Abstract
Dogs have been often chosen as a nonrodent species for preclinical development of small molecule drugs mainly due to availability and relative ease of handling. Recently, focus has increased on the minipig as a potential alternative to the dog, based on either scientific rationale or public opinion concerns. There are, however, other factors influencing nonrodent choices, in particular drug amount and synthesis time, which differ between species and therefore may impact the milestones of a drug development program. To assess the magnitude of compound need, a retrospective internal survey was conducted on drug amounts used in dog studies which were translated into the requirements for minipigs. Compound need approximately doubles if minipigs are used. Costs of compound are accordingly higher, and synthesis times are slightly increased. In our company, the differences were not considered significant enough to preclude the use of minipigs if the later preclinical program might benefit from improved human risk prediction.
Keywords
Introduction
Pigs have a long tradition in biomedical research due to their anatomical and functional similarities to humans, especially in the field of cardiovascular and dermatology (Montagna et al. 1964; Hughes 1986; Swindle and Smith 1998; Mortensen, Brinck, and Lichtenberg 1998; Authier et al. 2011; Summerfield, Meurens, and Ricklin 2015). For this reason, minipigs are being increasingly discussed as a potential alternative to dog as nonrodent species in drug safety assessment (Svendsen 2006; Bode et al. 2010; Ganderup et al. 2012; Swindle et al. 2012). Species selection is a critical step in drug development as it should translate efficacy and toxicity data from animal to human, thereby narrowing or preventing unexpected side effects in patients (Morton 1998). The species should be chosen based on a pharmacokinetic and metabolism profile similar to those in humans (European Medicines Agency 2010), which makes the minipig a viable nonrodent species. However, relatively few companies are using minipigs for general toxicity testing (Bode et al. 2010). In contrast, dogs are often selected as the nonrodent species due to extensive experience and background data and their cooperative interaction with humans during handling and treatment (Hasiwa et al. 2011). Factors related to species selection such as increased compound requirements, costs, and potential delays are impacting many aspects of investigational new drug (IND) program milestones. Based on a higher body weight, the increased drug amount is frequently challenged and perceived as one of the major drawbacks when discussing the minipig as a toxicity species (Bode et al. 2010). At our company, the Göttingen minipig was chosen as an alternative nonrodent species to complement the current preclinical safety portfolio. We were interested in the magnitude and impact of increased compound need when using these animals. In an internal retrospective survey, pharmacokinetic and toxicity studies for small molecule drugs in dogs were assessed for estimation of animal numbers and compound need. Based on the dog studies, drug amounts and costs were calculated for using minipigs in a preclinical safety program, supporting a compound up to the study for the IND application and beyond.
Methods
Compound Need Using Dogs
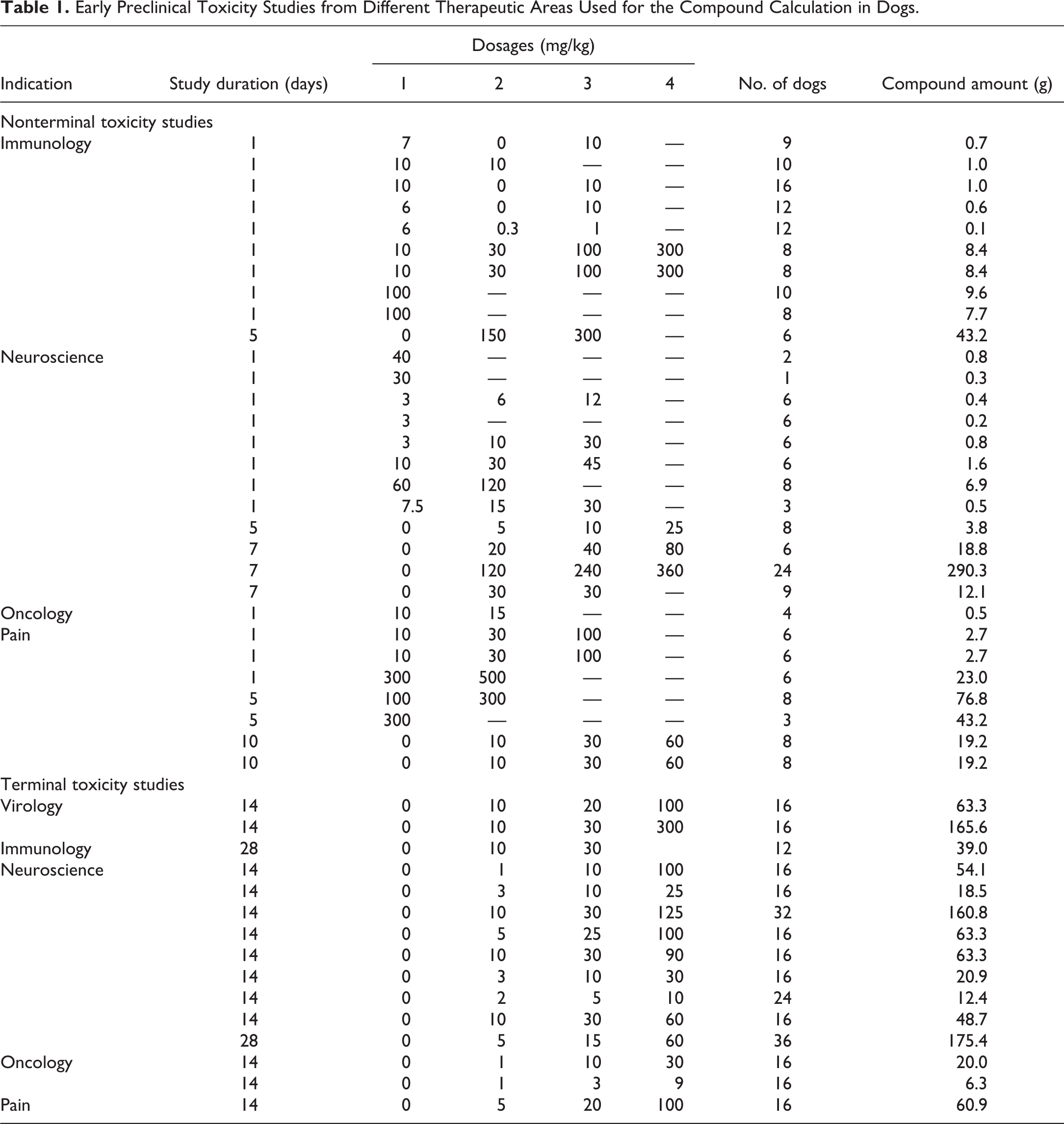
Dog studies preceding the milestone decision for the IND-enabling good laboratory practice (GLP) study were reviewed in 5 therapeutic areas over a period of 5 years. The compound amounts were calculated for terminal toxicity studies with histopathological end points and for nonterminal toxicity and pharmacokinetic studies conducted with repeatedly used pool animals. For terminal studies, beagle dogs were used at the age of 9 to 11 months. The corresponding mean body weights for compound calculation were taken from the provider Marshall BioResources (2015), that is, 9.6 kg for males (n = 1,563, SD = 0.3 kg) and 7.8 kg for females (n = 1,477, SD = 0.1 kg) with an overall mean of 8.7 kg (SD = 0.97 kg). For nonterminal studies with older dogs, the body weights were determined from internal sources (Figure 1A) due to the lack of data from the provider at ages older than 12 months. The data were derived from 50 pool dogs aged from 7.7 to 71.7 months, including 26 males (525 body weight recordings; mean weight = 10.4 kg, and SD = 1.3 kg) and 24 females (502 body weight recordings; mean weight = 8.7 kg, and SD = 1.0 kg) with an overall mean body weight of 9.6 kg (SD = 1.4 kg). The toxicity studies and corresponding dose levels underlying the compound calculations are depicted in Table 1. For pharmacokinetic studies conducted with pool dogs, compound amounts were calculated from 37 studies, involving an internal standard design with 6 dogs and dosages of 0.5 to 1.5 mg/kg. The mean animal number of 36 dogs in the IND-enabling GLP study was derived from standard study designs with 3 dose groups using 3 to 4 dogs/sex/group plus 2 recovery dogs/sex in 1 to 4 groups. The corresponding dose levels were deduced from the evaluated dose range finding studies.
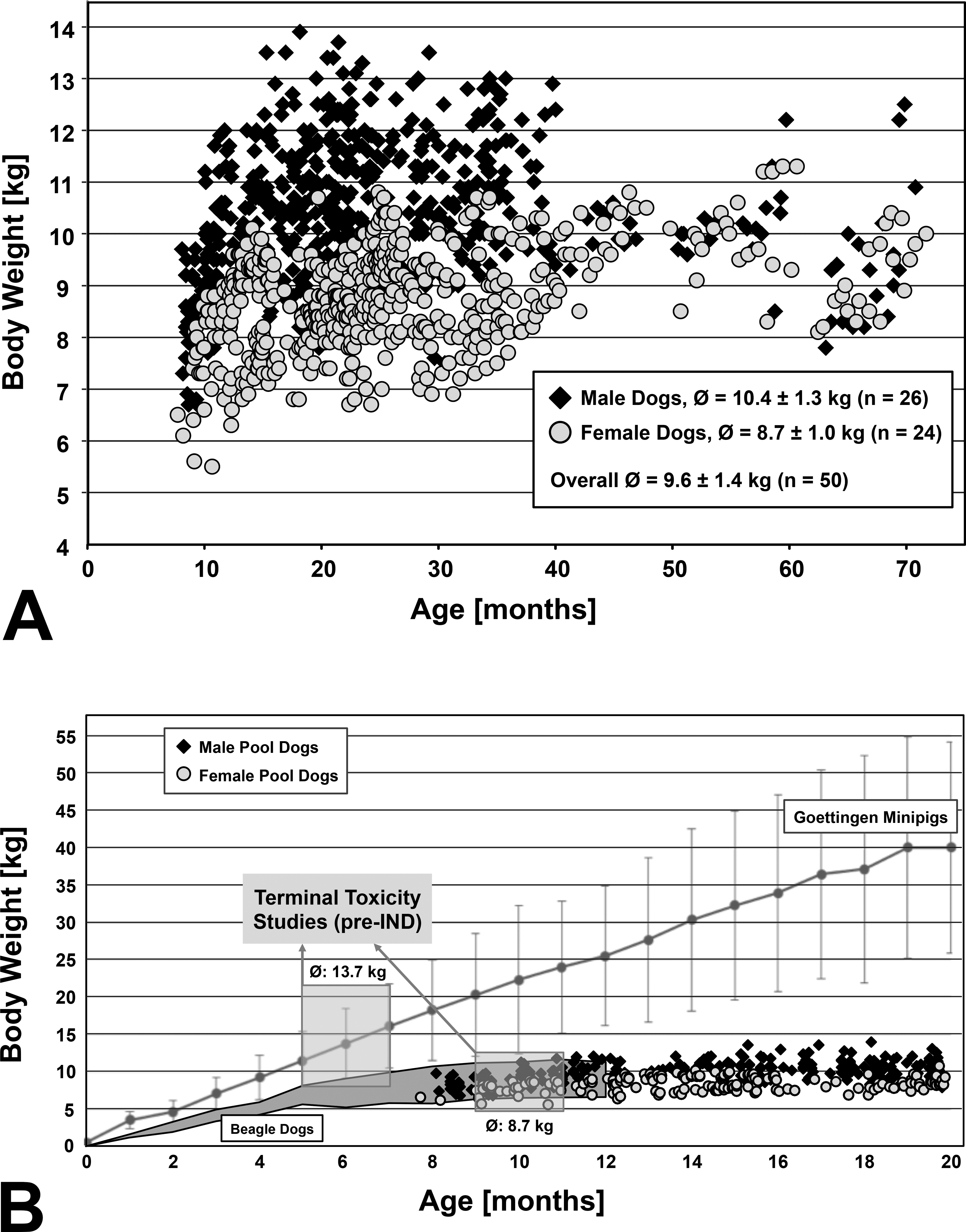
(A) Company-derived growth data for pool dogs. Body weights from 50 beagle dogs (26 males and 24 females) kept for repeated study use were measured every 2 to 4 weeks within an age range from 7.7 to 71.7 months. The mean weights were 10.4 kg for males (SD = 1.3 kg) and 8.7 kg for females (SD = 1.0 kg) with an overall mean of 9.6 kg (SD = 1.4 kg). (B) Growth data for beagle dogs and Göttingen minipigs. The mean (Ø) body weight at relevant age for early terminal toxicity studies preceding the investigational new drug application (pre-IND) were calculated for beagle dogs (8.7 kg, data from Marshall BioResources, U.S. colony; gray area spanning the 95% reference ranges of males and females) and for Göttingen minipigs (13.7 kg, data from Ellegaard Minipigs A/S; mean for males and females with 95% reference range). The body weight development for pool dogs at our company is indicated as squares (26 males) and circles (24 females).
Early Preclinical Toxicity Studies from Different Therapeutic Areas Used for the Compound Calculation in Dogs.
Compound Need Using Minipigs
For minipigs, a comparable set of internal growth data was not available and the body weights were derived from the provider Ellegaard (Ellegaard Göttingen Minipigs A/S 2015; Figure 1B). Based on these body weights, the drug amounts were calculated using the corresponding dog study designs and dose levels. For compound calculations in terminal studies, mean body weights were taken for 5- to 7-month-old minipigs, that is, 13.8 kg for males (n = 6,707, SD = 2.5 kg) and 13.7 kg for females (n = 7,631, SD = 2.4 kg) with an overall mean of 13.7 kg (SD = 2.4 kg). For nonterminal studies, mean body weights were calculated for a hypothetical pool of 5- to 20-month-old minipigs, that is, 22.6 kg for males and females (n = 6,707 and 7,631, SD = 4.0 kg and 4.5 kg, respectively) with an overall mean of 22.6 kg (SD = 4.3 kg). For pharmacokinetic studies, the drug amounts were calculated considering the use of the same pool of minipigs as for the nonterminal toxicity studies (5 to 20 months of age). The compound need for the 9-month GLP study takes into consideration the body weight development up to the age of 16 months at the end of the study.
Compound costs were calculated using a standard valuation of approximately US$50/g, which was determined by taking an average cost of materials, time, and labor across a broad number of programs at a variety of scales.
Results
During early drug development, a series of animal study types was conducted to assess the metabolic and toxicological profile of new compounds. In nonterminal pharmacokinetic, single-dose or short-term repeated dose toxicity studies, the test species may be reused. In contrast, terminal toxicity studies to define the dose for the IND-enabling toxicity study have a histopathological end point. When estimating substance need, terminal and nonterminal studies involve a different subset of animals with regard to age and body weight, and these are the most significant factors that influence compound amounts. Generally, it is the aim to keep the body weight as low as possible by concomitantly assuring advanced animal development especially with respect to sexual maturity. Minipigs are regarded as sexually mature from the age of 2 months in males (Navratil et al. 2015) to more than 6 months in females (Tortereau, Howroyd, and Lorentsen 2013; de Rijk et al. 2014), whereas dogs reach this stage at 8 to 14 months of age (Allen 1992; Chandra and Adler 2008; Goedken, Kerlin, and Morton 2008). Minipigs can therefore be used at a younger age in routine toxicity studies. In terminal pre-IND toxicity studies with a typical duration of up to 4 weeks, 5- to 7-month-old minipigs are generally used, corresponding to a mean body weight of 13.7 kg (Figure 1B). In contrast, dogs are at a comparable developmental stage at the age of 9 to 11 months, corresponding to a mean body weight of 8.7 kg. For nonterminal studies, a pool of animals exists for repeated use. In our assessment, each individual pool dog was on average in 12 studies and the replacement rate was approximately 1.5 to 2 years. For calculating compound need using the minipig, the published body weight data of up to 20 months were used, covering the identified turnover of pool animals. This results in a mean weight range of approximately 13 kg (95% reference range: 7.6 to 15.6 kg) at 5 months of age to 40 kg at 20 months (95% reference range: 23.1 to 54.0 kg) with an overall mean of 22.6 kg (SD = 4.3 kg). In contrast, a body weight limit is normally reached after 12 to 13 months in dogs (Parcher et al. 1970; Reinwald and Burr 2008). This was confirmed by internal dog data (Figure 1) where a mean body weight of 9.6 kg for pool dogs was determined. In the same assessment, the mean body weight of dogs at the age for terminal toxicity studies (9 to 11 months) was 8.5 kg (SD = 1.2 kg) which is in agreement with the published Marshall data (8.7 kg, SD = 0.97 kg).
The estimation of compound was done on selected therapeutic areas for pre-IND studies under non-GLP conditions (Table 2). Terminal toxicity studies involved 280 dogs over 5 years, corresponding to a compound amount of 0.97 kg. Assuming the use of minipigs with a 1.6-fold higher mean body weight (13.7 kg for minipig vs. 8.7 kg for dog), 1.56 kg of compound is needed. In nonterminal studies conducted with pool animals, a 2.4-fold higher body weight has to be considered for the minipig (22.6 kg for minipig vs. 9.6 kg for dog), resulting in compound amounts of 1.45 kg instead of 0.6 kg in nonterminal toxicity studies and in 5.0 g compared to 2.1 g in pharmacokinetic studies in our survey.
Comparison of Compound Need Using Dogs or Minipigs in Early Preclinical Studies.
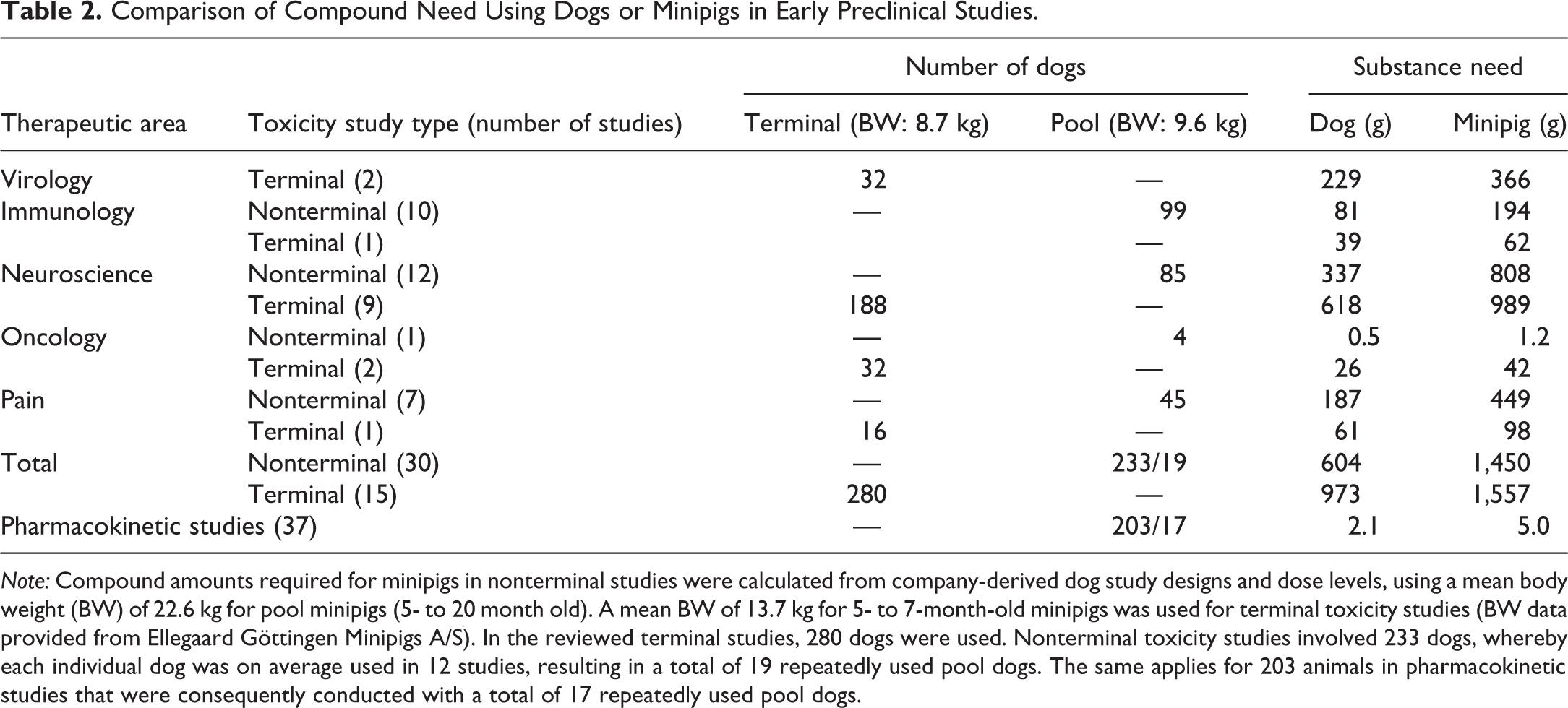
Note: Compound amounts required for minipigs in nonterminal studies were calculated from company-derived dog study designs and dose levels, using a mean body weight (BW) of 22.6 kg for pool minipigs (5- to 20 month old). A mean BW of 13.7 kg for 5- to 7-month-old minipigs was used for terminal toxicity studies (BW data provided from Ellegaard Göttingen Minipigs A/S). In the reviewed terminal studies, 280 dogs were used. Nonterminal toxicity studies involved 233 dogs, whereby each individual dog was on average used in 12 studies, resulting in a total of 19 repeatedly used pool dogs. The same applies for 203 animals in pharmacokinetic studies that were consequently conducted with a total of 17 repeatedly used pool dogs.
For estimating compound costs, no universal formula can be applied, as the expenditures depend on the chemical structure, complexity of synthesis, and yield. Furthermore, the substance need differs for the respective study types during drug development, and costs and speed also depend on the corresponding advancement of the manufacturing step. Generally, the average compound costs are estimated to increase proportionally with the additional material whereby the synthesis time is not significantly affected.
The costs for a preclinical package were calculated from 18 compounds in discovery of which 3 progress to the milestone decision for the IND-enabling GLP study (Table 3A). The program was translated into animal numbers and compound expenses (Table 3B) based on the 5-year assessment (Table 2). Using minipigs instead of dogs, the overall compound need would amount to 488 g instead of 204 g in terminal studies and 834 g instead of 521 g in nonterminal studies. To further develop this calculation for later drug development stages, one 13-week and one 9-month GLP study were used (Table 3C). In the 13-week study, 1.9 kg of compound would have been needed using the minipig as opposed to 1.2 kg in the dog. In the 9-month GLP study, approximately double the amount of compound would have been needed using the minipig (7.7 kg compared to 3.7 kg).
Comparison of Compound Costs Using Dogs or Minipigs in a Preclinical Program.
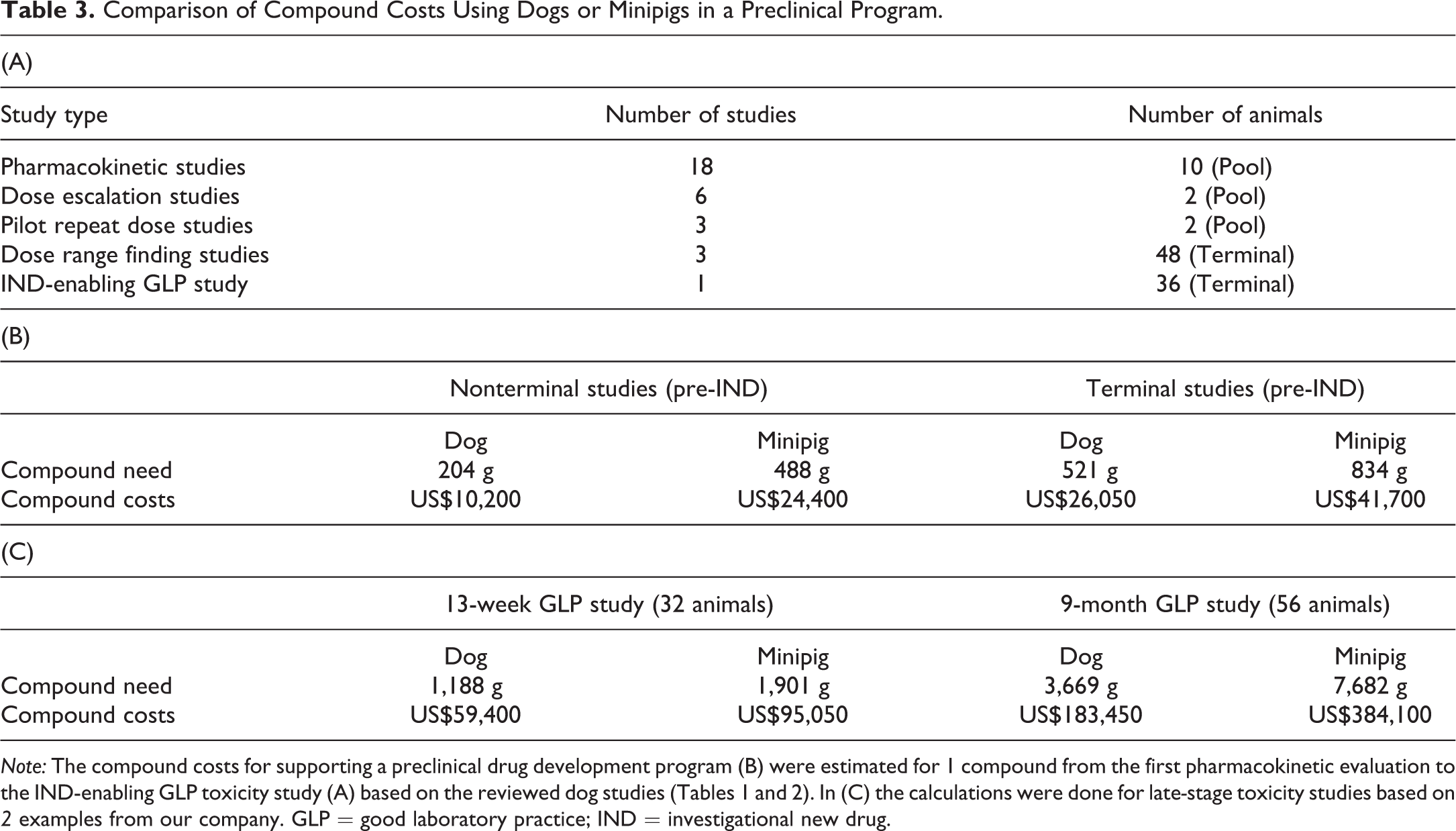
Note: The compound costs for supporting a preclinical drug development program (B) were estimated for 1 compound from the first pharmacokinetic evaluation to the IND-enabling GLP toxicity study (A) based on the reviewed dog studies (Tables 1 and 2). In (C) the calculations were done for late-stage toxicity studies based on 2 examples from our company. GLP = good laboratory practice; IND = investigational new drug.
Discussion
For preclinical safety testing of small molecules, dogs are often chosen as the first nonrodent species based on experience and extensive control data. The minipig may however be more suitable from a scientific rationale, regarding anatomy, physiology, or metabolism (Swindle and Smith 1998; Anzenbacher et al. 1998; Soucek et al. 2001; Bode et al. 2010; Swindle et al. 2012; Dalgaard et al. 2014; Oesch et al. 2014; Gutierrez et al. 2015). Pharmaceutical research and development is a competitive environment with aggressive timelines and high expenditures. Toxicology and pharmacokinetic studies account for approximately one-third of the overall costs of a preclinical program (Stergiopoulos, Kim, and Getz 2013). Arguments against choosing the minipig as a general toxicity species are often based on economic considerations. The increased compound requirements due to the higher body weight of minipigs are regarded as a disadvantage and may favor the dog (Bode et al. 2010). The present study compared the use of minipigs instead of dogs in early toxicity and pharmacokinetic studies to assess the impact on compound amounts, costs, and synthesis times for our company.
The minipig would require approximately 1.6 more substance in early terminal toxicity studies compared to the dog. The situation is different in nonterminal toxicity and pharmacokinetic studies that are generally conducted with a pool of repeatedly used animals. Our study revealed a replacement rate of approximately 1.5 to 2 years for pool dogs. In dogs, there are no essential body weight changes expected after the age of 12 to 13 months when they have reached full skeletal maturity (Parcher et al. 1970; Reinwald and Burr 2008; internal data, Figure 1). In contrast, the mean body weight of minipigs increases to 40 kg in the same time period (Figure 1, 20 months of age) and mean weights of up to 53 kg have been reported for mature Göttingen minipigs (Koehn et al. 2007); consequently, an incremental substance need has to be considered when keeping them longer. Based on the pool animal turnover, 2.4-fold more compound would be needed for nonterminal studies using minipigs. Drug amount and synthesis time, however, are not proportional, and the additional substance required for use in minipigs will affect costs more than time.
In summary, additional efforts regarding drug amounts and costs have to be considered when using minipigs instead of dogs in preclinical studies. In the whole context of a drug development program, these efforts were considered not significant enough for our company to preclude the minipig as a nonrodent species.
Footnotes
Acknowledgments
We would like to thank Raimund Geil for supporting data collection, Steve Wittenberger for providing advice regarding chemistry-related costs and estimations, and Lynda Nolan for proofreading the manuscript.
Author Contribution
Authors contributed to conception or design (KS, SR, JB, and PG); data acquisition, analysis, or interpretation (KS, HH, and PG); drafting the manuscript (KS); and critically revising the manuscript (SR, HH, JB, and PG). All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Authors’ Note
All authors are employees of AbbVie. All animal studies conducted at AbbVie are according to the relevant German and European guidelines and are approved by the authorities in Rhineland-Palatinate.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This publication was sponsored by AbbVie. AbbVie contributed to the design, research, and interpretation of data, writing, reviewing, and approving the publication.
