Abstract
There is a great need for improved diagnostic and prognostic accuracy of potential cardiac toxicity in drug development. This study reports the evaluation of several commercially available biomarker kits by 3 institutions (SRI, Eli Lilly, and Pfizer) for the discrimination between myocardial degeneration/necrosis and cardiac hypertrophy as well as the assessment of the interlaboratory and interplatform variation in results. Serum concentrations of natriuretic peptides (N-terminal pro-atrial natriuretic peptide [NT-proANP] and N-terminal pro-brain natriuretic peptide [NT-proBNP]), cardiac and skeletal troponins (cTnI, cTnT, and sTnI), myosin light chain 3 (Myl3), and fatty acid binding protein 3 (FABP3) were assessed in rats treated with minoxidil (MNX) and isoproterenol (ISO). MNX caused increased heart-to-body weight ratios and prominent elevations in NT-proANP and NT-proBNP concentrations detected at 24-hr postdose without elevation in troponins, Myl3, or FABP3 and with no abnormal histopathological findings. ISO caused ventricular leukocyte infiltration, myocyte fibrosis, and necrosis with increased concentrations of the natriuretic peptides, cardiac troponins, and Myl3. These results reinforce the advantages of a multimarker strategy in elucidating the underlying cause of cardiac insult and detecting myocardial tissue damage at 24-hr posttreatment. The interlaboratory and interplatform comparison analyses also showed that the data obtained from different laboratories and platforms are highly correlated and reproducible, making these biomarkers widely applicable in preclinical studies.
Keywords
Drug failures both in clinical development and following postmarket approval contribute significantly to the rising cost of drug discovery and development, with amortization of drug failures leading to the cost of development of new drugs being estimated as high as US$5 billion (Herper 2013). Drug-induced cardiac injury is a major cause of drug development failures, and 28% of drug withdrawals in the United States have been associated with negative cardiovascular effects (Gwathmey, Tsaioun, and Hajjar 2009; Feenstra et al. 1999; Ferri et al. 2013). Cardiotoxicity is also an important consideration for drugs with proven effectiveness in high-risk diseases such as HIV/AIDS, as some patients may be more susceptible to drug-related cardiovascular complications than others (Fantoni, Autore, and Del Borgo 2001; Lambert et al. 2015; Lipshultz et al. 2012; Bozkurt 2004). There is therefore a significant need for development of improved tools for both preclinical and clinical research that provide more sensitive predictors of potential cardiac effects of candidate drugs.
Serum biomarkers provide a minimally invasive measure for diagnosing cardiac effects of drugs. Several myocardium-specific proteins have been reported as useful biomarkers for congestive heart failure, myocardial infarction, and other heart diseases. These biomarkers include cardiac troponins I and T, cardiac natriuretic peptides, creatine kinase isoenzyme MB (CK-MB), and lactate dehydrogenase (Selvais et al. 1998; Babuin and Jaffe 2005; Lott and Stang 1989; O’Brien 2008; Lim et al. 2011). Among the biomarkers, cardiac troponins are considered the gold standard for cardiac injury considering their sensitivity and specificity; however, the troponins have a half-life of about 2 hr, which makes them difficult to use for routine evaluation in toxicology studies (Babuin and Jaffe 2005; Kehl et al. 2012; Dunn et al. 2011). The early clinical laboratory biomarkers for assessing cardiomyocyte degeneration/necrosis, Myl3, and FABP3 are abundant in myocyte cytoplasm and rapidly released with cell injury; however, they are present in both heart and skeletal muscle, thus limiting these biomarkers with regard to tissue specificity, and are rapidly cleared from the blood stream as well (Tanaka et al. 1991; Ravkilde et al. 1995; Tonomura et al. 2012). One major problem associated with these current biomarkers is that they typically only show significant changes in the first 12- to 24-hr postdose, returning to normal levels after 24 hr (Rajappa and Sharma 2005; Maynard, Menown, and Adgey 2000). Therefore, there is an urgent need for cardiac biomarkers that can evaluate drug-induced cardiotoxicity over longer periods of time and be incorporated in routine preclinical toxicology studies.
The natriuretic peptides, ANP and BNP, are cardiac hormones involved in plasma volume homeostasis, cardiovascular remodeling, and counter regulation of the renin–angiotensin–aldosterone system. ANP is produced primarily in the cardiac atria in response to cardiac ischemia, myocardial stretch, or cardiac hypertrophy due to pressure or volume overload (Mukoyama et al. 1991; Levin, Gardner, and Samson 1998). The messenger RNA transcript for ANP is approximately 1 kb in size and encodes a precursor protein (pro-ANP) of 126 amino acids. BNP was originally identified in extracts of porcine brain, but there is considerably more present in the cardiac ventricles. Human pro-BNP contains 108 amino acids. Amino-terminal fragments derived from the ANP and BNP precursors, NT-proANP and NT-proBNP, have been identified as predictors of cardiac dysfunction in many clinical settings including congestive heart failure, myocardial infarction, or septic shock (Mukoyama et al. 1991; Jarai et al. 2009; Meyer et al. 2007). The natriuretic peptide plasma levels are strong prognostic markers of 30-day mortality in patients with decompensated heart failure from ischemic and nonischemic origins (Luers et al. 2013; Scrutinio et al. 2013). In clinical studies of myocardial infarction, plasma BNP has been correlated with the severity of left ventricular dysfunction and remodeling (Morrow and Braunwald 2003) and also served as a predictor of future heart failure or other cardiovascular events in asymptomatic patients without obvious heart failure (Wang et al. 2004). When evaluated in combination with serum cardiac troponins, NT-proANP and NT-proBNP have been shown to be promising biomarkers bridging preclinical and clinical testing for chronic and/or functional cardiac injury (Walker 2006; Gaggin and Januzzi 2013).
MNX, a direct vasodilator, is capable of reducing blood pressure in patients with resistant hypertension; however, its effect can be limited because of its association with idiosyncratic onset of a pericardial effusion, plasma volume expansion, and aggravation of myocardial ischemia leading to left ventricular hypertrophy (Sica 2004; Bryan et al. 1977; Grim et al. 1979; Sanz et al. 1990). MNX is used in many preclinical studies for cardiac toxicity evaluation to induce cardiac hypertrophy by volume overload without primary myocardial injury (Turcani and Jacob 1998; van der Velden, Borgdorff, and Stienen 1999; Engle and Watson 2016).
ISO, a beta-adrenergic agonist used to treat bradycardia or heart block, has been used as a model compound to induce myocardial infarct-like lesions. Various studies have been conducted to evaluate myocyte damage following exposure to this drug (York et al. 2007; Kaufman 1985; Hasic et al. 2011; Acikel et al. 2005; Brady et al. 2010). We have previously reported on a variety of serum proteins showing significant changes in African green monkeys, Rhesus macaques and Cynomolgus macaques; these proteins can potentially be used for predicting long-term cardiac injury (Liu et al. 2013; Song et al. 2014) but are not currently available in standardized kits.
A wide range of cardiac effects that may eventually lead to the clinical syndrome of heart failure includes myocyte stretch/hypertrophy, myocardial injury, oxidative stress, neurohormonal activation, inflammation, apoptosis, and so on (Gaggin and Januzzi 2013). Of these, this study focuses on assessing a set of biomarkers that can distinguish cardiac hypertrophy (as evidenced by increased heart weights) from direct myocardial injury (degeneration/necrosis). Utilizing biomarkers that are insult-specific and tissue-specific, the improved diagnostic and prognostic accuracy of potential cardiac toxicity that may have not yet symptomized may become more feasible and reliable in both preclinical and clinical studies. As different laboratories use different platforms, or even subtle differences in application of the same platform, it is also important to assess the reproducibility of these biomarkers across different laboratories using the same platform as well as across different technology platforms. Recently, a cross-laboratory comparison study presented results demonstrating the ELISA detection of serum NT-proANP in rats to be technically adequate with acceptable intra- and interassay and interlaboratory precision and accuracy (Vinken et al. 2016). There have been reports of several successful uses of NT-proANP and NT-proBNP to detect cardiotoxicity in preclinical settings (Dunn et al. 2016; Stokes et al. 2015; Colton et al. 2011; Casartelli et al. 2011).
Here we report the results of the evaluation of several commercially available biomarker kits for the discrimination between myocardial degeneration/necrosis and cardiac hypertrophy. In addition, we compare the results from different laboratories, with both the same and different assay technologies, to assess the interlaboratory and interplatform variation in results.
Materials and Methods
Animal Care
Animal care and housing were in accordance with the National Research Council Guide for the Care and Use of Laboratory Animals, 8th edition (2011) and the Animal Welfare Standards incorporated in 9 CFR Part 3, 1991. All use of animals was approved by the Institutional Animal Care and Use Committee and conducted at SRI International (Menlo Park, CA) in a facility accredited by the Association for the Assessment and Accreditation of Laboratory Animal Care International.
Study Design
Three groups of Sprague-Dawley rats, each consisting of 10 males and 10 females, were administered 1% methylcellulose in sterile water (vehicle control, oral daily doses for 7 days), MNX at 30 mg/kg/day (oral daily doses for 7 days), or ISO at 5 mg/kg (subcutaneous single dose on day 1), respectively (Table 1). Oral dose administration was by gavage. Blood samples were drawn prestudy and on days 2 (main group, 5 animals/sex/group) and 8 (recovery group, 5 animals/sex/group) for determination of cardiac toxicity biomarker concentrations.
Study Design.
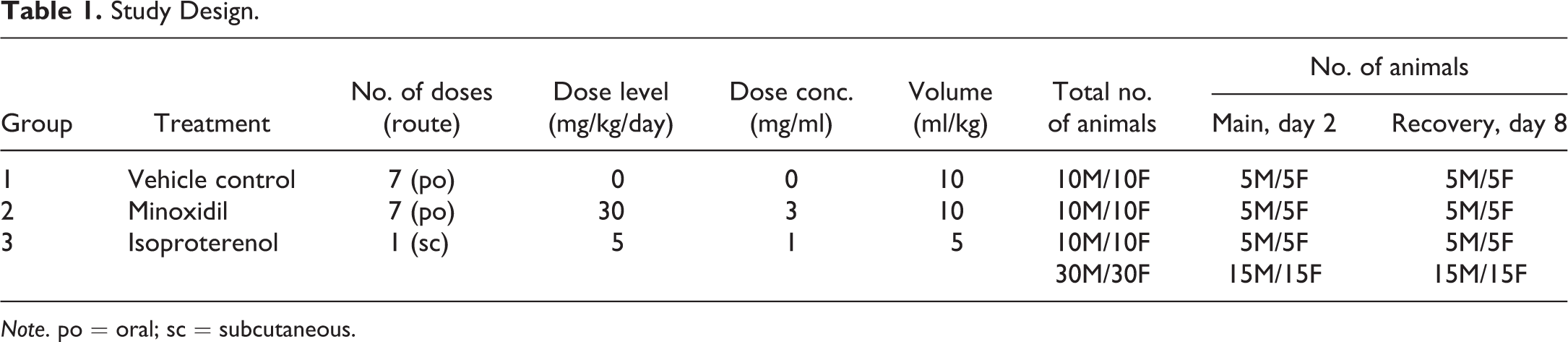
Note. po = oral; sc = subcutaneous.
Serum was prepared from the blood samples by centrifugation within 30 min of collection, then flash frozen in liquid nitrogen, and stored at ≤−60°C until analysis. The samples were analyzed by the participating laboratories (Table 2) using commercially available assays described under Biomarker Analysis section.
Assay Platforms Used by Each Laboratory for Cardiac Toxicity Biomarker Evaluation.
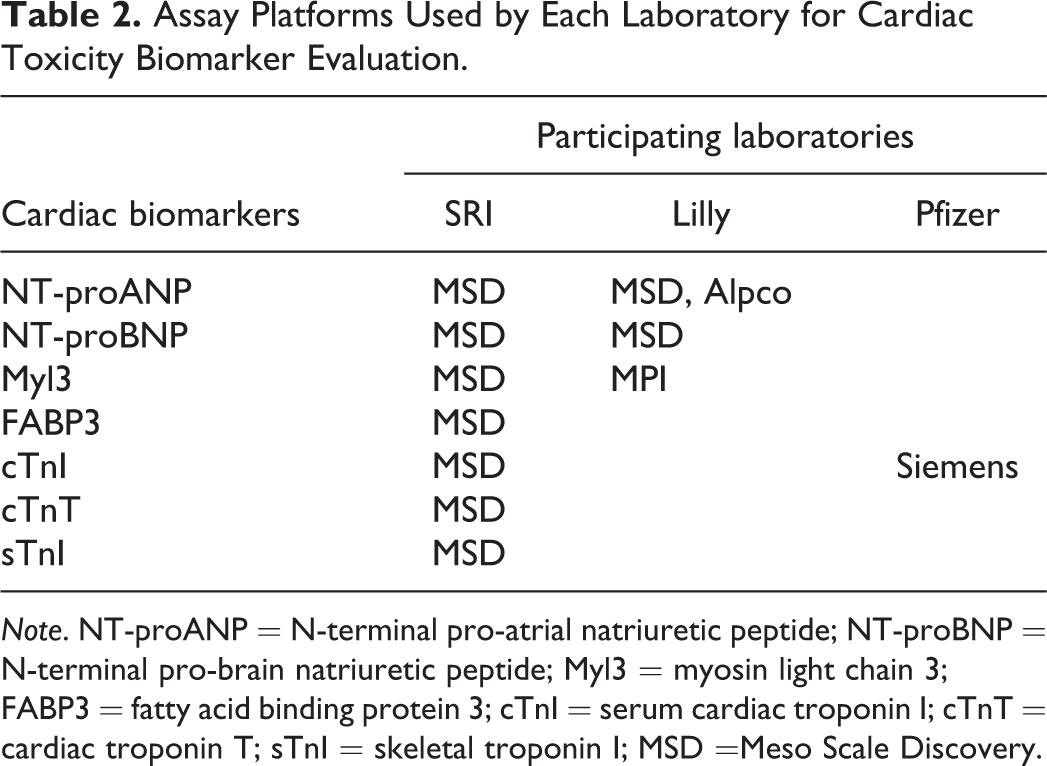
Note. NT-proANP = N-terminal pro-atrial natriuretic peptide; NT-proBNP = N-terminal pro-brain natriuretic peptide; Myl3 = myosin light chain 3; FABP3 = fatty acid binding protein 3; cTnI = serum cardiac troponin I; cTnT = cardiac troponin T; sTnI = skeletal troponin I; MSD =Meso Scale Discovery.
Biomarker Analysis
NT-proANP, NT-proBNP, Muscle Injury Panel 1 (MIP-1; Meso Scale Discovery [MSD])
Cardiac biomarkers were evaluated using electrochemiluminescent assays, including the rat MIP-1 assay (Myl3, FABP3, cTnI, cTnT, and sTnI) kit K15181C, rat NT-proANP kit K153MBD, and rat NT-proBNP kit K153JKD from MSD (Gaithersburg, MD). Serum samples were diluted 1:4 and 25 µl of the diluted sample (for MIP-1) or 50 µl of diluted sample (for rat NT-proANP and rat NT-proBNP) were dispensed and tested in duplicate according to the manufacturer’s protocol. After the completion of the procedure based on MSD assay protocol, the plates were analyzed using an MSD Sector Imager, Model 2400. The average of the 2 values was used to report the final results.
NT-proANP (Alpco)
NT-proANP was also measured by ELISA (Cat# 04-BI-20892; Alpco Diagnostics, Salem, NH; Vinken et al. 2016). Ten microliter of serum per sample was dispensed and tested in duplicate according to the manufacturer’s protocol. Plates were analyzed on a Molecular Devices (Sunnyvale, CA) Spectramax Plus 384 Microplate Reader using Softmax Pro data acquisition and analysis software, version 4.6. Results were reported as the average of duplicate values.
Myl3 (MPI)
Myl3 was quantitated in rat serum on a custom assay by MPI Research (Mattawan, MI). Samples (5 µl) were analyzed in duplicate on Gyrolab™ Bioaffy 1000 CDs (Gyros, Uppsala, Sweden) using the Gyrolab xP workstation with a biotinylated (EZ Link™ Micro Sulfo-NHS-LC biotinylation kit, Thermo Scientific, Rockford, IL) capture antibody (BiosPacific, Emeryville, CA) and a fluorescently labeled (Alexa Fluor® 647 labeling kit, Invitrogen, Carlsbad, CA) detection antibody (Abcam, Cambridge, UK). Prior to analysis, samples were diluted 1:4 in Rexxip A buffer (Gyros). Concentrations were interpolated from a 7-point standard curve using a recombinant; his-tagged rat-specific myl3 expressed in-house (Eli Lilly and Company, Indianapolis, IN) as a calibrator and fit with a 5-parameter logistic curve. Capture antibody was diluted in phosphate-buffered saline (PBS) + 0.01% Tween 20, detection antibody was diluted in Rexxip F buffer (Gyros), and standards were diluted in Rexxip A. Prior to sample analysis, the performance of the assay was characterized using positive controls (assay calibrator spiked into rat serum) at 6 different concentrations (approximately midway between each standard concentration) ran on 3 different CDs on 3 different days to assess precision and accuracy. Myl3 was quantifiable from 0.05 ng/ml to 200 ng/ml with <12.5% coefficient of variation (CV) and −5 to −12% relative error.
cTnI (Siemens)
The Siemens Advia Centaur cTnI-Ultra assay (Tarrytown, NY) was used to measure cardiac troponin I. It is a 3-site sandwich immunoassay using direct chemiluminometric technology that was optimized to detect human cardiac troponin I but has been shown to cross-react in multiple species including rats (Apple et al. 2008). Serum samples were tested for cTnI on the Siemens Advia Centaur according to the manufacturer’s protocol.
Organ Weights and Histopathology
Hearts were collected from all animals at necropsy and weighed. Left and right ventricular free walls were dissected from hearts and retained in 10% neutral-buffered formalin. Sections of the tissues were embedded in paraffin, cut approximately 5-µm thick, stained with hematoxylin and eosin, and evaluated by a board-certified veterinary pathologist. Each lesion was listed and coded by the most specific topographic and morphologic diagnoses, severity, and distribution, using International Harmonization of Nomenclature and Diagnostic Criteria for Lesions as a guide. A 4-step grading system (minimal, mild, moderate, and marked) was used to define gradable lesions for comparison between treated and control groups.
Statistical Analysis
Data were analyzed using 2-way analysis of variance (ANOVA) followed by Dunnett’s test for post hoc multiple comparison. Results were expressed as mean, standard deviations (SDs), %CV, and correlation coefficient (r). The intra-assay %CV was defined as the ratio of the SD and the mean of the measurements within each run. The interplatform and interlaboratory %CV was defined as the ratio of the SD and the mean of the group mean values from each platform or laboratory, respectively. For correlation analysis, the 2-tailed Pearson test was used. Statistical analysis was performed using GraphPad Prism, version 6, for Windows (La Jolla, CA).
Results
Serum NT-proANP and NT-proBNP
Two laboratories, SRI International (SRI) and Eli Lilly and Company (Lilly), analyzed the natriuretic peptides, NT-proANP and NT-proBNP, using assays from MSD (SRI/Lilly) or Alpco (Lilly). The results from all analyses showed that the group mean NT-proANP levels at 30-mg/kg/day MNX for 7 daily doses were increased on days 2 and 8 in both male and female rats (a 2.0- to 3.0-fold increase over the concurrent vehicle control mean value [p < .001; p < .01 on day 8 males from Alpco assay]; Figure 1). After a single dose of 5-mg/kg ISO, the group mean NT-proANP level was elevated on day 2 in both male and female rats (a 2.6- to 4.3-fold increase over the concurrent vehicle control mean value [p < .001], Figure 1). The intra-assay %CVs of NT-proANP ranged between 7.5 and 29.9% from SRI-MSD analysis, 6.1 and 58.2% from Lilly-MSD analysis, and 15.4 and 43.68% from Lilly-Alpco analysis (Supplemental Table 1). Statistically significant increases in group mean NT-proBNP levels at 30-mg/kg/day MNX for 7 daily doses on days 2 and 8 in male rats were shown in results from both laboratories (a 2.4- to 2.7-fold increase over the concurrent vehicle control mean value [p < .01], Figure 2A; a 2.2- to 2.8-fold increase over the concurrent vehicle control mean value [p < .05 on day 2; p < .01 on day 8], Figure 2B). At a single dose of 5-mg/kg ISO, the group mean NT-proBNP level was higher on day 2 in both male and female rats (a 2.4-fold increase over the concurrent vehicle control mean value for males [p < .01 from SRI; p < .05 from Lilly]; a 5- to 5.5-fold increase over the concurrent vehicle control mean value for females [p < .05], Figure 2) and on day 8 in male rats (from SRI analysis only; a 2.4-fold increase over the concurrent vehicle control mean value [p < .01], Figure 2A). The intra-assay %CVs of NT-proBNP ranged between 11.6 and 102.1% from SRI analysis and 15 and 93.5% from Lilly analysis (Supplemental Table 2).
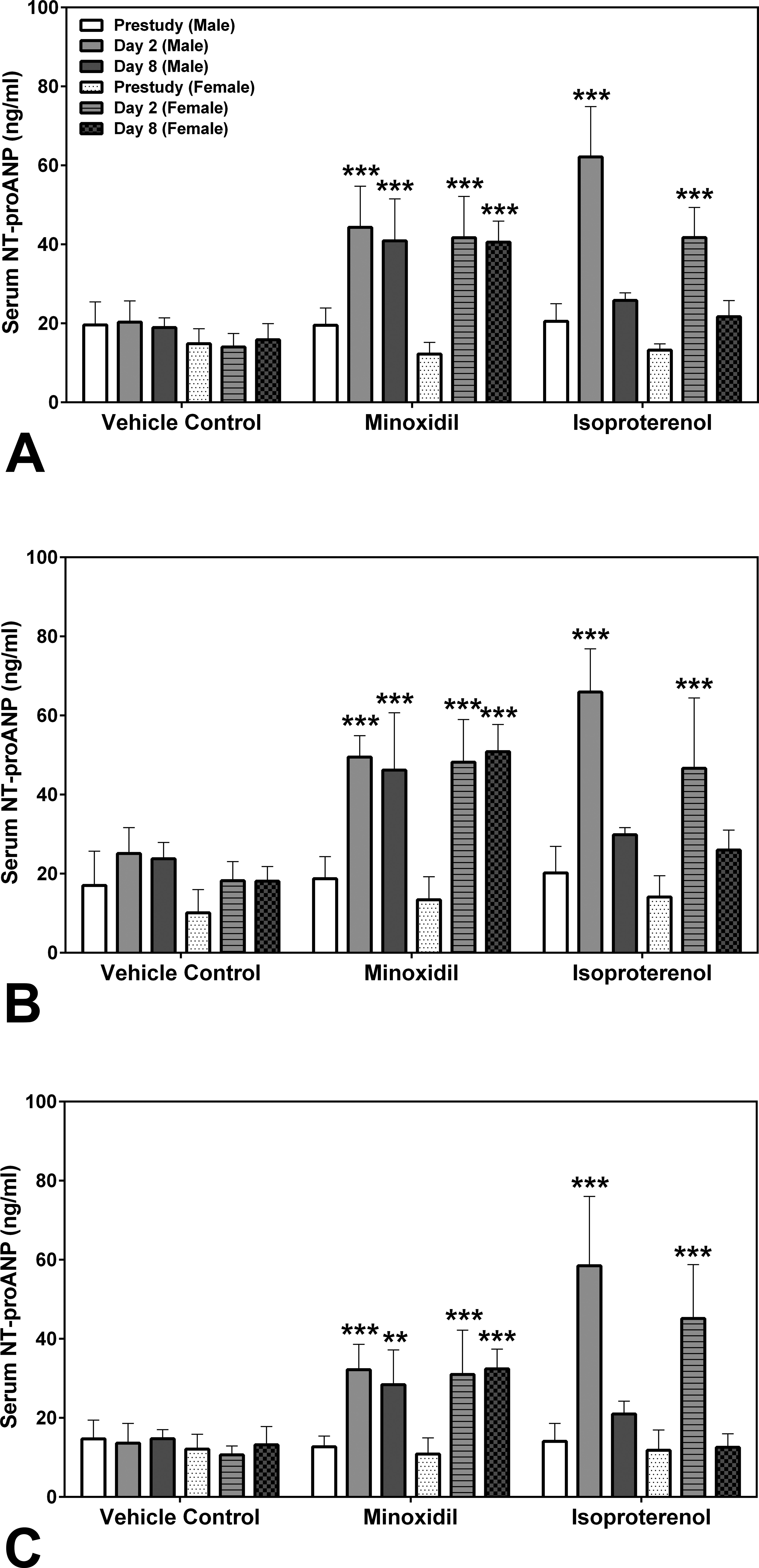
Serum N-terminal pro-atrial natriuretic peptide levels at prestudy, days 2 and 8 in rats treated with vehicle control, 7 daily oral doses of minoxidil at 30 mg/kg/day, or a single subcutaneous dose of isoproterenol at 5 mg/kg, analyzed by (A) Meso Scale Discovery (MSD) kit at SRI, and (B) MSD, or (C) Alpco kits at Lilly. Values are means, SD as error bars; n = 5. **Significantly different from the concurrent vehicle control, p value < .01 and ***p value < .001.
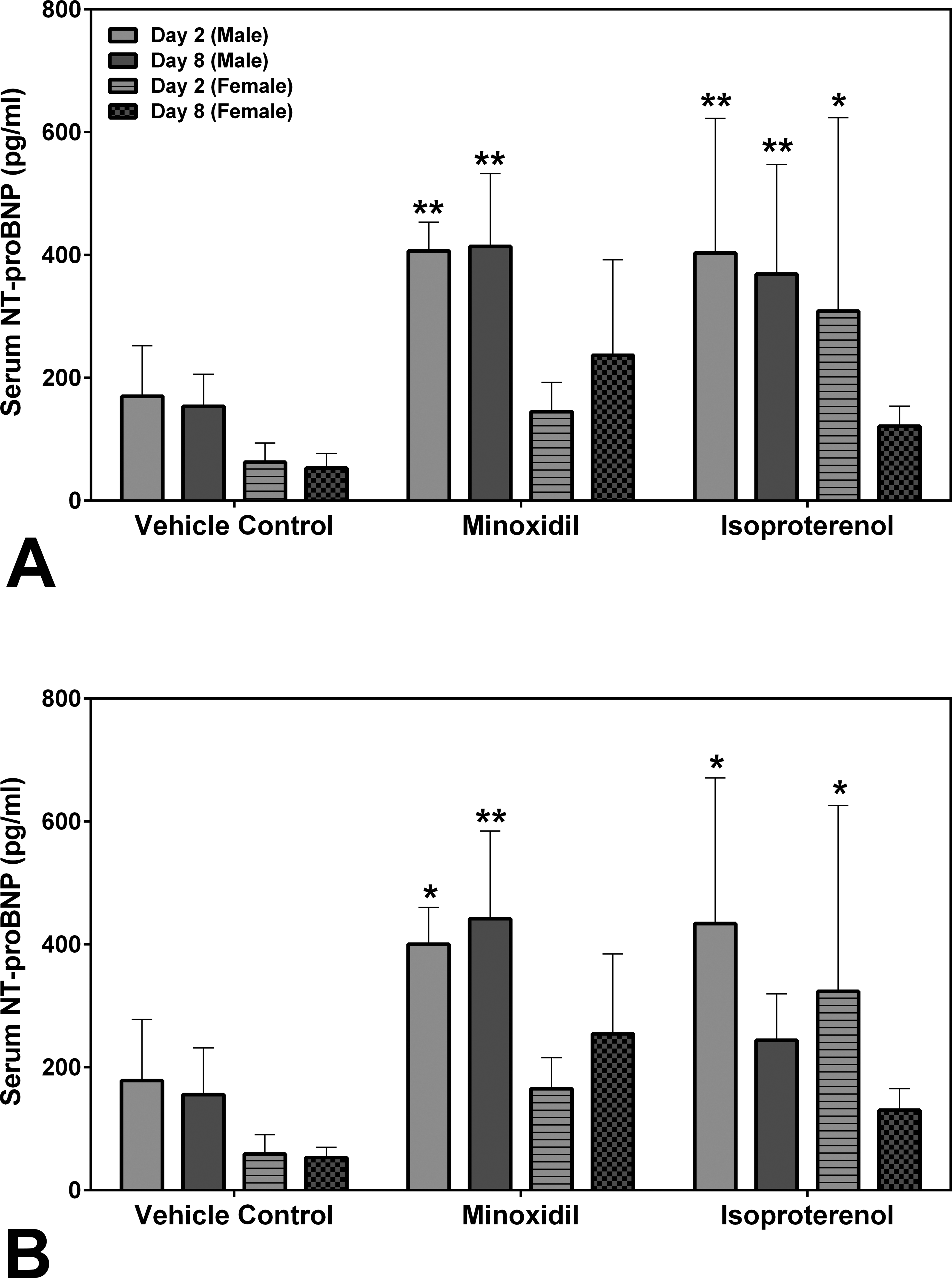
Serum N-terminal pro-brain natriuretic peptide levels on days 2 and 8 in rats treated with vehicle control, 7 daily oral doses of minoxidil at 30 mg/kg/day, or a single subcutaneous dose of isoproterenol at 5 mg/kg, analyzed by Meso Scale Discovery kits at (A) SRI and (B) Lilly. Values are means, SD as error bars; n = 5. *Significantly different from the concurrent vehicle control, p value < .05 and **p value < .01.
Serum Myl3 and FABP3
Myl3 was analyzed by SRI and Lilly using assays from MSD or MPI, respectively. FABP3 was analyzed by SRI using an assay from MSD. The results from both laboratories showed that the group mean Myl3 levels after a single dose of 5 mg/kg ISO were increased on day 2 in both male and female rats (a 73- to 108-fold increase over the concurrent vehicle control mean value for males [p < .001]; a 30-fold increase over the concurrent vehicle control mean value for females [p < .001], Figure 3). The intra-assay %CVs of Myl3 ranged between 25.3 and 56.4% from SRI analysis and 22.3 and 56.3% from Lilly analysis (Supplemental Table 3). No statistically significant differences were noted in the group mean FABP3 levels by MNX or ISO treatment in this study (Figure 4). The intra-assay %CVs of FABP3 ranged between 33.6 and 134.6% (Supplemental Table 4).
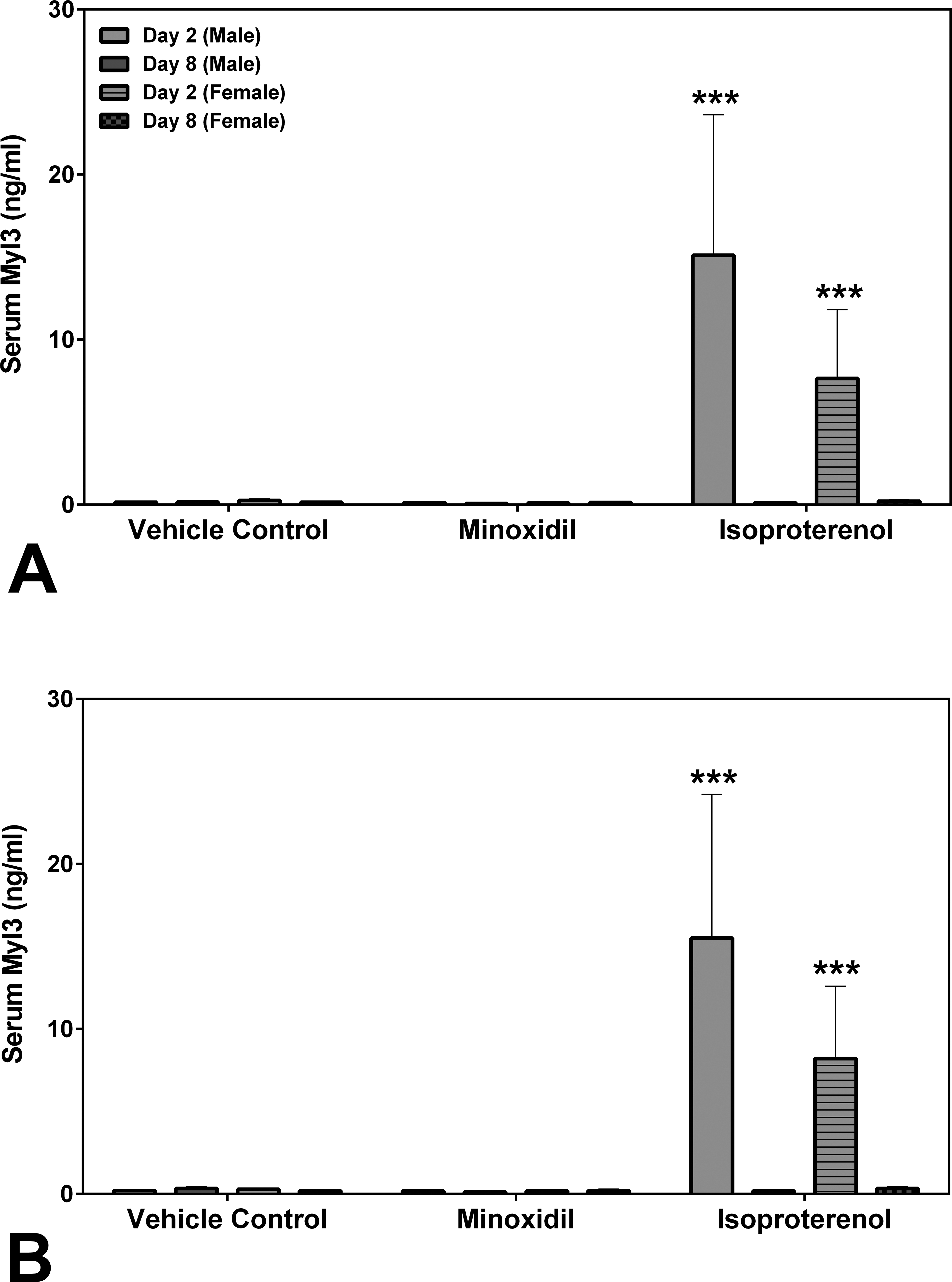
Serum myosin light chain 3 levels on days 2 and 8 in rats treated with vehicle control, 7 daily oral doses of minoxidil at 30 mg/kg/day, or a single subcutaneous dose of isoproterenol at 5 mg/kg, analyzed by (A) Meso Scale Discovery kit at SRI and (B) MPI kit at Lilly. Values are means, SD as error bars; n = 5. ***Significantly different from the concurrent vehicle control, p value < .001.
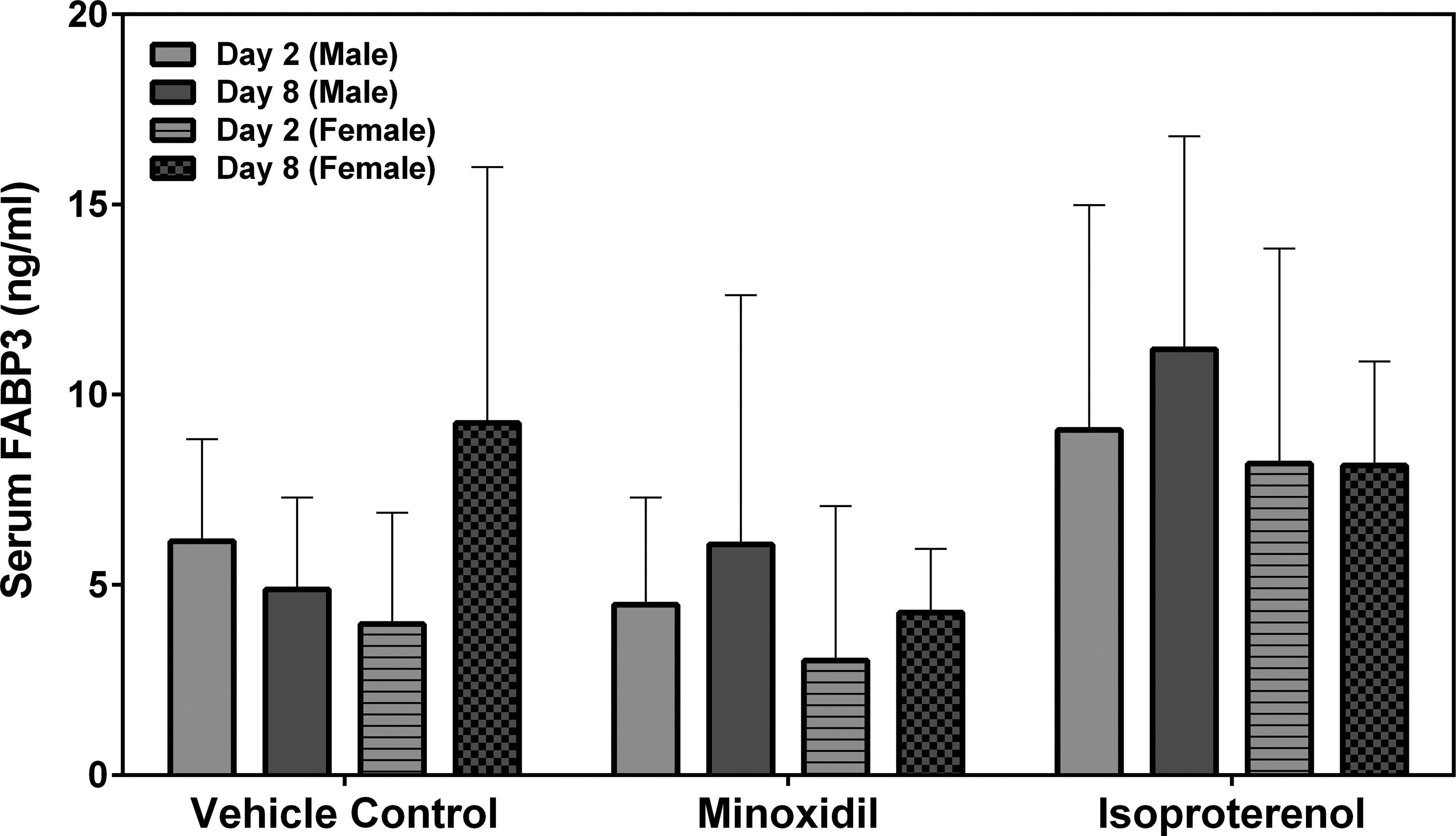
Serum fatty acid binding protein 3 levels on days 2 and 8 in rats treated with vehicle control, 7 daily oral doses of minoxidil at 30 mg/kg/day, or a single subcutaneous dose of isoproterenol at 5 mg/kg, analyzed by Meso Scale Discovery kit SRI. Values are means, SD as error bars; n = 5.
Serum Troponins
cTnI, cTnT, and sTnI were analyzed by SRI and Pfizer (cTnI only) using assays from MSD or Siemens, respectively. Statistically significant increases in cTnI levels after a single dose of 5 mg/kg ISO on day 2 were observed in both male and female rats by both laboratories (a >449-fold increase over the concurrent vehicle control mean value for males [p < .01 from SRI and p < .05 from Pfizer]; a >56-fold increase over the concurrent vehicle control mean value for females [p < .001], Figure 5A and B). The intra-assay %CVs of cTnI ranged between 76.7 and 223.6% from SRI analysis and 81.8 and 223.6% from Pfizer analysis (Supplemental Table 5). The group mean cTnT level after a single dose of 5 mg/kg ISO was increased on day 2 in male rats (a 159-fold increase over the concurrent vehicle control mean value [p < .001], Figure 5C). The intra-assay %CVs of cTnT ranged between 49 and 223.6% (Supplemental Table 6). The group mean sTnI level after a single dose of 5 mg/kg ISO was increased on day 8 in male rats (an 8-fold increase over the concurrent vehicle control mean value [p < .05], Figure 5D). The intra-assay %CVs of sTnI ranged between 34.4 and 223.6% (Supplemental Table 7).
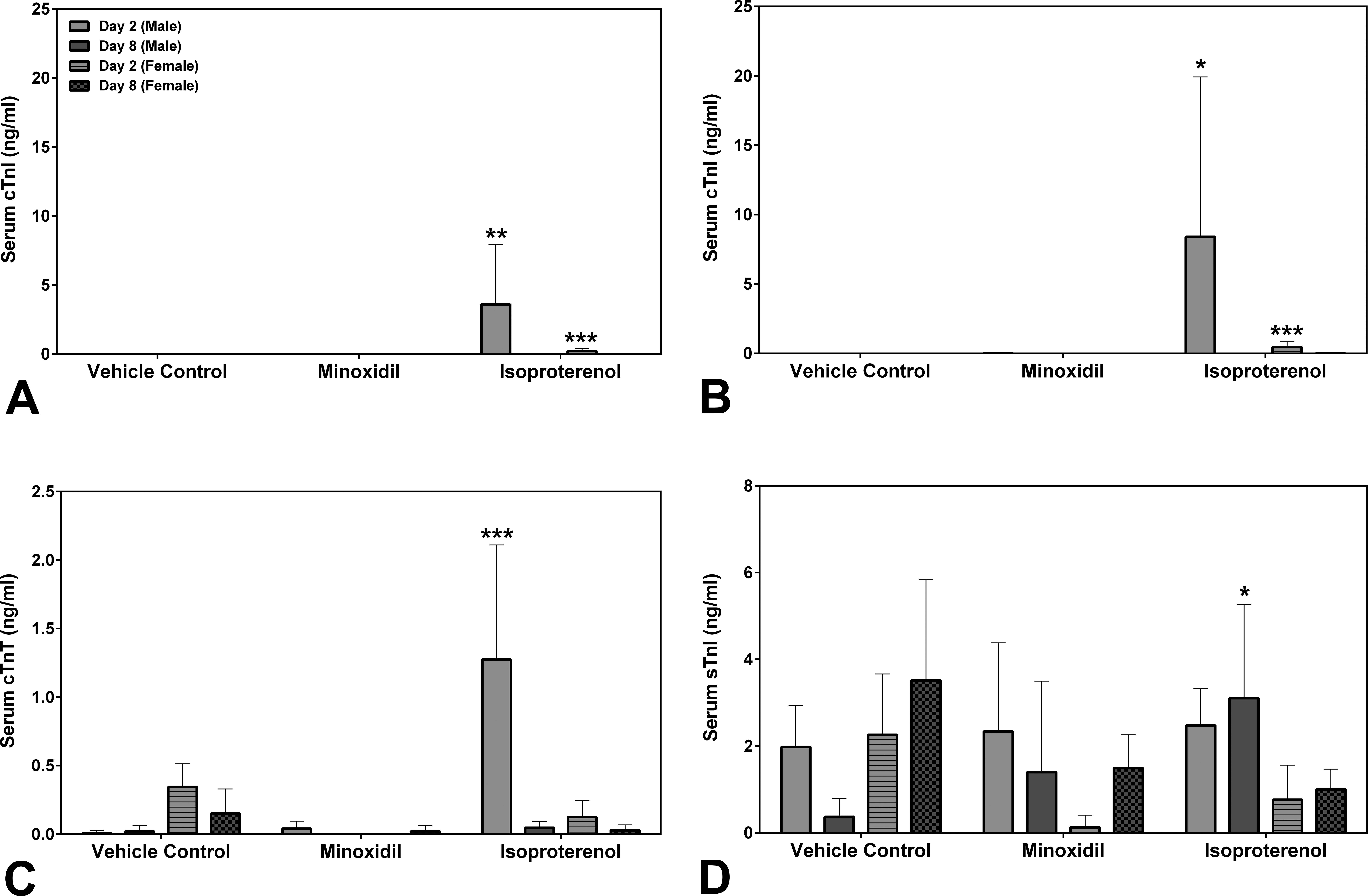
Serum cardiac troponin I, cardiac troponin T, and skeletal troponin I levels on days 2 and 8 in rats treated with vehicle control, 7 daily oral doses of minoxidil at 30 mg/kg/day, or a single subcutaneous dose of isoproterenol at 5 mg, analyzed by (A), (C), (D) Meso Scale Discovery kit at SRI, and (B) Siemens kit at Pfizer. Values are means, SD as error bars; n = 5. *Significantly different from the concurrent vehicle control, p value < .05, **p value < .01, and ***p value < .001.
Heart Weight and Histopathology
Statistically significant increases in absolute heart weights at 30 mg/kg/day MNX for 7 daily doses on day 8 (a 1.2-fold increase over the concurrent vehicle control mean value [p < .05]) and after a single dose of 5 mg/kg ISO on day 2 (a 1.2-fold increase over the concurrent vehicle control mean value [p < .01]) were observed in male rats (Figure 6). Histopathology findings included ventricle leukocyte infiltration, ventricle fibrosis, and necrosis in the ISO-treated animals but not in any of the MNX-treated animals (Figure 7). Cardiac hypertrophy was diagnosed based on the increase in both absolute and relative heart weight; however, hypertrophy was not detectable from histopathologic examination in this study, as histopathological changes of hypertrophy are difficult to detect unless the impact is severe. Females also showed better overall recovery from cardiac muscle injury from ISO compared with males on day 8 (Table 3).
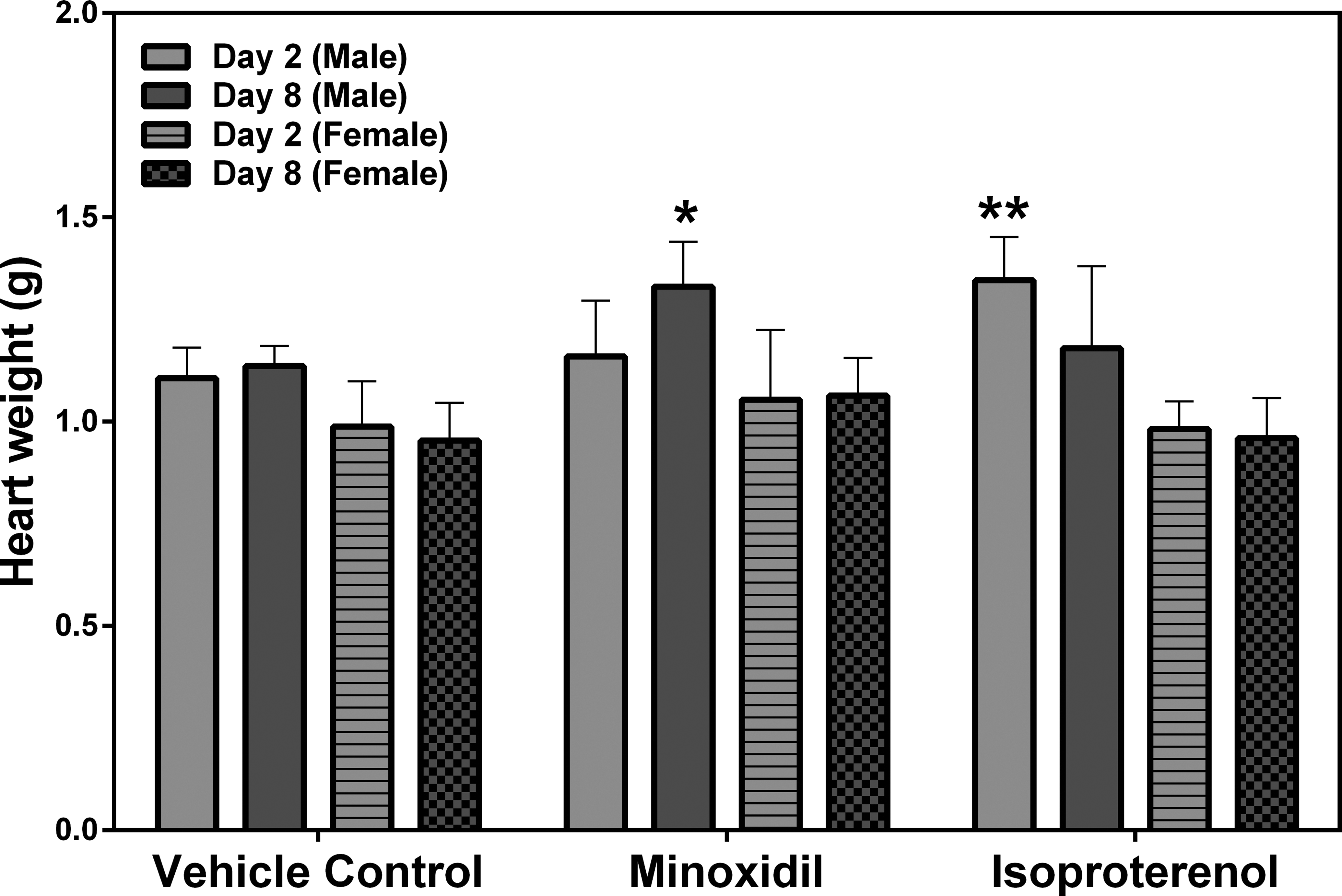
Heart weights at necropsy on days 2 and 8 in rats treated with vehicle control, 7 daily oral doses of minoxidil at 30 mg/kg/day, or a single subcutaneous dose of isoproterenol at 5 mg/kg. Values are means, SD as error bars; n = 5. *Significantly different from the concurrent vehicle control, p value < .05 and **p value < .01.
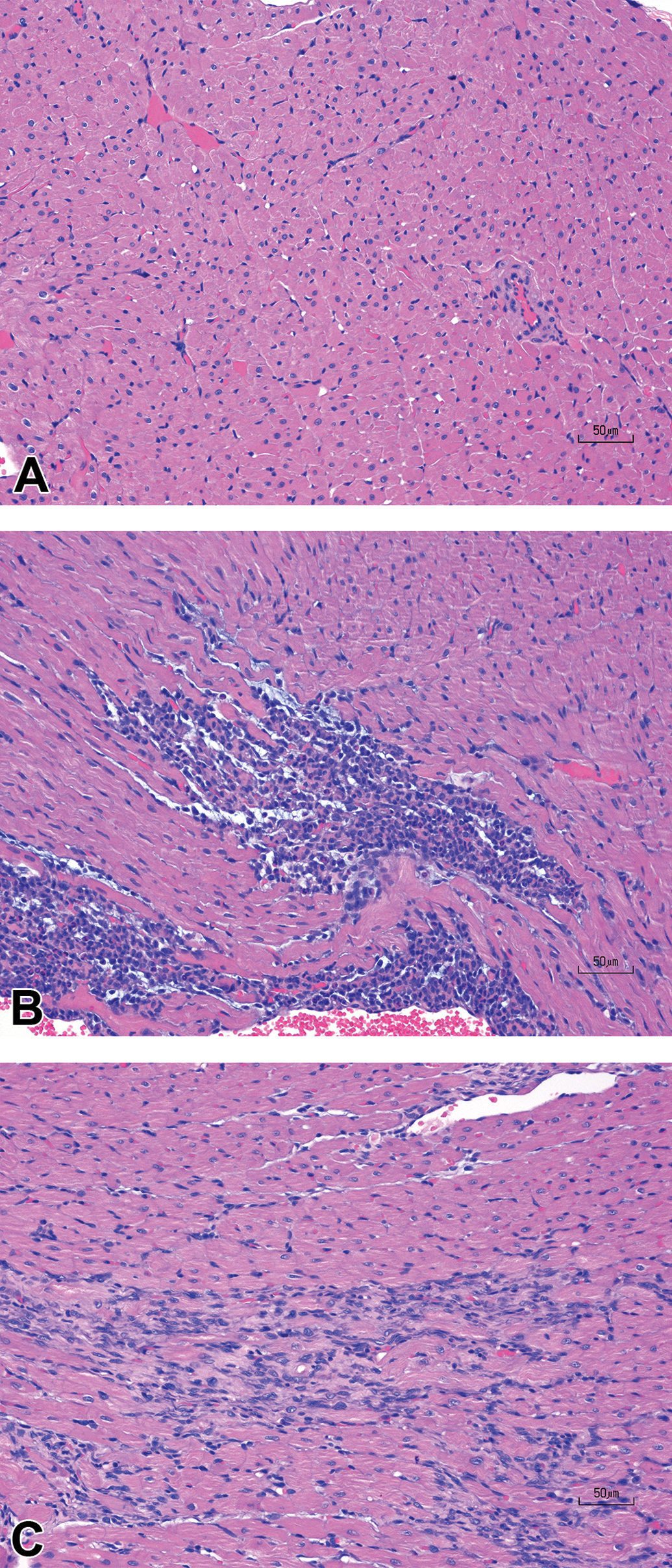
H&E images of rat ventricular myocardium after treatment with isoproterenol in rats showing (A) normal, (B) progression of moderate ventricle leukocyte infiltration and necrosis, and (C) progression of mild ventricle multifocal fibrosis. Original objective 20×.
Summary Histopathology Findings.
Interlaboratory and Interplatform Statistical Analysis
Interlaboratory and interplatform correlation and precision analyses of each data point on serum NT-proANP, NT-proBNP, Myl3, and cTnI levels were performed. Correlation analysis results are presented in Supplemental Table 8, and CV% and mean values are presented in Supplemental Table 9. Both NT-proANP and NT-proBNP data sets from 2 laboratories were well correlated (r = .9460 for NT-proANP and r = .9125 for NT-proBNP). The precision analysis showed highly reproducible NT-proANP levels with %CVs of <20% from all groups except female prestudy vehicle samples (26.9%). NT-proBNP data sets also showed strong precision and reproducibility with %CVs of <10% from all groups except male day 8 ISO samples (28.8%). Two sets of NT-proANP levels each from MSD and Alpco assays, both analyzed by Lilly, were well correlated (r = .8777) with %CVs ranging between 2.3 and 49.2%. Myl3 and cTnI also showed high correlation between the data sets from 2 different assays (MSD vs. MPI for Myl3, r = .9982; MSD vs. Siemens for cTnI, r = 0.9970). The precision of data was poor at low levels of Myl3 (<1 ng/ml) with %CVs ranging up to 50.1%; however, analysis showed great reproducibility of data at higher concentration of Myl3 in male and female ISO groups on day 2 with %CVs of less than 5%. The %CVs of cTnI ranged above 50% at concentrations higher than 0.3 ng/ml and above 100% at lower levels of cTnI.
Discussion
Based on the results of this study using commercially available kits, a multimarker strategy is beneficial in distinguishing the underlying cause of cardiac insult with or without apparent tissue damage (e.g., hypertrophy vs. myocyte necrosis). Also the changes in the levels of these cardiac injury markers were detectable over longer periods of time at 24 hr (day 2) or 168 hr (day 8; e.g., NT-proANP) posttreatment providing a wider time frame to identify the cardiac insult as well as the sex differences in the elevation of these markers. The addition of the natriuretic peptides to the cardiotoxicity biomarker panel along with the direct myocyte injury biomarkers in animal models would enable the researchers to stratify the risk of potential cardiac myopathy caused by various mechanisms of action at early stages of drug development.
While assessing serum biomarkers is a promising strategy for early preclinical detection of potential drug-induced cardiac injury, some of these serum markers have limitations with poor cardiac specificity, sensitivity, and short half-life as well as inconsistency across species and low relevance to clinical outcome. Reverse translational application of established clinical biomarkers such as cardiac troponins and natriuretic peptides to preclinical models increases clinical relevance, and a multimarker approach offers the advantage of a more comprehensive assessment of risk (Pinsky and Zhu 2011; Taqui and Daniels 2013). Prominent elevation of NT-proANP and NT-proBNP concentrations detected at 24-hr postdose following single and multiple doses of MNX, a vasodilator that causes pericardial effusion in man and left ventricular hypertrophy (Moravec et al. 1994; Reichgott 1981), suggest that these biomarkers can be used to detect the presence of or possibly predict the development of myocardial hypertrophy in the absence of direct cardiomyocyte injury. Similar results, where early changes in NT-proANP after only 2 doses reflect the eventual development of cardiac hypertrophy after 14 or 28 days in rats treated with a PPAR agonist, have also been reported (Engle et al. 2010).
NT-proANP and NT-proBNP concentrations were also elevated at 24 hr after a single ISO administration that caused direct myocardial injury as indicated by ventricular leukocyte infiltration, myocyte fibrosis, and necrosis in histopathologic examination. During the course of cardiac injury by ISO, the natriuretic hormones respond as protective hormones that counteract the physiologic abnormalities of heart injury and dysfunction as also demonstrated in several clinical studies (Maisel et al. 2002; Richards et al. 2003; de Lemos et al. 2001; Eggers et al. 2009). The clinical biomarker Myl3 was elevated in response to the direct myocardial injury with a single ISO treatment at 24-hr postdose but showed no change with MNX-mediated hypertrophy. While FABP3 was shown to be a good early marker (i.e., 2- to 3-hr postinsult) for detecting acute/active cardiomyocyte injury in rat models (Tonomura et al. 2012; Clements et al. 2010), no changes in protein levels of FABP3 were detected in this study, where the serum samples were collected at a later time point (24 hr) after ISO treatment, likely due to its rapid clearance in circulation. The concentrations of the most widely established biomarkers for myocardial injury, cTnI and cTnT, were also elevated only in rats treated with ISO at 24-hr postdose showing their specificity for myocardial degeneration and necrosis. Although increased cardiac troponin concentrations classically indicate cardiomyocyte necrosis, their elevation does not offer further information about the preceding events and also has poor sensitivity in the absence of infarction in people (Balk et al. 2001). The sex differences in some of the biomarkers were also presented with higher serum biomarker levels in male rats induced by the treatments (e.g., NT-proBNP, Myl3, cTnT, cTnI). The males also showed poorer recovery from ISO treatment compared with females on day 8 on histopathologic evaluation. The intra-assay variation with wide ranges of %CVs were presented in some of the groups or time points from NT-proBNP, FABP3, and serum troponin assays presumably due to relatively low basal or response levels to the treatments.
Interlaboratory correlation and precision analyses were performed for each data point on serum NT-proANP and NT-proBNP levels assessed by SRI and Lilly. Pearson r, which quantifies the direction and magnitude of correlation between 2 data sets, shows both NT-proANP and NT-proBNP data sets from 2 laboratories were well correlated and followed close trend lines (Figure 8). The precision of the data (%CV) presents the reproducibility of the biomarker levels from each laboratory, and the results showed precise and reproducible NT-proANP levels with %CVs lower than 20%. NT-proBNP data sets also showed strong precision and reproducibility with %CVs lower than 10%. This interlaboratory comparison analysis shows that the biomarker data obtained using the MSD kits at 2 participating laboratories are highly reproducible, thus enabling these assays to be used across different institutions to obtain comparable results in preclinical studies.
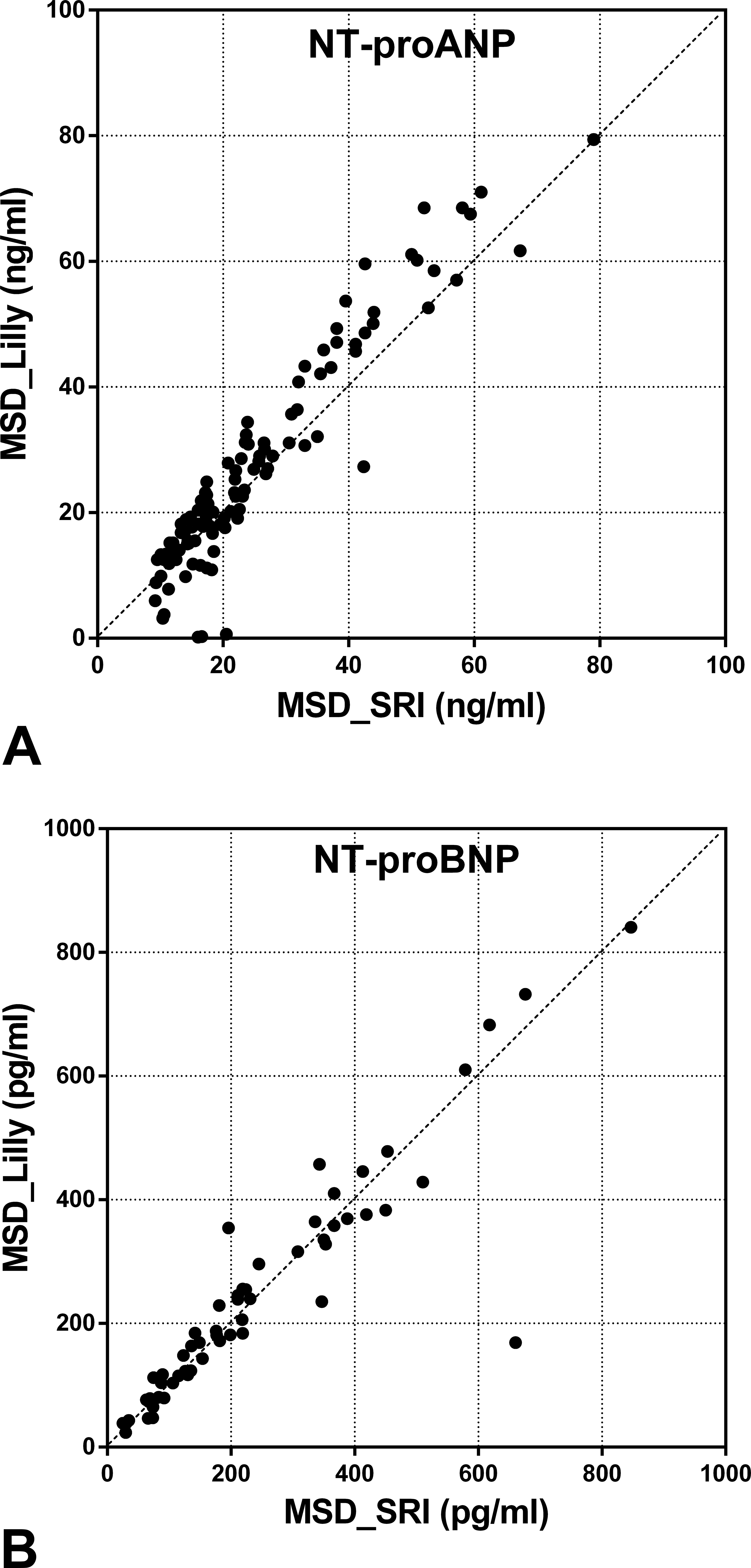
Interlaboratory comparison of (A) serum N-terminal pro-atrial natriuretic peptide levels and (B) serum N-terminal pro-brain natriuretic peptide levels in rats treated with vehicle control, 7 daily oral doses of minoxidil at 30 mg/kg/day, or a single subcutaneous dose of isoproterenol at 5 mg/kg, analyzed by SRI (x-axis) and Lilly (y-axis) using assays from Meso Scale Discovery.
Interplatform comparison was performed on NT-proANP, Myl3, and cTnI levels analyzed using 2 different assay kits as shown in Table 2. NT-proANP levels from MSD and Alpco assays, both analyzed by Lilly, were well correlated (r = 0.8777) with moderate precision and reproducibility (Figure 9A). Myl3 and cTnI also showed high correlation between 2 data sets from different assays (Figures 9B and C). The precision analysis showed great reproducibility of Myl3 data only in male and female ISO groups that had elevated Myl3 concentrations. Although the variability between biomarker levels obtained from different assays is considered to be inevitable and exaggerated with wide ranges of %CVs when the basal and/or expression levels of analytes are relatively low at the time of sample collection, the high correlation between each data set gives confidence in utilizing these biomarkers in preclinical studies using commercially available assay kits to interpret the evidence of cardiac toxicity across different studies and platforms. Although the high %CVs at baseline concentrations of the analytes suggests that subtle changes may not be detected, the advantages of using these kits are still creditable as the meaningful myocardial injury would induce the detectable changes in the levels of biomarkers within a reasonable time frame (∼24 hr) of sample collection serving as preliminary diagnosis and/or supplemental toxicity data along with the traditional cardiac toxicity evaluation. In cases where quantitative differences were significant (e.g., the Pfizer Siemens cTnI assay was clearly more sensitive than the corresponding MSD assay), the qualitative differences were not, and all assays correctly identified the cardiac toxicity induced by the drugs at the doses used in this study.
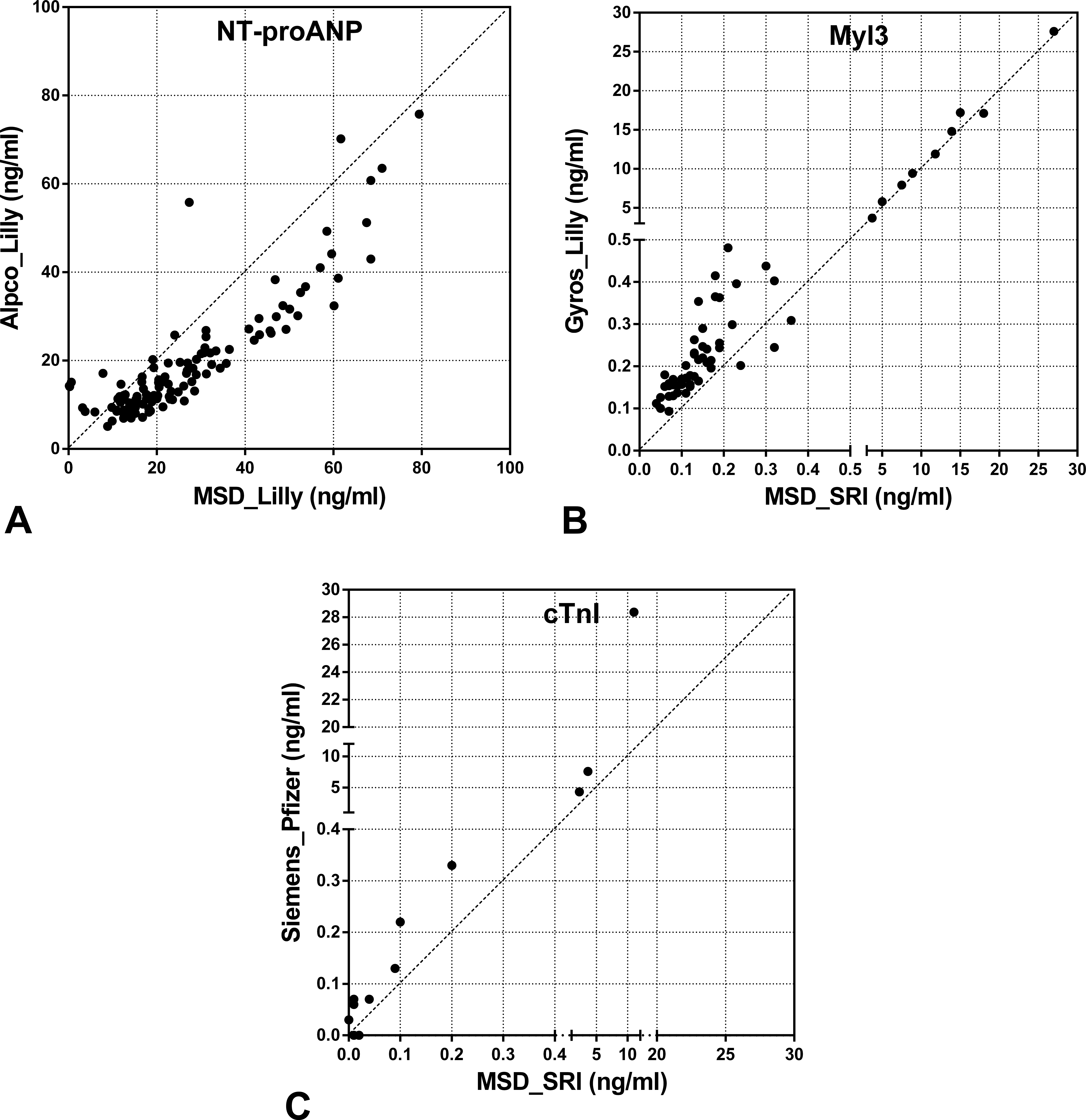
Interplatform comparison of (A) serum N-terminal pro-atrial natriuretic peptide, (B) serum myosin light chain 3 levels, and (C) serum cardiac troponin I levels in rats treated with vehicle control, 7 daily oral doses of minoxidil at 30 mg/kg/day, or a single subcutaneous dose of isoproterenol at 5 mg/kg, analyzed by SRI (B and C, x-axis, Meso Scale Discovery [MSD] assay), Lilly (A, MSD and Alpco assays; B, y-axis, MPI assay), or Pfizer (C, y-axis, Siemens assay).
In summary, we have evaluated several commercially available kits for the assessment of biomarkers of direct cardiac injury as well as cardiac hypertrophy, assessing both different assay platforms and interlaboratory reproducibility of results using the same platform. All cardiac markers performed as expected and demonstrated excellent differentiation between cardiac hypertrophy (based on increased heart weights) and cardiac injury. Interlaboratory reproducibility of the MSD kits was excellent, and interplatform reproducibility, while not as tight as results from the MSD platform in different labs, showed very good comparability. These results indicate that the use of these commercially available biomarker kits for assessment of cardiac toxicity could provide robust and reproducible data to identify drug-induced cardiac injury and provide improved prognostic indication of cardiac liabilities of candidate drugs.
Footnotes
Acknowledgments
The authors gratefully acknowledge Dr. Hao Zhang, NIAID, for his support and insightful suggestions. Diana Vuong, Lynn Chellarajen, Amanda Fields, and Lauren Haberland provided expert technical assistance in the conduct of the animal studies. This material is based upon work supported by Critical Path Institute’s (C-Path) Predictive Safety Testing Consortium (PSTC). The authors would like to acknowledge and thank the members of PSTC for their scientific, financial, and in-kind contributions that supported these research activities as well as the input from FDA and EMA scientists who serve as advisors.
Authors’ Contribution
Authors contributed to conception or design (KK, SE, WR, SS, JM); data acquisition, analysis, or interpretation (KK, NC, DF, SE, WR, SS, JM); drafting the manuscript (KK, NC, DF, JM); and critically revising the manuscript (KK, SE, WR, SS, JM). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project has been funded in part with federal funds from the National Institute of Allergy and Infectious Diseases, National Institutes of Health, and Department of Health and Human Services, under Contract Nos. HHSN272200700043C and HHSN272201400006.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
