Abstract
The current studies demonstrate the effect of low-dose intraperitoneal (IP) administration of isoprotenerol (ISO) and subcutaneous (SC) versus IP routes of administration of ISO on serum cardiac troponin I (cTnI) levels in female Hanover Wistar rats, providing additional evidence to support acceptance of cTnI as a cardiac biomarker. At 2 hr postdosing with 0-500 μg/kg ISO, mean serum cTnI levels were increased in a dose-related fashion at ≥10 μg/kg with no evidence of cardiac pathology. At 24 h, cTnI concentrations were generally at control levels, but histologic cardiomyocyte injury was evident in a proportion of the animals given ≥10 μg/kg. In a second experiment, rats given SC ISO at 5,000 μg/kg and necropsied at 0, 1, 2, and 4 hr postdosing had higher levels of serum cTnI than animals given the same dose IP.
In humans, serum/plasma concentrations of cardiac troponin I (cTnI) and cardiac troponin T (cTnT) are widely employed in the detection and evaluation of acute myocardial infarction and other cardiac conditions (Apple et al. 2005). However, the use of cardiac troponins (cTn) in the assessment of cardiac injury in toxicological investigations is not yet widely accepted (Wallace et al. 2004; York et al. 2007), and further evidence is required to support the use of these potential new serum biomarkers by investigating their absolute and temporal responses in a range of different types of cardiac injury induced with a variety of “model” cardiotoxic compounds (O’Brien et al. 2006; York et al. 2007; Brady 2008).
Isoproterenol (ISO), a powerful nonselective β-agonist, is widely used as a model agent in toxicological studies to induce cardiac muscle injury, through an exaggerated pharmacological effect (O’Brien et al. 2006; York et al. 2007; Mikaelian et al. 2008; Schultze et al. 2008; Zhang et al. 2008; Clements et al. 2009). This results in a rapid heart rate and a fall in blood pressure with a resultant local tissue hypoxia/anoxia leading to myocardial necrosis (Rona et al. 1959). These ISO-induced effects in the heart are associated with the release of cTn from the myocardium, resulting in increases in serum cTnI and cTnT within 4 hr of administration. Serum levels of cTn are closely associated with the degree of myocardial injury (Bertinchant et al. 2000; Wallace et al. 2004). For the induction of myocardial lesions in the rat the majority of studies have been performed in 6- to 8-week-old male rats; ISO has been administered by a variety of routes including the intraperitoneal (IP), subcutaneous (SC), and intravenous (IV) routes; but differences in cTn release and the degree of cardiac injury between these routes does not appear to have been reported (O’Brien et al. 2006).
The aims of the present studies were to investigate the sensitivity of serum cTnI in detecting cardiac damage at low dose levels of ISO and to provide information on the changes in serum cTnI levels in response to ISO administration by the IP and SC routes. Female rats were used to reduce the potential impact of background myocardial lesions that are more common in male rats (Lewis 1992).
Preliminary findings on these studies have been recorded in abstract form (Brady et al. 2005) and in a more comprehensive report (Brady 2008).
Female Hanover Wistar rats (Harlan UK Ltd, Bicester, Oxfordshire, UK) were housed in communal cages in groups of five, bedded on wood shavings, with diet (Harlan Teklad Ltd, Bicester, Oxfordshire, UK) and mains drinking water ad libitum. A temperature of 20°C (± 2°C) was maintained with a relative humidity of 40% to 60%, and a light:dark cycle of 12:12 hr (lights on at 07.00 h). All animal procedures were carried out under local Ethical Committee guidelines and approval for UK Home Office Project and Personal Licences. For experiment 1, (-)-isoproterenol hydrochloride (CAS number 5984-95-2, ISO; Sigma Chemical Co, Poole, Dorset, UK) was dissolved in de-ionized water (vehicle); solutions were prepared fresh to avoid oxidation, of 0 (vehicle control), 0.5, 2.4, 4.3, 21.5, 43.0, and 198.8 μg/ml. Rats (n = 80; age 8–9 weeks, mean body weight 176.7 g) were administered a single IP dose at levels of 0 (vehicle control; n = 20), and 1, 5, 10, 50, 100, and 500 μg/kg (n = 10 per dose level group). Animals from each control (n = 10) and ISO dose level group (n = 10) were killed by IP injection of pentobarbitone sodium (Euthatal, Rhône Mérieux Ltd, Harlow, Essex, UK) and necropsied at 2 hr (n = 5) and 24 hr (n = 5) postdosing, and abdominal aortic blood was taken for serum preparation and hearts were removed and placed in phosphate buffered formalin. Fixed hearts were processed and sectioned routinely as previously described (York et al. 2007), and a single standard H&E section was examined microscopically by one histopathologist and heart lesions graded (scored) as follows: no abnormalities detected (NAD), minimal (grade 1, scattered lesions, involving small numbers of isolated fibers, or small foci of several fibers), mild (grade 2, increased numbers of isolated fibers involved, or small foci of several affected fibers, extending deeper from the subendocardial myocardium into the midventricular myocardium), and moderate (grade 3, larger foci of fibers affecting more extensive regions of the myocardium) according to criteria of increasing lesion severity. For experiment 2, ISO dosing solutions were prepared at concentrations of 0 (vehicle control), 1,366.7 μg/ml (IP administration), and 1,383.3 μg/ml (SC administration). Female Hanover Wistar rats (n = 40; age 4–5 weeks, mean body weight 84.7 g) were given vehicle (control n = 5) or ISO at 5,000 μg/kg (n = 15) by either SC or IP routes. Control animals were necropsied at 0 h (time of dosing) and n = 5 ISO-treated animals each from the IP and SC groups at 1, 2, and 4 hr postdosing and blood was taken for serum preparation and subsequent assay for cTnI. Harvested serum from both experiments was stored at –80°C until analysis. Serum cTnI levels were assayed by automated immunochemiluminescence on the ACS: 180S (Bayer Healthcare Diagnostics, Newbury, Berkshire, UK), as described previously (York et al. 2007; Clements et al. 2009).
ISO-treated and control groups were compared using a one-way analysis of variance (ANOVA) followed by Dunnett’s test for post hoc multiple comparisons. Statistical analysis was performed using GraphPad Prism version 3.0 for Windows (GraphPad Software, San Diego, CA, USA).
Rats treated with a single IP injection of ISO at dose levels from 0 (vehicle-treated controls) to 5,000 μg/kg or a single SC injection at 5,000 μg/kg showed no clinical signs of toxicity or significant effects on body weight.
In experiment 1, dose-dependent increases in mean serum cTnI were detected at doses of ISO ≥ 10 μg/kg at 2 hr postdosing, reaching statistical significance at 100 μg/kg (Table 1 ). The dose level at which increased serum cTnI could be detected was therefore considered to be 10 μg/kg ISO at 2 hr; however, by 24 hr postdosing cTnI concentrations had fallen to baseline control values, except in 2/5 animals dosed with 500 μg/kg. This result indicates the importance of understanding the kinetics of biomarker release in a given model system when planning sampling schedules. All vehicle-treated control animals (n = 10) and those dosed with ISO at 1 (n = 5) and 5 (n = 5) μg/kg recorded serum cTnI levels at 0.03 μg/L, the lower limit of detection.
Experiment 1: Serum cardiac troponin I (cTnI) findings (μg/L) in individual control (vehicle-treated; 0 μg/kg; n = 10) female Hanover Wistar rats, and in rats treated with isoproterenol (ISO; n = 5/dose level group) at dose levels from 1 to 500 μg/kg at 2 hr postdosing a and group mean serum cardiac troponin I (cTnI) findings at 2 and 24 hr postdosing (at 24 hr all animals had cTnI levels of 0.03 μg/L except 2 animals given 500 μg/kg that showed levels of 0.04 and 0.13 μg/L).
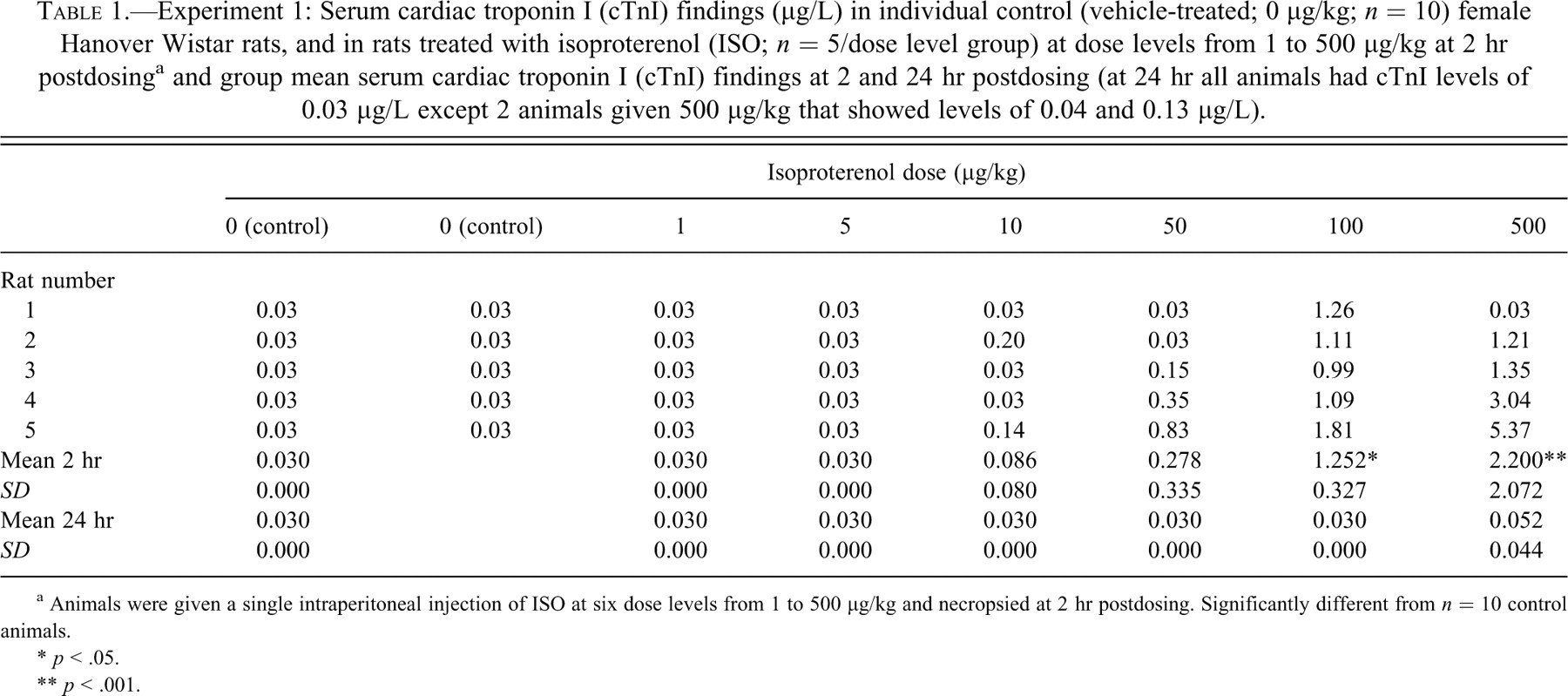
a Animals were given a single intraperitoneal injection of ISO at six dose levels from 1 to 500 μg/kg and necropsied at 2 hr postdosing. Significantly different from n = 10 control animals.
* p < .05.
** p < .001.
No histologic changes were apparent in the hearts of ISO-treated rats at the 2 hr time point in Experiment 1. However, at 24 hr postdosing (Figure 1 ), microscopic changes (characterized by myocardial degeneration/necrosis and myocyte loss) were detected in the myocardium of animals given doses of ISO ≥ 10 μg/kg (Table 2 ), demonstrating that under these experimental conditions 10 μg/kg was the lowest dose for identification of microscopic cardiac damage in routine H&E stained sections. This time course for the identification of microscopic lesions is consistent with those previously reported in an earlier investigation from this laboratory with higher doses of ISO between 250 to 20,000 μg/kg (York et al. 2007). When the mean cTnI levels at 2 h and scores of histologic cardiac injury at 24 hr are compared (Figure 2 ), the apparent association between these parameters can be seen and presents further evidence of the “temporal disconnect” previously described (York et al. 2007), with significant cTnI responses preceding the manifestation of microscopic change. This suggests that these timings for cTnI and microscopic lesion detection are consistent across a wide range of single doses of ISO at 2 and 24 hr postdosing.
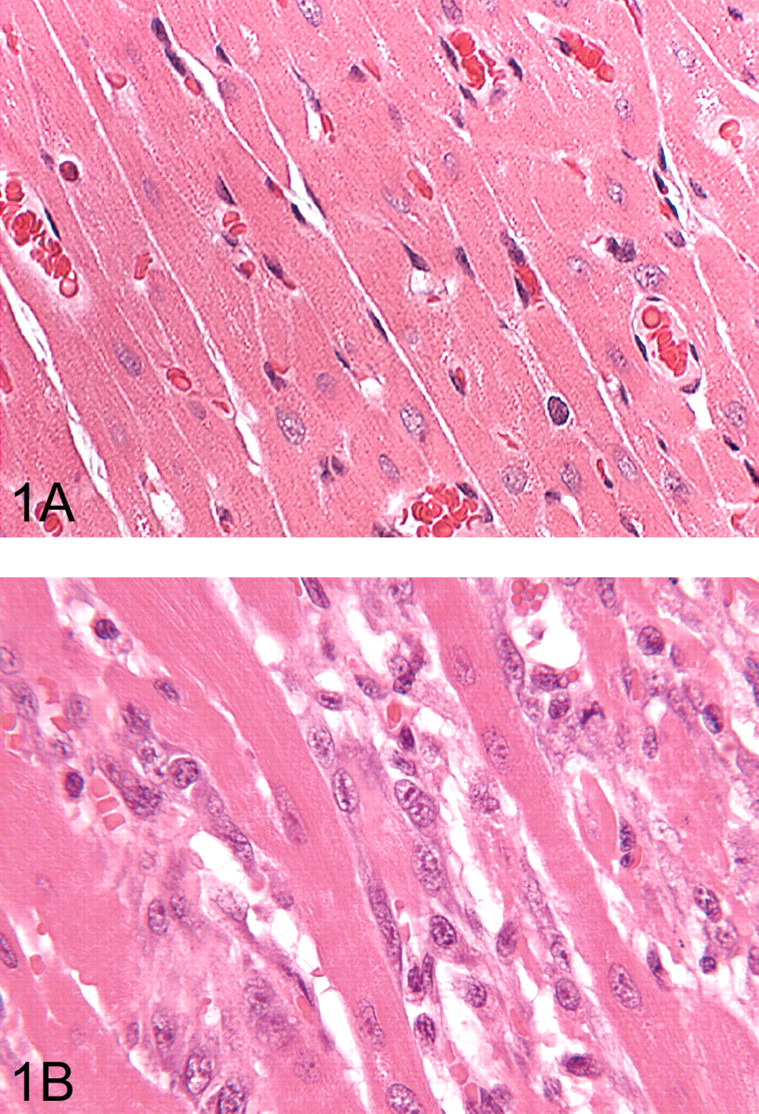
Experiment 1: (A) Myocardium from a control rat to show the normal appearance and arrangement of cardiomyocytes. (B) Representative section from a rat given a single intraperitoneal dose of isoproterenol and killed at 24 hr postdosing. There are multifocal areas of myocardial degeneration/necrosis, with an infiltration of mononuclear cells and fibroblasts. H&E, (A) ×400 original magnification (OM); (B) ×400 OM.
Experiment 1: Incidence of microscopic findings in the hearts of control (vehicle-treated; 0 μg/kg) female Hanover Wistar rats, and in rats treated with isoproterenol (ISO) at dose levels from 1 to 500 μg/kg at 24 hr postdosing. a
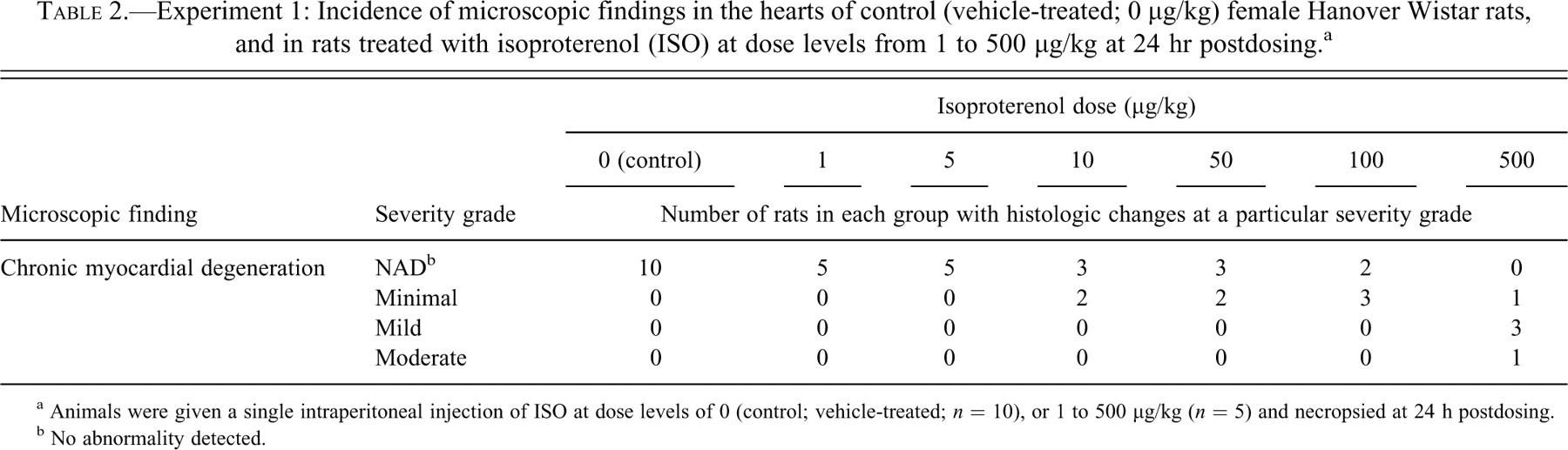
a Animals were given a single intraperitoneal injection of ISO at dose levels of 0 (control; vehicle-treated; n = 10), or 1 to 500 μg/kg (n = 5) and necropsied at 24 h postdosing.
b No abnormality detected.
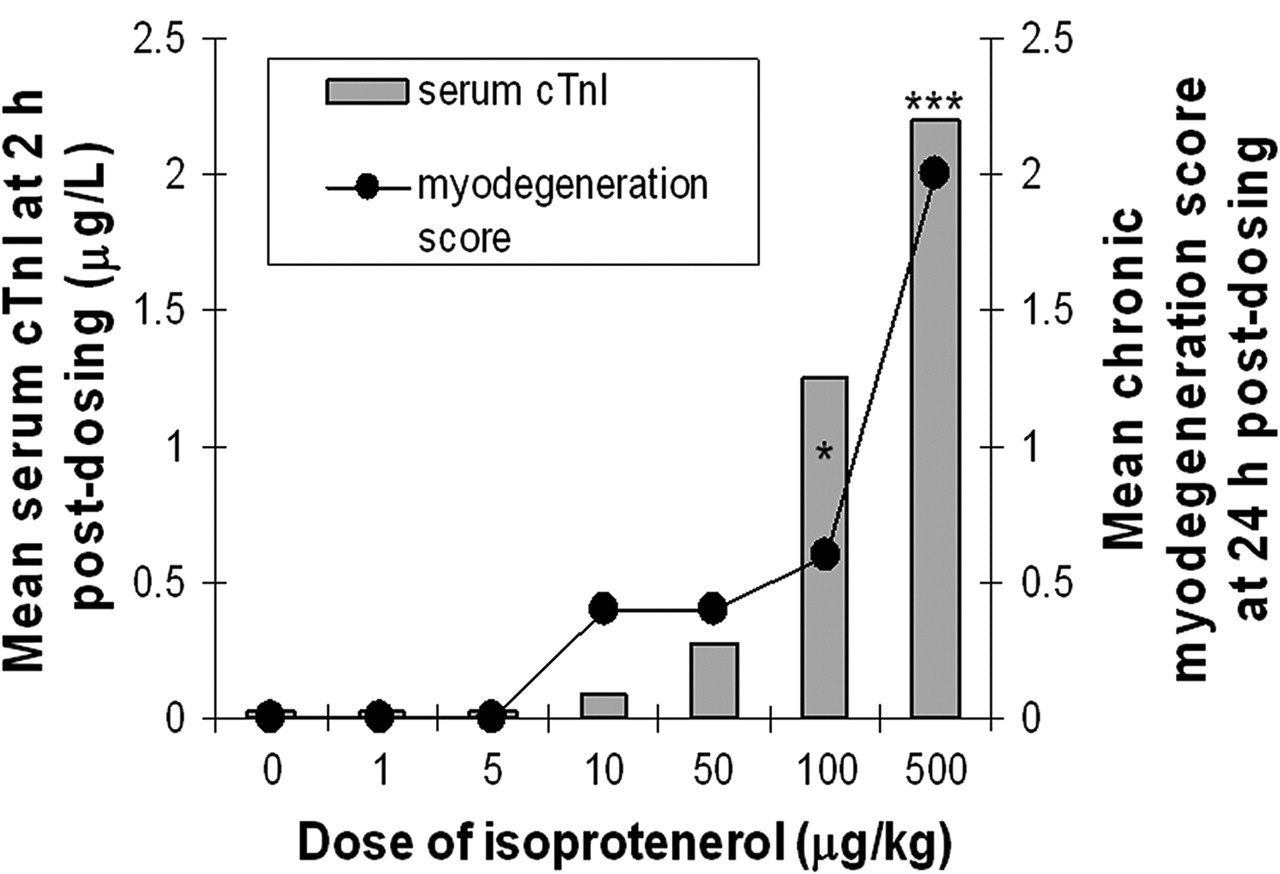
Experiment 1: Comparison of group mean cardiac troponin I (cTnI) levels at 2 hr postdosing with isoproterenol (ISO) at 1 to 500 μg/kg and the group mean microscopic cardiac lesion scores (severity grading) at 24 hr postdosing in female Hanover Wistar rats. There were 5 rats in each ISO dose level group, and 10 control (vehicle-treated) animals were sampled at 2 and 24 hr postdosing. Values for cTnI are measured in μg/L; chronic myodegeneration scores (severity grading): 0 = no abnormalities detected; 1 = minimal; 2 = mild; 3 = moderate. *cTnI levels significantly different from control, p < .05; ***p < .001. cTnI levels for ISO-treated and control groups were compared using a one-way analysis of variance (ANOVA) followed by Dunnett’s test for post hoc multiple comparisons.
Previous investigators have shown both different time courses and dose thresholds for induction of cardiac injury following acute singe doses of isoproterenol (O’Brien et al. 2006; Mikaelian et al. 2008; Schultze et al. 2008; Zhang et al. 2008; Clements et al. 2009). Whilst a combination of approaches have been used in these studies for scoring the severity of lesions over time, all investigations demonstrate that the severity of the microscopic lesion is influenced by dose and time point of collection. Full development of myocardial injury is generally observed at 24 hr postdosing and characterized by areas of acute necrosis, inflammatory cell infiltration, edema, interstitial fibrosis, and myocardial basement membrane alterations, primarily located in the left ventricle, septum, papillary muscle, and the apex of the heart.
Experiment 2 of the current investigation aimed to look at the effect of route of administration on subsequent levels of cTnI in serum. When rats were treated with ISO at 5,000 μg/kg either via the IP or SC routes (Table 3 ), elevations in cTnI (not statistically significant) were detected at 1 hr, peaked at 2 hr, and were decreasing at 4 hr postdosing. However, at all time points the levels of cTnI were higher when ISO was administered by the SC route. This experiment suggests that the route of administration may not significantly impact the timing of cTnI release, but it may affect the degree of cardiac injury, as it is assumed that the cTnI levels will reflect the extent of cardiac myofiber damage. In humans, the metabolism of ISO and conversion to active or inactive metabolites is dependent on the route of administration, with more inactive metabolites being formed following administration via the oral and inhaled routes, in comparison with the IV route (Sweetman 2006). In the current studies, the route of administration may have affected the metabolism of the drug, with greater glucuronidation (and therefore inactivation) occurring with the IP route of dosing (Dollery, Davies, and Connolly 1971; Brady 2008), resulting in lower levels of cardiac injury (making it harder to detect microscopically in standard histologic sections) and lower cTnI levels.
Experiment 2: Serum cardiac troponin I (cTnI) findings (μg/L) in individual control (vehicle-treated; 0 μg/kg; n = 5) female Hanover Wistar rats sampled at 0 hr, and in rats treated with isoproterenol (ISO; n = 5/time point) at 5,000 μg/kg by (A) intraperitoneal (IP) or (B) subcutaneous (SC) injection and sampled at 1, 2, and 4 hr postdosing. a
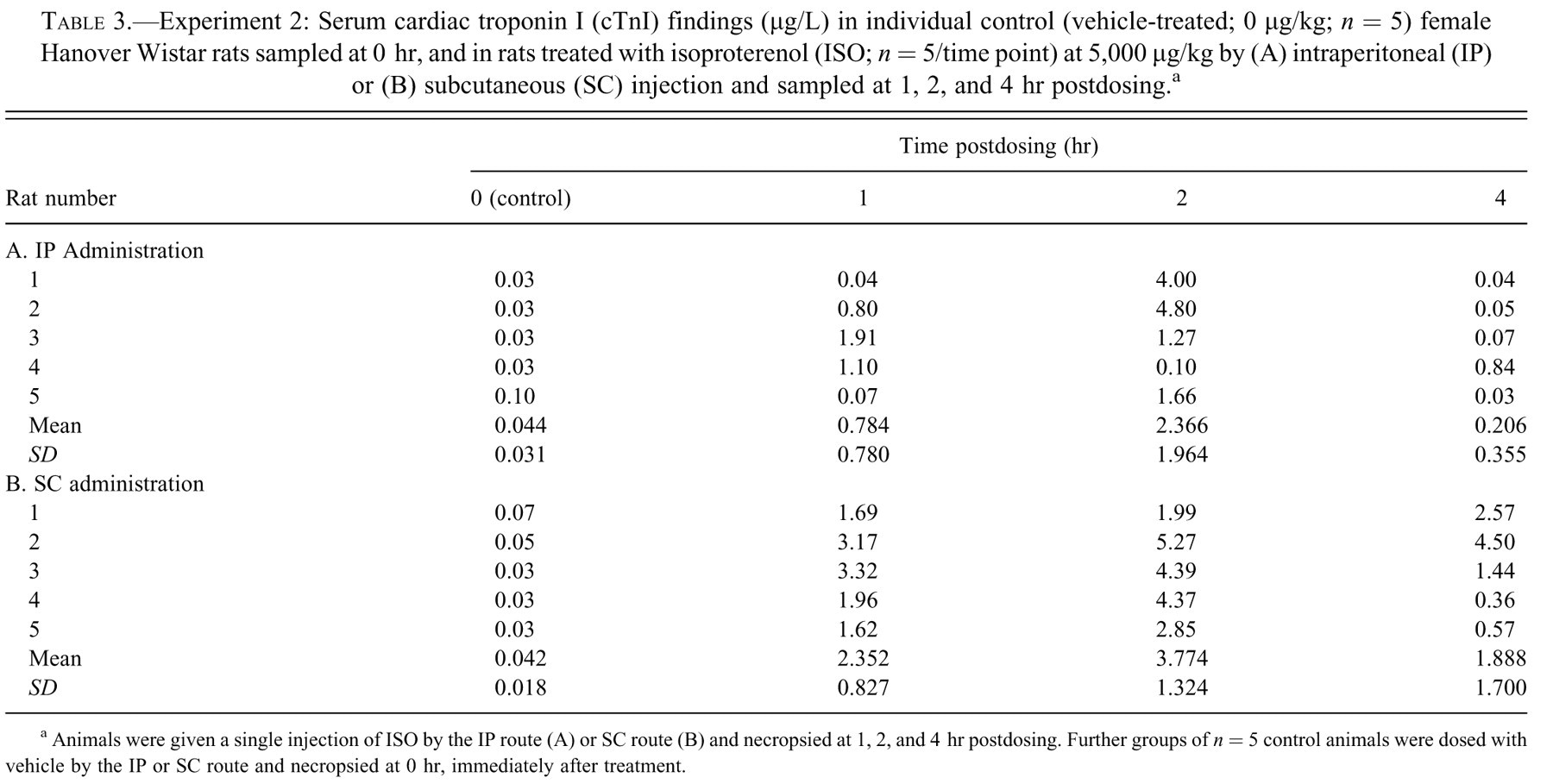
a Animals were given a single injection of ISO by the IP route (A) or SC route (B) and necropsied at 1, 2, and 4 hr postdosing. Further groups of n = 5 control animals were dosed with vehicle by the IP or SC route and necropsied at 0 hr, immediately after treatment.
Recently, Zhang et al. (2008) described two apparently different patterns of microscopic pathology in 8-week-old male Sprague Dawley rats dosed with low levels of ISO administered SC. At 3 and 6 hr postdosing (at doses of 8 to 64 μg/kg), minimal to mild lesions were described as a “Type 1 response” in the heart (characterized by minimal myofibrillar loss, cytoplasmic vesicles, minimal inflammatory cell infiltration, and a small amount of interstitial fluid). A more severe “Type 2 response” (characterized by myocyte necrosis, the presence of macrophages and leukocytes, fluid accumulation in interstitial spaces, interstitial fibrosis) was seen at dose levels of 125 to 500 μg/kg ISO, with necropsies at 3 to 24 hr postdosing. Zhang et al. suggested that the Type 1 response could indicate potentially reversible lesions, whereas the Type 2 response represented irreversible cell damage. However, the study did not investigate the potential progression of the Type 1 lesions induced by the lower doses after the 6 hr time point. The lesions seen in the current study at 24 hr with comparably low ISO doses are similar to those described as the Type 2 response by Zhang et al., suggesting progression over 24 hr to an irreversible lesion. It is considered that further studies are therefore needed before a reversible ISO-induced lesion can be confirmed.
Cardiac troponins are being intensely evaluated by the pharmaceutical industry as possible translational biomarkers to bridge the identification of cardiotoxicity from preclinical animal investigations to subsequent monitoring of potential early cardiac toxicity in human clinical trials (Wallace et al. 2004; O’Brien et al. 2006; Apple et al. 2008). One of the important factors in the evaluation of the sensitivity and usefulness of cTnI as a biomarker of cardiomyocyte injury is an understanding of the timing and lowest dose level at which both the tissue injury of interest, and the potential biomarker, can be detected. The present study provides evidence of a lowest effect level of 10 μg/kg ISO IP in Hanover Wistar rats for the detection of both injury by routine H&E histopathology and cTnI detection using the ACS180S assay. The investigations also highlight the importance of understanding the temporal disconnect between cTnI release and visible microscopic cardiac injury when using routine H&E sections and the potential effects of different routes of administration when using cTnI in the preclinical setting.
Footnotes
Acknowledgments
We acknowledge with thanks the cooperation of the technical staff at the School of Pharmacy for their care of the animals. We acknowledge the technical support provided by the Clinical Pathology and Histology Groups at GlaxoSmithKline, UK. SB acknowledges the support of the School of Pharmacy and GlaxoSmithKline, UK.
