Abstract
Tetrabromobisphenol A (TBBPA) is a brominated flame retardant that induces endometrial adenocarcinoma and other uterine tumors in Wistar Han rats; however, early molecular events or biomarkers of TBBPA exposure remain unknown. We investigated the effects of TBBPA on growth factor receptor activation [phospho-receptor tyrosine kinases (RTKs)] in uteri of rats following early-life exposures. Pregnant Wistar Han rats were exposed to TBBPA (0, 0.1, 25, and 250 mg/kg bw/day) via oral gavage on gestation day 6 through weaning of pups on postnatal day (PND) 21. Pups were exposed in utero, through lactation, and by daily gavage from PND 22 to PND 90. Uterine horns were collected (at PND 21, PND 33, and PND 90) and formalin-fixed or frozen for histologic, immunohistochemical, phospho-RTK arrays, or western blot analysis. At PND 21, the phospho-RTKs, fibroblast growth factor receptor 2 and 3 (FGFR2 and FGFR3), neurotrophic tyrosine kinase receptor type 3 (TRKC), and EPH receptor A1 (EPHA1) were significantly increased at different treatment concentrations. Several phospho-RTKs were also significantly overexpressed at PND 33 which included epithelial growth factor receptor (EGFR), FGFR2, FGFR3, FGFR4, insulin-like growth factor receptor 1 (IGF1R), insulin receptor (INSR), AXL receptor tyrosine kinase (AXL), MER proto-oncogene, tyrosine kinase (MERTK), platelet derived growth factor receptor alpha and beta (PDGFRA and PDGFRB), ret proto-oncogene (RET), tyrosine kinase with immunoglobulin-like and EGF-like domains 1 and 2 (TIE1 and TIE2), TRKA, kinase insert domain receptor (KDR;VEGFR2), fms related receptor tyrosine kinase (FLT4; VEGFR3), and EPHA1 at different treatment concentrations. EGFR, a RTK overexpressed in endometrial cancer in women, remained significantly increased for all treatment groups at PND 90. Erb-B2 receptor tyrosine kinase 2 (ERBB2) and IGF1R were overexpressed at PND 33 and remained increased through PND 90, although ERBB2 was statistically significant at PND 90. The phospho-RTKs, FGFR3, AXL, TYRO3 protien tyrosine kinase (TYRO3; DTK), HGFR, TRKC, FLT1/VEGFR1, and EPHB2 and 4 were also statistically significant at PND 90 at different treatment doses. The downstream effector, phospho-MAPK44/42, was also increased in uteri of treated rats. Our findings show RTKs are dysregulated following early-life TBBPA exposures and their sustained activation may contribute to TBBPA-induced uterine tumors observed in rats later in life.
Introduction
Tetrabromobisphenol A (TBBPA) is the most common brominated flame retardant used worldwide in consumer products, mainly within circuit boards. 1 Initially, TBBPA was thought to have low reproductive toxicity when no adverse effects were observed in rodent studies. 2 Although in earlier in vitro studies, TBBPA had been shown to act as an endocrine-disrupting chemical (EDC) having estrogenic activity in an estrogen response element (ERE) luciferase reporter assay in Michigan Cancer Foundation-7 (MCF-7) human breast cancer cells. 3 However, more recently, female Wistar Han rats in a National Toxicology Program (NTP) 2-year bioassay were reported to develop endometrial adenoma or adenocarcinoma, or malignant mixed Müllerian tumors after exposure to high doses (250, 500, or 1000 mg/kg) of TBBPA. 4 The early and low dose effects and the molecular mechanism(s) by which TBBPA elicits its carcinogenic activity in the uterus remain largely unknown. Dunnick et al. 4 have proposed that TBBPA may interfere with estrogen homeostasis through competitive binding of estrogen and TBBPA to sulfotransferases, which are important in estrogen metabolism and elimination. They suggested that decreased estrogen excretion may result in increased estrogen bioavailability for estrogen receptor binding in the uterus.
Receptor tyrosine kinases (RTKs) are growth factor receptors that upon interaction with their respective ligands are involved in multistep processes that send signals from the cell surface to the cytoplasm and nucleus. 5 RTKs regulate growth, differentiation, cell-cycle progression, and transcriptional activities of normal and cancerous cells. 5 Studies have shown that RTKs are overexpressed and/or activated in both benign and malignant tumors affecting both the myometrial and endometrial compartments of the uterus.6–8 It has been reported that hormone stimulation in the endometrium can lead to overexpression of RTK proteins, thereby contributing to tumor formation in female reproductive organs.9–11 In women, endometrial cancers are often classified into two major categories, type I and type II. Type I endometrial cancer is typically considered low-grade, occurring predominantly in perimenopausal and premenopausal women in response to unopposed estrogen stimulation and often preceded by endometrial hyperplasia.7,12 Type II endometrial cancers often occur in menopausal women, are estrogen independent, and are commonly diagnosed as high grade serous carcinoma, malignant mixed Mullerian tumor, or clear cell carcinoma. 12 Evidence exists that growth factor receptors [insulin-like growth factor receptor 1 (IGF1R), VEGFR, fibroblast growth factor receptor 2 (FGFR2), epithelial growth factor receptor (EGFR), Erb-B2 receptor tyrosine kinase 2 (ERBB2), and EPH receptor A2 (EPHA2)] can be overexpressed and are important regulators of endometrial cancer in women. 13 Epidermal growth factor receptor 2 (HER2: ERBB2), a known regulator of cell proliferation and anti-apoptotic pathways, was shown to be increased in TBBPA-induced uterine endometrial carcinomas taken from Wistar Han rats in a 2-year NTP bioassay. 14 Additionally, insulin-like growth factor 1 (Igf1) gene expression was found to be increased in uteri following short-term (5 days) TBBPA (250 mg/kg) exposures in adult rats, 15 and IGF1 is known to be involved in tumorigenesis through RTK signaling pathways. 16 In this study, we were interested in determining the effects of TBBPA on activation of growth factor RTKs following in utero and early-life exposures in Wistar Han rats.
Methods
Wistar Han rats
Pregnant Wistar Han [Crl: WI(Han)] rats were exposed to TBBPA (Sigma-Aldrich, cas#79-94-7, 97% purity, lot #MKBG4059) at 0, 0.1, 25, and 250 mg/kg/day, via oral gavage at gestation day 6 (GD6) through weaning of pups postnatal day (PND) 21. Pups were exposed to TBBPA in utero, through lactation period and then post-weaning (PND 22 to PND 90) by daily oral gavage. There were six rats per dose group for the RTK array studies. The six rats were from time pregnant Wistar Han [Crl: WI(Han)] rats obtained from Charles River, Raleigh, NC. Pregnant rats were received on gestation day 4 (GD4) and acclimated for 2 days prior to dosing with TBBPA at 5 mL/kg to administer 0, 0.1, 25, and 250 mg/kg bw/day. A random generator was used to randomly assign groups to a TBBPA group. The day after birth, GD21 was identified as PND 1. Standardization of litters was done to consist of 8 (4:4 sex ratio, if possible) on PND 4. On PND 21, pups were weaned and sorted into same sex and treatment groups of two or three littermates per cage. In this study doses 0, 0.1, 25, and 250 mg/kg/day were selected based on those used in the NTP 2-years bioassay (NTP 2014) (0, 250, 500, and 1000 mg/kg/day), whereby uterine endometrial adenocarcinomas, adenomas, and other uterine tumors were detected. Furthermore, TBBPA concentration of 0.1 mg/kg/day is relevant to human exposure levels (0.03–126 ng/g weight were found in fish, and 0.05–713 ng/g weight were found in infants). 17 Uterine horns were collected from pups at PND 21, 33, and 90, and frozen or formalin-fixed. All PND 33 and PND 90 samples were collected from rats, and histologically staged for estrous cycle phase. Only rats in the estrus phase were used for this study. All animals were cared for, handled, and used with approved protocols of the NIEHS Animal Care and Use Committee in accordance with the NRC Guide for the Care and Use of Laboratory Animals. 18 Animals were housed in controlled rooms with a 45–60% average humidity, 25°C temperature, and a standard 12-h light cycle. Food and reverse osmosis/deionized (RO/DI) water was provided ad libitum.
Tissue fixation and histology
The right horn of the uterus was removed including the vagina and cervix. A transverse section was taken through the midpoint of the right uterine horn, and longitudinal sections of the proximal and distal uterus including the cervix and vagina were collected and placed in 10% neutral buffered formalin for 24–48 h. Fixed tissues were embedded in paraffin, sectioned at 5 microns, and stained with hematoxylin and eosin (H&E). All PND 33 and PND 90 samples were collected from rats and histologically staged for estrous cycle phase. Only rats in the estrus phase (Supplementary Figure 1) as evidenced by light microscopic evaluation/staging of ovary, vagina, and uterus were used for this study. 19 No distinct histopathologic changes were observed in treated versus control groups at a light microscopic level (see Supplementary Figure 1). Procedures for digital slide scanning and capturing of images of H&E slides are described below in the immunohistochemistry section. Animal terminal body weights were not significantly different between all groups (Supplementary Table 1).
Protein extraction
Samples of 50–100 μg of frozen uterine tissue taken from the left horn of 4–6 individual rats per dose group were collected in lysis buffer with proteinase inhibitors (10 μg/mL aprotinin, 10 μg/mL leupeptin, and 2 μg/mL phenylmethylsulfonyl fluoride). The samples were then minced using a 30-s burst of a homogenizer, followed by centrifugation at 13,000 r/min for 30 min at 4°C. The supernatants were collected and stored at −80°C.
Phosphorylation of receptor tyrosine kinases array
Expression of RTKs was detected using the Proteome Profiler Mouse Phospho-RTK Array Kit (# ARY014, R&D Systems, Minneapolis, MN). Four hundred μg of pooled total protein from uterine tissue of 4–6 rats in each dose group were incubated with RTK array membranes spotted with 39 phospho-RTK antibodies. The procedures were performed according to the manufacturer’s protocol. A densitometer (Fluor ChemTM8900, α Innotech, San Leandro, CA) was used for quantitation of spot intensities. Data represent a composite of four replicates.
Western blotting (phospho-receptor tyrosine kinases)
The lysate of frozen rat uterine horn from 4–6 Wistar Han rats in each dose group as those used in the RTK array studies were collected in cold solubilization buffer as described above in the RTK array section. Western blot analysis was done as previously described. 20 Briefly, Western blots were incubated overnight with the following specific primary antibodies diluted as indicated: 1:1000 for total and phosphorylated mitogen-activated protein kinase (MAPK) p44/42 tyrosine 204 (Cell Signaling Technology, Cat# 9102 and 9101); 1:1000 for total and phosphorylated EGFR tyrosine 845 (Cell Signaling Technology, cat# 2232 and 2231); 1:1000 for total and phosphorylated ErbB2 (Her2) tyrosine 1221/1222 (Cell Signaling Technology, Cat# 4290 and 2243); 1:1000 for total IGF-1R and phosphorylated IGF-1R tyrosine 1131/1146 (Cell Signaling Technology, Cat# 3027 and 3021); and 1:500 for hypoxanthine-guanine phosphoribosyltransferase 1 (HPRT) (Santa Cruz Biotechnology, cat# sc-20975). A densitometer (Fluor ChemTM8900, Innotech, San Leandro, CA, USA) was used for quantitation of band intensities.
Immunohistochemistry of phospho-mitogen-activated protein kinase44/42
Immunohistochemical staining for phospho-MAPK was done using five-micron thick, formalin-fixed paraffin-embedded rat uterine sections placed on positive-charged slides and heated in a 60°C oven for 30 min prior to staining. Slides were de-paraffinized with xylene and hydrated through descending grades of alcohol to Tris/HCl buffer. Antigen retrieval was performed using a Decloaker (Biocare Medical, city, state) with citrate buffer pH 6.0 for 15 min at 110°C. Slides were cooled for 10 min and then placed in Tris/HCl wash buffer for 10 min. Sections were treated with 3% hydrogen peroxide for 15 min to quench endogenous peroxidase in tissues. Normal donkey serum (10%) was applied to the tissue sections for 20 min to block nonspecific binding sites followed by incubation with an avidin/biotin reagent to block endogenous biotin in the sections. Rabbit monoclonal anti-phospho-p44/42 MAPK (Erk 1/2) (Cell Signaling cat #4370S, lot #17, 0.5 mg/mL) antibody was applied to the sections for staining at a dilution of 1:150 for 1 h. Negative controls received normal rabbit IgG (Abcam cat #ab125938, lot #GR279962-1, 0.2 mg/mL) at the same protein concentration. Tissues were incubated with a donkey anti-rabbit (Jackson Labs, 1.1 mg/mL) biotinylated secondary antibody (Vector Labs) for 30 min followed by Vector RTU ABC for 30 min. After a brief tap water rinse, sections were treated with 3,3’ diaminobenzidine (DAB) (Dako, Carpinteria, CA) for 6 min to develop the chromogenic reaction. Sections were counterstained with hematoxylin for 20 s, rinsed in distilled water, and dehydrated through ascending grades of alcohol to xylene and coverslipped using a permanent mounting medium.
Slides stained for H&E and p-MAPK were cleaned with an isopropanol solution to prepare for digital slide scanning. The slides were then scanned using the Leica Biosystems Aperio AT2 Digital Whole Slide Scanner (Leica Biosystems, Inc. 1700 Leider Lane Buffalo Grove, IL). This machine uses line scanning technology to capture seamless, true-color, high quality, ultra-resolution digital slide images. After scanning, the resulting images were viewed using Aperio® ImageScope v. 12.0.1.5027 (Aperio Technologies, Inc. 1360 Park Center Dr Vista, CA), a viewing program designed to display and capture digitally scanned images.
Mitogen-activated protein kinase quantitative morphometry and artificial intelligence analysis
Twenty-four digital whole slide images (WSI) scanned on an Aperio (svs) slide scanner at 40X were analyzed at Charles River Laboratories, Inc, Durham, NC. Images were transverse sections of rat uteri. The set of 24 slides were immunohistochemically labeled for phospho-p44/42 MAPK. MAPK targets were visualized with 3,3’ DAB chromogen against a hematoxylin-stained background. Several slides had serial sections from the same animal mounted on them for a total of 38 uterine sections evaluated blindly without knowledge of control or dose groups. Scanned images of the MAPK labeled sections were first imported into the Visiopharm Image Analysis software (v.2019.09). With guidance from a board-certified veterinary pathologist the myometrium was manually segmented from the endometrium in a subset of the images. This was used by Dr Michael Staup (Charles River Laboratories, Inc, Durham, NC) to develop a convolutional neural network (CNN) for artificial intelligence (AI) and deep machine learning algorithms that could be applied to analyze the immunohistochemical stains (Supplemental Figure 2). Briefly, a post-processing step was used to automatically identify the mucosal (luminal) and glandular epithelium in the endometrium and segmentation of the myometrium (see Supplemental Figure 2). Labeling indices for MAPK were provided as a single area fraction for each compartment from each slide. The area fraction of MAPK expression was calculated for the mucosal (luminal) epithelium and glandular epithelium uterine tissue compartments of the endometrium.
RNA isolation
Adult (5–6-week-old) female Wistar Han rats were treated with 0, 25, 250, and 1000 mg/kg TBBPA by oral gavage in corn oil, 5 days per week for 13 weeks, 21 and uterine horns were collected after treatment and flash frozen. RNA extractions from frozen uterine horns were done as describe by Dunnick et al. 21 RNA from the original samples was extracted as described above and was used for real-time RT-PCR analysis in this study.
Real-time RT-PCR analysis
To determine Egfr, Erbb2, and Igf1r gene expression levels induced by TBBPA at 13 weeks, real-time RT-PCR was performed. Two micrograms of total RNA were used to prepare cDNA and primed with Egfr, Erbb2, Igf1r, and Gapdh primers (housekeeping gene as control) and reverse-transcribed with Superscript II (Invitrogen, Carlsbad, CA). The following primer sets specific for Egfr, forward primer 5’-ACCAGCAGGACTTCTTTCCCA-3’, and reverse primer 5’-TAAACTCACTGCTTGGCGGTG-3’; for Erbb2, forward primer 5’-ACATCTCAGCATGGCCAGACA-3’ and reverse primer 5’-TGTCAATGAGTACGCGCCATC-3’; for Igf1r, forward primer 5’-GCTACGTGAAGATCCGCCATT-3’ and reverse primer 5’- GCTGCAAGTTCTGGTTGTCCA-3’; and for Gapdh, forward primer 5’-ATCACCATCTTCCAGGAGCGA-3’ and reverse primer 5’- AGCCTTCTCCATGGTGGTGAA -3’, were used for gene expression studies. The Power SYBR Green PCR Master Mix (Applied Biosystems, Warrington, UK) was used, and RT-PCR was run by QuantStudio seven Flex (Applied Biosystems). The data analysis was based on the ΔΔCt method with normalization to GAPDH, and the results were expressed as fold changes as compared to untreated control groups.
Statistics
As most of the data were not normally distributed, a nonparametric statistical method, the Mann–Whitney ranked test, was used in the comparisons. 22 This method was used for RTKs, Western blot, IHC, and RT-PCR results. R version 3.5.3 was applied to analyze the data. The analysis of RTKs dot intensity value and IHC staining value were based on four replicates by way of one-way test. The analysis of Western blot densitometry value and RT-PCR fold changes were based on three replicates by way of one-way test. The differences were considered significant when a P-value of less than .05 was achieved.
Results
Phosphorylated receptor tyrosine kinases were differentially overexpressed in the uteri of tetrabromobisphenol -exposed rats at postnatal days 21, 33, and 90
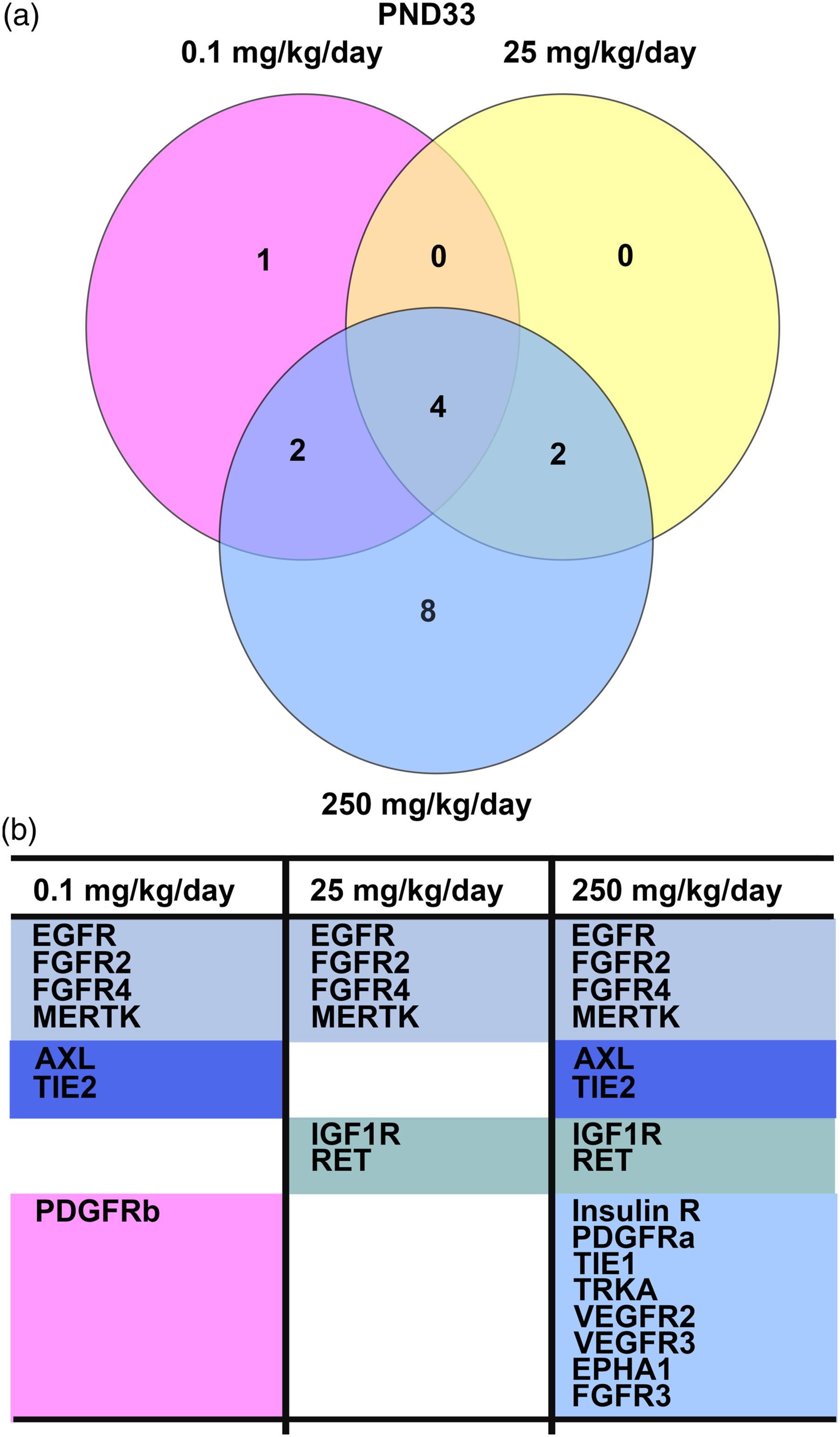
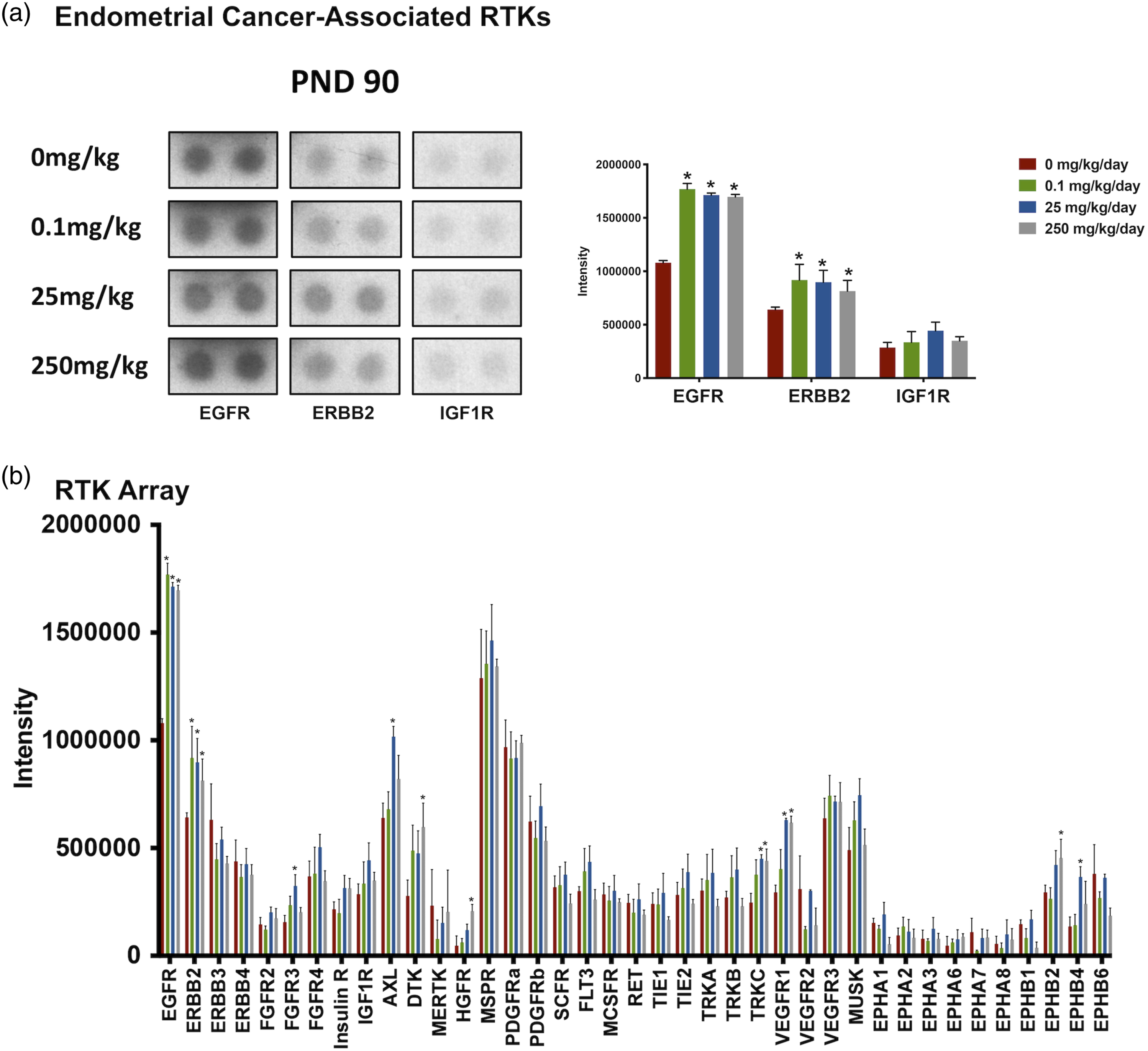
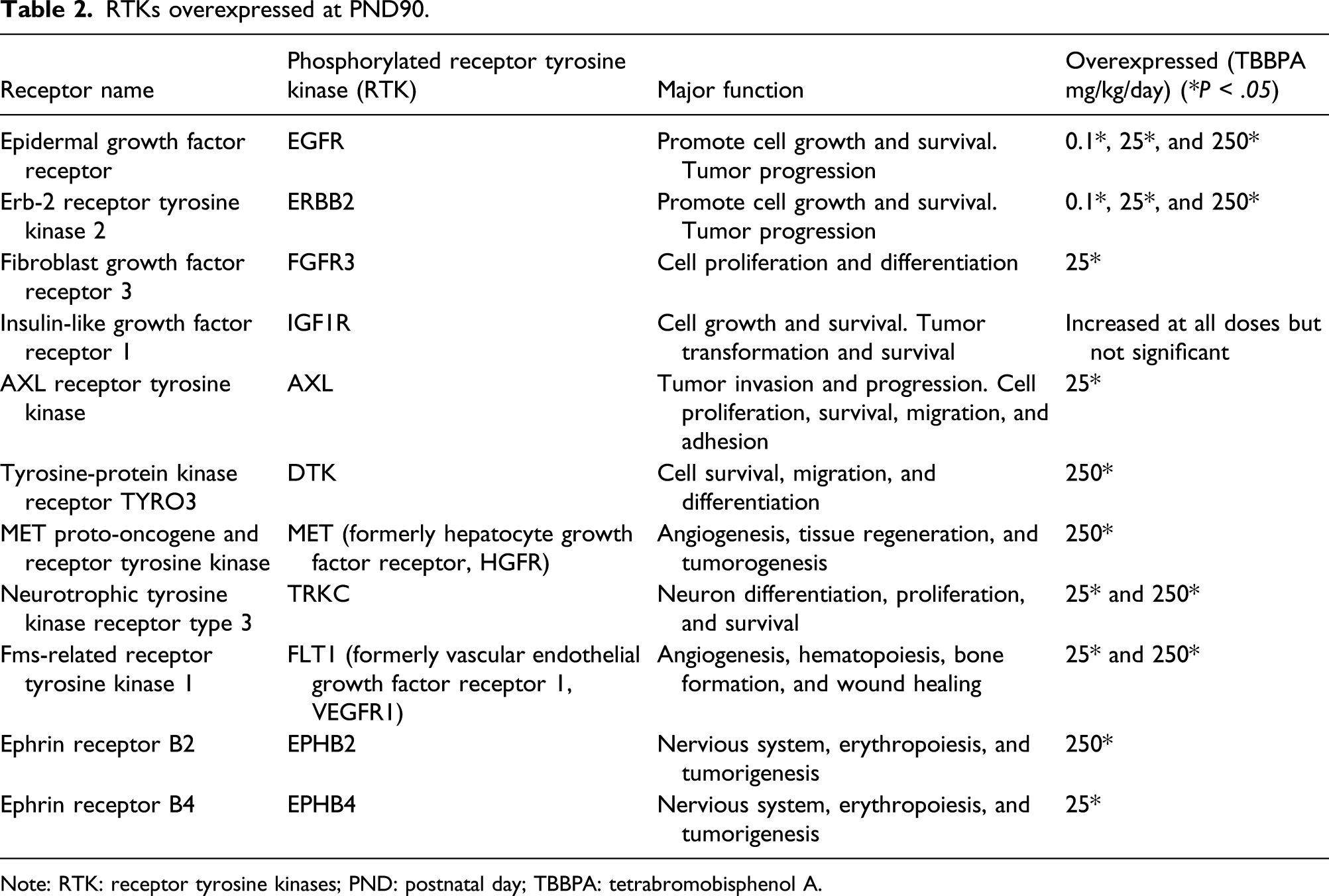
Phospho-receptor tyrosine kinase (phospho-RTK) arrays were done to determine the expression of activated growth factor RTKs in tissue from the uterine horns of Wistar Han rats following TBBPA treatment. At PND 21, animals had been exposed in utero and lactation period and were sexually immature at time of evaluation. Three RTKs most commonly overexpressed in endometrial cancer in women, EGFR, ERBB2, and IGF1R, were minimally increased, but not significantly compared to controls (Supplementary Figure 3(a)). The PND 21 RTK array showed activated receptors FGFR2, FGFR3, and TRKC significantly increased at 250 mg/kg TBBPA, whereas EPHA1 was significantly increased at 0.1 mg/kg TBBPA compared to controls (Supplementary Figure 3(b)). The RTK array at PND 21 also showed increased activated receptors including macrophage-stimulating protein receptor (MSPR; now MST1R) and ephrin receptors (EPHB2, EPHB4, and EPHB6), but none were significantly expressed (Supplementary Figure 3(b)). No overlap of significant RTKs was observed at PND 21 between the dose groups (Supplementary Figure 4). By PND 33, the activated RTKs, EGFR, and IGF1R, associated with human endometrial cancer, were significantly increased at all TBBPA dose groups for EGFR and the mid- and high doses of TBBPA for IGF1R, compared to controls (Figure 1(a)). There was also increased expression of ERBB2, another receptor important in endometrial cancer in women, at all doses compared to controls; however, this change was not significant. The RTK array at PND 33 showed significantly (P < .05) increased expression of the following activated receptors at different treatment concentrations: FGFR (2, 3, and 4), insulin R, AXL, MER proto-oncogene, tyrosine kinase (MERTK), platelet-derived growth factor (A and B), RET, TIE (1 and 2), TRKA, VEGFR (2 and 3), and EPHA1compared to controls (Figure 1(b); Table 1). The activated receptors EGFR, FGFR4, FGFR2, and MERTK were significantly overexpressed in all dose groups, whereas expression PDGFRb was significantly increased at 0.1 mg/kg TBBPA at PND 33 (Figure 2(a) and (b)). IGF1R and RET were overexpressed at 25 and 250 mg/kg TBBPA, and TIE2 and AXL expression was increased at 0.1 and 250 mg/kg TBBPA at PND 33 (Figure 2(a) and (b)). Insulin R, PDGFRa, TIE1, TRKA, VEGFR2, VEGFR3, EPHA1, and FGFR3 were activated at 250 mg/kg at PND 33 (Figure 2(a) and (b)). At PND 90, EGFR and ERBB2 were significantly increased at all doses compared to controls (Figure 3(a) and (b) and Table 2). MET proto-oncogene, receptor tyrosine kinase (MET; formerly hepatocyte growth factor receptor, HGFR), and Fms-related receptor tyrosine (FLT1; formally, vascular endothelial growth factor receptor 1, VEGFR1), FGFR3, AXL, TRKC, EPHB2, and EPHB4 were significantly (P < .05) increased at different doses compared to controls (Figure 3(b) and Table 2). Insulin-like growth factor receptor 1 was also activated compared to controls through PND 90, although not significantly (see Figure 3(a); Table 3). The activated receptor EGFR and ERBB2 was overexpressed in all dose groups, whereas VEGFR1 and TRKC showed increased expression at 25 and 250 mg/kg TBBPA at PND 90 (Figure 4(a) and (b)). AXL, EPHB4, and FGFR3 were activated at 25 mg/kg TBBPA, whereas DTK, EPHB2, and HGFR were activated at 250 mg/kg TBBPA at PND 90 (Figure 4(a) and (b)). Only EGFR remained significantly (P < .05) increased from PND 33 through PND 90 for all dose groups (see Table 3). Receptor tyrosine kinases phosphorylation in Wistar Han rats after treatment with 0, 0.1, 25, and 250 mg/kg/day TBBPA at PND 33. RTKs overexpressed at PND33. Note: RTK: receptor tyrosine kinases; PND: postnatal day; TBBPA: tetrabromobisphenol A. Overlapping of significantly activated RTKs in Wistar Han Rats after treatment with 0, 0.1, 25, and 250 mg/kg/day TBBPA at PND 33. Receptor tyrosine kinases phosphorylation in Wistar Han rats after treatment with 0, 0.1, 25, and 250 mg/kg/day TBBPA at PND 90. RTKs overexpressed at PND90. Note: RTK: receptor tyrosine kinases; PND: postnatal day; TBBPA: tetrabromobisphenol A. RTKs remain overexpressed in various dose groups from PND33 to PND90. Note: RTK: receptor tyrosine kinases; PND: postnatal day; TBBPA: tetrabromobisphenol A. Overlapping of significantly activated RTKs in Wistar Han rats after treatment with 0, 0.1, 25, and 250 mg/kg/day TBBPA at PND 90. 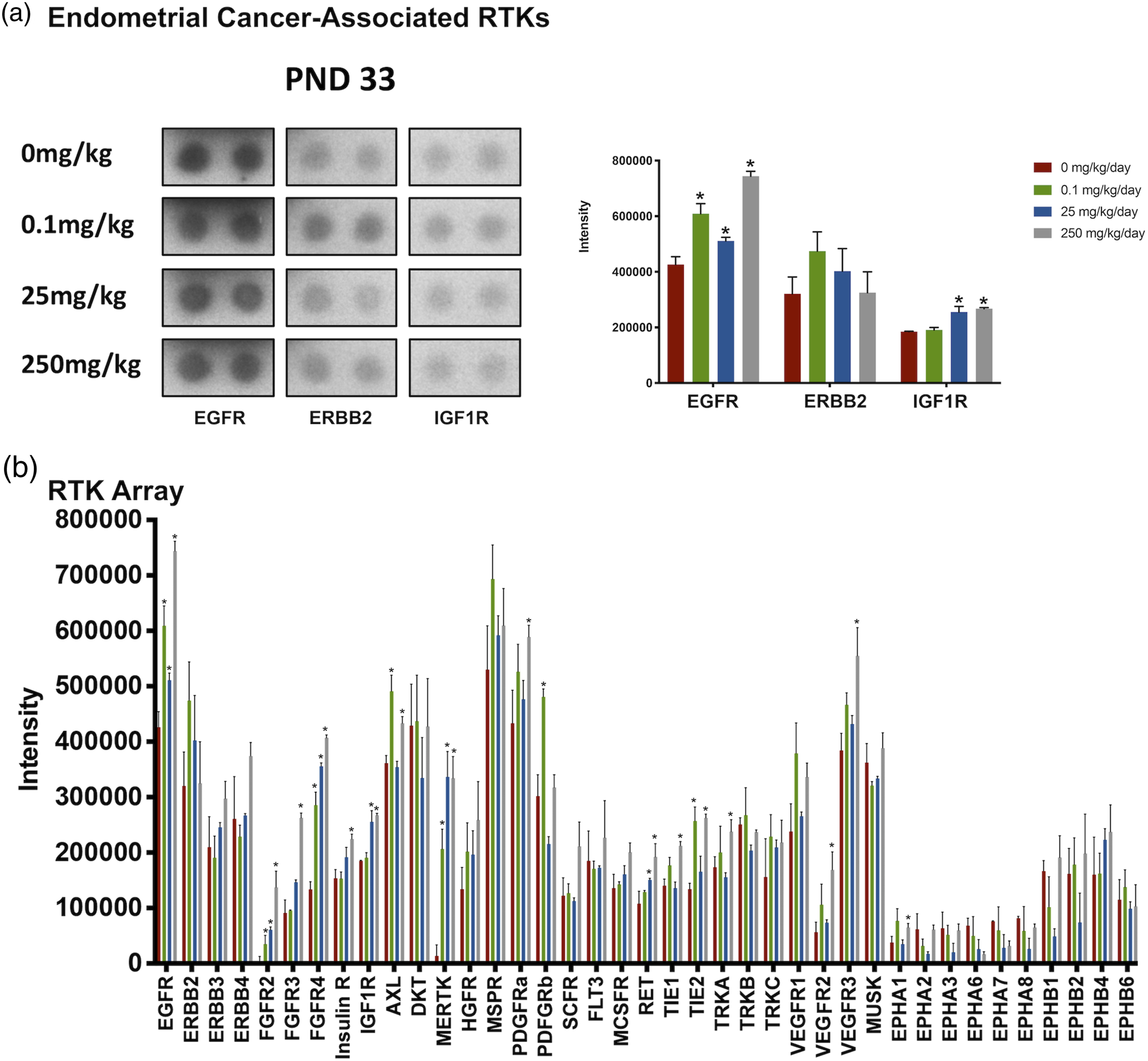


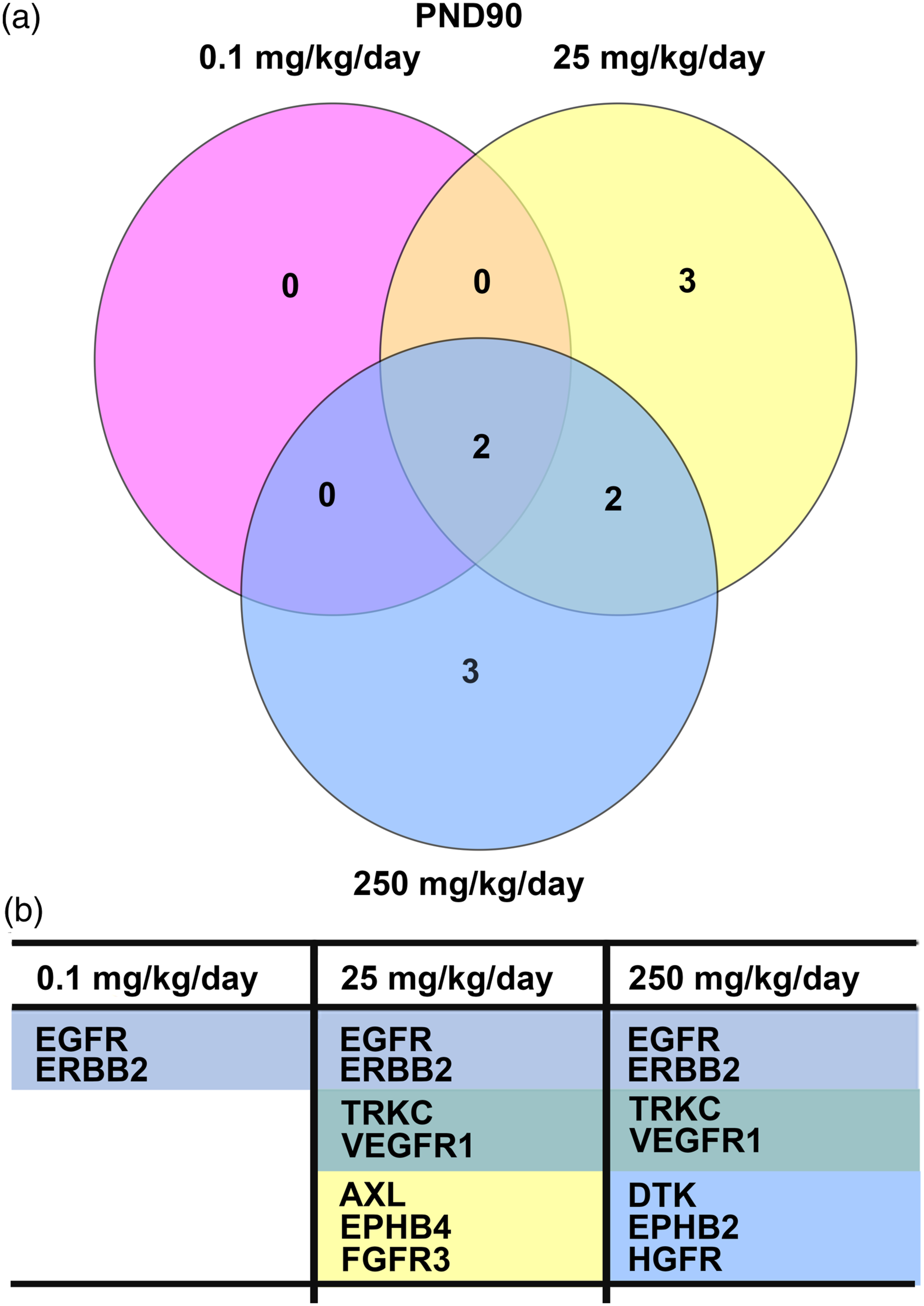
Phosphorylated epithelial growth factor receptor, Erb-B2 receptor tyrosine kinase 2, and insulin-like growth factor receptor 1 expression was increased in the uteri of tetrabromobisphenol A-exposed rats at postnatal day 90
Further studies were done to confirm the activated receptor expression levels of phosphorylated EGFR, ERBB2, and IGF1R observed in uterine samples at PND 90 due to their importance in endometrial cancer in women and reported increases in mRNA expression of Erbb2 in TBBPA-induced tumors from rats in the NTP 2-year bioassay
14
and Igf1 growth factor expression in adult rat uteri following TBBPA administration.
17
Western blot analysis was conducted to confirm the expression of activated EGFR, ERBB2, and IGF1R at PND 90 for all groups (Figure 5). The RTK array results represented a broad spectrum of activated tyrosine kinases for EGFR, ERBB2, and IGF1R; however, for Western blots, we selected a specific receptor tyrosine site for each receptor that leads to the activation of the downstream effector phosho-p44/42 MAPK pathway. We found that the ratio of phosphorylated to total EGFR protein expression was increased in all dose groups and significantly (P < .05) increased at 0.1 and 25 mg/kg compared to the control group (Figure 5(a)). However, the ratio of phosphorylated to total Erb-B2 (Figure 5(b)) and IGF-1R (Figure 5(c)) protein expression was significantly (P ≤ .05) increased at all treatment group levels compared to the respective control groups. These results confirmed the increased phosphorylation of EGFR, ERBB2, and IGF1R in the uteri of rats at PND 90 in TBBPA-treated groups compared to controls observed in the RTK arrays (see Figure 3). Protein expression of phospho-EGFR, phospho-ERBB2, and phospho-IGF1R after treatment with 0, 0.1, 25, and 250 mg/kg/day TBBPA at PND 90. 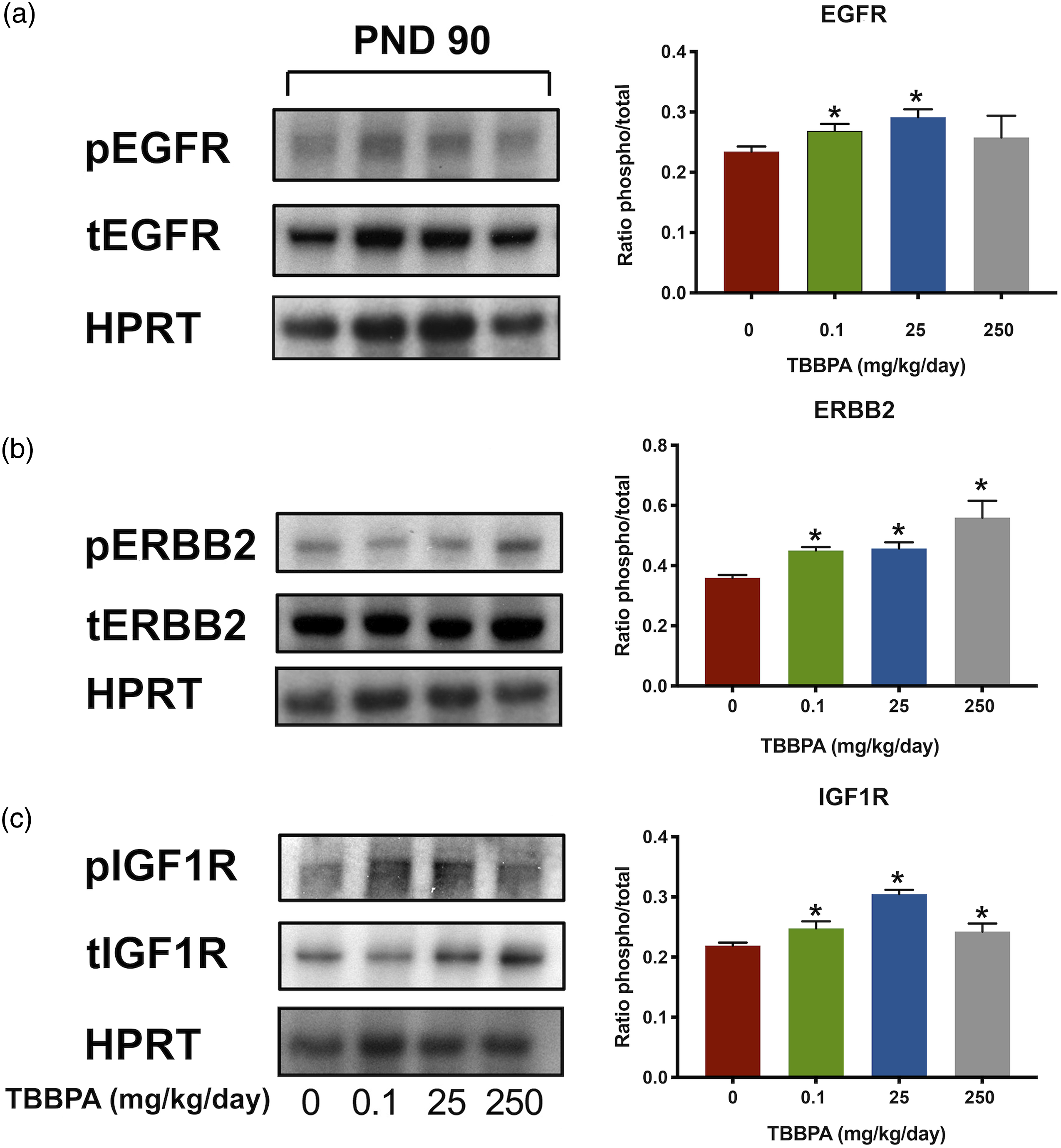
The receptor tyrosine kinases downstream effector protein and mitogen-activated protein kinase 44/42 expression was increased in the uteri of tetrabromobisphenol A-exposed rats at postnatal day 90
To determine the involvement of the downstream effector MAPK44/42 on the effects of differentially activated RTKs in uteri of TBBPA-treated rats at PND 90, Western blot analysis was done for all dose groups. The phosphorylated MAPK44/42 protein expression was significantly (P < .05) increased at PND 90 in uteri from all dose groups compared to controls (Figure 6). Protein expression of phospho-MAPKp44/42 after treatment with 0, 0.1, 25, and 250 mg/kg/day TBBPA at PND 90. Western blot analysis of phospho-MAPK p44/42. Phospho-MAPK p44/42 was significantly (*P < .05) increased at all dose groups compared to controls. Experiments were repeated independently in triplicate. Note: MAPK: mitogen-activated protein kinase; TBBPA: tetrabromobisphenol A; PND: postnatal day.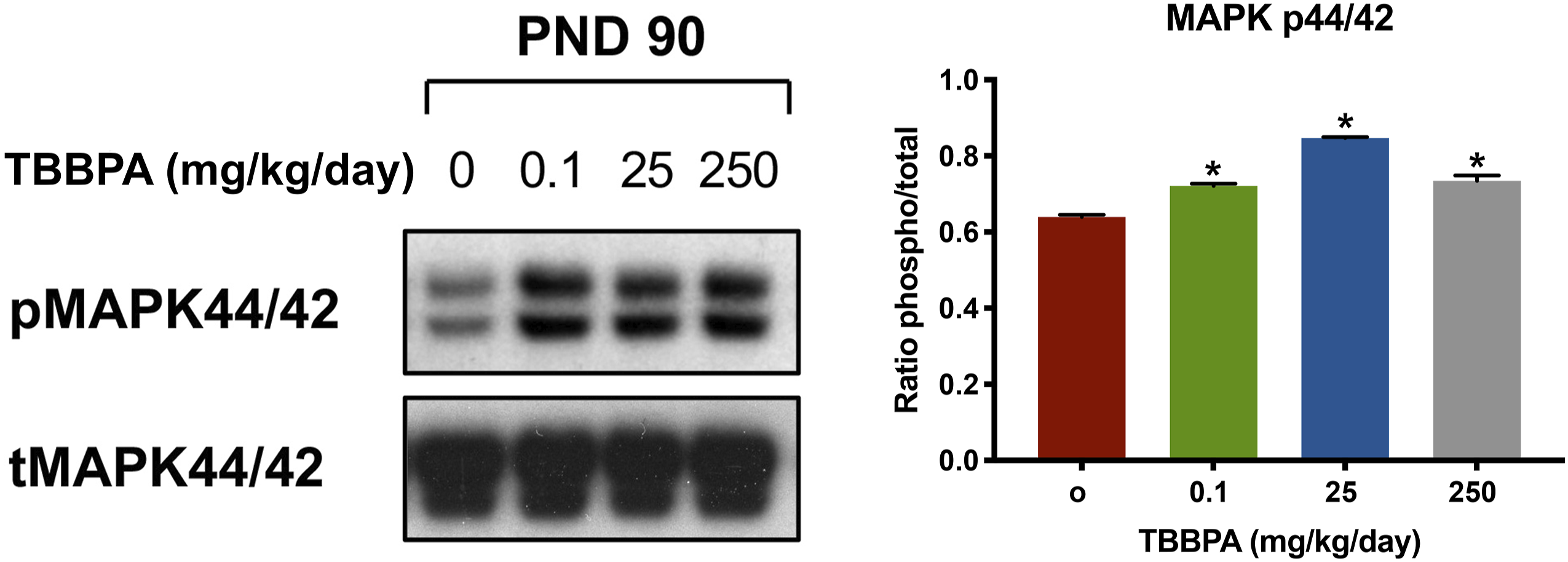
Mitogen-activated protein kinase protein, expression was increased in the luminal and glandular epithelium of the uteri of tetrabromobisphenol A exposed rats at all dose groups at postnatal day 90
We evaluated the expression of MAPK in the target cells of endometrial cancer, the endometrial luminal, and epithelial cells. Immunohistochemical images showed MAPK expression was located in the nuclei of the endometrial glandular and luminal epithelial cells in all dose groups compared to respective controls (Figure 7(a)–(c)). Deep machine learning and AI analyses of uteri from rats at PND 90 were done for all dose groups and controls, to determine the area fraction of MAPK-positive staining of endometrial luminal and glandular epithelium of the rat uteri. The luminal epithelium showed a MAPK staining increase of 3.3% at 0.1 mg/kg/day, 2.1% at 25 mg/kg/day, and 1.8% at 250 mg/kg/day compared to 1.2% at 0 mg/kg (control). However, the MAPK percentage staining was significantly (P < .05) increased in the luminal epithelium at 0.1 mg/kg TBBPA. The glandular epithelium showed a MAPK staining increase of 2.2% at 0.1 mg/kg, 1.9% at 25 mg/kg, and 1.3% at 250 mg/kg compared to 0.8% at 0 mg/kg (control). However, the MAPK percentage staining at 0.1 mg/kg TBBPA was significantly (P < .05) increased in the glandular epithelium. These results support the MAPK pathway as one of the downstream effectors activated by the RTKs and it was activated in the endometrial luminal and glandular epithelial cells which are the target cells in many of the uterine tumors (adenoma and adenocarcinomas) observed in rats in the TBBPA 2-year NTP bioassay, and similar to proposed target cells of endometrial cancer observed in women. Immunohistochemical images and AI analysis of MAPK positive staining of the endometrial glandular and luminal epithelium of TBBPA-treated (0.1, 25, and 250 kg/kg/day) Wistar Han rats at PND 90 and evaluated using DML and AI analysis. 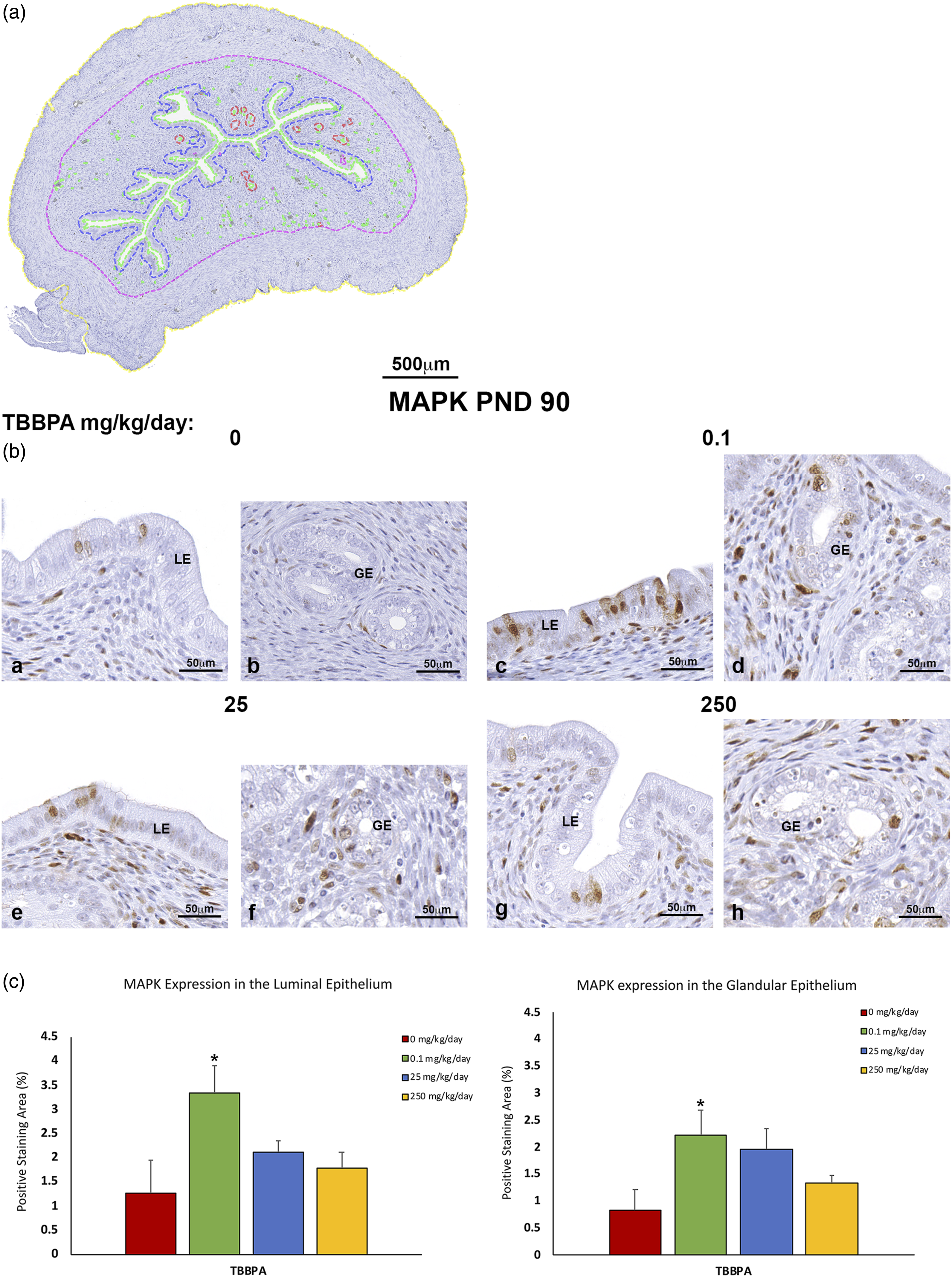
Epithelial growth factor receptor, Erb-B2 receptor tyrosine kinase 2, and insulin-like growth factor receptor 1 gene expression was increased in adult Wistar Han rats treated with tetrabromobisphenol A for 13 weeks
To determine if TBBPA-induced overexpression of Egfr, Erbb2, and Igf1r was a phenomenon noted only after in utero or early developmental exposures, we conducted gene expression analysis of frozen uterine samples taken from adult rats exposed to TBBPA for 13 weeks (oral gavage in corn oil, 5x/week) (Figure 8). Egfr was significantly (P < .05) increased for all dose groups, and Erbb2 was significantly increased (P < .05) in the 0.1 and 25 mg/kg/day groups compared to the controls. Igf1r was significantly increased (P < .05) at all dose groups compared to controls. mRNA expression of Egfr, Erbb2, and Igf1r in Wistar Han rats treated at 5–6 weeks of age with 0, 25, 250, and 1000 mg/kg/day TBBPA for 13 weeks. Egfr was significantly (*P < .05) increased at all dose groups compared to the control group. Erbb2 was significantly increased at 25 and 250 (*P < .05) mg/kg/day TBBPA compared to the control group. Igf1r was significantly (*P < .05) increased at all dose groups compared to the control group. Note: Egfr: epithelial growth factor receptor; Erbb2: epidermal growth factor receptor 2; Igf1r: insulin-like growth factor receptor 1; TBBPA: tetrabromobisphenol A.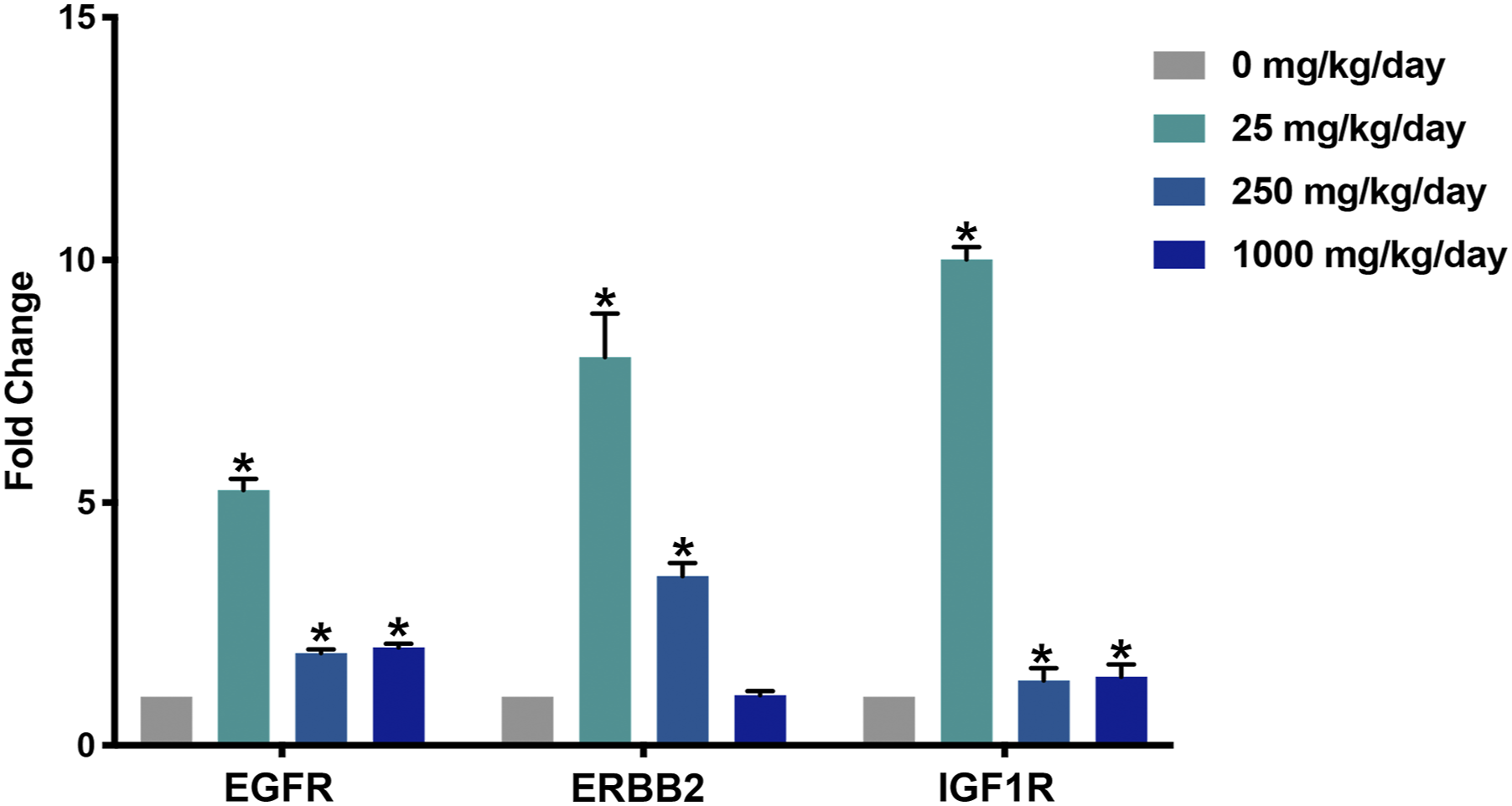
Discussion
A two-year bioassay conducted by the NTP found that Wistar Han rats developed endometrial adenoma, adenocarcinoma, atypical hyperplasia, and malignant mixed Müllerian tumors following exposures to TBBPA. 4 The mechanisms by which TBBPA induces tumor formation in the uterus of Wistar Han rats is still unclear; however, there is some evidence that suggests TBBPA may compete with estrogen for binding to sulfotransferases, which are important in estrogen metabolism and elimination, resulting in elevated levels of estrogen in the uterus. 4 Elevated estrogen levels in the uterus have been linked to increased risk of uterine cancer in women.23–25 Others have found that female Wistar Han rats treated with TBBPA for 5 days have upregulation of estrogen receptors (Esr1 and Esr2) in the uterus and changes in thyroid hormone receptors alpha and beta (Thra and Thrb), peroxisome proliferator-activated receptor alpha (Ppara), and glucocorticoid receptor gene (Nr3c1) expression. 15 These receptors are all involved in the regulation of estrogen homeostasis.
The RTKs are growth factor receptors that are thought to play a role in the promotion of tumor growth observed in TBBPA-treated rats. Harvey et al. 13 found a 30-fold overexpression of Erbb2 transcripts in uterine tumors taken from Wistar Han rats exposed to TBBPA in an NTP 2-year bioassay. Additionally, a significant upregulation of Igf1, the gene encoding IGF1, was found in the proximal and distal regions of the uterus of adult Wistar Han rats orally dosed with 250 mg/kg TBBPA for 5 days. 15 In the present study, Wistar Han rats exposed to TBBPA in utero and early in life up to PND 90 had dysregulated phosphorylated RTK expression of EGFR, ERBB2, VEGFR1 (FLT1), and HGFR (MET) in addition to others that were increased at PND 33 with EGFR remaining significantly increased through PND 90 compared to controls. EGFR is associated with many cancers including breast, endometrial, colorectal, and lung cancers. EGFR also plays an important role in regulating cell proliferation, inhibition of apoptosis (cell survival), and tumor progression.13,26–29 EGFR and ERBB2, another dysregulated RTK in uterine tissue, are both in the epidermal growth factor receptor family and are often overexpressed in breast and type II endometrial cancers.14,30 Other growth factor receptors that were found to be dysregulated in this study following TBBPA treatment were VEGFR1 (FLT1) and FGFR2-4, important in angiogenesis and proliferation, respectively, and both found to be overexpressed in endometrial cancer.13,31 The receptor tyrosine kinase AXL, that was significantly expressed at PND 33 and PND 90 in this study, is expressed in endometrial, breast, ovarian, and prostate cancers and is involved in tumor cell proliferation, survival, and at later stages of cancer in migration, invasion, and metastasis. 32 Overexpression or abnormal activation of RTKs has been linked to tumor formation and the development and progression of many different types of cancers.33–36
In the present study, rats were exposed to TBBPA in utero and up to PND 90 through lactation and by gavage at lower concentrations compared to animals in the NTP 2-year bioassay, although there was a group of rats in the current study treated at a concentration at which uterine tumors were observed (250 mg/kg) in the NTP 2-year bioassay. Overexpression of several RTKs were observed after TBBPA treatment; however, we conducted additional confirmation studies at PND 90 to further evaluate EGFR, ERBB2, and IGF1R, all heavily implicated in endometrial cancer in women
37
and Erbb2 also found to be overexpressed in endometrial adenocarcinomas in TBBPA-treated rats.
14
Additionally, we found that uterine samples from adult rats exposed to TBBPA for 13 weeks at concentrations (25, 250, and 1000 mg/kg/day) similar to those used for our in utero studies (25 and 250 mg/kg/day) and the NTP 2-year bioassay (250 and 1000 mg/kg/day) had significant increases in expression of Egfr, Erbb2, and Igf1r transcripts at the different treatment concentrations compared to controls. This points to the fact that regardless of age of exposure (in utero or adult) EGFR, ERBB2, and IGF1R protein and Egfr, Erbb2, and Igf1r transcripts are all overexpressed in the uterus in response to TBBPA treatment. The epidermal growth factor receptor family receptors consist of EGFR, ERBB2, HER3 (ERBB4), and HER4 (ERBB4). The epidermal growth factor RTKs are cytoplasmic membrane-anchored proteins that have similar structure and sequences and contain an extracellular ligand-binding, a transmembrane and an intracellular tyrosine kinase domain.38,39 We found that EGFR and ERBB2 expression was increased in the uteri of Wistar Han rats after treatment with TBBPA. ERBB2 has no known ligands but is able to bind other EGFR family members and activate downstream signaling pathways. Some of the signaling pathways that ERBB2 can affect are cell proliferation, cell survival, and cell differentiation.
30
Overexpression of ERBB2 has also been found in breast, lung, bladder, and gastric cancers.
39
EGFR more specifically has been shown to play a role in cell proliferation, cell survival, cell adhesion, and cell migration when tightly regulated; however, if overexpressed it can lead to the growth of tumor cells and tumor progression.
37
Our studies confirmed that EGFR was significantly upregulated at PND 33 at all doses and remained significantly increased at all dose groups compared to controls, at PND 90. The western blot analysis showed specific phosphorylation of EGFR at tyrosine site 845 (Y845) a tyrosine site that can be activated by Src and leads to transactivation of EGFR with downstream MAPK pathway activation that can result in cell proliferation or tumorigenesis (Figure 9). The mitogen-activated protein kinase p44/42 (MAPK) pathway is an essential component of the downstream signaling network of RTKs which results in cell proliferation.
40
We observed increased protein expression of activated MAPK compared to controls in uteri of TBBPA-treated rats and increased MAPK expression in uterine endometrial epithelial cells, the target cells of endometrial cancer. Proposed RTK activation pathway by TBBPA. TBBPA can cause phosphorylation of RTKs which in turn activate MEK 1/2 pathways resulting in activation of the downstream effector, MAPK p44/42. Note: RTKs: receptor tyrosine kinases; TBBPA: tetrabromobisphenol A; MAPK: mitogen-activated protein kinase.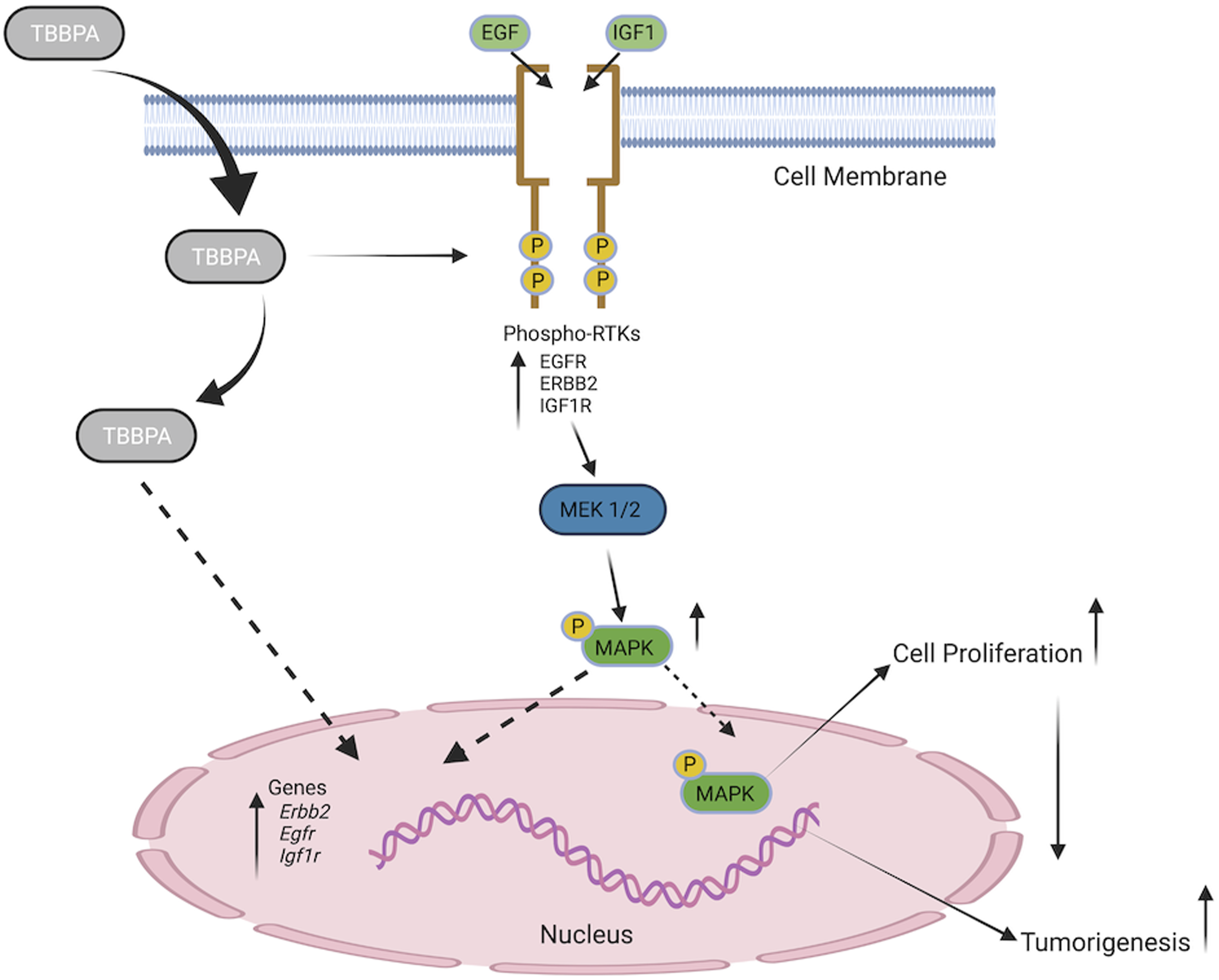
In summary, the results of this study show that TBBPA induces dysregulation of phosphorylated RTKs following in utero and early developmental exposures in Wistar Han rats. The sustained significant overexpression of EGFR, and to a lesser extent ERBB2 and IGF1R at PND 33 through PND 90 suggests that dysregulation of RTKs associated with endometrial cancers in women may be an early event preceding observable histopathologic changes in TBBPA-induced hyperplasia or endometrial cancer in Wistar Han rats that may be persistent during chronic exposures, since overexpression of Erbb2 was observed in endometrial tumors from rats in the NTP 2-year bioassay. 14 It is also important to take into consideration the limited tissue samples and group sizes used in this study, in addition, the mid- and high dose levels were much higher than environmental or occupational exposures for humans, although these higher concentrations were found to induce endometrial cancer in Wistar Han rats in a long-term bioassay. Further studies are needed to delineate a molecular mechanism, whereby TBBPA activates RTKs and determine the role of early dysregulation of RTK activation in the initiating and key events important in the pathogenesis of TBBPA-induced endometrial cancer using relevant human in vitro models.
Supplemental Material
sj-pdf-1-tor-10.1177_23978473211047164 – Supplemental Material for Differential receptor tyrosine kinase phosphorylation in the uterus of rats following developmental exposure to tetrabromobisphenol A
Supplemental Material, sj-pdf-1-tor-10.1177_23978473211047164 for Differential receptor tyrosine kinase phosphorylation in the uterus of rats following developmental exposure to tetrabromobisphenol A by Lysandra Castro, Jingli Liu, Linda Yu, Alanna D Burwell, Trey O Saddler, Lindsay A Santiago, William Xue, Julie F Foley, Michael Staup, Norris D Flagler, Min Shi, Linda S Birnbaum and Darlene Dixon in Toxicology Research and Application
Supplemental Material
sj-tiff-2-tor-10.1177_23978473211047164 – Supplemental Material for Differential receptor tyrosine kinase phosphorylation in the uterus of rats following developmental exposure to tetrabromobisphenol A
Supplemental Material, sj-tiff-2-tor-10.1177_23978473211047164 for Differential receptor tyrosine kinase phosphorylation in the uterus of rats following developmental exposure to tetrabromobisphenol A by Lysandra Castro, Jingli Liu, Linda Yu, Alanna D Burwell, Trey O Saddler, Lindsay A Santiago, William Xue, Julie F Foley, Michael Staup, Norris D Flagler, Min Shi, Linda S Birnbaum and Darlene Dixon in Toxicology Research and Application
Footnotes
Acknowledgments
The authors sincerely thank Drs Schantel Bouknight and Kristen Hobbie for their critical review of the article. This work was funded by the Intramural Research Program of the NIH, NIEHS, and DNTP. The authors also kindly thank Ms Sagi Encole Gillerand and Dr Suzanne Fenton for making provisions for rat uterine tissues, and Ms Elizabeth Ney for composing the MAPK immunohistochemistry plate. The authors appreciate the expert assistance of Dr Jianping Xue with statistics and the NIEHS immunohistochemistry and histology core laboratories with slide preparation and staining.
Author contributions
DD, LC, JL, and LY designed experiments and developed research concepts. LSB conceived and designed an NIEHS-wide TBBPA developmental exposure study. JL, LC, LY, LAS, ADB, JFF, NDF, MS, TOS, and DD performed experiments, collected, and/or analyzed data. LC wrote the initial manuscript draft. DD, LY, and JL contributed to manuscript revisions.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Intramural Research Program of the NIH, NIEHS, and DNTP (ES021196-27).
Supplementary material
Supplementary material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
