Abstract
The main objective of this study was to investigate whether cardiac troponin (cTn) and N-terminal, protein B-type natriuretic peptide (NT-proBNP) can be useful as indicators for amitriptyline cardiotoxicity which is a known drug having sublethal toxic cardiac effects. At the same time, this study looked at detecting potential histopathological changes specific to irreversible cardiac injuries in a rat model of amitriptyline cardiotoxicity. Male Wistar rats were randomly divided into 2 groups, control (saline) group and amitriptyline group (100 mg/kg body weight intraperitoneally, equivalent for lethal dose at 50%). Blood was collected 30 minutes after the administration. The cTn was measured using 3 different methods (2 methods designed for human use and a sandwich enzyme immunoassay specific for rat cTnT). The brain natriuretic peptide was measured by 2 different methods (1 for human and 1 specific for rats). Electrocardiography showed that the QRS complex (P < .0001) and the QT interval (P = .002) were significantly prolonged for amitriptyline-treated animals. Troponin T and NT-proBNP had significantly increased levels in all the rats but showed positive results only when using rat-specific quantitative measurement. In certain rats, the histopathological examination identified a few small foci of acute myocardial necrosis. In conclusion, elevation of cTnT and NT-proBNP are early indicators of cardiotoxicity, yet the significance of irreversible myocardial damage in amitriptyline cardiotoxicity needs to be further understood.
Introduction
In clinical practice, numerous serum biomarkers are used or proposed for use in the diagnosis of myocardial injury, including drug-induced cardiotoxicity. Troponin is known as a “preferred biomarker” given its nearly absolute myocardial tissue specificity and high sensitivity, with the possibility to reflect microscopic areas of necrosis. 1,2
Several studies indicate that troponin (subunits cardiac troponin [cTn] T and cTnI) can be used as a biomarker when irreversible myocyte injuries are present (myocyte death with loss of sarcolemmal integrity) and levels are often correlated with histological changes. 3,4,5
Wu 6 showed that slightly elevated levels of troponin can also appear in unstable angina. His study does not clarify whether troponin is released as a result of nonlethal cellular lesions or whether angina is accompanied by a certain degree of myocyte death. There are several studies, which show that nonlethal cell injury such as arrhythmias, altered electrical activity, hypertrophia, and other pathological changes do not lead to troponin release in the absence of irreversible cellular lesions; however, more proof is required. 3,4,7,8
The use of troponin as a biomarker of toxic cardiac injury has been investigated previously, 9 and the results show that cTnT and cTnI are potent cardiotoxicity markers regardless of the cardiotoxic agents involved (sympathomimetics or chemotherapeutic drugs) in both humans and laboratory species.
Natriuretic peptides are released from the heart in response to pressure and volume overload. They have proven to be reliable biomarkers for heart failure. Brain natriuretic peptide (BNP) is synthesized and secreted by the ventricular cardiomyocytes. The BNP is synthesized and stored as full-length prohormones and, on secretion, is cleaved to yield equimolar amounts of N-terminal protein B-type natriuretic peptide (NT-proBNP) and the active BNP hormone. Although BNP is the active neurohormone, both BNP and NT-proBNP have been described as useful markers for the diagnosis and exclusion of congestive heart failure. 10,11
The BNP plasma levels reflecting left ventricular dysfunction were also noticed in preclinical settings such as cardiac toxicity after anthracyclines. 12,13
The association between BNP and troponin shows promise as a potential biomarker for cardiac functional injuries. 14
The cardiovascular complications of amitriptyline overdose are common. Although these effects are generally described as reversible cellular lesions induced by amitriptyline’s action on various receptors or ionic channels, there were 2 reported cases of amitriptyline-induced acute myocardial infarction. 15,16 These cases were diagnosed using electrocardiographic records and elevated cardiac enzymes. Other studies reported either electrocardiographic signs of ischemia 17,18,19 or elevated myocardial cytolysis enzymes, 20 but none of these findings were associated with myocardial necrosis.
The main purpose of this study was to investigate whether cTn and BNP can be useful as indicators for amitriptyline cardiotoxicity which is a known drug having sublethal toxic cardiac effects. At the same time, this study looked at detecting potential histopathological changes specific to irreversible cardiac injuries in a rat model of amitriptyline cardiotoxicity.
Materials and Methods
All animal experimental procedures employed in this study were strictly in accordance with the European Community guidelines regarding ethics and approved by the Ethics Committee of the “Gr. T. Popa” University of Medicine and Pharmacy, Iasi.
The animal breeding facility at the “Cantacuzino” National Institute for Research and Development for Microbiology and Immunology Bucharest supplied adult male Wistar rats with an average weight of 250 ± 20 g. The animals were housed in a temperature-controlled room (22°C ± 2°C) with 12-hour/12-hour light/dark cycle, humidity 50% ± 10%, and 2 rats per cage and were allowed to acclimatize for 14 days before use, with free access to food and water.
Drugs Administration and Sample Collection
Different groups of rats were injected intraperitoneally (IP) with one of the following formulations: (1) amitriptyline—Arena Group S.A., Bucharest, Romania, (n = 18), 100 mg/kg body weight (equivalent for IP lethal dose at 50% [LD50]) 21 in 1 mL NaCl 9% saline vehicle and (2) 1 mL saline for the control group (n = 6).
Light ether anesthesia was administered, and blood was collected from the retro-orbital sinus 30 minutes after administration. This study used 2 different methods for sample collection: Blood of 1 mL with EDTA as an anticoagulant for plasma collection; samples were centrifuged for 15 minutes at 1000 × g at 2°C to 8°C within 30 minutes of collection. Blood of 1mL allowed to clot in serum separator tubes at room temperature for 60 minutes; samples were centrifuged for 20 minutes at approximately 1000 × g.
Troponin Measurement
Troponin (cTn) was measured using 3 different methods: On Site Rapid Test (CTK Biotech Inc, San Diego, California) using plasma samples; it is a lateral flow chromatographic immunoassay used for qualitative detection of cTnI, and its complex in human plasma is ≥1ng/mL. BIOSITE Triage Meter Pro equipment (Biosite Inc, San Diego, California) using plasma samples; it is a fluorescence immunoassay method designed to provide quantitative results of Troponin I using plasma samples. Enzyme-linked immunosorbent assay (ELISA) kit for rat Troponin T (cTnT), (Uscn Life Science Inc Wuhan, China), an immunoassay for in vitro quantitative measurements for cTnT in serum samples. This method ensures excellent sensibility and specificity with no significant cross reactivity (minimum detectable cTnT level less than 5.2 pg/mL).
Brain Natriuretic Peptide Measurement
The BNP was measured using 2 different methods: BIOSITE Triage Meter Pro equipment using plasma samples: it is a method for the rapid quantitative measurement of NT-ProBNP in human whole blood or plasma specimens using EDTA as the anticoagulant. ELISA kit for rat BNP, Uscn Life Science Inc, an immunoassay for in vitro quantitative measurements of BNP in serum samples (minimum detectable level less than 0.038 pg/mL).
Electrocardiographic and Blood Pressure Measurements
Light ether anesthesia was used for the electrocardiographic and blood pressure measurements. The electrocardiographic measurements were taken 60 minutes after the administration of amitriptyline using a computer-based data acquisition system (BIOPAC SYSTEMS MP 150, Biopac Systems Inc, California). The universal interface module UIM 100C was used to connect HLT 100-series amplifier modules and signal cables to the MP150 system. The ECG100C Electrocardiogram Amplifier records electrical activity generated by the heart and were connected to the MP150 system. Electrocardiograms were recorded using LL-110 Lead-Lock electrode, designed for the difficult adhesion conditions. The electrodes were placed at the right and left front paws and the left hind paw.
Blood pressure measurements were carried out using the Small Animal Tail Noninvasive Blood Pressure System (NIBP 200A, Biopac System, Inc), based on the tail-cuff technique.
Data acquisition software (AcqKnowledge 3.9.0., Biopac) was used to analyze the following parameters: heart rate, duration of QRS complex, QT interval, PR interval, and systolic and diastolic blood pressure. For the electrocardiographic parameters, the values represent the average of 5 nonconsecutive complexes.
Histological Analysis
Necropsy was performed at 2 hours after dosing, and rats were anesthetized with ether, and the hearts were collected. Tissue samples were fixed in 4% formaldehyde, paraffin-embedded, and sectioned at 4 µm (serial longitudinal sections every 30 µm). Paraffin-embedded sections were stained with hematoxylin and eosin.
Statistical Analysis
Results were expressed as mean ± standard error of the mean. Comparisons between the groups were performed using 1-way analysis of variance followed by Bonferonni post hoc test for multiple comparisons. All P values were calculated using 2-tailed statistical tests. Differences were considered relevant for a significance level of P < 0.05. SPSS for Windows 18.0 was used for the data analysis.
Results
Of 18 animals, 6 animals receiving amitriptyline died within 10 minutes after drug administration.
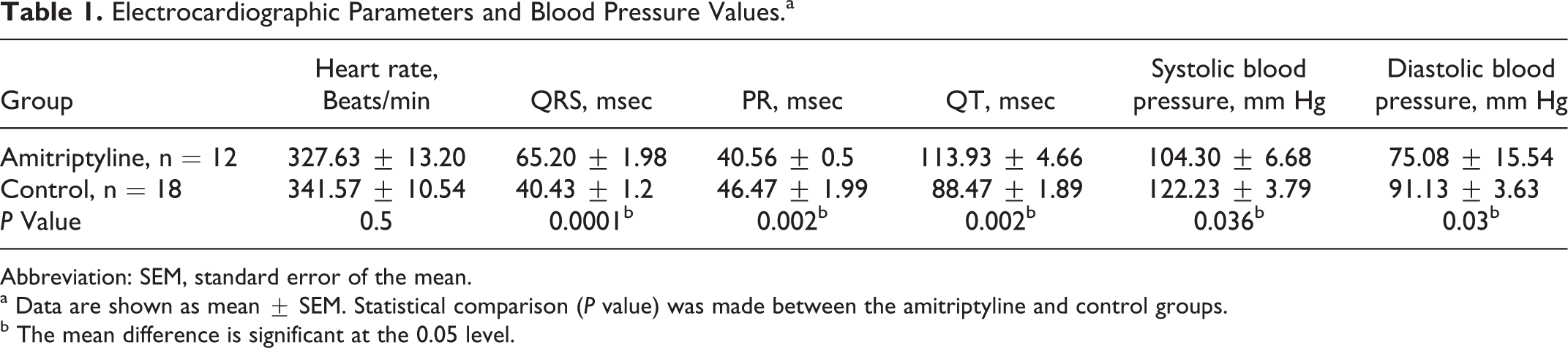
The results of the electrocardiographic and blood pressure measurements are illustrated comparatively between amitriptyline and control groups in Table 1.
Electrocardiographic Parameters and Blood Pressure Values.a
Abbreviation: SEM, standard error of the mean.
a Data are shown as mean ± SEM. Statistical comparison (P value) was made between the amitriptyline and control groups.
b The mean difference is significant at the 0.05 level.
The heart rate did not show significant difference between the 2 groups, with a range between 312 and 382 beats/min for the control animals and 253 to 402 beats/min for the amitriptyline-treated animals. Of the remaining 12 animals, 3 treated with amitriptyline had heart rate values significantly below the levels of the control group (281, 283, and 253 beats/min, respectively).
The PR interval was significantly shorter (P = 0.002) when animals were treated with amitriptyline. The QRS complex (P < 0.0001) and the QT interval (P = 0.002) were significantly prolonged for the animals treated with amitriptyline (Figure 1). Blood pressure decreased significantly when animals received amitriptyline for both systolic (P = 0.036) and diastolic (P = 0.03) measurements. In 30% of the animals (n = 4) that received amitriptyline, the electrocardiographic recordings showed signs of ischemic injuries (elevation of the ST-segment).
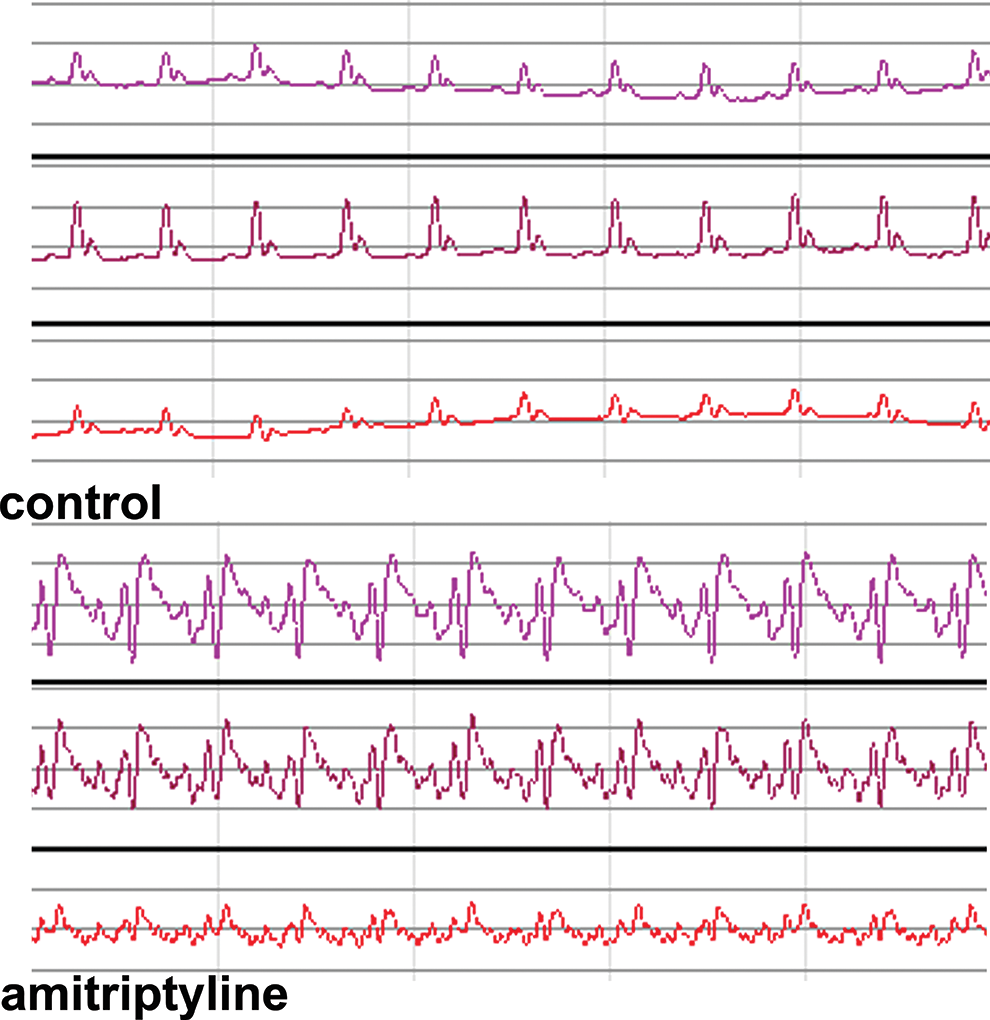
Electrocardiographic changes in rats treated with amitriptyline compared with saline control. Animals treated with amitriptyline show elongation of the QRS complex and of the QT interval.
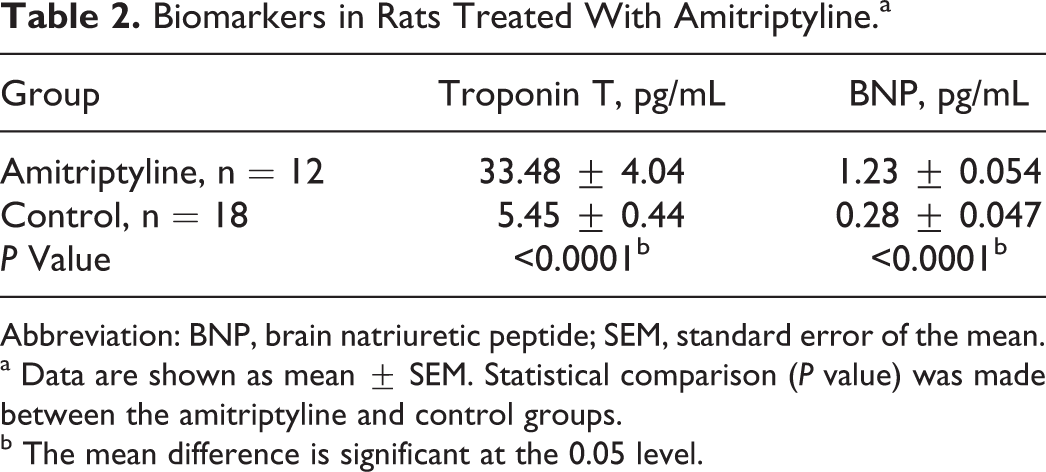
The troponin measurement done using the On Site Rapid Test, CTK Biotech Inc, did not show positive results in either of the 2 groups tested. Similarly, using the BIOSITE Triage Meter Pro equipment, the levels of the biomarkers we investigated were below the minimum detectable (cTnI < 0.05 ng/mL, BNP < 5.0 pg/mL). The troponin T (TnT)serum levels measured using ELISA kit for rat TnT revealed a range between 4.2 and 6.9 pg/mL for the control group, with an average of 5.45 ± 0.44 pg/mL. The group treated with amitriptyline showed a range between 18.8 and 59.8 pg/mL, with an average of 33.48 ± 4.04 pg/mL, significantly higher than the control values (P < 0.0001; Table 2).
Biomarkers in Rats Treated With Amitriptyline.a
Abbreviation: BNP, brain natriuretic peptide; SEM, standard error of the mean.
a Data are shown as mean ± SEM. Statistical comparison (P value) was made between the amitriptyline and control groups.
b The mean difference is significant at the 0.05 level.
The proBNP serum levels measured using ELISA kit for rat NT-ProBNP revealed a range between 0.20 and 0.50 pg/mL for the control group, with an average of 0.28 ± 0.047 pg/mL. The group treated with amitriptyline showed ranges between 0.90 and 1.5 pg/mL, with an average of 1.23 ± 0.054 pg/mL, significantly higher than the control values (P < 0.0001; Table 2).
The histological examination of heart tissue samples for the control group showed a normal microscopic structure—mononucleate fibers with clearly visible striations. Similar characteristics were visible when an increased magnification was used (Figure 2A).
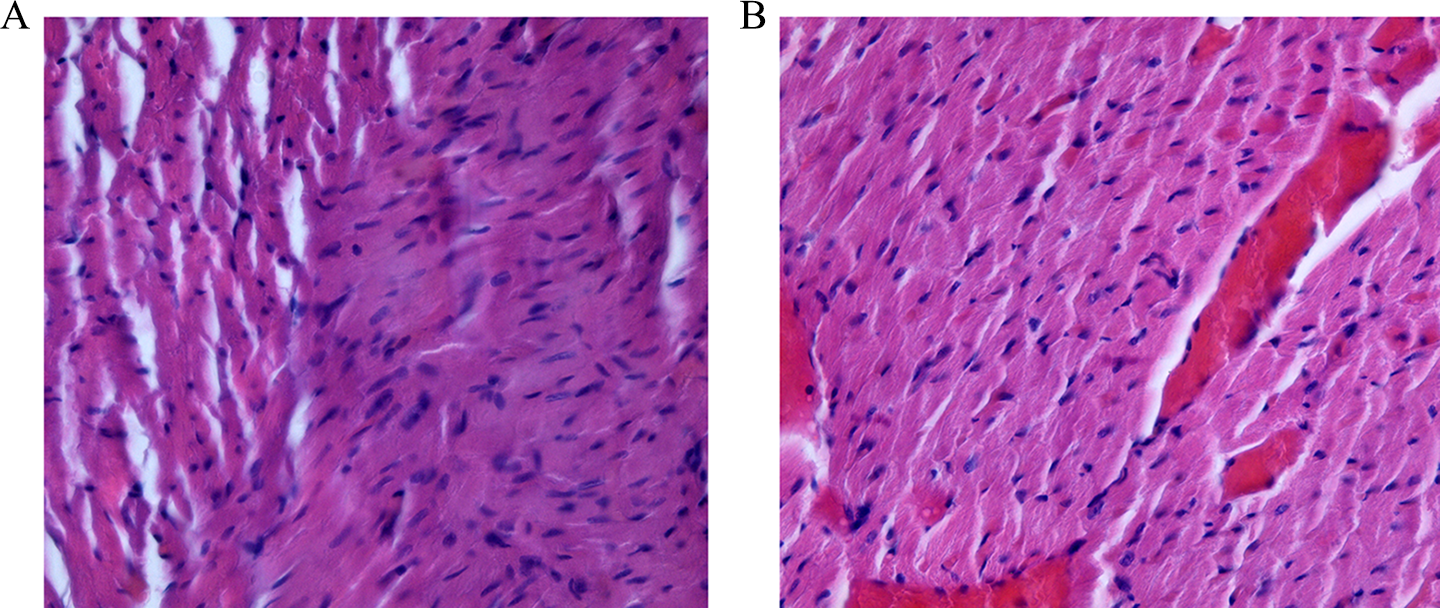
Hematoxylin and eosin staining on heart sections. A, Control group with normal microscopic structure. B, Amitriptyline-treated rats with dilated capillaries and microareas of necrosis with disappearance or shrinkage of nuclei.
The amitriptyline administration induces diffuse myocardial alterations, such as dilated capillaries, hemorrhagic areas with variable sizes, and, in over 30% of the treated animals, few small foci of acute myocardial necrosis characterized by pyknosis or karyolysis (Figure 2B). Histopathological alteration specific for myocardial necrosis was correlated with the rats that had the highest cTnT concentration.
Discussion and Conclusion
This study confirmed that cTnT is an early marker of cardiotoxicity for amitriptyline, an antidepressant with known nonlethal cardiomyocytic toxicity. According to our knowledge, no other preclinical study about biomarkers of amitriptyline cardiotoxicity has been reported until now. These results point out that acute myocardial necrosis can be the result of amitriptyline toxicity as previously reported in the literature 15,16 ; however, a larger animal sample is necessary to strongly confirm this hypothesis.
In order to avoid biased sources that might influence the validity of the data, it is important to use validated experimental methodologies.
In acute toxicity short-term studies, high doses of drugs are recommended, maximizing the potential for identification of organ damage. 22 Also, it is well known that cardiac toxic effects of tricyclic antidepressants occur after high doses. 23 The LD50 value for IP administered amitriptyline was used as described in the literature. 21,24
We preferred to deliver the drug IP, based on the fact that this route of administration (IP injection) requires minimal skill and does not result in lesions or signs of pain when drugs are administered. Also, IP injection has the advantage of being both simple and also causes minimum distress to the rats. 25 However, amitriptyline distribution in the heart is independent of the route of administration. 26 Furthermore, anesthesia has a number of effects on the cardiovascular system. For example, general anesthesia alters autonomic tone, whereas inhalation anesthetics, including halothane, enflurane, and methoxyflurane, have been shown to have pro- or antiarrhythmic effects and inhibit critical Ca2+ channels (ie, halothane and isoflurane) in the heart. 27 Light ether anesthesia was used due to the fact that studies regarding optimal anesthesia in experimental cardiology recommend this method over other anesthetics. 28
The time of blood collection was established at 30 minutes after the amitriptyline injection, this decision being based on 2 facts: enzymes are released early from the cardiomyocytes in toxic injuries 29 and TnT half-life is relatively short—about 120 minutes. 1 The half-life of BNP is only 22 minutes, whereas the half-life of NT-proBNP is much longer, around 120 minutes for a normal glomerular filtration rate status. In vitro, BNP is less stable than NT-proBNP if blood is not collected in plastic tubes containing EDTA as an anticoagulant. 22
The animals were sacrificed, and hearts were collected 2 hours post dosing, the biological half-life of amitriptyline in rats being approximately 2 hours. 26
Another possible source of bias in the method used for blood collection is that heparin can link with cTn molecules leading to decreased immunoreactivity and false-negative results. 30 It is also noteworthy that concentrations of troponins measured in heparin plasma are markedly lower than in serum in some cases. 31 Because of this, the blood collection in the present study was made with EDTA as an anticoagulant for plasma collection and in blood tubes with clot activator for serum collection. To avoid low or absent reactivity of rat blood tested for cTn or BNP using methods designed for human use, a rat-specific immunologic method was comparatively utilized. In the present study, the rat-specific ELISA assay proved to be the only method sensitive to quantitative measurements in vitro, while the human assay was not suitable for troponin or BNP measurement in rat.
The lack of reactivity of commercial grade human biochemical methods when used in animal species in preclinical studies was mentioned by other authors. 14,32 However, previous studies 33 used human-developed assays for immunological detection of the TnT and TnI in rats, assessing that these assays could be used effectively in the different preclinical species, due to the fact that troponin sequences are preserved between species. Recent published medical scientific literature brings information that not all the human cTn assays were found to be suitable for cTn measurement in each laboratory species, and individual assay characterization is needed for each animal species. 22,32
Cardiac troponins are the most sensitive and specific biochemical markers of myocardial damage, but there have been some reports indicating false-positive troponin value caused by analytical interferences with this assay: fibrin clots, microparticles in sample, heterophile and human antianimal antibodies, rheumatoid factor, macroimmunocomplex formation, and analyzer malfunction. 34,35 In general, analytical interferences responsible for false-positive troponin results are associated with a specific manufacturer’s troponin assay and are not encountered in all troponin assays. 34
To prevent these issues, the samples were checked for possible microparticles or clots, and the specimens were centrifuged according to the assay manufacture’s specification.
Another known possible source of bias is the fact that in patients having end-stage renal failure, cTnT, and cTnI may be increased in serum without clinical signs of acute myocardial damage. The detection of cTn during renal failure was suggested to be indicative of myocardial damage due to the hemodynamic changes associated with kidney failure. However, troponin elevation indicates the presence, not the mechanism, of myocardial injury. 1,36 It has also been proved that amitriptyline has no direct renal toxic effect. 23 In the present study, the possibility of renal failure induced by other factors (stress from sample procedures, IP injection, or anesthetic), with secondary rises in biomarkers level was not reliable, because the procedures were identical in both control and amitriptyline groups, but the levels of the biomarkers were significantly higher in the amitriptyline group.
The experimental amitriptyline toxicity replicated the known electrocardiographic and blood pressure changes that characterize tricyclic antidepressants toxicity. Our recordings showed prolonged QRS complex and QT interval; shortening of the PR interval; arterial hypotension; and in 4 of the animals, signs of acute myocardial ischemia—elevation of the ST segment.
Most importantly, TnT and BNP were significantly increased in the animals treated with amitriptyline. This elevation was manifested early, 30 minutes after injecting the drug, a situation previously described in the literature for experimental toxicity with isoproterenol. 29
The electrocardiographic ischemic changes were recorded in rats with histological pattern of myocardial necrosis. This is indicative of the fact that amitriptyline toxicity can lead to irreversible cellular lesions, a result similar to certain clinical observations already noted in the literature. 15,16 Myocardial necrosis is recognized as a possible complication in tricyclic antidepressants toxicity, as reported previously. In 1988, Chamsi-Pasha and Barnes 15 presented the case of a 22-year-old with anteroseptal myocardial infarction 24 hours after ingesting 300 mg amitriptyline. Some authors 18,37,38 reported abnormal atrial and ventricular repolarization, resembling myocardial injury after tricyclic antidepressant drug intoxication, but the level of myocardial enzymes was normal. The electrocardiographic pattern described was the Brugada syndrome.
Electrocardiographic changes suggestive for myocardial infarction were described in the literature 17,19 in the acute poisoning with dothiepin, although Steeds considers that the cause was the blockade of potassium and rapid sodium channels rather than a real myocardial injury.
In 2006, the case of a 33-year-old female patient was published 16 following the ingestion of 300 mg amitriptyline. The patient met all 3 elements for the diagnosis of acute myocardial infarction: clinical signs, electrocardiographic changes, and elevated myocardial enzymes (creatine kinase [CK], creatine kinase MB [CK-MB], TnI).
Certain cases of isolated elevation of enzymatic levels were previously described in this toxic pathology. Guthrie and Lott 20 observed increased levels of CK and CK-MB in amitriptyline toxicity, but these changes were not accompanied by changes in electrocardiographic or coronarographic recordings.
The current study showed enzymatic changes in all the animals treated with amitriptyline in toxic doses, but myocardial necrosis was observed only in 4 of these animals. This raises the hypothesis of an enzymatic release even when the irreversible myocardial injuries are absent. In this context, the troponin measurement appears to be a sensitive method for the early diagnosis of cardiotoxicity.
The proBNP, a known marker for cardiac failure, is also elevated in this model of acute amitriptyline poisoning, suggesting that proBNP could also be used as a biomarker for cardiac toxicity.
The NT-proBNP proved to be a helpful diagnostic indicator for identifying asymptomatic acute and subacute ischemic myocardial injury. 39
Increases in NT-proBNP preceded anatomical and functional changes associated with cardiotoxicity. 22
This study sustains the previous observations that troponin and BNP could be used in association as markers for cardiac functional injuries. 14
In conclusion, the present results provided evidence to attest the fact that cTnT and NT-proBNP should be considered as early biomarkers for cardiotoxicity induced by amitriptyline.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
