Abstract
Chronic administration of drisapersen, a 2′-OMe phosphorothioate antisense oligonucleotide (AON) to mice and monkeys resulted in renal tubular accumulation, with secondary tubular degeneration. Glomerulopathy occurred in both species with species-specific characteristics. Glomerular lesions in mice were characterized by progressive hyaline matrix accumulation, accompanied by the presence of renal amyloid and with subsequent papillary necrosis. Early changes involved glomerular endothelial hypertrophy and degeneration, but the chronic glomerular amyloid and hyaline alterations in mice appeared to be species specific. An immune-mediated mechanism for the glomerular lesions in mice was supported by early inflammatory changes including increased expression of inflammatory cytokines and other immunomodulatory genes within the renal cortex, increased stimulation of CD68 protein, and systemic elevation of monocyte chemotactic protein 1. In contrast, kidneys from monkeys given drisapersen chronically showed less severe glomerular changes characterized by increased mesangial and inflammatory cells, endothelial cell hypertrophy, and subepithelial and membranous electron-dense deposits, with ultrastructural and immunohistochemical characteristics of complement and complement-related fragments. Lesions in monkeys resembled typical features of C3 glomerulopathy, a condition described in man and experimental animals to be linked to dysregulation of the alternative complement pathway. Thus, inflammatory/immune mechanisms appear critical to glomerular injury with species-specific sensitivities for mouse and monkey. The lower observed proinflammatory activity in humans as compared to mice and monkeys may reflect a lower risk of glomerular injury in patients receiving AON therapy.
Keywords
Introduction
Antisense oligonucleotides (AONs) are single-stranded, synthetic deoxy-, or ribonucleotide sequences designed to hybridize to specific and complimentary messenger RNA (mRNA) sequences and inhibit their expression. Currently, numerous AONs are being evaluated in clinical trials for treating cancer, inflammation (allergic, autoimmune, and other inflammatory diseases), metabolic diseases (diabetes and high cholesterol), neuromuscular disorders, or viral diseases. Newer generation AONs have benefitted from structural chemical modifications to improve their stability, potency, and bioavailability. The toxicologic target organ profiles of these AONs are relatively similar, although there may be significant quantitative differences in the expected toxicities between specific structural groups (Henry et al. 2008). The second-generation AONs, including a compound (drisapersen) in development by GlaxoSmithKline (GSK) and Prosensa as a potential treatment for Duchenne’s muscular dystrophy, have stereotypical class-related toxicity responses, including proinflammatory activity and renal toxicity that are relatively independent of sequence and may be highly correlated with the agent’s pharmacokinetic properties. Accumulation of cytoplasmic granules in epithelial cells from a range of organs and tissues is a class effect of AONs observed in all species (Henry et al. 2008). Ultrastructural studies and immunohistochemical staining have demonstrated that the granular material found in epithelial cells represents the test compound or associated material contained within endosomal or lysosomal vesicles or vacuoles (Monteith et al. 1999). Renal effects in short-term toxicity studies with second-generation AONs have been observed in association with this renal accumulation of test material in the form of basophilic granules, most often identified in the proximal tubules and only rarely in the glomeruli (Henry et al. 2008). GSK has recently completed chronic administration of drisapersen to mice and monkeys over periods of 27 and 39 weeks, respectively. This article describes additional renal pathology involving the glomeruli, which is observed in mice and monkey following chronic administration of drisapersen, including dedicated mechanistic investigations to better understand the pathogenesis and its clinical translation.
Materials and Methods
Study Descriptions
Drisapersen was administered as a subcutaneous injection for 27 weeks in male CD-1 mice followed by a 20-week (high-dose group) or 35-week (control and other dose groups) off-dose period. The doses were 0 (control), 6, 18, or 72 mg/kg/injection, and the test compound was administered subcutaneously twice a week for the first 2 weeks, then once weekly at the end of the treatment period. The vehicle was 20 mM phosphate buffer in 0.8% saline for injection. Mice from each group were killed and examined at necropsy at the end of the treatment period and after the off-dose period. Plasma samples taken in weeks 5, 13, and 26 of treatment were examined for monocyte chemotactic protein 1 (MCP-1) and interleukin 6 (IL-6) cytokine levels as well as samples for clinical pathology to assess kidney function.
Drisapersen was administered to male CD-1 mice as 10 repeated subcutaneous injections at weekly intervals at doses of 0 (control), 30, 100, and 300 mg/kg mg/kg/injection. The vehicle was 20 mM phosphate buffer in 0.8% saline for injection. Mice from each group were killed and examined at necropsy at the end of the treatment period.
Drisapersen was administered as a subcutaneous injection to male cynomolgus monkeys for 39 weeks followed by a 39-week off-dose period at doses of 0 (control), 2, 6, and 12 mg/kg/injection twice weekly for the first 2 weeks and then weekly. The vehicle was 20 mM phosphate buffer in 0.8% saline for injection. Monkeys from each group were killed and examined at necropsy at the end of the treatment period and after the off-dose period. Samples of right kidney from 6 controls, four 6 mg/kg/injection, and one 12 mg/kg/injection monkeys were used for immunohistochemistry (IHC) and electron microscopy. The animals in drisapersen-treated groups were selected on the basis of the presence of glomerulopathology observed by H&E in the original study. Plasma samples were taken on weeks 1, 5, 7, 9, 11, 15, 18, 22, 26, 31, 35, and 39 and examined for a range of inflammatory biomarkers as well as samples for clinical pathology to assess kidney function. In the off-dose period, blood sampling was performed monthly. Urinalysis was performed monthly using both standard parameters and urinary albumin levels.
All animal studies were ethically reviewed and carried out in accordance with European Directive 86/609/EEC and the GSK Policy on the Care, Welfare and Treatment of animals or after review by Institutional Animal Care and Use Committee in accordance with the GSK Policy on the Care, Welfare and Treatment of Laboratory Animals and were in accordance with the Guide for the Care and Use of Laboratory Animals.
Kidney Sampling and Preparation
Kidneys from two mouse studies and one monkey study were routinely processed in buffered formalin and embedded in paraffin, with a section of right kidney cortex bisected and half embedded in optimal cutting temperature (OCT) compound and the other half processed for electron microscopy as described subsequently. The cortex in the monkey was collected separately for electron microscopy. Formalin-fixed kidneys were embedded in paraffin wax, sectioned, and stained with hematoxylin and eosin (H&E). For the periodic acid Schiff (PAS) procedure, formalin-fixed paraffin-embedded (FFPE) sections were placed on the Sakura DRS, deparaffinized, rehydrated, and oxidized with 0.5% periodic acid and incubated in cold PAS reagent (PolyScientific R&D Corp, Bayshore, NY) for 15 min. Sections stained with Masson’s trichrome or Congo red (Sigma Aldrich, St Louis, MO) were placed on the Artisan Staining System (Dakocytomation, Carpenteria, CA), deparaffinized, rehydrated, and either oxidized (Gomori methenamine silver [GMS]) or treated with mordant (Masson’s trichrome) overnight at room temperature. Separate sections were also stained with toluidine blue to better define cytoplasmic granules. Age-matched normal multitissue controls were included in each staining run and examined to validate the procedure.
Immunohistochemistry
FFPE or OCT frozen-embedded mouse kidney sections were placed on the Ventana Discovery XT System® (Ventana Medical Systems, Inc, Tucson, AZ), deparaffinized, and incubated with primary antibody (rat anti-mouse CD68; Abcam Ltd, Cambridge, MA, 0.1 μg/mL; rabbit anti–von Willebrand’s factor [Vwf]; Dakocytomation, 1:1,000; rabbit antisynaptopodin; Sigma, 3 μg/mL or guinea pig antinephrin; Progen Biotechnik GmbH, Heidelberg, Germany, 10 μg/mL; and anti-mouse immunoglobulin (Ig) G and IgM; Vector Laboratories, Inc, Burlingame, CA) for 1 hr. Slides stained for nephrin were subjected to an avidin/biotin block prior to application of primary antibody. Slides stained for synaptopodin, nephrin, and vWF were subjected to either an enzyme (protease 1) or heat-induced epitope retrieval using an EDTA-based buffer (CC1). Sections were incubated with appropriate secondary horseradish peroxidase–conjugated antibodies and reacted with DAB CM™ (Ventana Medical Systems, Inc) chromagen before counterstaining with hemotoxylin. Age-matched normal mouse multitissue controls were included in each staining run and examined to validate the procedure.
Frozen OCT-embedded monkey kidney sections were cut at 7 μm and incubated with the following primary antibodies: C4d (LifeSpan Biosciences, Inc, Seattle, WA), C3c (Dako North America, Inc, Carpinteria, CA), IgG (Dako), and IgM (AbD Serotec, Raleigh, NC). A polymer-based chromogenic method was used for C4d, and immunofluoresence (IF) was used for the other antibodies using either direct (C3c fluorescein isothiocyanate [FITC]) or indirect methods (Alexa Fluor 488 conjugated secondary antibodies). For IF staining, 4′,6-diamidino-2-phenylindole (DAPI) was used as a nuclear counterstain.
Transmission Electron Microscopy
Formalin-fixed tissues in paraffin-embedded blocks were examined from selected control and high-dose mice from the 27-week mouse study. Blocks were melted, and the tissues were removed for deparaffinizing in xylene. After rehydration in serial alcohols and phosphate buffer, the samples were fixed in 2.5% glutaraldehyde in phosphate buffer, postfixed in 1% osmium tetroxide, stained en bloc with uranyl acetate, dehydrated, and embedded in epoxy resin. In the investigative 8-week mouse study and 39-week study in monkey, fresh kidney was harvested at necropsy. A portion of 1 kidney from selected animals, including controls, in all 3 studies was trimmed to approximately 1 mm3 pieces and fixed with 2.5% glutaraldehyde/2% formaldehyde in 0.1 M cacodylate buffer for 24 hr, postfixed with osmium tetroxide, dehydrated, and embedded in epoxy resin. Sections were cut, stained with uranyl acetate and lead citrate, and examined with either an FEI Tecnai 20 transmission electron microscope operating at 120 kV or a Hitachi 7500 transmission electron microscope operating at 80 kV. Representative digital images were captured using either a Gatan UltraScan™ 1000 (Gatan, Inc, Pleasanton, CA) or an AMT XR41™ camera (for transmission electron microscopy), Woburn, MA.
Gene Expression Analysis (TaqMan™)
Mouse kidneys were separated into 3 groups: controls, those given 300 mg/kg/injection of drug with histologic evidence of glomerular injury, and those given 300 mg/kg drug without evidence of glomerular injury by routine microscopic examination. A portion of fresh kidney from mice treated for 8 weeks was placed in OCT media. Eight 10-μm OCT sections from 6 control animals, 6 histologically affected treated animals, and 3 histologically unaffected treated were cut onto slides using an RNase/DNase-free microtome. Excess OCT was removed from the slides using a sterile razor before the tissue was scraped into a labeled, sterile 1.5-mL microcentrifuge tube. All samples were homogenized for ∼1 min in 300 μl of working lysis buffer. RNA was isolated and concentrated using the Absolutely RNA Microprep Kit and the RNA Clean & Concentrator™ (Products Division, La Jolla, CA) with quality assessment using Agilent RNA 6000 Bioanalyzer (Agilent Technologies, Inc, Santa Clara, CA) and quantity assessment using the Quant-iT™ RiboGreen® Kit. The RNA was quantified using the Quant-iT RiboGreen Kit (Invitrogen, Carlsbad, CA) and qualitated using the Agilent RNA 6000 Nano Reagents and Analyzer (Agilent Technologies). Complementary DNA (cDNA) was created from the RNA using the High-Capacity RNA-to-cDNA Kit and then was loaded onto the Taqman® Gene Signature Immune Array for gene analysis. The Taqman raw data were evaluated qualitatively using RQ Manager Software and quantitatively assessed using the Data Assist™ Software (Life Technologies, Grand Island, NY) using the comparative CT (ΔΔCT) method for calculating relative quantitation of gene expression. The Ct values for all samples were normalized to 18S, and β-actin and the fold changes were created by comparing the control to treated. TaqMan Universal PCR Master Mix (2×; Applied Biosystems, Foster City, CA) and 7900HT Real-Time Fast PCR System (Applied Biosystems) were used to perform TaqMan Analysis according to the manufacturer instructions. TaqMan was performed for each gene using 50 ng of amplified single-stranded cDNA as previously described (Dalmas et al. 2005, 2008). Negative controls included a no template control for each gene of interest. Data were evaluated using the ▵▵CT method and as described in Applied Biosystems User Bulletin 2: ABI Prism 7700 Sequence Detection System and reported as fold change relative to respective controls.
Results
In general, the results of these nonclinical studies were consistent with the results from other AON compounds and have been well described in the literature (Henry et al. 2008; Monteith and Levin 1999) and for brevity therefore will not be presented in this article except where relevant to the kidney findings.
Macroscopic Pathology and Light Microscopy
Drisapersen administration induced renal findings typical of the class of (modified) phosphorothioate AONs. These findings were evident against a class-related background of widespread treatment-related inflammatory activity typified by activated granular macrophages in multiple tissues.
Mouse
Mice given 300 mg/kg/injection drisapersen for 8 weeks had notable renal pathology. Minimal to mild basophilic granules were seen within the proximal convoluted tubules, and in a few animals, there was also minimal secondary degeneration of the tubular epithelium. These tubular changes consisted of basophilia, rare cell sloughing, and occasional dilation of tubules. Glomerular changes were only noted in 6 of the 22 animals by routine H&E staining and were characterized by slightly increased mesangial matrix, increased glomerular cellularity, occasional inflammatory cells or nuclear debris, and rare intraglomerular basophilic granules (Figure 1A–F). Basophilic granules were better visualized using toluidine blue staining as compared to routine H&E. In addition to granules within most proximal tubules corresponding to those noted with H&E, toluidine blue staining also revealed occasional dark blue-staining granules within endothelial cells, podocytes, or mesangial cells of glomeruli. The glomeruli were negative for amyloid by Congo red stains and negative for fibrosis/collagen deposition by Masson’s trichrome stains. GMS and PAS stains both demonstrated slightly thickened basement membranes in the glomeruli of a few mice. No renal effects were seen at 100 mg/kg.
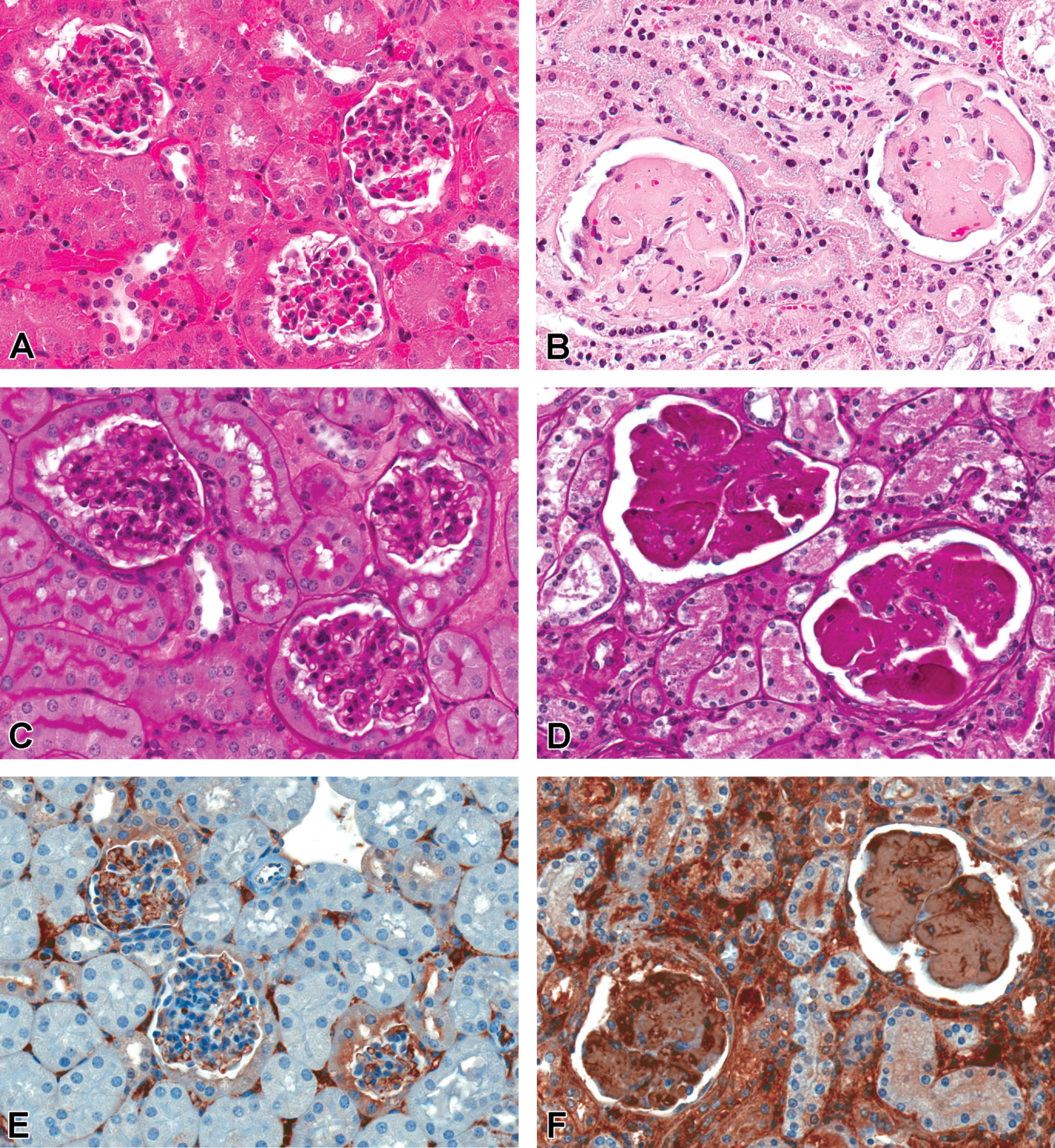
Mouse kidney. (A) H&E control mouse kidney with normal glomeruli. (B) H&E. Note marked accumulation of eosinophilic material effacing glomeruli in mouse given 72 mg/kg/injection for 27 weeks with 20-week off-treatment period. (C) PAS of control mouse kidney, with negative staining of glomeruli. (D) Note PAS-positive material in the glomeruli of a mouse given 72 mg/kg/injection for 27 weeks with 20-week off-treatment period. (E) Immunohistochemical stain for IgG/IgM in mouse control kidney, demonstrating generally negative or very light background staining in glomeruli. (F) IgG/IgM immunostaining in mouse given 72 mg/kg/injection drisapersen for 27 weeks with 8-week off-treatment period. Note strong dark staining in glomeruli, consistent with murine hyaline glomerulopathy as well as staining in interstitium. H&E = hematoxylin and eosin; Ig = immunoglobulin; PAS = periodic acid Schiff.
Mice given 72 mg/kg/injection drisapersen for 27 weeks had tubular basophilic granules and some secondary degenerative changes associated with drug accumulation as well as findings in the glomeruli and papilla. Increased glomerular mesangial matrix was characterized by diffuse, segmental to global accumulation of a homogenous, eosinophilic material in the glomerular tufts with normal or decreased glomerular cellularity. Weakly positive Congo red stains and minimally to mildly increased Masson’s trichrome and PAS staining at the 27-week time point suggested this matrix was a mixture of amyloid, fibrosis, and other hyaline material. Minimal to marked papillary necrosis was also noted in a few mice in association with amyloid deposits within the interstitium. Two mice that died or were euthanized prior to the end of 27 weeks of treatment had marked bilateral papillary necrosis, with loss of collecting ducts, thin loops of Henle, and vasa recta throughout the distal portion of the medullary papilla. These marked lesions were associated with casts, marked tubular basophilia/atrophy in the cortex, minimal single-cell necrosis of tubular cells and slight tubular dilation (ascending necrosis from functional nephron loss), and the presence of tubules lined by low cuboidal epithelium with cytoplasmic basophilia, nuclear crowding, and occasional mitoses (regeneration and cellular repair processes). An increased incidence of tubular cysts was considered a consequence of the chronic degenerative changes. Basophilic granules were also noted in the proximal tubules of most mice given 18 mg/kg/injection, but there was no evidence of degenerative tubular changes or glomerular lesions in these kidneys. Activated granular macrophages were found within multiple organs of mice given ≥18 mg/kg/injection, including the kidney. No drug-related renal changes were noted (including no basophilic granules) in mice given 6 mg/kg/injection.
At the end of the 20-week off-dose period, the glomerular changes in mice given 72 mg/kg/injection for 27 weeks were more severe than seen in those killed at the end of the treatment period, with mild to marked matrix deposition and loss of cellularity. These changes were associated with moderate to marked papillary edema and necrosis, interstitial amyloidosis, and tubular degeneration. Tubular basophilic granules were still noted at the end of the off-dose period and a few contained activated granular macrophages. Tubular basophilic granules were rare at the end of 35 weeks’ off-treatment period in mice given 18 mg/kg/injection but treatment-related changes in the glomeruli (increased matrix) were noted with a higher incidence (6 of 10 vs. 3 of 10) and severity as compared to controls and similar staining characteristics to animals given 72 mg/kg/injection. In contrast, mice given 6 mg/kg/injection and sacrificed after 35 weeks’ off-treatment period had glomerular changes of similar incidence (3 of 10), severity, and character (minimally thickened membranes in tufts only) as controls and were therefore considered most likely spontaneous, age-related glomerular membrane changes. H&E and toluidine blue staining of the slides from the most severely affected mice demonstrated marked global diffuse glomerular changes including enlarged acellular glomeruli filled with homogenous material that stained eosinophilic with H&E or PAS stains and light blue with Toluidine blue (Figure 1A and B). Some small areas of light pink congophilic material were noted in glomeruli and in the interstitium with Congo red stains, but green birefringence was largely negative under polarized light, suggesting amyloid is not the principal accumulating component. Seven mice given 72 mg/kg/injection died during the off-dose period, with most having moderate to severe kidney lesions and/or amyloid-related papillary necrosis, which were considered the cause of death. As noted previously, minimal amyloid was also noted in the kidneys of a few control mice at the end of the off-dose period with special stains in conjunction with age-related glomerular changes.
Monkey
In addition to the presence of basophilic granules in tubular epithelial cells (without secondary degenerative tubular changes), there was minimal to slight glomerulopathy noted in 2 of the 6 monkeys given 12 mg/kg/injection and 4 of the 8 monkeys given 6 mg/kg/injection for 39 weeks. This change was multifocal or diffuse and segmental to global. The glomeruli were enlarged and characterized by variably increased cellularity of the tufts and increased mesangium. Some basophilic granules and small numbers of neutrophils were occasionally present in the affected glomeruli. At the end of the 39-week off-treatment period, minimal thickening of the mesangium was noted in only a single monkey given 6 mg/kg/injection, with no other accompanying renal changes, suggesting at least partial reversibility of the glomerular lesion. In addition, there was a marked decrease in the incidence and severity of the basophilic granules in tubular epithelial cells, as they were noted largely confined to macrophages of the lymph nodes and rarely in macrophages in other organs.
There were no significant alterations in routine clinical chemistry parameters referable to the kidney at any time during the study, but there were significant changes in systemic proinflammatory markers as noted subsequently. Neither quantitiative nor qualitative urinalysis mean parameters were significantly altered in treated versus control monkeys for the duration of the study. At week 39, only a single animal with minimal glomerulopathy (observed at the end of the off-dose period) had increases in urinary albumin levels.
Electron Microscopy
Mouse
Ultrastructural examination of the glomeruli from mice given 300 mg/kg/injection drisapersen for 8 weeks revealed occasionally thickened and irregular endothelial cell linings, although fenestrations were maintained. Endothelial cells were hypertrophied with expanded cytoplasm and/or pyknotic nuclei. Frequently, endothelial cells, mesangial cells, and podocytes had numerous membrane-bound lysosomal vesicles containing electron-dense material. Basement membranes were mildly thickened and contained cellular debris and occasionally demonstrated longitudinal “layering” or “splitting” and/or reduplication. Rarely, the basement membrane and intercellular junctions between the endothelium and the podocytes contained electron-dense material, and electron-dense deposit (EDD) and electron-lucent deposit were also seen adjacent to these areas of altered glomerular basement membrane. No ultrastructural abnormalities were detected in the kidneys of the control mice after 8 weeks of dosing.
Ultrastructurally, the glomeruli of some mice treated with 72 mg/kg/injection drisapersen for 27 weeks were effaced by curvilinear fibrils of approximately 12 to 15 nm in diameter and approximately 200 to 800 nm in length, sometimes in layers, whorls, or fingerprint patterns, and consistent with the murine syndrome of hyaline glomerulopathy (Figure 2). These fibrils were noted along basement membranes and filled with the mesangium, separating remnant cell populations and effacing glomerular tufts. Glomeruli generally lacked cellularity, and some less affected glomeruli contained slightly thickened basement membranes and rare EDDs. In rare areas of a few glomeruli, there were masses of randomly arranged straight fibrils 7 to 10 nm wide of various lengths embedded in a granular matrix and consistent with amyloid (Figure 2C). Collagen fibers were noted rarely in glomeruli, distributed in a few collagen fibrils between mesangial cells and their associated basement membrane. Tubules contained intralysosomal inclusions within proximal epithelial cells. No findings were noted in the glomeruli of controls examined ultrastructurally.
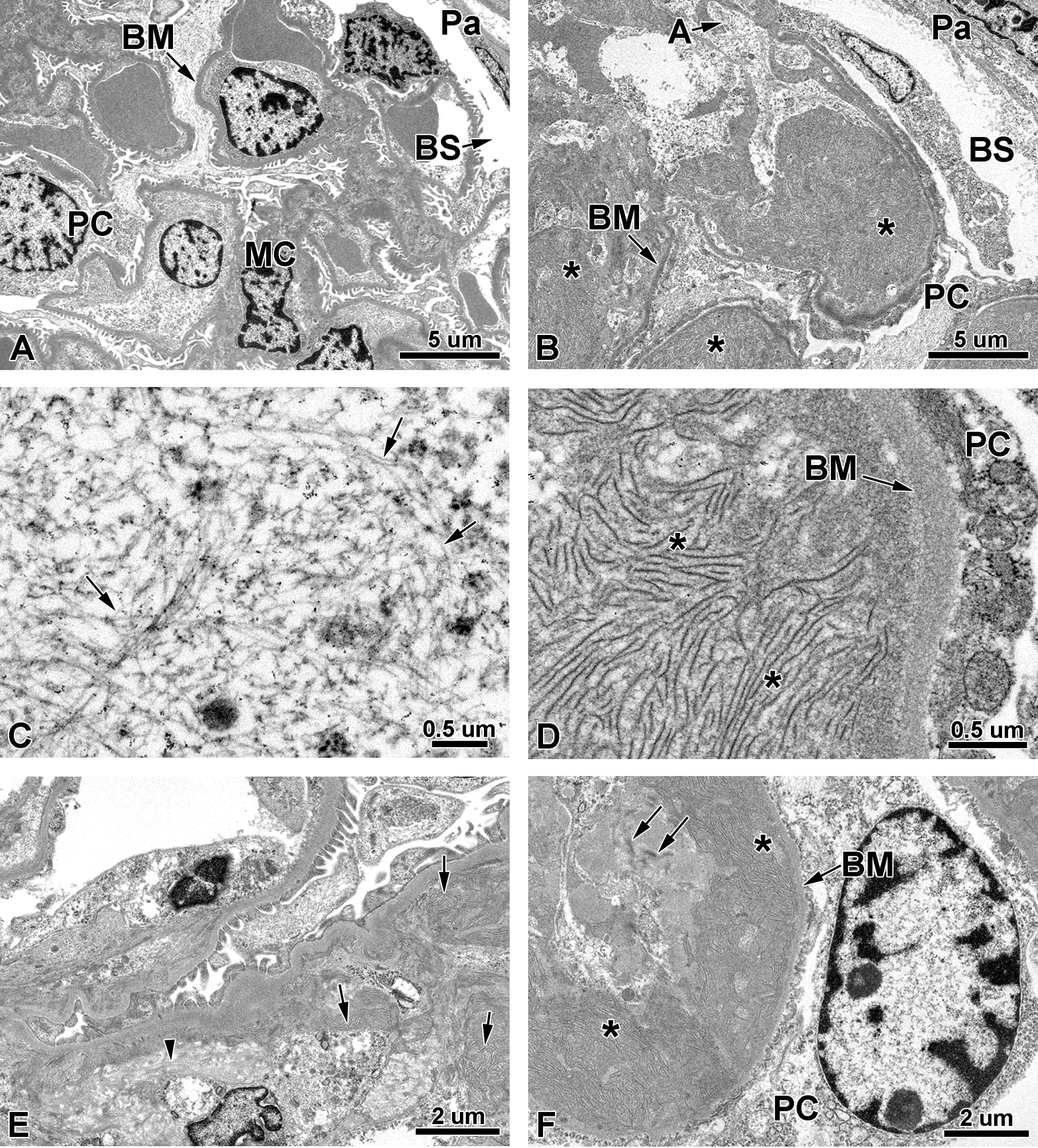
Transmission electron micrographs of glomeruli of a control mouse (A) and a mouse given 72 mg/kg/injection for 27 weeks followed by an 8-week off-treatment period (B). Note the intact basement membrane (BM) surrounding acellular areas within tufts effaced by bundles and whorls of curvilinear fibrils (*) have replaced normal architecture. Note the characteristic randomly arranged straight fibrils of amyloid measuring 7 to 10 nM in diameter within granular background observed in a treated mouse (C). Higher magnification (D) of curvilinear fibrils (*) in treated mice revealed ∼15 nM diameter lamellae characteristic of mouse hyaline glomerulopathy. Both hyaline glomerulopathy (arrows) and minimal collagen accumulation characterized by larger diameter banded fibers (arrowhead) were observed (E). (F) Electron-dense deposits and redundant or remnant membranes on the subendothelial side of the basement membrane were seen in the treated mice (arrows). A = amyloid fibrils; BM = basement membrane; BS = Bowman’s space; MC = mesangial cell; Pa = parietal epithelial cell; PC = podocyte.
Monkey
Ultrastructural examination of the kidneys from monkeys treated with drisapersen for 39 weeks demonstrated a spectrum of changes that varied slightly between animals (Figure 3A–F). There was an increased cellularity within the glomeruli characterized by an increased number of mesangial cells, with occasional infiltration of inflammatory cells including mononuclear cells (lymphocytes, macrophages, and plasma cells) and rare neutrophils, and this was accompanied by hypertrophy of the endothelial cells, and less commonly, hypertrophy of podocytes. The plump endothelial cells often contained tubuloreticular bodies and/or enlarged lysosomes. The mesangium was thickened, and there were intramembranous and subepithelial amorphous, finely granular, EDDs noted along the basement membrane, which resulted in some areas with effacement of podocytes and foot processes. Dense lysosomal inclusion bodies were occasionally noted within the cytoplasm of multiple cell types of the glomeruli. Osmiophilic lysosomal inclusion bodies were also noted in proximal tubule epithelium, typical of those associated with the basophilic granules of drug substance. Vacuolation of podocytes was rarely observed. There were no significant ultrastructural abnormalities noted in kidneys from control monkeys.
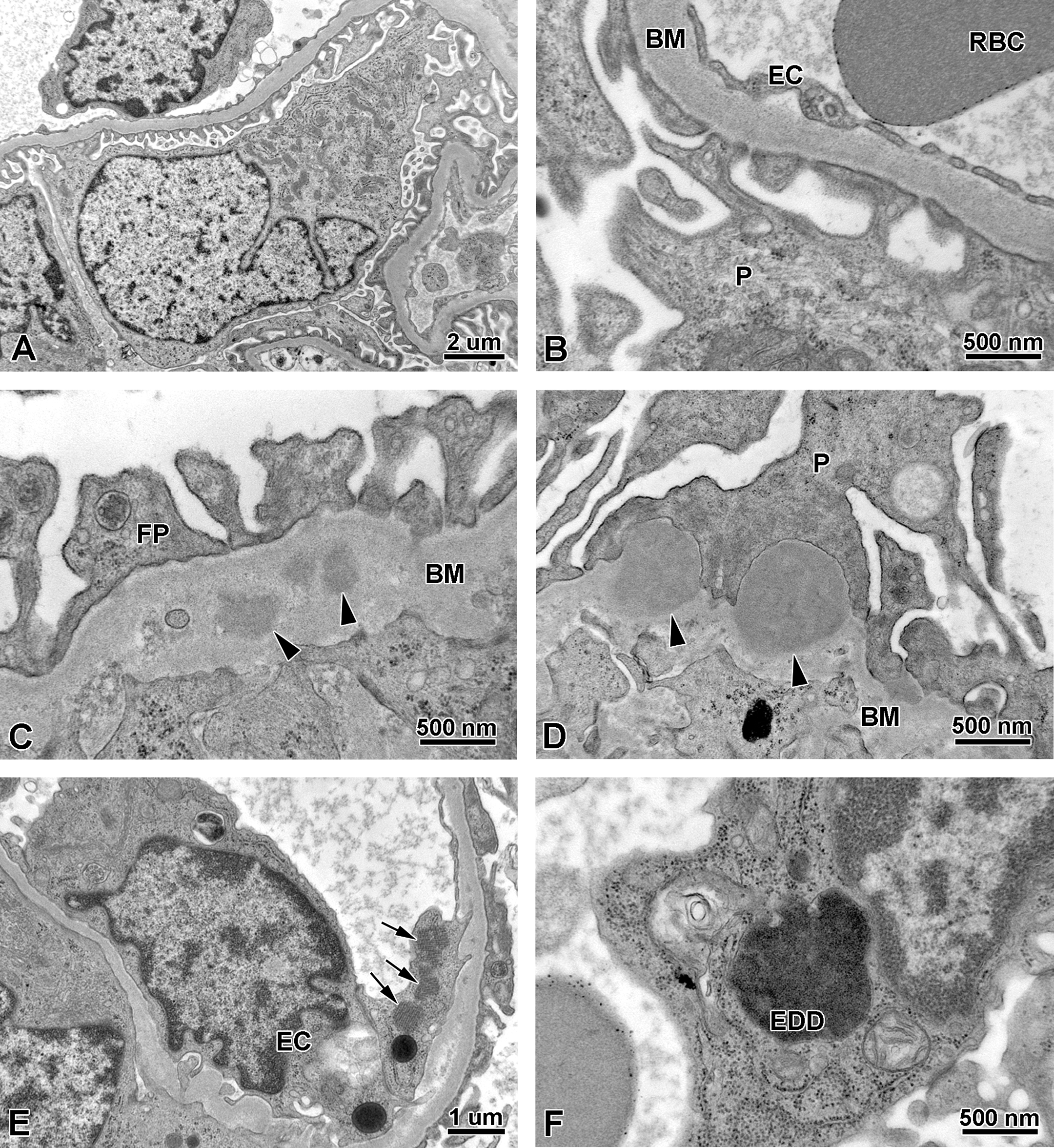
Electron microscopy of monkey kidneys in the 39-week toxicity study. (A and B) Normal glomerulus from vehicle control. (C and D) Glomerulus from monkey treated with drisapersen for 39 weeks. Note subepithelial-dense deposits (arrowheads) within and along the thickened basement membrane characteristic of immune deposition. (E and F) Glomerulus from monkey treated with drisapersen for 39 weeks. Note endothelial tubuloreticular bodies (arrows), dilated lysosomes, and an osmiophilic, membrane bound, electron-dense deposit (EDD), potentially representing drug accumulation, adjacent to the nucleus. BM = basement membrane; EC = endothelial cell; FP = foot processes (podocyte pedicels); P = podocyte; RBC = red blood cell.
Immunohistochemistry
Mouse
No differences between controls and mice given 300 mg/kg drisapersen for 8 weeks were noted for immunohistochemical stains for nephrin or synaptopodin, suggesting that podocytes were not significantly affected by the compound at this time point. Immunohistochemical stains for vWF, which stains glomerular and interstitial endothelial cells, demonstrated increased staining within glomeruli in several mice given 300 mg/kg as compared to the controls. These areas of increased staining likely corresponded to areas of endothelial hypertrophy, degeneration, or pooling of vWF protein within cytoplasmic spaces. CD68 immunohistochemical staining was markedly positive in treated mouse kidneys, including those with no histologic evidence of glomerular changes, while controls were largely negative for the same marker. Staining was most prominent in the peritubular interstitium, with only minimal staining within glomeruli. Positive CD68 immunostaining correlated with increased CD68 mRNA in the genomic data.
In mice given drisapersen for 27 weeks, IHC was strongly positive for IgG/IgM throughout the affected glomeruli of treated mice as well as within the cortical interstitium and vessels (Figure 1E and F). Control mice shared some of the interstitial and vascular staining but generally lacked staining of glomerular elements other than endothelium.
Monkey
In monkey, anti-C3c IHC revealed an increase in linear, granular-type staining within the glomeruli of test article-treated animals (Figure 4A–D). This was particularly prominent in 3 monkeys given 6 mg/kg/injection. IHC staining with anti-C4d, IgG, and IgM demonstrated no difference in glomerular staining between control and GSK2402968-treated animals
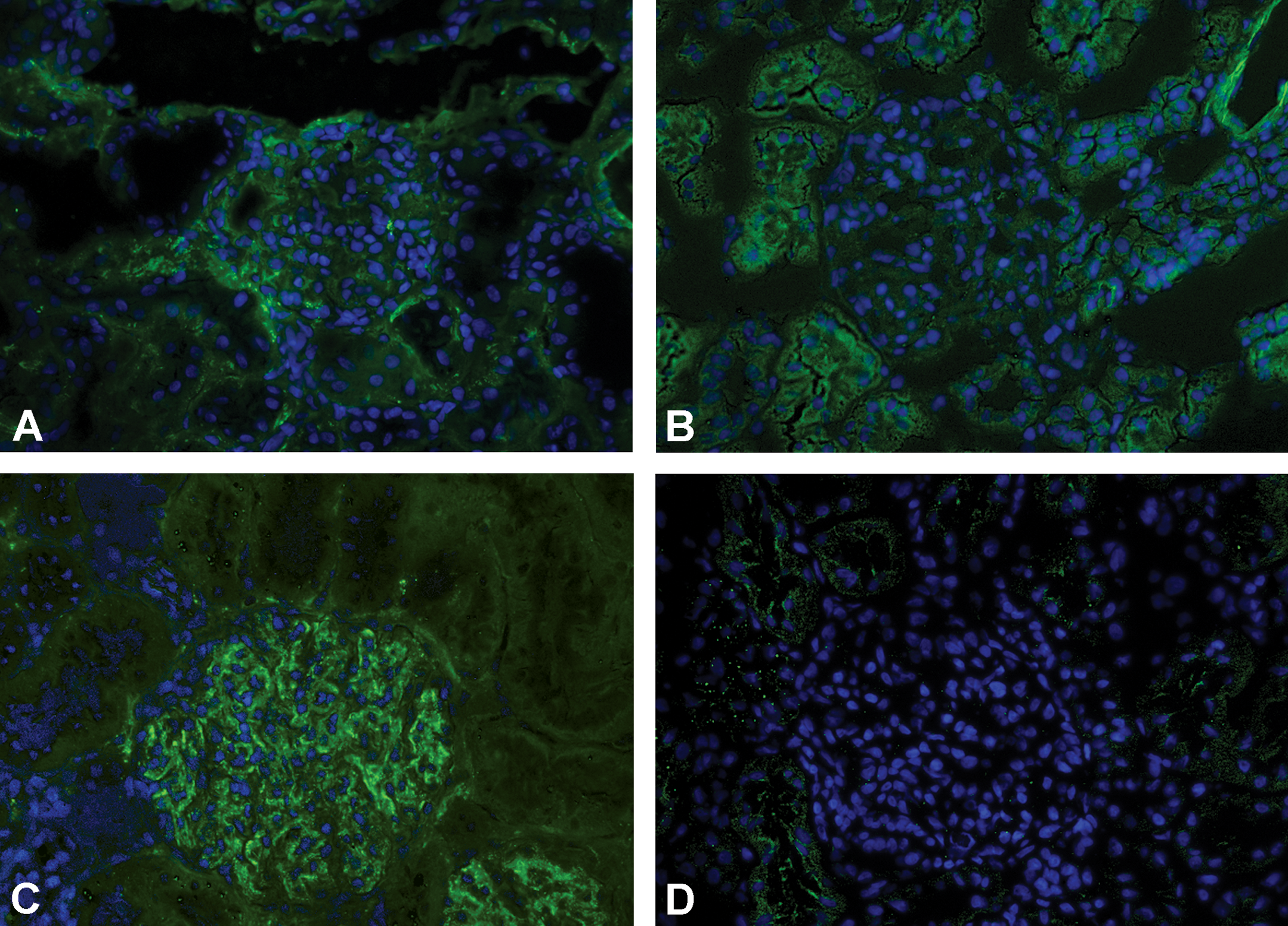
Kidney sections of monkey stained with C3C (A) or FITC-conjugated isotype control antibody (B) compared with kidney sections of monkey treated with 6 mg/kg drisapersen for 39 weeks stained with C3C (C) or FITC-conjugated isotype control antibody (D). FITC staining green, DAPI (nuclear) staining blue. Note moderate positive C3C staining only in (C). FITC = fluorescein isothiocyanate; DAPI = 4′,6-diamidino-2-phenylindole.
Gene Expression
In mice given 300 mg/kg/injection drisapersen for 8 weeks, an increase in immune response was seen in the following genes: IL-10, Ccl3, Ccl2, IL-6, IL-1a, Gzmb, Cxcl10, Cd68, IL-17, Tnf, IL-1b, Ptprc, Cd4, Nos2, Stat1, Ifng, Ccl5, Csf2, Socs1, Ctla4, Csf3, Vcam1, Csf1, C3, B2m, IL-12a, Sele, Cd8a, and Ptgs2 based on at least a 2-fold change in message when normalized to 18S and β-actin (Figure 5). The histologically unaffected (no evidence of glomerular injury) treated group followed the same trend as the histologically affected treated but with slightly less magnitude.
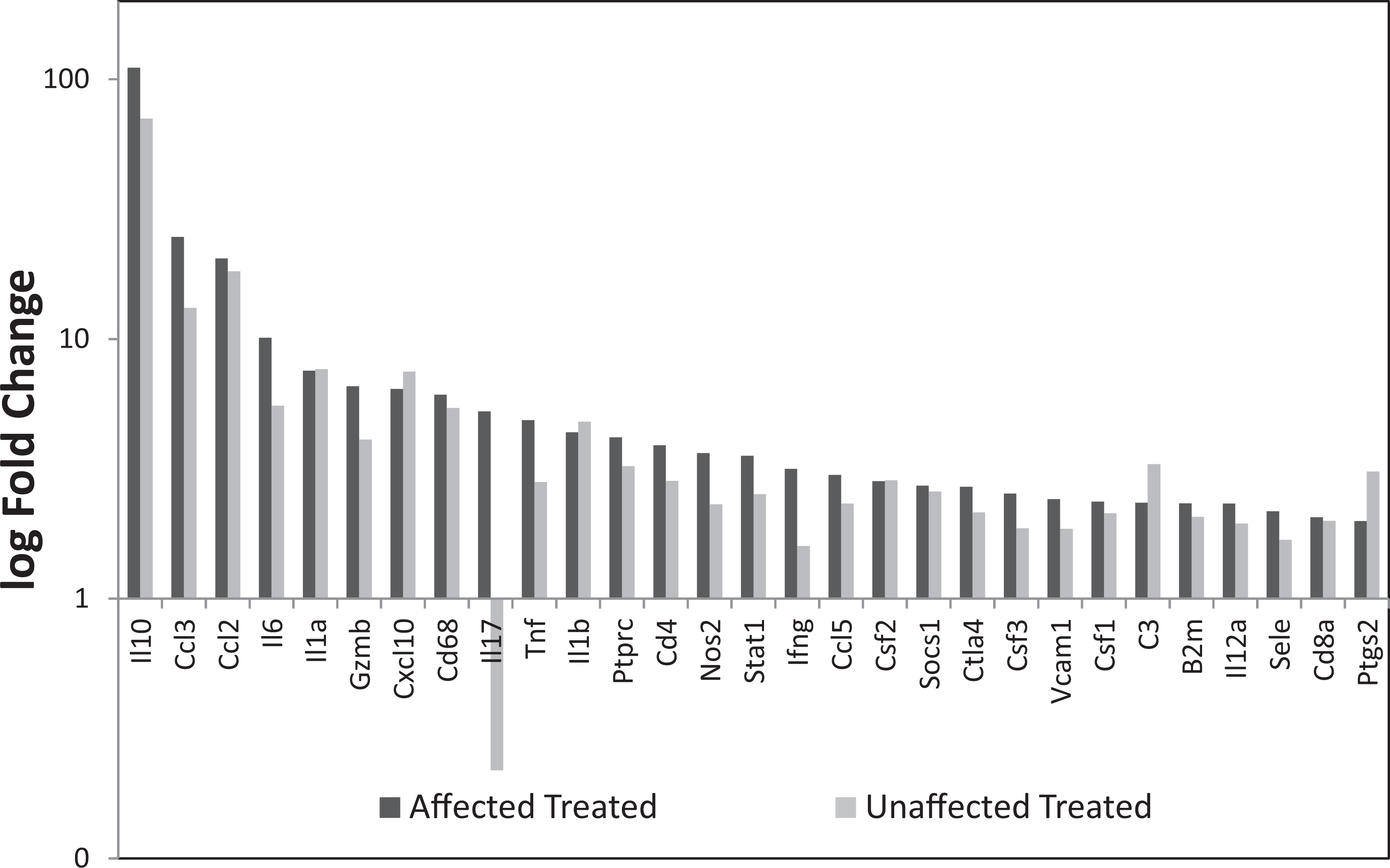
Relative mRNA levels after extraction from mouse renal cortex. Immune response observed in the affected treated and unaffected treated when normalized to 18S and β-actin when compared to controls. mRNA = messenger RNA.
Systemic Inflammatory Markers
Mice
In mice treated for 27 weeks, there was evidence of systemic inflammatory activity in the form of a dose-related increase in MCP-1 at doses of 6 mg/kg and more (Table 1). IL-6 was unaffected (data not shown). Significant increases in MCP-1 levels were noted in all samples from mice given 72 mg/kg/injection in weeks 5 to 26 at all time points. Increased MCP-1 was also noted in weeks 5 and 13 in mice given 18 mg/kg/injection, and in week 26 in mice given 6 mg/kg/injection. Levels of MCP-1 and number of animals affected increased with prolonged duration of treatment at all dose levels. By the end of the off-dose period, MCP-1 levels were below the limit of quantitation (LOQ) in all animals. Values for control animals always fell below the LOQ.
Mean MCP-1 serum levels in mice given drisapersen for 27 weeks.
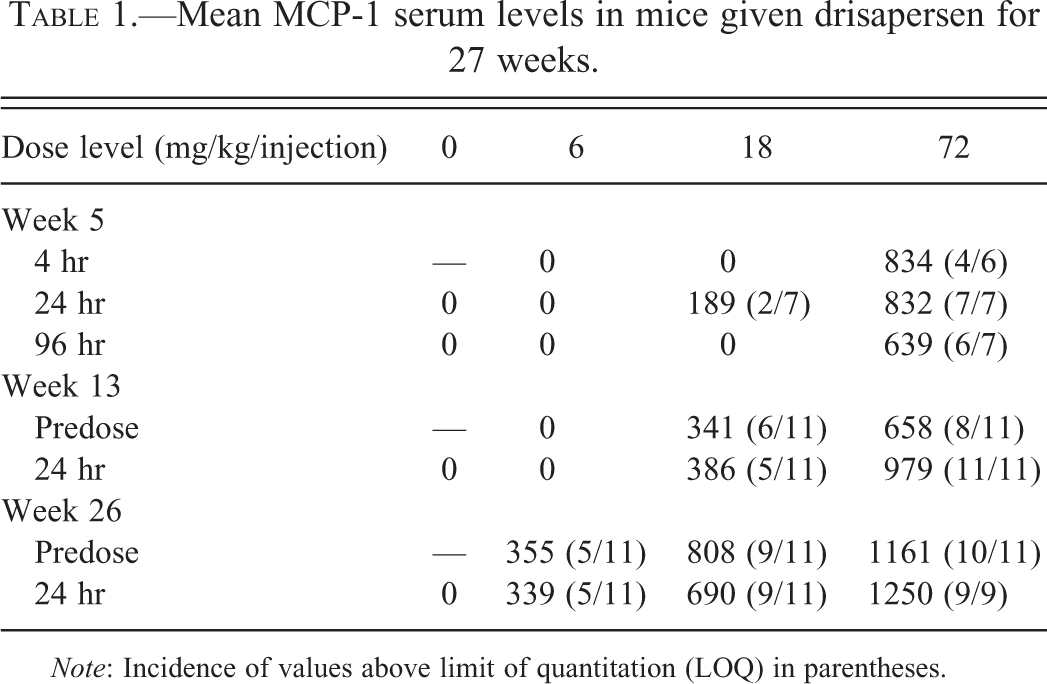
Note: Incidence of values above limit of quantitation (LOQ) in parentheses.
Monkey
In the 39-week study in monkeys, evidence of an inflammatory response was initially noted at 12 mg/kg/injection with the earliest significant changes occurring in MCP-1 at week 4 (Table 2). Mild to marked increases in MCP-1 were noted at multiple time points in all monkeys given 12 mg/kg/injection, but MCP-1 serum levels in control monkeys generally hovered near the baseline (Table 2 and Figure 6). By week 26, most monkeys given
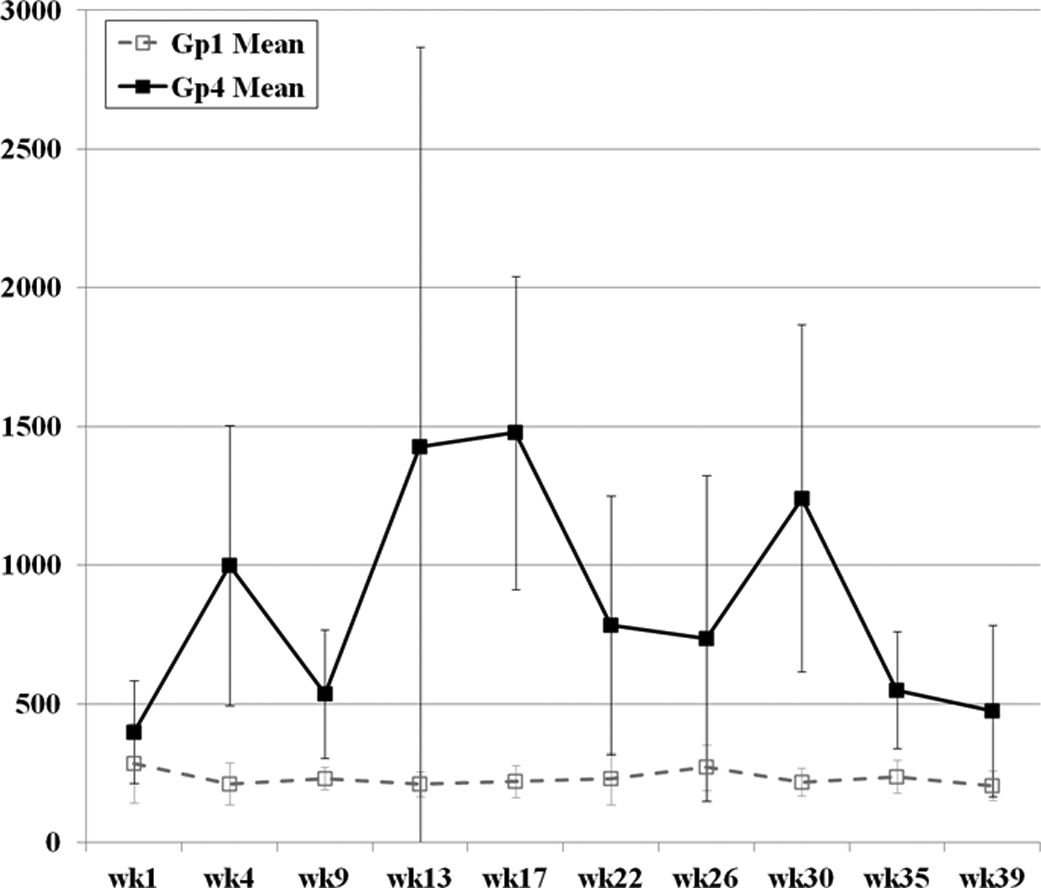
Mean serum MCP-1 values of control (group 1) and monkeys treated with 12 mg/kg/injection Drisapersen (group 4) for 39 weeks. MCP-1 = monocyte chemotactic protein 1.
Serum MCP-1 data in control monkeys and those given 12 mg/kg/injection drisapersen.
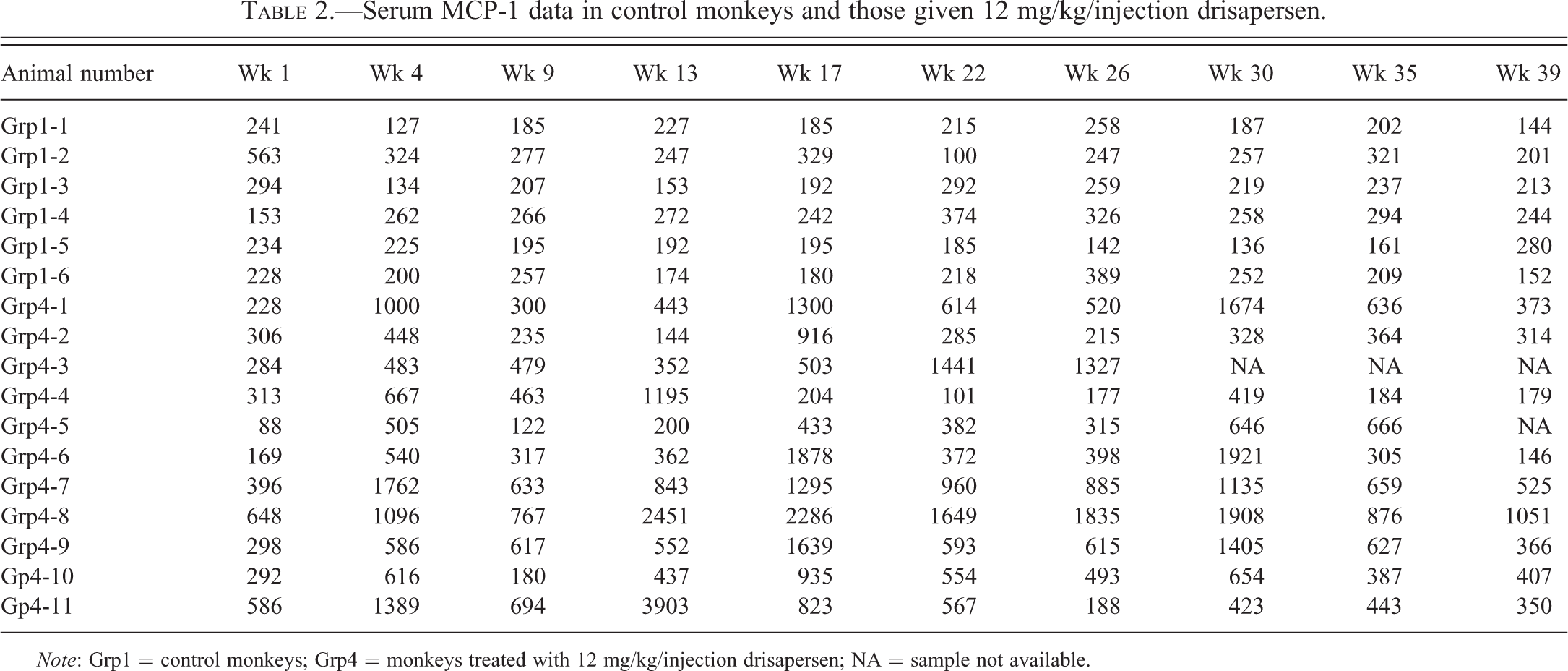
Note: Grp1 = control monkeys; Grp4 = monkeys treated with 12 mg/kg/injection drisapersen; NA = sample not available.
Discussion
Due to the overwhelming past experience of AON-related kidney toxicity being derived from subacute and subchronic studies, the descriptions and proposed pathogenesis have largely focused on tubular effects (Henry et al. 1999, 2008; Marquis and Grindel 2000; Monteith et al. 1999). AONs are primarily excreted by the kidneys and accumulate within the proximal tubule cell lysosomes, resulting in susbstantial renal tissue levels. In the toxicity studies with GSK2402968 described in this publication, lysosomal drug accumulation in the form of basophilic granules was abundant within proximal tubular epithelium of kidneys in both mice and monkeys, with slight tubular degenerative changes seen only in mice. In contrast, basophilic granules within glomeruli were not commonly identified at light microscopy in either mice or monkeys given drisapersen.
Minimal glomerular changes noted in several mice given 300 mg/kg/injection as early as 8 weeks after starting AON treatment were not associated with any evidence of matrix accumulation. Only at or after treatment of mice for 27 weeks did glomeruli demonstrate significant matrix accumulation and loss of cellularity, and these lesions progressed further during the off-dose period, indicating that matrix effects in mice are chronic, irreversible sequelae to initial glomerular injury. Glomerular lesions in monkeys given drisapersen for 39 weeks were much less severe than in the mouse, lacking the pronounced matrix accumulation and demonstrating (partial) reversibility following an off-dose period (see Figure 3 for comparison of species). Drisapersen has an elimination half-life of approximately 27 days based on the mean plasma data, and therefore it should be noted that exposure continued during the off-dose period.
Glomerular matrix accumulation in chronically treated mice was predominantly composed of fibrils of ∼15 nM diameter that are characteristic of murine hyaline glomerulopathy. The hyaline glomerulopathy was confirmed by negative Congo red stains, positive PAS, Masson’s trichrome, and strong positive IgG/IgM immunostaining. Similar fibrils and fingerprint-like patterns have been noted as a spontaneous age-related lesion in mice that are considered to be formed from Ig fragments (Frazier et al. 2012; Wojcinski, Albassam, and Smith 1991). In the spontaneous form, immunological processes in normal aging mice produce circulating antigen: Ig complexes that localize in kidneys along glomerular capillary walls and are only cleared slowly (if at all) through phagocytosis by mesangial cells (Linder, Pasternack, and Edgington 1972). There is a distinct human syndrome of immunotactoid glomerulopathy, which has also been referred to as hyaline glomerulopathy, but it is characterized by larger sized (20–30 nM) microtubular structures within the mesangium and therefore is unrelated to the syndrome in mice (Schwartz 2007b). PAS-positive hyaline fragments have also been noted in some cases of focal segmental glomerulosclerosis (FSGS) in people, but FSGS lacks other morphologic features of the mouse syndrome. Although antibody fragments are noted in human diseases such as light chain amyloidosis and Bence Jones protein glomerulopathy with myeloma, the ultrastructural morphology and staining characteristics are also quite different, and murine hyaline glomerulopathy is therefore considered a rodent-specific syndrome (Frazier et al. 2012; Wojcinski, Albassam, and Smith 1991). This is further supported by the absence of glomerular matrix accumulation in monkeys receiving chronic treatment with drisapersen.
Amyloidosis is one of the major disorders of aging mice (especially CD-1 strains), where the kidney is frequently a target organ. Although some amyloid accumulation may be expected in mice of this age, it is more likely that, like hyaline glomerulopathy, interstitial amyloidosis is a result of the ongoing, treatment-related, immune stimulation in these mice. Existing amyloid fibrils can act as a seed for further amyloid progression, and many factors including age, strain, concurrent inflammation, and amyloid/precursor protein levels in the serum can all affect fibril formation and deposition within tissues (Gise, Christ, and Bohle 1981). The combination of glomerular dysfunction and interstitial deposition of amyloidosis likely resulted in the secondary papillary changes that occurred with drisapersen and resulted in premature deaths. Interstitial amyloidosis is a well-recognized cause of papillary necrosis in mice (Frazier et al. 2012), possibly related to ischemia of the distal medulla via progressive loss of the vascular supply through occlusion of the medullary vasculature (Frazier and Seely 2013). Lysosomes are intimately linked with amyloid fibril formation and provide an environment conducive to the transition from helical to β-pleated sheet structure. Hence, there may be some connection between lysosomal accumulation of AONs and the eventual concentration of amyloidosis in the cortex and medulla. The amyloidosis and papillary necrosis seen in mice are unlikely to occur following clinical dosing as amyloid is not deposited in human kidneys in response to inflammatory activity without concurrent genetic (familial) predisposing abnormalities (Faccini, Abbot, and Paulus 1990).
Further support for an immune-based mechanism in mice is provided by marked upregulation of many immune system-related cytokines (noted in the murine renal microarray data and increased CD68 immunostaining) and the increased systemic cytokine levels in animals treated for 8 weeks. The proinflammatory potential of AONs is well recognized (Henry et al. 1999), with known quantitative differences based on the backbone structure and/or base sequence (Krieg 1998). Immunomodulatory effects involve stimulation of multiple receptors of the innate immune system, such as Toll-like receptors (TLRs), leading to stimulation of the innate immune system with resultant release of cytokines and chemokines and complement activation via the alternative pathway. Cellular patterns of TLR expression vary widely between different species, such that results of TLR stimulation preclinically may not be predictive of what will occur in humans or even in another preclinical species (Richardt-Pargmann and Vollmer 2009), and this may be an explanation for the somewhat different histomorphologic and systemic expression of inflammatory stimuli between mice and monkeys in our studies.
The initial cellular site of injury in the mouse glomerulus after 8 weeks of treatment appears to be the glomerular endothelial cell, rather than the podocyte or mesangial cell. The endocapillary endothelial cell represents a potential target for circulating cytokine-mediated injury, whereas podocytes (which appeared much less affected in mice at this early stage) are affected only secondarily via basement membrane effects. Alterations in the basal lamina include the presence of EDDs along the basement membrane. This type of change is indicative of an immune-mediated pathogenesis of glomerular injury (Jones et al. 1984; Sachs, Zhous, and Sheerin 1996) rather than reflecting direct nephrocytoxicity. The marked increase in CD68 immunostaining correlated with increased CD68 gene data in mice, demonstrating concordance of genomic and proteomic upregulation. Importantly, many of the upregulated genes including CD68 have been associated with both TLR activity and amyloid formation, suggesting that immune stimulation has both an important role in the early pathogenesis of glomerular injury in the mouse and a possible role in the tendency for the mouse to later develop progressive hyaline glomerulopathy and renal amyloidosis.
The glomerular changes in the monkey are less pronounced than in the mouse and resemble features of human syndromes of both membranous (MGN) and membranoproliferative glomerulonephritis (MPGN). MGN is characterized by intramembranous and subepithelial EDDs of complement and complement fragments and has been associated with a variety of drug-induced glomerulopathies, including those due to penicillamine, captopril, and lithium (Schwartz 2007a, 2007b). Thickened tuft walls, increased mesangial matrix, and hypercellularity are all features shared between drisapersen-treated monkeys and the human diseases. In particular, tubuloreticular structures within endothelium noted in monkey have been identified previously in human cases of idiopathic and hepatitis B-related MGN (Schwartz 2007a). Inflammatory cell infiltrates, as seen in the monkey glomeruli, are however generally absent in human MGN but are a recognized feature of human syndromes of MPGN.
The glomerular lesions in the monkey stain positively for complement C3 fragments but not for Igs or complement C4 fragments. This differential staining pattern is a common feature of C3 glomerulopathies described in man (Barbour, Pickering, and Cook 2013; Pickering and Cook 2008) and animal models of factor H deficiency. The pathogenesis is linked to dysfunction of the alternative complement pathway via interference with a key regulator protein, factor H (e.g., through genetic defects and/or autoantibodies). It is noteworthy that the proinflammatory effects of AONs in the monkey are characterized by selective activation of the alternative complement pathway, through transient inhibition of factor H (Henry et al. 1997, 2008), leading to increased circulating complement split factors and resulting in a progressive decline in complement C3 activity upon chronic treatment. The commonality in dysfunction of factor H and specific morphologic characteristics of the kidney pathology (i.e., dense deposits, selective C3 staining with no Ig staining) between the monkey and C3 glomerulopathies recognized in man and animal models of factor H deficiency makes it tempting to classify the glomerular lesions in the monkey as a C3 glomerulopathy. Monkeys are particularly sensitive to AON-induced complement activation (Kwoh 2007) and a similar direct AON-induced complement activation has not been observed in humans or other species.
Proteinuria has been occasionally noted in animals as well as in humans in clinical trials with AONs (Rao et al. 2004). Lysosomal accumulation of drug within tubules is an expected consequence of AON therapies and at high doses can induce mild, reversible tubular injury. Tubular effects and proteinuria may therefore potentially occur in patients, but it should not be assumed that glomerular injury will also accompany tubular changes when proteinuria is observed. Given the distinct mechanisms of injury between tubular degeneration and glomerular lesions associated with AON administration, the two types of renal lesions probably occur independently. It is important clinically to attempt to distinguish between tubular and glomerular injury when proteinuria is identified in a patient receiving AON treatment. Large increases (e.g., >1.5 g/L) or the presence of large molecular weight proteins in urine may aid in identifying glomerular origin and signal a risk of clinical glomerulonephritis. A kidney biopsy would then normally be considered. The characteristic expression of glomerulopathy may well depend on the inflammatory response specific for each species, and this differs both in character and in magnitude between humans and laboratory animals with AON treatment (Henry et al. 1997, 2008; Monteith et al. 1999). A lower risk of glomerular injury in patients with this type of therapy may therefore parallel lower proinflammatory activity in humans as compared to either mice or monkeys. However, clinical risk assessment necessitates monitoring, as in other cases where preclinical renal risks are identified. Since glomerular damage in preclinical species appears to be so intimately related to immune pathogenesis, monitoring systemic inflammatory activity/markers in addition to routine renal and glomerular functional assays is essential in AON clinical trials to assess the potential for drug-related glomerular injury.
Footnotes
Acknowledgments
The authors wish to thank Jan Kane, Roberta Thomas, Deon Hildebrand, and Anna Hughes for technical assistance and especially the staff at CiToxLab and HLS for their help in running the 9-month monkey toxicity study, 6-month mouse toxicity study, and male fertility study in mice.
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Several of the authors are employees of GlaxoSmithKline or Prosensa, and these two companies are jointly developing GSK2402968 as a potential therapeutic treatment.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
