Abstract
Over a 3-y period, 12 adult New Zealand white (NZW) rabbits were presented for postmortem examination following variably long periods of inappetence and soft-to-liquid stool production. Postmortem findings included serosanguineous fluid in abdominal and thoracic cavities, dark-red-to-white renal foci, reddened intestinal serosa, and pulmonary edema. Microscopically, mesangial changes and thrombi were observed in renal glomeruli, and mild-to-severe enteritis was observed. These findings resemble hemolytic uremic syndrome, which typically follows enterocolitis associated with Shiga toxin (Stx)-producing Escherichia coli infection. In our case series, various gram-negative bacteria, most commonly E. coli, were isolated from the intestinal tracts; however, Stx production was not demonstrated. Evidence of Encephalitozoon cuniculi infection, a common cause of renal disease in rabbits, was also not found. Our cases suggest that gram-negative enteric bacteria should be included in the differential diagnosis of renal disease in NZW rabbits, especially in cases with an accompanying clinical history of gastrointestinal disorder.
Enteric disease is a common problem in rabbits. Several infectious organisms have been associated with intestinal disease, including coccidia (Eimeria spp.), Clostridia spp., and coliform bacteria. 7 Stasis of the intestinal tract may lead acutely to reversible prerenal azotemia. However, chronic renal disease in rabbits is usually associated with the microsporidian parasite Encephalitozoon cuniculi, characterized histologically by granulomatous interstitial nephritis.10,12
Escherichia coli is a common nonpathogenic commensal inhabitant of the intestine of many species. 19 In contrast, this bacterium is not commonly isolated from the intestinal tract of healthy adult rabbits.15–17 Several E. coli serotypes, including O128:H2, have been reported to cause diarrhea in preweaning and weaned rabbits in Europe and America.3,4,15,16 Postmortem findings associated with infection with toxin production–negative E. coli (e.g., serotypes O153 and O15) include diarrhea and slightly excessive serosanguineous fluid in the peritoneal cavity. Histologically, edema and hemorrhage of the cecal lamina propria and submucosa in addition to epithelial degeneration and loss have been reported. Coccobacilli are found attached to epithelial cells.4,17 Renal lesions consisting of hydropic change in the proximal tubular epithelium were also noted in one report. 17
Dutch Belted rabbits infected with an enteropathogenic E. coli (EPEC) O145:H− or coinfected with EPEC O145:H− and enterohemorrhagic E. coli (EHEC) O153:H− developed variable degrees of enterocolitis associated with microangiopathy and nephropathy. 8 These lesions can be produced experimentally by single infections with EHEC O153 or O157:H7, which has been proposed as a model of hemolytic uremic syndrome (HUS) in humans. 9
A series of 12 adult New Zealand white (NZW) rabbits from a commercial production facility in Oregon State, USA were presented for postmortem examination over a 3-y period. Clinical signs included inappetence, often accompanied by the variable production of soft-to-liquid stools. The period of clinical illness was 1–13 d. Because of their worsening clinical course, all rabbits were euthanized via barbiturate overdose. Antemortem blood collection was carried out for hematology (6 rabbits; cases 1, 3, 5, 7, 8, 10) and clinical chemistry (5 rabbits; cases 1, 5, 7, 8, 11). Hematology findings included anemia in all 6 rabbits and heterophilia in 4 rabbits (cases 3, 5, 7, 8). Monocytosis was found in 4 rabbits (cases 3, 7, 8, 10) and thrombocytopenia in 1 rabbit (case 3; Suppl. Table 1). Clinical chemistry results consistent with renal disease were observed in all 5 rabbits from which serum was collected (Suppl. Table 2).
At postmortem examination, variable amounts of fecal staining were noted around the perineum and over the ventral abdomen. Serosanguineous fluid (5–25 mL) was present in each of the thoracic and abdominal cavities in 9 of 12 rabbits (cases 1, 3–9, 12; Fig. 1A). Abdominal fluid was analyzed from 2 rabbits (cases 1, 8) and classified as modified transudates (nucleated cell count 0.11–0.44 × 109/L, protein 42–54 g/L, specific gravity 1.026–1.027). The capsular and cut surfaces of the kidneys of 10 rabbits contained dark-red-to-white foci (cases 1–3, 5, 7–12; Fig. 1B). Urine was obtained from 6 rabbits by cystocentesis (cases 1, 2, 7–9, 11; Suppl. Table 3). Blood was detected in the urine from all cases. Acidic urine was found in 3 rabbits (cases 2, 7, 11), which may be associated with anorexia. Protein in association with dilute urine (specific gravity <1.020), indicative of renal disease, was observed in 3 cases (cases 1, 7, 8). 13 The serosa of the small intestine was segmentally reddened in 5 rabbits (cases 3, 4, 6, 10, 11), and the cecum was also reddened in 3 rabbits (cases 4, 10, 11). Case 2 had mucoid intestinal contents. The lungs of 8 of the rabbits were mottled and appeared edematous (cases 1–6, 8, 10). Specimens were obtained from 9 rabbits for aerobic and anaerobic bacterial culture (cases 2, 4–6, 8–12) and from case 1 for serologic testing for E. cuniculi. E. coli was cultured from the intestinal tract of 4 rabbits (cases 8–11) and from the kidney of case 5. Two isolates were submitted for further characterization (cases 5, 11). Unfortunately, the isolate from case 5 failed to grow and was lost prior to characterization. Serotyping and PCR from 1 E. coli isolate (case 11) yielded a O128:H2 serotype that was positive for eae, the gene for the intimin adhesion molecule, and was Shiga toxin (Stx) negative. The O128:H2 serotype has been characterized as an EPEC. 14 Klebsiella oxytoca and Citrobacter spp. were also isolated from the intestinal tract of 2 of the E. coli–positive rabbits (cases 10, 11). No Clostridium, Campylobacter, or Salmonella species were isolated. The fifth rabbit (case 10) from which cultures for bacteriology were obtained had been treated with metronidazole for 4 d prior to death. Heavy growth of Bacteroides fragilis was isolated from the intestine of the sixth rabbit (case 12). Serology results were negative for E. cuniculi.
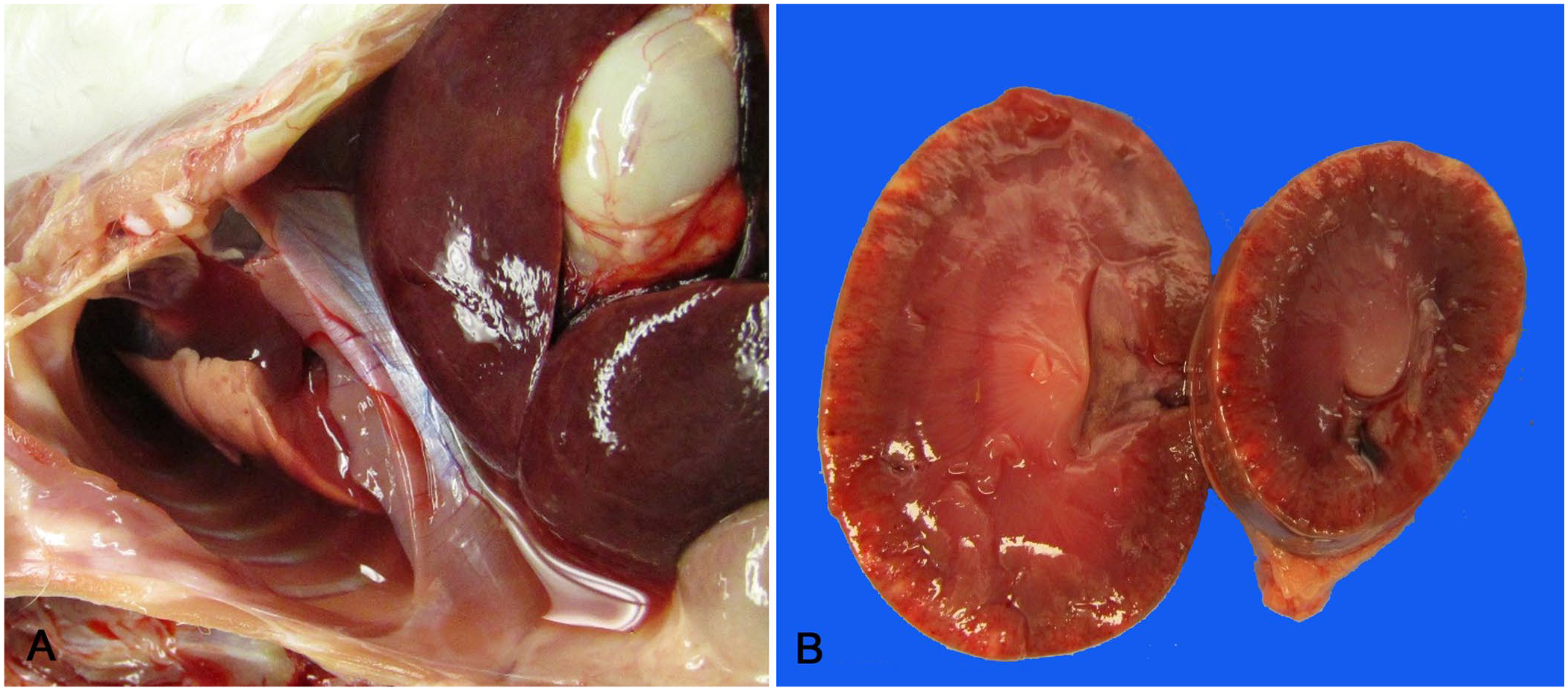
Gross findings in New Zealand white rabbits with enteritis-associated nephropathy.
Microscopic evaluation of the kidneys revealed changes in all rabbits ranging from mild focal segmental glomerular hypercellularity to diffuse glomerulopathy with expansion of the mesangium and mesangiolysis, thickening of capillary walls and duplication of basement membranes, thrombosis, and hemorrhage and fibrin exudation into the urinary space. Tubular changes included dilation, with variable vacuolation of epithelial cytoplasm and epithelial necrosis. Low numbers of heterophils and lymphocytes were variably present in the interstitium (Fig. 2).
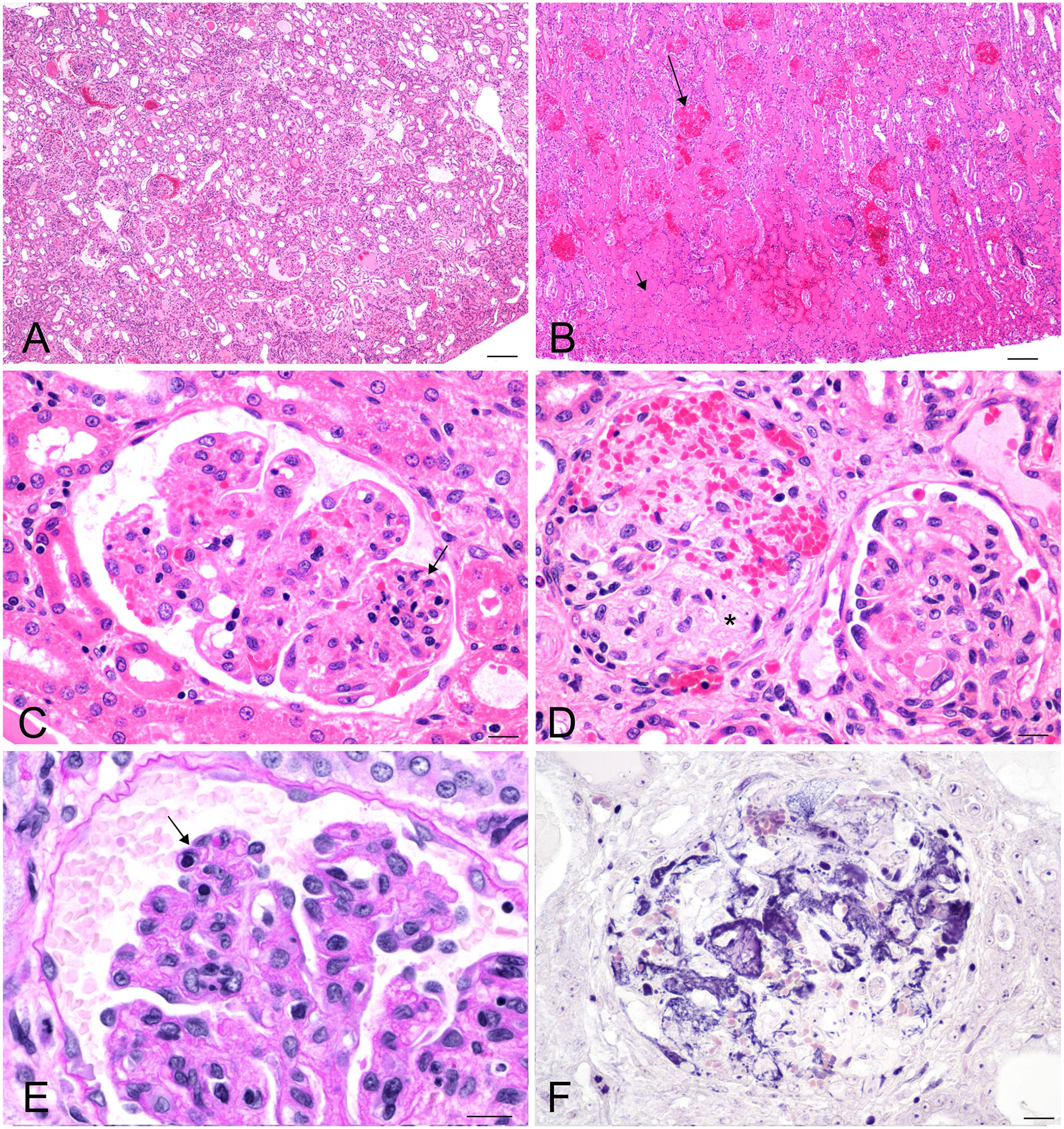
Kidneys from New Zealand white rabbits with enteritis-associated nephropathy, 4–5-µm sections.
Various amounts of fibrin, edema fluid, and hemorrhage were observed in the airways and alveolar spaces of 10 of 12 rabbits (cases 1, 3–10, 12; Fig. 3A). Low-to-moderate numbers of heterophils were present in the alveolar septa and alveolar spaces. Changes in the intestine of 7 of the rabbits (cases 1–3, 9–12) ranged from a mild increase in mixed inflammatory cells in the lamina propria of the small intestine (case 10) to severe acute necrotizing enteritis and typhlocolitis with mucosal and submucosal edema and rare hemorrhage (case 11). Colonies of gram-negative bacteria were observed adhered to enterocytes in the intestine of 7 rabbits (cases 2, 6–11; Fig. 3B). Giemsa and Warthin–Starry silver stains on the intestines did not reveal additional pathogens. Postmortem autolysis complicated evaluation of the intestines of the remaining rabbits.
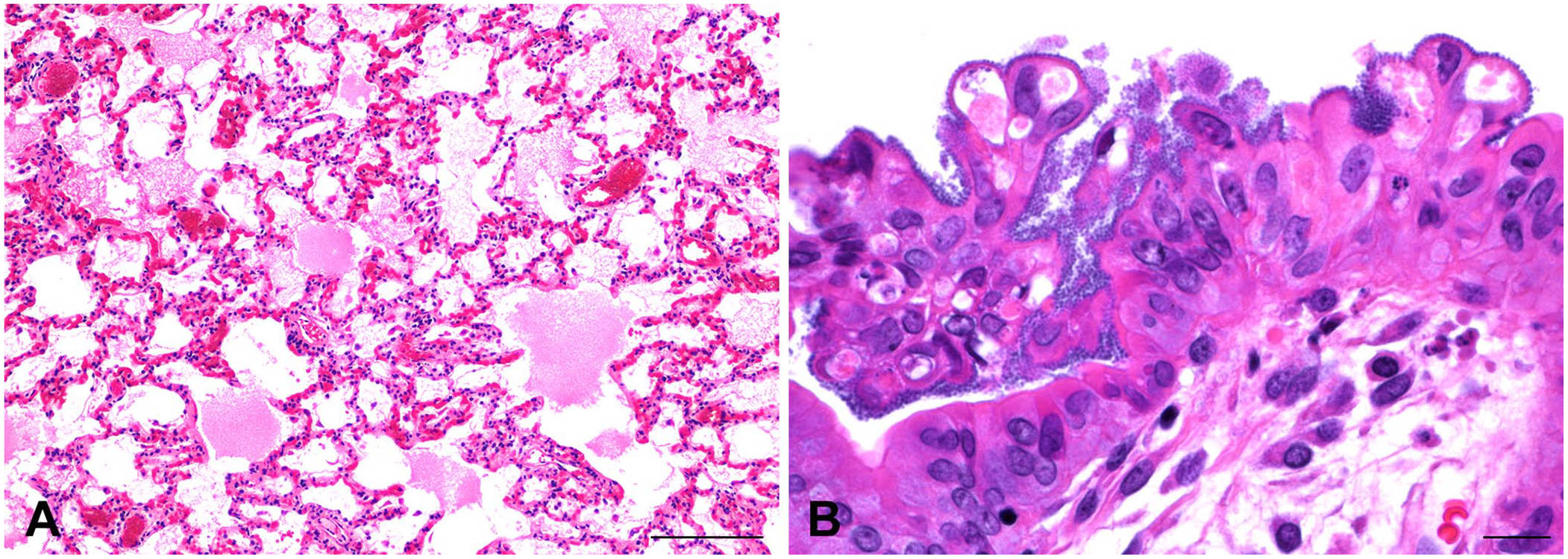
Tissues from New Zealand white rabbits with enteritis-associated nephropathy.
HUS is a thrombotic microangiopathy and is characterized by the triad of thrombocytopenia, anemia, and acute renal disease with endothelial cell injury and thrombosis. Chronic lesions may exhibit multilayered capillary basement membranes. HUS typically follows enterocolitis associated with infections of Stx-producing organisms such as Shigella dysenteriae type 1 or E. coli O157:H7; however, other infectious agents such as Streptococcus pneumoniae, Legionella pneumophila, and B. fragilis have also been associated with HUS.5,6,11 Additional causes of HUS include genetic dysregulation of the complement cascade, a variety of systemic conditions, such as autoimmune disease or neoplasia, and exposure to a variety of drugs. 6
Changes consistent with HUS have been reported in Dutch Belted rabbits infected with E. coli O157:H7, 8 and we observed similar lesions in the kidneys and intestines of the NZW rabbits of our report. However, only case 7 had thrombocytopenia (Suppl. Table 1); therefore, most of our cases do not fully fit the criteria for HUS. However, our cases do fit a diagnosis of partial HUS, in which the platelet count is in the normal range, and lesions, including renal damage, are similar to HUS. 18 Our cases also differed from the Dutch Belted rabbits in having serosanguineous fluid in the body cavities as well as pulmonary edema, most likely related to hypoalbuminemia (cases 5, 7, 8; Suppl. Table 2). E. coli was isolated from 5 of 6 samples submitted for culture (cases 5, 8–11); the isolate from case 11 was serotyped as O128.H2, an EPEC, rather than O157:H7, an EHEC. The E. coli isolated from case 11 may have produced Stx initially, but lost the ability as a result of excision of stx-bearing bacteriophages during the course of infection. 2 It is also possible that endotoxin, rather than Stx, was responsible for the nephropathy observed in our cases. B. fragilis, isolated from case 12, is a gram-negative organism that also possesses an endotoxin and has been associated with nephropathy.11,20 In an experiment using male NZW rabbits, a 5-h infusion of E. coli endotoxin produced acute renal failure associated with anemia and decreased platelet numbers. Within the glomeruli, capillary endothelial damage was followed by transient heterophilic infiltration, thrombosis, and fibrin deposition. Moderate degenerative changes were observed in the renal tubules. 1 It is possible that longer exposure to endotoxin results in more severe renal damage, as observed in our cases.
E. cuniculi is often associated with chronic renal disease in rabbits, and the lesions produced by this organism grossly resemble the kidney changes observed in our cases. However, our cases show that gram-negative enteric bacteria should be included in the differential diagnosis of similar renal disease, especially when there is an accompanying clinical history of gastrointestinal disorder.
Supplemental Material
sj-pdf-1-vdi-10.1177_10406387221115139 – Supplemental material for Acute renal injury from thrombotic microangiopathy associated with enteritis in New Zealand white rabbits
Supplemental material, sj-pdf-1-vdi-10.1177_10406387221115139 for Acute renal injury from thrombotic microangiopathy associated with enteritis in New Zealand white rabbits by Kimberly S. Waggie, Jessica M. Snyder and Piper M. Treuting in Journal of Veterinary Diagnostic Investigation
Footnotes
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
