Abstract
Minipigs are increasingly being used as an alternative to dog or monkey in nonclinical safety testing of pharmaceuticals since they share similar anatomical and physiological characteristics to humans. Integrative assessment of pharmacodynamic and pharmacokinetic data sets of drug candidates from in silico, in vitro, and in vivo investigations form the basis for selecting the most relevant nonrodent species for toxicology studies. Developing anticancer therapeutics represents a special challenge for species selection due to their effects on multiple organ systems. The toxicological profile of anticancer drugs can be associated with steep dose–response curves, especially due to dose-limiting toxicity on the alimentary, hematopoietic, and immune systems. Selection of an appropriate species for toxicology studies is of importance to avoid an inappropriately low (without benefit for the late-stage cancer patient) or high clinical starting dose (with a risk of unexpected adverse reactions). Although the minipig has been the preferred species to develop drugs applied topically, it is only rarely used in anticancer drug development compared to dog and monkey. In this context, we discuss the potential of minipigs in anticancer drug development with examples of programs for oral and dermal administration, intravascular application in drug-eluting stents, and local chemotherapy (chemoembolization).
Minipigs are increasingly being used as an alternative to dog or monkey in nonclinical safety testing of pharmaceuticals since they share similar anatomical and physiological characteristics in cardiovascular, urinary, integumentary, ocular, and digestive systems to human. A number of more recent publications covered general and specific aspects of the use and potential of minipigs in biomedical research and drug development (Foster et al. 2010a; McAnulty et al. 2012; Swindle et al. 2012). Comparison of genome sequences and gene expression profiles of pharmacological targets as well as enzymes and transporters involved in drug metabolism and disposition in nonrodent species including minipig are increasingly considered for selection of the most relevant species to predict pharmacodynamic and pharmacokinetic drug characteristics in humans (Foster et al. 2010b; Vamathevan et al. 2013; Helke and Swindle 2013; Dalgaard 2015). Integrative assessment of pharmacodynamic and pharmacokinetic data sets of drug candidates from in silico, in vitro, and in vivo investigations form the basis for selection of the most relevant nonrodent species for drug development.
Safety Evaluation of Anticancer Drugs
Although the evaluation of efficacy and safety in animal models for drug development in general and anticancer drugs specifically has many features in common, there are also differences based on the specific use of anticancer drugs in clinical practice (e.g., treatment in cycles and in combination with other drugs). Anticancer drugs comprise a variety of different classes of therapeutics, ranging from small molecules to biologic entities or their combinations. Their mode of action includes eradication of rapidly multiplying tumor cells by impairment of cell division based on cytotoxic effects (e.g., alkylating agents, antimetabolites, antimicrotubule agents, topoisomerase inhibitors, and antibody drug conjugates); inhibition of cell proliferation by targeting specific signaling pathways key to tumor growth (e.g., tyrosine kinase inhibitors, farnesyltransferase inhibitors, matrix metalloproteinase inhibitors, and antibodies against tumor-specific antigens); destruction of tumor-supporting tissues (e.g., angiogenesis inhibitors and vascular disrupting agents); or by prevention of tumor cell migration, homing, and metastasis (e.g., protease inhibitors and matrix metalloproteinase inhibitors). Nonspecific cytotoxic agents generally cause dose-dependent/dose-limiting toxicity due to inhibition of cell proliferation in various tissues (e.g., alimentary, ingumental, reproductive, hematopoietic, and lymphatic systems), whereas molecularly targeted agents are generally better tolerated with a specific toxicity profile.
Anticancer therapeutics are often applied in combinations, considering specific treatment schedules to increase efficacy, to reduce adverse reactions and to overcome drug resistance. The latter phenomenon is a major cause of therapy failure due to adaptations of the cancer cells (e.g., upregulation of drug efflux pumps, gene amplification, defective apoptosis pathways, or upregulation of DNA repair mechanisms). In view of the specific properties of individual tumor types, personalized treatment strategies based on molecular tumor characterization and patient risk profile are increasingly applied to further improve treatment responses and patient survival. In contrast to drug development for other indications, initial clinical trials with anticancer drugs are generally performed with late-stage cancer patients whose disease is refractory or resistant to available therapy or where current therapy is not considered to be providing benefit (Guidance for Industry, ICH S9, FDA 2010).
Animal studies with anticancer drugs are performed to provide evidence of pharmacological activity of the drug in suitable tumor models, to optimize treatment schedules and procedures (e.g., dosages, routes of administration, treatment cycles, and drug combinations), and to identify pathways and biomarkers in connection with efficacy and tolerability. Based on the evaluation of the toxicity profile, severity and reversibility of adverse effects in relation to the pharmacokinetic profile, a safe starting dose for clinical trials is defined. Selection of appropriate animal species for toxicology studies is of importance to avoid an inappropriately low clinical starting dose without benefit for the late-stage cancer patient or an inappropriately high starting dose with a risk of unexpected adverse reactions including death (e.g., see Narang and Desai 2009). Further animal studies are performed in compliance with regulatory requirements to support the progress of the clinical program and finally market authorization of the new drug. In addition, animal models are developed to explore the efficacy and/or safety of specific applications of anticancer drugs such as chemoembolization or nanoparticle formulations. Challenges in animal studies with anticancer drugs, especially cytotoxic agents, can be a steep dose–response curve with severe dose-limiting toxicity, for example, on gastrointestinal tract, bone marrow, and lymphatic system and, in connection with the latter, exacerbation of background diseases as a consequence of overimmunosuppression. Poor general health condition due to a combination of toxic effects and exacerbation of background diseases may also be associated with variable exposure, for example, after intestinal lesions or dysfunction. Animal models with limited utility may therefore lead to over- or underestimation of the toxicity of anticancer drugs.
Nonrodent Species Selection for Safety Evaluation of Anticancer Drugs—Pharmacokinetic Aspects
For selection of the nonrodent species, early in silico and in vitro evaluations, covering pharmacodynamic as well as pharmacokinetic aspects, are used in comparison to human data. Minipigs are considered increasingly in these test systems, however, in view of gaps in experience and background data not on a routine basis by most pharmaceutical companies (Brewster et al. in press ). Species differences in drug metabolism are well known. Although in general nonhuman primates show the closest homology of drug metabolizing enzymes compared to humans, drugs can be metabolized by various enzymes and eventually the profile of metabolites is important for the selection of an appropriate animal model, especially if species-specific enzymes dominate metabolism of the drug. The expression of CYP2C76 in monkeys but not in humans can result in a different metabolite and toxicity profile (Dalgaard 2015). For species selection, in vitro and in vivo correlations of ADME parameters including metabolic profiles and involved enzymes, subcellular fractions of drug-metabolizing tissues (liver and gut) including cytosol, S9, and microsomal fractions are used to address various questions and cover nearly all drug-metabolizing enzymes such as membrane-bound cytochromes (CYPs) and conjugation enzymes uridine 5′-diphospho-glucuronosyltransferase or cytosolic N-acetyl transferase (NAT), glutathione transferase, and aldehyde oxidase (AO; Zhang et al. 2012). An example of species-specific metabolic differences is the involvement of AO, a cytosolic molybdenum hydroxylase, involved in the oxidative and reductive metabolism of drugs containing aromatic heterocyclic nitrogen compounds. AO also serves as an important cellular source of reactive oxygen/nitrogen species in cell signaling and regulation of physiological functions. In humans, a single AO (AOX1) is functional, which is more active than in rodents with 4 active AOX genes, whereas dogs do not have functional AO (Barr, Choughule, and Jones 2014). Clinical studies with a small molecule, selective, and potent c-Met (a receptor tyrosine kinase) inhibitor revealed renal toxicity even at subtherapeutic doses due to insoluble metabolites generated by AO activity, although there was no indication of renal toxicity in nonclinical safety studies with rats and dogs (Lolkema et al. 2015). A similar potential of another c-Met inhibitor to cause acute renal failure in patients, due to intracellular crystal formation of an AO metabolite, was not detected in rat and dog studies but in monkeys (Barr, Choughule, and Jones 2014). In pigs, AO activity has been demonstrated with various substrates, and expression of orthologous AOX1 is predicted based on genomic data (Dalgaard 2015). Further examples of species-specific differences in metabolizing enzymes are the absence of NAT1 and NAT2 or CYP2C9-like enzymes in dogs but not in pigs. On the other hand, sulfation by 3-phosphoadenosyl-5-phosphosulfotransferase, an important pathway in human metabolism, appears to be less prominent in pigs (Dalgaard 2015).
Species-specific differences in membrane transporters in various organs including blood–brain barrier are of importance for the assessment of pharmacological and toxic effects of drugs. Especially in anticancer therapy, the development of drug resistance is one major reason of treatment failure and disease progression. Drug resistance can be a consequence of drug extrusion from the cell by energy-requiring efflux pumps belonging to the ABC (adenosine triphosphate-binding cassette) transporter superfamily such as P-glycoprotein (multidrug resistance protein MDR1/ABCB1), MDR-associated protein (ABCC1), and breast-cancer resistance protein (ABCG2) or of reduced cellular uptake of drugs by another transporter superfamily, solute carrier (SLC) transporters (F. S. Liu 2009). The latter include ion-coupled transporters, exchangers, and passive transporters and serve as uptake mediators of essential nutrients for tumor growth and survival but are also involved in the transfer of anticancer drugs into the cell. These transporters are expressed in various tissues such as small intestine, liver, and kidney and may be of particular importance for drug disposition, and based on their individual variation, contribute to variability in drug exposure and pharmacodynamic as well as adverse effects (Li and Shu 2014). In addition, SLC transporters may show different expression profiles in tumor cells compared to normal cells in order to support their specific demand of nutrients for sustained growth. In view of the importance of transporters for the pharmacological activity as well as the toxicological profile in connection with systemic exposure, early evaluation of drug candidates as potential transporter substrates may help to understand opportunities and risks in connection with specific tumor types, tissues, and species selection for drug development.
Selected transporters in monkeys, pigs, and dogs have been reviewed in comparison to humans (Dalgaard 2015), indicating that transporters in monkeys were highly comparable to humans with amino acid sequences higher than 90% identical, and in pigs and dogs largely comparable with amino acid sequences higher than 72% identical. The organic anion transporting polypeptides SLCO1B1 (OATP1B1) transporter, highly expressed in human liver, however, has no orthologue in pigs or dogs. SLCO1B1 activity may be important in hepatic disposition of anticancer drugs such as irinotecan, and genetic polymorphism may contribute to the known individual variation in the drug disposition (Li and Shu 2014). In addition, the organic anion transporter SLC22A8 (OAT3) is hardly expressed in dog liver (Bleasby et al. 2006; Dalgaard 2015). SLC22A8, present in most animal species including pig, is highly expressed in human kidney and could be mainly responsible for renal methotrexate excretion and nephrotoxicity as a consequence of reduced renal clearance (Li and Shu 2014).
Nonrodent Species Selection for Safety Evaluation of Anticancer Drugs—Pharmacodynamic Aspects and Challenges
Safety flags from early in silico and in vitro evaluations can influence the choice of the nonrodent species, based on similarities of the target in homology, tissue distribution, and pathway cross functionality to humans. Early in vivo data are mostly generated in rodents in connection with pharmacological tumor models to optimize dose and treatment schedules in combination with the assessment of safety parameters. Safety data in these models can give a first idea about potential target organs and some of the dose-limiting toxicities in later nonrodent studies. The dog is commonly used for testing of small molecule anticancer drugs, also in view of historical data with similar compounds in the product pipeline or from competitors. The nonhuman primate is commonly used for biologic anticancer drugs, whereas the minipig is generally the species of choice for drugs with dermal applications and for special indications supporting local administration of anticancer drugs, for example, in intravascular stents or by chemoembolization. Based on the literature, the minipig is only rarely used in the development of anticancer drugs with oral or parenteral routes of administration, although the dog as standard species often shows dose-limiting toxicity in the range of or even below human dose/exposure equivalents. In dogs, excessive vomiting, disturbance of the immunological barrier function (intestinal tract and lungs) in connection overimmunosuppression, and gastrointestinal intolerability are often noted as dose-limiting factors. Decreased resistance to infection and gastrointestinal intolerability are also observed in nonhuman primate studies, leading to moribundity and loss of animals. In contrast to dogs and nonhuman primates, minipigs are bred under strictly controlled hygienic conditions and provided in specific-pathogen-free (SPF) quality and therefore may have a lower risk for exacerbation of background infections compared to the other nonrodent species. Reasons for the only rare use of minipigs in studies with oral or parenteral administrations may be the lack of historical data with specific classes of anticancer drugs as well as the only sporadic involvement in early profiling investigations for species comparison in the selection process to identify the most predictive species for humans.
Species Comparison Based on an Example with an Anticancer Drug
The following example of the use of dogs, monkeys, and minipigs in the in-house development of a small molecule with antiproliferative, antiangiogenic, and immunosuppressive activities based on kinase inhibition is presented to give an idea of the advantages and limitations of each species without detailed description of the study designs, materials, methods, and results. These studies were performed in accordance with the respective national animal welfare regulations and approved animal study licenses and, with the exception of the early dose-range findings studies, in compliance with the principles of good laboratory practice (GLP). Study designs were based on internationally accepted test guidelines to support the clinical programs and fulfilled requirements for submission to health authorities to obtain market authorization. Initially, the dog was selected as a standard nonrodent species. Gastrointestinal mucosal ulcerations and hemorrhagic bronchopneumonia, however, were observed in the oral (gavage) rising-dose study after a few administrations of the compound resulting in early termination of the study. Findings suggested effects secondary to immunosuppression and excluded the dog for investigation of the toxicity profile in studies with longer duration and at higher doses. Comparable results with a similar compound of this class in dogs, but a better tolerability in humans, justified the change to a more appropriate species. Monkeys tolerated the compound better than dogs after oral administration in terms of doses and duration of treatment. Myocardial lesions without clear correlation to systemic exposure in individual animals suggested a relationship to viral infection, which was demonstrated in one study as an exacerbation of cardiotropic Coxsackie virus infection in the plasma and heart of treated animals. Inflammatory changes in the gastrointestinal tract contributed to the early termination of the high-dose group in a long-term toxicity study with monkeys. The monkey was a suitable species for exploring the toxicity profile of the compound including in long-term studies; however, higher doses were not tolerated due to gastrointestinal problems. The minipig was introduced in the development program to evaluate tolerability of the drug after oral administration at higher doses, and, as expected, myocardial lesions as seen in individual monkeys were not present in minipigs. The minipig tolerated the compound better than the dog in terms of doses and duration of the treatment. Diarrhea due to exacerbation of coccidial infestation (Figure 1) and associated lesions in the intestine (Figure 2) required early termination of individual high-dose animals in a mid-term toxicity study. Exacerbation of coccidial infestation and inflammatory, ulcerative lesions in the small intestine (Figure 2) suggested a consequence of immunosuppression and a reduced ability of the intestinal mucosa to regenerate in view of the antiproliferative activity of the drug. During the study, the minipigs were kept under optimal hygienic but not SPF conditions. The results of the study showed that the minipig was a suitable species for exploring the toxicity profile of the compound, but similar to monkey, higher doses were not tolerated due to effects secondary to overimmunosuppression. In general, exposure toxicity profiles were similar in both species with the exception of differences in the organ manifestation of effects secondary due to immunosuppression. Therefore, both monkey and minipig were appropriate species to support to the clinical development program and registration of this drug. Careful and extensive prescreening of animals used for toxicity studies concerning immunological and infectious status is of advantage for animal selection or explanation of study findings, which may be easier to define and standardize in the Göttingen minipig compared to monkeys of different strains and background.
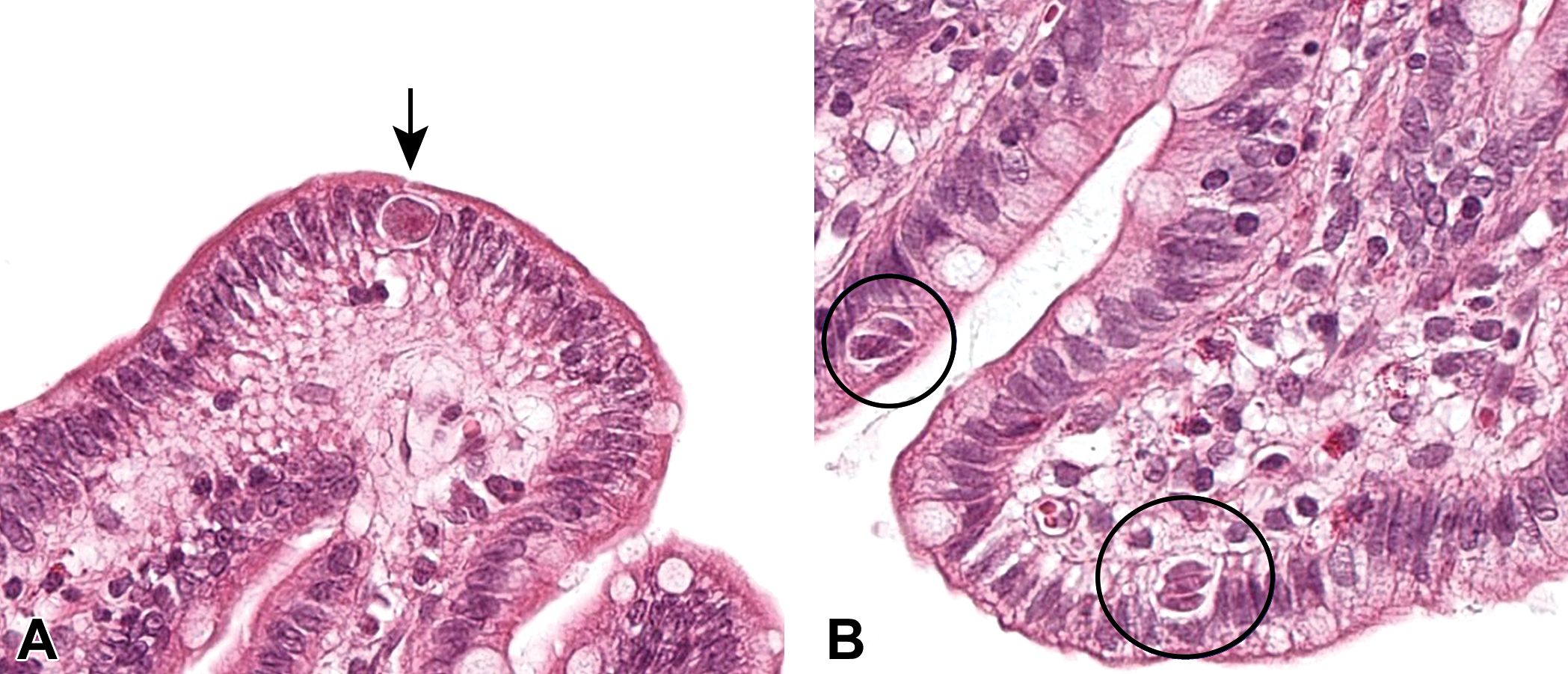
Parasitic infestation in the small intestine: Coccidiosis (intraepithelial parasites at different stages of its life cycle [arrows and circles]) in the Göttingen minipig treated with an anticancer drug. 10×, H&E.
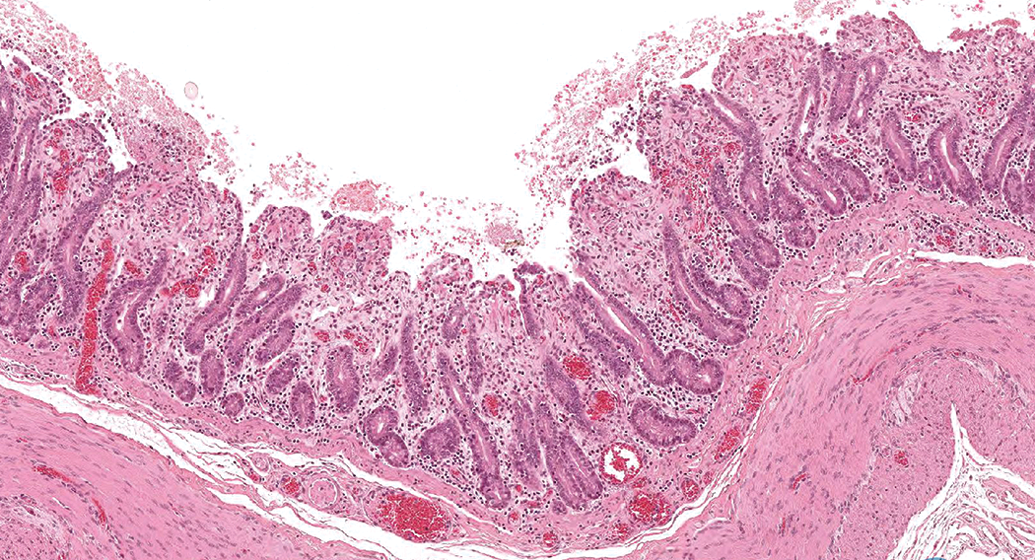
Marked ulcerative, necrotizing mucosal inflammation with villous atrophy in the small intestine (jejunum) of a Göttingen minipig treated with an anticancer drug due to parasitic manifestation. 4×, H&E.
The Minipig in Local Administration of Anticancer Drugs: Topical Treatment
The minipig is an established model for the investigation of local applications of anticancer drugs and is accepted or even requested by health authorities. The use of minipigs for dermal administrations in drug development has already a long history driven by the similarities between porcine and human skin (Lakver et at. 1991; Mortensen, Brinck, and Lichtenberg 1998; Mahl et al. 2006; Makin, Mortensen, and Brock 2012). An example of an integrated assessment of a new antiproliferative drug with topical administration in comparison to marketed standard therapeutics by in vitro investigations using human reconstructed epidermis and by in vivo evaluations after dermal administration to minipigs with respect to mechanistic pathway analysis and skin tolerability is given by Medina et al. (2003). A further minipig study with this drug after topical treatment over 2 weeks confirmed the results of the initial investigations with arrest of keratinocytes in the M phase of the cell cycle as demonstrated by laser scanning cytometry (Figure 3). Consequent selective induction of apoptosis was shown by histopathology (Figure 4) and laser scanning cytometry (Figure 5). These investigations showed that the minipig was a suitable model to assess pharmacological effects of the drug as well as local and systemic tolerability in connection with corresponding exposure data.
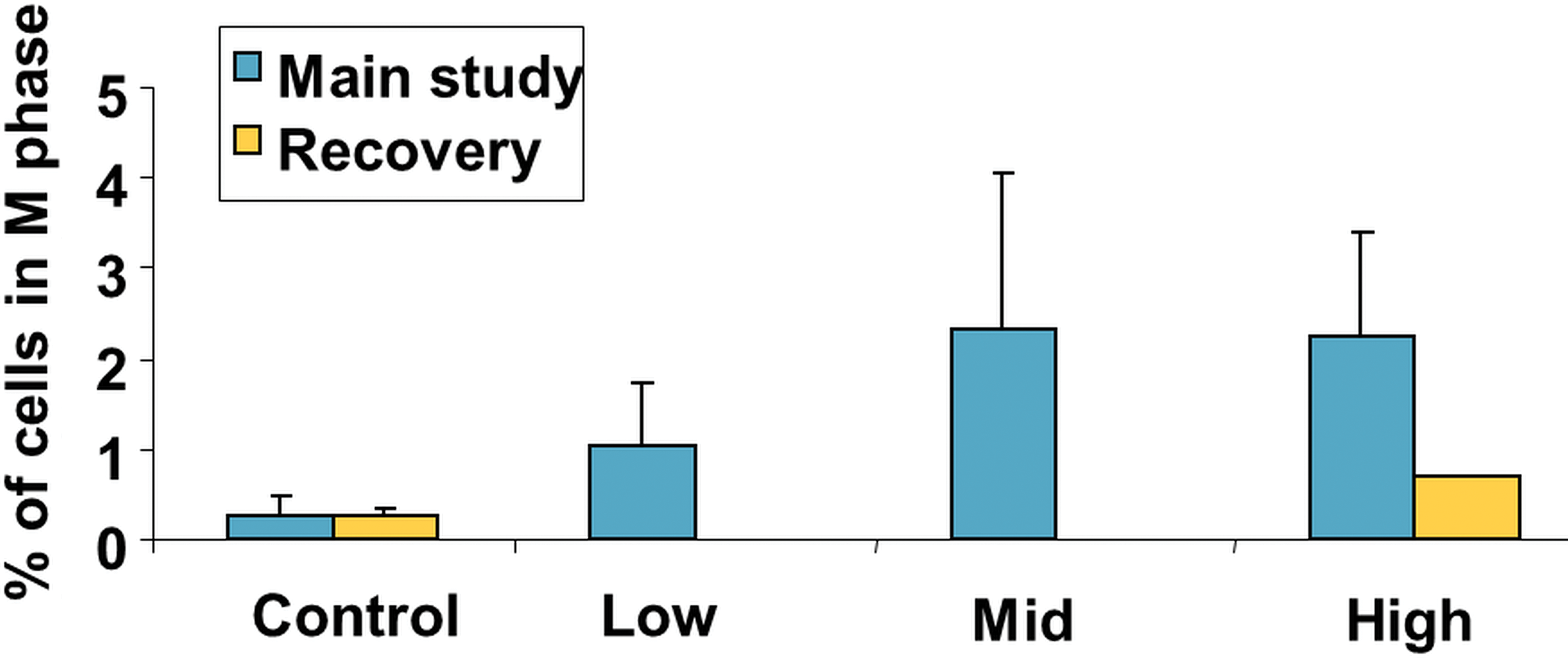
Cell cycle evaluation by laser scanning cytometry in the Göttingen minipig skin after 2 weeks of treatment with an anticancer drug, followed by 2 weeks of recovery period.
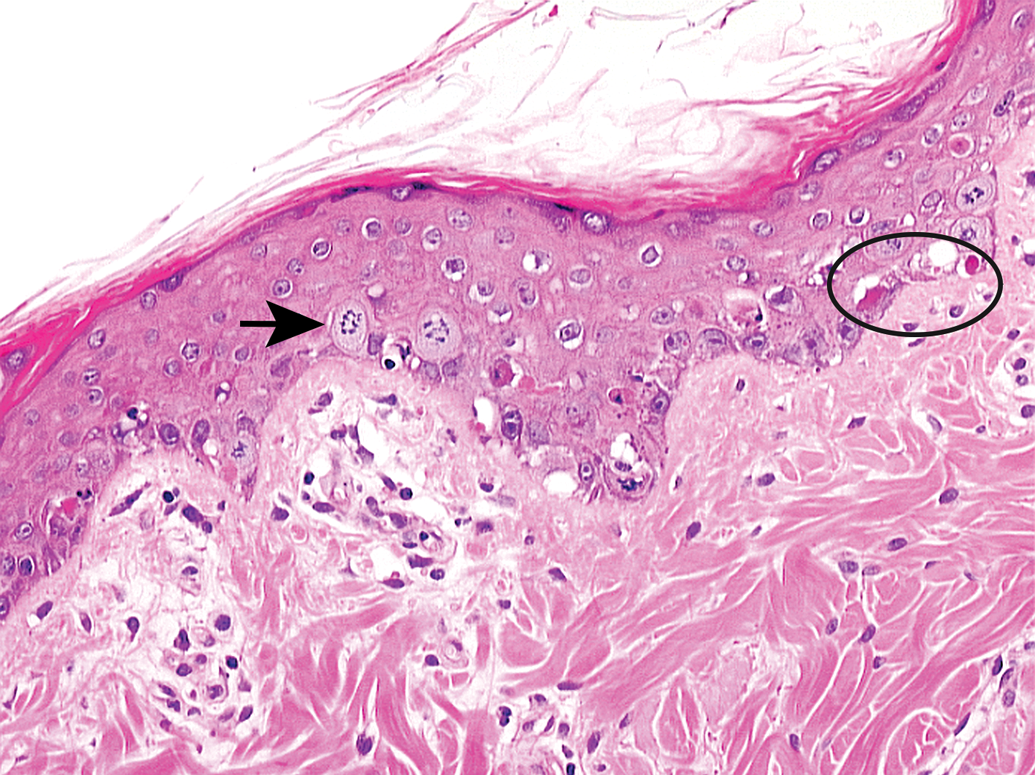
Epidermal apoptotic bodies (circle) and mitotic figures (arrow) in the Göttingen minipig skin treated with an anticancer drug. 20×, H&E.
Apoptosis evaluation by laser scanning cytometry in the Göttingen minipig skin after 2 weeks of treatment with an anticancer drug, followed by 2 weeks of recovery period.
Minipig in Local Administration of Anticancer Drugs: Intravascular Stents
Although applied in a different indication, drugs with antiproliferative, anti-inflammatory, and antineoplastic activities represent potential candidates for the use in drug-eluting stents to prevent restenosis of coronary or peripheral arteries with atherosclerotic lesions and their consequences like myocardial infarction, stroke, or other organ failure. Sirolimus, everolimus, zotarolimus, and paclitaxel are examples for anticancer drugs with clinical application in drug-eluting stents. Although vessels in the pig models lack atherosclerosis, they are still considered one of the best approaches for the investigation of tolerability and assessment of potential risks associated with new drug-eluting intravascular stents. These models also allow a comparison of different stents in clinical use over different implantation periods under standardized conditions (Hiranuma et al. 2014). Selected examples of these minipig models using various in-life and postmortem investigations are given further below. Investigations in minipigs were performed on the long-term local stent-mediated delivery of everolimus and the reduction of neointimal hyperplasia in the iliac arteries (Zhao et al. 2012). Furthermore, minipigs were used to assess fluctuations in arterial drug delivery from intravascular stents implanted in the coronary artery in relation to thrombus formation (Balakrishnan et al. 2008). For the development of a new type of sirolimus-eluting stents with asymmetrical coating to optimize target tissue exposure at the abluminal side of the stent and minimize drug effects at the luminal side, coronary arteries of minipigs (Shen et al. 2012) or domestic piglets (Sojitra et al. 2015) with different types of stents were investigated by various methods, for example, angiography, intravascular ultrasound, vasomotor function in response to acetylcholine, and postmortem scanning electron microscopy and histopathology examinations. In-life optical coherence tomography in comparison to histomorphometry was used to evaluate the coronary arteries of minipigs for up to 4 years after implantation of everolimus-eluting bioresorbable vascular scaffolds (Gogas et al. 2012). A porcine model was also used to evaluate safety and efficacy of a new sirolimus-eluting stent with bioabsorbable polymer by angiography, quantitative coronary angiography, histomorphometry, and electron microscopy (Wu et al. 2012). Minipigs were used to determine plasma and tissue concentrations of paclitaxel after placement of cobalt–chromium stents with nanoscale coating and different paclitaxel doses in the coronary arteries to assess drug kinetics and safety (Radeleff et al. 2010). Since sirolimus- and paclitaxel-eluting stents are less effective in the prevention of restenosis in patients with diabetes, a diabetic minipig model was used to investigate pathways involved in the pathogenesis of restenosis and to identify proteins involved in enhanced neointima formation in diabetes after vascular injury (Yang et al. 2013).
Minipig in Local Administration of Anticancer Drugs: Chemoembolization
Transarterial chemoembolization is a combination of local chemotherapy and target tissue embolization for treatment of patients with uterine fibroids, arteriovenous malformations, as well as nonresectable, benign or malignant renal and hepatocellular tumors (Stampfl et al. 2007a). The advantage of this therapy is induction of ischemic necrosis, reduced systemic distribution and toxicity of the chemotherapeutic agent by embolization of the tumor vasculature, and an increased local drug exposure in the tumor. Embolization techniques and materials vary considerably and are constantly adapted and further developed to improve therapeutic success. Current procedures for embolization of malignant liver tumors include the use of lipiodol in combination with various cytotoxic drugs; however, inhomogeneous uptake or late loss of lipiodol correlated with higher local recurrence rates and therefore required development of new treatment concepts (Stampfl et al. 2007b). Minipigs were considered an appropriate model with respect to similarities in their organ systems to humans. Although the nontumoral porcine models allow no assessment of the efficacy of transarterial chemoembolization, these models are suitable to compare different treatment concepts in terms of local and systemic toxicity in relation to their exposure profiles in target tissues and systemic circulation. Minipigs were used to evaluate the vascular distribution pattern of 40 to 120 µm-trisacryl gelatin microspheres (embospheres) in acute whole kidney embolization or acute arterial bed occlusion in the liver, and the durability and biocompatibility in chronic partial kidney or liver embolization by quantitative angiography and histomorphometry (Stampfl et al. 2007a, 2007b). Transcatheter hepatic arterial chemoembolization was also performed in miniature pigs to investigate the systemic exposure profile of doxorubicin using different matrices such as doxorubicin alginate microspheres, doxorubicin–lipiodol, and doxorubicin infusion over 8 weeks as well as embolization results with digital subtraction arteriography, computerized tomography, and histopathology (D. Liu et al. 2006). Comparison of cisplatin pharmacokinetics in plasma and liver of minipigs after intravenous, intrahepatic arterial administration, and intrahepatic arterial administration with partial embolization using absorbable gelatin or complete embolization showed lower systemic exposure to cisplatin and decreased plasmatic terminal half-life after embolization, whereas hepatic exposure and terminal half-life were increased compared to intravenous administration. These results are in agreement with similar observations in patients after chemoembolization with other materials (Chabrot et al. 2012). In this respect, the minipig models are supportive to explore and compare new chemoembolizaton procedures in order to increase local exposure of anticancer drugs in target tissues and reduce systemic toxicity.
Concluding Remarks
The minipig is the species of choice for the development of antiproliferative drugs with the dermal route of administration as well as for special local applications of anticancer drugs such as in drug-eluting stents or for chemoembolization. Although minipigs are increasingly being used with respect to similar anatomical and physiological characteristics to humans as an alternative to dog or monkey in nonclinical safety testing of pharmaceuticals in general, they are only rarely involved in the nonclinical development program for anticancer drugs with oral or parenteral administration. The reason for the current undervaluation of that species in nonclinical safety testing is most likely the lack of its consideration in early pharmacodynamic and pharmacokinetic testing rather than lack of suitability based on scientific criteria. Therefore, exploration of the potential of the minipig in the selection of the most relevant nonrodent species for the safety testing of anticancer drugs would add value to better predict human safety.
Footnotes
Acknowledgment
We thank Drs. D. Lapadula and W. Kluwe, Novartis Pharmaceuticals Corp., for their review and valuable comments on this article.
Authors’ Contribution
All authors (AM, ZD, and PH) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
