Abstract
Toxicologic pathologists must evaluate tissues of immature animals from a number of types of nonclinical toxicity studies. The pathologist who is familiar with normal postnatal organ development is in a better position to appropriately detect and differentiate between abnormal, delayed, or precocious development. Vacuolation and apoptosis in multiple tissue types are normal components of development that could influence the interpretation of some tissues. Unique postnatal features such as the germal matrix in the brain, gonocytes in the testes, and saccules in the lung may complicate the histopathological evaluation. With the knowledge of normal organ development and critical windows therein, it is possible to design targeted studies to identify xenobiotic toxicity.
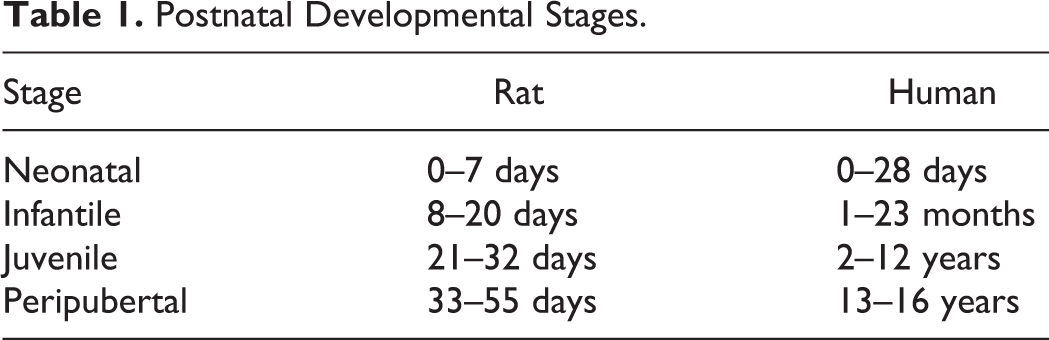
Toxicologic pathologists often evaluate tissues from immature animals in certain types of nonclinical safety toxicity studies, such as juvenile toxicity studies, pubertal and thyroid function assays, developmental neurotoxicity studies, developmental immunotoxicity studies, and multiple types of developmental and reproductive toxicology studies. Immature animals refer to animals during any of the defined postnatal developmental stages, which include neonatal (birth to postnatal day [PND] 7), infantile (PND 8–20), juvenile (PND 21–32), and peripubertal (PND 33–55) stages (Table 1). The term “juvenile” is commonly used in a general rather than specific sense to refer to animals at any one of these stages, and pathologists reading the literature should be aware of this common usage. When a pathologist examines tissues from immature animals, often those tissues are from unscheduled death animals, for which there are no concurrent controls for comparison purposes. Without age-matched concurrent controls, the pathologist must be knowledgeable about organ development in order to appropriately detect precocious or delayed (yet otherwise normal) development and differentiate this from abnormal development. The former generally indicates endocrine disruption and/or stress, while the latter commonly indicates direct toxicity. The pathologist who is knowledgeable about organ development will be in a position to help design targeted studies to identify test substance–related toxicity.
Postnatal Developmental Stages.
This article will provide a concise overview of development of several organ systems, focusing on those changes or findings that could be misinterpreted as toxicity. Table 2 is an overview chart of the postnatal date when selected organs achieve adult histomorphology.
Timinga for Selected Organs to Reach Adult Histologic Maturity.
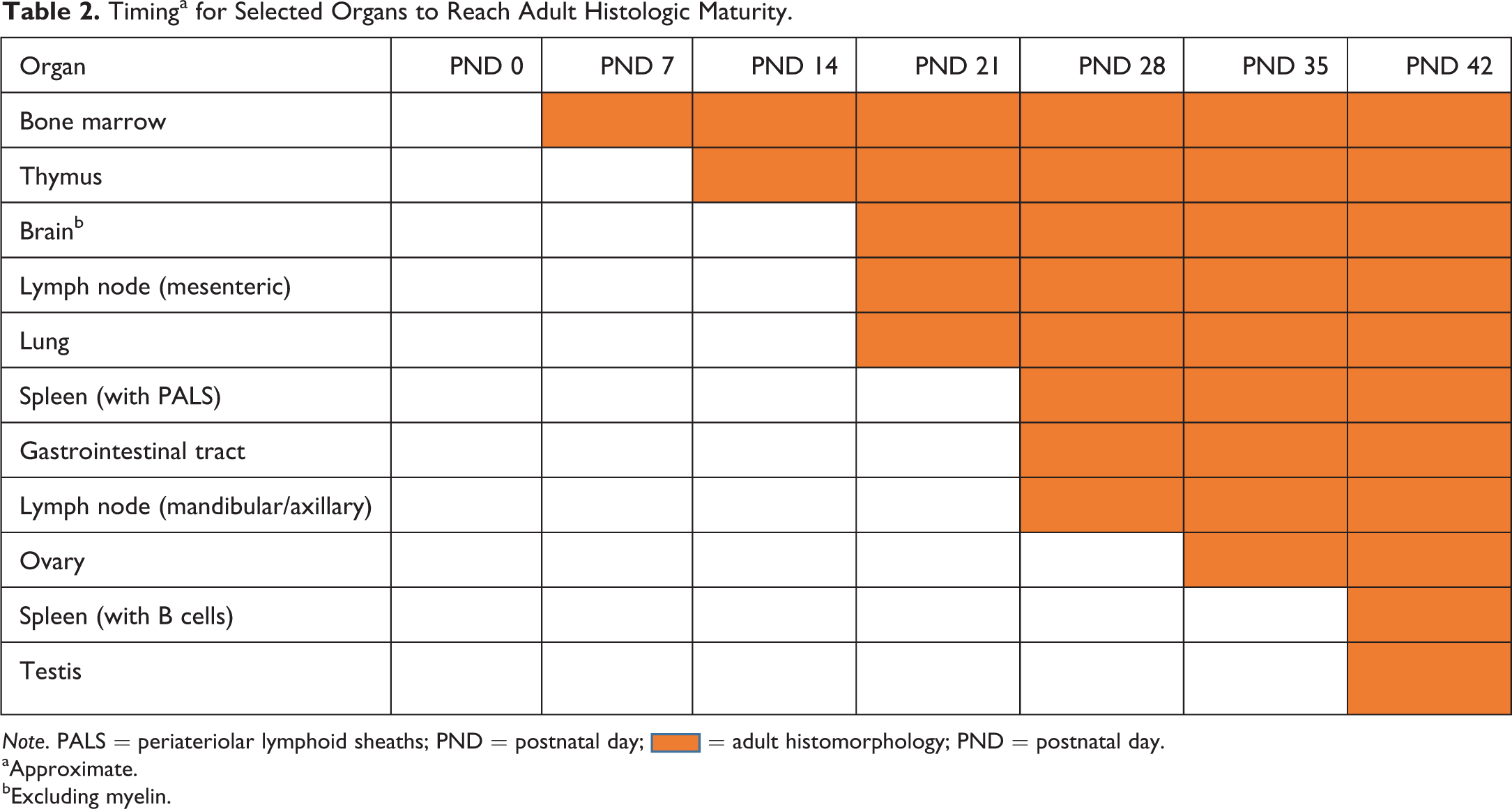
Note. PALS = periateriolar lymphoid sheaths; PND = postnatal day;  = adult histomorphology; PND = postnatal day.
= adult histomorphology; PND = postnatal day.
aApproximate.
bExcluding myelin.
Methods
All animals used in this study were acquired from a licensed laboratory animal vendor and were housed and maintained in an Association for Assessment and Accreditation of Lab Animal Care International (AALAC)-accredited laboratory animal facility. The procedures followed in the study were compliant with the facility’s standard operating procedures, and the study protocol was approved by the facility’s Institutional Animal Care and Use Committee.
Results
Brain
The rat brain undergoes profound postnatal development. At birth, virtually all of the neurons in most sites are formed and have migrated into position; yet, there are three regions of the brain with significant postnatal neuronal development. These include neurons of the granular layer of cerebellum, the olfactory bulbs, and dentate gyrus of the hippocampus. The brain appears histologically mature by PND 21, even though myelination is not complete until PND 37. Apoptosis associated with normal neuronal pruning can be profound and presents a significant complicating factor in interpreting brains from animals from birth through PND 14. There is massive apoptosis of neurons in the cortex, thalamus, superior and inferior colliculi, and deep cerebellar nuclei from PND 1 to PND 14, with a peak in apoptosis at PND 7 (Figure 1). In addition, the pathologist should expect to see unique structures in the developing brain. Primary and secondary germinal matrices are zones of densely packed neuroblasts lining the ventricles and covering the superficial surface of the cerebellum, respectively (Figure 2). The primary germinal matrix gives rise to the neurons of the hippocampus. It also protrudes into the olfactory bulbs as the rostral migratory stream which is the subregion of the primary germinal matrix that gives rise to neurons of the olfactory bulb. Primary germinal matrix and the rostral migratory stream should be expected to be visible up until PND 14. The secondary germinal matrix gives rise to the granular cell neurons of the cerebellum that develop postnatally. This layer of germinal matrix expands at PND 7 and only thereafter dwindles away by PND 21. At PND 21, only a small amount of germinal matrix may remain on the lateral aspects of the cerebellum. Vacuolation of choroid plexus, especially within the lateral ventricles, is prominent at birth and may remain until PND 14 to a small degree (Figure 3). Vacuolation of this structure should not be interpreted as a lesion in these neonatal and infantile animals.
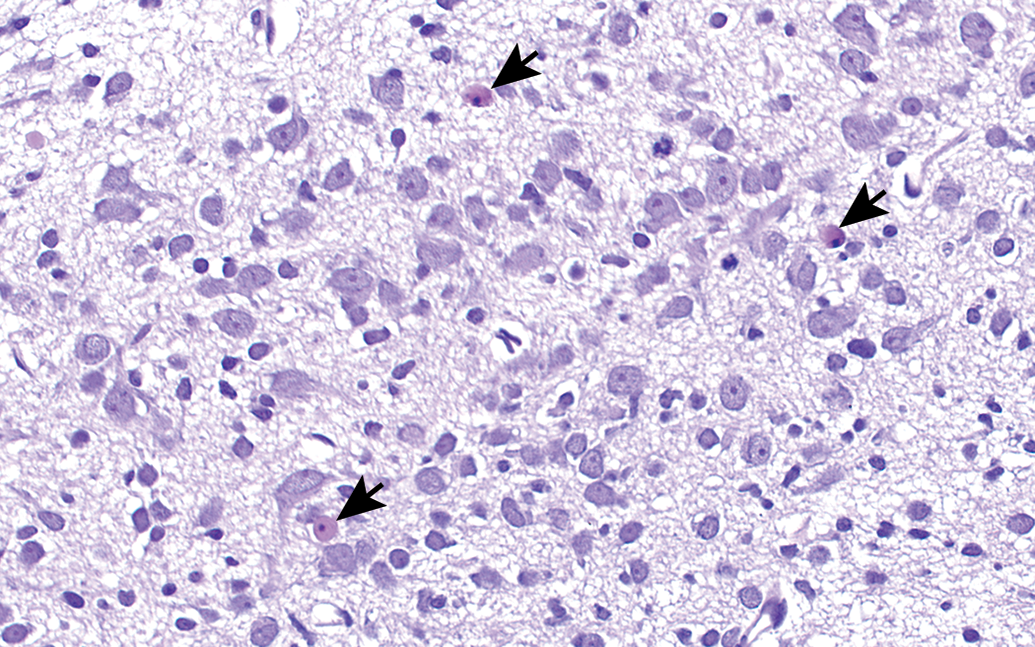
Deep cerebellar nucleus of rat brain at postnatal day 7. Note the apoptotic neurons (arrows), which are considered normal for this stage of development. H&E. Original objective 40×.
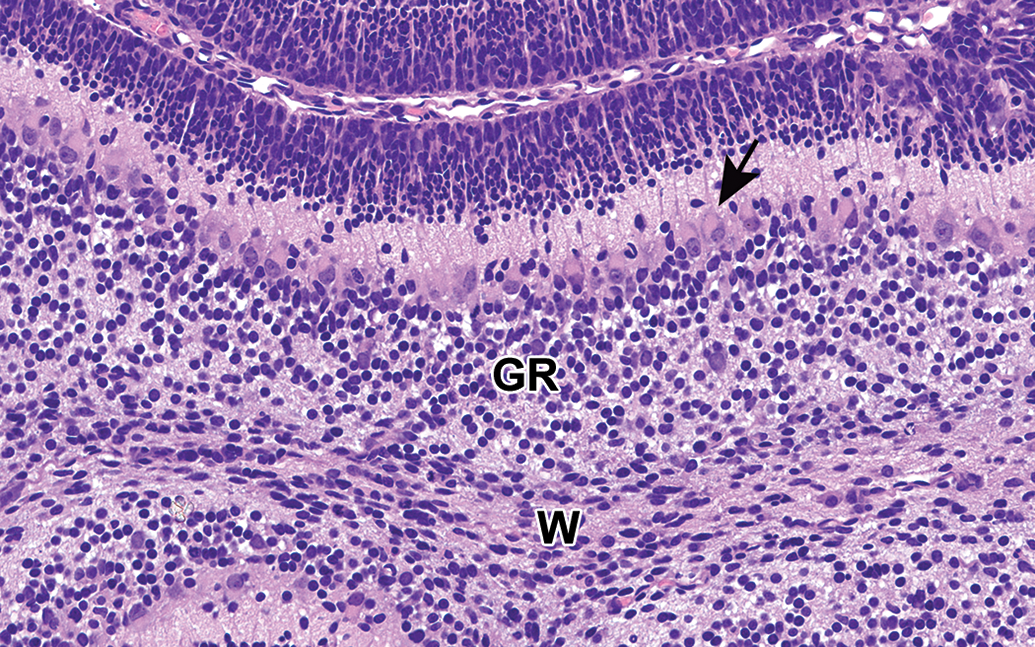
Cerebellum from rat brain at postnatal day 7. The secondary germinal matrix lies beneath the pia mater. Neuroblasts within this matrix develop into granular cell neurons and migrate to the granular cell layer. Many Purkinje cells may have eosinophilic intracytoplasmic globules (arrow) at this stage. W = white matter, GR = granular cell layer. H&E. Original objective 40×.
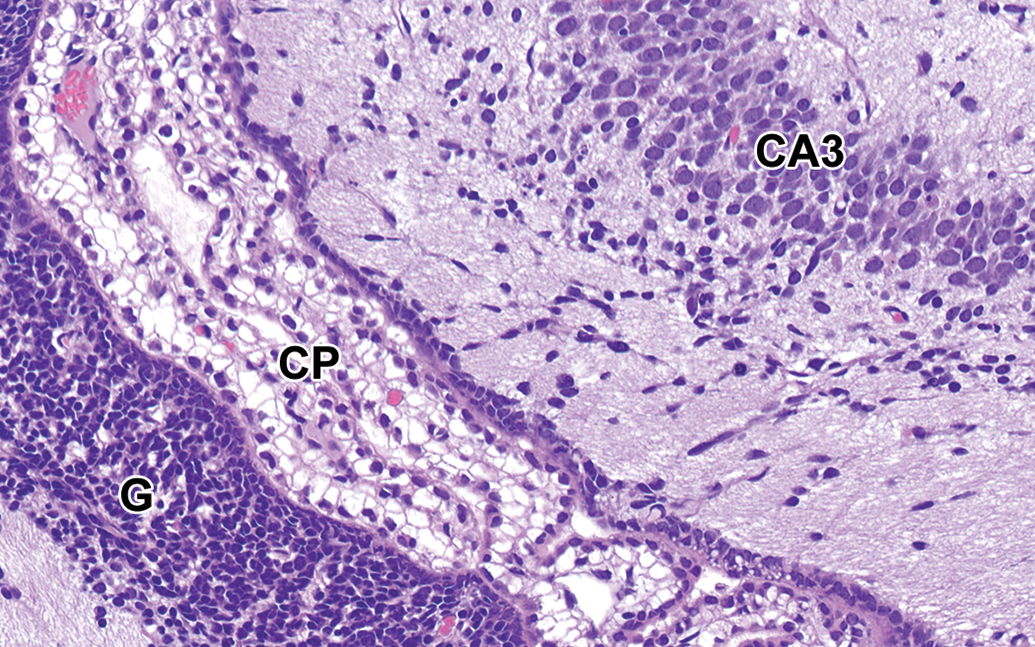
Rat brain at postnatal day 1. The choroid plexus within the lateral ventricle is composed of highly vacuolar epithelium. There is prominent primary germinal matrix at this time, and this germinal matrix gives rise to neurons of the dentate gyrus. Region III of the hippocampus proper (CA3) is present. CP = choroid plexus, G = germinal matrix. H&E. Original objective 40×.
Testis
The immature rat testis also presents diagnostic challenges. The period between PND 5 and PND 18 is considered to be a critical window because the testis at this stage has mitotically active spermatogonia, Sertoli cells, and Leydig cells. The spermatogonia and Sertoli cells are highly visible as the predominant cell types and form multiple cell layers within the seminiferous tubules. This period could be opportune to perform and evaluate short-term targeted studies to identify toxicity against these susceptible and visible cell populations. If such studies are performed, the pathologist must be aware that spermatogenic epithelium is not organized in the same way as in the adult testis. In immature seminiferous tubules, spermatocytes (from preleptotene through pachytene) constitute the innermost cell layers of the tubules, and also Sertoli cell nuclei are not in the expected location at the base of the germinal cell layers. This more luminal location for spermatocytes and Sertoli cells is in contrast to their more basilar location in adult seminiferous tubules.
Apoptosis is a normal feature of the developing testis as in the brain. Apoptosis of spermatogonia and Sertoli cells occurs as a result of high cell density during the late infantile period (PND 15–18). Hormonally dependent apoptosis affects pachytene spermatocytes during the juvenile period (approximately PND 22–27). Apoptotic cells may be difficult to distinguish from the developing germ cells: preleptotene, leptotene, and zygotene spermatocytes have chromatin patterns that could appear karyorrhectic or apoptotic. In particular, preleptotene spermatocytes have unusually coarse, clumped chromatin, and leptotene and zygotene spermatocytes have nuclei with very dense chromatin. Given the more central and visible position for these developing spermatocytes in the tubules, one should not mistakenly identify these cells as necrotic or apoptotic. A paucity of Leydig cells may be expected from PND 10 to about PND 25, when the robust testosterone-producing fetal Leydig cells are no longer present, and the small progenitor and immature Leydig cells are still maturing. This paucity of Leydig cells should not be misconstrued as atrophy or an indication of hormonal disruption.
Lung
The lung of the rat is in a saccular stage from birth to PND 4, and its light microscopic appearance should not be confused with inflammation or atelectasis. The wall of the saccule is thick, with abundant mesenchyme and fibroblasts, and flanked on each side by a capillary. From PND 4–21, the saccule undergoes deconstruction (with apoptosis and thinning of the mesenchyme) and reconstruction (with formation of primary and secondary septa). During this construction process, termed bulk alveolarization, it is not uncommon for saccules and alveoli to coexist (Figure 4). The interstitium of these developing alveolar walls appears foamy due to the presence of glycogen-filled progenitor type 2 cells. Microvascular growth is superimposed on the process of alveolarization. Capillaries form by a process called intussusceptive growth, and at PND 14 one may expect to see an abundance of empty capillaries that have yet to receive blood supply, which contributes to the vacuolar or foamy appearance to the tissue. The lung reaches histologic maturity by PND 21.
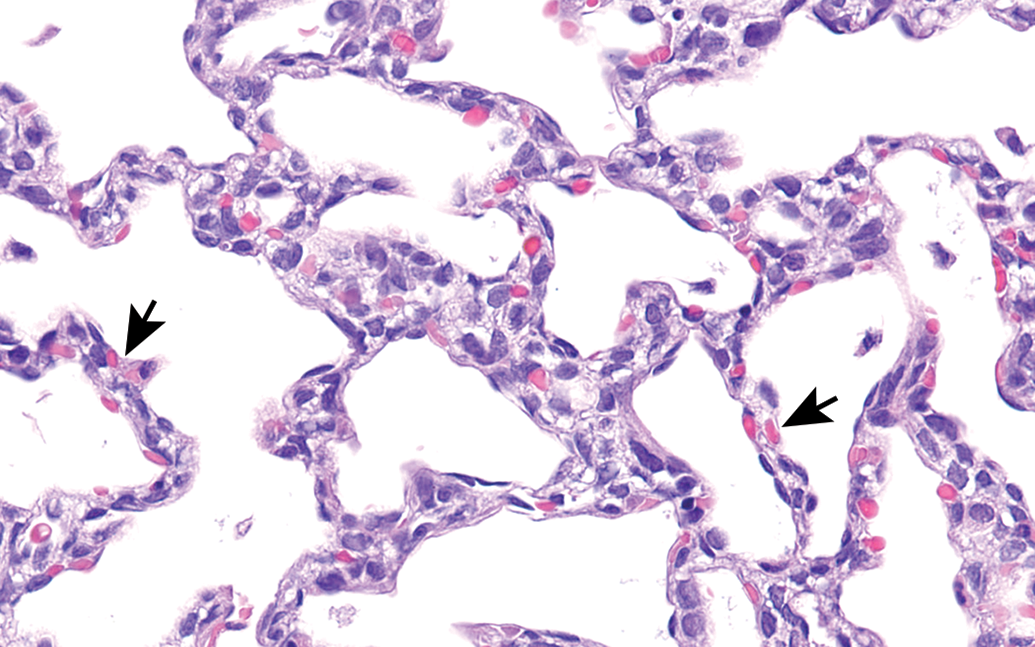
Lung from rat at postnatal day 7. Most of the airspaces are still saccules at this stage with double capillary walls (arrows). The foamy appearance of the interstitium is due to glycogen-filled progenitor type 2 cells. H&E. Original objective 40×.
Ovary
The rat ovary also presents diagnostic challenges to the pathologist who is unfamiliar with immature tissues. The ovary begins to form tertiary follicles with antral fluid after PND 10, yet these tertiary follicles have unique features that are unlike the early tertiary follicles in the adult ovary. The early tertiary follicles in the immature ovary have granulosa cells with poor adhesion and oocytes devoid of a zona pellucida (Figure 5). Hormonally dependent atresia of follicles can be massive around PND 25–27, and this process, while required for the depletion of redundant follicles in the center of the ovary, should be recognized as normal. Corpora lutea appear by approximately PND 38, indicating previous ovulation, yet the vagina may “histologically mature” prior to this time. Typical histologic features of proestrus or estrus stage vaginal lining may be present by PND 32, almost 1 week prior to ovulation or formation of corpora lutea in the ovary. The discrepancy between the vagina and ovary should be expected and not misconstrued as endocrine imbalance.
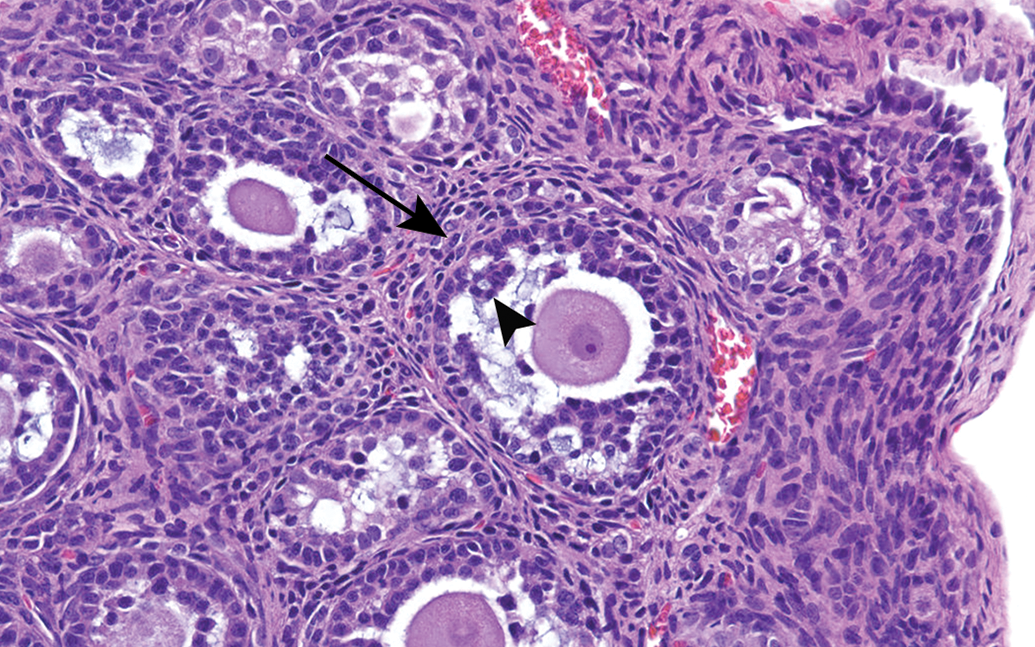
Ovary from rat at postnatal day 13. Note the atypical early tertiary follicle. This follicle is characterized by granulosa cells with poor adhesion (arrow head) and little discernment between the theca interna and the theca externa (arrow). The oocyte is devoid of a zona pellucida. H&E. Original objective 40×.
Gastrointestinal (GI) Tract
The GI tract presents some unique histologic features that should not be confused with toxicity. The stomach is “bland” with pH 7 from birth to PND 7, absence of mucus producing cells, and nonproductive parietal cells. The pH slowly declines to pH 4 by PND 21 and then to pH 2 by PND 42, as parietal cells (producing HCl) and chief cells (producing pepsin) develop. The lack of pepsinogen and HCl, along with deficient production of pancreatic enzymes before PND 14, allows for normal colonization of the mucosal surface by bacteria. During this early time of limited protein or lipid digestion, there is a physiologic need for intestinal epithelial cells to resorb intact protein and lipid. Such resorption results in formation of vacuoles in the intestinal tract that could be confused with toxicity. Clear lipid resorption vacuoles are common at the base of villi in the duodenum at PND 0–2 and near the tips of villi after PND 10. Vacuoles filled with a homogenous eosinophilic globules are common in the duodenum and ileum from PND 0–14 and represent receptor-mediated endocytosis of protein, much of which is intact maternal immunoglobulin (Figure 6). Clear vacuoles containing a dense eosinophilic central globule occurs in the ileum, cecum, and colon through PND 14, and these types of vacuoles represent nonspecific resorption of protein followed by intracytoplasmic lysosomal digestion of this protein (Figure 7). The intracellular digestion results in the clear space around a central dense eosinophilic globule. Intracellular digestion of protein in the ileum is required to compensate for inadequate production of proteases by the pancreas and inadequate production of pepsinogen by the stomach.
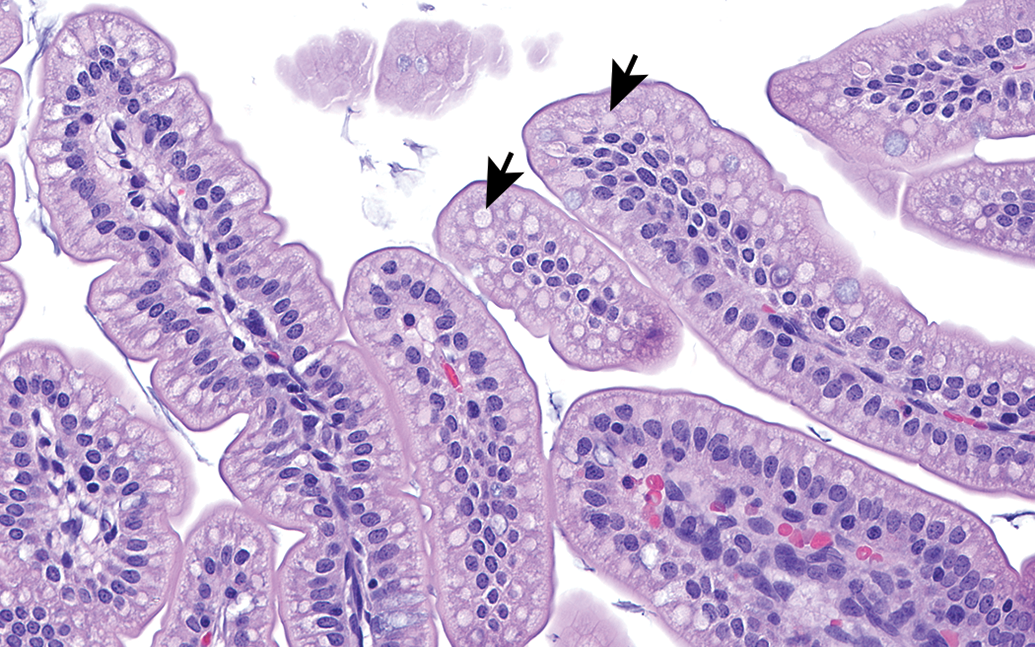
Jejunum from rat at postnatal day 7. The villus epithelium contains abundant vacuoles filled with pale eosinophilic protein (arrows). These vacuoles represent the result of receptor mediated endocytosis of intact protein. H&E. Original objective 40×.
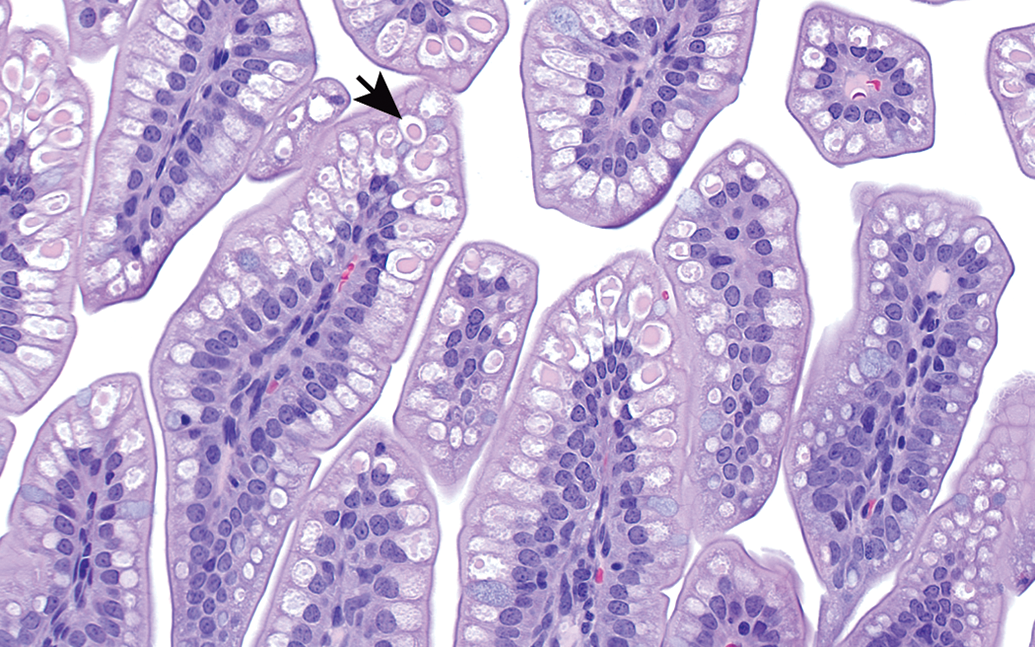
Ileum from rat at postnatal day 7. The villus epithelial cells have clear vacuoles (arrow), each containing a central condensed eosinophilic globule. This is the result of nonspecific absorption of intact protein (globule) followed by intracytoplasmic digestion due to lysosomal hydrolases (clear space). H&E. Original objective 40×.
The development of the GI tract is under control of glucocorticoid and thyroxine, and in instances of stress, precocious development (or maturation) of the mucosa may be expected. Stress-induced precocious development may be an evolutionary safeguard when pups in the wild are orphaned, and under this circumstance, hastened GI development would provide a survival advantage. By PND 21 when the pH is approximately 4, the entire GI tract develops a hyperplastic appearance, which resolves by PND 28. This appearance of hyperplasia is due a deep basophilia of the mucosa with a high mitotic rate affecting chief and parietal cells in the stomach, crypt cells in the small intestine, and mucosal epithelial cells of the cecum and colon. This appearance of hyperplasia should be recognized as normal postnatal development and not mistaken for a xenobiotic-related tissue alteration.
Lymphoid Organs
The primary lymphoid organs (bone marrow and thymus) undergo genetically programmed development early in the postnatal period. The bone marrow has essentially normal histomorphology by PND 7 and the thymus by PND 14. The secondary lymphoid organs (spleen, lymph nodes, and mucosa-associated lymphoid tissue [MALT]) have a basic genetically programmed structure with superimposed components that result from exposure to environmental influences. The earliest and most pronounced antigenic load is from the GI tract; therefore, mesenteric lymph nodes are the first to acquire “adult” histomorphology by PND 21. Secondary lymphoid organs, such as the spleen and somatic lymph nodes, which are sheltered from the antigenic load from the GI tract, develop subsequently. Immune system organs have histologic features of maturity at PND 42, but there is imperfect correlation between histological and functional indicators of maturity in immune system organs.
Summary
In order for a pathologist to evaluate and interpret histologic changes in tissues from juvenile animals, it may be beneficial to include additional control group animals that could be sacrificed as age-matched control animals in the event of early deaths on a given study. In addition, laboratories should consider collecting digital scans of tissues from control animals from various ages to form a resource library of control tissues that could be used for comparison purposes.
Footnotes
Author Contributions
All authors (CP, GP) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
