Abstract
Due to potential misdiagnosis of hyaline glomerulopathy (HG) for amyloidosis, a retrospective study of B6C3F1 mice from the National Toxicology Program (NTP) archives was undertaken to determine whether HG had occurred in prior NTP studies and, if so, whether these 2 glomerular lesions could be routinely discriminated. Kidney slides from 7 amyloid-positive control mice, 2 HG-positive control mice, 3 normal or negative control mice, and 41 potential HG mice (with renal-only deposits previously diagnosed as amyloid) were evaluated using hematoxylin and eosin (H&E), periodic acid Schiff (PAS), Congo red (CR), and Masson’s trichrome (MT) stains. Utilizing these techniques, HG was reliably distinguished from amyloidosis. All 41 potential HG mice had glomerular deposits histochemically inconsistent with amyloid; the deposits were PAS positive and CR negative. Four of the 41 mice were selected for transmission electron microscopy of the glomerular deposits; ultrastructurally, the deposits in these animals were consistent with HG and not amyloid. Our findings indicate that HG is a spontaneous lesion in B6C3F1 mice of low occurrence, is commonly misdiagnosed as amyloidosis, and is more likely than amyloid to cause glomerular deposits in mice without evidence of deposits in other tissues. Also, HG can be distinguished from amyloid on H&E evaluation; however, the distinction is improved with use of PAS or CR staining and/or ultraviolet evaluation.
Keywords
Introduction
Hyaline glomerulopathy (HG) was identified as a unique test article–related pathologic change in both male and female B6C3F1 mice and F344 rats in the National Toxicology Program (NTP) 2-year (105 week) carcinogenicity study of pulegone (Adams et al. 2011; NTP 2011). Pulegone is a monoterpene ketone used as a flavoring agent and present in the leaves and flowering tops of several members of the mint family Lamiaceae (International Agency for Research on Cancer [IARC] Pulegone Monograph). The NTP decision was to use the term “hyaline glomerulopathy” to characterize the glomerular changes because the morphologic changes present in this pulegone study, as determined by light and electron microscopy, were similar to those described by Wojcinski, Albassam, and Smith in 1991, which was the first published report that fully characterized this lesion. The presence of HG as a potential test article–related change in these studies, as well as the report of these changes occurring spontaneously in B6C3F1 mice (Wojcinski, Albassam, and Smith 1991), raised the question of the prevalence of spontaneous HG in control B6C3F1 mice in prior NTP studies. However, in order to determine the prevalence of HG, it first needed to be determined if HG could be reliably distinguished from renal glomerular amyloid.
The term hyaline glomerulopathy is proposed to be morphologically defined as the accumulation of nonamyloid, eosinophilic material expanding the glomerulus. Additional adjunctive staining techniques and ultrastructural analysis may further categorize the nature of the glomerulopathy. In the B6C3F1 mice from the 2-year pulegone study and the Wojcinski, Albassam, and Smith study, the material deposited within the renal glomeruli was periodic acid Schiff (PAS) positive and Congo red (CR) negative (NTP 2011; Wojcinski, Albassam, and Smith 1991). The ultrastructural findings in a mildly affected pulegone-treated mouse were composed of amorphous, finely granular deposits, and irregular basement membrane (BM) thickening (Adams et al. 2011; NTP 2011). Ultrastructural analysis of the deposits from a markedly affected pulegone-treated mouse and the spontaneous HG lesions in the Wojcinski, Albassam, and Smith report revealed well-structured, fibrillar, curvilinear, nonbranching deposits oriented in parallel rows often swirling and creating a “fingerprint” pattern (Adams et al. 2011; NTP 2011). The findings in the markedly affected animal from the pulegone study were the same as those reported by Wojcinski, Albassam, and Smith who described spontaneous HG deposits as loosely packed material composed of linear structures that were straight to curved and formed single- or double-layered lamellar structures (fibrils) that were 6.1 to 17.01 nm in diameter, with an intralamellar distance of 18.4 to 24.4 nm (Wojcinski, Albassam, and Smith 1991).
Morphological alterations consistent with HG in mice have been reported sporadically in the literature. In addition to the abovementioned report of HG as a spontaneous lesion in B6C3F1 mice (Wojcinski, Albassam, and Smith 1991), there was a recent report of mixed hyaline and amyloid deposits in CD-1 [Crl:CD1(ICR)] mice (Frazier et al. 2014). HG has also been described in young ddY (DDY/JclDidSeyFrkJ) mice (Kouchi et al. 2011). Additional reports of deposits morphologically reported to be consistent with HG include glomerulopathy in NNT-1/BSF-3 mice (Senaldi et al. 2002), idiopathic glomerulopathy in a young ICR (CRJ:CD-1) mouse (Shibuya et al. 1990), spontaneous glomerulosclerosis in aging RF (RF/UP) mice (Gude and Upton 1960), and spontaneous glomerular lesions in ICGN (ICR-derived strain with glomerulonephritis) mice (Ogura et al. 1989).
Renal glomerular amyloidosis has also been described as amorphous, eosinophilic extracellular glomerular deposits (Seely 1999) and thus would be expected to have a similar morphologic appearance to HG on routine hematoxylin and eosin (H&E)-stained kidney sections. Amyloidosis is a condition in which there is accumulation of abnormally folded proteins within tissues, and in mice it commonly accumulates within the renal glomerulus as well as other tissues such as the small intestine, mesenteric lymph nodes, ovaries, adrenal glands, heart, liver, thyroid, pancreas, parathyroid gland, and gall bladder (Frith and Chandra 1991). A number of histochemical stains have been utilized to identify amyloid in paraffin-embedded and/or frozen tissue sections including CR, sirius red, crystal violet, and thioflavine-T (Cooper 1969), but CR is most commonly used (Picken 2010). Amyloid deposits react in a particular way with CR and should appear as apple green birefringence upon examination under polarized light; however, small deposits can be difficult to visualize. Evaluating CR-stained slides with ultraviolet (UV) light, known as CR fluorescence, reportedly will aid in the detection of smaller amyloid deposits (Linke 2000; Sen and Basdemir 2003). Ultrastructural evaluation allows for conclusive identification of amyloid, and it can be distinguished by the characteristic delicate filaments of variable length and approximately 50 to 140 Å (5 to 14 nm) in width arranged either haphazardly or within bundles (Cohen and Calkins 1959).
Amyloid deposition in multiple sites, including renal glomeruli, has been cited as a common background finding in certain mouse strains including CD-1 mice (Engelhardt, Gries, and Long 1993; Frith and Chandra 1991). A search of the NTP databases (Carcinogenesis Bioassay Database System [CBDS; 1971 to 1982] and the Toxicology Data Management System [TDMS; 1982 to present] at http://cebs.niehs.nih.gov) revealed that the most common sites of amyloid deposition in B6C3F1 mice include the kidney, spleen, and liver, with variability in which and how many organs were affected in each mouse (Supplemental Tables 1 and 2).
Herein we report the results of the investigation into the occurrence and identification of glomerular changes, morphologically similar to amyloid, in B6C3F1 mice from chronic studies conducted through the NTP. The objectives of this study were to determine if HG could be discriminated from glomerular amyloid in mouse kidneys and, if so, to determine the occurrence, if any, of spontaneous HG in B6C3F1 mice from NTP 2-year carcinogenicity bioassays, in effect assessing the possible misdiagnosis of glomerular amyloidosis as a spontaneous background lesion. We hypothesized that HG may account for a portion of the renal-only amyloid cases and that HG could not be readily distinguished from glomerular amyloidosis with H&E evaluation alone but would require additional histochemical stains. As such, a panel of stains including CR, PAS, and Masson’s trichrome were applied in addition to the routine H&E stain. Kidney sections stained with H&E and CR were also evaluated with UV fluorescence to determine if there were any differences in autofluorescence between the glomerular deposits. A select number of cases were also evaluated with transmission electron microscopy (TEM) for ultrastructural analysis of the deposited material.
Materials and Methods
Selection of Control Mice
Eight B6C3F1 mice, 4 of each sex, were selected as positive controls for glomerular amyloidosis based upon the diagnosis of amyloid deposition in the kidney and at least one other organ. One female was excluded as the glomerular deposits were CR negative, and this animal was moved to the potential HG test group. Two B6C3F1 mice, 1 of each sex, were selected from the pulegone chronic study (NTP, 2011) to act as positive controls for HG. In addition, 4 B6C3F1 mice, 2 of each sex, with no prior diagnoses in the kidney, were selected from an unrelated chronic study to act as normal mouse kidney controls. One of these 4 was removed due to renal disease that was not apparent on H&E but was diagnosed with special stains. Therefore, 3 animals, 1 male and 2 females, were used as normal mouse kidney controls. The initial and final animal selection data are summarized in Table 1.
Initial and Final Animal Selection Data.
Selection of Potential HG Cases
Our investigation focused on the identification of B6C3F1 mice in which amyloid deposition was diagnosed solely in the kidney. Due to the somewhat similar appearance of glomerular amyloidosis and HG on H&E-stained slides resulting in potential for misdiagnosis, it was thought that animals with solely renal amyloidosis would provide the best pool of potential HG cases. The selection bias caused by the restriction of the pool of candidates was considered acceptable due to the proposed objectives of the study. A search of the NTP databases resulted in the identification of 191 cases of renal-only amyloidosis (90 males and 101 females) from the 92,005 B6C3F1 mice represented in the CBDS database (1971 to 1982) and 113 cases of renal-only amyloidosis (19 males and 94 females) from the 89,052 B6C3F1 mice represented in the TDMS database (1982 to present).
Initially, a group of potential HG mice were selected for additional evaluation ideally to be divided equally between males and females in order to eliminate gender-based bias; however, this study was not designed to investigate gender-related differences in glomerular deposits. Forty mice, including 23 females and 17 males, were ultimately selected from 27 NTP chronic studies for additional evaluation based upon tissue availability and quality of tissue preservation (Table 1). One female mouse from the amyloid-positive control group was diagnosed with Congo-negative renal deposits and moved to this potential HG test group, resulting in a total of 41 mice for evaluation. The initial evaluation of the potential HG mice included microscopic examination of the original H&E-stained sections of kidney, spleen, and liver to ensure that amyloid deposition was not present in these other organs during the primary study evaluation. Kidney sections were also assessed for adequate tissue quality, and moderately to markedly autolyzed tissue sections were excluded from the study. Individual animal details of the mice used in this retrospective study are available in Supplemental Table 3.
Additional Mice
An additional 21 mice were evaluated for use as either amyloid-positive controls or potential HG cases. The majority of these animals were not selected, predominantly based upon poor tissue preservation or lack of tissue availability. However, 1 mouse had mixed glomerular deposits, and this mouse was chosen for additional evaluation.
Histology
Paraffin-embedded blocks from the NTP archives were sectioned at both 5- and 8-µm thicknesses. Although 3-µm sections are often preferred for specific evaluation of renal glomerular BMs, 5-µm thick sections were utilized, as this thickness is more commonly used for routine kidney evaluation. Five-µm thick sections from each mouse were stained with H&E, CR, MT, or PAS. A single 8-µm thick section was also stained with CR, which was within the manufacturer recommended range of 6 to 10 µm. Staining with H&E was performed according to standard laboratory protocol. The PAS (Catalog #38016SS4; Leica Biosystems, Buffalo Grove, IL 60089 United States), CR (Catalog #38016SS9; Leica Biosystems, Buffalo Grove, IL 60089 United States), and MT (Catalog #25088; Polysciences, Inc., Warrington, PA 18976 United States) stains were performed according to the manufacturer’s instructions. Appropriate positive control slides were used for the PAS, CR, and MT staining processes.
Histopathology
Routine light microscopy was used to evaluate the H&E-, PAS-, CR-, and MT-stained slides. CR-stained slides were also evaluated for birefringence under polarized light. All of the newly prepared H&E-stained slides were evaluated by 2 American College of Veterinary Pathology (ACVP) board-certified veterinary pathologists (JSH and CLJ), and any mice affected with HG were independently scored for lesion severity by both pathologists. Discrepancies in severity scores were resolved by consensus decision between the 2 pathologists.
Severity of the lesions from the potential HG mice was graded on a 5-point scale (0 =
Summary of Hyaline Glomerulopathy Severity Scoring.
aPresence of other glomerular/periglomerular pathology would result in an increased severity score.
UV Evaluation
UV evaluation was performed on all H&E- and CR-stained slides, and tissues were assessed for the presence or absence of autofluorescence. UV evaluation has been shown to be a simple method to enhance CR evaluation of amyloid deposits (Sen and Basdemir 2003). The evaluation was performed utilizing an Olympus BX41 fluorescence microscope. Two filters were used including the “green” HG.TR (Texas Red) filter set (Chroma #41004), which has an absorption maximum of 560 nm (green) and an emission maximum of 645 nm (red), and the “blue” U-MNIB filter set (Olympus FITC [fluorescein isothiocyanate]), which has an absorption maximum of 470 to 490 nm (blue) and an emission maximum of 515 nm (green).
TEM
Six mice were selected for TEM evaluation. Five mice were selected randomly, 1 from the amyloid-positive control group and also 1 mouse from each severity group of presumed HG. Only mice from studies performed in the past 15 years were included in the random selection process primarily due to tissue availability and potential issues with tissue quality. Randomization was completed by the random number generator at www.randomizer.org. The 6th mouse selected for TEM evaluation had evidence of an unusual and unexpected mix of different glomerular deposits on H&E, CR, and UV evaluation that warranted further investigation.
Archived formalin-fixed kidneys were processed according to standard TEM protocol. In brief, the renal cortices were trimmed into approximately 1 mm × 1 mm × 1 mm cubes, given an additional overnight stabilization fixation in 0.1 M pH 7.3 phosphate buffered 2.5% glutaraldehyde, processed through an industry standard buffer rinse, osmium postfixation, graded alcohol–acetone dehydration series, and embedded in Spurr’s epoxy resin blocks. The blocks were thick-sectioned on an ultramicrotome at approximately 1-µm thickness, placed on glass slides, and stained with toluidine blue. The thick sections were evaluated using light microscopy to determine the areas of interest. Each selected region was processed into a thin section at approximately 80 nm thickness and placed on a 200-mesh copper grid, stained with uranyl acetate and lead citrate using standard procedures, and examined on a Zeiss EM 900 TEM.
Results
Amyloid-positive Control Mice
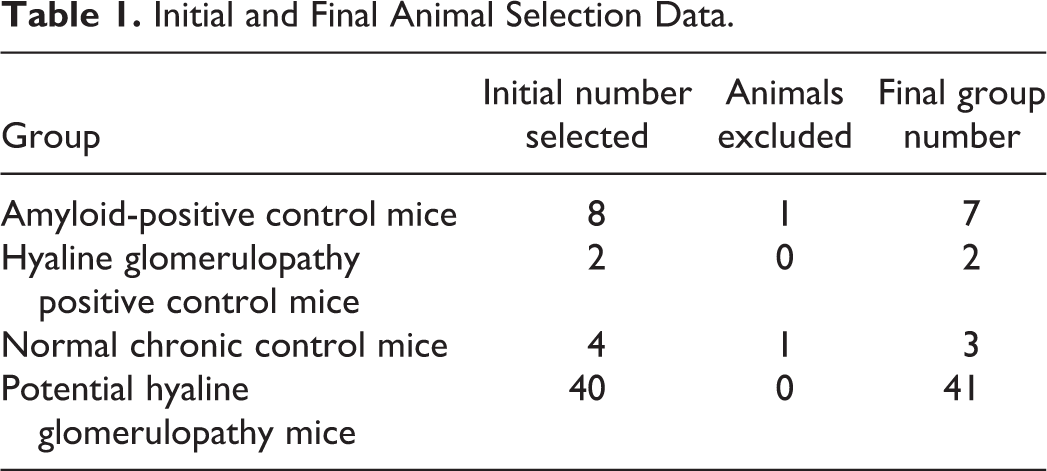
H&E stained amyloid positive-control kidney sections had amphophilic deposits present within the glomeruli and, in some mice, sporadically within the renal interstitium (Figure 1A; white arrowheads). The glomerular changes in all of the amyloid-positive control mice were quite extensive and, although this finding was not specifically graded for each animal, all would have been considered at least moderately affected. Glomerular deposits were fairly uniform throughout all glomeruli in the kidney and relatively equally distributed throughout each glomerulus within each mouse. The nuclei present within the glomeruli did not appear to be increased in number compared to controls and were also equally distributed throughout the glomerulus, although often displaced to the periphery of the loops by the deposits. In general, the visceral and parietal layers of Bowman’s capsule were in close apposition, resulting in little or no Bowman’s space surrounding the glomeruli, although determining if true synechiation of the layers was present was often difficult. Typically, Bowman’s parietal epithelium was hypertrophied, usually with minimal to mild expansion of Bowman’s capsule by collagen and amyloid (Figure 1A; black arrowheads). These changes were often accompanied by concurrent nonspecific renal changes consisting of a varying combination of tubular degeneration and regeneration, tubular atrophy, interstitial fibrosis, lymphoplasmacytic interstitial infiltrates, and intraluminal tubular proteinaceous casts (not shown).
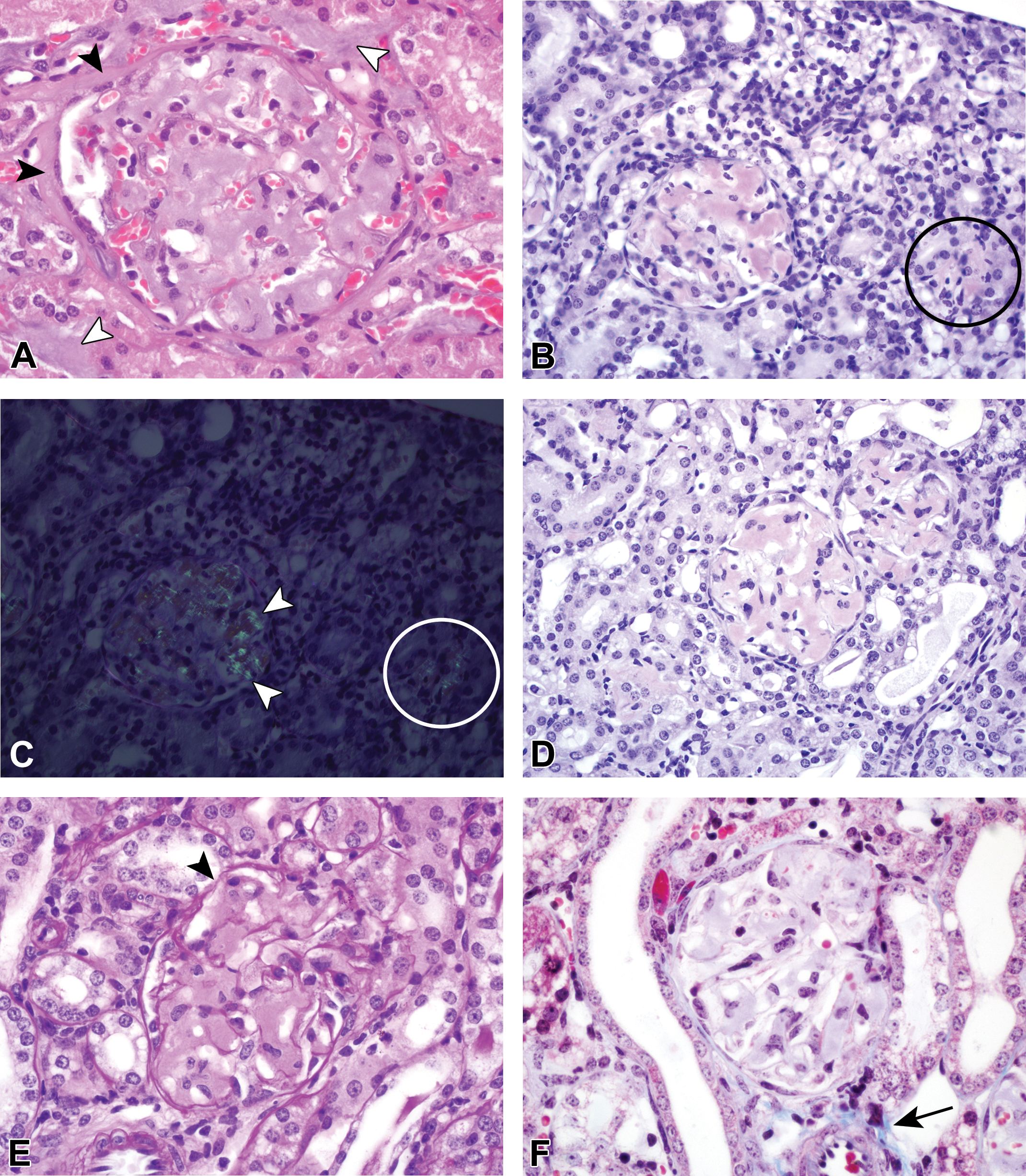
(A) Hematoxylin and eosin–stained amyloid deposits within both the interstitium (white arrowheads) and glomerulus are amphophilic and amorphous. The glomerular deposits fairly evenly expand the capillary loops. In general, the parietal and visceral Bowman’s layers are in close apposition or adhered. There is occasional minimal expansion of Bowman’s parietal epithelium (black arrowheads) by both fibrosis and amyloid. Congo red (CR) positive renal amyloid deposits (B–D) present both within glomeruli and occasionally the interstitium (circles) are characterized by pinkish-red coloration with CR staining (B and D) and green birefringence under polarization of CR-stained slides (C). Note the increased staining intensity of the amyloid deposits in the 8-µm thick section in panel B compared to the 5-µm thick section in panel D. Renal amyloid deposits are pale pink on PAS staining (E) and pale blue on MT staining (F). The basement membranes (arrowhead) in the PAS-stained section in panel E are deep pink to magenta, which is expected for these structures. The normal bright blue staining of collagen with MT is indicated by the arrow in panel F.
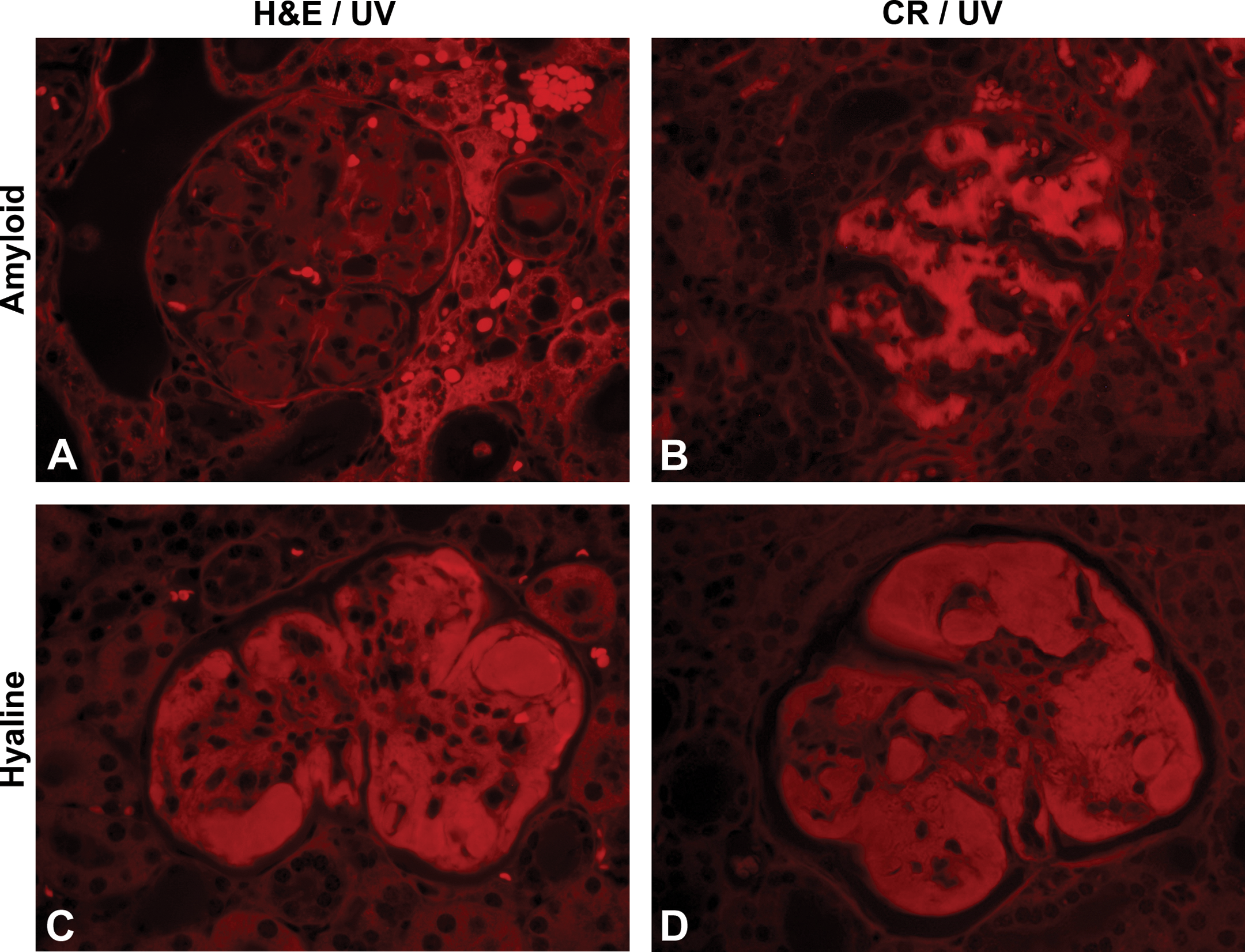
On the 8-µm sections, glomerular amyloid deposits were considered “positive” with CR when, with light microscopic evaluation, the glomerular loops and sporadic regions within the interstitium were pinkish-red (Figure 1B), and under polarization, the deposits exhibited apple green birefringence (Figure 1C). All 7 amyloid control mice exhibited CR positive staining and birefringence under polarized light. Although it was possible to distinguish the appearance of the amyloid deposits from HG deposits on the 5-µm CR-stained sections, the amyloid deposits were not as vibrant in the 5-µm sections (Figure 1D) when compared to the 8-µm sections, nor did the deposits exhibit any significant birefringence on polarization with microscopic examination (not shown). Very faint apple green birefringence could occasionally be captured with digital photography of the 5-µm sections (not shown). The glomerular amyloid deposits were diffusely pink with PAS staining (Figure 1E) and were uniformly very pale blue with MT staining (Figure 1F).
HG Control Mice
The glomerular deposits from the 2 mice (male and female) selected from the pulegone study (NTP, 2011) as HG positive controls had similar patterns of staining, and representative HG-positive control glomeruli are depicted in the second row of Figure 2. The glomerular deposits were eosinophilic on H&E, bright pink to magenta with PAS staining, variably blue to pink with MT staining, and CR negative with light microscopy. The CR-stained sections did not exhibit birefringence under polarization (not shown).
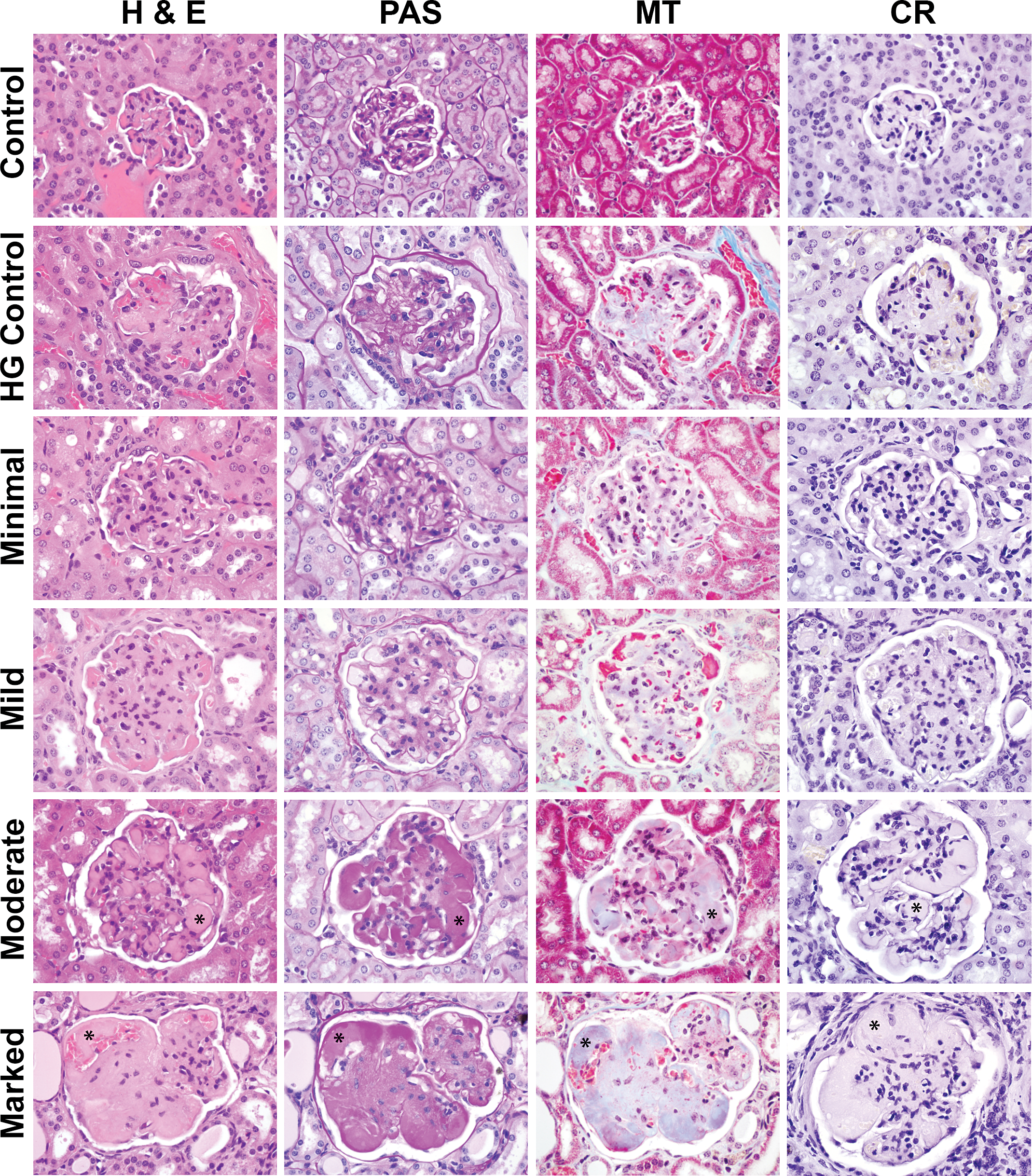
Glomeruli from a normal age-matched B6C3F1control, a hyaline glomerulopathy (HG) positive control of minimal severity from the National Toxicology Program pulegone study and from spontaneous HG B6C3F1 mice with minimal, mild, moderate, or marked glomerular changes. Note the overall enlargement of the glomeruli in affected mice when compared to the age-matched control. On routine hematoxylin and eosin staining, all HG mice have eosinophilic deposits expanding the mesangial region and glomerular basement membranes. Intracapillary deposits or “hyaline thrombi” were present in few mildly affected and all moderately and markedly affected mice (asterisks). The mesangial and intracapillary deposits are periodic acid Schiff positive (bright pink to magenta) and Congo red negative. The deposits were typically light blue to pale purple with Masson’s trichrome staining, the purple coloration due to the presence of numerous, small (1 to 2 μm) bright pink granules also present within some of the glomerular deposits.
Spontaneous HG Mice
The glomeruli from all 41 potential HG mice, including the one that was added after being removed from the amyloid-positive control group, exhibited similar staining to the HG-positive controls and dissimilar staining to the amyloid-positive controls; thus, the renal deposits in these 41 mice were considered to be consistent with spontaneous HG and inconsistent with amyloid. Figures 2 (first column) and 3 contain representative photomicrographs of H&E-stained kidneys with spontaneous HG at low (Figure 3A) and high (Figure 2, first column and Figure 3B) magnification. At low magnification, the glomeruli were distorted to variable extents by eosinophilic material, which at high magnification variably expanded the mesangium and glomerular loops, and formed discrete intracapillary amorphous eosinophilic deposits or “hyaline thrombi” (Figures 2 and 3B, asterisks). Relatively paucicellular areas were intermixed with regions of mesangial hypercellularity.
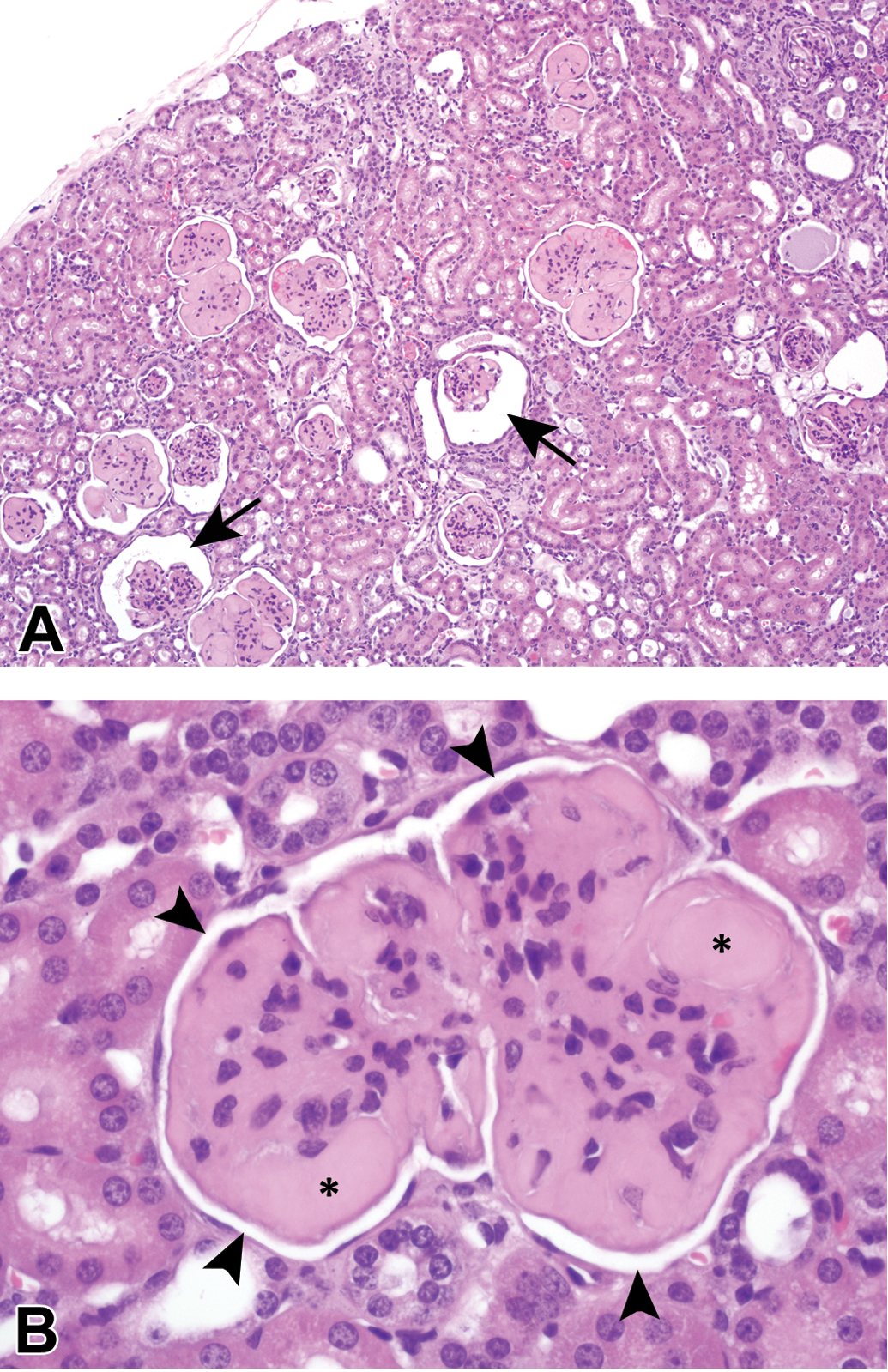
Hyaline glomerulopathy at low (A) and high magnification (B) in a moderately affected mouse. The deposits are eosinophilic with hematoxylin and eosin staining and are predominantly mesangial and subendothelial, but also form the so-called hyaline thrombi (asterisk) within capillaries. The mesangium is often hypercellular in the affected mice. Typically, Bowman’s space was at least slightly distended resulting in a small separation of the visceral and parietal Bowman’s layers and creating a white space surrounding the glomerulus (panel B, arrowheads). Bowman’s space was occasionally considerably expanded (panel A, arrows), although, it was rarely cystic.
The HG deposits within the glomeruli of minimally and mildly affected mice were typically relatively equally distributed throughout the entire glomerulus and equally affected all glomeruli throughout the kidneys. Minimally and mildly affected glomeruli usually had increased eosinophilic material expanding both the mesangial and capillary loops, generally accompanied by an overall increase in glomerular size and cellularity. More intra-animal variability was present in the extent of deposition within the glomeruli of moderately and markedly affected mice.
Other associated glomerular changes such as synechiation, sclerosis, and obsolescence often noted in the amyloid-positive control mice were uncommon to rare in the HG mice. It was not uncommon, however, to see variable thickening of Bowman’s capsule and reaction of the parietal epithelium. In general, Bowman’s space was easy to visualize as a thin, complete “halo” surrounding each glomerulus (Figure 3B, arrowheads), and occasionally the space was dilated (Figure 3A, arrows), although rarely cystic in appearance.
Other relatively nonspecific kidney changes were variably present in HG affected mice. Changes consisted primarily of, but were not limited to, renal tubular degeneration and regeneration, tubular dilatation with or without intraluminal hyaline casts, thickened tubular BMs, interstitial fibrosis, and lymphoplasmacytic infiltrates (not shown). It was not uncommon for the kidneys to appear smaller, with decreased spacing between glomeruli (Figure 3A), likely due to tubular atrophy and dropout.
The HG deposits were bright pink to magenta on PAS staining (Figure 2, second column). The deposits varied in color with MT staining (Figure 2, third column), and some regions were pale blue and others were bright pink. Staining coloration with MT is based predominantly upon pore size of the material stained, and as it is thought that there is entrapped fibrin and other plasma proteins within the larger HG deposits, a mixed staining result is not unexpected. Seven of the 41 mice with spontaneous HG had predominantly bright pink staining throughout the glomeruli with MT staining and as discussed more thoroughly in the UV evaluation section below, the brightly pink-stained material correlated to intracytoplasmic granules within mesangial cells or possibly macrophages. The HG deposits were CR negative on light microscopy (Figure 2, last column) and did not exhibit birefringence under polarization (not shown).
Severity Scoring of HG
Severity of the HG lesions from the 41 mice was graded independently by 2 ACVP board-certified veterinary pathologists (JSH and CLJ) as described in the Materials and Methods section and summarized in Table 2. Severity scores for each animal are included in Supplemental Table 3. Concordance between the pathologists was high (70%), the severity grades assigned by both pathologists were within 1 grade for each mouse for the remaining 30%, and the final reported severity scores reflect the consensus opinion of the 2 pathologists. Representative glomeruli at each severity grade, in addition to a normal age matched control and a pulegone-treated HG-positive control, with each stain are depicted in Figure 2. In all affected mice, the glomeruli were notably enlarged compared to the normal controls. In moderately and markedly affected mice, the glomeruli were considerably enlarged and distorted due to numerous variably sized deposits.
UV Evaluation
None of the H&E-stained amyloid deposits autofluoresced with UV illumination (Figure 4A). Amyloid deposits exhibited bright autofluorescence with UV illumination of CR-stained slides (Figure 4B). All HG deposits exhibited some degree of autofluorescence when the H&E slides (Figure 4C) or CR-stained slides (Figure 4D) were evaluated under UV light with either a TR or an FITC filter set. The degree of auto-fluorescence was dependent upon the extent of the deposits. In 5 mice, the glomerular loops contained rare to occasional autofluorescent wispy or nodular protrusions extending out along the subepithelial surface (Figure 5A). Although these protrusions resembled the spike-like protrusions typically associated with subepithelial immunoglobulin deposits, they were ultrastructurally inconsistent with immunoglobulin deposition (Figure 5B) and will be discussed more extensively in the TEM results section below. In the 7 spontaneous HG mice with predominantly pink deposits with MT staining, the glomerular loop deposits were brightly fluorescent and often granular with or without accompanying hyaline thrombi. In these 7 mice, the palely autofluorescent mesangium was similar to the other HG mice; however, there were occasional intracytoplasmic granules within mesangial cells or possibly macrophages (Figure 6A, inset, white arrow). With MT staining, the granular deposits were bright pink and were present either throughout the glomerular loops, in larger 3- to 4-μm diameter droplets within the mesangium, and/or occasionally present within the cytoplasm of mesangial cells (Figure 6B, inset, black arrow).
Ultraviolet (UV) evaluation of spontaneous amyloid (A and B) and hyaline glomerulopathy (HG; C and D) at high magnification (Texas Red filter set). Amyloid deposits stained with hematoxylin and eosin (H&E; A) do not exhibit autofluorescence but are autofluorescent when stained with Congo red (CR, B). UV evaluation of spontaneous HG exhibits bright autofluorescence with both H&E (C) and CR (D).
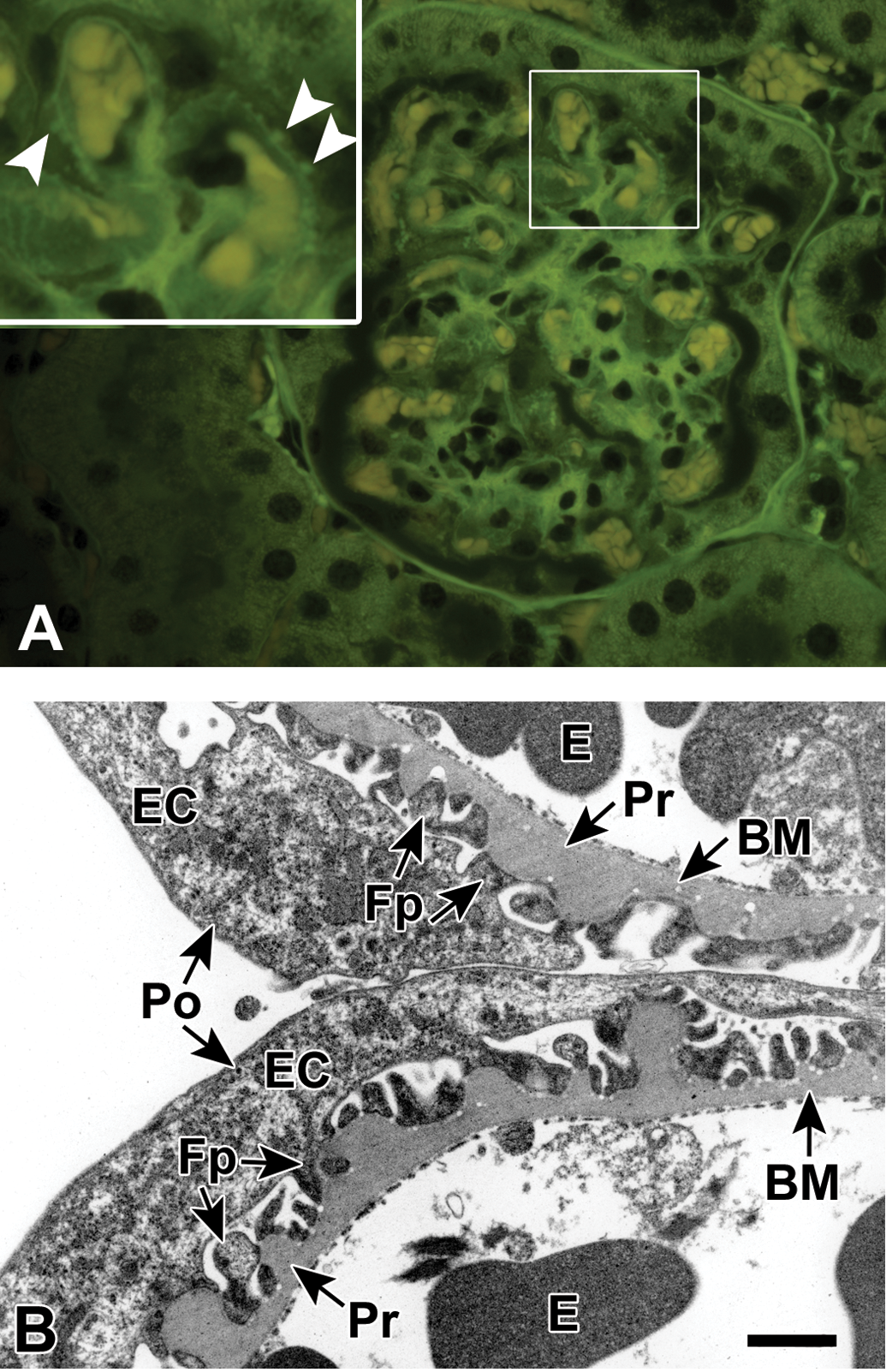
Ultraviolet illumination of a hematoxylin and eosin–stained kidney minimally affected with spontaneous hyaline glomerulopathy (A, FITC filter set). The mesangium is minimally expanded by fibrillary deposits and a slight increase in glomerular cellularity. A small number of punctate protrusions extend from the glomerular capillary basement membranes (BMs; white arrowheads, inset). Transmission electron micrograph of a glomerulus at 15,400× (B) from the same mouse depicting portions of 2 glomerular capillary loops with intraluminal erythrocytes and associated visceral epithelial cells with podocytes. Multiple variably sized thickenings of the BM are present, extending toward the epithelial (podocyte) side of the BM. The thickenings are composed of material indistinguishable from the adjacent unaffected BM. There is a minimal multifocal podocyte fusion predominantly over the thickenings. BM = basement membrane; E = erythrocyte; EC = epithelial cell; Fp = foot process; Po = podocyte; Pr = protrusion. Scale bar = 1.0 µm.
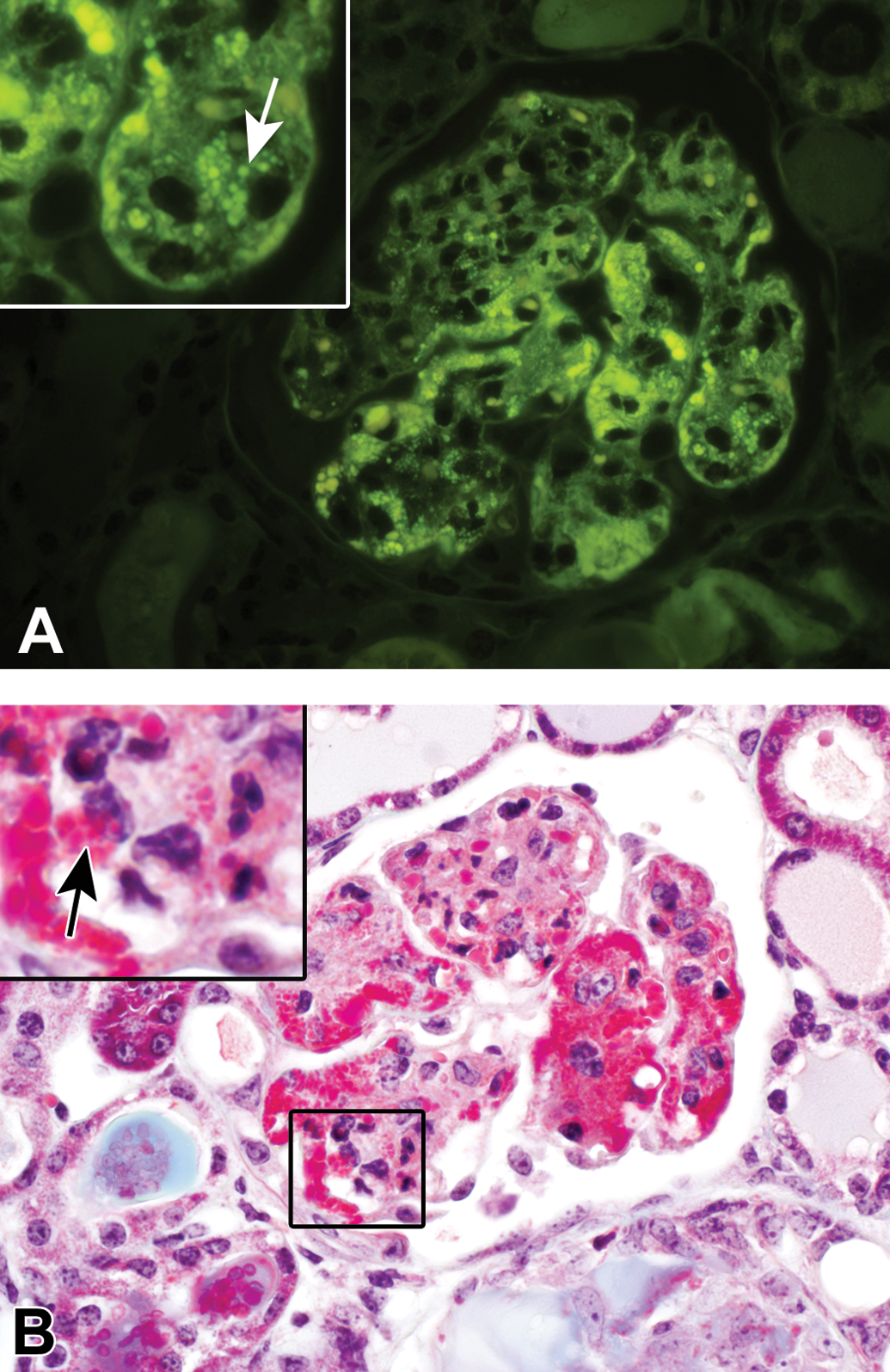
Ultraviolet illumination of a hematoxylin and eosin–stained kidney mildly affected with spontaneous hyaline glomerulopathy (A, FITC filter set). Inset at higher magnification: distinct, bright autofluorescent granules appear to be within the cytoplasm of mesangial cells or other phagocytic cells (white arrow). These autofluorescent granules correlated with bright pink to magenta granular deposits evident on Masson’s trichrome staining (B). Inset at higher magnification: again, these deposits often appeared to be within the cytoplasm of mesangial cells or other phagocytic cells (black arrow).
Nonglomerular and Extrarenal Deposits in the Amyloid-positive Control Group
Four of the 7 amyloid control mice had papillary necrosis secondary to dense deposition of amyloid within the renal medullary interstitium (not shown). Six of the 7 controls had characteristic amyloid deposits in the liver (Figure 7A, B, and C) and spleen (Figure 7D, E, and F), which were amphophilic with H&E staining (Figure 7A and D), did not autofluoresce with UV evaluation of H&E-stained sections (Figure 7B and E), and were CR positive (Figure 7C and F). The 7th amyloid control had amyloid deposits in the glandular stomach and adrenal cortex (not shown).
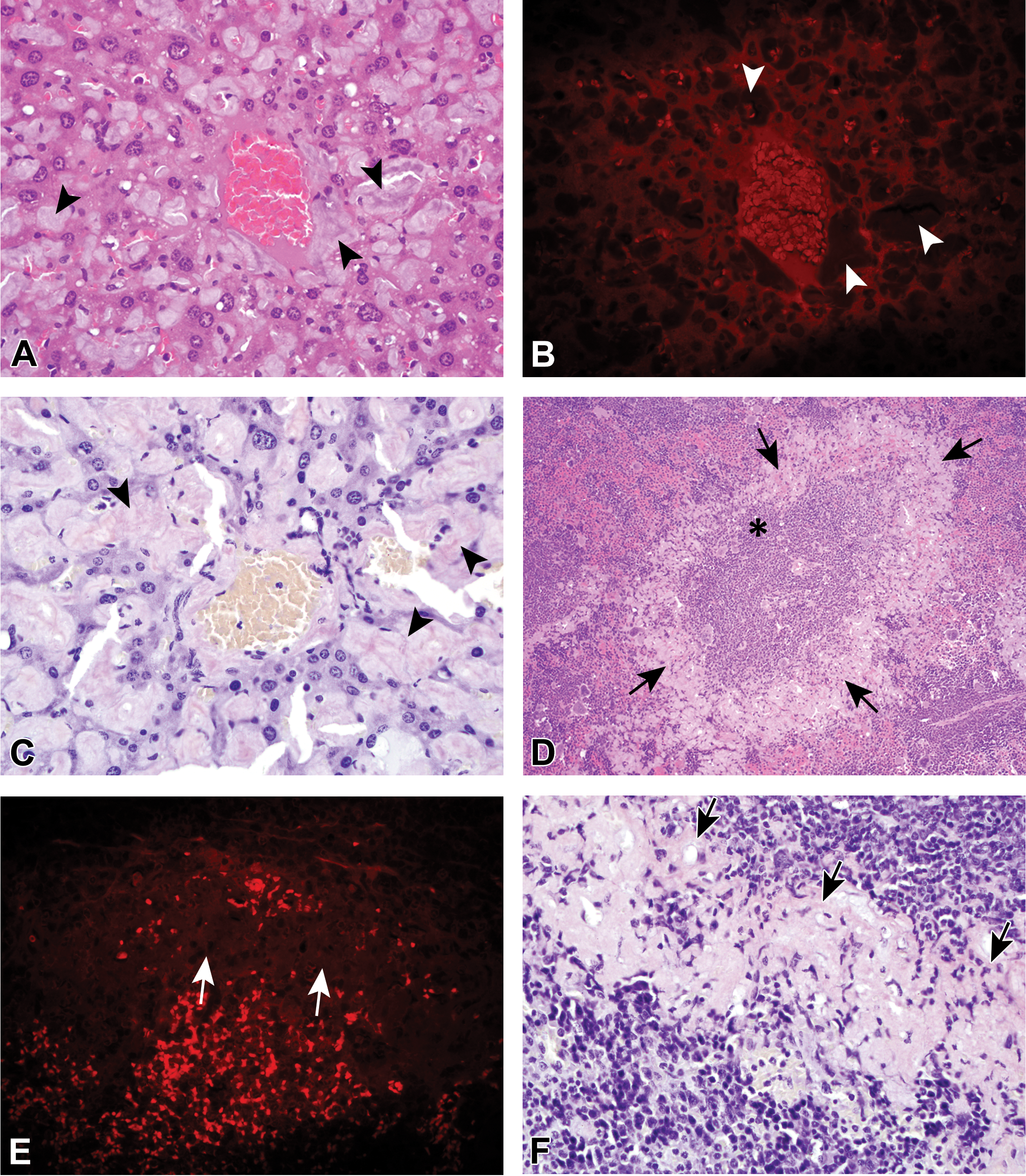
Extrarenal amyloid deposition in the liver (A–C) and in the spleen (D–F) was very common in amyloid-positive control mice. Amyloid deposits in the liver (arrowheads) were predominantly within the sinusoids and around smaller veins. The splenic amyloid deposits (arrows) were most often within the mantle zone surrounding the periartiolar lymphoid sheaths and lymphoid follicles (panel D, asterisk). Hematoxylin and eosin–stained amyloid deposits (panels A and D) were amphophilic and did not autofluoresce under ultraviolet evaluation (B and E). Congo red–stained amyloid deposits were pinkish (C and F).
Although amyloid deposition was previously diagnosed in 2 organs (kidney and lymph node) from one of the proposed amyloid-positive controls, the material deposited within the renal glomeruli was CR negative by both normal light microscopy and with polarization and was PAS positive, consistent with HG, not amyloid. The lymph node section was not processed for further evaluation; however, previously undiagnosed eosinophilic material was present within splenic vessels on the H&E-stained slide (Figure 8A). Upon further review, the material present within the vessels exhibited autofluorescence under UV evaluation (Figure 8B), was bright magenta with PAS staining (Figure 8C), and was CR negative (Figure 8D). As such, this mouse was removed as an amyloid-positive control and was added to the potential HG group.
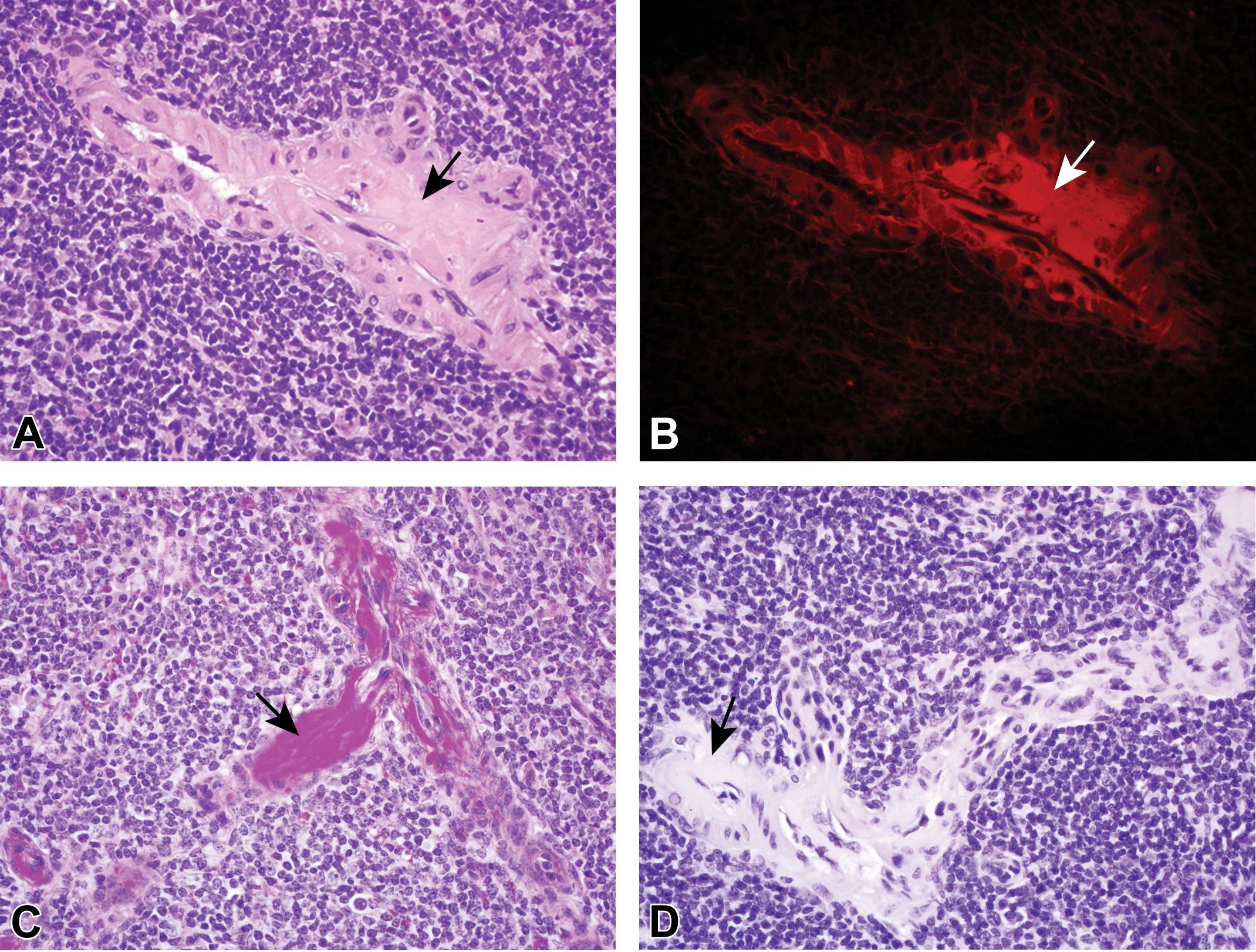
Deposits (designated in all panels by arrows) expanding vessel walls in the spleen stained with hematoxylin and eosin are eosinophilic with light microscopy (A) and autofluorescent with ultraviolet evaluation (B, Texas Red filter set). The deposits are bright pink to magenta with periodic acid Schiff staining (C) and negative with Congo red staining (D).
Based upon the discovery of PAS positive hyaline deposits within the vessels of this mouse, additional assessment was done to determine if similar deposits were present in other mice. Reevaluation of PAS-stained slides was undertaken to identify any other unusual PAS-positive deposits within nonglomerular sites. Five mice from the potential HG group were identified with extrarenal deposits within vessel walls, similar to those depicted in Figure 8, including in the kidney, spleen, urinary bladder, testes, and ovary (not shown). The search for extrarenal deposits was not exhaustive and was limited to the other tissues (typically liver, spleen, and some intestinal sections) present on the PAS-stained slide.
TEM
The ultrastructural characteristics of the deposits seen in the amyloid-positive control were consistent with what has been previously described for amyloid (Cohen and Calkins 1959). Deposits were composed of randomly arranged, occasionally bundled, nonbranching, relatively straight, slender, 5 to 10 nm diameter fibrils of variable length (Figure 9A and B). In this mouse, the deposits were predominantly subendothelial and mesangial, rarely subepithelial and often disrupted the paramesangial capillary BMs. There was highly variable podocyte fusion and effacement, with occasional regions of complete loss of primary and secondary foot processes (Figure 9A). Podocytes are highly specialized epithelial cells that cover the glomerular basement membrane with their numerous interdigitating foot processes.
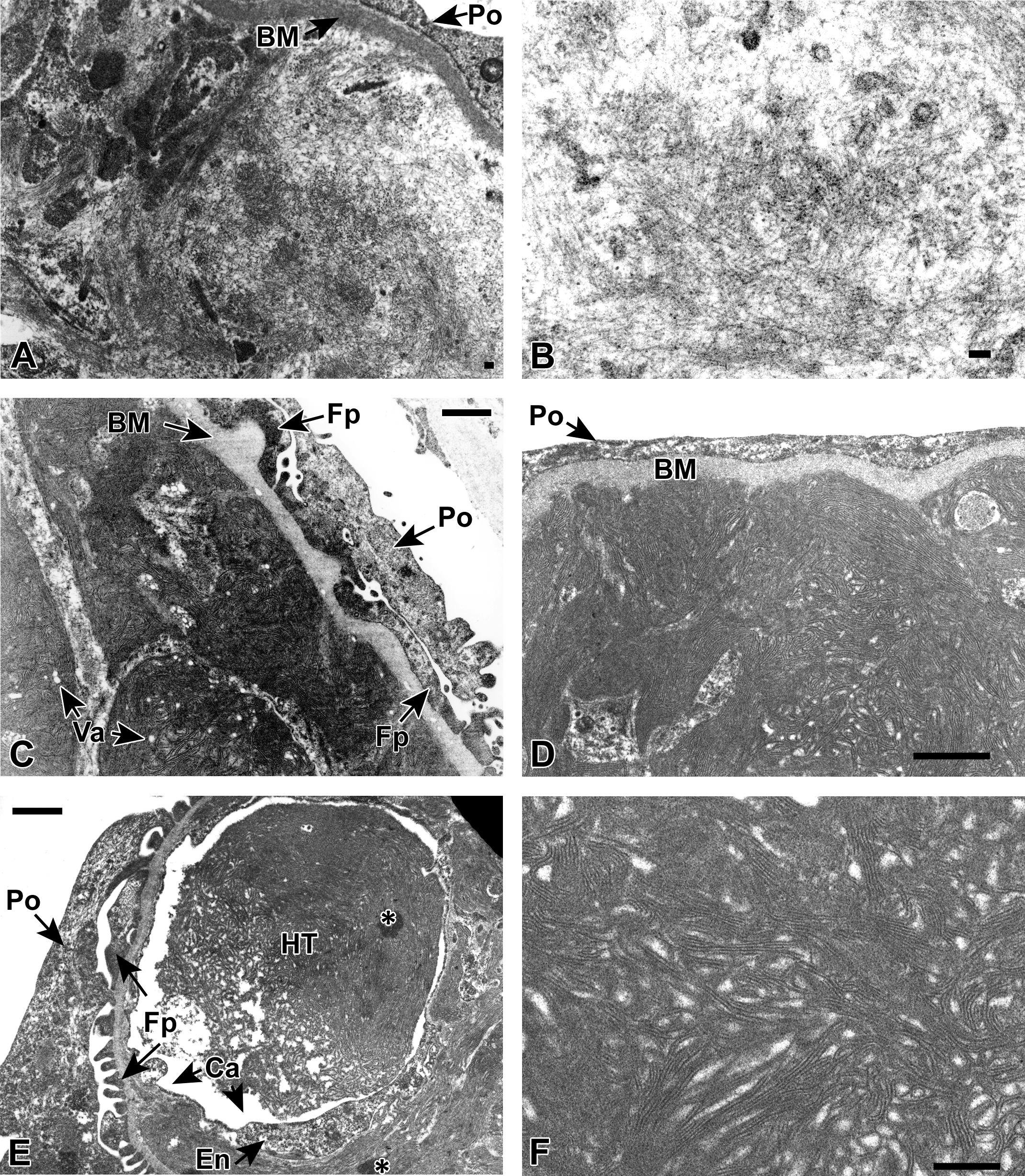
Transmission electron microscopy (TEM) of an affected glomerulus from an amyloid-positive control mouse at 35,500× (A, scale bar = 0.1 µm) and 70,300× (B, scale bar = 0.1 µm) reveals the typical ultrastructural appearance of amyloid fibrils within the glomerulus. The fibrils are slender (5 to 10 nm in diameter), relatively straight, nonbranching, randomly arranged, and occasionally bundled. In panel A, the deposit is extensive and abuts the basement membrane (BM) underlying a podocyte (Po) that has lost all foot processes (Fps) (effacement). TEM of a spontaneous hyaline glomerulopathy (HG) mouse at 22,000× (C, scale bar = 1.0 µm) and 34,000× (D, scale bar = 1.0 µm). The deposited material is composed of long, nonbranching curvilinear fibrils approximately 15 nm in diameter often forming whorls or fingerprint patterns. The deposited material was predominantly expanding the mesangium and subendothelial regions and often obliterated most normal glomerular structures with the exception of the glomerular loop BM. The overlying podocytes (Po) often have blunting, fusion, or loss of Fps. Small, circular, smooth lined vacuoles (Va) are present within the HG deposits. TEM of a spontaneous HG mouse at 22,000× (E, scale bar = 1.0 µm) and 56,700× (F, scale bar = 1.0 µm). Often, moderately and markedly affected spontaneous HG mice had discrete deposits, or hyaline thrombi (E, HT), that appeared to be within an endothelial cell-lined (E, En) capillary (E, Ca). The Po and Fps in panel E are relatively normal. Artifact related to tissue processing is indicated by the asterisks (*) in panel E.
The ultrastructural characteristics of the deposits seen in the spontaneous HG mice in which the deposits were considered to be of mild, moderate, or marked severity were all similar in appearance. The deposits were predominantly subendothelial and mesangial. In the moderately and markedly affected mice, the normal glomerular structure was severely distorted, commonly with obliteration of capillary lumens and a lack of conspicuous normal glomerular features, although the BM could typically be distinguished. The deposits were generally well circumscribed, variably sized although often extensive, and composed of moderately electron dense curvilinear, nonbranching, closely packed fibrils often organized into characteristic whorls or fingerprint patterns (Figure 9C–F) as previously described (Adams et al. 2011; Frazier et al. 2014; Wojcinski, Albassam, and Smith 1991). The fibrils were, on average, approximately 15 nm in diameter with an average intrafibril distance of 25 nm. The deposits were occasionally associated with entrapped, variably sized, smooth-margined vacuoles (Figure 9C, “Va”), also as previously described (Adams et al. 2011), that could be entrapped organelles. The hyaline thrombi seen with light microscopy correlated to well-circumscribed deposits (Figure 9E, “HT”) entirely contained within the capillary lumen (Figure 9E, “Ca”). There was also considerable podocyte (Po) foot process fusion and effacement in the evaluated glomeruli, as seen in panels 9C and D.
The ultrastructural characteristics of the deposits seen in the minimally affected spontaneous HG mouse were distinct from the 3 other spontaneous HG mice described above. The predominant ultrastructural alteration was the presence of numerous variably sized, often knob-shaped thickenings of the BM (Figure 5B), which likely correlated to the small protrusions evident on UV fluorescence (Figure 5A). The composition of the thickenings was indistinguishable from the composition of the adjacent unaffected BM, and the thickening always extended toward the epithelial (podocyte) rather than endothelial side of the membrane. There was minimal multifocal podocyte foot process fusion and effacement throughout the evaluated glomeruli.
Additional Mice
An additional 21 mice were evaluated for use as either amyloid-positive controls or potential HG cases. All animals, except one, were not selected predominantly based upon poor tissue preservation or lack of tissue availability. However, 1 mouse had mixed glomerular deposits, and this mouse was chosen for additional evaluation (Figure 10).
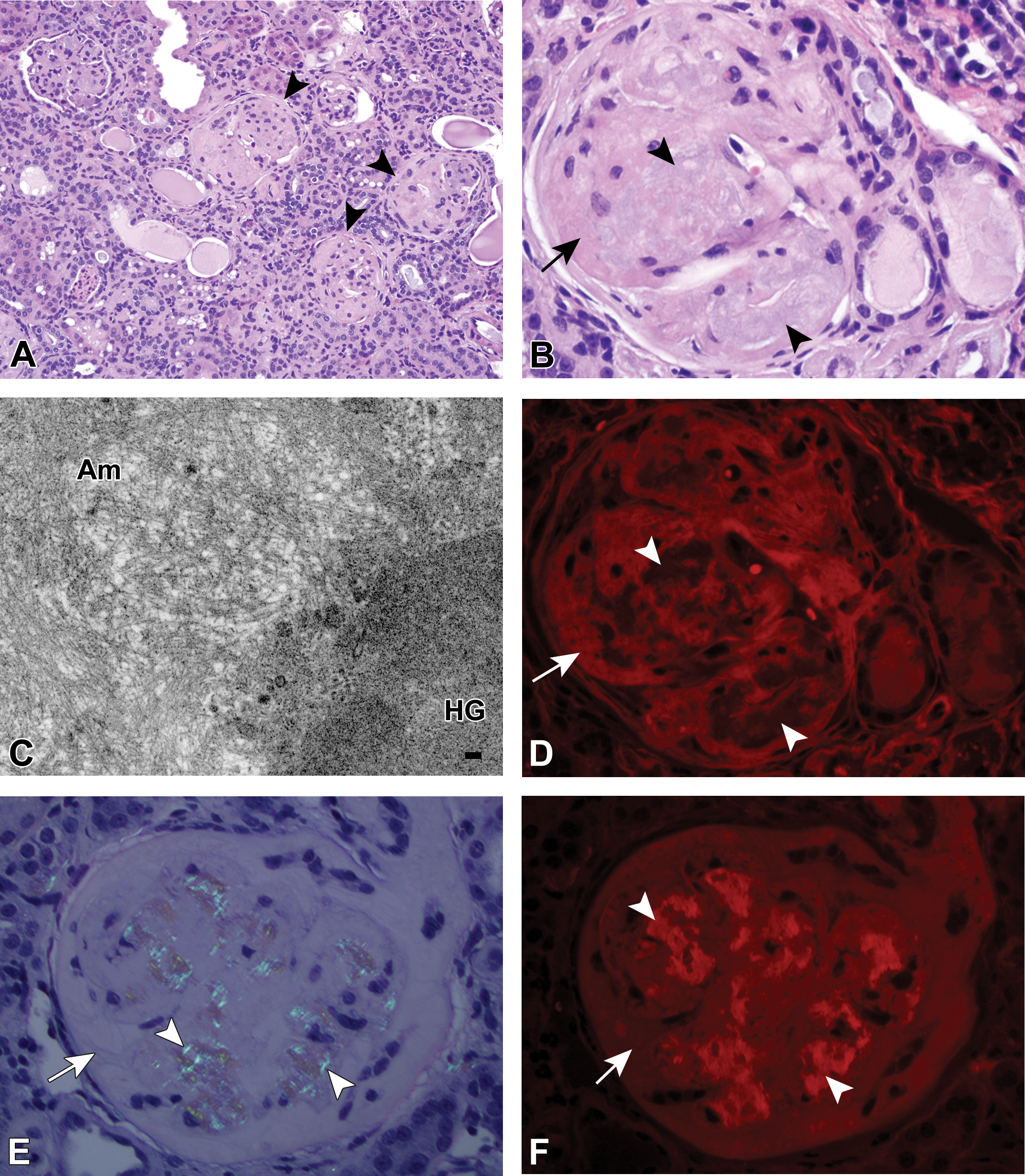
A single mouse out of the 75 in total evaluated for potential inclusion in this study had 2 admixed but distinct renal glomerular deposits. On routine hematoxylin and eosin (H&E) staining at low (A) and high magnification (B) the renal glomerular deposits were variably amphophilic and eosinophilic and ranged from hypercellular to paucicellular. There was more synechiation and periglomerular reaction (panel A, arrowheads) than seen with the majority of the mice with hyaline glomerulopathy (HG); however, there were also a fair number of glomeruli in which there was considerable expansion of the Bowman’s space (not in image), which was not typical of mice with renal amyloidosis. By ultrastructural analysis (C; 35,500×), the relatively loosely and haphazardly arranged amyloid deposits (Am) in the upper left and central portion of the figure were easily distinguished from the second, densely granular deposit (HG) in the lower right corner of the panel, scale bar = 0.1 µm. Although this deposit did not have the curvilinear fibrils seen with many of the other spontaneous HG mice, due to the histochemical similarities of this deposit with the other HG deposits, it was considered to fall within the HG spectrum. The eosinophilic deposits seen in the high-magnification H&E image (panel B, arrow) exhibited autofluorescence under ultraviolet (UV) evaluation (panel D, arrow, Texas Red [TR] filter set), whereas the amphophilic deposits (panel B, arrowheads) did not exhibit autofluorescence with this stain (panel D, arrowheads; TR filter set). With Congo red (CR) staining (E and F), the renal glomerular deposits had regions consistent with amyloid exhibiting pinkish-red staining (not shown), and the CR-positive deposits also exhibited birefringence on polarization (panel E, arrowheads) and autofluorescence with UV evaluation (panel F, arrowhead; TR filter set). The HG deposits did not exhibit birefringence upon polarization of the CR-stained section (panel E, arrow) and were less autofluorescent than the amyloid deposits on CR fluorescence (panel F, arrow).
In this mouse, the deposits were consistent with both HG and amyloid with H&E staining (Figure 10A and B). Ultrastructural analysis confirmed the presence of amyloid fibrils (Figure 10C, “Am”) and a second deposit composed of electron dense granular material (Figure 10C, “HG”), consistent with HG. The nonamyloid deposits were autofluorescent by UV evaluation of H&E-stained sections (Figure 10D, arrow) and the amyloid deposits did not autofluoresce. CR-stained amyloid deposits exhibited birefringence with polarization (Figure 10E, arrowheads) and autofluorescence with UV evaluation (Figure 10F, arrowheads).
Discussion
Recently, HG was identified as a unique test article–related pathologic change in both male and female B6C3F1 mice and F344 rats in the NTP 2-year (105 week) carcinogenicity study of pulegone (Adams et al. 2011; NTP 2011). HG has some similarity on routine H&E evaluation for what has been reported as glomerular amyloidosis, highlighting the potential misdiagnosis of HG for amyloidosis. We evaluated kidneys from 41 mice with a prior diagnosis of renal-only amyloidosis and compared them to 2 positive HG controls selected from the pulegone study and 7 amyloid-positive controls selected from the NTP database. As amyloid is well characterized and has distinct staining/birefringence with CR staining, 7 positive controls were deemed sufficient for our purposes.
Surprisingly, the renal glomerular deposits in the 41 B6C3F1 mice selected as potential HG mice from the NTP archives were all histochemically and ultrastructurally inconsistent with amyloid and instead were consistent with HG. Importantly, these mice only had glomerular deposits previously diagnosed as amyloid and did not have amyloid diagnosed in other tissues.
Although H&E staining of amyloid deposits in rodents is classically described as eosinophilic, in this review, it was amphophilic, a characteristic that was confirmed by performing the staining at 2 different histology labs by different personnel (Charles River Laboratories, Inc., Durham, NC, and National Institute of Environmental Health Sciences Histology Core Laboratory, Research Triangle Park, NC). In addition, the amyloid deposits on an archived H&E-stained slide from a more recent NTP study were amphophilic (not shown). Upon reevaluation of the original H&E-stained slides, it was evident that staining intensity and/or coloration varied greatly and that there was an overall paucity of hematoxylin staining when compared to the more recently processed tissues. Due to the age of some of the specimens, fading of the stain may be a factor, or the variation in appearance may reflect alterations in staining protocols over the years. Amyloid deposits on older H&E-stained slides tend to be a paler orange-pink than the adjacent renal tubule epithelial cells, which provide an excellent internal control for eosinophilia.
The examples of renal amyloidosis evaluated in this study were from 2-year carcinogenicity studies and were all fairly advanced. Due to this lack of range in the extent of the amyloid deposition in the amyloid-positive control mice, a direct comparison of glomerular amyloidosis and HG at all severities was not possible. It is possible that differentiating less advanced cases of amyloid from HG may be problematic if evaluation of H&E-stained slides is the sole method used. That being said, the general characteristics of HG at all severities were quite similar and differed from the amyloid-positive controls used during this study.
Upon comparison of the findings seen by light microscopy evaluation, the distribution of the amyloid and HG deposits within the glomerulus were fairly distinct. In amyloid-affected glomeruli, the deposits were typically diffuse, affecting over 75% of the glomerular capillary loops and approximately even in the degree of deposition over the affected regions. No significant increase in glomerular cellularity was noted, and nuclei were, in general, evenly distributed throughout each glomerulus. In the minimally and mildly affected HG mice, the deposits were diffuse and relatively evenly distributed throughout the glomerulus. In a few mildly affected HG mice, there were small, distinct, round to ovoid intracapillary deposits or hyaline thrombi, but these deposits were more numerous in moderately and markedly affected mice. These “thrombi” appeared to be inspissated material within the glomerular capillary loops and were consistent with what was observed in the affected kidneys in the pulegone study (NTP 2011). In the more severely affected glomeruli, the accumulation of this material seemed to obliterate the capillary loops. The irregular distribution and variance in size of these distinct deposits in the moderately and markedly affected mice often resulted in distortion of the glomeruli. Also, in the more severely affected HG mice, there were regions that appeared hypercellular and those that appeared paucicellular.
In general, amyloid deposits evaluated in the 7 amyloid-positive control mice appeared to be in close apposition or adherence (synechiation) to the visceral and parietal Bowman’s epithelium. Glomeruli in HG mice tended to have an easily visible Bowman’s space, often with minimal to mild distension. Minimal hypertrophy of Bowman’s parietal epithelium was not uncommon in HG mice, synechiation was rare, and sclerosis infrequently occurred in markedly affected mice.
Renal cortical interstitial deposition of amyloid within the kidney was not a prominent feature of most of the mice evaluated; however, greater than 50% had evidence of renal papillary necrosis secondary to marked medullary amyloid deposition. There was no evidence of papillary necrosis in any HG mice.
The mice with confirmed amyloid glomerular deposits had amyloid in at least 1 additional organ such as liver, spleen, adrenal, stomach, or intestine, which suggests that amyloidosis may generally be a multiorgan disease and that if renal-only amyloid does exist, it might be a rare finding. The mouse removed as an amyloid-positive control had original diagnoses of amyloid deposition in the kidney and thymus, and the nonrenal deposits in this animal were located solely within vessel walls. The results of this study indicate that material histochemically similar to that seen in the renal glomeruli of HG-affected mice can be found as subintimal deposits of vessels within multiple organs including the spleen, ovaries, uterus, testes, and kidneys. However, an exhaustive search for these deposits in other tissues from mice affected with HG was not performed.
Staining with both PAS and CR provided the ability to discriminate amyloid and HG deposits. Amyloid deposits were diffusely pink with PAS staining, whereas HG deposits were bright pink to magenta. Conversely, HG deposits did not stain pinkish-red with CR, whereas amyloid deposits were pinkish-red in CR-stained sections under light microscopy conditions. Importantly, the sections utilized for CR staining should be 8 μm rather than 5 μm, as more consistent amyloid staining was achieved with 8-μm sections in this study. In addition, amyloid deposits in the 8-μm sections exhibited birefringence under polarizing conditions, and hyaline deposits did not. Although there was some difference in the bluish coloration of the amyloid and HG deposits on MT staining, this was not a reliable stain to easily distinguish the 2 glomerular deposits.
UV evaluation of H&E-stained slides provided excellent discrimination of amyloid and HG deposits as H&E-stained amyloid deposits did not autofluoresce on UV illumination with either the TR or FITC filters whereas HG deposits did autofluoresce. Evaluation of UV illuminated CR-stained slides did not allow for discrimination of the 2 glomerular deposits as both HG and amyloid deposits exhibited autofluorescence. UV evaluation, particularly using the FITC filter, allowed for fine detail evaluation such as the presence of small nodular “protrusions” on the glomerular capillary BM and phagocytized autofluorescent material. The former could occasionally be found with PAS staining and the latter could be distinguished by the unique bright orange-red coloration with MT staining. For most routine rodent studies, additional stains may not be necessary to distinguish between amyloid and HG deposits if a UV light source is available and if the presence or absence of deposits in other organs is considered.
Ultrastructural analysis of the deposits via TEM easily distinguished amyloid and nonamyloid deposits, although this analysis is not practical in every animal with glomerular deposits due to costs involved. However, if a treatment effect is noted, then TEM on a subset of affected animals is recommended, even if only formalin-fixed tissues are available.
The ultrastructural appearances of the spontaneous HG deposits were not all alike, but all were considered to be within a diagnostic spectrum for HG (Adams et al. 2011; NTP 2011; Wojcinski, Albassam, and Smith 1991). In the minimally affected mouse evaluated ultrastructurally, the changes were almost exclusively composed of irregular BM thickenings, similar to those that had been reported in a pulegone-treated mouse study (Adams et al. 2011; NTP 2011). Another ultrastructural appearance of an HG deposit in this review was the granular deposit that occurred in the mouse with the mixed deposits, as was seen in pulegone-treated animals (Adams et al. 2011; NTP 2011).
The glomerular deposits seen in a majority of mice in this study were morphologically and ultrastructurally consistent with the deposits reported as a spontaneous lesion in B6C3F1 mice (Wojcinski, Albassam, and Smith 1991), reported to be associated with pulegone administration in B6C3F1 mice (Adams et al. 2011), and recently reported to be associated with administration of a second-generation antisense oligonucleotide in CD-1 mice (Frazier et al. 2014). The Wojcinski, Albassam, and Smith article indicates that the intraglomerular deposits were positive with a polyclonal antimouse immunoglobulin (IgG-IgM-IgA) cocktail (1991) and the Frazier et al. paper indicates that the deposits were positive with mouse IgG/IgM (2014). Renal glomerular deposits were positive multifocally for IgM, IgG, and IgA and negative for C3 in the pulegone study (Adams et al. 2011); however, based upon the multifocal distribution of the immunoreactivity and lack of reactivity in the regions corresponding to the eosinophilic deposits on H&E staining, it was speculated that the immunoreactivity may have been due to the immunoglobulins within trapped serum. For this reason, staining with immunoglobulins was not pursued in this retrospective study.
In our review, there was 1 animal that had mixed renal glomerular deposits, and the Frazier et al. paper reported the affected mice on their study also had mixed deposits (2014). In both studies, 1 deposit was histochemically and ultrastructurally consistent with amyloid, and the 2nd deposit had histochemical characteristics of HG. Although the nonamyloid renal deposit in the mixed deposit mouse in our review was ultrastructurally inconsistent with the curvilinear fibrils originally described for HG (Frazier et al. 2014; Wojcinski, Albassam, and Smith 1991), the deposits were similar to the granular deposits seen in a mildly affected mouse from the pulegone chronic study (Adams et al. 2011; NTP 2011). Similarly, the minimally affected spontaneous HG animal in this review did not have curvilinear fibrils, but instead had multifocal BM thickening, which was also described in the same mildly affected pulegone-treated mouse (Adams et al. 2011; NTP 2011). These different ultrastructural presentations, distinct from amyloid, are all thought to fall within the HG spectrum. Only 1 animal with mixed deposits was identified in this study; however, as we focused on animals with renal-only lesions, there was selection bias against animals that would have had systemic amyloidosis.
There are a small number of additional publications describing renal glomerular lesions in mice with at least some similarity to spontaneous HG in B6C3F1 mice. These publications include reports of spontaneous lesions in aging RF mice (Gude and Upton 1960, 1962), spontaneous nephritic lesions in a mutant strain of ICR mice (Ogura et al. 1989), glomerulopathy in a 33-day old ICR mouse (Shibuya et al. 1990), and young ddY mice with tubulo-fibrillary deposits (Kouchi et al. 2011).
Gude and Upton (1960, 1962) reported on glomerulosclerosis that occurred as a spontaneous lesion in aging RF mice. The lesion was reported to be a spontaneous renal glomerular hyaline degeneration which had no definitive connection to amyloidosis (Gude and Upton 1960). Further histopathologic and histochemical description of this finding by the same authors revealed similarities to the spontaneous HG lesions seen in B6C3F1 mice (Gude and Upton 1962). Gude and Upton (1962) reported a progressive thickening of the capillary wall, followed by accumulation of hyaline material within the mesangium, ultimately resulting in some instances in complete sclerosis. The accumulations were reported to be red with PAS staining (PAS+), to have no color change with CR, and were blue with MT (Gude and Upton 1962).
Ogura et al. reported lesions consisting of thickened capillary loop BMs with irregular “spike-like” protrusions and some mesangial enlargement without cellular proliferation (1989). No CR or TEM was performed on this study; however, the material expanding the mesangium was reported to be PAS positive (Ogura et al. 1989). There was granular staining with antimouse IgA, IgG, and IgM, with overlapping patterns to these 3 antisera, and staining with antimouse C3 was faint and only present in the glomeruli of severely affected mice (Ogura et al. 1989).
Shibuya et al. reported a single case of glomerular hyalinization that occurred in a young, 33-day old ICR mouse and was characterized by PAS positive, CR negative deposits expanding the capillary BMs and mesangium (1990). The ultrastructural description of the deposits in this mouse differed from what was seen with spontaneous HG in our study. The ICR mouse deposits were described as amorphous material within the capillary loop BMs and subendothelial areas as well as the mesangium subepithelial regions of Bowman’s capsule and also in Bowman’s space with fusion and flattening of the podocytes. These deposits were not immunoreactive with antimouse IgG, IgM, or IgA (Shibuya et al. 1990).
Kouchi et al. reported the presence of tubulo-fibrillary deposits in 2 young female 14-week old, ddY mice resulting in HG. These deposits were PAS positive, CR negative, red with MT staining, and were strongly immunoreactive with antimouse IgG, IgM, and IgA and less immunoreactive with antimouse C3. Ultrastructurally, these deposits were composed both of microtubular structures (80 to 100 nm in diameter) and fibrillary (9 to 16 nm in diameter) structures. The fibrillary structures were ultrastructurally consistent with what was seen in several mice in our study as well as by others (Adams et al. 2011; Frazier et al. 2014; Wojcinski, Albassam, and Smith 1991).
Anecdotally, mouse glomerular amyloid often does not stain CR positive, and this may, along with their morphological similarities on H&E staining, result in a misdiagnosis of HG as amyloidosis. These 2 lesions may appear indistinguishable by H&E staining without prior knowledge of the subtle morphological and staining differences for each, or without a side-by-side evaluation. A comparison of histologic and histochemical characteristics of these 2 lesions is summarized in Table 3.
Comparison of Typical Features of Renal Glomerular Amyloid Deposition and Renal Hyaline Glomerulopathy in B6C3F1 Mice from National Toxicology Program Chronic Studies.

Important take home points based upon the results of this retrospective study are: The spontaneous background lesion of HG in B6C3F1 mice may be misdiagnosed as amyloid. Spontaneous, age-related glomerular deposits in B6C3F1 mice are more likely due to HG than amyloid when there is no evidence of similar deposits in other tissues such as the liver, spleen, adrenal glands, or intestines. It may be possible, although difficult, to distinguish HG from advanced renal amyloidosis using H&E evaluation alone. The diagnosis is improved with the use of PAS or CR staining and/or UV evaluation. TEM, however, remains the gold standard for definitive diagnosis.
Hyaline glomerulopathy is proposed as an encompassing diagnostic term for different lesions with similar morphologic appearance that are not composed of amyloid. The use of the term “hyaline glomerulopathy” does not imply the etiology, and, in fact, the etiology of HG in the pulegone study and in the 41 mice used in this study remains undetermined.
Footnotes
Acknowledgments
The authors would like to acknowledge the efforts of the NIEHS Histology Core Laboratory and the Charles River Laboratories, Inc., North Carolina Histology Laboratory. In addition, the authors would like to acknowledge the Charles River Laboratories, Inc., North Carolina Electron Microscopy Group, particularly Dr. Robert Keys for all of his assistance.
Authors’ Contribution
Authors contributed to conception or design (SE, JH, CJ, and JM); data acquisition, analysis, or interpretation (SE, JH, CJ, and JM); and drafting the manuscript (JH). All authors critically revised the manuscript, gave final approval, and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported [in part] by the Intramural Research Program of the National Institutes of Health (NIH) and the National Institute of Environmental Health Sciences (NIEHS).
