Abstract
Noncommunicable diseases, including cardiovascular disease, diabetes, chronic respiratory disease, and cancer, are the leading cause of death in the world. The cost, both monetary and time, of developing therapies to prevent, treat, or manage these diseases has become unsustainable. A contributing factor is inefficient and ineffective preclinical research, in which the animal models utilized do not replicate the complex physiology that influences disease. An ideal preclinical animal model is one that responds similarly to intrinsic and extrinsic influences, providing high translatability and concordance of preclinical findings to humans. The overwhelming genetic, anatomical, physiological, and pathophysiological similarities to humans make miniature swine an ideal model for preclinical studies of human disease. Additionally, recent development of precision gene-editing tools for creation of novel genetic swine models allows the modeling of highly complex pathophysiology and comorbidities. As such, the utilization of swine models in early research allows for the evaluation of novel drug and technology efficacy while encouraging redesign and refinement before committing to clinical testing. This review highlights the appropriateness of the miniature swine for modeling complex physiologic systems, presenting it as a highly translational preclinical platform to validate efficacy and safety of therapies and devices.
Keywords
Introduction
Noncommunicable diseases (NCDs, chronic diseases) cause 7 of every 10 deaths worldwide (Centers for Disease Control and Prevention 2013; Go et al. 2014), representing 68% of all annual deaths (56 million deaths). The cumulative economic loss during 2011–2015 is estimated at US$7 trillion (World Health Organization 2014) and accounts for 86% of health care costs (Go et al. 2014). Juxtaposed to this is the increasingly unsustainable and economically inefficient process of therapy and device development targeted at ameliorating these diseases. Currently, excluding government contribution (DiMasi, Grabowski, and Hansen 2015), the private sector invests between US$1.8 and US$2.6 billion (Morgan et al. 2011; Avorn 2015) for drug and between US$75 and US$94 million for medical device (Makower, Meer, and Denend 2010) development. Drug and device development require approximately 13 (Morgan et al. 2011) and 4.5 years (Makower, Meer, and Denend 2010), respectively, from inception to approval. This high investment cost is in part due to the stringent regulatory requirements (U.S. Department of Health and Human Services 2009, 2010, 2011) stipulated by agencies for approval. However, the lack of a clear understanding of how to appropriately meet those requirements in an efficient and cost-effective manner is a major hurdle. Despite time, cost, and effort, new therapies experience a high failure (90–95%) rate during the process (Khanna 2012), primarily in the course of clinical development phases II and III (Arrowsmith 2011a, 2011b). These obstacles have led investors to search for medical areas of less regulatory uncertainty where the investment return may be potentially expanded and more lucrative. These factors subsequently translate directly to a reduction in innovation (Kaitin and DiMasi 2011). The potential failure rates can be mitigated if, in preclinical stages, efficacy and safety can be validated in an environment providing a highly translational platform to mimic the human population. This would require the use of a relevant animal model that closely models human disease not just during the preclinical phase but as early as possible in the target discovery phase.
Complexity of Disease and the Importance of a Translational Animal Model
NCDs, which include cardiovascular disease, diabetes, chronic respiratory disease, mental illness, and cancer, are complex in etiology and progression and are associated with comorbidities (Marrero, Bloom, and Adashi 2012). Overarchingly, human health and disease are influenced by complex and still largely enigmatic interactions between genetics/epigenetics, environment, and the microbiome (Figure 1). Interactions between these factors affect not only (a) causality of disease but also (b) presentation of symptoms and biomarkers, (c) course of disease progression, (d) development of comorbidities, and (e) efficacy and safety of preventative and therapeutic strategies. As these interactions vary from individual to individual and population to population, so do the effect they have. Such complexity poses seemingly insurmountable challenges in the prevention and treatment of these diseases. Animal models are intended to provide an anatomical and physiological platform that mimics the complex physiological responses that exist in a disease process. Animal models may be divided into 3 categories according to their ability to reproduce human disease:
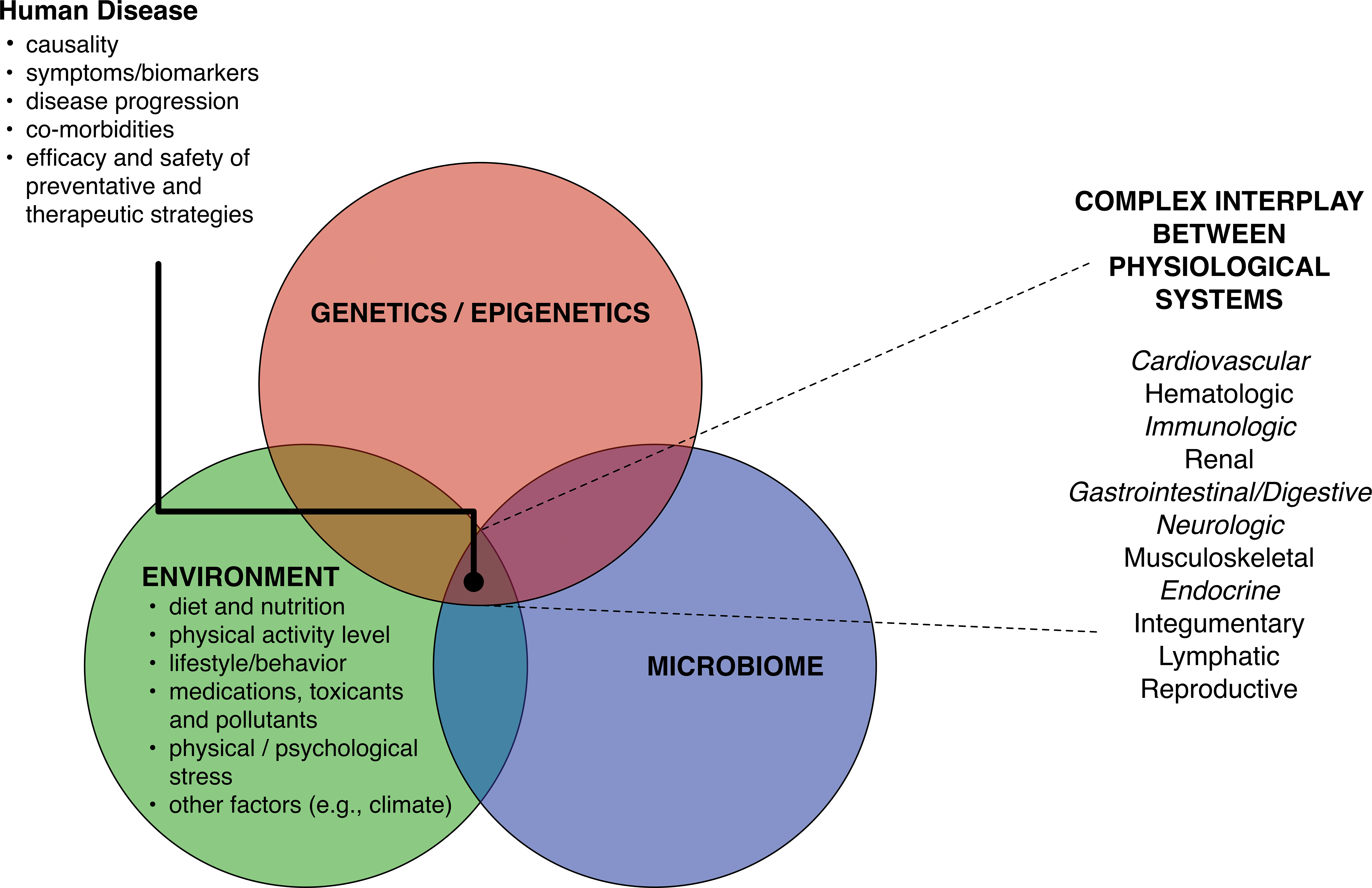
Intrinsic and extrinsic factors influence complex interplay of physiological systems in human disease. The
Miniature Swine
Beyond the conventional swine breeds’ genetic, anatomic, and physiologic similarities to humans (Wernersson et al. 2005; Schook et al. 2005; Schwartz et al. 2008), mini-swine also have the additional advantages of human size and slower growth curves. At full maturity, conventional breeds can reach 249–306 kg (550–675 lb) and pose challenges in husbandry and handling in the biomedical research setting. In contrast, at equivalent maturity, mini-swine weigh 68–91 kg (150–200 lb), the weight of an average human male (Swindle et al. 1994). Currently, the most widely used mini-swine breeds in U.S. research laboratories and contract research organizations include the Hanford™ (Ho et al. 2010), Sinclair™ (Misfeldt and Grimm 1994), Yucatan™ (Panepinto and Phillips 1986), and Göttingen (Simianer and Kohn 2010). Other mini-swine breeds such as Tibetan (Fan et al. 2003), Banna (Sun et al. 2004), Ossabaw (Hamamdzic and Wilensky 2013), and the Rapacz familial hypercholesterolemic (RFH; Granada et al. 2009) swine are used for specific advantages they possess in a given area of study. While other mini-swine breeds, such as the Wisconsin Miniature Swine™ at University of Wisconsin–Madison, continue to be developed and refined for specific characteristic advantages, there have been efforts to further miniaturize the mini-swine. The microminipig (MMPig) developed in Japan (Miura et al. 2014; Kaneko et al. 2011), weighing around 7 to 10 kg at maturity, is thought to be ideal for toxicological (Guruge et al. 2016) and diet-induced atherosclerosis (Miyoshi et al. 2010) studies. While the MMPig loses the advantage of human size offered by mini-swine breeds, particularly in medical device research, it enables toxicological studies without requiring large quantities of novel drugs.
The human size and the friendly temperament of mini-swine offer advantages in husbandry and handling; the animals can be easily trained to tolerate minor procedures without sedation (e.g., blood draws and noninvasive imaging) and perform exercise regimens (Simmons et al. 2014). Their size also allows for the use of medical equipment and procedures that are used in human clinical settings, allowing researchers to employ state-of-the-art medical innovations in research (Wentland et al. 2015; Ge et al. 2012; Johnson et al. 2014), while giving direct translatability of experimental knowledge gained to humans. The size of mini-swine, when compared to small animal models, allows for repeated and larger volumes of sample collection and gives the statistical advantage of longitudinal study design. Swine are omnivores with similar nutritional requirements to humans and can be fed human diets (e.g., fast foods) with desired meal patterns. This is particularly valuable in studying the effects of human diets and nutrition on disease processes and drug metabolism, efficacy, and safety. The reproductive characteristics of swine, including early sexual maturity, large litter size, and short generation intervals (12 months; Swindle 1998), when combined with the availability of sophisticated gene-editing tools, allow for creation of novel genetic models and the exploration of transgenerational effects of epigenetics in a manner that is impractical in nonhuman primates (NHPs). Additionally, research in swine, because of the wide use of the animal for food, does not raise the ethical concerns faced by NHP research. In fact, the recent advances in the use of swine in neuroscience and cognition fields, until now considered the key domain of NHP (Weatherall 2006), have begun to invigorate the preference of swine over NHP. With commercial vendors providing a wide range of swine-specific products, including cell lines, antibodies, and assay kits, and with annotated swine-specific microarrays for gene expression studies (Zhao et al. 2005; Tsai et al. 2006), tools available for swine research are no different than those for more widely used animal species.
Genetics and Epigenetics
Genetics influence nearly every aspect of human biology and disease. Although other factors can dramatically affect how genetics presents phenotypically, a deep understanding of the similarities and differences between the genome of humans and swine is of importance when creating relevant swine models. The Swine Genome Sequencing Consortium first sequenced the genome of
Epigenetic modifications have emerged as a key mechanism by which extrinsic factors can permanently influence an organism’s cellular function and contribute to the pathophysiology of disease processes (Portela and Esteller 2010; Fernandez-Twinn, Constancia, and Ozanne 2015; Stegemann and Buchner 2015). The epigenome is especially sensitive to modification during fetal and neonatal developmental periods (Rosenquist et al. 2015). Evidence suggests that these programmed effects can be inherited and thus affect multiple generations (Fernandez-Twinn, Constancia, and Ozanne 2015; Stegemann and Buchner 2015). The epigenetic processes that affect genome function are now recognized to also include the regulatory effects of noncoding RNAs, including micro-RNAs (Tu et al. 2015) and long noncoding RNAs (Hung and Chang 2010); however, the understanding of how these compare between humans and swine is in its infancy (Paczynska, Grzemski, and Szydlowski 2015; Zhou et al. 2014b). The combination of similarities in genetics and physiology between swine and humans implies swine are an excellent model to study the influence of external factors such as nutrition, smoking, alcohol consumption, environmental toxicants, and stress on epigenetics. Global analysis of methylation in multiple tissues has identified differentially methylated regions (Yang et al. 2011) associated with differences in expression of genes involved with lipid metabolism and immune regulation (Li et al. 2012). A comprehensive survey of imprinted genes in swine revealed that, in general, imprinting patterns seen in swine were similar to humans but dissimilar to mice (Bischoff et al. 2009). The mouse has been used extensively to understand epigenetic principles but may be limited in its potential to elucidate specific epigenetic contributions for human diseases (Stoger 2008), such as obesity (Cheverud et al. 2008) or Albright’s hereditary osteodystrophy (Germain-Lee 2006). Contrastingly, recent work by Multhaup and colleagues suggest that epigenetic analysis in mice provides valuable information for understanding human obesity and diabetes (2015). Until our understanding of epigenetics and its role in disease evolves further, it will be difficult to translate epigenetic findings in mice to humans (Rosenfeld 2010). Certainly, before we can fully ascertain that the swine is a relevant model for exploring the role of epigenetics in human disease and for investigating therapeutic epigenetic manipulations, a comprehensive mapping of the swine epigenome and the development of relevant tools for manipulation must occur.
Environment
Environmental influences on health and disease are often difficult to directly address in research. This is, in part, due to challenges in manipulating certain environmental parameters, the subtlety of the influence, or a lack of mechanistic understanding of effects. These influences include (a) diet and nutrition; (b) physical activity level; (c) lifestyle/behavior; (d) medications, toxicants, and pollutants; (e) physical and psychological stresses; and (f) other factors (e.g., climate; Figure 1). Because environmental influences are complex and variable in their effects between individuals and species, it is crucial that accurate models are used. For example, a mouse exposed to crowded conditions may not exhibit the same stress response and subsequent pathosis as a canine exposed to similar conditions. Fortunately, the swine responds to many human environmental factors in a similar fashion. Swine can be fed the same diet as humans (Miller and Ullrey 1987), trained to perform exercise regimens (Laughlin, Overholser, and Bhatte 1989), raised in crowded or isolated social conditions (Keeling and Gonyou 2001), and/or subjected to specific stressors (Moberg and Mench 2000). Simple changes in environmental enrichment alone have been shown to modulate cognitive and endocrine systems in swine (de Jong et al. 2000), reflecting an adherence to the principle that has been well established in human populations (van Praag, Kempermann, and Gage 2000).
Microbiome
The microbiome is a dynamic symbiotic system that influences and is influenced by many factors including gastrointestinal–digestive physiology, diet, and the host immune system (Tremaroli and Backhed 2012; Kau et al. 2011; Ley et al. 2008; Purchiaroni et al. 2013). Recent technological advances (Bassis, Young, and Schmidt 2013) and collective efforts in characterizing the microbiome have led to the recognition of the central influence of the microbiome–host interactions in health and disease (Shreiner, Kao, and Young 2015; Cho and Blaser 2012). For example, both piglets and children require exposure to microbiota and a spectrum of pathogens to decrease their risk of developing allergies (Inoue et al. 2005). The microbiome can rightly be thought of as a primary physiological system that contributes to overall homeostasis. This underscores the importance of having similarities in microbiome between animal models and humans.
The microbiomes of swine and humans are highly similar (Heinritz, Mosenthin, and Weiss 2013). Their microbiome primarily consists of
Physiological Systems of Relevance to Most Prevalent NCDs
Genetics, environment, and the microbiome exert their influence through the modulation of complex interactions between multiple physiological processes (Figure 1). An ideal preclinical model has similarities to humans in most, if not all, of these processes and thus responds similarly to these 3 influences. Swine physiology resembles humans in multiple ways; particularly with respect to cardiovascular, pulmonary, immune, digestive, endocrine, and nervous systems (Swindle 2012). In this review, we focus on physiological systems that more directly influence the most prevalent NCDs.
Cardiovascular System
Cardiovascular disease is the leading cause of death in the United States, with an economic burden that is projected to triple by the year 2030 (Heidenreich et al. 2011). Relevant models are essential for effectively studying this NCD. Fortuitously, the swine heart is remarkably congruent to that of humans in anatomy and physiology (Bajorat et al. 2006; Crick et al. 1998; Hughes 1986) and has been a developmental platform for numerous diagnostic and interventional technologies (Hicks et al. 2009). The platform has led to the development of noninvasive clinical methods, including ones for evaluating electrophysiology (Smith et al. 1997), platelet physiology (Llanes et al. 2001), thrombus formation (Martini et al. 2008), and blood flow (Lee et al. 2011). These methods have further enhanced studies in which swine have been used to model a host of cardiovascular diseases, including atherosclerosis (Granada et al. 2009), arrhythmias (Park et al. 2015; Janse, Opthof, and Kleber 1998), acute (Feng et al. 1998) and chronic (White, Roth, and Bloor 1986) myocardial infarction (Dib et al. 2006; Ye et al. 2014), venous occlusion (Shi et al. 2015), hypertrophic cardiomyopathy (Lin et al. 2002), endocarditis (Christiansen et al. 2013), and valvular disease (Sider, Blaser, and Simmons 2011; Porras et al. 2016). The availability of these relevant models has facilitated the advancement of novel therapies for clinical evaluation. For example, a swine model of congestive heart failure treated with myoblast transplantation has directly enabled the progression of the therapy toward clinical trials (Opie and Dib 2006). A swine model also helped reveal the potential of mesenchymal stem cells for enhancing nerve regeneration and promoting cardiac remodeling and neovascularization (Pak et al. 2003). Other facilitated advancements include tissue engineering of vessels (Solan and Niklason 2005) and interventions for vessel occlusions (Jahan 2010).
Atherosclerosis is the most common underlying pathological process of cardiovascular disease (Heidenreich et al. 2011). The RFH swine (Rapacz et al. 1994; Hasler-Rapacz et al. 1998; Prescott et al. 1991), a model of atherosclerosis developed and miniaturized at the University of Wisconsin–Madison, produces plaques spontaneously as a function of age in a manner remarkably similar to the human disease in time course, location, and phenotypic complexity, making it a platform for the development of cardiovascular imaging and interventional devices (Granada et al. 2011; Tellez et al. 2010b). Mature RFH mini-swine (Thim 2010), like humans (Finn et al. 2010; Hansson and Libby 2006), display a large stenotic atherosclerotic burden with thin fibrous cap, a large necrotic core, presence of inflammatory infiltrates led by macrophages and foam cells, and intraplaque hemorrhages accompanied by adventitial and intraplaque neovascularization. As in humans, the arteries also contain multiple lesions at varying stages of plaque maturity. Beyond the study of plaque morphology and composition, the swine model has been valuable for studying molecular mechanisms of plaque development with direct translatability to humans (Jiang et al. 2014; Johnson et al. 2014; Bahls et al. 2011; Bahls et al. 2013). Furthermore, changes in elastophysiology (Ge et al. 2012) and blood flow dynamics (Wentland et al. 2015) that occur as a consequence of the disease can also be modeled in a manner that is observed in patients. The anatomical similarities to humans have allowed for the use of clinically available intravascular imaging technologies (e.g., intravascular ultrasound and optical coherence tomography) for
Pulmonary System
Respiratory diseases are one of the most common NCDs and the economic burden is great (Fendrick et al. 2003). Similarities in anatomy, physiology, and neonatal development (Winkler and Cheville 1985) of the lungs and airways between swine and humans have enabled studies of surfactant function, reperfusion injury, pulmonary hypertension (Brandler et al. 2005), and asthma (Turner et al. 2002; Thomas et al. 2011). As the majority of medical equipment and procedures used in human patients can be employed in mini-swine, preclinical studies of the pulmonary system are easily facilitated and can be conducted in a manner translational to clinical practice. Swine are easily anesthetized, ventilated, and managed for extended periods of time (Swindle 1998) using methods and protocols established in human medicine, allowing for studies relevant to acute and chronic management of patients in the health-care setting.
Infection and inflammatory processes exacerbate and drive the progression of many chronic pulmonary diseases (Parameswaran and Murphy 2007; Sethi and Murphy 2008), including chronic obstructive pulmonary disease and cystic fibrosis (CF). The first line of defense against lung infection involves responses from polymorphonuclear neutrophils, which when dysfunctional, can cause significant cell damage and further pathologies. Swine have been used to model this early local inflammatory response (Van Reeth et al. 1999), investigate potential neutrophil inhibitors (Brea et al. 2012), and model key aspects of cytokine activity in the lungs (Van Reeth et al. 1999). As the symptoms of CF are driven by inflammatory mechanisms mentioned above, swine models displaying relevant phenotypes of CF, such as gastrointestinal manifestations (Stoltz et al. 2010), are used to study the pathophysiology and treatments of the disease (Rogers et al. 2008). In contrast, genotypic mouse models of CF do not carry relevant phenotypes. Swine are also used to study acute lung injury (Okazaki et al. 2014), a life-threatening hypoxemic condition especially for neonates that is typically associated with reduced lung compliance, edema, increased surface tension, and inflammatory cascades. Swine studies of treatments such as sphingomyelinase inhibitors significantly improve lung function in terms of oxygenation, ventilation, compliance, and edema through mechanisms that are similar in humans (Preuss et al. 2012). In end-stage pulmonary disease, lung transplantation is often the only remaining management strategy despite the limited availability of human lungs for transplant. Swine experiments have helped elucidate the mechanisms of hyperacute rejection in xenotransplantation (Wiebe et al. 2000; Takahashi et al. 2002). This knowledge, along with current advancements in genetic engineering of swine, offers promise for making lung xenotransplantation a reality in the near future through industry-driven biotechnology initiatives (Harris et al. 2014).
Immune System
The immune system plays a key role in the pathophysiology of human disease and interacts with most other physiological systems. The immunome, comprising all genes and proteins involved with the immune system, is a regulatory network that serves as the decision-making circuit, and, as such, it must be genomically and functionally similar between humans and a preclinical model if inferences are to be made (Amit et al. 2009). While the immune genome of swine or mouse is ∼84% similar to that of humans, the number of orthologous gene families between swine and humans is 10-fold greater than between mouse and humans (Dawson et al. 2013). The differences in both innate and adaptive immunity between humans and mouse raise doubts as to the relevance of mice as preclinical models (Mestas and Hughes 2004) and beg for better models. The immune system of swine has been a topic of research for decades (Gustafson 1972) and has been well described using recently available resources (Mair et al. 2014).
The largest portion of the immune system resides in the mucosa-associated lymphoid tissue (MALT), the first line of immune defense, with the early postnatal period being critical for MALT development in both swine and humans (Bailey et al. 2005). Similarly to humans, swine house a large proportion of immune cells in the lymphoid tissues (Ramos et al. 1992), with the vascular-associated lymphoid tissues in particular being similarly distributed as in humans (Langohr et al. 2008); the gut-associated lymphoid tissues also show many similarities including toll-like receptor expression profile (Tohno et al. 2006). Swine also acquire pathologies of the lymphoid tissue, such as lymphomas with similar histological characteristics to those in humans (Ogihara et al. 2012). In contrast, clear differences that exist in the mucosal immune system of rodents and humans bring uncertainty to the relevance of the rodent model in the context of studying human disease (Gibbons and Spencer 2011; Korneychuk, Meresse, and Cerf-Bensussan 2015). For example, bronchus-associated lymphoid tissue while not constitutively expressed in humans, swine, and mice, is always present in rats (Pabst and Gehrke 1990; Taylor 2014). In a review of mouse and human intestinal immunity, Gibbons and colleagues discuss the similarities and differences that exist in the mucosal immune system of the 2 species (2011).
Swine also prove useful in studies of allergies, which are generally difficult to model. Swine can be sensitized to proteins such as ovomucoid (egg white protein ovomucoid) and show allergic response mediated by IgG with the presentation of IgE antibodies after oral challenge (Schmied et al. 2013). Swine studies of the complement system, another component of the innate immune response that is implicated in over 30 pathologic conditions (Sahu, Morikis, and Lambris 2003) including ischemia/reperfusion injury (Gong et al. 2015) and septic shock after traumatic blood loss (Charchaflieh et al. 2012), provide a translational framework for studies in humans (Szebeni et al. 2003; Ogura et al. 1994). Uniquely, gram-positive septic shock is not exhibited in NHP or rodents but is seen in swine as in humans (Miwa et al. 2006). Sepsis therapies previously developed in other animal models have had mixed results when applied to human populations (Dyson and Singer 2009). Mice, in particular, do not show similar gene-level responses to inflammatory stressors like trauma, burns, and endotoxemia (Seok et al. 2013).
Although infections are not considered NCDs, they play a significant role in the pathological mechanisms of many chronic diseases, including chronic pulmonary diseases (Parameswaran and Murphy 2007; Sethi and Murphy 2008) and gastrointestinal disorders (Mann and Saeed 2012; Lidar, Langevitz, and Shoenfeld 2009; Collins, Chang, and Mearin 2012), and thus, how infections affect an animal model is of importance. Additionally, the response of the animal model to infection, when compared to that of humans, gives insights into the similarities or differences in immune function. Swine respond to infection with cytokine and immune cell profiles that are similar to those of humans (Zakaryan et al. 2015). Acute phase proteins associated with innate immune response to infection are often used to distinguish between bacterial and viral infections in swine veterinary practice as done in human medicine (H. Murata, Shimada, and Yoshioka 2004). Swine present symptoms characteristic of human infections (Meurens et al. 2012) and are susceptible to many enteric human pathogens (Andrutis et al. 2000; Brady et al. 2011; Jeong et al. 2010; Sheoran et al. 2012; Steele et al. 2010; Widmer et al. 2000; Yuan et al. 1996). Swine are routinely used for studies of infections that burden human populations (Rajao and Vincent 2015) and for developing antibodies and vaccines (Sandbulte et al. 2014). Viral mutation rates in swine have been used to inform predictive models of viral behavior affecting humans (Chang et al. 2002). Swine manifest similar influenza pathogenesis as observed in humans (Khatri et al. 2010) and can serve as models of immunization to aide in the understanding of individual and population (herd) immunity (Masic et al. 2010; Hyland et al. 2004).
Swine have been widely used for studying the immune response associated with tissue rejection or graft-versus-host disease in transplantation, including recent work in mini-swine that highlighted the role of cardiac myosin in heart allotransplants (Veillette et al. 2011). Xenotransplantation of tissue from swine to humans has been making progress in overcoming hyperacute tissue rejection (Pierson et al. 1997) through creation of genetically engineered humanized swine (Lai et al. 2002), while the use of adipose derived stem cells from swine is being explored for its potential in enhancing tissue regeneration in processes like wound healing (Yun et al. 2012) and bone regeneration (Wilson et al. 2012; Monaco et al. 2011). Organ and cell xenotransplantation from genetically modified swine may be entering clinical trials in the near future (Ekser et al. 2012).
Digestive System
The impact of diet and nutrition on NCD is well established (World Health Organization 2003), thus warranting a model that is of high homology to humans in this context. The swine is perhaps the most humanlike model in reference to digestive physiology, nutrition, and dietary behavior. Swine are omnivores like humans with dietary requirements more similar than that of any other animal (Lewis and Southern 2001). They naturally eat meals rather than nibble throughout the waking hours and thus are a good choice for modeling the distinct patterns of nutrient flux in tissues and the associated physiological changes. Nutrient requirements of swine and humans are similar during infancy, adolescence, sexual maturity, and lactation (Miller and Ullrey 1987). Many similarities also exist with respect to early age digestive capacity and enzyme activity profiles (Xu et al. 1994). Young swine have been used to study neonatal nutrition on intestinal function (Puiman and Stoll 2008) and have been observed to display similar early onset of pathologies such as increased apoptosis, decreased proliferation of B cells in the pancreas, and poor gut barrier function, when optimal nutrition is lacking (Guilloteau et al. 2010).
The digestive system of swine is comparable to that of humans with respect to mesenteric vasculature, functional structural colon segments, and relative length. As such, it has been used to model a variety of conditions to study various aspects of gastrointestinal physiology. The swine pancreas is similar in density, capsule presence, vesicular and ductal topography, and the regulation of digestive secretions (Truty and Smoot 2008). The translational nature of swine has been further reaffirmed by studies examining gastrointestinal blood flow during exercise (Mortensen et al. 1998), gastrointestinal transit time of pharmaceuticals (Davis, Illum, and Hinchcliffe 2001), lumen perfusion and integrity during ischemic attack (Bustamante et al. 1994), peptic ulcers (Fuller and Boenker 1968), inflammatory bowel disease (Bassaganya-Riera and Hontecillas 2006), gastric dilation and bypass surgery (Nocca et al. 2005), pneumatosis intestinalis (Sangild et al. 2006), metabolic disorders (Torres-Rovira et al. 2012), and the gut microbiota (Heinritz, Mosenthin, and Weiss 2013).
Endocrine System
As the influence of the endocrine system is vast, we will limit our discussion to aspects related to obesity and metabolic syndrome (Grundy et al. 2004). It was estimated that obesity and related disorders account for almost 10% of all medical expenditures, about US$147 billion in 2008 (Hammond and Levine 2010), with childhood obesity alone accounting for US$14.3 billion (Trasande and Chatterjee 2009). Obesity and metabolic syndrome epitomize the physiological complexity of NCD and truly require a model that can replicate the influences of both internal and external factors in an integrated manner. The utilization of swine models in obesity-related research is particularly advantageous due to striking similarities between humans and swine in cardiovascular systems, fat accumulation, and obesity-related metabolic pathways (Spurlock and Gabler 2008). Swine and humans exhibit similar changes in metabolite profile in response to high-fat diets with swine developing a number of defining features of the metabolic syndrome, including insulin resistance, dyslipidemia, and hypertension (Neeb et al. 2010; Hanhineva et al. 2013; Dixon et al. 2002). The Ossabaw swine model in particular exhibits insulin resistance when challenged with a high-fat diet and has been used to study effects of weight loss on metabolic parameters (Sham et al. 2014). Transgenic swine with the dominant-negative gastric inhibitory polypeptide receptor genotype display characteristics of type 2 diabetes with a time course comparable to the disease in humans (Renner et al. 2010). Additionally, swine responses to exercise show corresponding changes in lipid metabolism, demonstrating the usefulness of the model for examining a well-known external influencer of obesity (Carey 1997).
Nervous System and Cognition
Translational neurological animal models are necessary to understand the neurological complications associated with NCD, such as chronic pain and impaired motor function, as well as in the context of stroke and mental illness. The study of neurobiology in the context of cognition and behavior, especially those implicated in higher brain function, has been mostly limited to NHP models. However, for ethical, economical and practical reasons, NHP do not lend themselves to large-scale or invasive studies. Swine, next to NHP, are the most humanlike neurological models described to date and thus are increasingly being used as an alternative to NHP (Lind et al. 2007).
The adult swine brain has comparable gyrencephalic structure and gray–white matter distribution to the human brain and also displays known gender structural variations (Conrad, Dilger, and Johnson 2012). Functional regions, neuroactive proteins, and cytoarchitecture of the prefrontal cortex of swine have been characterized (Jelsing et al. 2006a), including serotonin (Mezzomo, Cumming, and Minuzzi 2007) and dopamine systems (Rosa-Neto, Doudet, and Cumming 2004). Blood flow in the swine brain (Ostergaard et al. 1998) and spinal cord (Strauch et al. 2007) has also been characterized through anatomical and noninvasive imaging techniques. The rate of embryonic brain development is uniquely similar to that of humans (Jelsing et al. 2006b). The swine brain develops and matures similarly to humans with respect to myelination, biochemistry, and electrical activity (Sauleau et al. 2009). The major brain growth spurt extends from the late prenatal to early postnatal period in both the swine and humans and differs from other common animal models.
Swine are increasingly used to study and model humanlike cognition and behavior. In this context, the relevance of rodent models is limited compared to swine, where cognitive processes like learning and memory are more generalizable to humans. Swine self-establish social hierarchies (Meese and Ewbank 1973) and display sophisticated social behavior and self-recognition (Mendl, Held, and Byrne 2010). In recent years, validated cognitive tests that assess sensory awareness, classical and operant conditioning, spatial awareness, object recognition, and observational learning have been developed for swine (Gieling et al. 2013; Gieling, Nordquist, and van der Staay 2011) and allow for the evaluation of working long-term and spatial memory as well as learning (Kornum and Knudsen 2011). Swine have been used as a model of aging for examining the impact of stress and age on neurological function (Oostindjer and Amdam 2013). Swine models of neurological and neurodegenerative disorders, such as stroke, Alzheimer’s, Huntington’s, and amyotrophic lateral sclerosis (Dolezalova et al. 2014; Lorson et al. 2011), have been developed; stroke models have enabled studies of initiating events (Shao et al. 2014) and management (Platt et al. 2014). Swine have also been shown to be better than rats for studying physiologic responses following traumatic injury (Mytar et al. 2012) and exhibit humanlike expression levels of inflammatory markers after neurological insults (Zhou et al. 2014a).
Swine are an important model for neurosurgical testing and development (Sauleau et al. 2009). The anatomical and functional similarities, as well as their suitability for stereotaxic and image guided surgery, have enabled the development of neurosurgical techniques such as convection-enhanced delivery (Raghavan and Brady 2011; Miranpuri et al. 2013) and ultrasound-enhanced delivery (Liu et al. 2010). The physical size of mini-swine allows for quantitative structural magnetic resonance imaging (MRI), magnetoencephalography (Okada et al. 2005), positron emission tomography (Jensen et al. 2003), and functional MRI (Fang et al. 2006) using well-established swine-specific protocols. Several brain atlases developed for swine (Saito et al. 1998; Felix et al. 1999; Watanabe et al. 2001) facilitate many neurosurgical procedures.
Medical Device Development in Swine
The development and validation of medical devices in the context of safety and efficacy are challenging and require models that are both anatomically and physiologically relevant for the developmental pipeline to be efficient and translational. Swine have been widely used as a translational platform for the development of devices (Swindle et al. 2012; Tunev, Paulin, and Kirchhof in press ), including those for cardiovascular (Granada, Tellez, and Kaluza 2014), orthopedic (Eguchi et al. 2014), gastrointestinal (Demyttenaere et al. 2009), neurological (Federici et al. 2012), and general surgical (Pezeshki et al. 2014) applications. Advances in cardiovascular device development, particularly stents, vascular closure devices, and vascular imaging modalities, illustrate the importance of swine on modeling the combination of anatomical and physiological factors that are present in human patients. Canine models were largely used for the development of early stents, which were essentially metallic scaffolds that could maintain patency of arteries that underwent angioplasty (Granada, Tellez, and Kaluza 2014). However, as stent designs became more advanced, with the ability to deliver an antiproliferative pharmacologic agent to the arterial segment or to be absorbed into the vascular wall over time, evaluation of their safety, biocompatibility, and efficacy began to require models that more closely mimicked human physiological and pathological processes. The swine has outstandingly become the preferred model for stent development and evaluation. Their response to stent implantation is similar to humans and has been extensively characterized. For example, the vascular response to stent implantation observed at 1- and 3-month follow-up in the swine model is similar to that observed in human patients at 6 months and 1 year, respectively, after stent implantation (Virmani et al. 2003). Similarly, the use of swine for development of vascular closure devices to achieve hemostasis following percutaneous interventions has been accelerated because of similarities in vascular dimensions, arterial wall dynamics, and reabsorption rate (Tellez et al. 2010a). In the arena of vascular imaging devices, such as intravascular ultrasound and optical coherence tomography (Tellez et al. 2010a; Choi et al. 2012; Ghosn et al. 2008; Kim et al. 2011; A. Murata et al. 2010), swine have been instrumental for the validation of safety, physician technique training, and the refinement of computational algorithms to improve imaging specificity and sensitivity.
Future of Swine Models in Biomedical and Preclinical Research
The use of advanced genetic editing tools such as transcription activator–like effector nucleases (Carlson et al. 2012) and clustered regularly interspaced short palindromic repeats (CRISPR)/CRISPR-associated protein-9 nuclease (Redel and Prather in press ; Klymiuk et al. in press ; Whitworth et al. 2014) in swine has opened the door for the rapid creation of (1) novel genetic mini-swine models of human disease (monogenic and polygenic), (2) preclinical validation of gene and regenerative therapies, (3) humanized swine for xenotransplantation, and (4) preclinical platforms for targeted population and personalized medicine. Personalized medicine is the customization of medical resources to meet the needs of the individual patient, attempting to account for individual variability in disease cause, presentation, and response to treatment, which are influenced by genetics, environment, and microbiome. We believe that the rapidly increasing efficiency of swine research programs and facilities, coupled with advances in genetic editing techniques and their efficiencies, will enable the use of swine as surrogates for individual patients or select patient populations. For example, neurofibromatosis type 1 (NF1) is a unique disease that displays variable expressivity, pleiotropy, allelic heterogeneity, and age-dependent expression (Gutmann 2014). Consequently, knowing which interventions and therapies will be effective is incredibly difficult. It would be desirable to create a genetic surrogate swine with the specific NF1 mutation found in an individual patient or a group of patients and screen the efficacy of multiple therapies prior to applying the intervention in the clinic.
Summary
Modeling physiological complexity increases concordance between preclinical and clinical studies. This facilitates the selection of appropriately validated therapeutic targets saving time and costs. Failing quickly and early in the developmental process provides opportunity to redesign therapies and devices before committing to costly clinical trials. Academic institutions in the United States with well-established animal science programs, such as the North Carolina State University, University of Illinois Urbana–Champaign, Purdue University, University of Wisconsin–Madison, and University of Missouri have played a pivotal role in building a strong foundation for the use of swine in research. For example, the specific-pathogen-free swine research facility at the University of Wisconsin–Madison combines the (a) efficiency and cost-effectiveness developed by agricultural swine practices, (b) biomedical standards of hygiene and animal welfare, and (c) surgical and medical imaging capabilities to allow large-scale breeding, management, and use of mini-swine in biomedical research.
Footnotes
Acknowledgments
We would like to thank the Director (Dr. Thomas Crenshaw) and staff (Mr. Jamie Reichert, Mr. Sam Trace, and Dr. Ana Cecilia Escobar López) of the Swine Research and Teaching Center (SRTC), the Facility Manager (Ms. Catherine “Terry” Jobsis) and staff (Mr. Kim Trumble) of the Translational Research Facility, and Dr. Michael Maroney (Attending Veterinarian) at the University of Wisconsin–Madison for their roles in breeding, maintenance, and care of the biomedical swine lines and overall programmatic support that has enabled our research and knowledge incorporated into this review.
Authors’ Contribution
Authors contributed to conception or design (DTS, AT, and DS); data acquisition, analysis, or interpretation (DTS, AT, JM, FA, JW, DB, KD, SR, and DS); drafting the manuscript (DTS, AT, JM, FA, DB, KD, and DS); and critically revising the manuscript (DTS, AT, JM, FA, JW, DB, KD, SR, and DS). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
