Abstract
Over the past 3 decades minipigs have moved from being an obscure alternative to dogs and nonhuman primates to being a standard animal model in regulatory toxicity studies. This article covers the use of minipigs as a model in the context of nonclinical drug safety and provides an overview of the minipig’s developmental history and relates minipigs to other animal species commonly used in toxicology; and the minipig’s translational power is supported by 43 case studies of marketed drug products covered. Special focus is given to criteria for selecting minipigs in nonclinical programs supporting the development of new medicines; the use of swine in the assessment of food additives, agrochemicals, and pesticides; as well as a regulatory perspective on the use of minipigs in Food and Drug Administration (FDA)-regulated products. This article presents the main points conveyed at a symposium held at the 2010 American College of Toxicology meeting in Baltimore, Maryland.
Introduction
This publication presents the main messages from a session with the same name hosted by Dr Ken L. Hastings and Niels Christian Ganderup at the American College of Toxicology 2010 annual meeting in Baltimore, Maryland. The session dealt with the use of minipigs as a model in the context of nonclinical safety assessment and provided an overview of the domestic pig’s and minipig’s developmental history, its application in biomedical research, and its predictive value. Special focus was given to criteria for selecting minipigs in nonclinical programs supporting the development of new medicines; the use of swine in the assessment of food additives, agrochemicals, and pesticides; as well as a regulatory perspective on the use of minipigs in Food and Drug Administration (FDA)-regulated products. Swine is used collectively to describe both minipigs and domestic pigs. Domestic pig and minipig are used to explicitly describe 1 of the 2, and no distinction between breeds within the 2 groups is made, that is, domestic pigs can, for example, be Yorkshire or German Landrace, while a minipig could be a Yucatan micropig or a Göttingen minipig.
Introduction to Pigs and Minipigs as a Nonrodent Species (Niels-Christian Ganderup)
First the reader is introduced to the classification of swine and how animals commonly used in safety assessment are phylogenetically related to each other; a short overview of commonly used minipig breeds is provided. Subsequently, the biomedical use of swine, dog, and nonhuman primates (NHPs) in 4 major regions (Canada, European Union [EU], Japan, and the United States) is described and an assessment of the minipig’s ability to predict clinical outcome using cases studies is conducted (note 1).
The aim is to demonstrate that swine have a long history of use in biomedical research, and minipigs especially have been used extensively in safety assessment of new medicines and should, in fact, be considered alongside dogs and NHPs when selecting nonrodent species.
Classification, History, Breeds, and Basic Characteristics
Classification and Phylogeny of Swine
Swine, both domestic pigs and minipigs, are mammals belonging to the order of even-toed ungulates (Artiodactyla), meaning they are hoofed animals. Current classification places swine in the suborder of Suina where Suidae is 1 of 3 families. The family Suidae has 5 distinct genera, Sus, where modern swine are classified, is 1 of them.
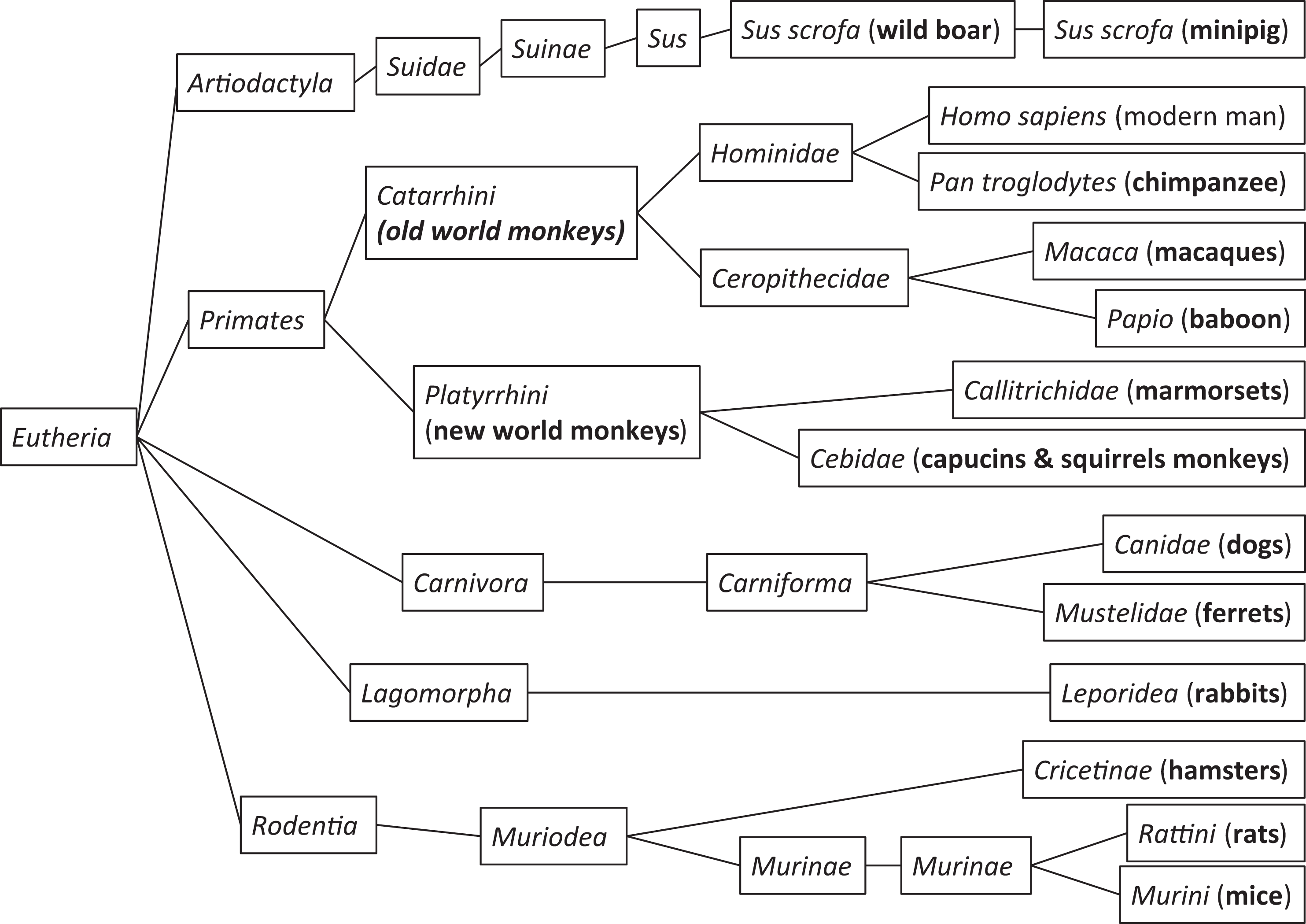
Schematic of the relationship between various species used in safety assessment. Man is included for reference only. All pigs and minipigs extend from
Minipigs breeds
There is no accurate account of the number of minipig breeds in existence throughout the world, the most comprehensive and up-to-date publication from 2011
4
names 16 breeds but also stresses that it is not exhaustive; numbers exceeding 20 have been reported.
5
The number of breeds actually used in toxicity testing is even smaller (Table 1). Requirement for large homogenous groups, availability of extensive background data, and good genetic management of breeding stock are key reasons for this; furthermore, Association for Assessment and Accreditation of Laboratory Animal Care International accreditation is also oftentimes demanded, and only few breeds meet all these criteria. For purpose of classification, all minipig breeds are classified as
Breeds of Minipigs in Europe, Japan, and North America.

The different strains of minipigs do vary in some of their basic characteristics (Table 2). The most stable denominator is age of sexual maturity that is comparable across the different minipig breeds. Weight at which sexual maturity is reached and weight of the fully grown animal varies significantly between breeds; as do the color of the skin and hair covering. Sexually mature animals are preferred in toxicity studies and the minipig does reach sexual maturity significantly earlier than other commonly used nonrodents.
Basic Characteristics of Minipig Breeds, Dogs, Marmosets, Cynomolgus, and Rhesus Monkeys Used in Safety Assessment.a
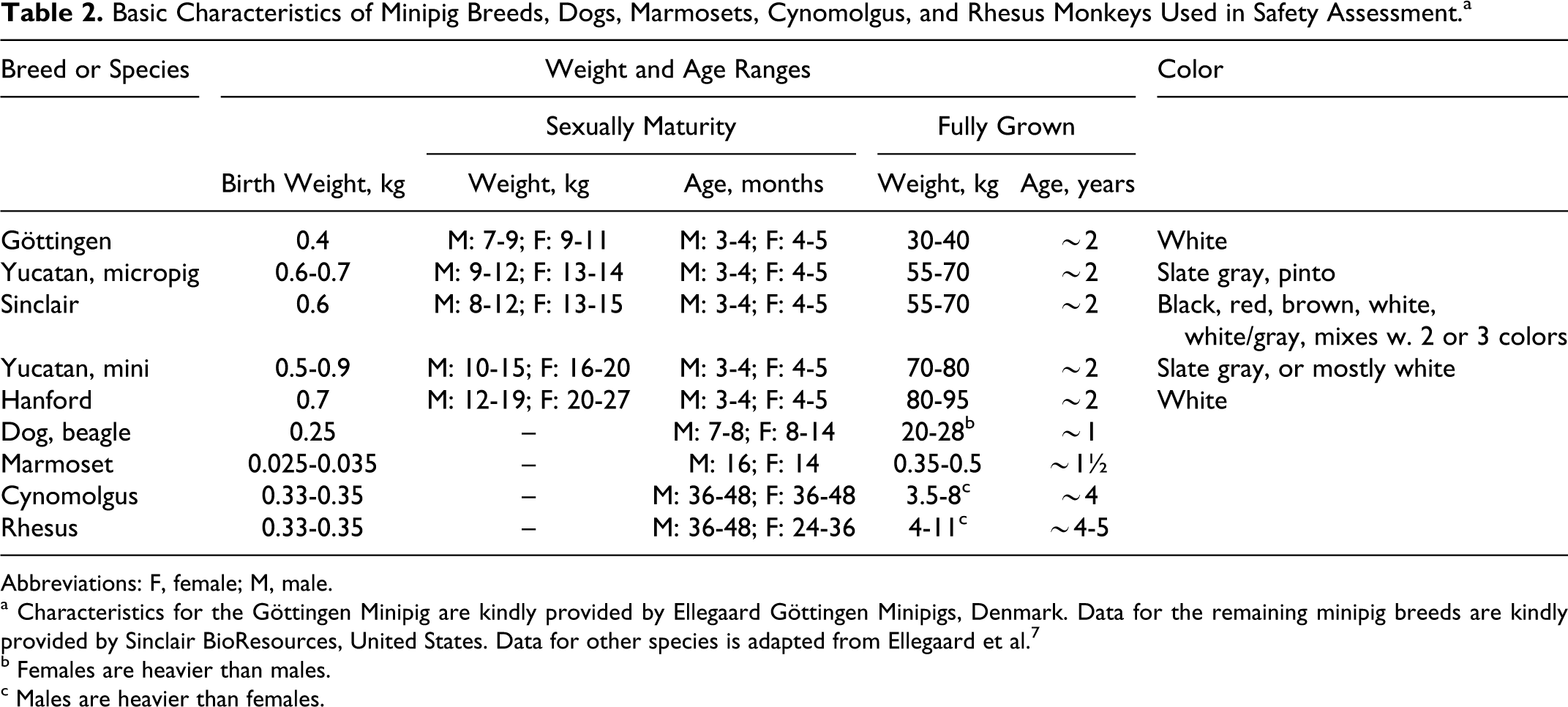
Abbreviations: F, female; M, male.
a Characteristics for the Göttingen Minipig are kindly provided by Ellegaard Göttingen Minipigs, Denmark. Data for the remaining minipig breeds are kindly provided by Sinclair BioResources, United States. Data for other species is adapted from Ellegaard et al. 7
b Females are heavier than males.
c Males are heavier than females.
Use of Swine in Early Biomedical Research
The use of animals to learn about the human body is not new. Most notably, Flemish anatomist Andreas Vesalius (1514-1565) and his famous work
Basis for Assessing Suitability and Relevance of a Species
Having outlined the classification of swine and introduced the different breeds of minipigs, we now turn the more concrete task of assessing the suitability and predictivity of the minipig in safety assessment. Relevance and suitability of an animal species or breed for scientific purposes can be ascertained in several ways; here 3 approaches are combined: (1) the extent of use of pigs in biomedical research across 4 major geographical regions with significant pharmaceutical research; (2) overview of number of publications/year where domestic pigs or minipigs were used; and finally (3) comparison of predictive value of minipigs in safety studies employing various routes of administration. Taken collectively, the 3 reasons mentioned above support the claim that swine are a suitable and relevant nonrodent species; for the relevance of minipigs in toxicology the predictive value (reason #3) is particularly relevant.
Data Collection
For each of the 3 points mentioned above, a short description of how data were gathered is provided.
Use of Dogs, Pigs, and NHP’s in Canada, EU, Japan, and the United States
Biomedical research only uses any given species if it is scientifically relevant and is likely to produce the answer/answers sought after. Implicitly this addresses questions such as similarity to human anatomy, physiology, biochemistry as well as suitability and predictivity of the species selected to mention some. This is why this overview of use of swine supports the statement of this publication that swine are relevant models in biomedical research (predictive power of minipigs in toxicology is covered later in the case studies). Data for the number of laboratory animals used in Canada, EU, Japan, and the United States is presented. The data available are fragmented because the type of data and frequency of collection and reporting varies significantly between regions.
There are several limitations of the data and some significant differences in the form of registration exist, they are addressed now. These data do not specify the aim of the experiment (except EU), distinguish between pigs and minipigs (except Canada), the frequency of data collection varies greatly between the 4 regions. Furthermore, the EU has expanded in size (from 12 to 27 member countries) that skews the retrospective use of the data to identify trends. France reports data 1 year earlier than the rest of the EU, and finally not all countries have reported since Eurostat (EU’s Statistics Office) made it mandatory. It is also important to note that data from the Canada, EU, and Japan describe the number of animals used (head count), but an animal may be used for more than one experiment, for example, a minipig used in 4 different pharmacokinetic studies counts as 1 minipig in the statistics. In the United States, the number of experiments using animals are reported (study count), thus as a single animal used in 2 separate studies will count as 2 animals in the statistical report. Therefore, the data do not present exactly the same information but both do support the argument this article makes.
Countries like China, India, and Pakistan use a sizeable number of dogs, swine, and NHPs for biomedical purposes, but such data are, at best, difficult to access, and accuracy cannot be ascertained, hence they are not included. Despite these shortcomings, the comparison of usage of the 3 main nonrodent species still provides insights to the extent of their use in those regions.
Publication History/Extent of Use
PubMed was used for the literature search. Data on total numbers of citations for pigs and minipigs (Figure 4) were found by repeating searches on PubMed for each publication year.
Case Studies
The case studies and discussions are gathered from the published literature and are discussed below in detail. Routes of administration employed in minipigs will also be discussed.
Discussion
Use of Dogs, Swine, and NHP’s in Canada, EU, Japan, and the United States
Data for swine, dogs, and NHPs are reported by region (see Figure 2) and as grand total for all regions with distribution among the 3 species (see Figure 3). The total use (all regions combined) of swine and NHPs has increased by 15% and 33%, respectively, whereas the use of dogs has decreased negligibly (2%) from 2002 to 2008. There are striking differences between the patterns of use of the 3 species among the regions. Canada and the EU have similar patterns of use of the 3 species, while Japan and the United States have altogether distinct patterns.
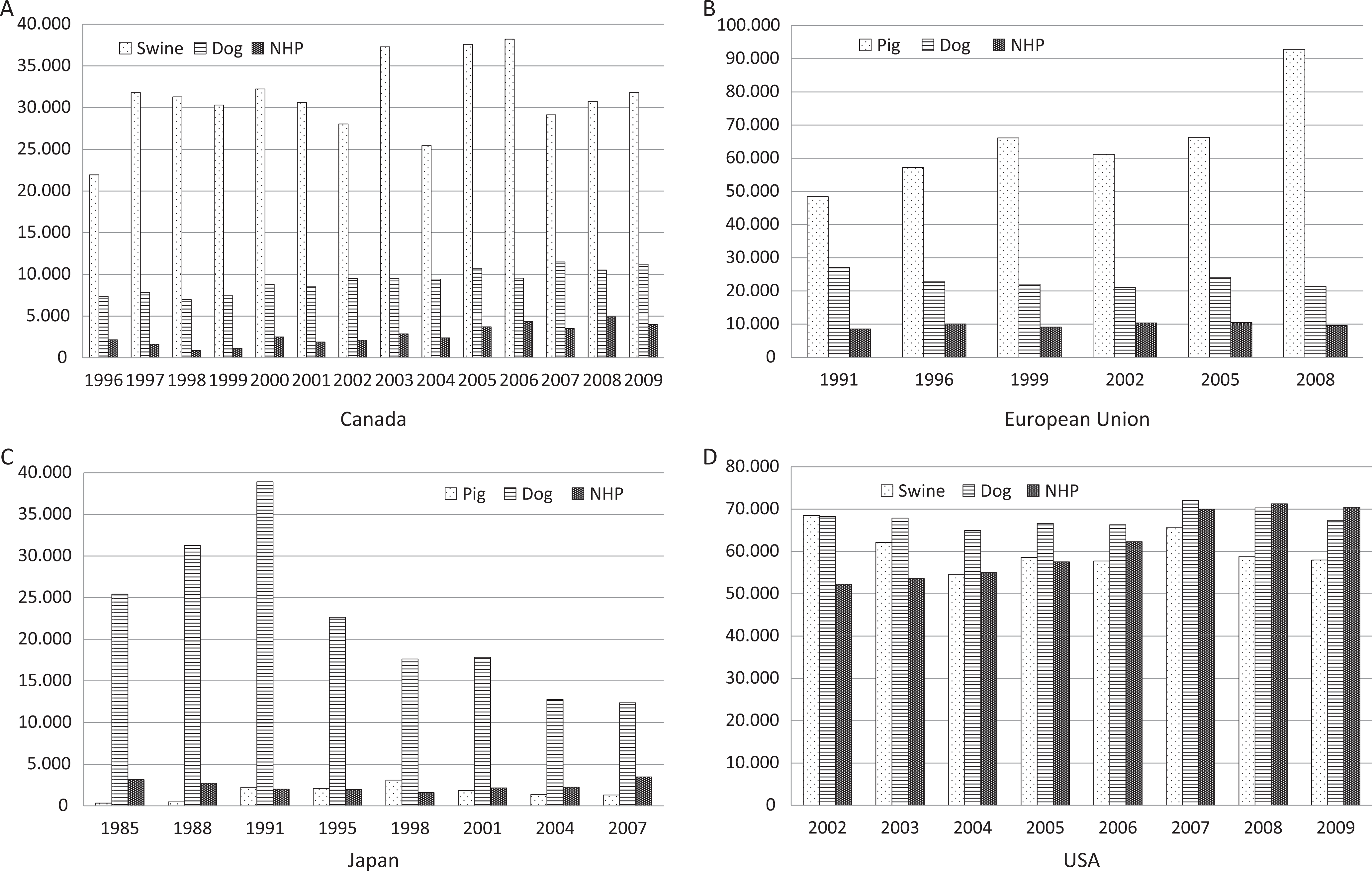
Use of dog, pig, and nonhuman primate (NHP) in Canada (panel A), European Union (panel B), Japan (panel C), and the United States (panel D). Sources used are Canada: the Canadian Council on Animal Care's Web site. 10 EU: Reports from the European Commission on the use of experimental animals in the European Union. 11 United States: Animal and Plant Health Inspectorate under the US Department of Agriculture (USDA). 171 Japan: Japanese Association for Laboratory Animal Science 172 data kindly extracted by Dr Naoki Hayashi, OYC, Japan.
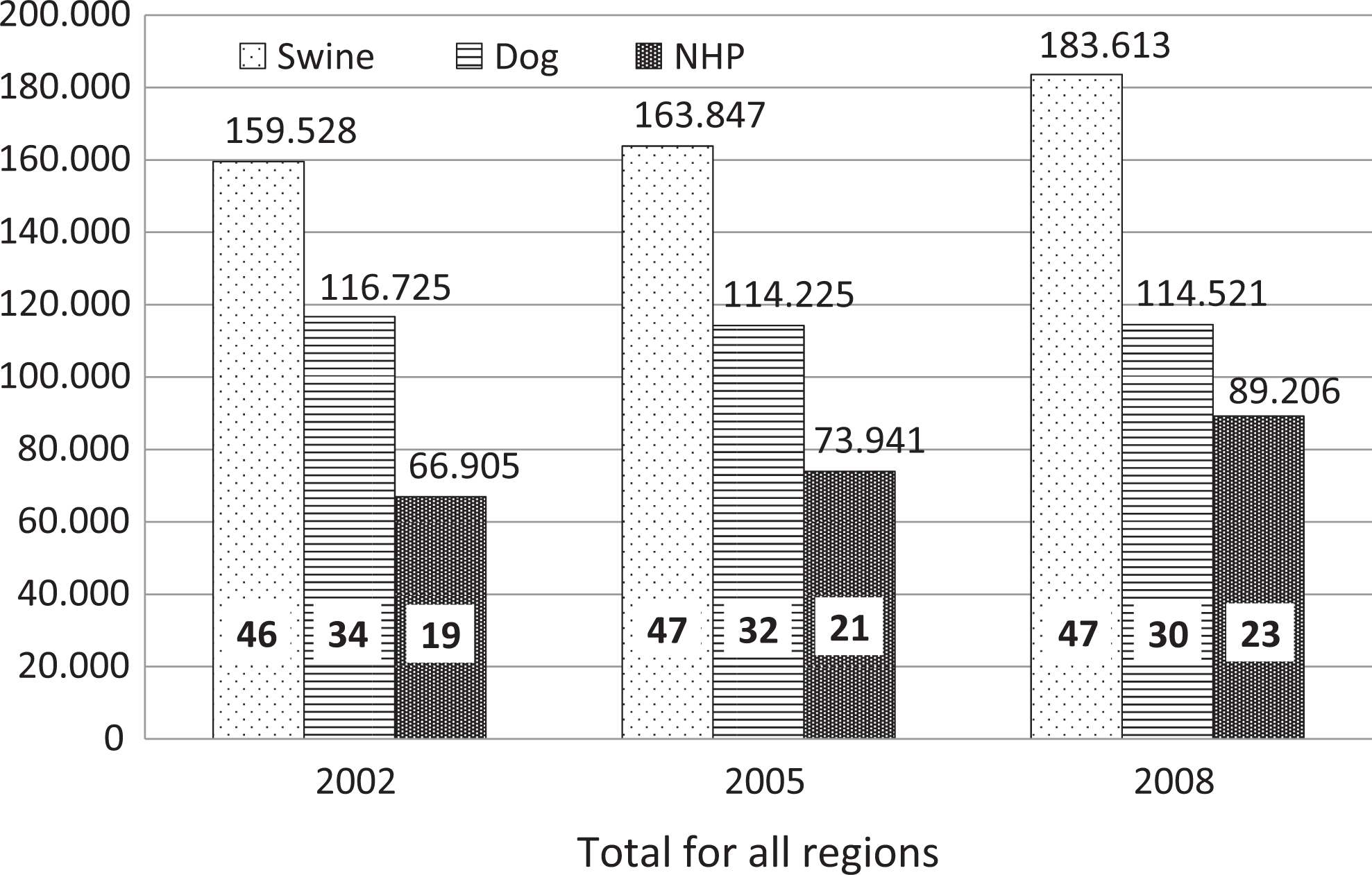
The total use of dog, swine, and nonhuman primate (NHP) in 4 regions for the years 2002, 2005, and 2008. From 2002 to 2008, the use of swine increased by 15% and the use of dogs was more or less constant (down 2%), while the use of NHPs grew 33%. The years depicted are the only years where data were available in all regions. Numbers above columns is the number of animals; numbers inside columns is percentage of a given year, total for each year equals 100%.
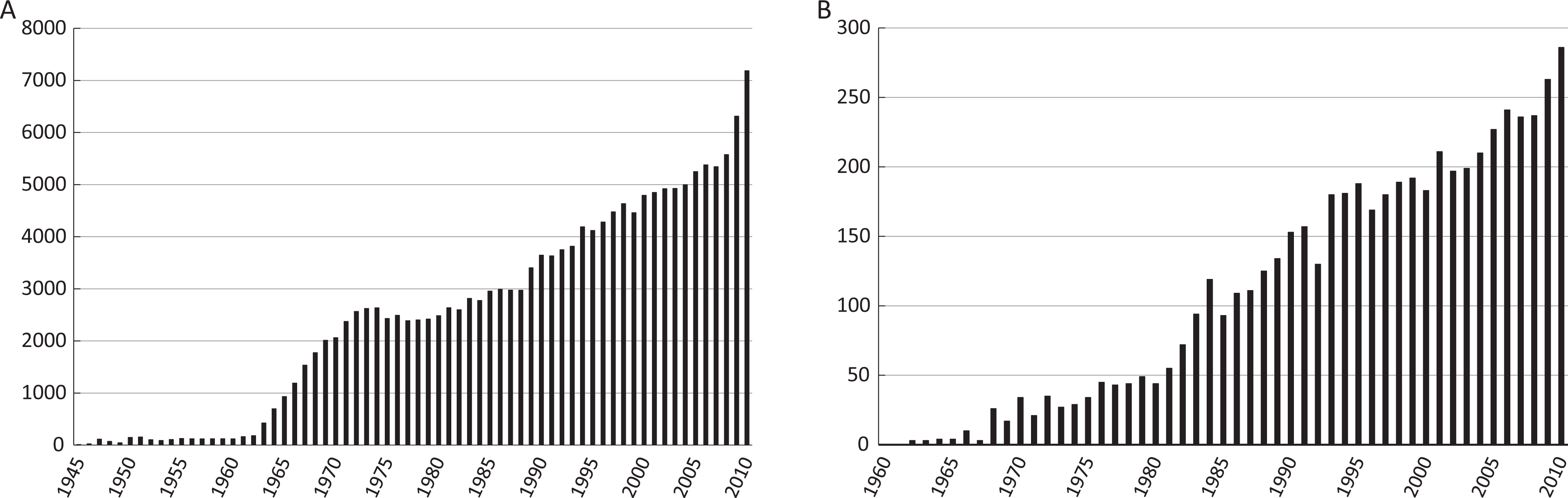
Number of publications per year using domestic pigs (panel A) and minipigs (panel B) as research model. The total number of publications cited on PubMed (including 2010) pertaining the domestic pigs and minipigs are 160 953 and 5475, respectively. For both graphs an increase in use over time is evident.
The EU has increased pressure to reduce the use of NHP’s that lead to a proposal for banning the use of great apes (chimpanzees, bonobos, orangutans, and gorillas) in 2007 12 and to the adoption of the new Directive 2010/63/ which increases the requirements for scientific scrutiny before allowing the use of other NHPs.
Pressure to move away from the dog, a companion animal, is rising in Europe (see eg,
Japan is unique in that dogs are used to a much greater extent than swine and NHPs. Use of dog has decreased dramatically over the past 2 decades (38 915 in 1991 vs 12 376 in 2007) and has not been replaced by use of minipigs or NHPs. Swine are only used in very small numbers in Japan, the reason for this is not known.
In the United States, 3 observations can be made (1) with the exception of 2007 and 2008, the use of dogs is fairly constant, (2) the use of swine has gone down 15% from 2002 to 2009, (3) the use of NHPs has increased by 35% over the same period. The increase in the use of NHP may well be caused by the rise in the development biopharmaceuticals/large molecules (Figure 3).
In Canada and the EU, the patterns of relative use are comparable; furthermore, the use of NHPs in the EU is constant, as opposed to Canada, where it has increased.
The extent of use of swine, dogs, and NHPs in Canada, EU, Japan, and the United States has been reviewed. The number of swine used in total across the regions is higher than dogs and NHPs, suggesting this species is indeed a relevant model in biomedical research and needs to be considered when selecting which species to use.
Publication History/Extent of Use
Another approach to demonstrate scientific relevance of an animal species is the extent to which it has been employed in research to gain understanding on basic anatomy, biology to issues of safety assessment of chemical and biological structures. The first publication pertaining to pigs referenced on PubMed dates from 1851 14 while the first minipig article on PubMed dates from 1954. 15 The references do include agricultural and veterinary research and their relevance for toxicology can be debated. However, in providing a bird’s-eye view of the use of domestic pigs and minipigs in biomedical research, it is deemed reasonable to include such publications.
Figure 4 shows the number of citations per year employing minipigs or domestic pigs; for both instances the scientific output has increased steadily. The total number of publications citing minipigs is lower than for pigs. The 2 main reasons for this is that minipigs have only been available since the 1960s, are more expensive than farm pigs, and acquiring minipigs is, at least looking from a historical perspective, not as easy as acquiring pigs. Equally important, vast amounts of data from investigations employing minipig conducted in pharmaceutical companies and contract research organizations (CROs) remain unpublished, which impacts especially minipigs, and is unfortunate as sharing data would reduce repetition of experiments (both futile and valuable), and reduce animal usage.
A large volume of literature employing swine to learn about human health and safety exists, and for both the domestic pig and the minipig there is a clear increase in the number of publications.
Case Studies
Minipigs has been used in a number of marketed drug products for both safety and efficacy assessment employing various routes of administration and its predictive value is comparable or better than what is reported for other nonrodent species. The suitability of minipigs in safety assessment and potential issues related to its use have been voiced many years ago 16,17 and a systematic approach to address suitability and considerations for use have been undertaken, as exemplified by the use of minipigs to address immunotoxic potential of food additives. 18 Literature describing in broad terms the use of minipigs in toxicology from different study types is abound. 7,19 –29 However, they all fail to address the predictive value of the minipig. Only 2 major publications accomplishing this have been identified: one deals with pharmaceuticals 30 ; while the other deals with agrochemicals, food additives, and industrial chemicals 31 (see below). Other articles do address the translational aspect but are limited to case studies which by their very nature compare one (or few) compounds or models, 32 –35 or they are very broad in their coverage not providing detailed discussions on each case. 36,37
This section presents more recent information on marketed drug products where the minipig was employed in the nonclinical development and compares the predictive value of the minipig to that of other nonrodents, before concluding by discussing the historical development routes of administration used in minipigs over the past two decades.
Marketed products (Table 3) cover more than 20 indications and 6 routes of administration (dermal, intradermal, oral, intravenous, subcutaneous, and intramuscular). The range of indications and study types also support the usefulness of the minipig both from a safety and efficacy perspective. A few nonstandard study highlights include the use of the minipig as an animal model to assess carcinogenic potential of a topical product; an embryo-fetal development/segment II study; and a product indicated for the treatment of renal carcinoma (from Ganderup et al 30 ).
Indications for Which Minipig Was Used to Varying Levels in Toxicological and/or Pharmacological Assessment.a
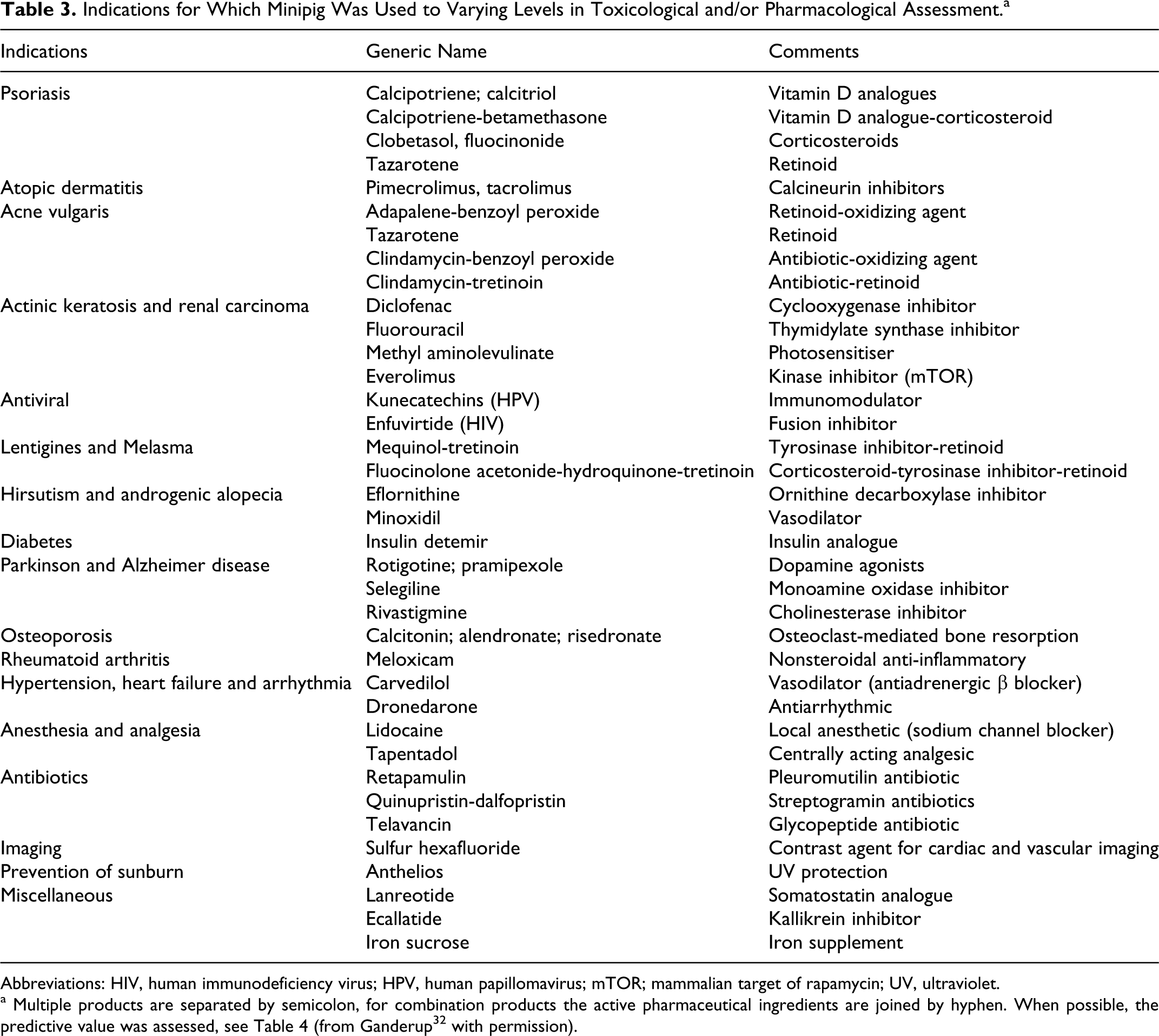
Abbreviations: HIV, human immunodeficiency virus; HPV, human papillomavirus; mTOR; mammalian target of rapamycin; UV, ultraviolet.
a Multiple products are separated by semicolon, for combination products the active pharmaceutical ingredients are joined by hyphen. When possible, the predictive value was assessed, see Table 4 (from Ganderup 32 with permission).
The assessment of predictive value of the minipig in safety and/or efficacy assessment of 43 marketed drug products is summarized in Table 4. The comparisons are made based on information extracted from the US FDA (Drugs@FDA) and the European Medicines Agency (European Public Assessment Reports [EPARs]) and analyzed using PharmaPendium and presented in-depth in Ganderup’s study. 30 Nonclinical and clinical data were compared for each product. The type and detail of data available varied between products and predictive value was only evaluated when there is overlap in data from clinical and nonclinical studies, that is, grading was not undertaken if data were insufficient. Three grades of predictive value were given (good, acceptable, and poor) and of the 27 drugs where minipig were used for safety assessment, only 11% nonconcordance was observed, that is, in 11% of cases adverse reactions seen in patients were not predicted from minipig studies. Olson et al 37 reports 30% nonconcordance between animal studies (rodent and nonrodents combined) and clinical studies, which increases to 37% nonconcordance when looking at nonrodent studies alone (primarily dog), a level significantly higher than what is reported for minipigs. Olson et al also state that nonrodent models employed (dog, NHP, rabbit, guinea pig, rat, and mouse) fail to accurately predict cutaneous reactions in humans; no mention of domestic pig or minipig is made. Pigs and minipigs have established themselves as the species of choice for the testing of topical products. 38 The minipig has good concordance to human responses, as mentioned above, and if the investigation is narrowed even further to include only safety assessment of dermally and intradermally administered products, a strong concordance from minipig to human reactions is still evident, which is in stark contrast to Olson et al 37 who report poor concordance of animal models for the topical route.
Predictive Value for Safety and Efficacy of the Minipig Based on an Analysis of 43 Marketed Drug Products.a
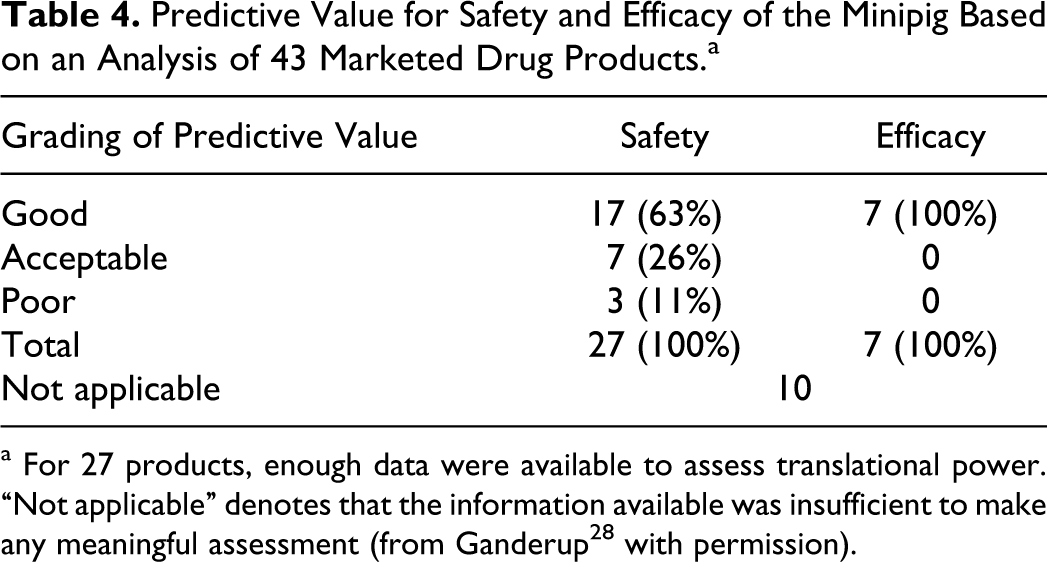
a For 27 products, enough data were available to assess translational power. “Not applicable” denotes that the information available was insufficient to make any meaningful assessment (from Ganderup 28 with permission).
The routes of administration employed in minipig studies have been reported by Ganderup, 30 in a survey of industry reported by Bode et al 23 and by Makin. 39 Other sources of such information could not be identified but may well exist. Table 5 shows that the minipig has moved away from being predominantly a dermal model to being used predominantly in oral toxicology studies, although dermal administration still accounts for one-third of all studies.
Route of Administration (Dermal Versus Nondermal) Used in Minipigs Over Time.a
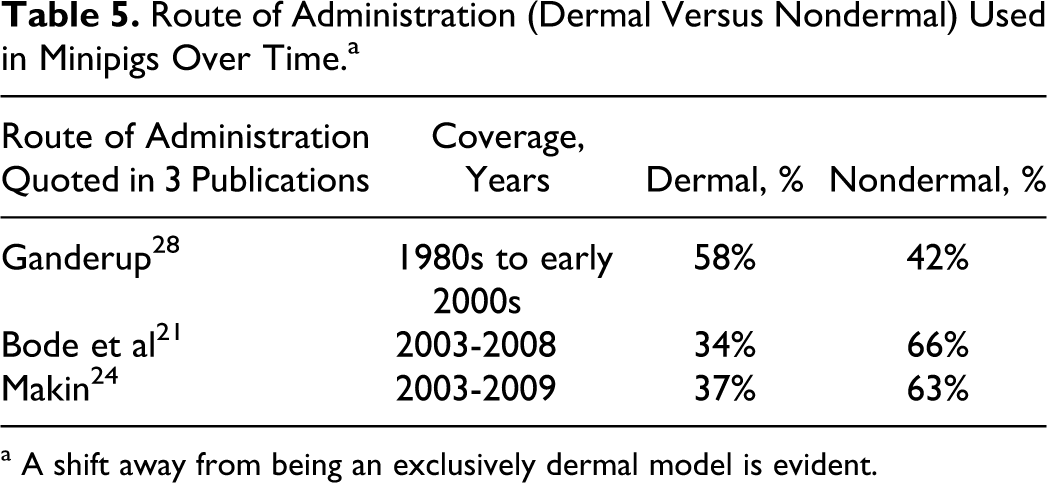
a A shift away from being an exclusively dermal model is evident.
Commonly Occurring External, Visceral, and Skeletal Malformations and Variations in the Gottingen Minipig (Gestation Day 110).a
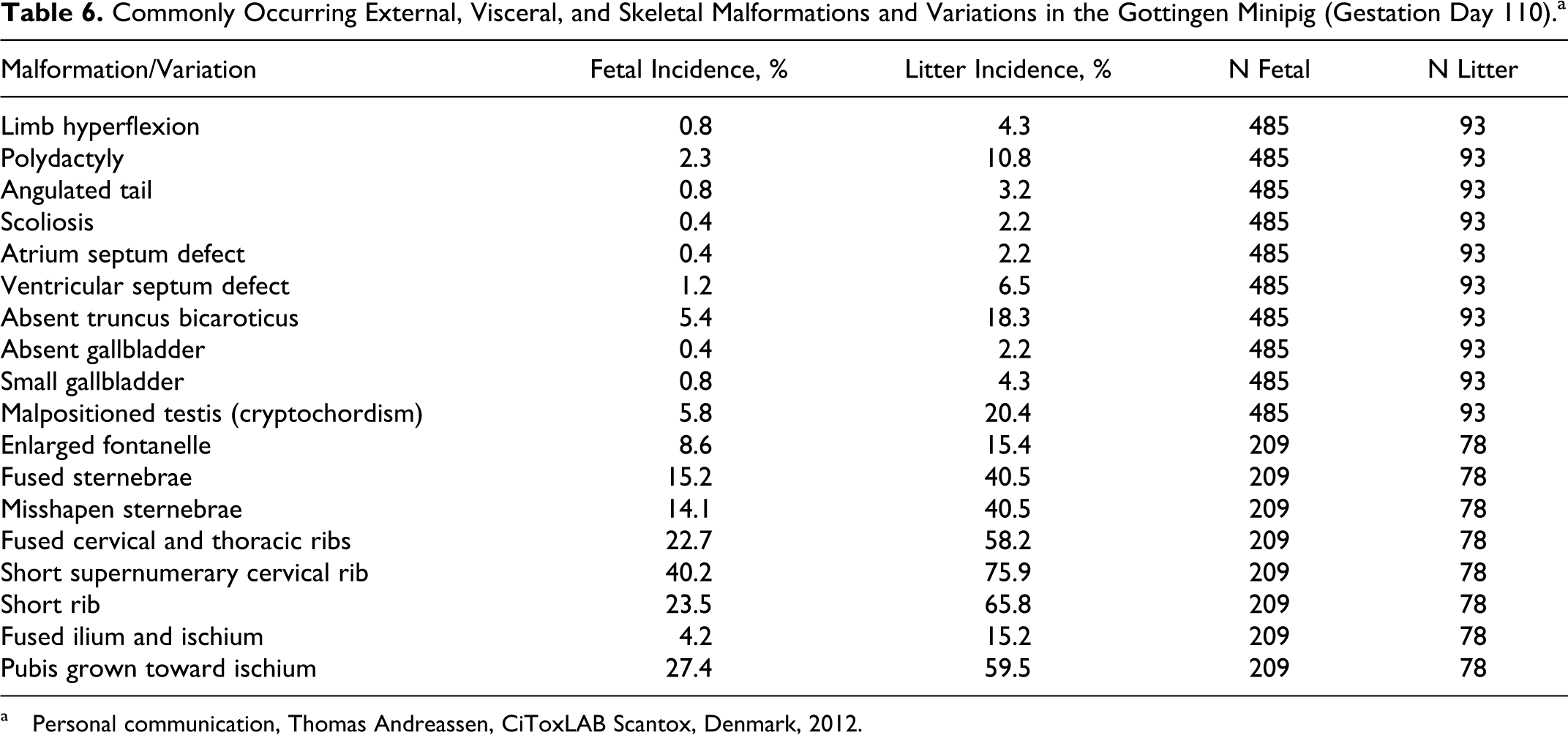
a Personal communication, Thomas Andreassen, CiToxLAB Scantox, Denmark, 2012.
Bode et al 23 conducted a survey of industry asking what routes of administration were employed in minipig studies by 22 pharmaceutical companies and 1 CRO, while Makin 39 includes general toxicology studies. One possible way to interpret this shift is that the industry has “operationalized” what this article claims: the minipig is a relevant and suitable model in safety assessment and all routes of administration can be utilized in the minipig.
Conclusions
There is evidence in the scientific literature supporting the claim that the minipig is a nonrodent species that has good predictive power and has a proven track record in supporting the nonclinical development of new medicines. The pattern of routes of administration used in minipig studies has changed and the fact that the dermal routes is giving way to nondermal routes of administration underlines the claim that the minipig is more than a good model for topical studies.
While the minipig has gained greater acceptance among nonclinical researchers, by virtue of history and habit, the dog still appears to be the nonrodent species used more often in the pharmaceutical industry. To advance even further, the use of domestic pigs and minipigs in safety assessment one should aim at filling gaps that still exist, such as development and qualifications of nonstandard biomarker (although most standard biomarkers are well established in minipgs 40 ), development of standardized kits and reagents for analysis of various parameters relevant for the toxicologist, and a more detailed analysis of its use in pharmacology.
The Use of Swine in Food Safety Assessment, Agrochemicals, Pesticides, and Other Nonpharmaceuticals (Warren Harvey)
The safety assessments of pharmaceutical and chemical compounds are typically performed in 2 animal species—1 rodent and 1 non-rodent species. The selected nonrodent toxicology species is usually determined based on pharmacokinetic, pharmacodynamic, or metabolic similarities based on the anticipated human response. It is recognized that the minipig offers distinctive biological similarities as a nonrodent model other than the dog or NHP in both the pharmaceutical and nonpharmaceutical sectors.
Swine are similar to man in many aspects in relation to skin morphology and physiology, cardiovascular anatomy and physiology, digestive anatomy and physiology, dental characteristics, renal morphology and physiology and eye structure, and visual acuity. 23 Swine are now established as models for man in a number of biomedical studies as well as in cardiovascular, pulmonary, gastrointestinal (GI) tract/nutrition, renal, immunological, metabolic, embryological/fetal, neonatal, and integumentary fields. 41
Swine are omnivorous mammals and have a remarkable similarity to man in GI tract anatomy, physiology of digestion, and associated metabolic processes. Thus, this species is often used in pediatric and biomedical research especially in the field of nutrition and other associated areas including digestion, absorption, metabolism, and immunology. 42
Comparative GI Tract Anatomy of Swine and Humans
Despite the many obvious external morphological differences between humans and swine, their internal anatomy and physiology are very similar. 43,44 Anatomically, there are differences such as the division between the duodenum, jejunum, and ileum is not as distinct in the porcine small intestine as they are in humans. The stomach is similar to other monogastric species, with the exception of the mucosa and a muscular outpouching of uncertain function termed the torus pyloricus. The most noticeable macroscopic difference between the human and porcine is the length of the intestine. The small intestine of adult domestic pigs is around 15 to 22 m, while the large intestine has an average length of 4 to 6 m. 43 The small intestine of a human adult averages around 5.5 to 7 m, while the large intestine is around 1.5 m in length. Another distinct difference between humans and swine is the spatial arrangement of the intestine within the abdominal cavity (Figure 5), particularly that of the large intestine. The greater proportion of the pig large intestine, consisting of the cecum and the majority of the colon, is found in a spiral conformation arrangement in a series of clockwise and anticlockwise coils, known as the spiral colon. 45
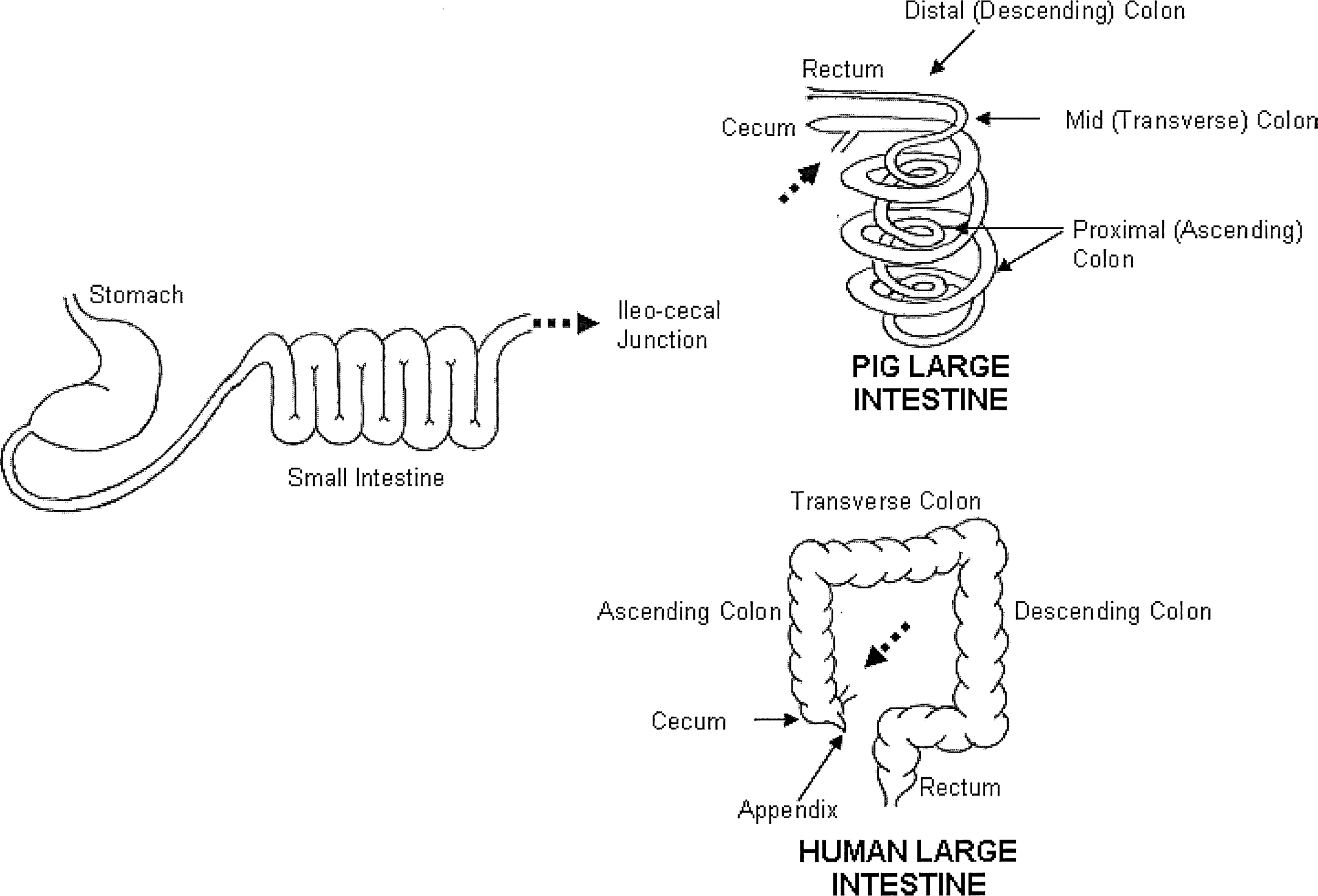
Comparative structure of the intestine of humans and pigs. The stomach and small intestine are very similar between humans and pigs; the large intestine is strikingly different in its conformation but not functionality. Taken from Patterson et al 45 with permission of the authors and publisher.
Even though there are differences in absolute intestinal length and the internal spatial layout of the alimentary canal, the porcine digestive and metabolic processes function in much the same way as those of a human, and digesta transit times are also similar between the 2 species. 46,47 The GI transit time in young domestic pigs is considerably faster compared with adult pigs. 48 Swine nutritional requirements and physiology of digestion are very similar to those of humans. 49 Both swine and humans are highly dependent on dietary quality, because symbiotic microorganisms in the gut play a relatively minor role in modifying ingested nutrients. 43
Swine in Nonpharmaceutical Research
The GI tract of the weaning swine closely resembles that of a 2- to 5–year-old child. The rapid growth rate of the domestic pig during the first 6 months of age and therefore, the high nutrient demands, noticeably exaggerate those nutrient demands in relation to humans. For this reason, changes in nutrient availability are readily detected in a growing domestic pig, which make the pig a very sensitive and an ideal animal model for nutritional studies. 173 The requirements of the weanling domestic pig for macro- and micronutrients are similar to or greater than human requirements, 43,50 so the domestic pig is a good model for subgroups of the population with high nutrient demands such as children and pregnant or lactating women. 51 By sexual maturity at approximately 6 to 8 months of age, domestic pig studies can be and are readily conducted covering the growth and developmental phases of pig’s life. Swine are also able to ingest, tolerate, and metabolize fat concentrations similar to those found in the human diet and exceeding human intake. Swine and humans have similar gastric cell types, similar gastric villi structure and component epithelial cell types. 52 This makes swine an ideal model for human nutritional studies investigating the bioavailability and digestibility of various dietary factors in different GI tract compartments.
As described above, the porcine model is particularly ideal for nutrient bioavailability and absorption studies because of the remarkable similarities between swine and humans with respect to their nutritional requirements as well as their digestive and metabolic processes. It is therefore not surprising that researchers are currently using the porcine experimental model to address scientific questions.
Food Additives
Food additives are among the most intensively tested products and are generally characterized by their intended function either as direct or indirect additives. Some of the functional classes found in the diet are direct additives including food colors, antioxidants, sweeteners, preservatives, emulsifiers and stabilizers, thickeners, and flavoring agents. The safety assessment of food additives usually includes the chronic testing in a nonrodent species. The domestic pig and minipig are ideal models for testing additives, rather than the dog or NHP, for the reasons as described above. Domestic pigs have been used extensively for testing food additives as well as provide supporting data in mechanistic studies as part of the regulatory process, for example, many food colors, flavorings, artificial sweeteners, pectins, fat substitutes, gums, and microalgae. Nevertheless, swine are rarely recommended as a potential nonrodent species in the test guidelines but are fully accepted by the regulatory authorities. They are recommended by the World Health Organization in “Guidelines for the Preparation of Toxicological Working Papers” for food additives 53 or contaminants 54 that specify where in the document swine data should be included and also refers to pigs in the appropriate appendix which provides information on how to calculate dietary concentrations of additive for various species. Furthermore, the FDA Food Ingredient Guidelines 55 advocates the use of dogs as the nonrodent species and does not promote the use of swine.
Pigs in Nutritional Research
Fat-Free Macronutrients
Atherosclerosis is associated with hyperlipidemia in humans. 56 Like humans, swine are classified as low-density lipoprotein (LDL) mammals, because LDL is the dominant cholesterol carrier and swine may develop spontaneous atherosclerosis similar to humans. Therefore, swine may be particularly useful as models for the study of human lipoprotein metabolism and cardiovascular disease 57 and given the similarities between human and porcine GI physiology and lipoproteins, porcine are a very attractive model for investigating nutritional effects in the safety assessment of fat-free macronutrients.
Olestra
Olestra (Olean, Procter and Gamble, Cincinnati, Ohio) is a mixture of polyesters formed from sucrose and fatty acids derived from edible fats and oils with similar taste and cooking characteristics to those of traditional fats and oils. Because Olestra is not hydrolyzed by gastric enzymes or absorbed intact from the GI tract, Olestra can serve as a dietary replacement for conventional fats and oils, thereby, contributing no calories or fat to the diet.
Animal and human studies highlighted GI disturbances with Olestra use and as a lipophilic compound, had the potential to interfere with the absorption of lipophilic nutrients or other dietary components, 58 as well as inhibition of the absorption of dietary lipid-soluble vitamins (A, D, E, and K). The weanling domestic pig was selected as the most suitable model because the vitamin stores and nutritional indices of the pig are known to be responsive to dietary change. Thus, any changes in stores and indices as a result of Olestra consumption would be clearly determined in these domestic pig studies. 59
A total of 6 studies as summarized by Peters and coworkers 60 from 4 to 39 weeks duration showed that changes in body stores of fat-soluble vitamins can be produced within the time frames of the studies. As a result of these studies, extra dietary amounts of vitamins A, D, and E were needed to offset the Olestra effects and supplementation of these vitamins was effective under normal and exaggerated intake study conditions. The authors recognized that studies conducted in the weanling domestic pig were able to monitor and assess the major growth and developmental phases as well as maturation in nutritional stores. This is significant when assessing the potential effects of Olestra or any other substance on the nutritional status of infants and young children. The pig studies were shown to be extremely predictive and resulted in the successful approval of the product in 1996 for use in replacing up to 100% of the fat used in the preparation of savory snacks, 61 although not without controversy.
Canola Oil
Canola oil (also known as low erucic acid rapeseed) is marketed for cooking and use in salads and considered as Generally Recognized as Safe by the US FDA. Due to its very low saturated fat and high monosaturated fat content, and beneficial omega-3 fatty acid profile, canola oil is claimed to promote good health. 62 Nevertheless, the US and UK health organizations do not recommend the use of canola oil in infant formulas because infants fed formula might consume higher amounts of erucic acid than would be provided in usual mixed diets, leading to possible accumulation of triglyceride in the heart. 63 High dietary intakes of erucic acid in the rodent can induce myocardial lipidosis and necrosis as well as structural and functional abnormalities of rat mitochondria. 64,65 Newborn piglets, birth weight >1 kg, were bottle fed diet formulas from birth containing various concentrations of canola or canola oil blends (eg, canola with bean, soy, palm, sunflower, and coconut oils) showed potential changes associated with blood phospholipid concentrations as well as the possible relationship of heart triglyceride accumulation and myocardial lipidosis. 64,66,67 Indeed, these studies demonstrated that induced erucic acid myocardial lipidosis in newborn domestic piglets was more severe than in weaned pigs. 68 More importantly, these studies confirm that cardiovascular toxicity with erucic acid was better modeled by the pig than the rat and that the domestic piglet was much less prone to oil-induced lipidosis and fibrosis than the rat. Unquestionably, swine is the animal model of choice in assessing the safety of these substances and accepted by regulatory authorities, as rats have shown that they are less able to digest vegetable fats (whether or not they contain erucic acid) than humans and swine.
Chemicals
The use of nonrodents in the safety testing of chemicals is not anticipated as the rodent is regarded as the species of choice. The data requirements for the classification, packaging, and labeling of substances as outlined in the Annexes to Directive 92/32/EEC, 7th amendment to Directive 67/548/EEC 69 specifies the use of one species and this is the rat as default.
Agrochemicals
As defined under the Annexes of the Directive 91/414/EEC 70 and Directive 94/79/EEC, 71 both the rat and dog are the recommended species of choice with no suggestion that swine or minipigs to be used. The regulations for a 90-day study in the nonrodent as outlined in Annex V to Directive 79/831/EEC 72 and the OECD Test Guideline 409 73 recommends the beagle dog as the nonrodent species but recognizes swine and minipigs as an alternative test model. However, the dog remains the most commonly used nonrodent species for pesticide or chemical testing. Minipigs are not the primary nonrodent species in standard regulatory toxicology studies for pesticide registration but would be considered acceptable with justified scientific rationale. 41
A literature review identified swine use as a “supporting” model included as part of the safety assessment of triflumuron, a benzoylurea insecticide that interferes with chitin synthesis
74
and quizalofop-
Organophosphorus Research
Swine have been used in organophosphorus (OP) research for decades 80 as more desirable to measure numerous physiological and biochemical parameters, which are difficult in small animals. Swine, compared to humans, express a similar clinical syndrome after poisoning 81 and have a similar cholinergic nervous system. 82
Administration of a single oral dose of tri-
Paraoxon, the active metabolite of the insecticide parathion, is one of the most potent AChE-inhibiting insecticides available and so rarely used due to the risk of poisoning to humans and other animals. A comparative study utilizing both the White Landrace pig and the Göttingen minipig to investigate the kinetic properties of swine in association with the inhibition of paraoxon demonstrated similar kinetic properties of AChE activities between White Landrace pig and minipig, providing a kinetic basis for extrapolation of data to humans. 85
Dimethoate, a commonly used OP insecticide in agriculture 86 is unlike any other OP pesticide poisoning, in that the majority of dimethoate deaths result from delayed cardiovascular shock after early respiratory failure. The reason for this clinical difference is not known, but hypotension is an important factor and the pathophysiology of dimethoate poisoning in the minipig was investigated. 87
Dimethoate poisoning in minipigs is similar to human self-poisoning characterized by early respiratory failure, distributive shock and lethal cardiovascular collapse, and severe AChE inhibition. 88 Anesthetized Göttingen minipigs treated orally with toxic doses of standard agricultural dimethoate, 40% emulsifiable concentrate or dimethoate 25% active ingredient in ethanol caused severe hypotension due to peripheral vasodilatation that was similar to human poisoning. 87
Conclusion
Swine may not as yet command the confidence of regulators as do the dog and macaque NHP in toxicology testing of certain substances but swine have been extensively used in testing food additives and substitutes in foods as well as human nutrition. There remains a reluctance to accept this species in the nonpharmaceutical sector and are rarely recommended in the guidelines.
Extensive studies in the domestic pig and minipig conducted to international regulatory guidelines have established whether or not additives are safe for humans. International guidelines as well as US and EU regulations on additives recommend the dog as first choice nonrodent species and not swine. Swine are not only able to confirm safety of the test substance in question but also the mechanisms of action. The dog is not anatomically or physiologically similar with respect to the digestive tract but remains accepted by international regulatory bodies for all nonrodent testing.
The domestic pig or minipig would be considered by the agrochemical sector as a nonrodent species if there were scientific justification, but data requirements for agrochemical products state the dog as the preferred nonrodent species. Given the complexity of worldwide registering of an agrochemical, nonpharmaceutical companies require minimal testing in a nonrodent species that will satisfy all agencies, in the favored dog species, to avoid any further testing or delayed marketing. To this end, there is a lack of enthusiasm for the minipig use from both a historical and financial perspective in the agrochemical industry. However, as these concerns do not involve a fundamental biological objection, no regulatory agency should dismiss the minipig, if scientifically based arguments and data support the minipig as the most appropriate nonrodent species.
Use of Minipigs in Safety Assessment of New Medicines (Jens Thing Mortensen)
Introduction
Over the last 10 to 20 years, the use of minipigs as a nonrodent species for safety testing of pharmaceutical drugs, food products, and related products increased steadily. Thus, in addition to the “traditional” rat + dog or rat + non-human primate (NHP) combination for a toxicology program, the minipig should also be considered for safety testing of pharmaceutical products.
It is well documented that domestic pigs—and minipigs—are physiologically and anatomically close to humans in many aspects, for example, both species are omnivores, the GI (see elsewhere in this publication) and cardiovascular systems 89 have many morphological and functional features in common and the skin is of similar structure in the two species. 90,91 Additionally, the porcine metabolism (see eg, 92 ) and the immune system (see eg, 93 –95 ) have been documented in great detail, allowing for a scientifically qualified choice of nonrodent species for a toxicology program. If one takes into account the increased quality of the breeding and supply systems, standardized procedures for the care and use of the minipig, including feeding, dosing, sampling, anesthetic and surgical procedures, and the fact that a substantive amount of background and validation data now exist, the minipig has the potential to improve the predictivity of a toxicology program and should be considered on par with the “traditional” nonrodent species, dog and NHP, for regulatory safety testing of pharmaceuticals. The choice must be based on scientific evidence and selection criteria to ensure that the most relevant nonrodent species is used.
Here a general view of the use of minipigs in safety evaluation of pharmaceutical drug candidates is provided. Food ingredients and other chemical products are covered elsewhere in this publication. For the different areas of use, the biological features that make the minipig specifically relevant and useful will be mentioned, important practical and technical aspects of the study designs will be discussed, and relevant pros and cons for the use of the minipig in comparison with the other nonrodent species will be pointed out. This article is not an exhaustive list of all references within the subject matter but rather a selection of key references which underline the various points made.
The ADME Studies
Extensive literature about the pig and minipig in absorption, distribution, metabolism, and excretion (ADME) studies has been published.
29,96
–99
Only key points of relevance to the toxicologist involved in drug development are presented here and a brief outline of important phase 1 and phase 2 reactions is given. In minipigs, the metabolic activity (relative to body weight) is higher than that of man by a factor of 2 to 3.
95
There is intrinsic activity of several of the pivotal cytochrome P450 isoenzymes (CYPs) in several tissues in the minipig, including the liver, the skin, and the gut. The CYP3A and CYP2E both have substrate specificity and activity very similar to humans,
29,100,101
while reports state that the domestic pig and minipig have very little or no CYP2D activity.
102
The CYP activity in the skin and liver of several species has been described by Rolsted et al.
103
The CYP activity in dermal microsomes, which may be of significance for the transdermal delivery of pharmaceuticals, was described to be in the order of 0% to 2% compared to hepatic microsomes. The composition of CYPs in dermal microsomes was described to differ from hepatic microsomes, and there are also differences between the species. The presence of esterases and other enzymes in skin may also be of importance for the activation or detoxification of some pharmaceuticals, for further discussion see for example Jewell et al,
104
Preusse and Skaanild,
97
and Makin et al.
91
Glucuronidation and acetylation are the most important phase 2 reactions present in domestic pigs and minipigs, but significant glutathione-
Dermal Toxicology
It is no exaggeration that the use of minipigs in dermal toxicology is the minipig’s “claim to fame” within regulatory safety testing. By simply looking at the skin of humans and swine, it is obvious that the skin in 2 species share visual properties. The skin is fairly thick and has sparse hair covering, it is carved by fine intersecting lines and may or may not be pigmented, depending on breed. 106 –108 In both species, the texture and thickness of the skin vary according to the body site. Under the microscope, skin from the 2 species also appears quite similar; the epidermis has approximately the same number of cell layers and is of similar thickness in swine and humans (approximately 70-140 μm in the swine, compared with 50-120 μm in humans). 109 In particular and importantly, the stratum corneum, being the main barrier for transdermal entry of chemical substances, has a similar structure in both species, making the minipig a good model for measurement of transdermal penetration and permeation of chemicals and drugs applied to the skin surface of humans. 110,111 In both species, the epidermis contains the antigen-presenting Langerhans cells of the immune system and pigment-producing melanocytes. The dermis contains collagen and elastic fibers and is vascularized in both the species. The structure of the dermis is slightly firmer in swine and the vasoconstrictor capability in the swine is more developed, 106 which should probably be seen in connection with its role in thermoregulation. Accordingly, the skin of swine generally has no eccrine sweat glands and the swine relies on vasoconstriction and external cooling to maintain a constant body temperature. 91,106 The surface pH of swine skin is slightly higher than in human skin. 112 –114
The minipig is a useful animal model for investigation of both local tolerance and systemic toxicity of dermal pharmaceutical products. 91 For local tolerance testing of dermal products, the test and reference substances are applied to standardized test fields (eg, 5 × 5 cm) on the dorsolateral part of the skin. In a routine study, typically 6 test fields can be accommodated in each animal without too high risk of cross contamination. The test formulations are applied once or several times daily in a standardized amount to each field, and the skin reaction is then evaluated clinically for erythema, edema, and other reactions using the standardized scale of Draize, and at the end of the study by microscopy of the treated skin. The advantages of using the minipig in comparison to the “traditional” species, the rabbit, are that several test compounds can be evaluated and compared in the same individual, thus reducing the interindividual variation, and that the similarity in skin structure between human and porcine skin makes the minipig a better predictor of skin irritancy. It is well known that due to the much thinner and more delicate skin, the rabbit has a tendency to overpredict the irritancy of chemicals applied, compared to human skin. 115
For systemic toxicity testing of topical drug candidates, minipigs are routinely treated with the test compound formulated for topical application (ideally the final drug product, ie, the same formulation as intended for clinical trial and marketing) on a clearly marked and clipped skin area in the dorsolateral region of approximately 10% of the total body surface. High enough dose levels for evaluation of local as well as systemic toxic reactions are achieved by a combination of maximizing the test compound concentration, the treated skin area, and the applied amount of test article. The amount of test material that can practically be applied to the treatment site depends on the type of topical vehicle used (cream, ointment, gel, lotion, foam, spray, etc). Practical experience has shown that for “sticky” formulations like an ointment or cream, up to 10 to 20 mg/cm2 of test material can physically be applied to the skin (corresponding to 5-10 g test material applied to a 500-cm2 test site in a 10-kg minipig with an approximate body surface area of 5000 cm2 [0.5 m2]). However, this is much more than the human patients would normally apply to the diseased skin, and it should be noted that for some types of vehicle, much of the applied test material would be absorbed by the bandage material or washed off at the end of the exposure period and would not in reality be available for penetration into the skin. For other more “runny” types of topical vehicles (eg, a lotion), such large amounts cannot sensibly be applied. In the evaluation of the actual dermal and systemic exposure to the test compound in a dermal toxicity study, it is important to take account of the physical and thermodynamic properties of the topical vehicle as well as of the pharmacokinetic measurements. If this is not done properly, the risk and safety assessment of the topical product may very well be skewed. In addition to this, it should also be considered that in human patients topical drugs are intended to be applied to diseased skin, which often has a compromised epidermal barrier (often involving inflammatory and hyper- or hypoproliferative processes). From a biological, practical, and animal ethical point of view, it is not easy to mimic such diseased skin conditions in a normal, healthy minipig in a toxicology study. However, it should be considered to include investigation of the effect of application of the test material to damaged skin (eg, tape stripped skin, skin with induced partial or full thickness wounds, or otherwise damaged skin). Relevant end points in studies with induced damaged skin barrier are transdermal absorption, local irritancy, and systemic toxicity in general of the topical drug candidate, and these investigations could either be part of the general dermal toxicity study, or be investigated in a specifically designed stand-alone study. It is important to prevent oral ingestion and cross-contamination with the test compound between groups by application of suitable non-occlusive or occlusive protective dressing to the treated skin area, and by appropriate seclusion/separation of the animals during the treatment period. It should be kept in mind that occlusion may affect dermal penetration and hence systemic bioavailability of the test compound and the dosing method chosen should most closely mimic the clinical condition. Appropriate measures should be taken to avoid physical accumulation and microbiological contamination of the applied topical formulation on the skin, for example, by regular gentle washing of the treated skin area with lukewarm (∼30°C-35°C) water or a solution of mild soap. The usual standard parameters are measured in a dermal toxicity study in minipigs: clinical observation, including local skin reaction, body weight, feed intake, clinical pathology, electrocardiography, ophthalmoscopy, 116 organ weight, and macroscopic and microscopic pathology. 117
The main advantage of using the minipig for toxicity testing of dermal drug candidates is that the similar skin structure in humans and swine provides for a realistic modeling of the dermal and transdermal exposure to the test compound and hence also a much more realistic biological response than seen in rabbits or rodents which have skin that is significantly different than human skin.
Wound Healing
The anatomical similarity between human and porcine skin makes the minipig a very useful model for wound healing research, 118,119 whether the intention is to investigate the efficacy or the potential adverse effects of a pharmaceutical or medical device product. Different types of wounds can be studied in the porcine model, including full skin thickness wounds induced by surgical incision or split thickness wounds performed with an electrodermatome or carbon dioxide laser. In addition, models of impaired healing, including chemically induced necrotic wound models or wound infection models applying relevant microorganisms have been described. Evaluation of the wound healing process is done by clinical observation (reepithelialization, inflammation, hemorrhage, exudation, and wound tensile measurements) and histopathology. For all wound-healing studies, it is imperative to work under the highest standards of animal welfare to avoid unnecessary suffering in the experimental animal.
General Toxicity Studies
The use of minipigs for products other than dermal drugs has developed considerably over the years. There are generally 2 reasons for this development:
The technical procedures for handling, dosing, observation, clinical and anatomical pathology measurements, and development of relevant background data, and so on have developed tremendously over the years making both day-to-day work and scientific aspects easier for the nonclinical experimenter.
The metabolism of the minipig, including the cytochrome P450 system, which is of utmost importance for selection of the optimal species for toxicity testing of small molecule compounds, has now been described in detail. Specific aspects of the porcine metabolism compared to humans are known and in vitro tissue needed for early screening and species selection is commercially available. For an overview of ADME studies in minipigs, the reader is referred to Preusse and Skaanild. 97
Routes of Administration
Administration of test compounds to the minipig by other routes than dermal is perfectly feasible, adding to the usefulness of this animal model in toxicology.
Oral administration can be achieved via a gavage tube inserted into the stomach. Alternatively, the minipigs can be trained to ingest the test compounds via the mouth in gelatin capsules or formulated in tablets, hidden in a small amount of wet food. However, it should be noted that even after training of the minipigs, on a few occasions the minipigs may chew on the capsule/tablet, thereby potentially affecting the pharmacokinetics of the drug or substance. This method is therefore not suitable for test compounds that are locally irritant or have an unpleasant taste, since accidental aspiration into the respiratory tract may have serious consequences, including aspiration pneumonia.
Subcutaneous or intramuscular injections are usually given in the neck region. Because the minipig’s skin is closely attached to the underlying structures, like man, much smaller relative volumes can be achieved than compared to, for example, rabbit, rat, or dog.
For intravenous injections, small volumes can be injected into the veins of the ear. However, due to the small size and fragility of the ear veins in the minipig, it is usually preferable to perform repeated and large volume intravenous injections via a surgically inserted vascular access port inserted into one of the large veins of the body and with an injection chamber placed subcutaneously.
It is also possible to treat the swine by the intranasal, 120 –123 rectal, 123,124 or intravaginal 125 –129 routes. For some of these routes, training of the minipigs to accept the treatment, for example, by positive reinforcement training, is worthwhile to increase the cooperativity of the animal and to reduce the stress induced by the dosing procedure. 130
Repeat-dose toxicology studies in minipigs are conducted in the same way and using the same end points as for toxicology studies in dogs or primates, as also briefly described above in the section on dermal toxicology studies. A more detailed account of dosing methods employed in minipigs can be found in the study by Clausing. 131
Safety Pharmacology
The minipig has been shown to be a relevant and useful species for cardiovascular safety pharmacology studies using telemetry technique for the measurement of electrocardiogram, heart rate, and blood pressure. 132,133 Cardiac repolarization is described in detail in minipigs, 134 as is the use of minipigs in ex vivo proarrhythmia testing. 135 Characterization of the minipig in respiratory safety pharmacology was recently published. 136,137 An overview of the use of domestic pigs and minipigs in safety pharmacology can be found in Milano, 138 which covers cardiovascular, central nervous system, and respiratory system (core battery) as well as GI and renal system. The traditional species for a telemetry study has been the dog or the NHP. However, if the minipig for one reason or the other has been selected as the most suitable nonrodent species for the general, repeat-dose toxicology studies, it is also prudent to use the minipig for the telemetry study.
The porcine cardiovascular system has many similarities with the human cardiovascular system 139,140 like in humans there is a limited collateral blood supply to the heart and the major myocardial ion channels determining the polarization/repolarization of the cardiomyocytes are present in the minipig. 141 Additionally, in relation to cholesterol metabolism and cardiovascular pathology, like in humans and in contrast to many other laboratory species, a significant amount of the cholesterol is carried in the LDL fraction, making the domestic pig or minipig a suitable model for atherosclerotic changes. 142
The relevance of the minipig for cardiovascular safety pharmacology testing has been confirmed and validated in a series of studies using marketed pharmaceutical drugs with known effects on the myocardial function (including QT prolongation), on the heart rate, and on the blood pressure (propranolol, isoproterenol, nifedipine, dofetilide, terfenadine, and others 143 ).
The surgical procedure required to insert the telemetry equipment (electrodes, transponder, and radio transmitter) is basically similar to the procedures used in dogs or NHPs. However, the rather fast growth rate of the young and adolescent minipig, compared to dogs or NHPs can be a challenge for the long-term placement and function of the electrodes, and special care has to be given to ensure the correct placement of electrodes in the minipig. The route of administration should in principle be similar to the intended clinical route. However, it is important to conduct the treatment in a way that does not stress the animals unnecessarily and interfere with the measurement of the cardiovascular reaction. For example, extensive bandaging of the treated skin area after dermal treatment to prevent oral ingestion can impact signal transmission and hence study integrity. It should also be remembered that feeding of minipigs significantly impacts cardiovascular parameters, postprandial increased heart rate, and feeding of the animals should be timed strategically in relation to the data collection. 144
Reproductive Toxicology
The pigs and minipig have been shown to be very useful for reproductive toxicology studies, as an alternative to the most commonly used nonrodent species, the rabbit. The domestic pigs and minipig’s reproductive biology is well characterized, 145 –147 and it has shown to be susceptible to a number of teratogens including pyrimethamine, 148 coumarin and troxerutin, 149 ethinyl-nitrosouera, 150 ethanol, 151 vitamin A, 152 –154 vitamin C, 155 thalidomide, 156 tretinoin, 157 tobacco, 158 an anti-gestagen (metallibure), 159 –163 and fungal food contaminants. 158 An overview of the topic can be found in the studies by Barrow et al 164 or Ganderup. 165 The minipig could be relevant if metabolism of the test compound in the rabbit has been shown to differ too much from humans, or for compounds that are not well tolerated by the rabbit (eg, some antibiotics given orally). The ease of housing and handling, the relatively large litter size, and the short time to sexual maturity and reproductive cycle time makes the minipig an attractive nonrodent model in reproductive toxicity testing.
Minipigs are sexually mature already at 4 to 5 months, the female estrus cycle is 21 days with an estrus period of 47 to 56 days, the pregnancy period is 114 days, with gestation days 11 to 35 being considered the period of organogenesis. 166 The litter size is 5 to 9 in multiparous females (4-6 in primiparous females). One important factor to keep in mind is that the placentation in swine is of the diffuse, epitheliochorial type with 6 epithelial layers between the maternal and fetal blood circulation being maintained throughout the pregnancy. Unlike in many other species (including humans) maternal antibodies are not transferred to the porcine fetus during pregnancy but only via the colostrum during the first few days of life. This should be kept in mind when considering using the minipig for fetal developmental studies with large molecules. However, it has been shown that fetal exposure occurs after oral treatment of pregnant minipig sows of some antiarrhythmic drugs.
In preparation for a fetal developmental study, female estrus is synchronized with altrenogest, a synthetic progestin, before being mated. The fetal developmental study design is very similar to a rabbit developmental toxicity protocol: group size >18 females, treatment of the pregnant sows via the clinically most relevant route during the period of organogenesis (gestation day 11-35), the fetuses are removed by caesarean section on gestation day 110 and examined for external and visceral changes via an ordinary necropsy. 164,166 Skeletal changes can be examined after preparation and alizarin staining of the fetuses; changes in the head are examined after cross-sectioning of the heads after staining with Bouin fluid.
Examples of more commonly occurring variations and malformations found in Göttingen minipig fetuses are cryptorchidism, “truncus bicaroticus,” open eyes, polydactyli, and cardiac ventricular septum defect. 166,167 Needless to say that is extremely important to have a detailed background database of “spontaneous” variations and malformations occurring in the specific minipig strain, including changes under the conditions of the laboratory conducting the study, in order to be able to interpret the findings in a specific study. 167 Due to the fact that the Göttingen minipig has actually been used for developmental toxicity studies for 10 to 15 years now, a substantial database of developmental variations and abnormalities does now exist, and the Göttingen minipig 166,167 can therefore be considered a fully valid and useful nonrodent animal model for developmental toxicity studies.
Juvenile Toxicology
The minipig is an attractive species for juvenile toxicity testing, in cases where a nonrodent species is preferable. Reasons to use a nonrodent species could be similar to those that are mentioned for other toxicology studies: presence of pharmacological effect of the test compound, more “human-like” metabolism of the test compound, or more “human-like” development of organ systems in relation to the time of birth, for example, central nervous system, GI tract, cardiovascular system, renal system, skin, and so on (see Beck et al 168 for comparative age categories based on CNS and reproductive development). Cross-fostering of the offspring is possible and allows proper genetic distribution of the individuals in the study between treatments, in juvenile studies with dosing started at a very early age. 164
Important milestones in the postnatal development of the minipig are weaning at 4 weeks of age and sexual maturity at 3 to 5 months, roughly corresponding to the ages of 1 to 2 years and 16 years in a human child, respectively. In consequence, a juvenile toxicology study in the minipig would be conducted within this time span, potentially continuing into adulthood, depending on the purpose of the study. 164
Regarding dosing methods, the same methods that are used in adult minipigs are generally possible in juvenile piglets, 131 although it should be kept in mind that during the lactation period (first 4 weeks of life) the piglets are very dependent on the sow, and especially so during the first 24 to 28 hours where colostrum is passed from sow to offspring, thus potentially stressful procedures, for example, continuous bandaging in a dermal toxicology study should be avoided or limited. For that same reason, procedures should be avoided the for the first 24 to 48 hours postpartum unless absolutely necessary. The observation procedures employed in a juvenile toxicity study in the minipig would generally be the same as in a repeat-dose toxicity study in adult minipigs, perhaps supplemented by measurements of growth (crown-rump length, bone length, and weight). Behavioral assessment 169 is often conducted in the developing animal (and in safety pharmacology studies) and such investigation are well described in domestic pigs and minipigs, for further information on this topic the reader is referred to an extensive review by Lind et al. 170
Use of Minipigs in Safety Testing
A review of the actual use of minipigs for safety testing over a 9-year period in an experienced laboratory illustrates the variety in the practical use of minipigs (Figure 6). It is evident that both in terms of type and length of the minipig safety studies and in terms of the dosing routes and other technical procedures applied in these studies, the minipig has now become a mature, established, and well-accepted animal model that is very useful for safety evaluation of a variety of pharmaceutical drugs, food ingredients, and other chemical products, irrespective of the intended use and route of exposure.
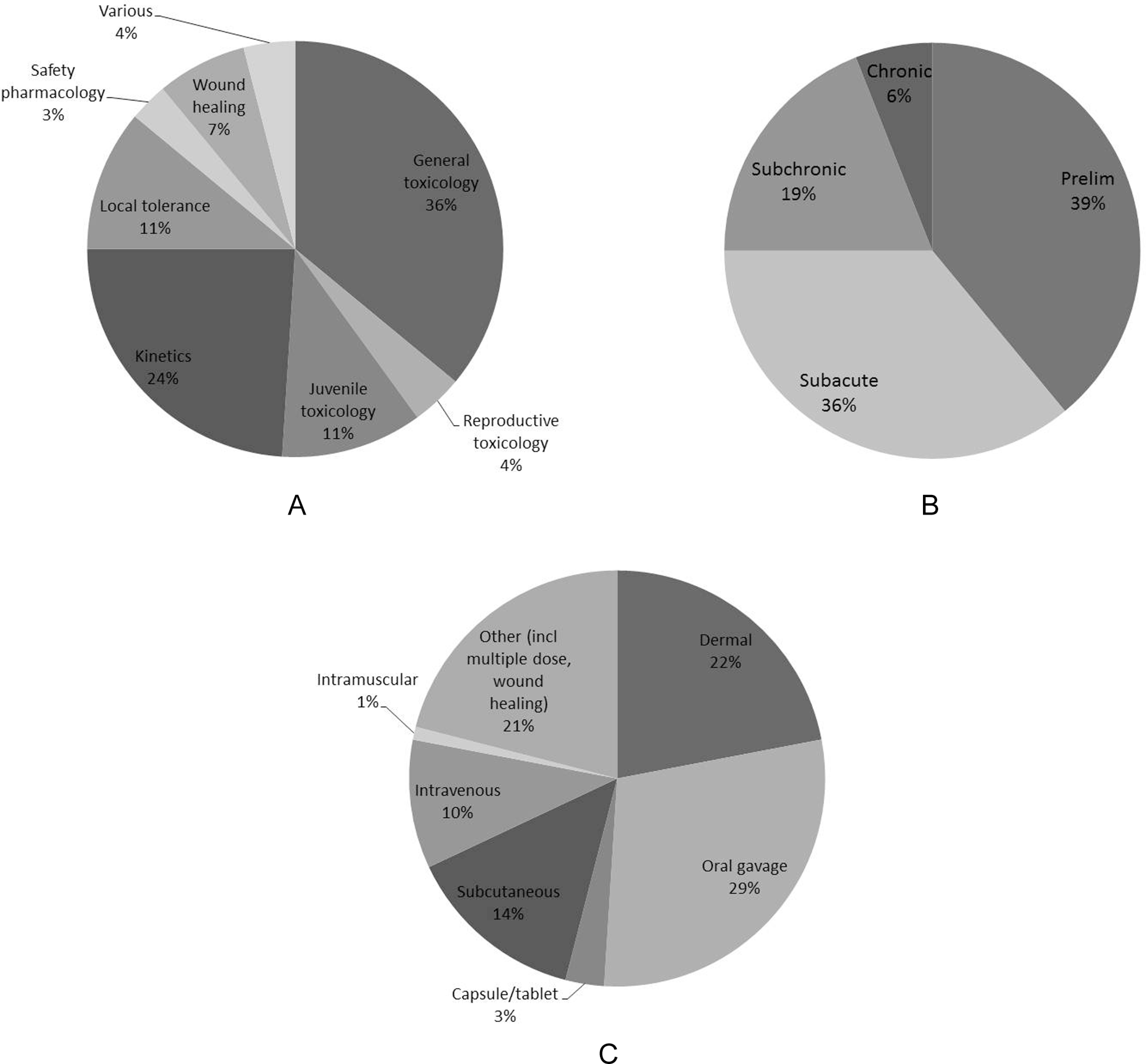
Variety in the use of minipigs in safety testing of pharmaceuticals in terms of study type (A), study length (B), and route of administration (C). Data courtesy of CIToxLAB, Scantox, Denmark, 2012.
In summary it can be said that the minipig is no longer a new nonrodent species in nonclinical safety. All doing routes commonly employed in toxicology are also usable in minipigs and in some areas covered here the minipig certainly appears to be the model of choice (Figure 6).
Regulatory Perspective: Advancing the Use of Minipigs in Safety Assessment (Wafa Harrouk)
According to the International Conference on Harmonization (ICH) guideline M3 (ICH M3 [R2]), repeat dose toxicity studies with pharmaceuticals are conducted for safety testing in 2 species (1 rodent and 1 nonrodent) to support investigative studies in humans and to support marketing of a drug product. This requirement exists to allow the best possible prediction of potential adverse effects in humans. The M3(R2) guidance does not specify which nonrodent species should be used for the evaluation of human drug products in order to provide flexibility in the choice of animal models. The choice of the nonrodent species depends on several factors, such as the ability of the chosen animal model to tolerate the drug being administered, comparative metabolism profiles between animals and humans as well as among animal species in order to extrapolate the results among species, and the pharmacokinetics and pharmacodynamics profile (eg, systemic exposure and ADME end points). The nonrodent animal models that have been used to date include the dog, rabbit, monkey, and the minipig. In the case of the monkey, some countries have imposed additional restrictions because of the interest in reducing the use of NHPs in toxicity studies.
The dog has been used traditionally as the nonrodent animal model in repeat-dose toxicity studies that are submitted to the FDA, but this model presents several potential hurdles including species-specific toxicities due to differences in metabolic profile, the tendency of dogs to vomit easily and the large amount of drug needed for the testing protocols. Other potential hurdles include differences between the dogs and humans in the skin and the GI tract properties.
The rabbit is another nonrodent animal model which is also used in repeat dose toxicity studies but is mostly seen as the model of choice for special toxicity studies (ie, irritation) and as the second species used in embryo–fetal developmental studies. Similar to the dog, the metabolic profile, the dermal and the GI tract of the rabbit could differ significantly from their human counterparts.
The use of the minipig has recently increased in nonrodent toxicity studies in part because of the similarities in many organ systems between the humans and minipigs. Several routes of administration (eg, oral and dermal) have been used in minipig toxicity studies. For pharmaceutical testing, the minipig has emerged as a useful model for in vivo and in vitro testing for dermal penetration and absorption studies due to the large resemblance of skin properties of the minipig to that of humans (see Table 3 and the dermal toxicology section above). These similarities also support the use of the minipig for the purpose of assessing dermal and systemic toxicity. Anatomic and physiologic similarities include light pigmentation, sparse hair coverage, the presence of large amounts of elastic tissue, Langerhans cells, phase I + II activities, and cellular turnover. Biochemically, the minipig shares with humans the presence of cytoskeletal proteins (integrins). Other characteristics of minipig skin are helpful in nonclinical testing such as the ability to test over a large dermal area, thus permitting the use of multiple testing sites, the ability to conduct long-term testing, and the ability to test various types of patches or occlusion.
New drug applications submitted to gain marketing approval for topically applied products can combine results from oral and dermal toxicity studies to fully characterize the toxicity profile of the topical product in question. For topical new molecular entities, a chronic dermal toxicity study is expected to be completed. In cases where systemic absorption is low, some studies of systemic effects might be abbreviated or waived. If the drug is not a new molecular entity by the topical route, a shorter toxicity study by the dermal administered route might be acceptable to bridge to existing information. The dermal toxicity study can address issues such as the total topical dose used, maximum area of exposure, percentage of drug absorbed through the skin, potency of drug and its metabolites, severity of potential adverse effects, and the integrity of the treated skin area. Although the minipig has become a standard animal model in dermal toxicity studies submitted to the FDA, the use of the mini-pig is not limited to dermal toxicity studies. Some of the challenges of using minipigs in toxicity testing include the size of the animals, which could have implications on the amount and cost of the drug used, and their longer life span which eliminates the possibility of using them in life-time bioassays for carcinogenicity studies (the minipig lifespan can last up to ∼15-20 years). With the growing use of the minipig as a nonrodent animal model, researchers and regulators are gaining a better understanding of this model and of potential species-specific adverse effects that may arise, if any. With increased experience, more robust historical databases are being established for the minipig thus making this animal model more accessible and accepted by sponsors and regulators in the drug development field.
Footnotes
Author’s Note
The opinions and information in this presentation are those of the author and do not necessarily reflect the views and policies of the U.S. Food and Drug Administration.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
