Abstract
Hyaline glomerulopathy with tubulo-fibrillary deposits was observed in two young female ddY mice. One of the mice showed gross systemic edema and bilateral enlargement and pale color of the kidneys, whereas no significant gross findings were noted in the other mouse. Microscopically, a large number of the glomeruli in both mice were enlarged because of diffuse and global deposition of amorphous eosinophilic materials. The deposits were negatively stained with Congo red and positively stained with IgG, IgM, IgA, C3, and periodic acid–Schiff. Electron microscopic examination revealed microtubular and fibrillary deposits with diameters of 80–100 and 9–16 nm, respectively, in the subendothelial space of the glomeruli. These features are histopathologically similar to immunotactoid glomerulopathy or fibrillary glomerulonephritis according to the classification of human glomerular lesions. Understanding of these characteristics of hyaline glomerulopathy in ddY mice is essential when evaluating pharmacological, pharmacokinetic, and toxicological studies using this mouse strain.
Glomerulonephritis (Kirschbaum 1944; Kobayashi et al. 2000; Sass 1986; Seely 1999), glomerulopathy (Shibuya et al. 1990; Wojcinski et al. 1991), and glomerulosclerosis (Gude and Lupton 1960) are spontaneous diseases that frequently occur in aging mice. Although the etiologies of these glomerular lesions have not been fully elucidated, ultrastructural and immunohistochemical findings strongly suggest immunological causes (Kobayashi et al. 2000; Sass 1986; Wojcinski et al. 1991). In addition, hyaline glomerulopathy has been recently reported as a response of chronic pulegone exposure in both mice and rats (Adams et al. 2011). ddY mice, which are affected by IgA nephropathy with aging (Imai et al. 1985; Muso et al. 1996), are used in various fields of research such as pharmacological, pharmacokinetic, and toxicological studies. In the present report, we describe spontaneously occurring hyaline glomerulopathy with the characteristic of tubulo-fibrillary deposits in two young ddY mice.
The experimental procedures and use of the animals followed accredited in-house animal welfare principles. ddY mice were purchased from a commercial supplier (Japan SLC Inc., Hamamatsu, Japan) for the purpose of microbial monitoring at four weeks of age. The mice were housed five mice per polycarbonate cage with wood chips and maintained in animal rooms with a controlled temperature of 23°C ± 2°C, relative humidity of 55% ± 10%, ventilation at fifteen times/hour, and a twelve-hour light/dark cycle. Rodent chow (CE2, Oriental Yeast Co. Ltd., Tokyo, Japan) and tap water were available ad libitum. All the mice were kept untreated until scheduled necropsy. Among these mice, the kidneys from two female mice were subjected to histopathological examinations because of unscheduled death with gross lesions including systemic edema, and bilateral enlargement and discoloration of the kidneys at fourteen weeks of age (Case 1) and for scheduled necropsy at fourteen weeks of age without gross abnormalities (Case 2). The kidneys were fixed in neutral-buffered 10% formalin, and slices were embedded in paraffin. Histological sections were cut at 3-µm thickness and stained with hematoxylin and eosin (H&E).
The kidneys were further examined by periodic acid–Schiff (PAS), Congo red, Masson trichrome, and periodic acid methenamine silver (PAM) staining. Immunohistochemical staining was performed according to a labeled polymer method using a Histofine Simple Stain Rat MAX-PO (MULTI) kit (Nichirei Biosciences Inc., Tokyo, Japan) and primary antibodies against mouse IgG, IgM, IgA, and C3 (MP Biomedicals, Cappel Research Reagents, Irvine, CA, USA), and the sections were counterstained with hematoxylin. For electron microscopic examination, small pieces of kidney fixed in neutral-buffered 10% formalin were further fixed in 2.5% glutaraldehyde, postfixed in 1% osmium tetroxide, and embedded in Epon resin in a standard manner. Ultrathin sections of selected areas were prepared, contrasted with hafnium chloride and lead citrate, and examined using a Hitachi 7600 transmission electron microscope (Hitachi High-Technologies Co., Tokyo, Japan).
Histopathologically, the kidneys of Cases 1 and 2 showed common glomerular lesions. Diffuse and global deposition of amorphous eosinophilic materials was prominent in the glomeruli of the bilateral kidneys (Figure 1 ). As a result, a large number of the glomeruli were enlarged. Collapse of capillary lumens and expansion of the mesangial region because of the deposits (Figure 2a) as well as fibrosis, mineralization, and adhesions of Bowman’s capsule were also observed in the glomerular lesions. Findings noted in other parts of the kidney included dilated and regenerative tubules, proteinaceous casts, mineralization of the arteriolar walls in Case 1, and interstitial mononuclear cell infiltration in Case 2. The glomerular, tubular, and arteriolar wall lesions in Case 1 were more severe than those in Case 2, and these differences may have contributed to the differences in conditions or gross findings of these mice. Neither proliferation of mesangial cells nor infiltration of inflammatory cells into the glomeruli was found in the kidneys. In addition, the deposits of eosinophilic materials were confined to the glomeruli, whereas the basement membrane of the renal tubules, interstitium of the kidney, and arteriolar walls were free of the deposits.
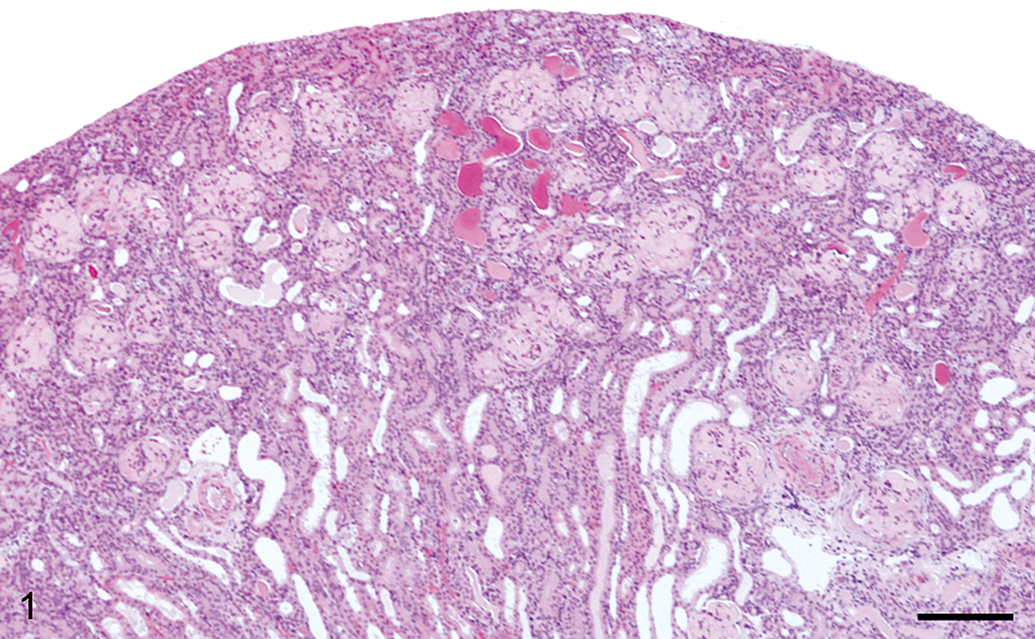
The glomeruli of Case 1 are enlarged owing to deposition of amorphous eosinophilic materials. Dilated renal tubules and proteinaceous casts were also observed. Hematoxylin and eosin; bar = 200 µm.
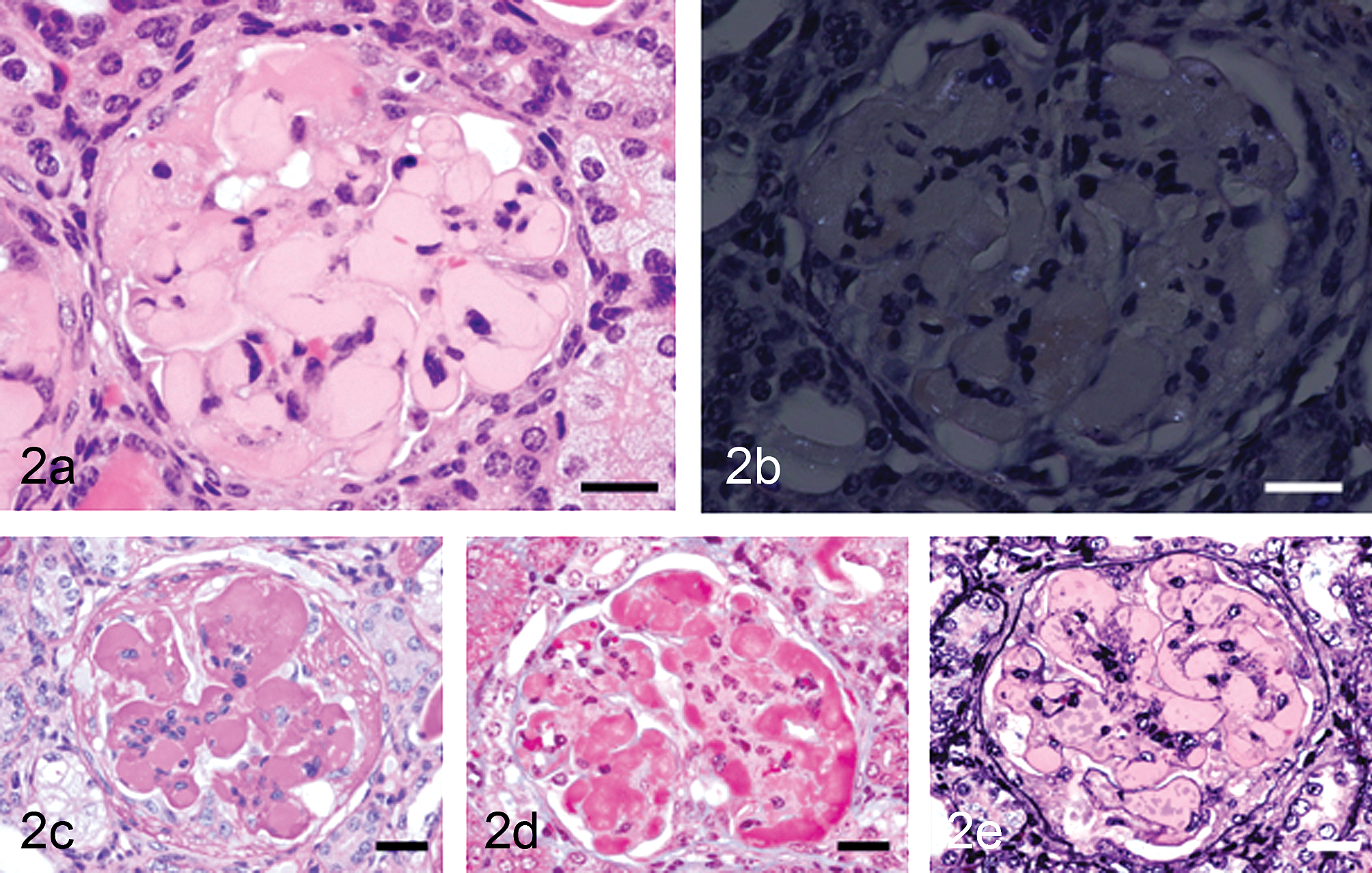
Glomeruli of Case 1. (a) An enlarged glomerulus with deposition of eosinophilic materials, collapse of capillary lumens, and expansion of mesangial region. Hematoxylin and eosin; bar = 50 µm. (b) The glomerular deposits are negatively stained with Congo red with polarized light. Congo red with polarized light; bar = 50 µm. (c) The glomerular deposits are positively stained with periodic acid–Schiff. PAS; bar = 50 µm. (d) The glomerular deposits are stained red with Masson trichrome. Masson trichrome; bar = 50 µm. (e) The basement membrane stained black with PAM is uniform in thickness, whereas the deposits observed in the inner side of basement membrane are not stained with periodic acid methenamine silver. Periodic acid methenamine silver; bar = 50 µm.
The glomerular deposits were negatively stained with Congo red (Figure 2a), positively stained with PAS (Figure 2c), and stained red with Masson trichrome (Figure 2d). The basement membrane stained black with PAM was uniform in thickness throughout the glomeruli (Figure 2e), whereas the deposits located in the inner side of basement membrane were not stained with PAM (Figure 2d). Immunohistochemically, the deposits showed strong reactivity for IgG (Figure 3a), IgM (Figure 3b), and IgA (Figure 3c), and they showed lower reactivity for C3 (Figure 3d), with approximately same degree of staining between Case 1 and Case 2.
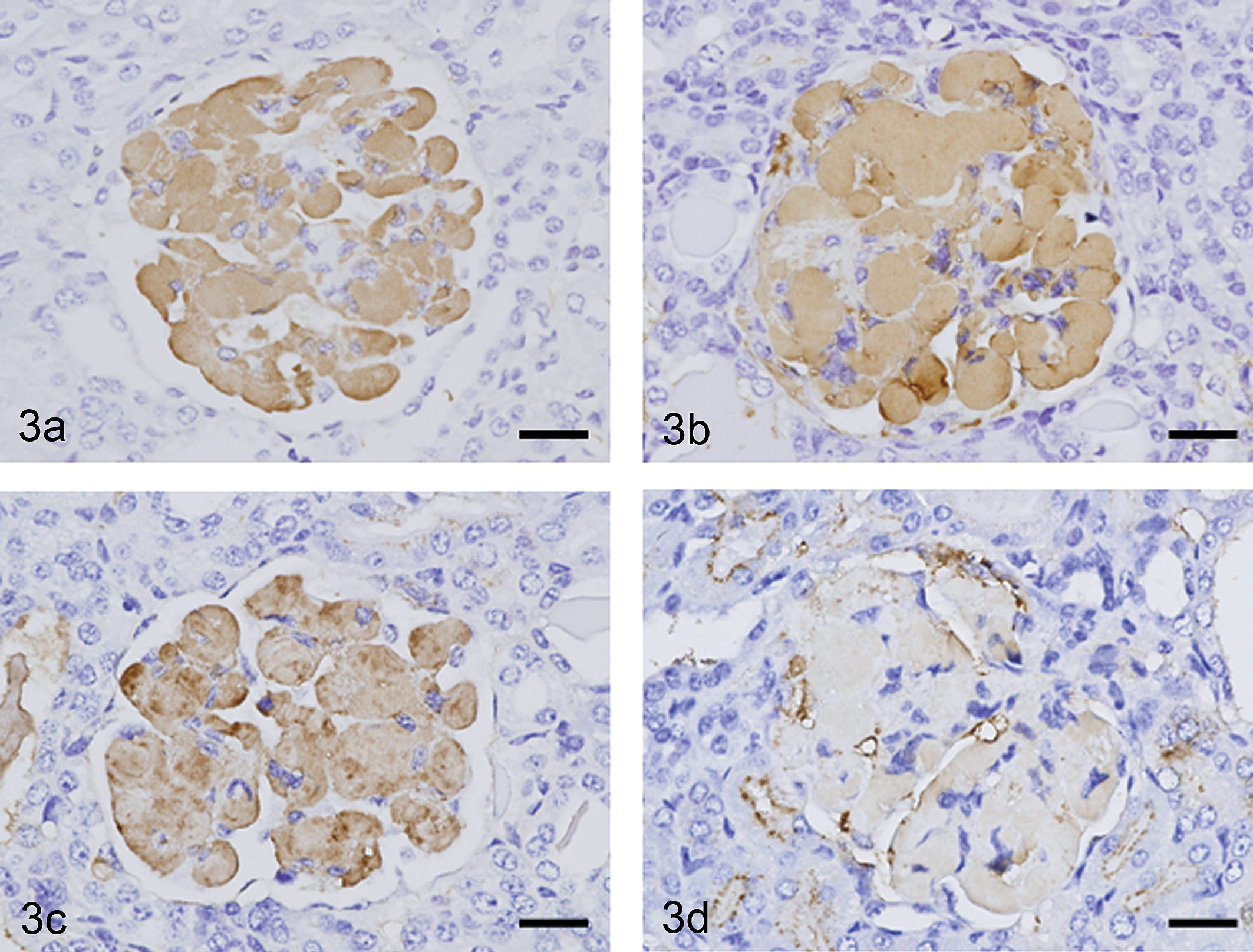
Glomeruli of Case 1 after immunohistochemical staining for mouse IgG, IgM, IgA, and C3. The glomerular deposits were positive for IgG (a), IgM (b), and IgA (c) and weakly positive for C3 (d). Bars = 50 µm.
Electron microscopic examination of the glomeruli of the two mice revealed numerous electron-dense deposits, primarily located in the subendothelial space at the site of the glomerular capillary basement membrane, and massive deposits that frequently expanded into the mesangial region (Figures 4a and 4b). At higher magnification, these deposits were composed exclusively of microtubular structures in Case 1 (Figure 4c), whereas they were composed of mixtures of microtubular and fibrillary structures in Case 2 (Figure 4d). The microtubular structures had diameters of 80–100 nm and hollow centers (Figures 4c and 4d). The fibrillary structures had diameters of 9–16 nm (Figure 4d). The structures were straight or curved linear structures, rather than bifurcating or branching (Figure 4c). The basement membrane was not thickened.
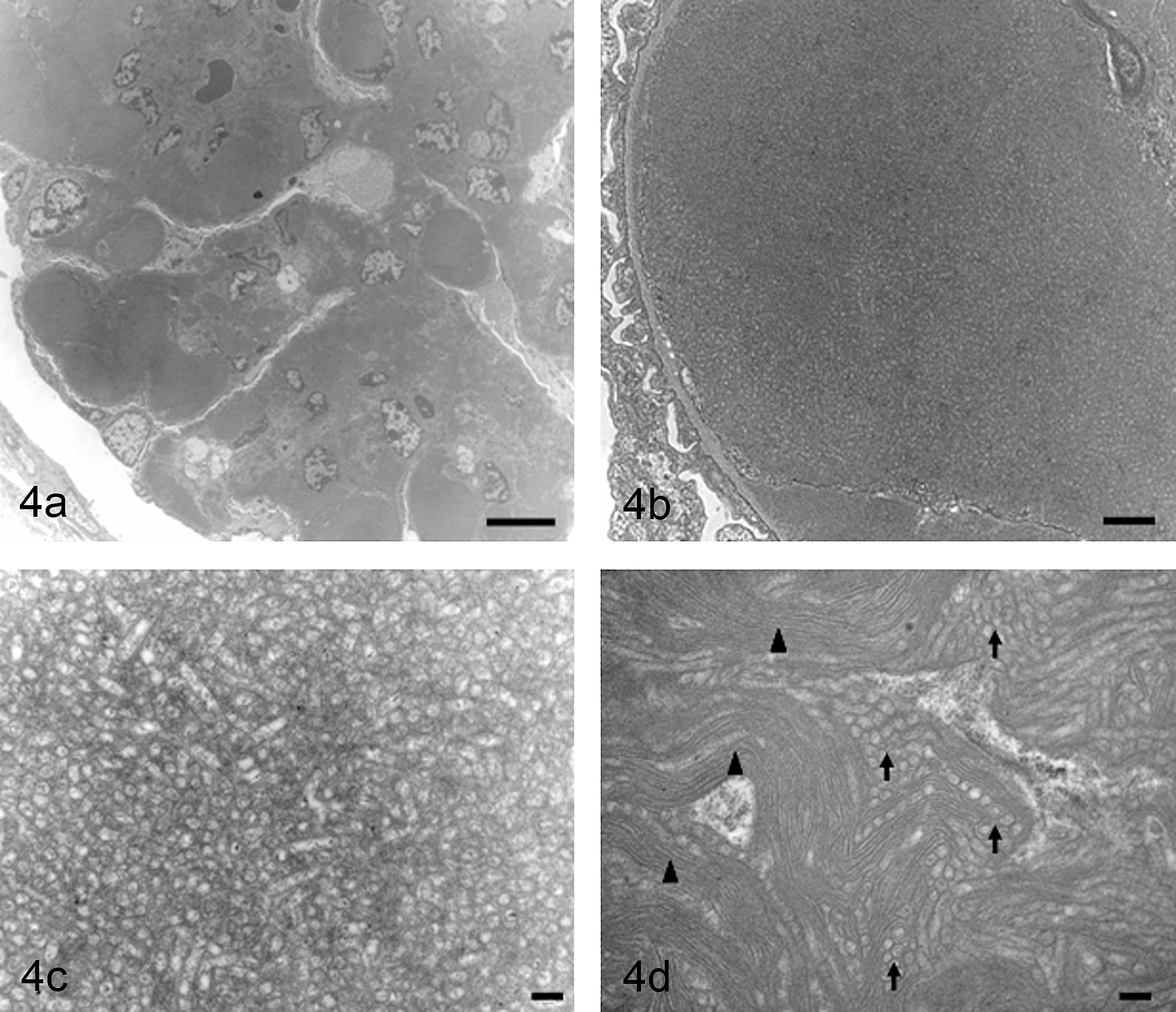
Glomeruli of Case 1 and 2 observed by electron microscopy. (a) Electron-dense deposits on an affected glomerulus in Case 1. Bar = 10 µm. (b) Higher magnification of (a). Deposits are located in the subendothelial space at the glomerular capillary basement membrane. Bar = 1 µm. (c) High magnification image of Case 1 reveals microtubular structures with diameters of approximately 80–100 nm and hollow centers. Bar = 200 nm. (d) High magnification image of Case 2 shows both microtubular (arrows) and fibrillary (arrowheads) structures with diameters of 80–100 and 9–16 nm, respectively. Bar = 200 nm.
The light microscopic findings of our H&E-stained sections resemble amyloidosis (Maita et al. 1988), and the light microscopic examinations may not enable us to differentiate our cases from hyaline glomerulopathy (Wojcinski et al. 1991) or glomerulonephritis (Kobayashi et al. 2000). The amyloidosis reported to occur in several mouse strains is readily distinguished by the facts that the deposits are positively stained with Congo red and appear as 10 nm wide and nonbranching filaments (Maita et al. 1988; Seely 1999). Hyaline glomerulopathy is spontaneous lesions found in aging female B6C3F1 mice; these lesions are related to pulegone exposure in rats and mice (Adams et al. 2011; Wojcinski et al. 1991). Glomerulonephritis is also reported as spontaneous lesions in a female
Although the histopathological characteristics of Cases 1 and 2 may represent one of the subtypes of the IgA nephropathy to which ddY mice are susceptible (Imai et al. 1985; Muso et al. 1996), there have been no reports describing such massive deposits of amorphous materials with tubulo-fibrillary structures in the glomeruli of ddY mice.
Light and ultramicroscopic findings for the glomeruli of the two ddY mice are also similar to immunotactoid glomerulopathy or fibrillary glomerulonephritis, according to the classification of human glomerular lesions showing tubular or fibrillary deposits in the glomeruli (D'Agati et al. 2005; Iskandar and Herrera 2002; Korbet et al. 1985; Rosenstock et al. 2003), although these classification have not been defined in rodents. Immunotactoid glomerulopathy and fibrillary glomerulonephritis are characterized by non-amyloid glomerulopathies in which the deposits are negative for Congo red and are instead composed of immunoglobulins such as IgG, IgM, IgA, and C3. These deposits are observed by electron microscopy as electron-dense deposits and are similar to amyloid in that they consist of nonbranching fibrillary structures (Iskandar and Herrera 2002; Korbet et al. 1985; Rosenstock et al. 2003). Immunotactoid glomerulopathy is defined by glomerular deposition of hollow, stacked microtubules of ≥30 nm, whereas fibrillary glomerulonephritis is defined by glomerular deposition of randomly oriented fibrils of < 30 nm in diameter (Iskandar and Herrera 2002; Korbet et al. 1985; Rosenstock et al. 2003).
In conclusion, all of the morphological findings obtained from Cases 1 and 2 support the notion that the glomerular lesions in these ddY mice with tubulo-fibrillary deposits are consistent with hyaline glomerulopathy reported in mice and rats, and they are also similar to immunotactoid glomerulopathy or fibrillary glomerulonephritis found in humans. Similar glomerular lesions have been reported in NNT-1/BSF-3-transgenic mice and were classified as immunotactoid glomerulopathy (Senaldi et al. 2002). The incidence of glomerular lesions in ddY mice has not been reported, and the etiology of the glomerular lesions has not been clearly elucidated. Therefore, recognition and further understanding of these unique electron microscopic features in the kidneys of ddY mice are essential when evaluating pharmacological, pharmacokinetic, and toxicological studies using this mouse strain.
Footnotes
Acknowledgment
We wish to thank the technicians at the pathology laboratory for their excellent technical assistance.
