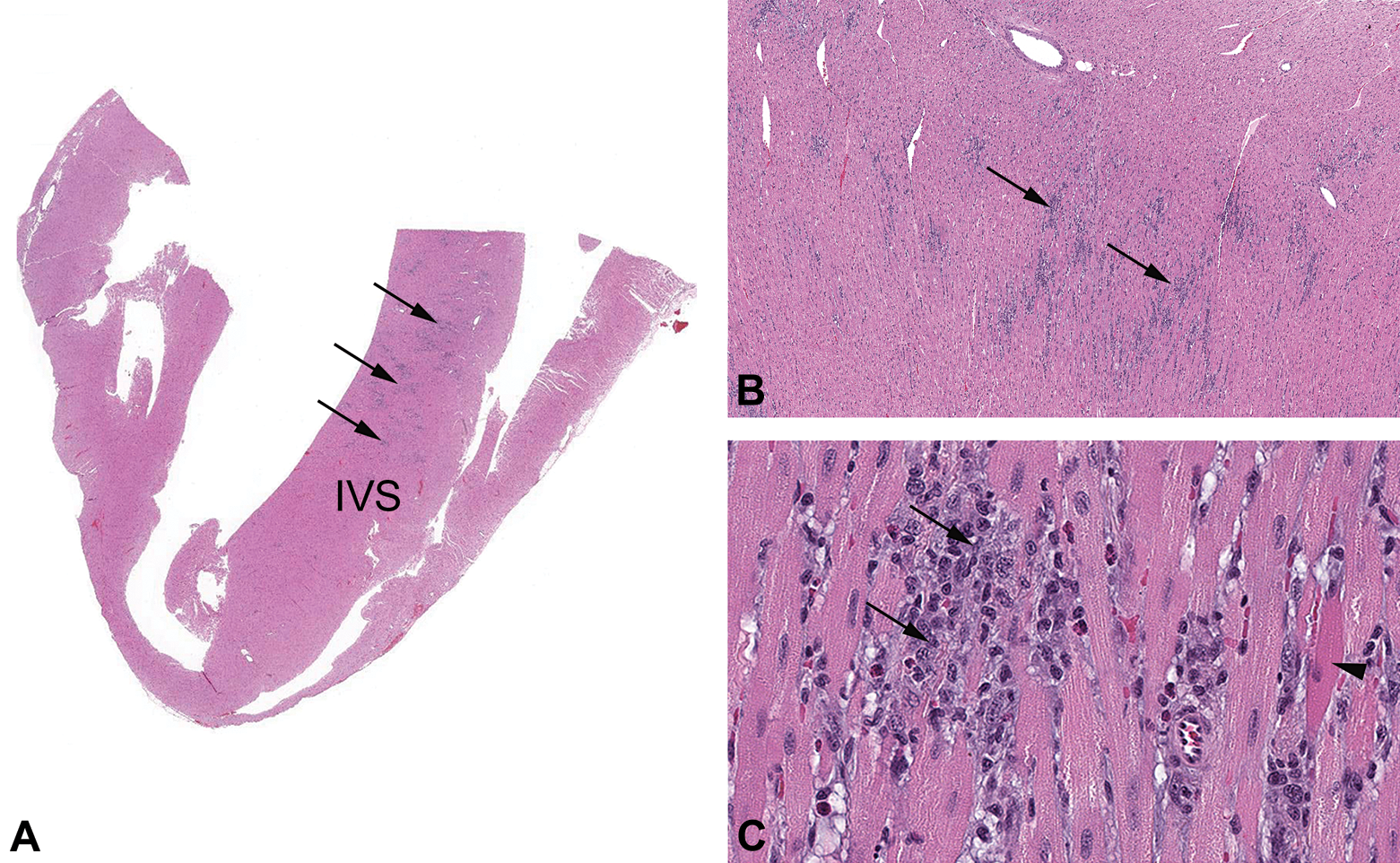
Photomicrographs of the heart from an increased procedures rabbit (group 1). (A) Low magnification of cardiac ventricles and interventricular septum (IVS). The IVS and left ventricle had multifocal widespread inflammatory cell infiltrates (arrows). (B) Higher magnification of the IVS shows the infiltrate distribution. (C) High magnification of the inflammatory cell infiltrates; infiltrates included macrophages (arrows) with occasional necrotic cardiomyocytes (arrow head). Infiltrates were often in a linear pattern, presumably along the length of necrotic individual cardiomyocytes. Hematoxylin and eosin. (A) Original objective 0.4× (B) original objective 4× and (C) original objective 20×.
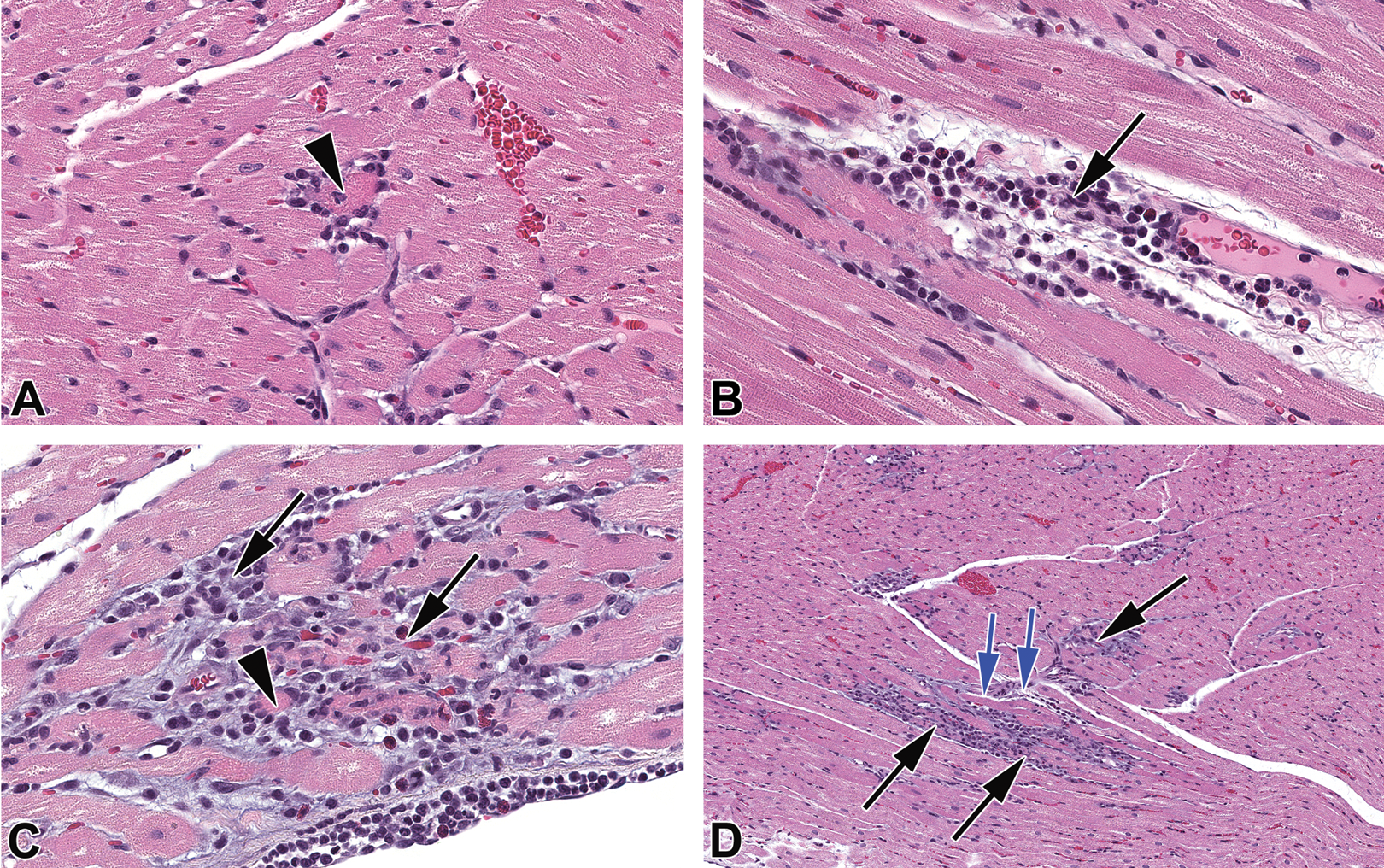
Photomicrographs of the heart from an increased procedures rabbit (group 1; different from those in Figure 3). (A) Necrotic cardiomyocyte (arrow head) associated with few inflammatory cells. (B) Inflammatory cell infiltrates (black arrow) associated with focal cardiomyocyte necrosis and interstitial edema (clear spaces). (C) Inflammatory cell infiltrates comprising macrophages and heterophils (black arrows) associated necrotic cardiomyocytes (arrow head) with basophilic matrix and early fibrosis. (D) Distribution of inflammatory cell infiltrates (black arrow) was occasionally closely associated with small vessels (blue arrows). Hematoxylin and eosin. (A–C) Original objective 20× and (D) original objective 10×.
Stress Biomarkers
Serum stress biomarkers were evaluated before the study start to obtain baseline values and at necropsy. Changes in serum stress biomarkers in response to handling and procedures (IM injection and 4 blood collections over 48 hr) were evaluated only in group 1. Changes were most evident with norepinephrine (NE) and cortisol (Figure 5A and C). NE concentrations gradually increased after time 0 (immediately prior to IM saline administration), with peak values typically occurring at 48 hr postadministration. Mean NE concentrations increased at each dosing interval compared to baseline, peaking at day 29, which coincided with the 3rd IM saline administration (peak at 1.7× baseline; Figure 5A). Serum cortisol concentrations mimicked the findings in NE (Figure 5C). However, elevations in cortisol often peaked 4 hr after IM saline administration. Serum cortisol concentrations typically decreased or returned to near baseline values just prior to the next dosing interval. There were similar changes in mean corticosterone values as those seen with cortisol but based on higher corticosterone baseline values, increases in mean values were slight and only identified on days 15 and 29 at 4 hr postdose (Figure 5D). The values were typically lower than baseline values at time 0 at each dosing interval as well as on day 59. Epinephrine also showed changes over time, but based on high baseline values, no conclusions could be made (Figure 5B). Variations in NE and epinephrine were high, possibly in part due to the nature of the stress response in the individual animals and/or to the very short half-life of NE and epinephrine (a few minutes) in the blood (Friedgen, Halbrugge, and Graefe 1993). To minimize handling and procedures in group 2 animals, blood was collected and stress biomarkers evaluated only prior to the initiation of dosing and prior to necropsy (Figure 5). Serum stress biomarker values were similar between groups 1 and 2 at comparable time points (i.e., prior to initiation of dosing and at necropsy).
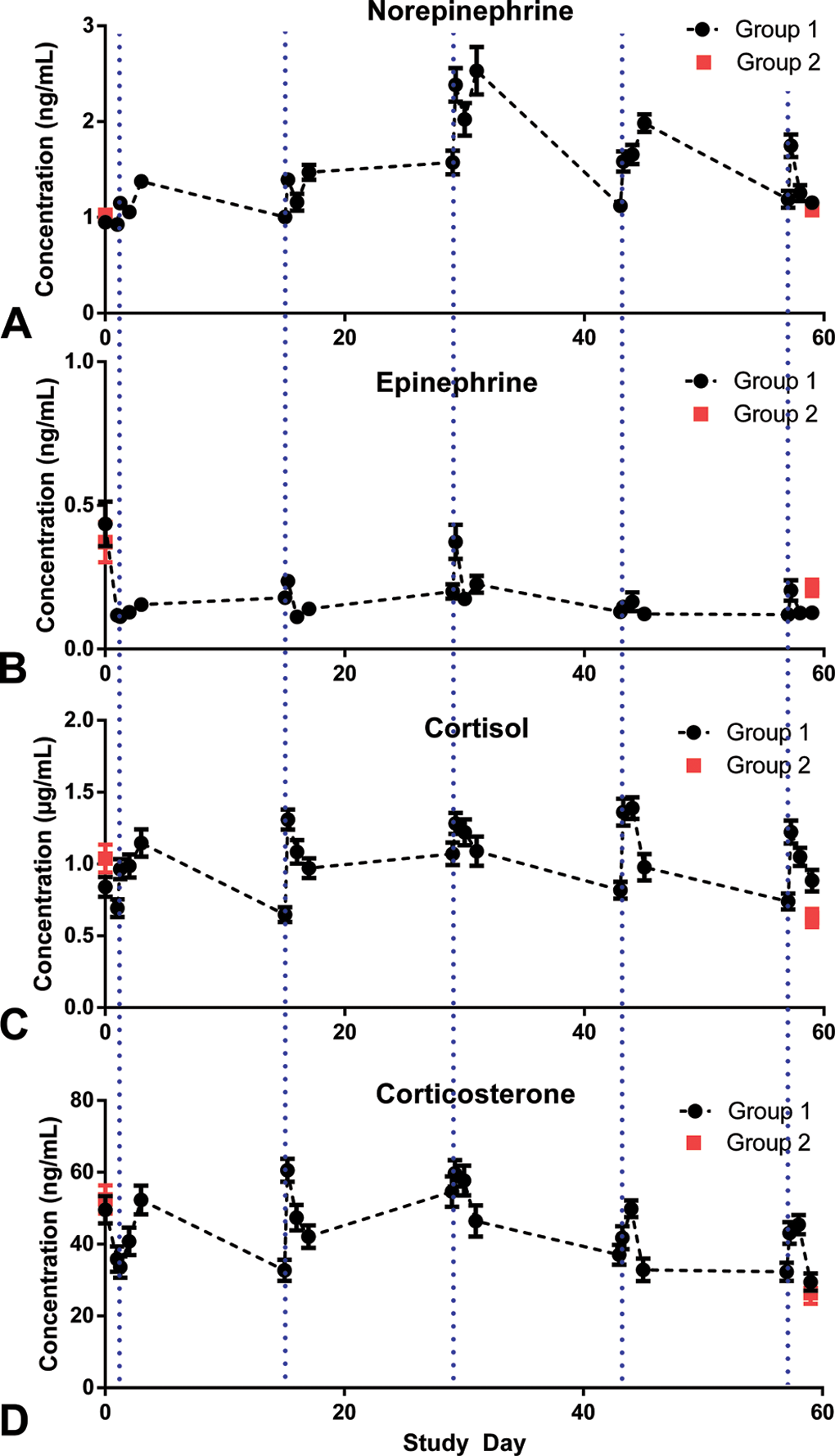
Plasma concentrations of stress biomarkers associated with dosing in groups 1 and 2. (A) Norepinephrine (NE), (B) epinephrine (EP), (C) cortisol, and (D) corticosterone. In group 1 (•), dosing days (1, 15, 29, 43, and 57) included 4 blood collections: 0 hr (predose, vertical dotted blue lines), 4-hr postdose, 24-hr postdose, and 48-hr postdose. Stress biomarker concentrations typically increased over 48 hr after saline administration as compared to the time 0 (predose) blood sampling. Maximal NE and EP levels over the course of the study occurred at day 29. Measurements were taken prior to the study initiation and at necropsy in group 2 (▪).
There were no notable changes in urine chemistry biomarker parameters (NE or epinephrine) at the end of study (day 59). The timing of the urine collections (day 59 only) allowed for the assessment of changes in the stress biomarkers only at the end of the study (data not shown).
cTnI
Based on internal Pfizer studies conducted to determine the suitability and sensitivity of using the human TnI-Ultra assay to measure rabbit cTnI, concentrations of cTnI up to 0.06 ng/ml were considered to be within the range of background. Individual cTnI concentrations >0.06 ng/ml were present in 23 (33%) of the 70 rabbits in group 1 on one or more of the study days. These elevations typically were present at 4 and/or 24 hr postdose and were indicative of cardiomyocyte injury with release of cTnI into the circulation. Elevations in cTnI did not correlate well with histological findings, as >90% of the animals in group 1 had inflammatory infiltrates in the heart, but elevations in cTnI were only evident in 33% of rabbits. The 2 animals with the highest elevations in cTnI (peak values of 2.04 and 1.69 ng/ml), which occurred on day 57, had the most severe inflammatory findings in the heart (moderate). Interestingly, the 2 rabbits with mild inflammatory cell infiltrates did not have cTnI > 0.06 ng/ml at any time point during the study, indicating that these lesions may have initiated during the intervals between sampling. More animals had cTnI > 0.06 ng/ml on day 57 (6 animals) as compared with day 1 (4 animals). There were no animals with cTnI > 0.06 ng/ml in group 2, the minimally handled animals (only evaluated prior to the initiation of dosing and day 59 [48 hr after the last dose]).
CRP and CK
Study procedure–related increases in CRP and CK, as compared to the baseline values, were evident in group 1 (increased procedures) and increases in CRP were also observed compared with baseline in group 2 (minimal procedures). In group 1, CRP increased up to 4.3× baseline and CK increased up to 2.9× baseline at multiple time points over the study (after the 1st, 3rd, 4th, and 5th doses). Both analytes typically started to increase 4 hr postdose, peaked at 24 hr postdose, decreased at 48 hr postdose, and returned to baseline or near baseline at the subsequent predose time point. The increases in CRP and CK were attributed to an acute phase response and muscle injury (possibly as a result of restraint, blood collection vascular trauma, and/or IM injection), respectively. Group 2 rabbits also had slight increases in CRP 1.8× baseline on day 59 (48 hr postdose) but did not have changes in CK.
Discussion
This report details the nature, incidence, and distribution of microscopic inflammatory findings in hearts from female NZW rabbits. Minimal scattered aggregates of macrophages, with or without heterophils or individual necrotic cardiac myocytes, represented the most common background finding in the rabbit heart. Rabbits subjected to more handling and procedures had more widespread myocardial inflammatory foci and, in a few animals, increased severity of these findings.
The type of infiltrate described in detail here is similar to that described previously in the heart of rabbits (Miller 1924; Osheroff et al. 2013; Weber and Van Der Walt 1975). These findings appear to fall along a continuum representing the chronicity of the lesion. The initiating cause may be individual cardiomyocyte necrosis which is rapidly followed by infiltration of macrophages and heterophils. Ultimately, if lesions are severe enough, the inflammation resolves with fibrosis. Presumably small lesions resolve with limited or no scarring. The presence of inflammatory cells tracking along the length of cardiomyocytes is consistent with underlying individual cardiomyocyte necrosis. Small and transient elevations in serum cTnI during the study may have been the result of cardiomyocyte perturbations or death underlying some inflammatory foci. cTnI had limited correlation with microscopic lesions except in the most severe cases; this may reflect timing of blood collection relative to tissue sampling as well as the region evaluated microscopically. The active inflammatory lesions described in this article may be distinguished from effects of direct cardiotoxins, as toxic insult tends to have more widespread cardiomyocyte degeneration or necrosis, evidence of vascular injury, or have toxicity in other organ systems (Dunnick et al. 2004; Hanton et al. 2008; Jokinen et al. 2011, 2005; Lamberti et al. 2014; Merrick, Robinson, and Condie 1991; Nyska et al. 2005; Pettersen et al. 2012; Varga et al. 2015).
The data presented here demonstrate that handling and procedures can increase both the distribution of inflammatory foci in the rabbit heart and, in a subset of animals, the severity of the inflammatory findings. The more severe (mild and moderate) inflammatory findings were morphologically similar to the minimal (background) findings except that they were more expansive or widespread. Thus, while background inflammatory findings are typically minimal, they could range in severity up to moderate in any given toxicology study. Mild and moderate inflammatory findings were also evident in of 3 out of 1,254 animals in the Pfizer historical database, indicating that lesions of this severity have also been seen in control animals historically.
In the published literature, heart lesions similar in nature and distribution (primarily left ventricle and papillary muscle) to those identified in this study have been associated with both stress and catecholamines in rabbits (Downing and Chen 1985; Weber and Van Der Walt 1975). Catecholamine toxicity in the heart has been attributed to effects on the vasculature (hypoxia/ischemia) as well as to direct cardiomyocyte damage from spillover of catecholamines released from local sympathetic nerve terminals (Fineschi et al. 2010; Samuels 2007; Y-Hassan 2014; Liaudet, Calderari, and Pacher 2014). Within hours of administration, catecholamines cause contraction band degeneration/necrosis; after 24 hr, the findings are primarily inflammatory, comprising macrophages with scattered polymorphonuclear cells (heterophils). The lesions caused by catecholamine administration in rabbits have been suggested to be α-adrenergic receptor driven (Downing and Chen 1985).
Stress-induced cardiac damage in humans is suggested to be due to direct injury by catecholamines (Liaudet, Calderari, and Pacher 2014). In this study, a one-to-one correlation between the serum stress biomarker concentrations and heart lesions was not identified. A relationship of the heart findings to local catecholamine release, however, cannot be ruled out; additionally, peak blood levels of these very short-lived stress hormones may have not have coincided with the blood collection schedule. It is also possible that heretofore unknown mechanisms may also play a role in the pathogenesis of background inflammatory cell findings in the rabbit heart.
As compared to other species, rabbits are known for being hyperexcitable and may be uniquely susceptible to the development of stress-associated heart lesions (Osheroff et al. 2013). Interestingly, catecholamine concentrations in the rabbit heart have been demonstrated to be notably higher (up to 3 times) than that of other species such as the rat or dog (Angelakos, Fuxe, and Torchiana 1963; Mukherjee et al. 1983). Additionally, there is well-documented interindividual variation in the ability to respond/adapt to stress in NZW rabbits which may account for the observed differences in the severity of myocardial inflammatory findings (Giannico et al. 2014; Hilton 1982; Oxley and Ellis 2015; Schadt and Hasser 1998; Trocino et al. 2013; Burhans, Smith-Bell, and Schreurs 2015; Smith-Bell, Burhans, and Schreurs 2012; Lidfors and Edström 2010; Verga, Luzi, and Carenzi 2007; Weber and Van Der Walt 1975). Variation in individual susceptibility and adaptability to stress could explain the presence of mild and moderate severity heart findings in only a small subset of rabbits after increased handling and procedures.
Myocardial inflammatory cell infiltrates have been well described in rats and to a lesser extent in other species, such as nonhuman primates and mini pigs (Jokinen et al. 2011, 2005; Kemi et al. 2000; Chanut et al. 2013; Helke et al. 2016; Sato, Doi, Kanno, et al. 2012). While the etiology of the myocardial lesions has not been clearly identified, vascular- or catecholamine-based mechanisms have been suggested (Berridge et al. 2016; Cross et al. 2015; Greaves 2007; Jokinen et al. 2011, 2005; Kemi et al. 2000; Yoshimura et al. 1989). A more detailed understanding of the progression of lesions with age as well as any sex-related differences in the rabbit is required to fully understand this background finding.
Conclusions
Female NZW rabbits have a high incidence of minimal myocardial inflammatory cell findings that can range in severity from minimal to marked, in which the predominant cell type is the macrophage. While some of these findings may represent incidental infiltrates common in tissues of animals (usually perivascular and lymphocytic), many appear to be the result of individual cardiomyocyte necrosis which is rapidly followed by inflammatory cell infiltration (typically macrophages with heterophils). In severe cases, these inflammatory cell foci may resolve as focal fibrosis. The chronicity of the lesion dictates the histological appearance: very early lesions have individual cardiomyocyte necrosis with little or no inflammatory infiltrate; intermediate lesions have mixed inflammatory cell infiltrates with or without necrotic cardiomyocytes; and late lesions consist of inflammatory cell infiltrates and fibrosis. The incidence and severity of these foci may be exacerbated by handling and procedures that occur in toxicology studies. Based on the data presented here and on the well-described catecholamine-related effects on rabbit hearts, we propose that the increase in distribution and severity of inflammatory cell foci in the rabbits subjected to increased procedures and handling could be mediated by a stress response. Additionally, a subset of NZW rabbits may be exceptionally vulnerable to stress responses and/or do not habituate to stressors, which may contribute to the subsequent development of myocardial inflammatory cell foci under stressful conditions. The development of heart lesions secondary to study procedure–related stress in routine repeat-dose toxicology studies may make differentiation between stress-associated heart lesions and test article–related findings in toxicology studies in rabbits challenging. Further efforts are needed to understand how age and sex (as only females were evaluated in this study) may impact the incidence and severity of the myocardial inflammatory cell infiltrates in rabbits, particularly in the face of study-related activities and stress.
Footnotes
Acknowledgments
Special thanks to Kimberly Ebersole for her careful review of the data included in this article and Alan Opshal for image analysis support.
Author Contribution
Authors contributed to conception or design (RSS, IP, GH, KK, RP, VM, CR, JC, WR, DC); data acquisition, analysis, or interpretation (RSS, IP, CR, JC, WR, DC); drafting the manuscript (RSS); and critically revising the manuscript (IP, GH, KK, RP, VM, CR, JC, WR, DC). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
