Abstract
Cardiovascular toxicity represents one of the major reasons for the termination of the development of drugs, even in late development phases. This growing issue is often not restricted to specific therapeutic areas, and it is gaining critical importance, in particular for chronically administered drugs, highlighting the limitations in terms of sensitivity of the current investigational paradigms. Furthermore, drug-related changes may become evident after long-term administration for different reasons, including accumulation of the drug in the heart. This article describes how the integrated use of investigational tools represents a powerful approach for the early identification and characterization of cardiotoxicity in preclinical development. Cardiac changes were observed in the dog after long-term oral administration of casopitant, a neurokinin 1 receptor antagonist, developed for the treatment of depression and anxiety. Different approaches and sensitive biomarkers were used in a time-course study to investigate the onset, progression, and reversibility of the lesion. The integrated evaluation of cardiovascular parameters, electron microscopy, troponin I, and natriuretic peptide results highlighted any minimal early changes, allowing the full and deep characterization of the lesion. The outcome of this study was the driver for drug development decision making on casopitant and backup drugs.
Keywords
Introduction
Cardiovascular toxicity is a growing issue in the preclinical development of new drugs. It accounts for approximately 27% of drug development failures (Car 2006), and it does not seem to be restricted to specific high-risk therapeutic areas. For example, the psychiatric/neurologic therapeutic area has the higher attrition rate, with a 22% failure rate in drug development, of which approximately 16% is related to cardiovascular causes (Olson et al. 2000). These data suggest low attrition caused by cardiovascular complications for CNS-active compounds; however, examples of cardiac toxicity have already been described in literature in the past, particularly for tricyclic antidepressants (Burckhardt et al. 1978; Jefferson 1975). Furthermore, the toxicological risk assessment of psychiatric or neurology drugs is even more critical, because of the chronic treatments often needed and the physical-chemical properties of candidate drugs that may lead to tissue accumulation of potentially toxic metabolites. The ability of drugs to permeate the blood–brain barrier requires specific physical-chemical properties that normally include high lipophilicity, which is often associated with high distribution volumes and tissue accumulation (Lafuente et al. 2009; Monro 1990); furthermore, the cationic amphiphilic structure often adopted by central nervous system (CNS)-active drugs can potentially cause phospholipidosis (Anderson and Borlak 2006; Ikeda et al. 2008; Rabkin 2006; Vonderfecht et al. 2004). Numerous drugs and chemicals containing a cationic amphiphilic structure, despite their diverse pharmacological activities, are capable of inducing phospholipidosis in animals and humans; the main features of this finding include the reversible accumulation of polar phospholipids in association with the development of lamellar bodies within cells (Nonoyama and Fukuda 2008; Reasor et al. 2006). Examples of cationic amphiphilic drugs (CADs) are described in the literature to affect several organs; even if skeletal muscles are sometimes reported to be affected, the heart has only very rarely been found to be involved (Drenckhahn and Lüllmann-Rauch 1979; Halliwell 1997; Jagel and Lüllmann-Rauch 1984; Roos et al. 2002; Shaikh and Downar 1987).
Several tools are available to mitigate the risk of potential cardiovascular liabilities at the early phases of preclinical drug development. However, many causes, including potentially toxic metabolite accumulation over time, can cause relevant target organ toxicity only in long-term toxicity studies and in a later stage of drug development. This was the case for casopitant, a potent and selective antagonist of the human neurokinin 1 (NK1) receptor developed for the treatment of depression and anxiety, whose cardiac toxicity was evident at the histopathological evaluations only after long-term administration in dogs. Neurokinin 1 receptors are widely distributed (also found in non-neural tissues such as endothelial and inflammatory cells, as well as gastrointestinal, respiratory, and genitourinary tissues), but this mechanism of pharmacological action is considered unlikely (directly or indirectly) to be related to the cardiovascular toxicity observed. In particular, oral administration of casopitant in a thirty-nine-week dog study caused increased heart weight and myocardial necrosis, degeneration, and inflammation associated with higher plasma levels of cardiac troponin I and creatine kinase MB mass (CK-MB) at the doses of 25 and 40 mg/kg/day. In addition, transmission electron microscopy (TEM) examination, considered the gold standard to confirm a diagnosis of phospholipidosis, showed ultrastructural changes in the heart that were considered to be suggestive of phospholipid accumulation. Previous short-term studies up to thirteen weeks in duration demonstrated that casopitant was extensively metabolized (Miraglia et al. 2010), widely distributed, and retained for longer periods in tissues (as such, and/or as drug-related material), but cardiac changes were limited to a minimal increase of heart weight. At this stage of drug development, the casopitant toxicological concerns were limited mainly to the cationic amphiphilic nature of the chemical structure, possibly leading to phospholipidosis-related changes (Anderson and Borlak 2006; Ikeda et al. 2008). Drug tissue accumulation with chronic administration provided the basis for the late-onset toxicity, and the cardiac changes were evident only in the thirty-nine-week study in dogs. In this scenario and at this stage of drug development, the characterization of the cardiac toxicity was investigated to evaluate the onset, progression, and reversibility of the cardiac changes to identify early markers of cardiac impairment with focus on their translatability to the clinical setting. The aim of this study was to provide a fully integrated evaluation of investigational tools to enable the early identification and characterization of casopitant-induced cardiac toxicity in the dog. In this context, it was also considered how to translate this information to a human setting to support the development of other backup drugs in the same chemical class.
Materials and Methods
Study Design
An investigative study was set up to provide a broad and complete toxicological characterization of a possible cardiac liability, likely to be associated with long-term administration of casopitant. The main objective of this study was to characterize the onset, progression, and reversibility of the cardiac lesion by using a number of standard and nonstandard evaluations, as described below. The investigative repeat-dose study required the oral administration of casopitant in male dogs for up to twenty-six weeks, and it included interim kills and a recovery period of twenty-two weeks. In detail, animals were randomly assigned to two treatment groups (n = 12 receiving vehicle alone, and n = 15 receiving casopitant at 40 mg/kg/day). All dogs received a daily oral gavage dose of casopitant or vehicle (dose volume of 10 mL/kg). A restricted group of animals (n = 2 receiving vehicle alone, and n = 3 receiving casopitant at 40 mg/kg/day) were similarly treated for thirteen weeks followed by a twenty-two-week recovery period (Figure 1 ). After six, thirteen, twenty, or twenty-six weeks of treatment, or following completion of a twenty-two-week recovery period, three treated animals and two or three control (vehicle) animals at each time point were deprived of food overnight and killed by intravenous injection (via the jugular vein) of sodium pentobarbitone 200 mg/mL, 0.75 ml/kg (Pentobarbital sodium, Sigma Aldrich, St. Louis, MO, USA) and sodium heparin 5,000 U/mL, 0.2 mL/kg (Clarisco Teofarma, Pavia, Italy) and then exsanguinated.
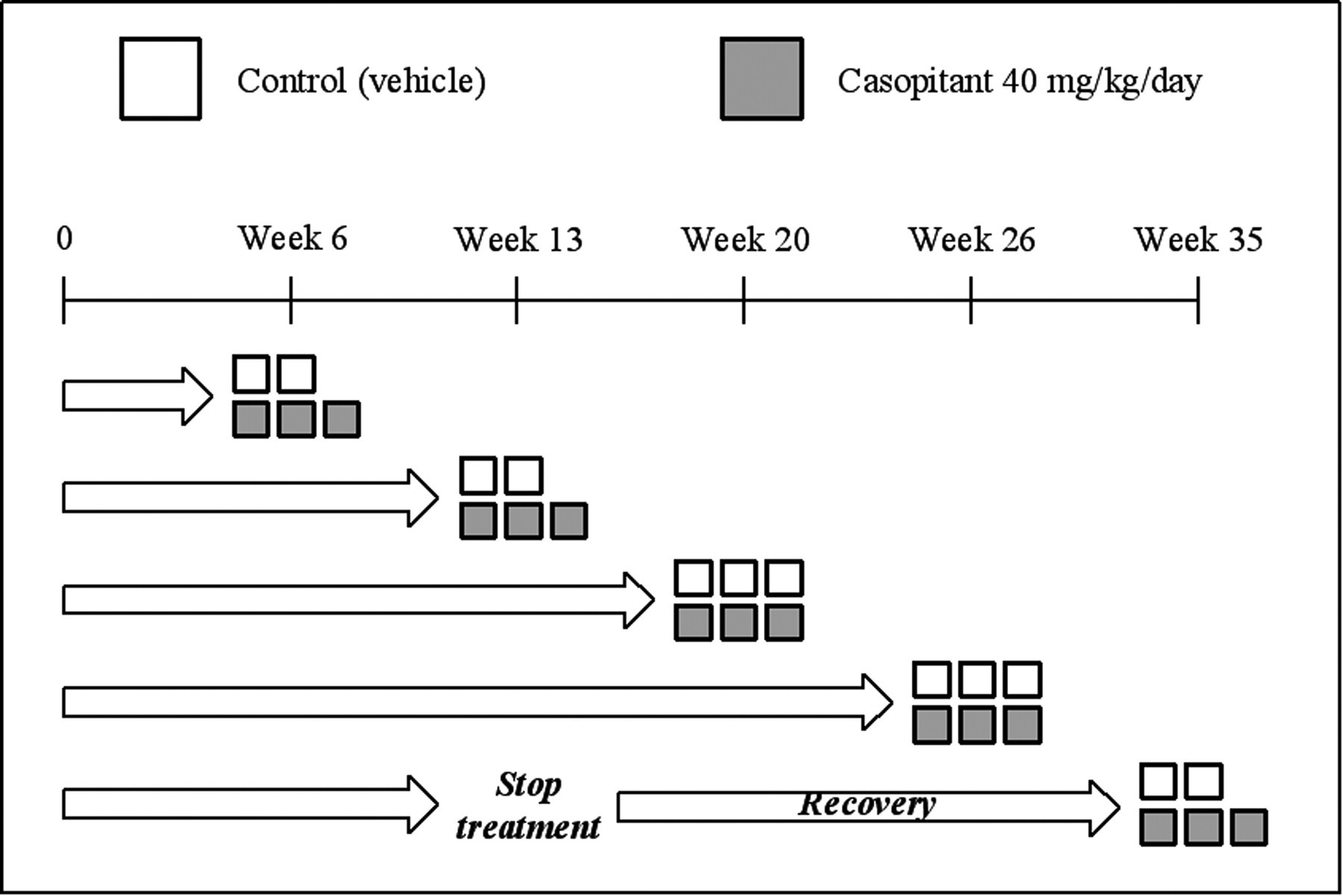
Study design and scheduled necropsies. Each box represents a single animal—white for control (vehicle) dogs and grey for casopitant-treated dogs.
The first day of dosing was designated Day 1; days in the pretreatment period were assigned negative numbers.
Animals and Maintenance
Twenty-seven male beagle dogs, obtained from Harlan Ltd., Blackthorn, UK, were used in the study. All animals were housed individually in concrete/solid-floored pens with sawdust litter. Following arrival, animals were acclimated to local housing conditions for approximately 27 days before the start of dosing, and on the first day of treatment, the animals were approximately eleven to twelve months old and weighed 10.5 to 13.7 kg.
Each dog was offered approximately 400 g of pelleted diet (supplier Harlan Teklad 2021, Blackthorn, Bicester, England) daily time. Food was not provided during the night before each bleeding occasion or scheduled necropsy.
Statement of Ethical Approval
All work involving animals was carried out in accordance with European directive 86/609/EEC governing animal welfare and protection, which is acknowledged by Italian Legislative Decree no. 116, 27 January 1992, and according to internal reviews performed by the GlaxoSmithKline Committee on Animal Research and Ethics (CARE) and to company policy on the care and use of laboratory animals.
Clinical Observations
A check of food consumption, as well as of the condition and behavior of all animals, was made daily starting from Day −21 (Week −3) and throughout the treatment period, whereas body weight was measured weekly.
Postmortem Evaluations
During necropsy sessions, a detailed macroscopic evaluation was performed on all animals killed at different time points. Hearts were taken from each animal (interim kill, terminal kill, and recovery kill), briefly washed in cool saline solution, and weighed using a XP1203S Mettler Toledo scale. Small pieces of heart tissue (left and right ventricle myocardium) were collected from all animals for TEM evaluation, fixed, and preserved in 4% formaldehyde/1% gluteraldehyde in phosphate buffer for at least forty-eight hours. The remaining heart tissue was fixed and preserved in 10% neutral buffered formalin for at least ninety-six hours.
Histopathology Examination
Postmortem analysis on hearts of dogs killed at different time points was performed by extensive histopathological evaluation. From all control (vehicle) and test article–treated dogs, cardiac tissue was selected, including both auricles, left ventricular wall, left papillary muscle, apex, interventricular septum, and atrioventricular valves. All selected samples were processed by an automated processor (Tissue-Tek VIP 5-Sakura, Torrance, CA, USA) and embedded into paraffin wax blocks. From these, 3-µm thin sections were produced using a rotative microtome (Micron HM355S, Haryana, India), stained with hematoxylin and eosin (Tissue-Tek DRS 2000 Slide Stainer, Medical Equipment Source, LLC, Mars, PA, USA), and microscopically examined with a light microscope. To further characterize cardiac histological changes, special stains (Azan’s trichrome, and phosphotungstic acid-hematoxylin PTAH) and immunohistochemical staining for cardiac troponin I (cTnI, cat. no. ab19615, mouse monoclonal, Abcam, Cambridge, MA, USA), caspase 3 cleaved (Cell Signaling, Santa Cruz, CA, USA, cat. no. 9661, rabbit polyclonal), brain natriuretic peptide (BNP; Peninsula Laboratories, Inc. via Bachem, Bubendorf, Switzerland, cat. no. T-4200, rabbit polyclonal), and atrial natriuretic peptide (ANP; Phoenix Pharmaceuticals, Burlingame, CA, USA, cat. no. H-005-06, rabbit polyclonal) were applied on representative myocardium sections taken from selected control and test article–treated animals.
The severity of degenerative changes was graded according to an estimate of the overall percentage of myofibers involved in each section. In this scheme, grade minimal represented involvement of 10% to 20% of myofibers; grade mild, 30% to 50%; grade moderate, 60% to 80%; grade marked, almost all myofiber involvement.
Clinical Pathology Analyses
Starting from Day −21 (Week −3) and every two weeks throughout the treatment period, approximately 5 mL blood were collected into tubes without anticoagulant for serum, and the following parameters were evaluated: cardiac troponin I (cTnI) and creatine kinase MBmass (CK-MB) were measured by using an ADVIA Centaur CP (Siemens, Munich, Germany) immunochemistry automated analyser; NT-pro BNP was measured by using the Canine CardioSCREEN EIA enzyme immunoassay (VETSIGN, Guildhay Ltd, Guilford, UK). Electrolytes (sodium, potassium, chloride, calcium, and inorganic phosphorus) were also evaluated at different time points throughout the study by using an ADVIA 1650 automated analyzer (Siemens, Munich, Germany). These biomarkers were chosen on the basis of their high sensitivity, specificity, and prognostic value, including the possibility of being applied to monitor human beings in clinical phases (Ishii et al. 2003; Morello and Januzzi 2006).
Transmission Electron Microscopy
Phospholipidosis-related changes were evaluated in the heart using TEM, as the ultrastructural examination remains the gold standard for phospholipidosis evaluation (Mortuza et al. 2003). Approximately 1 mm3 of cardiac tissue for each sample was trimmed, post-fixed in 1% osmium tetroxide, processed using an automated processor (EMP5160, RMC Products, Tucson, AZ, USA), and embedded into EPON 812 resin blocks. Semithin sections (nominal thickness 0.5 to 1 µm) were produced using an ultra-microtome (PT-PC Power Tome, RMC Products, Tucson, AZ, USA), mounted onto glass slides, and stained with 1% toluidine blue (aqueous solution) to select appropriate areas and tissue orientation for TEM evaluation. The slides obtained were checked by a light microscope. Ultrathin (about 60 nm) sections were cut using the same ultra-microtome and stained using an EM AC20 electron microscopy stainer (Leica, Wetzlar, Germany) using 0.1% uranyl acetate and 0.25% lead citrate aqueous solutions. The sections obtained for each sample were examined under a transmission electron microscope MORGAGNI 268D (Philips Healthcare, Netherlands) operating at 90KV.
Cardiovascular Functional Evaluations
The full characterization of the onset and progression of the lesion were also monitored using electrocardiogram (ECG) and echocardiography evaluations (unpublished data). Electrocardiograms and echocardiographic parameters were recorded from each animal once prior to the initiation of dosing and at approximately six to seven-week intervals during the treatment and recovery periods. Recordings were made prior to dosing and approximately one hour after dosing for ECG and only post-dose for echocardiography.
Electrocardiogram recordings were made using leads I, II, III, aVR, aVL, aVF, V1, V2, V3, V4, V5, and V6. All leads were analyzed to assess potential wave configuration and rhythm changes. Heart rate, PR and QT intervals, and QRS complex duration on at least five consecutive complexes were calculated.
Echocardiographic imaging was obtained using a Vivid 7i (General Electric, UK) ultrasound scanner equipped with a multiple set of transducers. A phased-array matrix transducer specific for cardiology application was used at frequencies ranging between 3.5 and 6 MHz (M4S, General Electric).
Toxicokinetic Evaluations
All of the above evaluations were also supported by the measurements of parent compound and main metabolite concentrations in both plasma and cardiac tissue, to evaluate the potential accumulation of drug-related material and its relationship with the pathological findings. Plasma samples were obtained from blood collected into K3EDTA tubes at the following nominal times: predose, thirty minutes, one, two, four, six, eight, ten, and twenty-four hours after dosing. All animals were bled on Day 1, and two or three animals were bled on the day prior to each necropsy at Weeks 6, 13, 20, and 26. In addition, during each necropsy occasion, samples of 3-5 g (each) of myocardium were collected from all animals, weighed, briefly washed in ice-cold saline, and frozen on solid carbon dioxide. Quantification of casopitant and selected metabolites in dog plasma and myocardium homogenate was performed by using two analytical methods based on protein precipitation with acetonitrile, followed by HPLC tandem mass spectrometry (HPLC-MS/MS) analysis (Pagliarusco et al., unpublished data).
Results
Clinical Observations
Instances of salivation were observed for all animals at 40 mg/kg/day from the first week of treatment. Red gums were first observed in one animal at Week 3 of treatment, and this effect subsequently progressed to all animals by Week 9, when mucosal erosion (around the incisors/canines) and thickening of the gums began to be observed. Red eyes were also evident from Week 5 of treatment.
Postmortem Evaluations
No additional findings at macroscopic examination were observed. Table 1 summarizes the differences noted in heart weights between treated animals and controls throughout the study, following completion of the indicated treatment period. Minimal increases in absolute and relative (to body weight) group mean heart weight were apparent from Week 6, with further increases at Weeks 13 and 20 (for the relative weight, up to 1.17, 1.39, and 1.47 times the control value, respectively). At the end of the treatment period (Week 26), there were no further increases in heart weight, with values at Week 26 remaining similar to those at Week 20 (up to 1.45 times the control values, relative weight). No complete recovery was seen after twenty-two weeks of treatment-free period, even if signs of a limited degree of reversibility were apparent (1.30 times the control values at the end of the recovery period).
Heart weights, absolute and percentage relative to body weight, at the different time points.
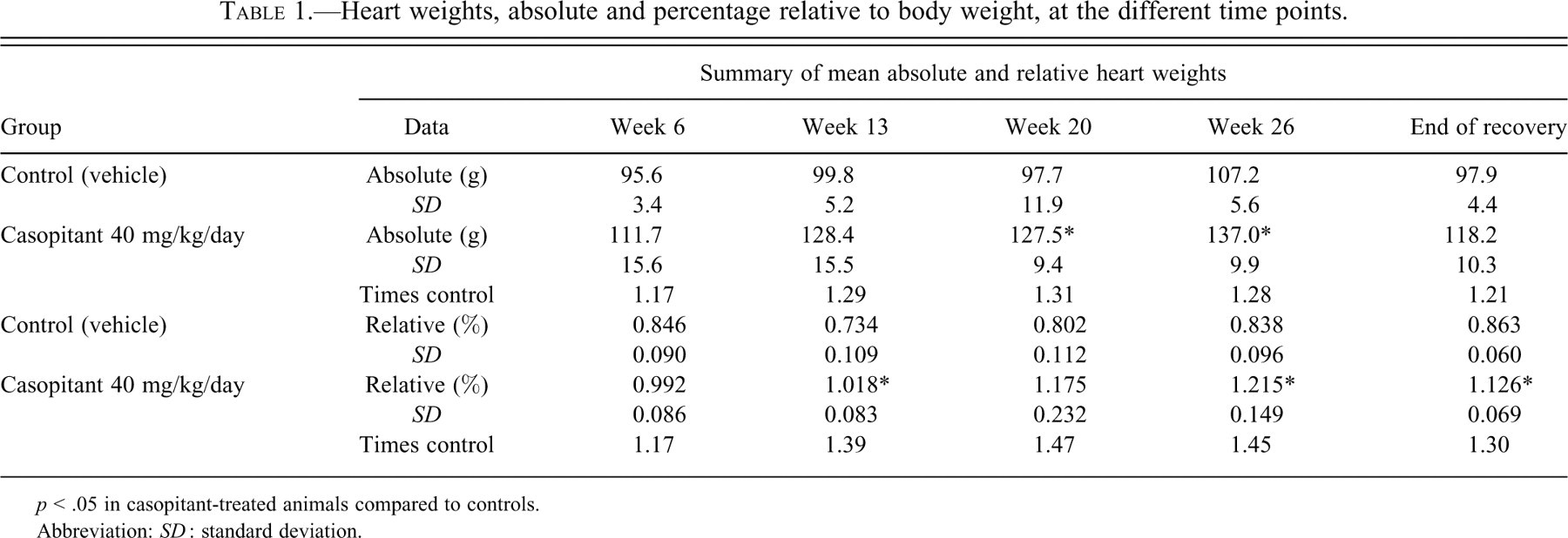
p < .05 in casopitant-treated animals compared to controls.
Abbreviation: SD : standard deviation.
Histopathology Examination
Test article–related microscopic findings were seen in dogs following twenty or twenty-six weeks of treatment; earlier time points showed a normal cardiac morphology. Cardiac findings consisted of myofiber degeneration/necrosis, minimal to moderate and multifocal (Figure 2a–d). In detail, two of three dogs were affected after twenty weeks and three out of three dogs after twenty-six weeks of treatment. The lesion affected all cardiac walls, with no particular prevalence. In sections stained with PTAH, degenerating/necrotic myocardiocytes observed at these two time points (after twenty or twenty-six weeks of treatment) were more clearly recognizable. No significant increase in the interstitial collagen (fibrosis) was detectable in sections stained by Azan’s trichrom. In sections stained using the immunohistochemical method for cardiac troponin I, degenerating/necrotic myocardiocytes were characterized by loss of positivity to the anti-cTnI antibody (Figure 2e). Immunohistochemistry for the detection of atrial ANP and BNP showed comparable distribution of immunoreactivity for ANP and BNP between control and casopitant-treated animals. In addition, no cleaved caspase–3–positive myocardiocytes were detected in either control or casopitant-treated dogs. The use of these special stainings was particularly useful to corroborate standard H&E: loss of positivity to the anti-cTnI antibody was seen in necrotic cardiomyocytes, confirming the origin of cTnI measured in serum, and the absence of cleaved caspase-3–positive myocardiocytes rejects the hypothesis of apoptotic pathway involvement.
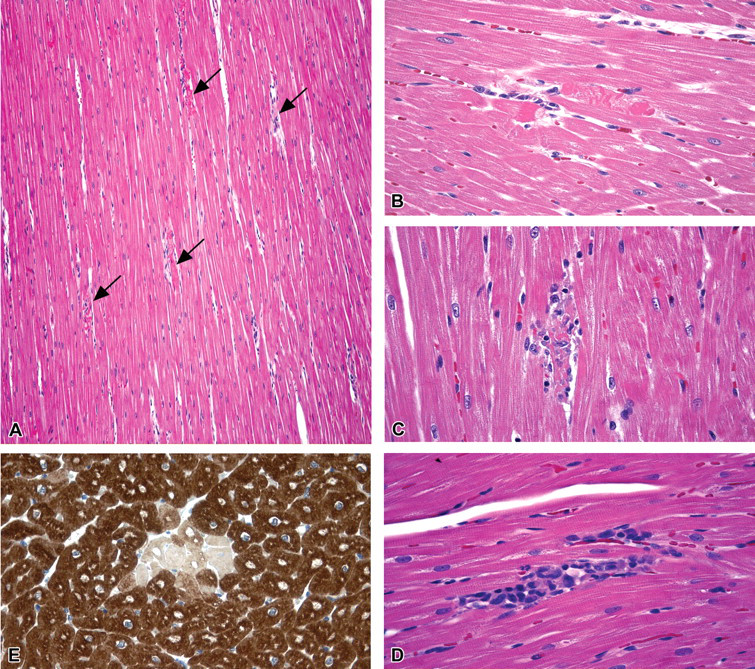
(a) Histopathological changes in casopitant-treated dogs. Papillary muscle (left ventricle) showing moderate degenerative changes multifocally affecting single cardiomyocyte (arrows). H&E; 10× original magnification. High magnification showing degenerative changes in cardiomyocytes, (b) cytoplasmic hypereosinophilic, transversely oriented contraction bands, sign of early damage; (c, d) infiltration and substitution of the necrotic cardiomyocytes, by macrophages and lymphocytes. H&E; 40× original magnification. (e) Immunohistochemical stain for cardiac troponin I (cTnI). Degenerating/necrotic cardiomyocytes characterized by loss of positivity to the anti-cTnI antibody, surrounded by unaffected (normal) cardiomyocytes, showing diffuse cytoplasmic positivity for cTnI (brown stain). Immuhistochemical method with mouse monoclonal, cTnI antibody, Abcam, cat. no. ab19615; 40× magnification.
Test article–related minimal myofiber degeneration/necrosis was seen in one animal given thirteen weeks of treatment and following twenty-two weeks of recovery, characterized by histological/immunohistochemical features similar to those observed in interim/terminal kill dogs.
Cardiac Biomarkers—Troponin I(cTnI), Creatine Kinase MB (CK-MB), and NT-pro Brain Natriuretic Peptide (NT-pro BNP)
Alterations and increasing trends of cardiac necrosis markers are summarized in Figures 3a, 3b, and 4.
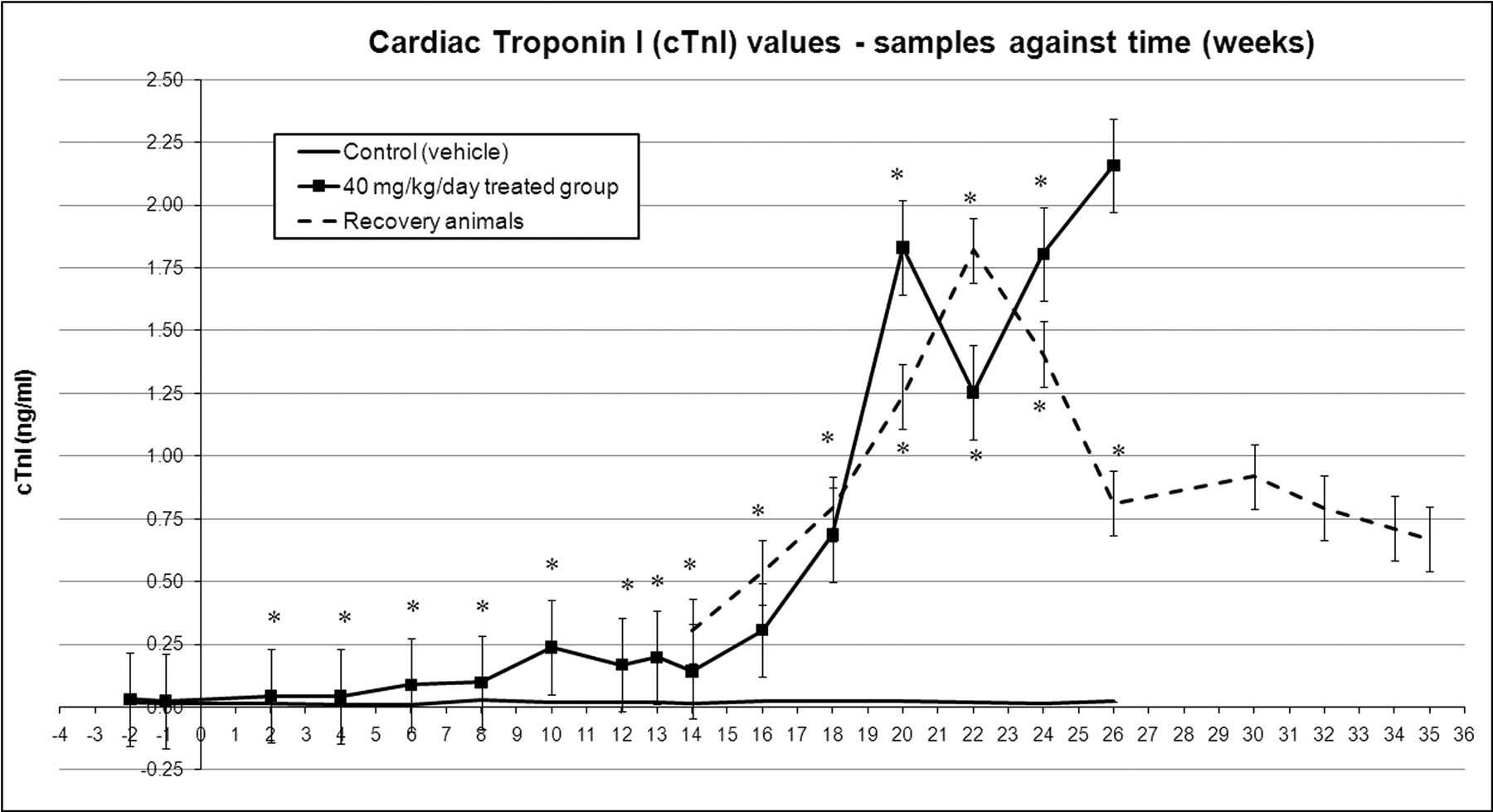
Cardiac troponin I changes across time, mean values with standard error bars. * p < .05 in casopitant-treated animals compared to controls.
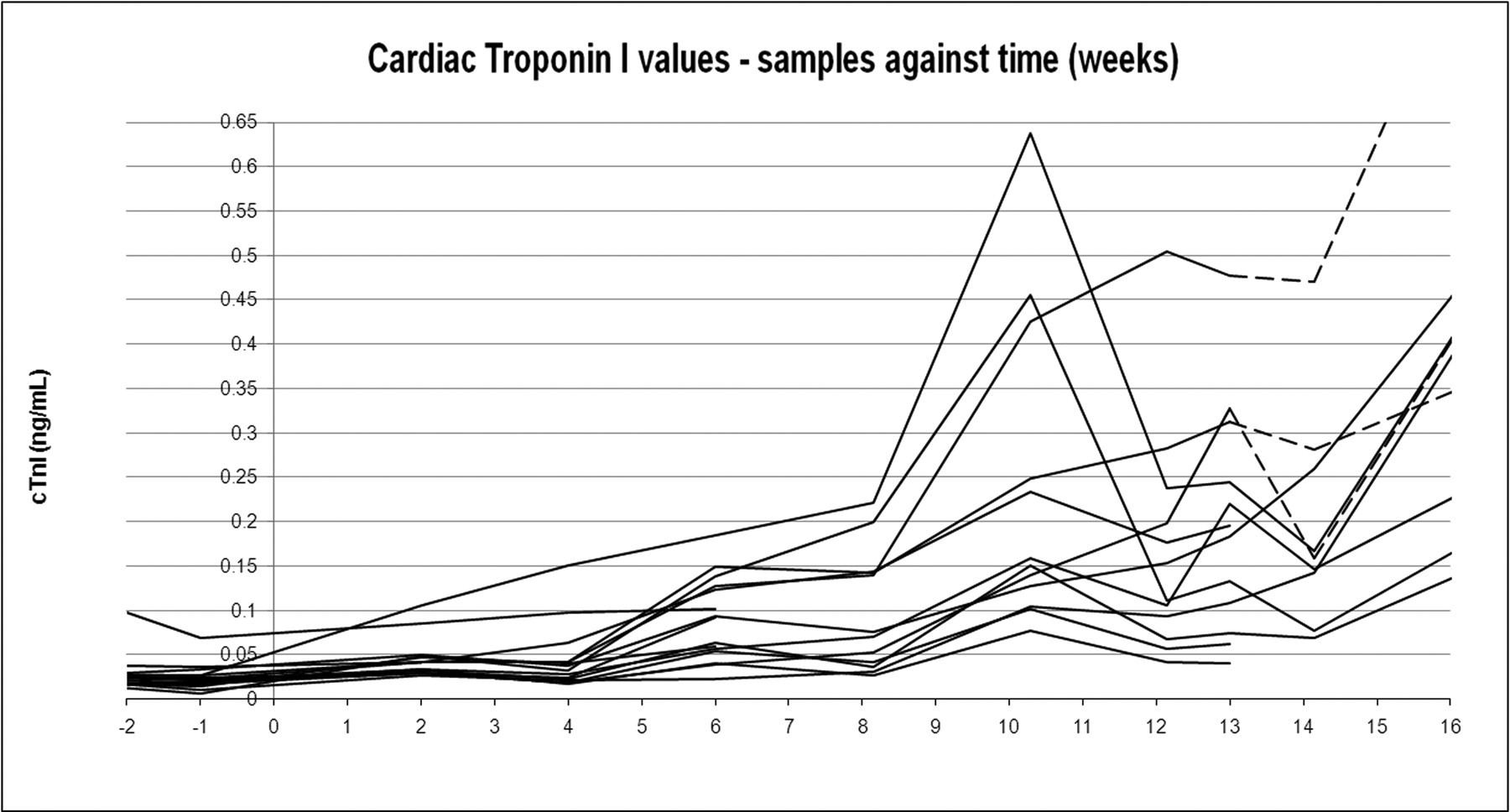
This figure reports the cTnI changes for treated animals only (individual trend) and for a limited period (up to Week 16), to highlight the detail of the early minimal changes reported in Figure 3a. This chart shows the differences of each individual trend in comparison to the corresponding predose periods (negative week numbers). Dotted lines represent the treatment withdrawal (recovery period).
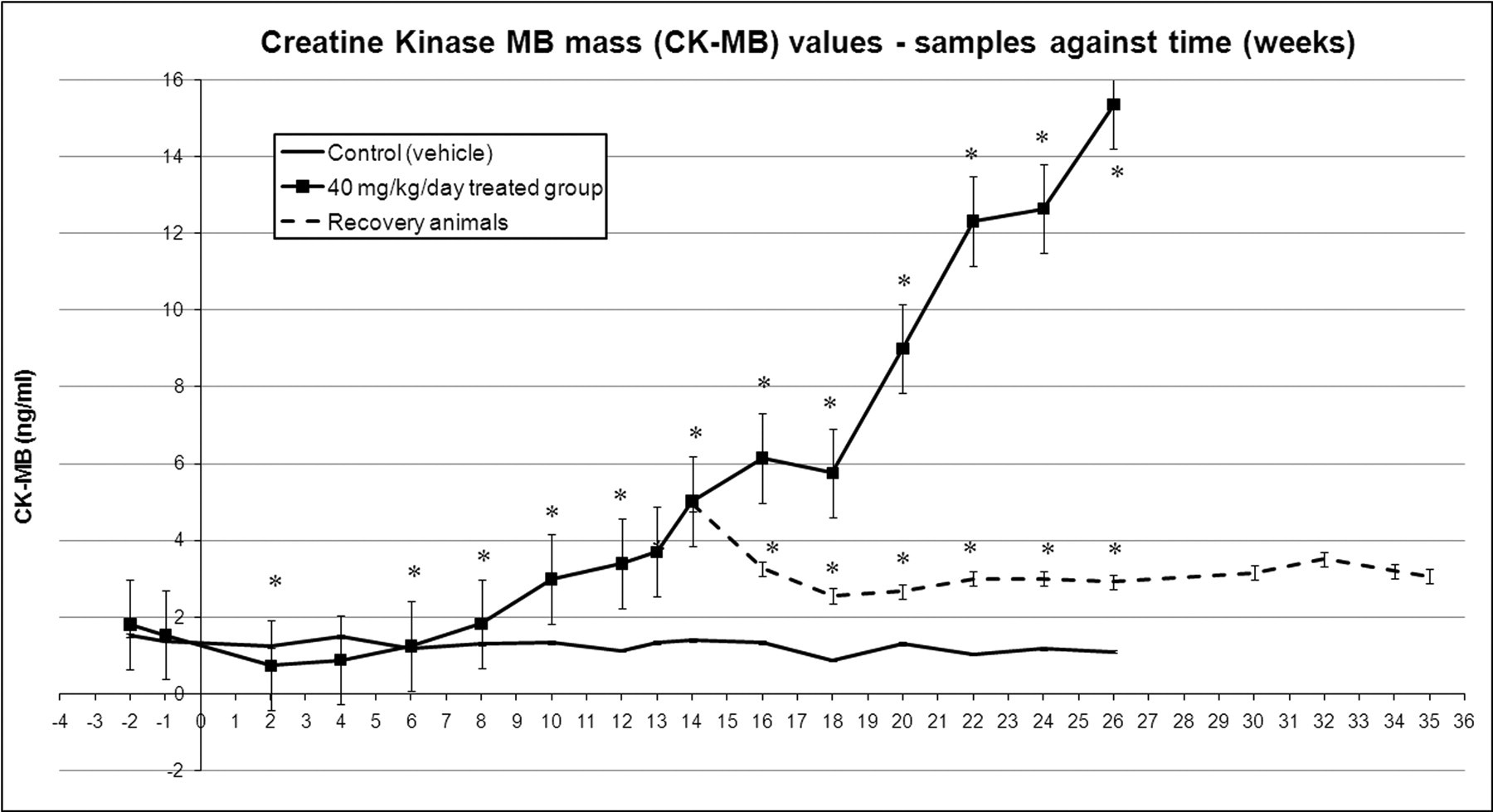
Creatine kinase MB mass changes across time, mean values with standard error bars. * p < .05 in casopitant-treated animals compared to controls.
Starting at Week 6, cardiac troponin I (cTnI) increased 3.5 times (mean value) compared to the predose value in animals treated at 40 mg/kg/day. Between Week 7 and Week 13, mean cTnI values continued the increasing trend in treated dogs, reaching up to ten times the predose value on Day 72 (Week 10); CK-MB started to increase from Week 10, with values up to 2.5 times the predose value. After Week 13, cTnI showed a further increase, reaching values of up to eighty-four times the predose value on Week 20 and up to 100 times predose on Week 26, whereas CK-MB increased up to 6.7 times predose on Week 20 and up to twelve times predose on Week 26. In the recovery animals (twenty-two weeks recovery, following thirteen weeks of treatment), the levels of cTnI showed the same increasing trend, reaching a peak after nine weeks of recovery on Day 155 (Week 22 of the study, sixty-seven times predose from fourteen times at the end of dosing). Following eleven weeks of recovery, from Day 169 (Week 24) of the study, cTnI values started to decrease, although they did not return to baseline (predose) values. CK-MB levels at the end of dosing were 2.4 times predose, whereas values at the end of the recovery period were slightly lower (i.e., 1.8 times predose).
Alterations and increasing trends of NT-pro BNP are summarized in Figure 5 .
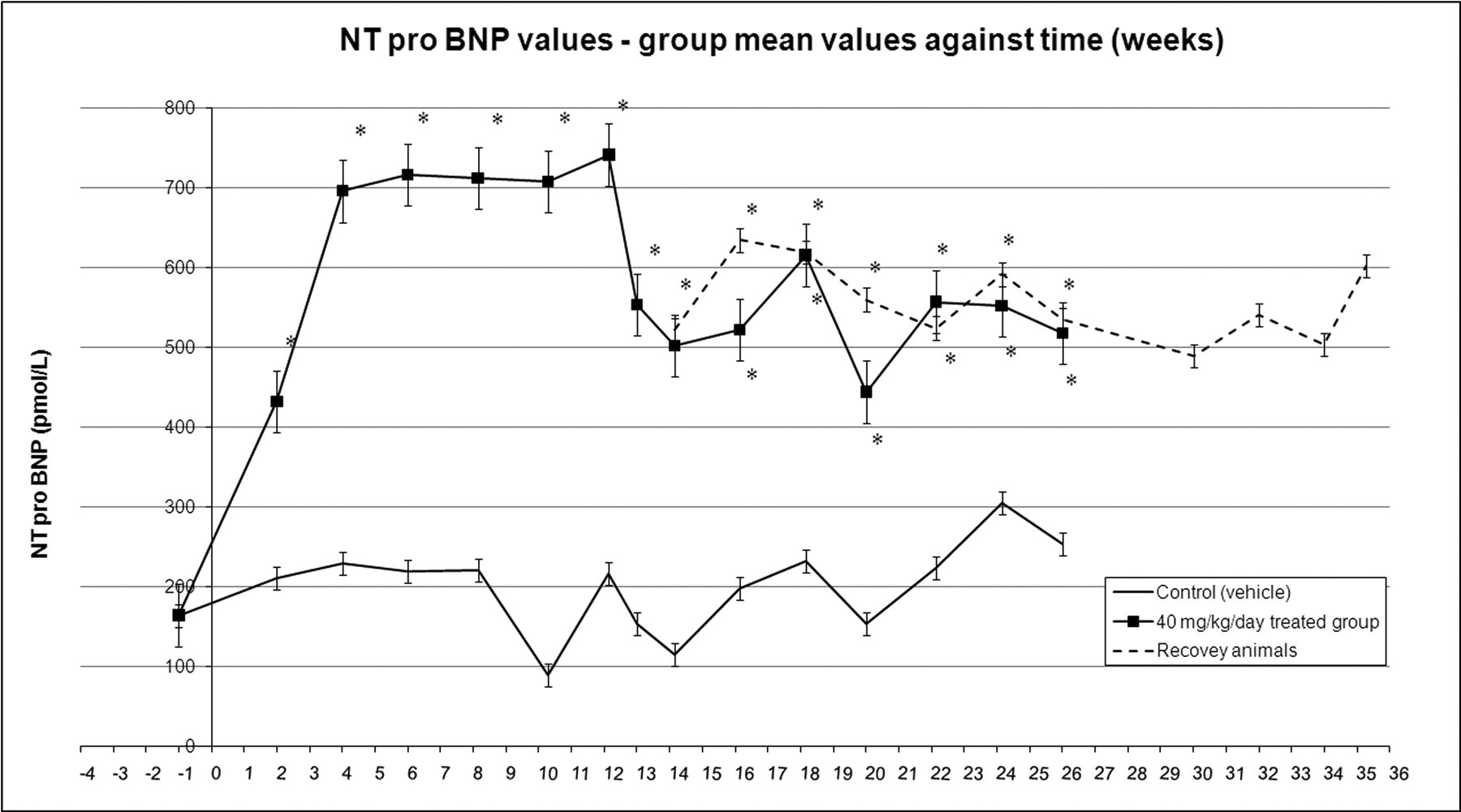
NT-pro BNP changes across time, group mean values with standard error bars. * p < .05 in casopitant-treated animals compared to controls.
For dogs treated at 40 mg/kg/day, the concentration of NT-pro BNP increased immediately from the first time point measured (2.6 times the predose value on Week 2, and up to 4.7 times predose on Week 12), and remained elevated for the duration of the study. Although there was a downward trend in values from Week 13 of treatment (3.5 times predose), NT-pro BNP values did not return to baseline (predose) values.
Compound-induced increases of NT-pro BNP were maintained up to the end of the recovery period (up to 4.2 times predose, from 4.0 times predose as measured at the end of the thirteen-week treatment period).
Transmission Electron Microscopy
Heart sample ultrastructural evaluation showed time course– and test article–related changes in all treated animals starting from Week 6 of the study (Figure 6 ). These changes consisted of intracytoplasmic multilamellar bodies in the sarcoplasm of myofibers and in endothelial or smooth muscle cells of blood vessels. Multilamellar bodies appeared variably arranged with concentric, highly ordered lamellae or disorganized and loosely arranged stacks of lamellae. Ultrastructural changes increased in severity at the subsequent time points, from scattered single intracytoplasmic multilamellar bodies at Week 6 of the study (first interim kill time point) to several accumulations of intracytoplasmic multilamellar bodies at Week 20 (interim kill) or Week 26 (terminal kill) of the study. At Week 26 some necrotic myofibre portions, filled with multilamellar bodies, were also observed. The majority of mitochondria and sarcoplasmic reticulum showed a normal ultrastructural configuration (shape and size).

Ultrastructural changes in the dog myocardium. (a) Normal myocardium. (b) Myocardium after six weeks of treatment showed scattered sarcoplasmic multilamellar bodies. (c) Myocardium after thirteen weeks of treatment showed numerous sarcoplasmic multilamellar bodies. (d) Myocardium after twenty weeks of treatment showed accumulation of sarcoplasmic multilamellar bodies, forming a cluster. (e) Myocardium after twenty-six weeks of treatment showed severe accumulation of sarcoplasmic multilamellar bodies and myofibrillar necrosis. (f) Myocardium after twenty-one weeks of recovery period again showed severe accumulation of sarcoplasmic multilamellar bodies.
Ultrastructural examination performed on animals after the twenty-two-week recovery period (corresponding to Week 35 of the study) did not show clear signs of recovery, with the persistence of intracytoplasmic, multilamellar bodies.
Cardiovascular Functional Evaluations
An increase in mean heart rate (HR) of 54 bpm (ranging from 48 to 65 bpm increase in comparison to predose values) was noted in animals treated with casopitant, starting from Day 39 (Week 6). This effect lasted for a similar magnitude during the dosing period, generally both before and after dosing, but by Day 137 (Week 20), mean HR had reduced to 30 bpm (ranging from 0- to 62-bpm increase in comparison to predose values), and by the end of the recovery period it was no longer apparent. Blood pressure was not evaluated in this study, but a similar (same study design) and concomitant cardiovascular study showed decreases in both systolic and diastolic blood pressure (up to 31 and 22 mmHg at Week 2, respectively) and in the calculated pulse pressure (up to 17 mmHg at Week 4). At later time points, pulse pressure tended to normalize (no alterations from Week 18 onward).
The QT interval increased by 26 msec (ranging from 0- to 40-msec increase in comparison to predose values) in casopitant-treated animals after twenty weeks of treatment and after the twenty-two-week recovery period (control values ranging between 150 and 205 msec). When corrected for HR, the QT interval also increased (41 msec) starting from Day 39 (Week 6), and again lasted throughout the dosing period for a similar magnitude; this increase was noted generally before and after dosing. This effect reduced to 36 msec by Day 137 (Week 20). Toward the end of the recovery phase, Day 219 (Week 19 of recovery), animals previously treated with casopitant still presented slightly higher mean values compared to pretreatment (19 msec), similar to those detected at the end of dosing for these animals (15 msec).
At the echocardiography evaluation, there was an increase in mass in treated animals (observed at Week 12 and Week 20) that was not consistently seen in all treated dogs, compared with controls. Left ventricle mass was the result of an evident increase of LV walls thickening, particularly the posterior wall (1.4 and 1.37 times the pretreatment in diastole and systole, respectively) compared to the anterior wall, even if the thickening could be considered symmetrical. All the other animals treated with casopitant showed lower magnitude increases with no substantial regional difference. Because of the variability of the data, it was not possible to draw any definite conclusions on the recovery of this finding.
Toxicokinetic Evaluations
Parent compound and all metabolites were characterized in both plasma and heart tissue. Results of the cardiac concentrations of parent compound and main metabolites are reported in Figure 7 . Some metabolites (e.g., M200 and M134) seemed to accumulate more than the parent compound in the cardiac tissue, and their concentration rose without reaching the steady state up to 26 weeks of treatment. These tissue concentrations did not mirror the corresponding plasma concentrations, as they had minimal or non-quantifiable concentrations in plasma (data not shown). Furthermore, M200 and M134 were still present in heart tissue after the recovery period, confirming their very slow clearance and elimination.
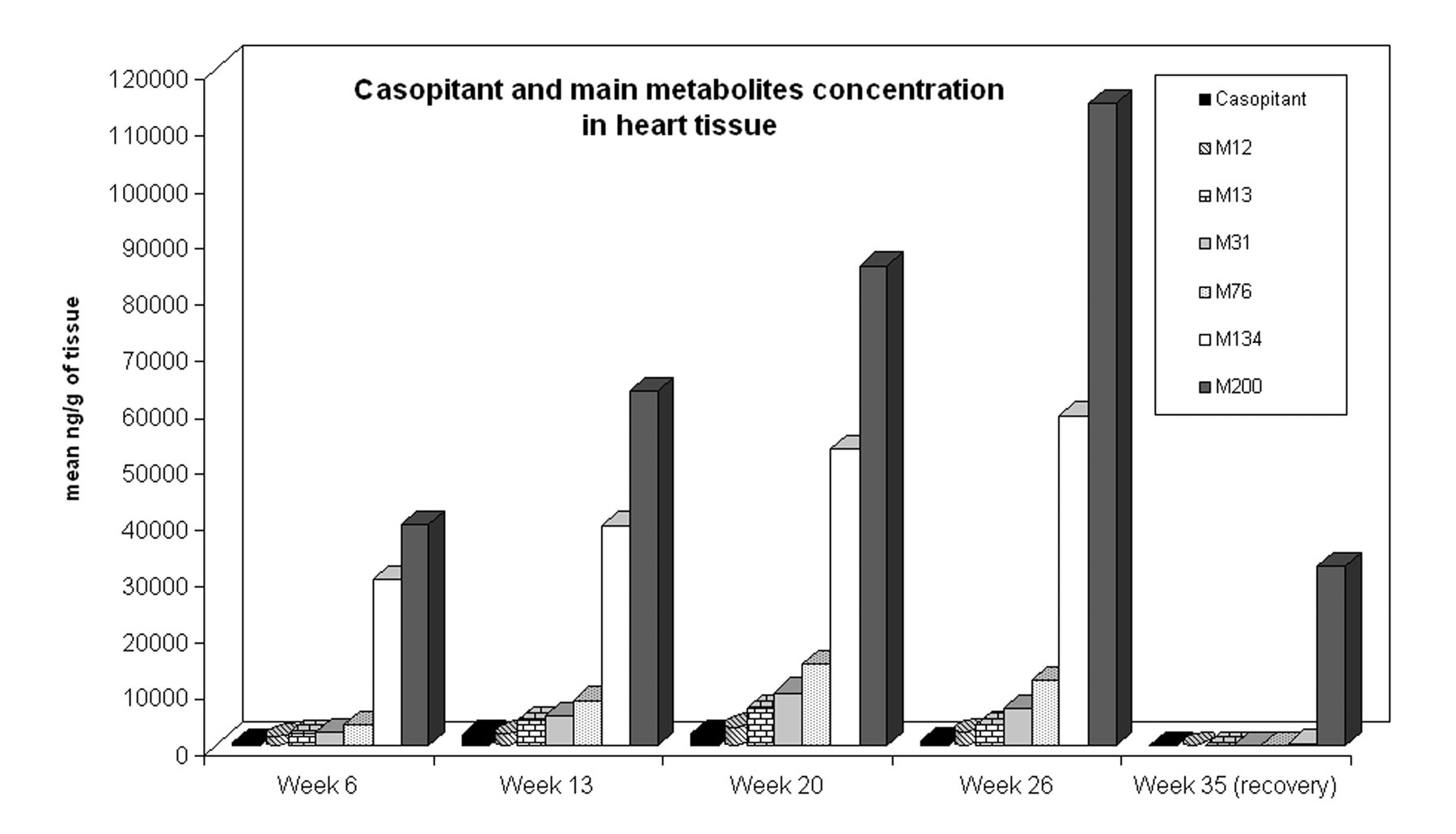
Mean concentration of casopitant and main metabolites (M) in the dog heart, measured as ng per gram of wet tissue.
Discussion
Cardiovascular toxicity is a key issue in the preclinical development of new drugs, and it requires early hazard identification and an accurate follow-up during preclinical drug development. Risk factors associated with pharmacological activity are well known to increase cardiovascular liability risk; however, other aspects such as physical-chemical properties, which may lead to the accumulation of a drug (or its metabolites) in specific target tissues, can increase the risk of unexpected chronic organ toxicity. In this context, cationic amphiphilic drugs are known to potentially accumulate in tissues (Vonderfecht et al. 2004), with morphological end points indicative of phospholipidosis. Target organs of accumulation may vary, as phospholipidosis can have a broad spectrum of different manifestations and severities in different organs, depending on the specific physical chemical properties of the drug and its interaction with the affected organ (Kacew et al. 1997; Lüllmann-Rauch 1979; Schneider 1992). Examples of cationic amphiphilic drugs that cause target organ toxicity are available in the literature (Drenckhahn and Lüllmann-Rauch 1979), and evidences of phospholipidosis in many tissues are often associated, even if the heart is reported to be only rarely affected (Halliwell 1997; Jagel and Lüllmann-Rauch 1984).
In this article, the example of casopitant, an NK1 receptor antagonist developed for depression and anxiety, is reported. The physical-chemical properties of this compound lead to its accumulation in tissues (Miraglia et al., 2010; Pagliarusco et al., unpublished data) as well as to phospholipidosis-related changes, as demonstrated in previous studies. Cardiovascular toxicity became evident only after long-term administration in dogs, and an integrated assessment of the onset and progression of the finding was needed to provide insights for risk assessment and to support decision making.
An oral investigative repeat-dose study in dogs, for up to twenty-six weeks with interim kills and a recovery period of twenty-two weeks was designed to characterize the cardiac changes by using a broad range of investigative tools. This article presents how the integrated approach of different disciplines and the applications of multiple integrated tools can constitute a powerful approach for the characterization of heart toxicity.
In the investigative dog study presented in this article, cardiac cTnI demonstrated to be the earliest and most powerful biomarker of damage to monitor the onset and progression of cardiac changes. Troponin I showed changes starting from six weeks of treatment, although minimal in magnitude and particularly evident for individual animals. Even if above our cutoff values in dogs, these early cTnI increases should not be evaluated on the basis of the statistic calculations alone, but rather, they assume a prognostic value when correlated with other evidence of cardiac changes. Electron microscopy findings and metabolite tissue accumulation together suggested progressive cardiomyocyte damage, potentially owing to metabolite accumulation, and in this scenario, serum cTnI evaluation provided an early and noninvasive evidence of isolated cardiac cell necrosis. Evaluation of cTnI and TEM showed high sensitivity in comparison to histopathological evaluations: cardiac lesions became apparent with light microscopy only following extensive sampling of all chambers of the heart after thirteen weeks of treatment in one animal only, and they were clearly present in all dogs at the twenty-week time point. A correlation between cTnI values and light microscopy evaluations was done: the latter was able to show relevant cardiac lesions (H&E staining) only when cTnI values reached approximately 1 ng/mL, corresponding to a very high value in comparison to the baseline cutoff calculated for our dog colony (about 0.07 ng/mL). The power of cTnI in terms of sensitivity and specificity was confirmed in this study, as well as in recent literature (Oyama and Solter 2004), making it an important marker for an accurate preclinical heart safety evaluation; on the other hand, CK-MB showed a lower sensitivity than cTnI, with later increases, starting from Week 10.
The most informative picture on cardiac changes was obtained by integrating cTnI alterations with TEM findings. Transmission electron microscopy examination revealed changes in cardiac cells with multilamellar bodies in sarcoplasm at the same time points at which cTnI showed the initial increases (six weeks of treatment). In this study, the application of this technique had an added value in identifying and characterizing the onset of the lesion. Similar to cTnI, TEM findings increased in severity in the different time points considered, suggesting a progressive general impairment and perturbation of the cardiomyocites. At Week 26, some necrotic myofibers, filled with multilamellar bodies, were also observed, in line with the light microscopy observations.
Heart weights were also affected early, starting from Week 6. Although this is a relatively nonspecific parameter and changes were minimal at this early time point, it can also be put into context by comparison with the accompanying morphological and functional parameters: at Week 6, treated dog hearts showed increased LV mass and wall thickening at the echocardiography evaluation, as well as increased HR and prolongation of the QTc interval (unpublished data). Taken alone, the nature and modest magnitude of the observed changes in HR and blood pressure (decreases of the latter observed in a separated cardiovascular study) were considered of insufficient severity to be responsible for the anatomical changes described above, but the prolongation of the QTc interval may reflect the cardiac remodeling likely to occur in the development of these changes (Kang, 2006). The LV mass increases noted at echocardiography were consistent with the increase in heart weight and with the elevations seen in NT-pro BNP levels, a biomarker for the onset and evolution of cardiac hypertrophy (Crivellente et al., unpublished data; Goetze et al. 2006; Panou et al. 2006). However, NT-pro BNP started to increase after two weeks of treatment, preceding most, if not all, the anatomical and functional changes described above. Furthermore, al cardiovascular observations above (LV mass increases, QTc interval, and HR) showed a trend toward recovery after treatment withdrawal, with HR completely recovered after fourteen weeks, whereas NT-pro BNP did not return to normal values twenty-two weeks after treatment withdrawal. This study showed NT-pro BNP alterations even in the absence of any change in related functional or morphological parameters; this finding strongly supports the sensitivity of NT-pro BNP, as it behaved as an early prognostic biomarker of functional cardiac impairment.
All of the above findings, correlated with the toxicokinetic results, confirm that the physical-chemical properties allowed the compound or a metabolite to penetrate and accumulate in the heart, as well as in other tissues. The cationic amphiphilic nature of the chemical increased the affinity of this compound and/or metabolites for the cellular membranous compartments, possibly leading to a perturbation of cytoplasmic organelles such as mitochondria (Kacew et al. 1997). Furthermore, chronic treatment increased drug/metabolite accumulation as well as cell pathology and caused progressive cell death, also confirmed by cTnI elevations. It is important to consider that no ultrastructural changes, other than the presence of intrasarcoplasmic multilamellar bodies, were observed at TEM evaluation. Specifically, no typical features of cardiotoxicity were observed; the hearts of these animals did not show myofilaments and sarcoplasmic reticulum degeneration (Cheville 1994), myocytolysis and detachments from the insertions on the Z line and A band forming aggregate masses in the sarcoplasm (Van Vleet et al. 1991), or lysosomes involved in autophagy of degrading senescent cellular organelles, namely, mitochondria, lipid globules, and myofilament components (Cheville 1994).
The progression of the cardiac changes seen in-life, and the continued presence of heart changes during and after the recovery period, correlated with the cardiac concentration of certain metabolites (e.g., M200; Figure 7). The cardiac concentrations of these metabolites showed no correlation with plasma concentrations, indicating a different kinetic for accumulation in the cardiac tissue than in plasma. These data suggested that plasma exposures to casopitant and its metabolites do not provide reliable monitoring tools for defining safety margins. Furthermore, the concentrations of some metabolites found in the heart matched the cardiac toxicity in terms of severity of the electron microscopy and plasma cTnI and NT pro BNP findings, suggesting the potential implication of one or more metabolites in causing cardiac toxicity. In this scenario, the measurement of cumulative exposure may be more appropriate to determine a safety margin for human use. Little information on the toxic potential of these metabolites is currently available, but some insights on their possible mechanism of action can be considered, including the chemical structure of some metabolites that are very concentrated in the heart (e.g. M200) that contain an ethylenediamine side chain derived mostly from the cleavage of a piperazine group of the parent compound. These structures have been described in the literature to potentially accumulate in tissues (Breyer and Gaertner 1974) as well as to cause cardiac toxicity in some cases. Further analyses of some of these active metabolites revealed a potential calcium antagonist activity (demonstrated for M31), and a calcium and iron chelating ability was also hypothesized. This finding suggested a possible role of casopitant metabolites in calcium homeostasis or in perturbation of mitochondria functionality; in fact, the impairment of calcium homeostasis can also be correlated with the mucosal erosions and red gums observed in the dogs, starting from the third week of treatment: this is a common finding observed, for example, after treatment with calcium channel blockers (Missouris et al. 2000). Even if the mechanism of toxicity was not fully elucidated and demonstrated, taken together, all these data suggest that drug-related material potentially able to induce toxicity was actually present in the heart at concentrations higher than expected from the plasma toxicokinetic data, and that is most likely linked to the observed late-onset toxicity.
This article shows how the integration of different conventional and investigative tools increased the prognostic power for the assessment of cardiac toxicity during the preclinical development of a new drug. The integration of different observations and disciplines, as well as the inclusion of nonstandard investigative measurements, provided a wider view of the treatment effects in the dog heart, providing a picture of the very beginning of treatment-related cardiac changes and accurately characterizing the onset and progression of the lesion. In this specific case, this investigative study highlighted the limitations in terms of sensitivity of the original standard regulatory toxicity studies, suggesting that the increased use of nonconventional tools would better characterize potential new drugs.
Footnotes
Acknowledgments
We would like to express particular thanks to all the pathology and in vivo support technical staff at GlaxoSmithKline Verona, who actively contributed to the successful realization of this complex study.
