Abstract
Vascular injury can be induced by different classes of drug candidates, and it can affect the mesenteric vasculature. Sampling of the mesenteric vessels in the rat is crucial for proper assessment of potential adverse or pharmacologic effects of drugs in nonclinical rodent studies. To date, several sampling and processing techniques for the histopathologic evaluation of the mesenteric artery in rodents have been described and used in studies with candidate drugs that may affect the vascular system. However, most of those techniques require a significant amount of time and effort. A less labor-intensive, time-consuming, and expensive technique that allows examination of the mesentery vasculature with abundant longitudinal and cross sections of the vessels when examined microscopically was developed and presented here.
Vascular injury can be an adverse effect identified during nonclinical toxicity testing within certain pharmacological classes of drug candidates. A few of the better known examples of drug-induced mesenteric vascular injury that has been reported in rats include inotropic vasodilator (Westwood, Iswaran, and Greaves 1990), phosphodiesterase III and IV inhibitors (Joseph, Rees, and Dayan 1996; Zhang et al. 2002; Zhang et al. 2008; Weaver et al. 2008; Weaver et al. 2010), the K+ channel opener minoxidil (Tsoporis et al. 1993), and the dopaminergic receptor subtype agonists fenoldopam (Dalmas et al. 2008). Acute injury of the mesenteric arteries is characterized by increase in inflammatory cell infiltration (Weaver et al. 2010), arterial medial necrosis and inflammation (Westwood, Iswaran, and Greaves 1990; Dalmas et al. 2008), or medial necrosis and hemorrhage (Joseph, Rees, and Dayan 1996), while chronic injury includes wall thickening and vasculopathy (Westwood, Iswaran, and Greaves 1990). Accurate and reproducible sampling of the mesenteric vessels in the rat is crucial for proper assessment of potential adverse or pharmacologic effects of drugs in nonclinical rodent studies. Most of the techniques used for harvesting the mesenteric arteries in rats require some additional time and effort. A less labor-intensive, time-consuming, and expensive technique was developed that would also allow microscopic examination of the mesentery vasculature with abundant longitudinal and cross sections of the vessels.
Methods and Results
Twenty male and female Sprague-Dawley rats (Rattus novegicus, Harlan Labs; 16–24 weeks of age at the time of the necropsy) were cared for in accordance with the Guide for Care and Use of Laboratory Animals (National Research Council 1996). The animals were housed at an American Association for Accreditation of Laboratory Animal Care–accredited facility. The animals incorporated in this research were from the control groups (saline) from a Good Laboratory Practice compliant preclinical toxicity study with a test article (injectable administration) potentially targeting the blood vasculature. The study complied with the Animal Welfare Act Regulations (9 CFR) and U.S. Public Health Service Office of Laboratory Animal Welfare Policy on Humane Care and Use of Laboratory Animals. In the new described technique, the entire mesenteric arcade, with gastrointestinal tract attached, was collected at necropsy and laid out flat on a cutting board (Figures 1 and 2). The gastrointestinal tract and mesenteric lymph node were removed from the mesentery with the use of a scissors (blunt/blunt tip; Figure 3A), rolled up with the help of a small thumb-dressing forceps until little balls of tissue approximately 6 to 7 mm in diameter were formed (Figure 3B). The mesenteric rolls were placed into a small tissue cassette between 2 sponges (Figure 3C), fixed in 10% neutral-buffered formalin, embedded in paraffin, sectioned at 4 μm, and stained with hematoxylin and eosin (H&E) for light microscopic evaluation. A single section of the tissue on each glass slide contained cross, longitudinal, and tangential sections of several mesenteric arteries. This method standardized and optimized the light microscopic evaluation of numerous mesenteric arterial profiles per section (Figures 4 and 5).
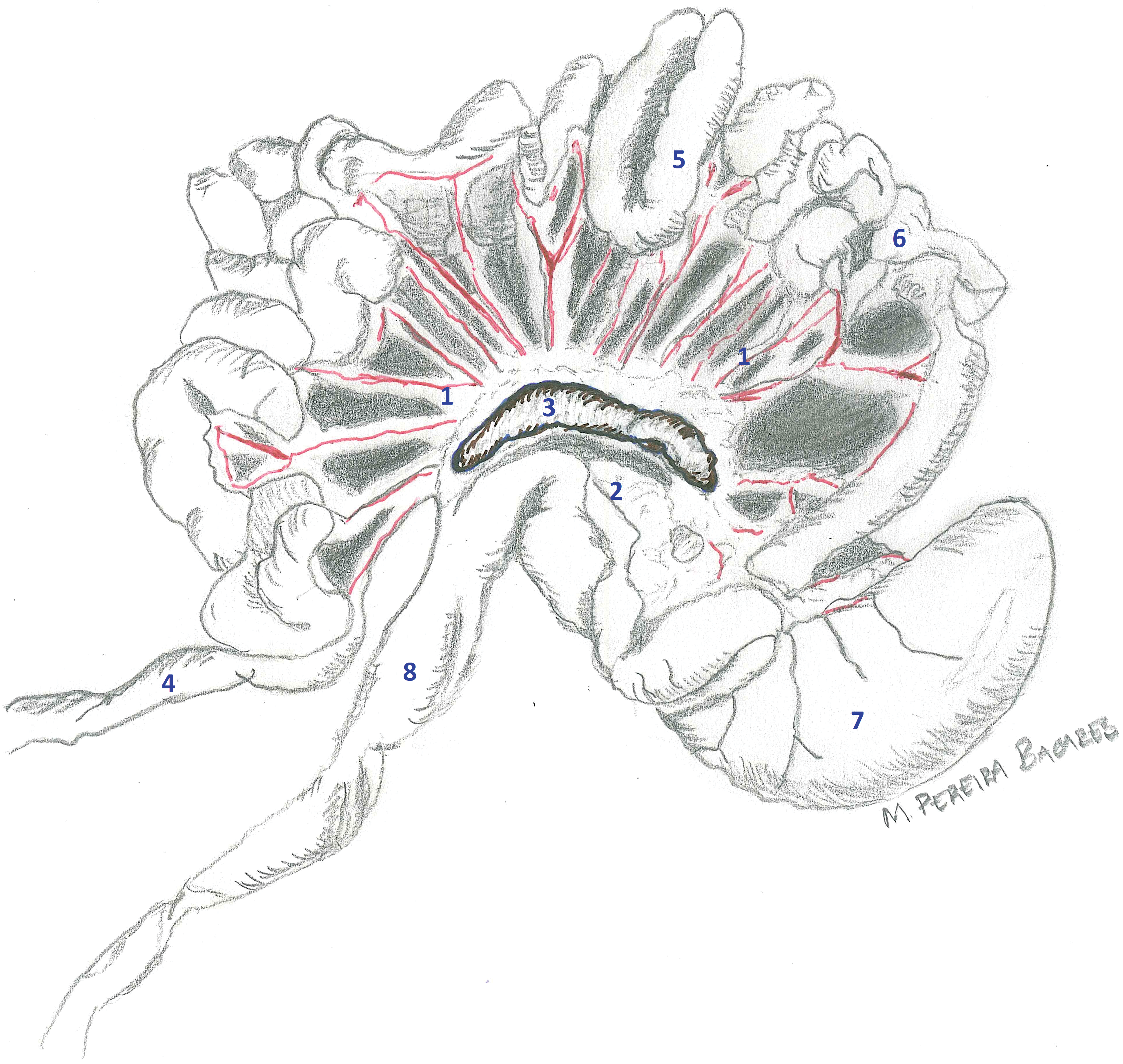
Gastrointestinal tract from a young adult male rat from a control group. The entire gastrointestinal tract was removed from the body cavity. Mesentery containing mesenteric arteries, veins, and lymphatics (1); mesenteric fat (2); and mesenteric lymph node (3) is still attached to the walls of jejunum (5); ileum (6); and cecum (7). Duodenum (4) and colon (8) are illustrated.
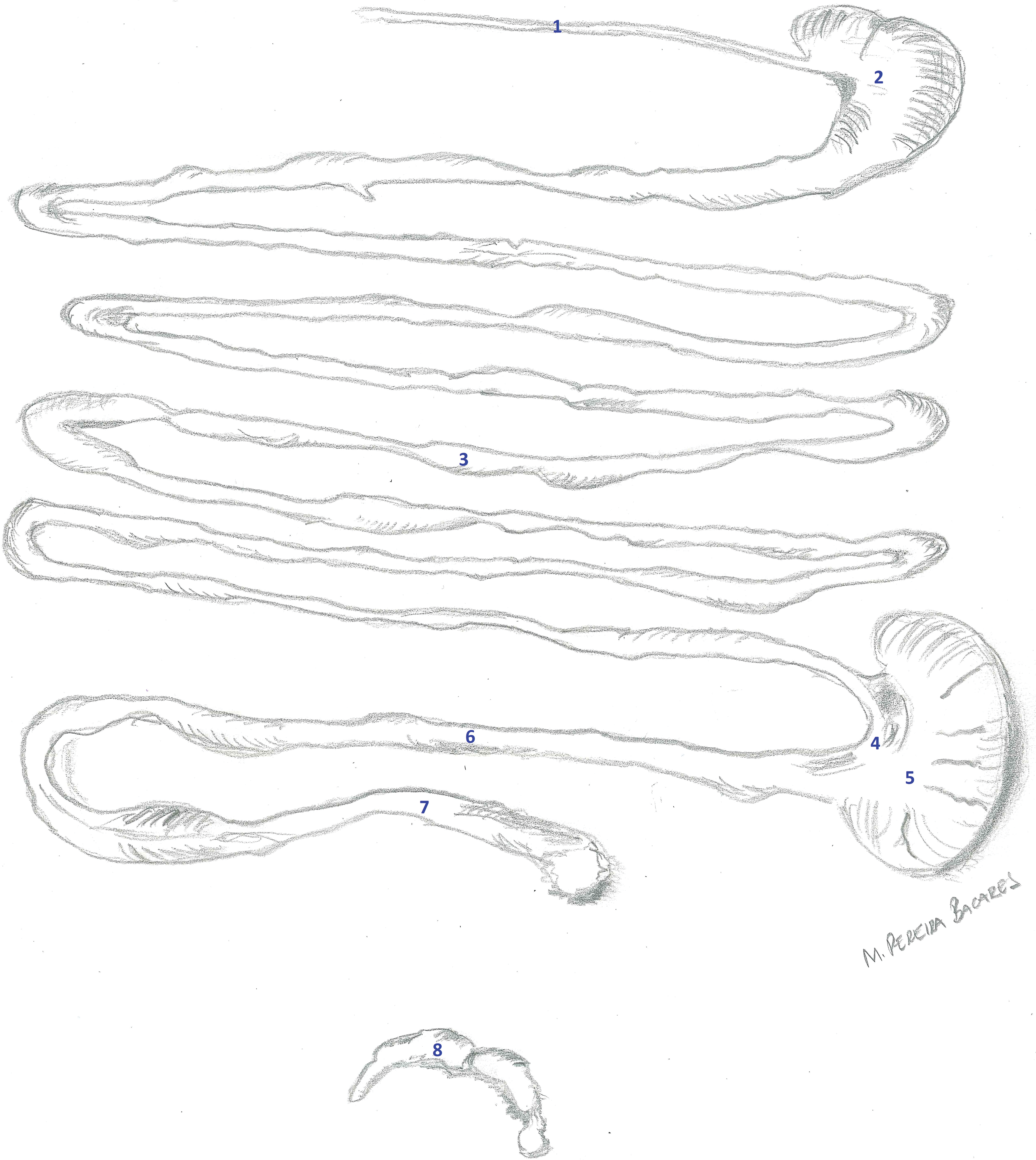
Gastrointestinal tract and mesenteric lymph node from a young adult male rat from a control group. The gastrointestinal tract and lymph node were removed from the mesentery. (1) Esophagus, (2) stomach, (3) small intestine, (4) ileocecal junction, (5) cecum, (6) colon, (7) rectum, and (8) mesenteric lymph node.
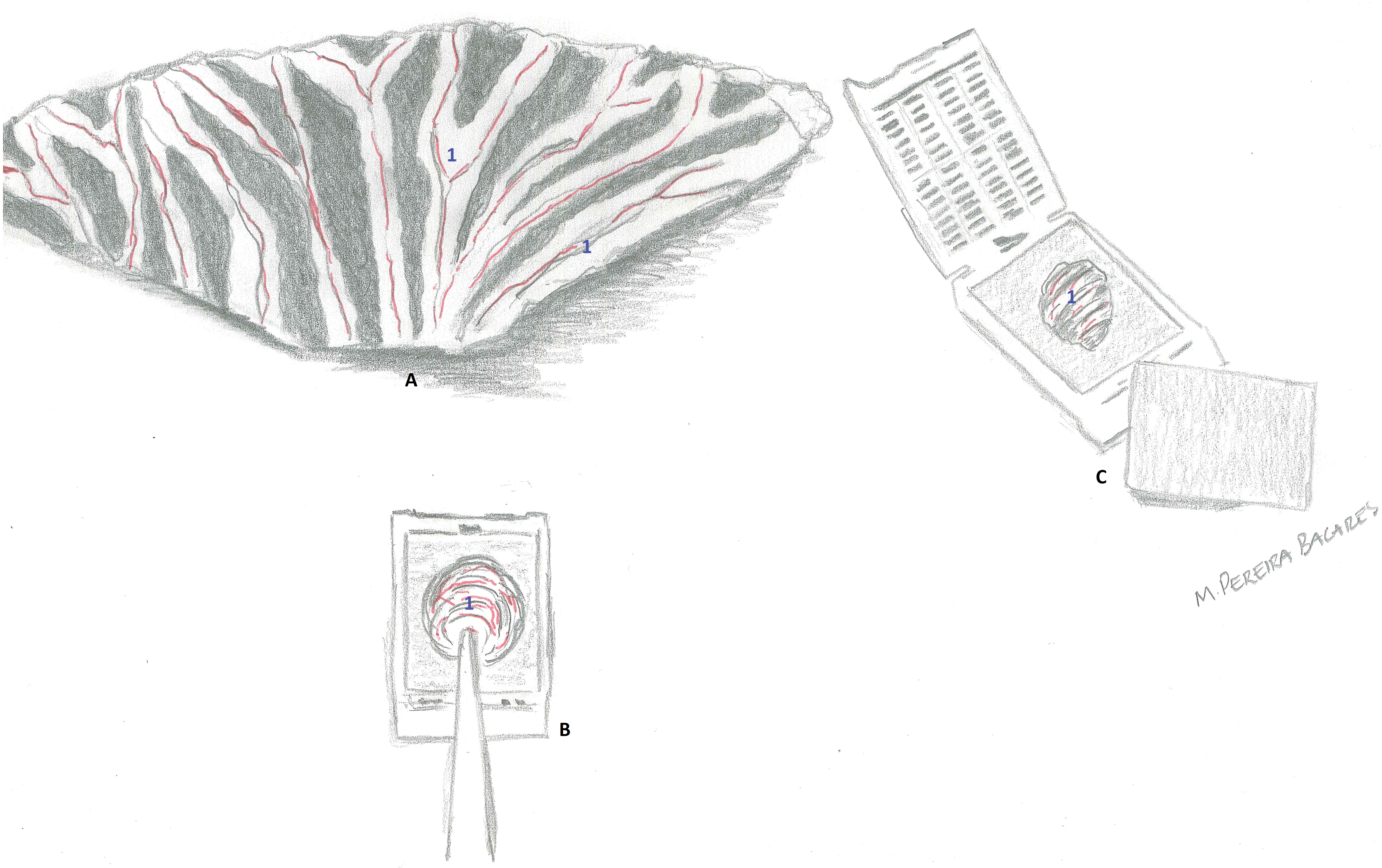
Mesentery from a young adult male rat from a control group. (A) The entire mesentery was freed from the gastrointestinal tract and is laid out flat on a cutting board. Mesenteric arteries (in red), lymphatics, and mesenteric fat (1) are evident. (B) The entire mesentery is rolled up with the help of a small thumb-dressing forceps until little balls of tissue were formed. (C) The mesenteric rolls were placed into a small tissue cassette between two sponges.
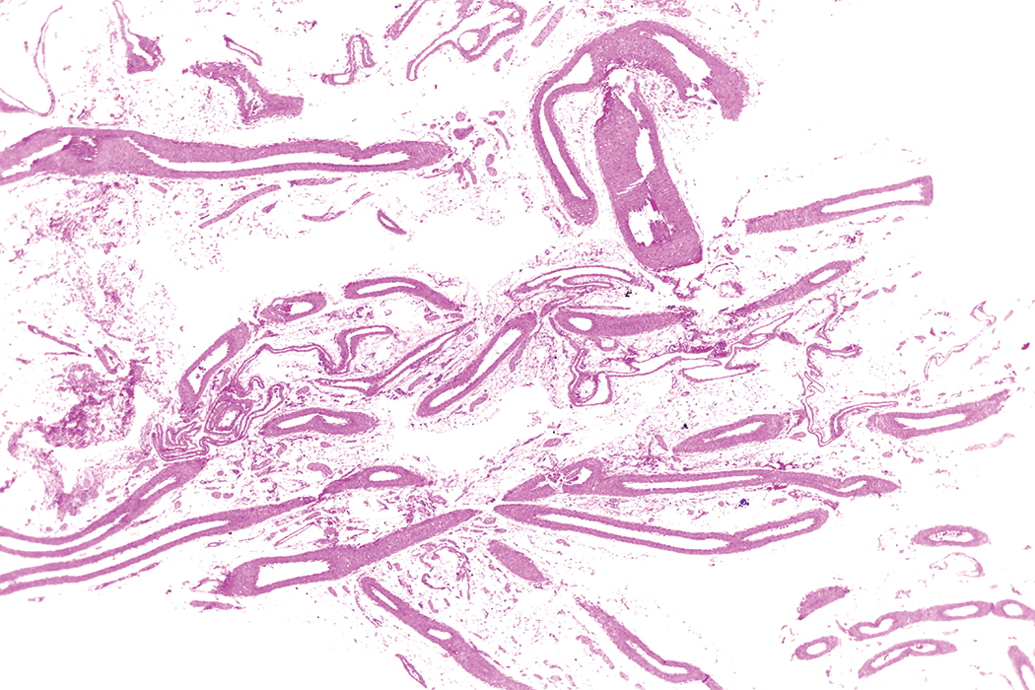
Lower magnification of longitudinal and cross sections of mesenteric arteries within the normal limits from a young adult male rat from a control group. Original objective 2×. H&E.
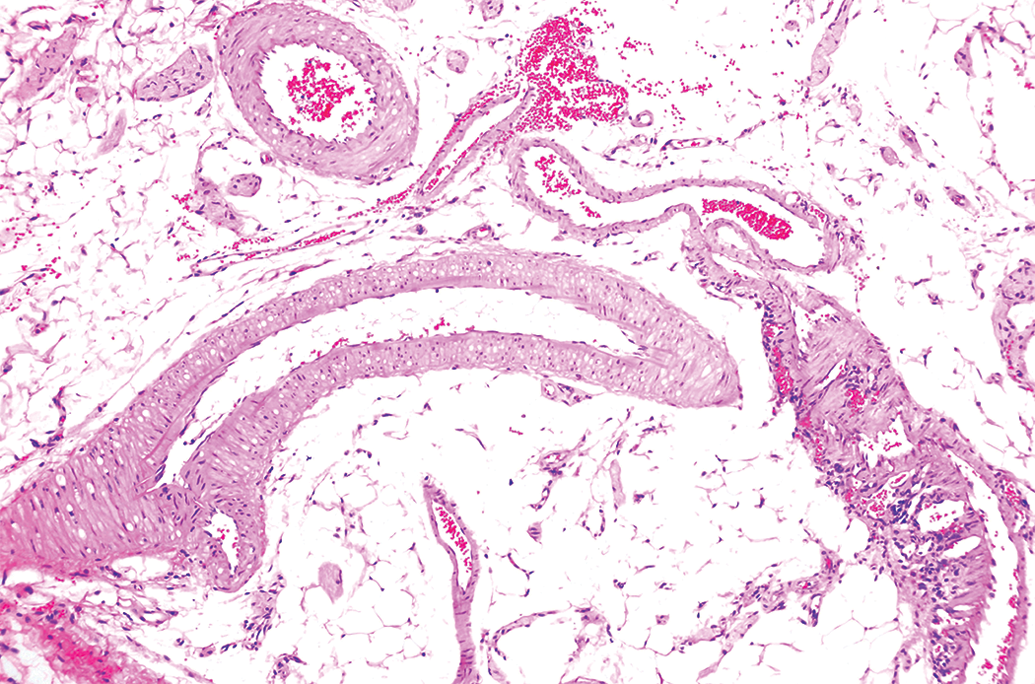
Higher magnification of longitudinal and cross sections of mesenteric arteries within the normal limits from a young adult male rat from a control group. Original objective 10×. H&E.
Discussion and Conclusion
To date, several sampling and processing techniques for the histopathologic evaluation of the mesenteric artery in rodents have been described and used in studies with candidate drugs that may affect the vascular system. Zhang et al. (2008) and Weaver et al. (2010) collected and fixed 3 fragments of the rat mesentery in 10% formalin solution, which were embedded in paraffin, sectioned, and stained with H&E for histopathological evaluation. Zhang et al. (2002) carefully spread the mesentery over a metal tray coated with wax, fixed the mesentery with 10% neutral formalin solution for 24 hr, sectioned the mesentery into 2 to 4 portions, embedded it in paraffin, sectioned the block at 5 μm, and stained the tissue with H&E. Similar to Zhang et al. (2002), Weaver et al. (2008) pinned out the mesentery at full extension, fixed it with 4% buffered formalin solution for 24 hr, sectioned the mesentery into 2 to 4 portions, and then subsequently embedded it paraffin, sectioned at 4 μm, and stained the tissue with H&E. Zhang et al. (2006) updated their technique by removing the entire small intestine with mesentery from the abdominal cavity and spreading it onto a wax-coated metal tray. Small intestine with the mesentery was then divided into 3 segments in the shape of a ring, each of which was secured on the tray using thumbtacks at the borderline between the intestine and mesentery. After fixation, the mesenteric tissue with a very narrow edge of intestine was saved for embedding in paraffin, in which the plane of unfolded mesenteric tissue was parallel to the cutting plane of a paraffin block, sectioned at 5 μm, and stained with H&E. Sheth et al. (2011) and McMahon Tobin et al. (2014) removed the gastrointestinal tract and laid it out flat to enable removal of the intestines and mesenteric lymph node. The mesentery tissue was rinsed with saline (NaCl 0.9%) and then rolled onto the plunger of a 1-cc syringe; the rolled tissue was placed in a small tissue cassette and fixed in 10% formalin for 24 hr prior to embedding in paraffin. The tissue was sectioned at 5 μm and stained with H&E. Dalmas et al. (2008) excised the entire mesentery, removed the mesenteric lymph, and cut in half the mesentery. Each half was rolled on a 3-mm-diameter plastic cylinder. One of the halves was placed in a Tissue-Tek® processing capsule, fixed in 10% neutral-buffered formalin, processed into paraffin blocks, sectioned at 5 μm, and stained with H&E. Newsholme et al. (2000) and Brott, Richardson, and Louden (2012) dissected the mesentery free from the viscera, rolled it on a 3-mm-diameter plastic cylinder, removed it from the cylinder, secured it in a plastic embedding cassette, and fixed it in 10% neutral-buffered formalin. Rolled mesenteries were routinely processed to 5-μm paraffin sections and stained with H&E. Joseph, Rees, and Dayan (1996) perfusion fixed the mesenteric vasculature with modified saline followed by Karnovsky’s fixative, spread the intestines on a cork board and held it in place by pins at the mesentery/intestinal border, and sampled up to 3 mesenteric arteries (20- to 40-mm longitudinal segments) subsequently obtaining 12 to 30 transverse serial 4-μm sections stained with H&E. Slim et al. (2003) sampled vasculature from the mesentery and secured it with staples to wax mounting strips, fixed the mesentery in 10% buffered formalin for 24 hr, embedded 3 representative samples in paraffin, sectioned at 3 μm, and stained the sections with H&E.
The new described technique is simpler than all other techniques used for the histopathologic evaluation of the mesenteric arteries in studies with candidate drugs that may affect the vascular system. One of the greatest advantages of this new technique is that the entire mesentery is sampled in a single roll, increasing the chances of having several cross, longitudinal, and tangential sections of several arteries in a single slide. Unlike some of the used techniques, where the mesentery needs to be spread over a tray for a 24-hr fixation in 10% formalin solution (Slim et al. 2003; Zhang et al. 2002; Zhang et al. 2006) or pinned out at full extension and fixed in 4% formalin solution (Weaver et al. 2008) or have the vasculature perfused with modified saline followed by Karnovsky’s fixative and have the mesentery pinned (Joseph, Rees, and Dayan 1996) prior sectioning, the new technique does not require any of these additional steps. The entire mesentery sampled in a single roll using only a pair of scissors to free the mesentery from the intestines and lymph nodes and a small thumb-dressing forceps to roll up the tissue, without any need for sectioning mesentery in half as in one technique (Dalmas et al. 2008) or rolling the tissue onto a plunger of a syringe (Sheth et al. 2011; McMarhon Tobin et al. 2014) or onto a plastic cylinder (Dalmas et al. 2008; Newsholme et al. 2000; Brott, Richardson, and Louden 2012) is advantageous. A sample of mesentery that resembles the Swiss roll technique contains not only more sections but also more sections of different mesenteric arteries in a single slide, without the need to section each mesenteric artery separately as described in some techniques (Zhang et al. 2002; Zhang et al. 2006; Weaver et al. 2010). Therefore, investigators would be able to evaluate not only a large number of sections from several mesenteric arteries in one slide but also the arterial walls in different angles, increasing the chance of detecting even very subtle drug-induced morphologic changes.
Considering the amount of time and effort required for the aforementioned techniques (e.g., pinning out the mesentery at full extension, fixing it in a pinned position for 24 hr, or rolling mesentery onto a plunger or plastic cylinder), a less labor-intensive, time-consuming, and expensive technique was developed that also would allow microscopic examination of the mesentery vasculature with abundant longitudinal and cross sections of the vessels. This technique is simpler than all other techniques described in the literature, and it does not require additional personnel or special instruments to perform and adds only a few minutes to the necropsy time, thereby limiting expenses. It is an ideal sampling technique when screening for potential drug-induced vasculitis.
Footnotes
Acknowledgment
I would like to thank all the necropsy and histology technical staff at Histo-Scientific Research Laboratories.
Authors’ Contribution
The author (MPB) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. The author gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
