Abstract
The INHAND Project (International Harmonization of Nomenclature and Diagnostic Criteria for Lesions in Rats and Mice) is a joint initiative of the Societies of Toxicologic Pathology from Europe (ESTP), Great Britain (BSTP), Japan (JSTP), and North America (STP) to develop an internationally accepted nomenclature for proliferative and nonproliferative lesions in laboratory animals. The purpose of this publication is to provide a standardized nomenclature for classifying lesions observed in the urinary tract of rats and mice. The standardized nomenclature of urinary tract lesions presented in this document is also available electronically on the Internet (http://www.goreni.org/). Sources of material included histopathology databases from government, academia, and industrial laboratories throughout the world. Content includes spontaneous developmental and aging lesions as well as those induced by exposure to test materials. A widely accepted and utilized international harmonization of nomenclature for urinary tract lesions in laboratory animals will decrease confusion among regulatory and scientific research organizations in different countries and provide a common language to increase and enrich international exchanges of information among toxicologists and pathologists.
Introduction
The INHAND Project (International Harmonization of Nomenclature and Diagnostic Criteria for Lesions in Rats and Mice) is a joint initiative of the Societies of Toxicologic Pathology from Europe (ESTP), Great Britain (BSTP), Japan (JSTP), and North America (STP) to develop an internationally accepted nomenclature for proliferative and nonproliferative lesions in laboratory animals. The purpose of this publication is to provide a standardized nomenclature for classifying lesions observed in the urinary tract of rats and mice. The standardized nomenclature of urinary tract lesions presented in this document is also available electronically at the goRENI website on the Internet (http://www.goreni.org/) and may include additional photomicrographs and potentially additional updated information. This document follows a similar anatomical approach. A widely accepted and utilized international harmonization of nomenclature for urinary system lesions in laboratory animals will decrease confusion among regulatory and scientific research organizations and provide a common language to increase and enrich international exchanges of information among toxicologists and pathologists.
Generally used preferred terms of systemic nonproliferative lesions across organ systems, such as hemorrhage, periarteritis, or thrombosis are included in separate INHAND manuscripts currently under preparation and are therefore not included in this document except where there are distinguishing features of the process relevant to the urinary tract. Similarly, systemic tumors such as lymphoma or histiocytic sarcoma are described in separate documents under the hematopoietic system and therefore are also not discussed in this document. Tumors that occur at many sites or locations within the body are described under the specific organ system considered the most appropriate such as schwannoma in the nervous system, or hemangiosarcomas under the cardiovascular system and which will be described in other sections of the INHAND guides. Since most of the pathologic lesions of the renal pelvis are similar or identical to those in the lower urinary tract, description for both nonproliferative and proliferative lesions of the renal pelvis are included with the lower urinary tract rather than with the kidney in this document and specific features related to pathology of the pelvis are highlighted within that section.
The urinary system in all mammalian species, including mouse and rat, is represented by the kidney and lower urinary tract (ureters, bladder, and urethra). Although the primary function of the kidney is to eliminate waste products of metabolism, ancillary functions include hormone elaboration, control of body fluid volume, electrolyte regulation, low-molecular-weight (LMW) protein turnover, and metabolic processes. The function of the lower urinary system is to transport (and store) urine from the kidney for elimination from the body.
Nonproliferative degenerative lesions of the kidney are commonly encountered in rodents with advancing age, including those associated with the syndromes of chronic progressive nephropathies (CPNs). These conditions may be aggravated or exacerbated by xenobiotic treatment, making interpretation difficult. While modern laboratory animal management practices have limited the incidence of infectious processes in the kidney, inflammatory conditions related to infectious disease may still occur. However, they are not described in detail in this document. Excellent reviews are published elsewhere on the effects of infectious disease, diet, and aging in the rat and mouse urinary tract (Barthold 1998; Hard and Khan 2004).
The kidney is an extremely common target organ for therapeutic and diagnostic agents. Renal injury may occur as a result of direct effects on tubules or glomeruli, or indirectly via altered hemodynamics. There is a common tendency for renal changes to occur in preclinical toxicity studies, due to high administered doses, high renal blood flow (resulting in high peak concentrations of drugs), predisposition for renal excretion of many drugs and/or their metabolites, metabolic activity, high oxygen consumption, and ability to concentrate drugs in the urinary solvate. The manifestation and pattern of renal injury are dependent on the nature of the inciting agent and its particular mode of action. However, it is important to consider that the nephron often responds to perturbation as a unit, rather than only at the specific site of injury.
Proliferative lesions in laboratory rodents may arise spontaneously or as a result of exposure to potentially toxic test materials such as classic genotoxic carcinogens (Hard et al. 1995). The diversity of agents resulting in the development of renal tumors suggests that there are a variety of different mechanisms underlying rodent kidney carcinogenesis (Hard 1998a). While most or perhaps all nongenotoxic renal carcinogens are nephrotoxic, the converse is not necessarily true and there are many nephrotoxins which have not been demonstrated to be renal carcinogens.
Since the lower urinary system of the mouse and rat (ureter, bladder, and urethra) are composed of similar tissue layers (urothelium, subepithelial connective tissue, and smooth muscle), they have the potential to share similar proliferative and nonproliferative xenobiotically induced lesions. However, while lesions in the urinary bladder are frequently encountered in rodents, lesions of the ureters and urethra are relatively rare. The reason for this difference has been ascribed to the rapid passage of urine through the ureters and urethra as compared to the longer contact time of material stored within the bladder (Hard et al. 1999). The anatomic orientation of the bladder within the pelvis of the rat and mouse and the fact that they are horizontal quadrupeds may also predispose the anterior wall to retention of microcrystals and other particles as compared to humans and therefore increased likelihood of developing proliferative lesions in rodents (DeSesso 1995). Acute toxicity of the urothelium can be produced by reaction of the xenobiotic or its metabolite with the urothelium, production of urinary solids or by significant alterations of normal urinary constituents and can range from effects on only the superficial urothelium to full thickness ulceration.
Morphology
The kidneys of the rat and mouse are unipapillate and retroperitoneal with the right kidney slightly cranial to the left. There is a single wedge-shaped papilla projecting into the renal sinus and surrounded by the renal pelvis, which is continuous with the ureter. The functional unit of the kidney, the nephron, is divided into the glomerulus, proximal tubule, descending and ascending limbs of Henle, distal convoluted tubules, connecting segment, collecting duct system, interstitium, and juxtaglomerular apparatus (JGA). There are approximately 30,000 nephrons in an adult rat kidney and 10,000 in the mouse kidney, but there is some variation by strain and age, with progressive loss over time. The rodent kidney can also be divided into five topographic zones: cortex, outer stripe of the outer medulla, inner stripe of the outer medulla, inner medulla, and papilla. Whenever possible, toxic renal responses should be classified on the basis of structure and topographical location.
The mature rat kidney weighs approximately 0.51-1.08% (mean 0.65%) of the body weight, while the mature mouse kidney weight varies considerably with age, breed and especially gender and is approximately 1.15-2.25% of bodyweight with males having greater kidney weights than females (Schlager 1968). The vascular supply arises from the renal artery, branching into the interlobar arteries, and continuing as arcuate arteries running parallel to the capsule along the corticomedullary junction. These continue as interlobular arteries and eventually to afferent arterioles and glomerular capillaries. Efferent arterioles that arise from glomeruli near the medulla give rise to vasa recta which supply the medulla. These vessels eventually coalesce to form arcuate veins.
The glomerulus consists of a capillary network projecting into a capsular space (Bowman’s capsule). The glomerular tuft is lined by endothelial cells, overlying a fenestrated basal lamina and opposite a layer of podocytes. There is an adjacent central region composed of mesangial cells. There is a single basement membrane, which is highly negatively charged. Passage of plasma proteins larger than 70 kD is normally restricted by this charge and size selective barrier. In mice, the glomerular size relative to total kidney weight is smaller than in other species, including rat. Glomerular size tends to increase with age and can vary among strains of rodents. In normal mature male mice, and occasionally also in mature male rats, proximal convoluted tubular epithelial cells extend into Bowman’s capsule.
Proximal tubules make up the majority (some 75%) of the structural subunits of the renal cortex. In the rat, the proximal tubule can be subdivided into the S1 and S2 (convoluted) segments and the S3 (pars recta or straight) segment. In the mouse, it is more difficult to discern the transition from the S2 to S3 segment than in the rat where it tends to be abrupt. The S1 segment is often noted with an oval or transverse profile in tissue section and has a thick, well-developed brush border and numerous basolateral elongated mitochondria. The S2 segment has a shorter brush border with fewer mitochondria, but more pronounced lysosomal bodies. Convolutions or transverse profiles are more prominent in tissue sections. The S3 or straight segment has both transverse and linear (longitudinal) profiles in section with sparse phagolysosomal droplets, but a slightly taller brush border than the other segments. Pars recta of juxtamedullary nephrons are located within the outer stripe, while those associated with short-looped nephrons are localized to both outer stripe and within medullary rays.
The thin limb of the descending loop of Henle transitions abruptly from the S3 segment, forming the boundary between the outer and inner stripes of the outer medulla. Juxatamedullary nephrons have long thin segments that can extend deep into the papilla, while short-looped (subcapsular) nephrons have thin limbs of Henle that are shorter and distributed throughout the outer medulla. These tubules are lined by flattened, amphophilic epithelium and very sparse microvilli. The outside diameter is markedly smaller than the proximal tubules, while the luminal diameter is only slightly smaller. The thin ascending limb becomes the thick ascending limb and returns to the cortex within a medullary ray. The thick ascending limb is considered part of the distal nephron, which is also made up of the macula densa and distal convoluted tubules. The thick limb ends near its own glomerulus, just past the macula densa. Cells lining the thick ascending limb are cuboidal and eosinophilic, with a prominent ovoid central nucleus, and are smaller than cells found in the proximal convoluted tubules. Tamm-Horsfall protein is affixed along the luminal membrane surface and immunohistochemical stains for Tamm-Horsfall prominently label these structures.
The JGA is located at one pole of the glomerulus and consists of the macula densa, efferent and afferent arterioles, the renin secreting granular cells of the afferent arteriole, and the extraglomerular lacis (mesangial) cells. Cells of the macula densa are low columnar with apical nuclei. The JGA is important in tubuloglomerular feedback control of renin secretion.
The distal convoluted tubules are short segments near the glomeruli which begin beyond the macula densa and extend to the connecting tubules which are continuous with the collecting ducts. The connecting tubules are ill-defined in the rat and mouse. Cells lining the distal convoluted tubules are somewhat taller than those of the thick ascending limb and the lumina are variably wider.
The collecting ducts extend from the cortex via the medullary ray through the outer and inner medulla to the tip of the papilla. The lining cells begin as low cuboidal but increase in height to low columnar in the papilla.
The renal pelvis is lined by transitional epithelium (urothelium), but as compared to the epithelium of the bladder, it is much thinner. There is a slightly altered form of epithelium lining the papilla, with only a single layer of cells present at the tip.
The interstitium is composed of a matrix of fibrocytes and dendritic cells and represents only about 7% of the cortical volume in the rat, while the medullary interstitium accounts for up to 29% of the volume of the medulla. The interstitium of the medulla (especially the papilla) is rich in mucopolysaccharides and contains several types of interstitial cells embedded within extracellular matrix elements. Type I (stellate) cells are lipid-rich and associated with prostaglandin production, while type 2 (monocytic) cells are round, contain large nuclei, and scant cytoplasm. Type 3 (pericyte) cells are flattened and are often associated with the vasa recta.
Localization of the lesion to a specific segment within the kidney may often be of great value in helping to determine the mechanism for xenobiotic-induced lesions in rodents. While morphologic differences as described in the preceding paragraphs will often provide segment specificity, immunohistochemistry can be a useful adjunctive tool for aiding in segment identification of lesions. For example, immunohistochemical staining for Tamm-Horsfall protein will label thick ascending limbs in all species, but this immunostain also labels distal convoluted tubule in rats and mice (Vekaria et al. 2006). Immunohistochemical stains directed against calbindin-D28 K specifically label distal convoluted tubules in rodents (Timurkaan and Tarakci 2004). Alpha-GST immunostains label proximal tubules including S1 and S2 segments of the proximal convoluted tubules as well as S3 straight segments. Aquaporin-2 immunohistochemistry can be used to label collecting ducts, both in the medulla and those extending into the cortex (Vekaria et al. 2006).
The lower urinary tract of the rodent is comprised of the paired ureters, the urinary bladder, and the urethra. The epithelial lining of all three organs is comprised of urothelium (transitional epithelium), which is formed by a series of three or more epithelial layers, and while normally of low mitotic activity, the urothelium is quite susceptible to hyperplastic responses. The most basal cuboidal layer is metabolically active, while the larger intermediate cell layers have multiple interdigitations and the superficial domed cells are large, polygonal, and have a thick asymmetric unit membrane. The urothelium is surrounded by a layer of variably thick connective tissue and two layers of smooth muscle (inner circular layer and outer longitudinal layer) as well as an outer adventitial covering. A basement membrane separates the urothelium from the underlying connective tissue.
The ureter is continuous with the kidney pelvis and enters dorsolaterally in the wall of the bladder where it extends obliquely through the muscular layers of the wall before emptying into the lumen at the trigone laterally. Because of constrictions at the ureteropelvic junction at the pelvic brim and at the entrance point to the bladder wall, these two areas are the most common sites of obstruction.
The urinary bladder of rodents is found within the pelvis, ventral to the colon. The anterior portion in rodents is called the dome, and the posterior segment at the exit of the urethra is referred to as the trigone. Maximum urine volumes under normal conditions in both rats and mice range up to 1 mL, but larger volumes occur with obstruction or neoplasia. Small numbers of inflammatory cells, especially mast cells and/or lymphocytes, are noted normally in the supepithelial compartment, particularly in the mouse.
The urethra is short in females and significantly elongated in males. In the male it extends from the bladder through the pelvic girdle and through the penis to its tip. In both male and female rodents, the vast majority of the lining epithelium is composed of (transitional) urothelium, but it can be occasionally replaced at certain points by stratified squamous epithelium, particularly near the external orifice.
Inhand Nomenclature of the Kidney
Congenital Lesions of the Kidney
Adrenal Rest: Capsule or Subcapsular Cortex
Species: rat, mouse
Synonyms: ectopic adrenal, adrenocortical choristoma
Pathogenesis/cell of origin A developmental abnormality that results from a cluster of cells which undergo disordered migration during organogenesis in the embryo
Diagnostic features Small aggregation of well-differentiated adrenocortical cells Attached to exterior of capsule or located subcapsularly
Differential diagnoses Neoplasia: invasive and not recognizable as well-differentiated adrenal gland Adrenal hamartoma: non-neoplastic but disorganized or less differentiated adrenal cells
Comment: Adrenal rests are a congenital anomaly and have little toxicologic significance. In humans they rarely may undergo malignant transformation, but this has not been conclusively demonstrated in rodents (Prentice and Jorgenson 1979; Goren, Engelberg, and Eidelman 1991).
Agenesis: Cortex and/or Medulla
Species: rat, mouse
Synonyms: unilateral hypoplasia, renal aplasia
Pathogenesis/cell of origin A unilateral developmental abnormality, where one entire kidney or a significant portion thereof fails to form from the metanephric ducts; bilateral agenesis is a lethal trait. May be a heritable defect in some strains. Ret kinase is involved in metanephric differentiation and drugs targeting this enzyme or closely related factors may induce renal agenesis, such as syk kinase inhibitors or other teratogenic drugs.
Diagnostic features Noted macroscopically as missing or markedly hypoplastic kidney Incidence varies markedly within breeds but is rare in most species used in toxicololgic testing
Comment: Renal agenesis is the gross macroscopic term for this condition. Renal aplasia usually refers to the complete lack of any identifiable renal parenchyma, while some remnants may be present and the term agenesis is more appropriate. Histologic diagnoses of “kidney, absent, unilateral” or “kidney, missing” are often employed in microscopic incidence tables. Transgenic mice lacking the Ret kinase gene as well as certain strains of mutant rats exhibit unilateral urogenital malformations including renal agenesis, and drugs that affect or inhibit metanephros differentiation are also associated with agenesis of the kidneys (Clemens et al. 2009; Chen et al. 1995; Amakasu, Suzuki, and Suzuki 2009).
Hypoplasia: Cortex and/or Medulla
Species: rat, mouse
Synonyms: none
Pathogenesis/cell of origin Renal hypoplasia is a quantitative defect caused by a reduced mass of metanephric blastema or by incomplete induction of nephron formation by the ureteral bud (Bush, Stuart, and Nigam 2004; Neiss 1982).
Diagnostic features Noted macroscopically as abnormally small kidney Incidence varies markedly within breeds but is rare in most rodent species Generally unilateral, with hypertrophy of the contralateral kidney; when bilateral it tends to result in early mortality Most often characterized by a reduced number of nephrons
Differential diagnoses Renal dysplasia: abnormal elements are present, including primitive mesenchymal tissues or primitive ectodermal structures resembling metanephric ducts. Renal agenesis: Complete absence of one kidney or small unrecognizable remnant. For the diagnosis of renal hypoplasia, there should be some relatively normal renal structures (nephrons remaining but they are reduced in size and/or number).
Comment: Renal hypoplasia is most often encountered in mutant strains of mice and rats such as the hgn strain, but it also is found rarely as a spontaneous lesion in reproductive toxicology studies and can occur with teratogenic agents affecting metanephric differentiation similar to those agents inducing renal agenesis (Ret kinase inhibitors or syk kinase inhibitors; Suzuki et al. 2007; Suzuki and Suzuki 1995; Clemens et al. 2009).
Dysplasia: Cortex, Medulla
Species: rat, mouse
Synonyms: none
Pathogenesis/cell of origin An abnormality that results from disorganized development of renal parenchyma associated with altered differentiation of specific cellular elements of renal tissue
Diagnostic features Presence of anomalous tissue within the cortex or medulla including any of the following: persistent primitive mesenchyme, persistence of ectodermal structures or atypical tubules, or tissues such as cartilage that are inappropriate for their location There may be interstitial fibrosis or amorphous hyaline material separating tubules Kidneys may be decreased in size and misshapen
Differential diagnoses Neoplasia: invasive and usually involves only one disordered cell type Hypoplasia: decreased nephrons and renal size but no abnormal tissue
Comment: Renal dysplasia has been recognized in both rats and mice, and while most cases are congenital, dysplasia may also rarely be associated with exposure to teratogenic agents during the prenatal or early postnatal period.
Epithelial (Tubular) Changes of the Kidney
Atrophy, Tubule (Figures 1 and 2): Proximal and Distal Tubules
Species: rat, mouse
Synonyms: none
Pathogenesis/cell of origin renal tubular epithelium generally limited to cortex. Since all renal components are interdependent, irreversible damage to a major percentage of either glomeruli or tubules will eventually result in impaired function, decompensation, and end stage kidney disease, the hallmark of which is marked tubular atrophy, glomerular obsolescence and shrunken, scarred kidneys; Diffuse cortical atrophy may also occur as a result of hydronephrosis with compression atrophy of the tubules
Diagnostic features Contraction and collapse of tubule with obliteration of lumen Variable peritubular interstitial fibrosis Tubular basal lamina is often thickened
Differential diagnosis Infarction: may involve both tubular atrophy and fibrosis, but in a wedge-shaped distribution due to its vascular pathogenesis
Comment: Tubular atrophy is a constant feature noted in rats and mice with progressive renal failure and is frequently present in late stage CPNs. In rodent models of progressive renal failure, apoptosis of the renal tubular epithelium has been shown to be an integral factor involved in the pathogenesis of tubular atrophy. Increased reactive oxygen species and a renal environment favoring proapoptotic signals contribute to cell death. TGF-beta has been shown to play a major role in the induction of fibrosis accompanying tubular atrophy, and involves a complex interaction between atrophic epithelium, basement membrane, interstitial fibroblasts, and epithelial–mesenchymal transdifferentiation (Frazier et al. 2000).
Degeneration, Tubule (Figures 3 and 4): Proximal and Distal Tubules, Collecting Ducts
Species: rat, mouse
Synonyms: degeneration/regeneration
Pathogenesis/cell of origin renal tubular or ductular epithelium Like necrosis, tubular degeneration can arise from a variety of agents involving hypoxia, disruption of ATP production, mitochondrial injury, free radical formation, peroxidation, or perturbed cell signaling Ultrastructural changes can include glycogen loss, loss of microvilli, vesiculation, nuclear clumping, or swelling of the endoplasmic reticulum Tubular vacuolation may be the first manifestation, followed by other histomorphologic changes
Diagnostic features Degeneration encompasses several morphologic changes in renal epithelial cells associated with loss of viability including tinctorial change, vacuolation, blebbing, cellular sloughing and other alterations including repair. When a particular component(i.e. vacuolation) is much more prominent or the even the exclusive type of change noted, then one of these more specific morphologic diagnoses can be utilized instead of the more general term of degeneration. Degeneration may represent a reversible change or may represent the early manifestations of irreversible necrosis.
Differential diagnosis Necrosis—irreversible cellular change with eventual sloughing and cell loss.
Necrosis, Single Cell (Figures 5 and 6): Proximal and Distal Tubules, Collecting Ducts
Species: rat, mouse
Synonyms: single cell death, apoptosis, apoptotic necrosis
Pathogenesis/cell of origin Renal tubular or ductular epithelium
Diagnostic features Involves individual cells, may be solitary or scattered Increased cytoplasmic eosinophilia Nuclear changes variable, may include peripheral condensation of chromatin, pyknosis, and fragmentation Absence of inflammatory response May progress to more pronounced tubular necrosis with prolonged administration or increased dose of toxicant
Differential diagnoses None, but affected epithelium may be very widely scattered making identification difficult and requiring careful examination of entire microscopic field Should be easily differentiated from early autolysis, which will be more widespread and usually involves karyorhexis rather than pyknosis, and will often have accompanying marked tinctorial changes in cytoplasm.
Comment: Single cell necrosis of the kidney shares features with single cell necrosis in other organs. Although apoptosis predominates, there may be a mixture of both apoptosis and necrosis in the same section. Morphologically, apoptosis is much harder to detect than necrosis because of its rapid progression and the rapid removal of dead cells. Therefore, “single cell necrosis” is the preferred term. Unlike tubule necrosis, there is no release of cellular contents with apoptosis and because of rapid phagocytosis by neighboring cells and shedding into lumina, there is no inflammatory component (Davis and Ryan 1998). Single cell necrosis is characterized by cell shrinkage, eosinophilia, and variably pyknotic nuclei. Surrounding tubular epithelium is generally normal, but there may be an accompanying increased mitotic rate. Apoptosis has a highly regulated role in the kidney associated with maintenance of vital renal functions, so rare apoptotic cells may be found in otherwise normal control kidneys (Davis and Ryan 1998). During the development of the thin loop of Henle, superfluous cells are deleted by apoptosis in the medulla. Apoptosis is an ATP-dependent process and can be initiated by a variety of stimuli including fatty acid synthetase (FAS) ligand, perforin, increased intracellular calcium, and a specific set of genes such as bax and bak. Once triggered, cytochrome C and specific caspases are responsible for downstream effects including cleavage of DNA and proteins, disassembly of cell structural components and eventual cell death (Davis and Ryan 1998; Jurgensmeier et al. 1998). Acute damage to the proximal convoluted tubules has been associated with single cell necrosis of the distal tubules (Davis and Ryan 1998; Bucci et al. 1998). Apoptotic epithelial cells can be positively identified by TUNEL, cleaved caspase 3, or Annexin V immunostaining or by electron microscopy, although this is rarely necessary in routine toxicology studies. Optimized TUNEL staining in kidney appears to require greater dilution and omission of amplification steps as compared to immunostaining protocols in other organs such as liver (Short 1998).
Necrosis (Figures 7 and 117): Proximal and Distal Tubules, Collecting Ducts
Species: rat, mouse
Synonyms: acute tubular necrosis, toxic nephrosis, oncotic necrosis, coagulative necrosis
Pathogenesis/cell of origin renal tubular or ductular epithelium
Diagnostic features Cytoplasmic eosinophilia and pyknosis or karyorrhexis of nuclei Sloughing of affected epithelium into tubular lumina or thinning/attenuation of the epithelial layer lining tubules Cellular casts and amorphous luminal debris are common Necrosis may often be associated with other degenerative lesions including tubular dilation (see below), vacuolation, or crystalluria. Acute inflammatory response may occur Repeated injury may also be associated with regenerative hyperplasia With chronic injury, there is loss of the basal lamina leading to tubular atrophy and/or interstitial fibrosis
Differential diagnoses Postmortem autolysis: uniform dissolution of entire tissue section with no change in organization or depth of cell layers Artifactual damage
Comment: Necrosis may occur as a direct adverse effect of a metabolite or xenobiotic on the tubules or it may occur secondary to ischemia, but the morphologic picture and sequelae are generally similar (Harriman and Schnellmann 2005). Direct effects may be region specific, and the proximal tubules are most commonly affected. Lesions can be multifocal or diffuse. Ischemia is more often zonal or involves a patchy distribution. The medullary thick ascending limb of the loop of Henle is particularly sensitive to anoxia, as is the S3/pars recta segments of the proximal tubules. The pars recta also has high CYP metabolic capability, which can generate reactive metabolites. Beta-lyase and gamma-glutamyl transpeptidase in tubular epithelium can deconjugate metabolites to generate toxicants locally. The cellular mechanistic pathogenesis of renal tubular necrosis is as varied as the wide variety of agents that induce it, but include oxidative stress, effects on ion homeostasis, cytoskeletal injury, lysosomal accumulation and breakdown, mitochondrial injury, phospholipidosis, and inactivation of signaling kinases (Almanzar et al. 1998; Choudhury and Ahmed 2006; Lameire 2005; van de Water, Imamdi, and de Graauw 2005). Stages of necrosis include loss of glycogen and microvilli, vesiculation, nuclear clumping and swelling of endoplasmic reticulum (which are all reversible), followed by loss of nuclear staining, mitochondrial dysfunction and swelling, ion pump dysfunction with cell swelling, and eventually digestion of cell contents. Inflammation is often variable, but atrophy and fibrosis or fibrous replacement are the eventual sequelae, regardless of cause. As with other types of toxic injury, the straight portion of the proximal tubule represents one of the most susceptible sites for necrosis, due to predilection for metabolic activation, transporter-mediated accumulation, and sensitivity to ischemic hypoxia or reperfusion. It is important to define which segments are affected in order to correlate the necrosis with functional changes and biomarkers.
Necrosis, Papillary (Figures 8–11): Medulla and Papilla
Species: rat, mouse (rat more commonly)
Synonyms: pyramidal necrosis, analgesic nephropathy
Pathogenesis/cell of origin Medullary interstitial cells, medullary collecting duct and loop of Henle/thick ascending limb epithelium.
Diagnostic features Earliest stage is loss of structural definition at papilla tip involving mainly interstitial cells Progresses to thrombosis, hemorrhage, loss of microvasculature, loops of Henle, collecting ducts, with replacement by homogeneous eosinophilic matrix Most severe form involves confluent necrosis extending from tip through full papilla Mineralization and/or inflammation may occur in transverse band between necrotic and viable tissue (abscission zone) Sloughing of necrotic papilla may occur at abscission zone, followed by reepithelialization of surface by transitional epithelial cells, which may be followed by transitional cell hyperplasia Secondary changes include pyelonephritis, cortical tubule dilatation, and/or hydronephrosis. Severity can be graded according to extent of papilla involvement; if papilla tip is out of plane of section, early minor lesions may be missed.
Differential diagnoses Postmortem autolysis: uniform dissolution of entire tissue section including all of medulla, especially in inner and outer stripe Pyelonephritis due to ascending infectious causes
Comment: The rat is particularly susceptible to chemically induced papillary necrosis, and there may be marked gender differentiation in susceptibility to the lesion precipitated by certain drugs. Histologic features of papillary necrosis vary with the agent and dose, from mild interstitial edema or mucoid change in the renal papillary matrix to frank necrosis and hemorrhage or to complete loss of the tip of the papilla. Among drugs, renal papillary injury is well recognized following NSAID treatment, and it is the best studied. The pathophysiologic mechanism involves the inhibition of vasodilatory prostaglandins and redistribution of medullary blood flow resulting in ischemia. Concentration of toxicants in the distal medulla and local metabolic activity (e.g., prostaglandin hydroperoxidase activity) may also play roles (Bach and Nguyen 1998). The initial targets are the medullary interstitial cells, followed by degenerative changes in the medullary capillaries, loops of Henle, and collecting ducts (Choudhury and Ahmed 2006; Schnellman 1998). Inflammation can be quite variable from sparse to severe suppurative infiltrates. As pyelonephritis is a possible sequelae of the more severe forms, in some cases it may be difficult to determine whether papillary necrosis or the inflammatory process was the initiating cause.
Infarct (Figures 12 and 13): Cortex
Species: rat, mouse
Synonyms: cortical scar
Pathogenesis/cell of origin Cortical nephron Necrosis of broad, wedge-shaped, well-demarcated areas of the cortex supplied by an arcuate artery Often associated with focal loss of vascular supply due to compromised blood flow, thrombosis, or renal arterial vascular disease resulting from or in association with chronic renal disease Can occur with metastatic tumors or with advanced mononuclear cell leukemia May be associated with administration of xenobiotic agents including acute nephrotoxicants and especially drugs affecting renal vasculature
Diagnostic features Recent lesions contain central, peripheral, and marginal zones; central zone consists of a wedge-shaped area of necrosis, while peripheral rim may have neutrophilic or monocytic infiltration and tubular degeneration, with congestion in the marginal zone With chronicity, there is variable scarring and replacement of interstitium with mature fibrosis, marked tubular atrophy, and tubular collapse with or without inflammation or dystrophic mineralization Overlying capsular surface is often depressed
Differential diagnoses Interstitial fibrosis from other renal disease; diffuse interstitial disease rather than wedge-shaped area as is not related to compromised blood flow
Hemorrhage: Cortex, Medulla
Species: rat, mouse
Synonyms: none
Pathogenesis/cell of origin Hemorrhage can occur from inflammation, tubular necrosis, vascular injury or from the presence of calculi or tumors.
Diagnostic features Blood-tinged urine or hemoglobin in urine The presence of extravasated red blood cells may occur, but this is less common with renal hemorrhage than lower urinary tract hemorrhage unless associated with neoplasia Often results in bright red hemoglobin casts within tubules of a single or cluster of nephron segments
Differential diagnosis Congestion
Comment: Hemorrhage often accompanies acute injury and can occur in the kidney as a primary lesion associated with nephrotoxicants without significant degeneration or necrosis. With subacute injury it may be accompanied by hemosiderin pigment within tubular cytoplasm or within interstitial macrophages. The presence of luminal hemorrhage implies either damage to the interstitial vascular supply and epithelial basal lamina or damage to the glomeruli as intact erythrocytes do not pass functioning glomerular filtration barriers.
Vacuolation (Figures 14–16): Proximal and Distal Tubules, Collecting Ducts
Species: rat, mouse
Synonyms: vacuolization, vacuolar degeneration,
Pathogenesis/cell of origin renal tubular or ductular epithelium
Diagnostic features Intracellular accumulation of fluid, lipid, or other material within epithelium resulting in a swollen, pale, or granular cytoplasmic appearance Discrete clear or translucent spaces of variable size Macrovesicular (large spaces) or microvesicular (many small spaces) forms May occur normally in outer cortical tubules, especially in CD-1 mice
Differential diagnoses Postmortem autolysis: cell swelling is diffuse and presents with granular or lacy appearing cytoplasm. Vacuoles often lack uniformity of size or discrete outlines within tubules. May be accompanied by rarefaction of nuclei and/or dissolution of entire tissue section. Accumulation, glycogen: clearing of cell cytoplasm and/or frothy appearance associated with hyperglycemia
Comment: Vacuolation may precede degeneration and necrosis but may also indicate a reversible change or occur in normal animals (Johnson et al. 1998). As a diagnostic term, it is best reserved for cases where it is the primary or sole degenerative process present. Accumulation of fat within tubules occurs as a consequence of disrupted cellular machinery due to toxic insult. Special stains such as oil red O, Sudan black, or osmium can be used to visualize lipid within cytoplasm, and other techniques such as electron microscopy or immunohistochemistry may be utilized to characterize other material within vacuoles. Dextran vehicles and contrast agents have also been associated with cytoplasmic vacuolar accumulations. Phospholipidosis of the kidney appears microscopically with routine HE staining as vacuolation of the epithelium. Ultrastructurally, phospholipidosis represents lysosomal accumulation of membranous whorls of electron dense material.
Accumulation, Glycogen: Proximal and Distal Tubules
Species: rat, mouse
Synonyms: clear cell, tubule; osmotic nephrosis; hydropic change
Pathogenesis/cell of origin renal tubular or ductular epithelium
Diagnostic features Intracellular accumulation of fluid and glycogen within epithelium resulting in a swollen, pale, or granular cytoplasmic appearance Discrete clear or translucent spaces of variable size May occur following administration of osmotically active compounds in “osmotic nephrosis”
Differential diagnosis Vacuolation: circumscribed (membrane bound) pale lucent areas
Comment: Accumulation of glycogen can occur as a consequence of distal tubular absorption of sugars or other hypertonic substances with diabetic nephropathy (Monserrat and Chandler 1975; Ahn et al. 1992), loss of lysosomal glycolytic activity (Bucci et al. 1998), or with osmotic nephrosis related to the intravenous administration of sugars such as dextrose or mannose. This is technically not true vacuolation, as the changes are largely cytoplasmic rather than lysosomal. Affected cells appear swollen and often have a clear or frothy appearance. This has historically been assigned a variety of diagnostic terms such as “vacuolation, microvesicular, tubule,” “cytoplasmic rarefaction,” “clear cell, tubule,” or “glycogenosis, tubular.” Affected cells stain positively with Periodic acid Schiff (PAS) stain (diastase-labile) or Best’s Carmin stains and ultrastructurally demonstrate abundant cytoplasmic glycogen particles by electron microscopy (Frank and Gray 1976). While descriptive terms such as vacuolation, cytoplasmic alteration or clear cells may be useful in describing these changes, in cases where glycogen has been definitively identified in tissues, it is appropriate to use the term “accumulation, glycogen” instead. This may be an important distinction as these changes associated with diabetic nephropathy have been considered a preneoplastic lesion in the kidney with the potential for progression to renal cell carcinoma or other renal tumors (Ahn et al. 1992; Dombrowski et al. 2007).
Accumulation, Hyaline Droplets (Figures 17 and 18): Proximal and Distal Tubules
Species: rat, mouse
Synonyms: hyaline droplet accumulation, eosinophilic droplets
Pathogenesis/cell of origin Renal tubular epithelium Usually represents cytoplasmic accumulation of LMW proteins in the secondary lysosomes of the proximal tubules Potentially the accumulating LMW proteins can be of various origin, but are known to include Alpha2u-globulin nephropathy and lysozyme (generated by generalized histiocytic sarcoma)
Diagnostic features Varying degrees of increase in eosinophilic, cytoplasmic droplets in proximal convoluted tubules, predominantly the second segment of proximal tubule In very severe cases (can occur with generalized histiocytic sarcoma) all parts of the proximal tubule can be involved Droplets tend to be of rounded form and of varying size, but can also be polyangular (in Alpha2u-globulin nephropathy) in male rats, see Alpha2u-globulin nephropathy May be associated with granular casts located at the junction of the outer and inner stripes of outer medulla
Differential diagnoses In the male rat only, hyaline droplet accumulation needs to be distinguished from the normal pattern of droplets, representing secondary lysosomes, in the S2 segment of proximal convoluted tubules Hemoglobin, myoglobin accumulation, or intratubular hemorrhage (may not be localized within epithelium and often associated with casts) Alpha2u-globulin nephropathy is the preferred term when the protein has been identified as such by immunohistochemistry, special stains, and/or when droplets are highly characteristic for that condition
Comment: Hyaline droplets represent LMW protein accumulation within lysosomes due to disturbance of the normal balance of tubular reabsorption and hydrolysis as a result of either increased filtered protein loads or decreased catabolism (Maak et al. 1979, Alden 1986). Rats and mice with histiocytic sarcoma demonstrate hyaline droplets of variable size which contain lysozyme and this can be demonstrated by immunohistochemistry (Hard and Snowden 1991). Droplets may represent xenobiotic: protein complexes that do not consist of either of the above proteins. Although alpha2u-globulin nephropathy is one type of hyaline droplet nephropathy, it is the preferred term in many or most cases in the rat where typical features are presented and particularly if special stains or immunohistochemistry has been performed for verification. Hyaline droplet nephropathy is the preferred diagnostic term when atypical features are noted, including variable- or large- sized droplets, high incidence in females, abnormal location within the nephron, or when special or immunohistochemical stains indicate alpha2u-globulin is not a primary component. Droplets and granular structures have been noted in the proximal tubules of rodents, dogs, and monkeys given antisense oligonucleotide therapeutics, but these tend to be more basophilic and irregularly shaped and should be diagnosed separately as “basophilic granules” even when a portion of these appear somewhat eosinophilic with HE (Marquis and Grindel 2000). They represent accumulation of degradation-resistant polyanionic oligonucleotide molecules within lysosomes rather than the proteinic composition of other types of hyaline droplets.
Alpha2u-Globulin Nephropathy (Figures 19–21): Proximal Tubules
Species: rat
Synonyms: eosinophilic droplets, alpha-2u-globulin nephropathy
Pathogenesis/cell of origin Chemically induced by noncovalent binding of certain chemicals, or their metabolites, to the circulating low-molecular protein, alpha2u-globulin, which is synthesized in the liver of male rats only Complexing of the chemical with alpha2u-globulin increases the long half-life of the protein after uptake into the S2 proximal tubules, and lysosomal accumulation results
Diagnostic features Varying degrees of increase in eosinophilic, cytoplasmic droplets in the S2 segment of proximal tubules in the cortex Exfoliation of sporadic cells into the tubule lumen; increase in mitotic figures involving affected portions of proximal tubules; may be some associated tubule basophilia in more severe cases Usually associated with formation of granular casts at the junction of outer and inner stripes of outer medulla, representing accumulation of cell debris where the S3 segment of proximal tubule narrows into the descending limb of Henle Accompanied by an exacerbation of spontaneous CPN
Differential diagnoses Accumulation associated with alpha2u-globulin nephropathy needs to be distinguished from the normal pattern of droplets, representing secondary lysosomes, in the S2 segment of proximal convoluted tubules Hyaline droplet accumulation: this term is preferred when there is question of whether the accumulated material making up the droplets is alpha2u-globulin or not (e.g., when there have been no special stains or immunostains performed and there are other mitigating circumstances such as high incidence in females, or marked irregularity in the size or shape of droplets such as in lysozyme associated granules with histiosarcoma) Increase in droplet number, angular forms, and disruption of the normal pattern of droplets characterize α2u-globulin nephropathy (Hard 2008)
Comment: The protein responsible for this condition was originally labeled Alpha-2u-globulin according to electrophoretic nomenclature with the “u” referring to “urinary” (Roy and Neuhaus 1966; Neuhaus and Lerseth 1979;). It has alternatively and erroneously been referred to in literature as Alpha-2 microglobulin or Alpha-2 mu-globulin. Alpha2u-globulin is androgen regulated and synthesized in copious amounts in the liver of male rats only. Thus, alpha2u-globulin nephropathy is a sex- and species- specific entity (Montgomery and Seely 1990; Short et al. 1989; Swenberg et al. 1989). Many xenobiotics bind to alpha2u-globulin and decrease effectiveness of lysosomal degradation (Alden et al. 1984). Visualization of these droplets can be enhanced by staining with Mallory Heidenhain stain, Martius scarlet blue, chromotrope-analine-blue stains, immunohistochemistry (De Rijk et al. 2003), or by examining H&E stained kidney under ultraviolet illumination (Hard 2008). While it should be unnecessary to routinely use special stains on all cases of hyaline droplets to confirm the diagnosis of alpha2u-globulin nephropathy in rats, this may be helpful where there are unusual presentations such as incidence in females or abnormal histologic features of the droplets. Because of the sustained proliferative effects secondary to overload-associated cellular loss, there is an increased incidence of renal tumors often accompanying chronic hyaline droplet nephropathy or alpha2u-globulin nephropathy in rats, and the increased cell turnover may potentiate rat CPN (Alden et al. 1984; Mattie et al. 1991).
Dilation, Tubule (Figure 22 and 23): Proximal and Distal Tubules, Collecting Ducts
Species: rat, mouse
Synonyms: simple tubular dilatation, tubular dilatation
Pathogenesis/cell of origin: renal tubular and ductular epithelium
Diagnostic features Tubules with mild-to-moderate expansion of lumina lined by relatively normal or minimally flattened epithelium Often associated with tubular necrosis Can contain luminal casts, cellular debris, or suppurative inflammation Radial or zonal distribution Can occur as a result of obstruction of the nephron by crystalluria, protein casts or cellular debris, and obstruction of the lower urinary tract Term should not be used with polycystic disease
Differential diagnoses Polycystic kidney disease or other congenital cyst
Comment: Tubular dilation most often accompanies other forms of renal damage (e.g., necrosis or degeneration), but tubular dilation without accompanying degenerative changes can occur following administration of a variety of agents including modified starches, lithium, and angiotensin-converting enzyme (ACE) inhibitors (Christensen and Ottensen 1986; Schetz et al. 2005). Interstitial inflammation or fibrosis is quite variable depending on the cause and the individual process. The pathogenesis of tubule dilatation has been linked to tubular stasis, excessive renal hemodynamic changes, or electrolyte and water loss (Gardner 1988; Lameire 2005). Certain xenobiotics, notably corticosteroids, may induce tubular dilation in young animals without evidence of other tubular damage, and agents which interfere with nephron development during the neonatal period may also cause tubular dilation and/or cyst formation (Perey, Herdman, and Good 1967). Tubular dilation due to luminal obstruction from crystalluria is associated with a number of poorly soluble drugs such as sulfonamide or quinolone anibiotics and purine analogues (Schetz et al. 2005). These substances may precipitate in the nephron causing a backup of filtrate and large increases in pressure that result in dilation of multiple segments. Crystals may be present within tubules or collecting ducts distal to the lesion or crystals or secondary urothelial hyperplasia may be noted in the pelvis to aid in identifying the cause. However crystals often are removed in processing.
Cyst (Figures 24 and 117): Cortex and Medulla
Species: rat, mouse
Synonyms: cystic tubular dilation, tubular ectasia, cystic tubule
Pathogenesis/cell of origin: Can occur in young rats with corticosteroid treatment as a result of dilated collecting ducts which fail to establish continuity with developing nephrons
Diagnostic features Tubules with markedly expanded lumina Lined by flattened single cell layer of epithelium Variable lumen contents Thin fibrous capsule is occasionally present Presence of peritubule inflammation occasionally
Differential diagnoses Dilated arcuate vein (erythrocytes may not be visible but veins always lined by endothelium) Dilatation tubule: multiple tubules are affected and tend to be much smaller lumina
Comment: Cystic tubules are a more severe manifestation of tubule dilation and are found commonly in late stages of CPNs of rats and mice. Cysts may also represent solitary congenital spaces or be related to congenitally acquired polycystic kidney disease in rats and mice (Perey, Herdman, and Good 1967; Smith et al. 2006). A spectrum of polycystic lesions in the kidney and hepatic biliary tree are noted in Caroli’s disease, which is an autosomal recessive condition in polycystic kidney disease (PCK) rats and found spontaneously in other strains (Nakanuma et al. 2010).
Nephropathy, Retrograde (Figures 25–27): Proximal and Distal Tubules, Medullary Ducts
Species: rat, mouse
Synonyms: reflux nephropathy, ascending pyelonephritis
Pathogenesis/cell of origin Ascending change from papilla to cortex caused by effects in the lower urinary tract, including increased urine reflux, partial or transient obstruction, increased pressure, or ascending infection (Vivaldi et al. 1959; Heptinstall 1964; Heptinstall 1965)
Diagnostic features Constellation of tubule changes extending from papilla to cortex Cortical lesions appear as irregular foci or patches of tubule basophilia often arrayed in a linear pattern following the course of the nephron; coupled with distal tubule dilation; tracts of basophilic and dilated collecting ducts traversing the outer and inner medulla, mainly at the periphery of the pyramid Cells/nuclei of affected collecting ducts are crowded (representing simple hyperplasia) and usually with increased mitoses; cortical basophilic lesions and medullary collecting duct tracts are connected Unless due to ascending infection, inflammatory cells are not prominent; can resolve into chronic scars which traverse the outer kidney zones into the inner medulla where collecting ducts remain dilated and hyperplastic (Mackenzie and Asscher 1986)
Differential diagnoses Pyelonephritis of hematogenous origin or septic thrombi from indwelling cannulas: similar cortical lesions but usually with polymorph neutrophils Obstructive nephropathy: crystal deposits in tubule lumen; granulomatous inflammation characterized by mononuclear cells, and sometimes epithelioid and/or multinucleate giant cells CPN: Relatively well-defined cortical foci of basophilic proximal tubules with conspicuously thickened basement membrane; associated with hyaline protein casts in the medulla Chronic infarct: Usually taper axially from the subcapsular cortex; do not connect with tracts of dilated collecting ducts extending through the medulla
Comment: In rats (and less so in mice), retrograde nephropathy is the renal lesion produced by dietary exposure to melamine (Hard et al. 2009). The pathogenesis and morphology of retrograde nephropathy differs from that of obstructive nephropathy. It appears to be related to reflux phenomenon associated with transient or partial obstruction or precipitation of material in the urinary tract resulting in irritation and perturbation along the nephron with degenerative effects such as tubular basophilia predominating along with dilation of tubules and ducts in the distal nephron.
In contrast, obstructive nephropathy is always a result of blockage of the outflow of the urinary filtrate (usually from crystals) and as such there is dilation and/or necrosis along the entire tract of adjacent nephrons without retrograde urine flow or reflux. (See also obstruction, urethra, and obstructive uropathy)
Basophilia, Tubule (Figures 28-30): Proximal and Distal Tubules, Collecting Ducts
Species: rat, mouse
Synonyms: Tinctorial change
Pathogenesis/cell of origin renal tubular and ductular epithelium
Diagnostic features Tubular epithelial cells with basophilic cytoplasm, but otherwise normal profiles May be slightly enlarged or plump (hypertrophy) Increased nuclear:cytoplasmic ratio with increased mitoses can occur as an early sign and only manifestation of CPN in rat in which there is associated thickening of the basement membrane
Differential diagnoses Staining artifact (which affects large numbers of adjacent tubules rather than being multifocal) Simple tubule hyperplasia—while this change results in basophilia of tubules, there is also focal crowding and an increase in the number of tubule lining cells Regeneration, tubule—has basophilia as a common feature following injury, but cells are very flat or low cuboidal with a rudimentary brush border and a high rate of mitosis and generally no thickened basement membrane; The terms regeneration, tubular hyperplasia, and tubular basophilia should not be used interchangeably as tubular basophilia can occur without reparative processes CPN is the preferred term when there are additional features associated with tubular basophilia such as nuclear crowding and thickened basement membranes, especially in subchronic and chronic studies in rats
Comment: Tubular basophilia is one of the most frequently encountered manifestations of induced nephron injury, particularly in repeat dose toxicity studies. It may be a sequel to degenerative conditions or represent excessive cellular turnover and presents as a tinctorial change in the epithelial cytoplasm (Gopinath, Prentice, and Lewis 1987). In young growing rats, a few basophilic cortical tubules are a normal feature. Tubular basophilia can represent tubular regeneration, but may also indicate early atrophy or persistent low-grade toxic injury. It is commonly associated with CPN coinciding with thickening of the basement membrane and occurs as a background change in an increasing percentage of rats and mice with age. In the absence of any other evidence of regeneration or degeneration within a kidney, tubular basophilia should be used as a preferred term, but it should not be used as an additional diagnosis as part of a more comprehensive degenerative or regenerative renal process. Pathophysiologic changes associated with the morphologic alterations include increases in endoplasmic reticulum, enlarged nuclei, and depending on the etiology, myeloid bodies or inclusions within lysosomes (Lameire 2005; Peter, Burek, and Van Zwieten 1986). Basophilic tubules in rats induced after chronic nitrosamine treatment stained positively forglyceraldehyde -3- phosphate dehydrogenase (GAPDH) and glucose-6- phosphate dehydrogenase (G6PDH) while demonstrating a reduction of staining for other enzyme activity when compared with controls and surrounding proximal or distal tubules (Tsuda et al., 1986).
Chronic Progressive Nephropathy (CPN) (Figures 31–34): Proximal and Distal Tubules
Species: rat, mouse
Synonyms: chronic progressive nephrosis, chronic nephritis, spontaneous nephrosis, chronic nephrosis, progressive glomerulonephrosis, glomerulonephrosclerosis, nephritis, glomerulonephritis, dietary nephritis, chronic progressive glomerulonephropathy, glomerulosclerosis, old rat nephropathy (Barthold 1998)
Pathogenesis/cell of origin
Spontaneous in origin and of unknown etiology
In the rat it is influenced by various physiological factors such as caloric intake, protein content of diet, and male hormones; however, recent studies have confirmed an effect of dietary protein content on severity of CPN but not incidence (Travlos et al. 2011)
Incidence and/or severity can be exacerbated by certain chemicals
Earliest lesion detectable in young adults is a basophilic tubule or evidence of regeneration in outer kidney, but soon thereafter basophilia is accompanied by crowded nuclei and/or thickened basement membrane; hyaline casts often first observed in outer medulla and variable mononuclear infiltrates
Will often progress into adjacent areas of parenchyma and can ultimately result in end-stage kidney Onset varies with strain
CPN is the preferred term for this condition, particularly in subchronic or chronic studies, when characteristic morphologic features such as tubular basophilia, nuclear crowding, and thickened basement membranes are all present; tubular basophilia is an appropriate diagnostic term to be used in studies if it is the only morphologic feature present, as in very early cases in young animals
Diagnostic features
Rat Foci or areas of basophilic proximal tubules, with or without simple tubule hyperplasia, and associated with conspicuously thickened basement membranes and nuclear crowding Prominent hyaline casts in medulla may be present, except in earliest lesions; with progression, tubule atrophy, tubule dilation, focal glomerular sclerosis, and glomerular atrophy; mononuclear cell infiltration mainly as perivascular clusters Transitional cell hyperplasia of renal pelvis lining occurs in very advanced disease All kidney parenchyma is involved in end-stage kidney and hyaline casts in dilated tubules are responsible for the roughened kidney surface seen grossly; in advanced disease, particularly if chemically exacerbated, occasional tubules may have a florid appearance with complex lining epithelium, but also with conspicuously thickened basement membranes Interstitial fibrosis can be observed in advanced cases
Mouse Not as well characterized as in the rat; a similar constellation of changes can occur, but often associated with glomerular dilation changes in the amino acid composition of the basal lamina have not been demonstrated in the mouse, so the pathogenesis is thought to be different
Differential diagnoses Atypical tubule hyperplasia: Usually solid, basophilic tubule profiles with prominent encirclement of connective tissue cells around the periphery; usually basement membrane is not conspicuously thickened Simple tubule hyperplasia and/or regeneration: Either may be a component of CPN, but these diagnoses should be reserved for lesions lacking other characteristic features such as thickened basement membranes and casts Pyelonephritis: Patchy cortical basophilia; may be associated with polymorph neutrophils Obstructive nephropathy: Crystal deposits in tubule lumen; granulomatous inflammation characterized by mononuclear cells, and sometimes epithelioid and/or multinucleate giant cells.
Comment: There are a number of comprehensive reviews of CPN (Gray 1977; Gray, Van Zwieten, and Hollander 1982; Barthold 1979; Goldstein, Tarloff, and Hook 1988; Hard and Khan 2004). Advanced CPN in the rat is associated with parathyroid gland hyperplasia (due to lack of activation of Vitamin D by the proximal tubules), and widespread metastatic mineralization. Because rat CPN is both regenerative and degenerative, with a high rate of cell turnover, advanced disease may be a risk factor for renal tumor development. Florid tubule profiles in advanced disease are not preneoplastic and need to be discriminated from atypical tubule hyperplasia (Hard and Seely 2006). There is often some confusion regarding the diagnosis of early lesions of CPN in young rats less than 12 weeks of age. Where possible, to accurately and appropriately diagnose even the early stages of CPN, the spectrum of changes including basophilic tubules, with conspicuously thickened basement membranes, crowded nuclei and/or the presence of hyaline cast(s) in the outer stripe of the outer medulla should be identifiable as confirmatory diagnostic signals. In young animals, where only a single attribute of the condition is present and where uncertainty exists as to whether changes represent early CPN or not, the component diagnoses (such as tubule basophilia alone) can be used at the discretion of the pathologist and with the understanding that these may in fact represent the earliest CPN lesion. CPN can be exacerbated by many chemicals that result in increased incidence and severity in chronic toxicity studies. An occasional chemical produces a response that has some features of CPN, but produces a renal tubular lesion that is far more widely distributed than occurs with CPN exacerbation. These can be differentiated based on specific features such as tubules that do not have the conspicuous basement membrane thickening, and hyaline casts that tend to be present in the cortex as well as more distally. Alterations in the amino acid composition, hydroxylation, and glycosylation of basal lamina surrounding tubules have been demonstrated and are thought to be important in the pathogenesis in the rat (Abrass 2000).
Casts (Figures 35 and 36): Proximal Tubules, Distal Tubules, Loop of Henle/thick ascending limb, Medullary Collecting Ducts
Species: rat, mouse
Synonyms: tubular proteinosis is true synonym; hyaline, granular, protein, myoglobin or hemoglobin casts are subtypes,
Pathogenesis/cell of origin tubular lumina
Diagnostic features uniform inclusion occupying a length or cross section of tubule The most common types of casts are hyaline or granular, but the preferred term “cast” can be used to describe all types Cellular casts can also occur when sloughed epithelial cells fill lumina and lose their cellular outlines
Cast, Hyaline
Diagnostic features Homogeneous eosinophilic contents filling tubule lumen Typically of protein composition
Cast, Granular
Diagnostic features Nonhomogeneous contents of granular particulate matter in tubule lumen Eosinophilic or chromophobic Typically consists of cell breakdown products and debris Typically occurs in Alpha2u-globulin nephropathy at the junction of the outer and inner stripes of the outer medulla
Differential diagnoses Tubular crystalluria Postmortem artifact Tamm-Horsfall mucoprotein (uromodulin) which is a normal constituent of filtrate produced in the thick ascending limb and distal tubules
Comment: Tubules filled with eosinophilic (hyaline) proteinaceous casts generally indicate that there is increased glomerular permeability in that nephron and are often associated with glomerular damage. However, large amounts of protein within tubules can induce damage to the surrounding tubular epithelium as well. Casts are a common feature accompanying chronic nephropathies in rats and mice and their number increases with advancing age (Alden 1986; Peter, Burek, and Van Zwieten 1986). Granular casts are more indicative of primary tubular injury, and they are more often associated with necrotic luminal cellular debris and potentially a mild inflammatory infiltrate. Casts that are comprised largely of hemoglobin occur in renal tubules following acute hemolytic crises due to erythrocyte destruction from causes such as copper poisoning, transfusion reactions, or drugs inducing severe hemolytic anemias (Ericsson, Mostofi, and Lundgren 1969). Similarly, casts comprised of myoglobin are noted in tubules secondary to marked muscular damage (Riggs, Schochet, and Parmar 1996). The latter two types of casts are often a darker red color and can be accompanied by intact erythrocytes within lumina.
Adipose Aggregate, Interstitial: Cortical and Medullary Interstitium
Species: rat, mouse
Synonyms: lipomatosis, lipomatous metaplasia
Pathogenesis/cell of origin Unknown: The interstitial cytoplasm normally contains variable numbers of lipid droplets and small numbers of mature adipocytes. These cells may proliferate or their increase may represent differentiation from type I interstitial cells of the renal medulla or other pluripotent mesenchymal stem cell
Diagnostic features Occurs in the interstitium of the cortex and especially medulla An unencapsulated focus of well-differentiated adipocytes which may replace other cells and expand the interstitium such that neighboring tubules may be mildly compressed Typically considered a spontaneous background lesion, but occasionally noted with increased frequency after xenobiotic treatment
Differential diagnoses Lipoma: expansile lesion which continues to grow over time and generally affects the architecture of surrounding cells; often well circumscribed; generally much larger than interstitial adipose aggregates Liposarcoma: invasive, less-differentiated vacuolated tumor cells
Comment: It is unclear what the cell of origin is for these lipomatous nodules within the interstitium. They may arise from normal resident adipocytes but they may also represent a hyperplastic or metaplastic response of type I medullary cells or adipocytic differentiation of an undifferentiated stem cell. They can be noted spontaneously, especially in obese rodent models such as the Zucker Diabetic Fat rat, or with increased frequency following drug treatment such as with some compounds affecting fat metabolism. The term lipomatous metaplasia has been applied to this lesion, and like cellular metaplasia in other tissues, this represents the reversible subsititution of one tissue for another. However, unlike true metaplasia, there is little evidence that these lesions are pre-neoplastic and undergo neoplastic transformation to liposarcomas, and their toxicologic significance is negligible as they generally do not adversely affect renal function.
Crystals (Figure 37): Proximal and Distal Tubules, Collecting Ducts
Species: rat, mouse
Synonyms: crystalluria, urolithiasis, tubular calculi
Pathogenesis/cell of origin tubular lumina
Diagnostic features Occurs in lumina of tubules (can dissolve out in fixation/processing; fixed frozen or fresh frozen sections may be required to demonstrate the crystals). Usually located in cortex or outer medulla Crystals sometimes made more visible by polarized light Shape of crystals may be engulfed by multinucleated phagocytic cells May lead to obstructive nephropathy
Differential diagnoses Casts: generally stain eosinophilic and are not birefringent Mineralization (nephrolithiasis due to calcium phosphate which tends to stain blue rather than clear or brown)
Comment: Administered compounds or their metabolites may precipitate in the urine filtrate, particularly if they are of low solubility and/or in high plasma concentration with a high percentage of renal clearance (Yarlagadda and Perazella 2008). Because of the concentration of urine and hypertonicity in the distal tubular segments, especially in the rat, crystalluria is more common in this region (Hagiwara et al. 1992). Examples of crystal-inducing drugs include quinolone or sulfonamide antibiotics and purine analogues such as acyclovir. Manipulation of the diet or administration of agents which alter urinary pH (e.g., carbonic anhydrase inhibitors) or alter intravascular volume depletion will increase the incidence of tubular crystalluria. Crystals may also form from complexes with minerals, as happens with the calcium oxalate crystals noted with ethylene glycol toxicosis (Robertson 2004; Li and McMartin 2009). A metabolite of ethylene glycol is oxidized by alcohol dehydrogenase to oxalic acid, which sequesters calcium within the renal tubules leading to its precipitation and eventually luminal obstruction (Hess, Bartels, and Pottenger 2004). Crystals from any cause may incite degeneration and necrosis in the adjacent tubular epithelium, or crystals may be associated with obstructive nephropathy due to blockage of urine flow. Tubular crystalluria may also accompany the presence of calculi within the renal pelvis as the etiopathogenesis of the two lesions is similar. Since many crystals are birefringent, the use of polarized light during microscopic examination often aids in visualization and proper diagnosis.
Nephropathy, Obstructive: Proximal Tubules, Distal Tubules, Thick Ascending Limb
Species: rat, mouse
Synonyms: crystal nephropathy
Pathogenesis/cell of origin Renal damage caused by precipitation of chemical as crystals in the tubule lumen Inflammatory process resulting from tubule blockage by the crystals (Chevalier 2006) In male mice can occur from blockage of urinary outflow in the bladder or urethra from the formation of a proteinaceous plug; can be associated with retained spermatozoa and evidence of paraphimosis or balanoposthitis
Diagnostic features Crystal deposits in tubule lumen; crystals may be birefringent; tubule dilatation proximal to the blockage Granulomatous inflammation characterized by interstitial infiltration of mononuclear inflammatory cells Epithelioid cells and multinucleated giant cells of Langhans type sometimes present; mild fibrosis; sometimes neutrophil infiltration Crystals sometimes observed within giant cells Presence of proteinaceous plug in male mice Blockage of the ureters, i.e., intra-abdominal tumors
Differential diagnoses Retrograde nephropathy: tubular basophilia and lesions in both cortex and medulla Pyelonephritis of hematogenous origin: neutrophilic inflammation and tubular dilation is rare and regional
Mineralization (Figures 1, 38, and 39): Medullary Collecting Ducts; Corticomedullary Junction; Proximal or Distal Tubules, Renal Pelvis
Species: rat, mouse
Synonyms: calcification, nephrocalcinosis, multilamellar bodies
Pathogenesis/cell of origin Can occur either as dystrophic calcification specifically in the renal tubules and collecting ducts or as metastatic calcification as a result of systemic calcium/phosphorus imbalance Both types are common and occur spontaneously in laboratory animals or as a consequence of drug treatment Occur with dietary imbalance of calcium/phosphorus ratio, particularly in female rats; this can include calcium or Vitamin D administration, oxalates, parathyroid hormone-like hormones compounds or with drugs which modify urinary pH, as well as many other types of drugs and agents (Ritskes-Hoitinga and Beynen 1992) Typically composed of calcium (and much less commonly magnesium) salts, phosphorus, and glycoprotein One common spontaneous form of mineralization is thought to be derived from shedding of microvilli and microvesicles from S1 proximal tubules and accumulation in the outer stripe of the medulla where this debris subsequently undergoes mineralization (Nguyen and Woodard 1980) May be visible macroscopically as white stippling on cut surface or microscopically as densely basophilic granular deposits In rats, there can be a much higher prevalence of mineralization in the outer stripe of the outer medulla in females due to a dietary imbalance of calcium:phosphorus ratio and incidence and severity increase with age (Clapp, Wade, and Samuels 1982; Ritskes-Hoitinga and Beynen 1992)
Mineralization, tubule
Diagnostic features Deposits replace tubular cytoplasm as a sequel to tubular degeneration
Mineralization, intraluminal
Diagnostic features Most commonly found along the corticomedullary junction and of mixed chemical composition, but can occur in cortex, medulla, or papilla Degenerative and/or necrotic changes can occur in adjacent tubules In papilla, often occurs in prebend segment of loop of Henle in a linear formation, and this form can be a feature of alpha2u-globulin nephropathy in chronic studies.
Mineralization, basement membrane Focal or linear basophilic deposits along basement membranes within interstitium Preferentially involves interstitium around proximal convoluted tubules and accompanying vasculature, as well as glomerular tufts
Mineralization, interstitial
Granular or rounded basophilic deposits within interstitium most commonly found in cortex and often appear along basal lamina.
Differential diagnoses Intratubular bacterial colonies: stain positively with gram stain and negative for mineral
Special stains Mineralization can be demonstrated with Alizarin Red S or Von Kossa’s stains
Comment: Mineralization is encountered frequently in rodent toxicity studies. The diagnosis of mineralization without a regional qualifier may be sufficient in many or most studies, but in some cases use of the subterms interstitial, tubule, basement membrane, and so on, may be important to denote. Many xenobiotic agents can affect calcium, phosphorus, or PTH regulation (Matsuzaki et al. 1997). Mineralization can also be a profound and early secondary consequence of renal failure. A deficit of calcitriol and an abnormality in the calcium sensor receptor may be the important factors initially, while later in advanced renal failure, hyperphosphatemia becomes an additional important pathogenic factor. More commonly, renal mineralization is found as a spontaneous lesion along the corticomedullary junction in rats as a background finding and is of no clinical consequence. Female rats are especially sensitive, while mice tend to be more resistant. A syndrome of mineralization is also encountered frequently in the renal pelvis of rodents. The mineralized lesions may be much more severe with agents affecting calcium regulation, or where a compound affects urinary pH such as with carbonic anhydrase inhibitors (Ritskes-Hoitinga and Beynen 1992; Nicoletta and Schwartz 2004).
Metaplasia, Osseous: Interstitium
Species: rat
Synonyms: ectopic bone formation
Pathogenesis/cell of origin Represents osseous tissue in the interstitium which displaces the tubules Pathogenesis is unrelated to mineralization May occur with chronic corticosteroid therapy
Diagnostic features Generally focal lesions that are present in the cortical interstitium Composed of osteoid islands and/or well-differentiated bone, with irregular boundaries Bone marrow may be present in the larger foci Rarely associated with secondary inflammation and is not considered preneoplastic
Differential diagnosis Mineralization: composed of basophilic amorphous crystalline deposits rather than osteoid and does not contain nuclei (osteoblasts)
Comment: Osseous metaplasia has most commonly been noted in the kidneys of Fischer F344 rats (Montgomery and Seely 1990). It is uncommon in other strains or species, but can occur with chronic corticosteroid therapy in any organ, especially the skin (Frazier et al. 1998), and has occasionally been noted in rodents with the chronic administration of some of the bone morphogenic proteins (BMPs).
Accumulation, Pigment (Figures 31, 40, and 41): Proximal and Distal Tubules, Collecting Ducts
Species: rat, mouse
Synonyms: lipofuscinosis, bilirubinuric nephropathy
Pathogenesis/cell of origin Represents intracellular deposits of lipofuscin, bilirubin, or resorbed blood iron pigments Several drugs potentiate the development of lipofuscin droplets within proximal epithelial cells or are themselves pigmented and result in accumulation of colored material Bilirubin can accumulate in renal tubules if systemic blood levels increase as occurs with liver disease or inhibition/dysfunction of biliary conjugating enzymes (UGT1A1) Hemosiderin pigment occurs in the cytoplasm of proximal tubules with immune-mediated or drug-induced hemolytic anemias, as well as with anemias associated with leukemia
Diagnostic features Yellow to brown stippling or granules in cytoplasm Lipofuscin is found in the kidneys of most laboratory rodents, especially rats, and is most often localized to the proximal tubules
Differential diagnosis The various types of pigments need to be distinguished by special stains
Special stains Lipofuscin is weakly PAS positive and can be identified using the AFIP Schmorl’s stain method Hemosiderin is positive for Perl’s stain or Prussian blue Bilirubin is positive using Hall’s method
Comment: The functional significance of pigment accumulation varies widely from lipofuscinosis (of limited or no functional significance) to hemoglobinuric nephropathy (hemolytic anemia–induced hemoglobin overload) which can result in severe renal dysfunction (Ichikawa and Hagiwara 1990; Ivy et al. 1990; Ikeda et al. 1985). Differentiation of the pigment into its components is therefore important.
Inclusion Bodies: Proximal and Distal Tubules, Collecting Ducts
Species: rat, mouse
Synonyms: none
Pathogenesis/cell of origin May occur in the nucleus, cytoplasm, and mitochondria of proximal convoluted tubules with lead toxicity (Navarro-Moreno et al. 2009) In the cytoplasm, may represent myeloid bodies (such as with gentamicin or other aminoglycosides), protein precipitates (as with bisphosphonates), giant mitochondria, or proliferating endoplasmic reticulum (Pfister et al. 2005) Cytoplasmic inclusions consisting of iron deposits have been noted with compounds affecting iron metabolism or transport, such as with chronic uranium nephrotoxicity (Donnadieu-Claraz et al. 2007) Viral inclusion bodies in renal tubules may occur as a result of accumulation of viral matrix proteins within the nucleus or cytoplasm such as occurs with cytomegalovirus
Diagnostic features Intracellular homogeneous profiles which are eosinophilic or basophilic Either intracytoplasmic or intranuclear depending on source
Differential diagnosis Hyaline droplets: brightly eosinophilic and multiple throughout cortex (esp. S2)
Special procedures The various types of inclusions may need to be distinguished by electron microscopy
Comment: Nuclear and cytoplasmic inclusions are frequently encountered in the renal tubules of rodents on toxicity studies, and their pathogenesis may be quite diverse. Inclusions have been noted with heavy metals, antibiotics, and a wide variety of other agents, and they may also occur with viral infections. Although phospholipidosis usually appears with routine HE staining microscopically as vacuoles, lysosomal inclusions consisting of membranous whorls can be particularly prominent in renal phospholipidosis and these can be visualized easily using Toluidine blue stains and acid phosphatase immunohistochemistry (Schneider 1992). Depending on pathogenesis and location, inclusions may be associated with further parenchymal injury and degeneration over time.
Basophilic Granules (Figure 42): Proximal and Distal Tubules
Species: rat, mouse
Synonyms: none
Pathogenesis/cell of origin Basophilic granules occur with regular frequency in the cytoplasm of proximal and distal tubules with repeated administration of antisense oligonucleotide therapeutics. The preferential urinary excretion pathway and polyanionic nature of these molecules favors their incorporation and sequestration into lysosomes of the proximal tubules where, due to their modified structure, they fail to be metabolized rapidly.
Diagnostic features Intracellular homogeneous or granular profiles which are generally strongly basophilic, but a small percentage of which may also appear eosinophilic with HE stains Most commonly noted in proximal tubules, but can be found in distal convoluted tubulesand rarely within mesangial cells of the glomeruli They are generally associated with degenerative changes in cells only at high doses or after extended dosing periods, but can eventually lead to necrosis of affected cells
Differential diagnoses
Hyaline droplets: brightly eosinophilic and multiple as well as being diffuse throughout cortex
Inclusion bodies: specific to the particular type of agent exposed to. Basophilic granules are the preferred term for only those associated with antisense oligonucleotide therapeutics
Special procedures The nature of the granules can be confirmed by nucleic acid stains or via electron microscopy, in which phagolysosomes contain globular electron dense material interpreted to be oligonucleotide associated material
Comment: The kidney is one of the most sensitive organs for accumulation of phosphorothiorate oligonucleotide compounds in all preclinical toxicologic species (Marquis and Grindel 2000, Henry et al. 1999). They are filtered by the glomerulus and then absorbed by proximal tubules across the brush border as well as by active transport from the circulation across the basolateral membrane, based on their polyanionic nature. However, because of trapping within lysosomes and inefficient efflux across the apical membrane into the urine filtrate, they tend to accumulate with time (Rappaport et al. 1995; Sawai et al. 1996). Some oligonucleotides have been reported to produce eosinophilic granules rather than basophilic granules, and these are often contained within vacuoles of proximal tubules in monkey and other species (Henry et al. 1999).
Amyloidosis, Interstitial (Figure 43): Interstitium, tubules
Species: mouse, rat (rarely)
Synonyms: none
Pathogenesis/cell of origin Interstitial or tubular amyloid deposition generally follows rather than precedes glomerular amyloidosis Occurs spontaneously with age in some mouse strains but can also occur following chronic inflammatory conditions Spontaneous disease characterized by the extracellular deposition of polypeptide fragments of serum immunoglobulin fragments or other defective serum proteins AA amyloid is derived from circulating serum amyloid A (SAA) proteins, and these are acute-phase apolipoproteins that are associated with high-density lipoprotein (HDL) particles
Diagnostic features Peritubular and/or interstitial deposits of homogeneous eosinophilic material Deposits of insoluble protein fibrils with a β-pleated structure Congo red positive apple green birefringence and positive with Thioflavine T, or immunohistochemical staining Spontaneous development with age Strain and sex differences in occurrence and onset When severe, may result in papillary necrosis by disruption of blood supply to medulla Extremely common in the CD-1 strain as well as other strains of mice, while it is rare in rats May involve tubules/collecting ducts and interstitium, and other organs such as intestine are also frequently affected
Differential diagnosis Hyaline or fibrinoid deposits which are usually more focal and do not show the birefringence with Congo red
Comment: Amyloidosis is progressive and generally irreversible. It is a complicating factor in the interpretation of chronic studies in mice such as 2-year carcinogenicity studies in the CD-1 strain and can be a common cause of morbidity. It is becoming less common in mice strains such as CD-1 with awareness by suppliers and breeders. Amyloid fibrils are generally composed of light immunoglobulin polypeptide chains. Type A fibrils are the major component in experimental animals (Röchen and Shakespeare 2002). The protein precursor of amyloid is SAA, a family of major acute-phase proteins produced by the liver under the influence of cytokines (Yu et al. 2000). The acute phase protein under normal circumstances is degraded, however in mouse, hamster and man this protein can be deposited as amyloid in the kidney with extensive alterations in the glomeruli, tubules and interstitium. Deposits in mice can be seen from the age of 8 months (or earlier depending upon the strain) and the incidence increases with advancing age (Higuchi et al. 1991). Amyloidosis may involve more than one sublocation (both tubules and glomeruli and interstitium).
Regeneration, Tubule (Figures 44 and 45): Proximal and Distal Tubules
Species: rat, mouse
Synonyms: tubular cell regeneration
Pathogenesis/cell of origin Regeneration of tubular epithelium following necrosis/exfoliation where the basement membrane remains intact
Diagnostic features Response to cell loss in the tubule Very flat basophilic cells spreading over basement membrane Low basophilic cuboidal cells lining tubules with a rudimentary brush border High rate of mitosis
Differential diagnosis Tubular basophilia is a common feature associated with tubular regeneration following chemical injury; however, the terms should not be used interchangeably as tubular basophilia can occur without reparative processes. In many cases, such as CPN, the basement membrane will show thickening which is not a normal feature in regeneration.
Comment: After necrosis in a nephron there is feed back, which shuts down the glomerulus preventing the necrotic tubule from dilating. After acute necrosis, regenerative cells spread over the basement membrane in areas where the necrotic epithelium has been shed (Cuppage and Tate 1967). If the cell loss is patchy, the adjacent non-necrotic cells spread to cover the bare basement membrane giving a flattened basophilic lining to the tubule. Where the cellular loss has been more severe there is an increase in mitotic activity in the lower part of the nephron and cells migrate up to cover the exposed basement membrane. Following an acute wave of necrosis, the proximal tubular epithelium can be reconstituted within 5 to 7 days. Initially the cells are basophilic with a low almost squamous appearance gradually becoming more cuboidal by day 5 but with only a rudimentary brush border. By 10 to 14 days the regenerative cells are of a more normal height and staining quality and have a relatively normal brush border. After 21 to 28 days, the tubules show a normal enzyme profile and are functionally normal. Damage to the basement membrane will preclude this resolution and may lead to fibrosis. Several growth factors and cytokines have been shown to be involved or upregulated in this regenerative process (Dube et al. 2004). Regenerating tubular epithelium is often resistant to the initial nephrotoxin, because effects such as diminished secretory carrier systems or loss of the brush border may prevent further accumulation of toxin. Completely regenerated tubules may again be susceptible resulting in cyclic waves of degeneration and regeneration. In such cases, degeneration/regeneration as combined diagnoses has been used. In the absence of any other evidence of regeneration or degeneration, tubular basophilia is the preferred term.
Karyomegaly (Figure 46): Proximal and Distal Tubules
Species: rat
Synonyms: nuclear enlargement
Pathogenesis/cell of origin Presumed to represent repeated nucleic acid replication without nuclear divisions or cytokinesis
Diagnostic features Marked nuclear enlargement in tubule cells, predominantly proximal tubules Nuclei may be slightly irregular in shape Usually sporadic but may be frequent with certain chemicals
Differential diagnosis Should not be confused with the modest nuclear enlargement representing DNA replication during the cell cycle
Comment: Karyomegaly (increase in nuclear size) and karyocytomegaly (increase in both cell size and nuclear size) are occasionally noted in rat tubular epithelium and are of uncertain pathogenesis. Affected nuclei may exhibit hyperchromatic staining, multiple nucleoli, and irregular outlines. Karyomegaly in proximal tubule cells has usually been observed as a chronic event after administration of renal carcinogens, other agents such as lysinoalanine, heavy metals, and some anticancer agents (Hard et al. 1995; Hard et al. 1999; Montgomery and Seely 1990). Some renal carcinogens (e.g., aflatoxin or ochratoxin A) induce frequent tubule karyomegaly in the rat as an early event (Boorman et al. 1992). A karyomegalic cell is not regarded as a preneoplastic cell that is necessarily destined to develop into a renal tubule tumor, so any relationship to carcinogenicity is uncertain (Hard et al. 1995; Montgomery and Seely 1990).
Hypertrophy, Tubule (Figures 24, 47–49): Proximal and Distal Tubules, Collecting Ducts
Species: rat, mouse
Synonyms: none
Pathogenesis/cell of origin Distal tubule hypertrophy can develop as a compensatory or an adaptive response to increased renal demand Associated with increases in ion transport capacity; can occur because of dietary excess of sodium chloride, or following high doses of certain drugs Sporadically observed in advanced CPN Collecting duct cellular changes are seen following altered potassium homeostasis (e.g., potassium dietary changes or loop diuretics)
Diagnostic features Increase in size of individual tubule cells without increase in cell number Cytoplasm usually brightly eosinophilic, but can be pale; single layer of tall cuboidal or columnar cells with broad bases; nuclei often located apically Seen mainly in cortex involving the distal convoluted tubule
Comment: Although hypertrophic tubules consist of a single layer of cells, tangential sections can sometimes make them appear misleadingly complex because of the enlarged nature of the cells. Hypertrophic tubules are not proliferative and are not preneoplastic lesions. The features of tubule hypertrophy are consistent with an adaptive response to an increase in active transcellular transport capacity (Ellison, Velazquez, and Wright 1989; Hard et al. 1999). Outer and inner medullary collecting duct cells are heterogenous with specialized roles in electrolyte, hydrogen ion, and bicarbonate transport (Kaissling et al. 1985). Adaptive increases in intercalated disc dark cells are seen with hypokalemia (Evan et al. 1980).
Glomerular Changes
Glomerulonephritis (Figures 50–55, 119, and 120): Glomeruli
Species: mouse, rat
Synonyms: none
Pathogenesis/cell of origin A sequela to chronic degenerative changes in the glomeruli and marked hemodynamic alterations. Complement activation leading to podocyte injury shown in Heymann nephritis model in rat (Cybulsky, Quigg, and Salant 2005)
Diagnostic features
Membranoproliferative form shows basement membrane thickening which may be uniform with antibodies directed against glomerular basement membrane antigen or “lumpy” with irregular thickening with circulating immune complex deposition. Glomerular basement membrane thickening confirmed on PAS, methanamine silver, or EM.
Crescentic form shows increased visceral and/or parietal glomerular epithelial cells two or more cells thick. Usually segmental and may form adhesions reducing Bowman’s space. Fibrocellular and fibrous forms of crescents more blue/green on trichrome stains. Mesangial cell proliferation Mononuclear inflammatory cell infiltration
Differential diagnoses Hyaline glomerulopathy: lacks mononuclear cell inflammatory change CPN with glomerulopathy component: has changes in other compartments such as tubules and basement membranes Bowman’s capsule metaplasia/hyperplasia Glomerulosclerosis: replacement of glomeruli with fibrosis without significant contraction Loss of nuclei generally is more common than with atrophy.
Comment: Membranoproliferative and crescentic features may both be present simultaneously. It is often difficult to specifically characterize glomerular injury with routine HE stains alone. In such cases, a simple diagnosis of glomerulonephritis or glomerulopathy without additional modifiers may be sufficient.
Glomerulopathy, Hyaline: Glomeruli
Species: B6C3F1 mouse, rat
Synonyms: none
Pathogenesis/cell of origin/cell of origin A sequel to chronic degenerative changes in the glomeruli with immunoglobulin deposition.
Diagnostic features Loss of cellularity of glomerular capillary tufts, usually diffuse and global Acellular deposition of immunoglobulin; PAS positive, Masson’s Trichrome positive, Congo Red negative
Differential diagnoses Glomerulosclerosis: associated with replacement of glomeruli with fibrosis rather than basement membrane thickening Membranoproliferative glomerulonephritis: associated with megangial cell proliferation and mononuclear cell infiltration Amyloidosis: basement membrane glomerular depositions are Congo red positive and birefringent
Comment: Hyaline glomerulopathy has usually been described as occurring in control mice, but it was recently identified as a response to chronic pulegone exposure in both rats and mice (National Toxicology Program [NTP] 2009). Electron microscopy showed the glomerular mesangium to be occupied by finely granular amorphous material suggestive of immune complex deposition (NTP 2009). In humans, hyaline glomerulopathy is termed immunotactoid glomerulopathy and is characterized ultrastructurally by organized microtubular structures in the mesangium (D’Agati, Jennette, and Silva 2005).
Glomerulopathy, Mesangioproliferative (Figure 56): Glomeruli
Species: mouse, rat
Synonyms: mesangial thickening
Pathogenesis/cell of origin Mononuclear phagocytes (CD68 positive) remove high molecular proteins leaking through glomerular basement membrane and proliferate if protein load increased.
Diagnostic features Mesangial cells hypertrophic Some mitotic cells may be present Increased mesangial matrix, initially axial, PAS positive No capillary changes Need thin sections and special stains to confirm
Differential diagnoses Glomerulonephritis: has additional features with basement membrane thickening and a mononuclear cell inflammatory component Hyaline glomerulopathy: is more acellular
Comment: Morphine-induced mesangial cell proliferation is mediated via kappa opioid receptor in rats (Weber et al. 2008). Thrombopoietin transgenic mice develop a mesangioproliferative glomerulopathy (Shimoda et al. 2007). In some cases, it may be sufficient to use the simpler terms of glomerulopathy or glomerulonephritis in preclinical toxicity studies, without additional modifiers where further classification cannot be established.
Glomerulosclerosis (Figures 1 and 33): Glomeruli
Species: mouse, rat
Synonyms: includes focal segmental glomerular sclerosis
Pathogenesis/cell of origin A sequel to chronic degenerative changes in the glomeruli and marked hemodynamic alterations
Diagnostic features Shrinkage and contraction of one or more glomerular capillary tufts, generally with coincidental enlargement of Bowman’s space; Commonly associated with advanced CPN in the rat and chronic nephropathies in mice (Hard and Seely 2005; Ma and Fogo 2003); Can be focal or multifocal, segmental, or global in distribution
Differential diagnosis Glomerular atrophy: shrinkage of glomeruli without fibrosis
Comment: Early minimal change disease is thought to progress to focal segmental glomerular sclerosis, e.g., puromycin aminonucleoside nephrosis (Hagiwara et al. 2006. Once hemodynamic or degenerative changes in the glomerulus are initiated, a complex sequence of events within the mesangium and podocytes are initiated, mediated by TGF-beta and CTGF, which result in the stimulation of fibroblast proliferation and collagen formation with the eventual replacement of normal architecture (Frazier et al. 1996; Kriz and Lehir 2005; Lee and Song 2009). Glomerulosclerosis may be induced by several toxicants including N-nitrosomorpholine in rats, and associated histologic and ultrastructural alterations have been described in detail (Romen, Bannasch, and Aterman 1975).
Mesangiolysis (Figure 57): Glomeruli
Species: mouse, rat
Synonyms: Hemolytic uraemic syndrome
Pathogenesis/cell of origin a degenerative lesion associated with endothelial injury, complement activation and loss of patency of capillary wall
Diagnostic features Necrosis of mesangial cells and/or endothelial cells within the glomerulus Proteinaceous leakage into Bowman’s space; Focal and segmental Fibrin deposition within capillary tufts Crescent formation and sclerosis follow
Differential diagnosis Glomerulosclerosis: replacement of glomeruli with fibrosis without significant contraction; loss of nuclei is more common than with atrophy
Comment: Mesangiolysis is uncommonly encountered in preclinical toxicity studies and is most often associated with vasculotoxic compounds or in diabetic models. The Anti-Thy-1 rat model may exhibit mesangiolysis in the early stages (Kriz et al. 2003). It is also associated with complement activation, e.g., some snake venoms. Special stains and/or electron microscopy may be necessary for diagnosis and distinction from other forms of glomerular injury.
Amyloidosis, Glomerular (Figure 43): Glomeruli
Species: mouse, hamster
Synonyms: none
Pathogenesis/cell of origin Spontaneous disease characterized by the extracellular deposition of polypeptide fragments of serum immunoglobulin fragments or other defective serum proteins AA amyloid is derived from circulating SAA proteins, and these are acute-phase apolipoproteins that are associated with HDL particles
Diagnostic features Glomeruli are hypocellular and contain amorphous pale pink deposits of amyloid protein that stain positively with Congo Red and show apple green staining under polarized light The glomeruli are most frequently involved but amyloidosis can progress to include tubules/collecting ducts and interstitium; other organs such as intestine are also frequently affected (Gruys and Snel 1994) Ultrastructural study reveals extensive involvement of mesangial cells and basal lamina by amyloid fibrils
Differential diagnoses Membranous glomerulonephritis with basement membrane thickening with immunoglobulin, immune complexes, and complement deposition Hyaline glomerulopathy; PAS positive, Congo red negative, immunostains for IgG, IgM, or IgA.; glomerulosclerosis: obliteration of glomeruli with fibrosis; trichrome positive while Congo red staining is negative
Comment: Amyloidosis is progressive and generally irreversible. It is a complicating factor in the interpretation of chronic studies in mice such as 2-year carcinogenicity studies and can be a significant cause of mortality. Historically, amyloidosis was extremely common in CD-1 strains but is becoming less so as suppliers and breeders are aware of the condition. It is still common in some other strains of mice, while it is rare in rats. See Interstitial Amyloidosis for more details on pathophysiologic mechanism.
Atrophy, Glomerular (Figure 1): Glomeruli
Species: mouse, rat
Synonyms: collapsing glomerulopathy
Pathogenesis/cell of origin Glomerular atrophy is a sequel to chronic degenerative changes in the glomeruli and subsequent hemodynamic alterations; glomerular atrophy is also observed in streptozotocin-induced diabetes in rats with podocyte apoptosis (Menini et al. 2007)
Diagnostic features Shrinkage and contraction of one or more glomerular capillary tufts, generally with coincidental enlargement of Bowman’s space Can be focal or diffuse, segmental or global in distribution Collapsing glomerulopathy of kd/kd mice shows loss of synaptopodin and WT-1 podocyte markers and increased Ki67 expression (Barisoni et al. 2005)
Differential diagnosis Glomerulosclerosis has replacement of glomeruli with fibrosis without significant contraction; loss of nuclei is more common than with atrophy
Comment: This lesion is indicative of chronic glomerulopathy and is a hallmark of end-stage disease (Hard et al. 1999). Atrophy is also present with atubular glomeruli of polycystic kidney disease (autosomal dominant polycystic kidney disease [ADPKD]) rats (Tanner et al. 2002).
Dilation, Bowman’s space (Figures 1 and 58): Glomeruli
Species: mouse, rat
Synonyms: enlargement of Bowman’s space
Pathogenesis/cell of origin Dilation of Bowman’s space may occur as a consequence of increased hydrostatic pressure within Bowman’s capsule due to glomerular hyperfiltration or as a consequence of shrinkage of the capillary tufts due to atrophy. It can also be a result of ascending intra-tubular reflux
Diagnostic features Pronounced increases in the diameter of Bowman’s capsule without concomitant increased size of the glomerular tufts, resulting in widened Bowman’s space Commonly associated with infarction of adjacent parenchyma or advanced chronic nephropathies (Hard and Seely 2005; Hard et al. 1995) Can be focal or multifocal, segmental, or global in distribution
Differential diagnosis Glomerulopathy: there are no other significant changes in the glomerular cellular elements with dilation of Bowman’s space
Comment: This lesion is indicative of alteration of the intraglomerular fluid pressures and will lead to degenerative sequelae in the glomeruli and accompanying tubules (Hard et al. 1999).
Metaplasia/Hyperplasia, Bowman’s Capsule (Figure 59): Glomeruli
Species: mouse, rat
Synonyms: metaplasia, Bowman’s capsule; hyperplasia of parietal epithelial cells
Pathogenesis/cell of origin The cells of the parietal epithelium of the capsule are under hormonal influence in mature male mouse and alterations in circulating levels of testosterone may affect their morphology
Diagnostic features Replacement of flat squamous parietal cells of Bowman’s capsule in female mice by tall cuboidal epithelium; hypertrophy of Bowman’s epithelium in male mice is normal; In rats, the single cell layer of squamous parietial epithelium may also undergo a similar change to cuboidal cells resembling proximal tubules Increased number of parietal epithelial cells accompanies change in morphology
Differential diagnoses Handling artifact involving herniation of proximal tubular epithelium into the capsular space: epithelium is indistinguishable from surrounding PCTs Indicators of altered sexual dimorphism may be evident in other organs, e.g., adrenal
Comment: Bowman’s capsule metaplasia/hyperplasia may occur spontaneously in older rats and is more commonly noted in males than in females (Hard et al. 1999). In contrast, metaplasia of Bowman’s capsule is generally limited to female mice. Parietal epithelium may undergo hypertrophy and/or hyperplasia with glomerular disease, particularly in glomerulosclerosis in rats (Peter, Burek, and Van Zwieten 1986). It has been noted in spontaneous hypertensive rats as well, but it is unclear if the change is related to hypertension (Haensley et al. 1982). Either hyperplasia or metaplasia could be used as the diagnosis, on a case-by-case basis. These metaplastic/hyperplastic changes are not considered preneoplastic lesions.
Hyperplasia, Mesangial: Glomeruli
Species: mouse, rat
Synonyms: mesangial cell hypertrophy
Pathogenesis/cell of origin Mesangial cells increase in number, size, and cytoplasmic volume as a response to certain stimuli including hyperglycemia and with accumulation of certain drugs (Wehner and Petri 1983)
Diagnostic features Increased number of mesangial nuclei within the glomeruli, with or without concomitant increase in cell size Over time and with further injury, mesangial hyperplasia may be associated with an increase in the secretion of mesangial extracellular matrix proteins and marked proteinuria, where it is more properly labeled mesangioproliferative glomerulonephritis These hyperplastic changes are not considered preneoplastic lesions and have not been associated with renal tumors
Differential diagnoses Mesangioproliferative glomerulonephritis or other related glomerulopathies where there is an increase in mesangial matrix and extracellular matrix proteins, potentially without a concurrent increase in mesangial cell number and cell volume Glomerulonephritis should be accompanied by proteinuria and tubular casts, while this is not a feature of simple mesangial hyperplasia.
Comment: Mesangial hyperplasia has been reported commonly in the streptozotocin-induced diabetic rat model, as well as with some agents inducing phospholipidosis such as chloroquine, where they are accompanied by lysosomal accumulations in podocytes and mesangial cells (Wehner and Petri 1983). Other drugs, such as some growth factor inhibitors or anticancer agents, also have resulted in mesangial inclusion bodies, and these changes may also be associated with mesangial hyperplasia. Elevated serum ammonia levels or administration of NH4Cl induces hypertrophy but not hyperplasia in mesangial cells (Ling et al. 1998). This hypertrophy is thought to be caused by the reduction of protein degradation in the absence of enhanced protein synthesis in mesangial cells under these conditions.
Inflammation in the Kidney
Infiltrate, Inflammatory Cell, Interstitium (Figures 33, 49, and 60): Interstitium of Cortex and Medulla
Species: rat, mouse
Synonyms: inflammation, focal
Pathogenesis: unknown
Diagnostic features Small foci of inflammatory cells within the interstitium with mononuclear cells (lymphocytes, plasma cells, and/or macrophages) and/or small numbers of neutrophils Acute and chronic forms
Comment: Inflammatory cell infiltrates are extremely common in rats and mice and are often of no toxicologic significance. The number of inflammatory cell foci increases with age and/or with the presence of CPN in rats or chronic nephropathy in mice.
Interstitial Nephritis: Interstitium of Cortex and Medulla
Species: rat, mouse
Synonyms: none
Pathogenesis/cell of origin unknown, but in some cases may be associated with antibody complexes directed at the basement membrane or extracellular matrix elements; interstitial nephritis has been associated with drug administration of certain compounds such as succinimides (Barrett, Cashman, and Moss 1983), lithium, methacillin (Linton et al. 1980), allopurinol and with a specific syndrome related to chronic NSAID treatment (independent of COX activity); interstitial nephritis is also one component of CPN in rats and is a consistent feature of chronic nephropathy in mice.
Diagnostic features More generalized inflammation of the interstitium with mononuclear cells (lymphocytes, plasma cells, and/or macrophages) and variable degrees of edema Acute and chronic forms May be an increase in fibrocytes resulting in subsequent fibrosis
Differential diagnoses Inflammatory infiltrate, interstitium: focal, primarily lymphocytic infiltrates rather than the extensive, mixed inflammation noted with interstitial nephritis Chronic pyelonephritis: the inflammatory infiltrate is generally more severe in the papilla and medulla and also involves tubular lumina; acute pyelonephritis has a predominance of neutrophils Lymphoma: a uniform and homogenous population of large or less differentiated lymphocytes CPN: typically focal and associated with alterations of the basement membrane; tubules basophilic; interstitial nephritis is only minor component of this syndrome
Comment: Interstitial inflammation may accompany numerous renal conditions such as those associated with antibodies against the basement membrane, immune complex diseases, or the chronic nephropathies, but the term interstitial nephritis is reserved for generalized inflammatory lesions of the interstitium, regardless of cause. With focal forms of interstitial inflammatory infiltrates, there are solitary, scattered, or perivascular accumulations of well-differentiated lymphocytes, and the preferred term for these foci is “inflammatory infiltrate, interstitium.” The generalized form of interstitial nephritis in rodents is characterized by diffuse or patchy distributions of lymphocytes, plasma cells, and usually fewer macrophages. This distinction is an important one, as the focal form is likely to have significantly less impact on renal function and is less likely to be related to test article treatment. Primary intersitital disease in rats, unlike in dogs or humans, is relatively uncommon, and consistent or perpetuated, minimal, chronic renal toxicity in rats is expressed as exacerbated CPN rather than chronic interstitial nephritis alone. However, interstitial nephritis is a recognized complication of drug treatment in rodents and may represent the predictable renal lesion expression of specific agents (Linton et al. 1980). The pathophysiologic mechanism of these types of changes with agents such as the succinimides or methacillin, and the reason for their localization to the interstitium is not well understood.
Microabscess: Cortex, Medulla
Species: rat, mouse
Synonyms: tubulitis, renal abscess, suppurative nephritis, pyelonephritis (in diffuse or severe cases)
Pathogenesis/cell of origin Solitary or multifocal Discrete microscopic aggregations of neutrophils (either intraluminal in tubules, in Bowman’s space of glomeruli, or in the interstitium) When present in interstitial tissue, usually associated with bacterial emboli Represents focal bacterial infection
Diagnostic features Frequently formed around a nidus of exfoliated cells in tubular lumen A punctate focus of acute inflammatory cells with a central focus of necrotic debris, sometimes with demonstrable bacteria In interstitium associated with bacterial emboli. Numerous neutrophils are seen filling renal tubules or interstitial space; these leukocytes may form into a cast within the tubule The neutrophils can coalesce in the distal tubules and collecting ducts, then be passed in urine as WBC casts The cut surface of the kidney reveals yellowish appearance in both cortex and medulla in severe cases Affected tubules have conspicuously basophilic, hyperplastic (simple type) epithelium, are surrounded by an inflammatory cell infiltrate, and contain a plug of cell debris (mainly degenerating neutrophils) in the lumen
Differential diagnoses Chronic inflammation: Cellular infiltrate is a mixture of granulocytic, lymphocytic, histiocytic cells, fibrosis Tubulo-interstitial nephritis (obstructive nephropathy): Along with crystal formation, inflammation of the renal tubule or interstitium, plus foreign-body giant cells and hyperplasia of tubular epithelial cells, is observed Acute inflammatory focus: Usually more focally extensive or diffuse in nature and lacking central area of necrosis and bacteria
Comment: Microabscesses are characterized by discrete microscopic aggregations of neutrophils, usually in the interstitial tissue. They may be either solitary or multifocal, and are often bilateral. Renal microabscesses are usually the result of systemically disseminated bacterial infection, occurring by direct embolism of pyogenic microorganisms in intertubular capillaries or glomerular loops. Microabcesses can be associated with intravenous administration of drugs where infected thromboemboli are formed. Bacteremia may give rise to microabscesses centered on the glomeruli. Microabscesses may also occur in the tubule lumen where stasis and/or cellular debris present a good medium for bacterial growth. Pyelonephritis or pyelitis may also present as microabscessation (Duprat and Burek 1986), and represents acute inflammation of the renal pelvis and parenchyma usually caused by ascending bacterial infection (refer to pyelonephritis for more information). Solitary proximal tubules affected with microabscessation often occur in advanced stages of CPN, in which setting they need not be diagnosed separately. If sectioned tangentially, such a lesion may appear as a solid, basophilic tubule, mimicking a focus of atypical tubule hyperplasia. The presence of neutrophils in and around the focus identify the lesion as an inflammatory profile, and not preneoplastic.
Edema, Interstitial: Cortex, Medulla
Species: mouse, rat
Synonyms: none
Pathogenesis/cell of origin Interstitial tissue of cortex, medulla, and papilla
Diagnostic features Widening of perivascular and intertubular spaces (interstitial edema) Pink-staining homogenous material in interstitium and occasionally in tubular lumen with dilatation
Differential diagnosis Postmortem autolysis: Uniform dissolution of entire tissue section with no change in organization of cellular components
Comment: Edema is characterized by the presence of eosinophilic proteinaceous fluid in the interstitial space in cortex, medulla, and papilla. It may occur independently or it may accompany acute inflammation. Mild interstitial edema and inflammation, associated with acute tubular injury, is a frequent feature of nephropathy associated with NSAID toxicity (Hard and Neal 1992).
Pyelonephritis (Figures 61 and 62): Medulla (Intersititum and Collecting Ducts)
Species: rat, mouse
Synonyms: papillitis, pyelitis (when restricted to renal pelvis)
Pathogenesis/cell of origin May be a result of ascending bacterial infection from the lower urinary tract or rarely as an extension of drug-induced interstitial nephritis originating in the cortex or outer medulla, but more commonly in toxicity studies is a sequela to papillary necrosis such as occurs with chronic analgesic (NSAID) toxicity (Bach and Bridges 1985a; Bach and Nguyen 1998); may also result from ulcerative lesions of the urothelium associated with urolithiasis of the pelvis; pyelonephritis can increase in severity and incidence with increasing age, but the spontaneous acute form is rare in the Fischer F344 rat
Diagnostic features Suppurative inflammation of the interstitium and tubular lumina of the collecting ducts in the inner medulla and papilla, with radial extension into cortex Tubular basophilia of the cortical tubules or those in the outer medulla Secondary necrosis of the collecting ducts; Subsequent progression to chronic inflammation with infiltration of lymphocytes, plasma cells, and moncytes with interstitial fibrosis Tip of papilla may become necrotic and ulcerated; Tubule epithelium adjacent to intraluminal neutrophils is basophilic and reversibly hyperplastic Macroscopically in severe cases, the renal pelvis may be dilated with suppurative exudates; the adjacent medulla is hyperemic, and whitish or yellow radial streaks may be visible in the cortex Microscopically, ulceration and necrosis of the papilla is present and marked to severe infiltration of the medullary and cortical parenchyma by neutrophils is characteristic, particularly within cortical tubule lumens
Differential diagnoses Interstitial nephritis: the inflammatory infiltrate is generally predominated by lymphocytes and macrophages and involves the cortex as well as the medulla. Retrograde nephropathy: characterized by tubule dilation but lacks suppurative inflammation
Comment: Pyelonephritis is considered a tubulointerstitial disease and is characterized by suppurative or mixed inflammation and often necrosis of the pelvis and distal renal parenchyma, which when chronic, results in interstitial fibrosis and loss of collecting ducts (Heptinstall 1964). Pyelonephritis is the preferred term for the spectrum of lesions involved, and individual diagnostic terms such as tubular basophilia, inflammatory infiltrates, casts, and so on, can be lumped under this diagnosis except in cases where there is some question as to whether they are separate versus related processes. Rodents are prone to spontaneous (infectious) cases of pyelonephritis, and several different rodent models of pyelonephritis are used to assess antibiotic therapy for the condition in humans. Ascending infection is a common consequence of cystitis in rats due to spontaneous vesicoureteral reflux, but descending infection from hematogenous origin also occurs (Heptinstall 1965). Several factors likely predispose the medulla toward infection including relative hypoxia (due to low perfusion in the vasa recta), hypotonicity-associated depression of leukocytic phagocytic activity, and continuity with ureters and urinary bladder where bacteria may be harbored. Inflammatory changes in the medulla extend radially into the cortex. Although commonly associated with primary papillary necrosis, pyelonephritis without concomitant papillary injury is also occasionally encountered as a drug-induced lesion in toxicity studies, particularly with immunosuppressive compounds such as cyclosporine which affect T cell function and cell mediated immunity (Miller and Findon 1988). Specific virulence factors have been identified which enhance the pathogenicity of the bacteria, with E. coli and Proteus species as the most commonly isolated pathogens taken from lesions in rodents (Pichon et al. 2009; Rice et al. 2005).
Fibrosis, Interstitial (Figures 1, 8, and 33): Cortex, Medulla
Species: rat, mouse
Synonyms: peritubular fibrosis
Pathogenesis/cell of origin Formed as a response to damage to adjacent tubules
Diagnostic features Interstitial accumulation of fibrous collagen with an increase in interstitial cells, particularly myofibroblasts Extracellular matrix accumulation surrounding degenerate tubules, and appearing following the tubular change Localized or diffuse depending on extent of tubular damage End-stage fibrous scar with depression of kidney surface
Differential diagnoses Interstitial nephritis: which has more lymphocytes, plasma cells, and macrophages Differentiate from inflammatory foci and early interstitial nephritis (Inflammatory infiltrate, interstitium)
Comment: The cortical interstitium is normally composed of a network of fibroblasts and dendritic cells with only a small number of lymphocytes or macrophages (Kaissling and Le Hir 2008). In chronic interstitial fibrosis, the interstitial cells have myofibroblast characteristics which are considered to be derived from the proliferation and differentiation of the residual fibroblasts (Yang and Liu 2001). The timing of the appearance of these cells and their localization adjacent to damaged tubules indicates that the interstitial reaction is stimulated by changes in the tubules (Eddy 1996; Frazier et al. 2000). The transformed cells secrete large amounts of metalloproteinase-2 which specifically degrades basement membranes. TGF-β has been shown to cause the transformed cells to release α-smooth muscle actin and initiate the reorganization of actin filaments in the interstitial matrix (Eddy 1996). Proteinuria can lead to increased glomerular filtration of growth factors such as HGF and TGF-β giving high tubular levels of growth factors. These in turn can activate cytokines in tubular cells which stimulate interstitial fibrosis (Wang, Lapage, and Hirschberg 2000). Studies indicate that the myofibroblasts and fibrosis reaction is part of a reparative process and blocking of their transformation can increase renal dysfunction. The lesion progresses chronically, which can eventually result in depressions of the capsular surface.
Proliferative Lesions (Non-Neoplastic)
Hyperplasia, Tubule (Figures 63 –65): Proximal and Distal Tubules, Collecting Ducts Hyperplasia, Tubule, Simple (Figure 63)
Synonyms: Focal tubule hyperplasia Increase in the number of tubule lining cells but not intraluminally or beyond a single layer Cytoplasm is usually basophilic May appear as focal crowding of lining cells in tangential sections Size is variable due to lumen dilation or increased cell number.
Differential diagnoses Atypical tubule hyperplasia: More than a single cell layer forming a solid tubule, papillary projections or a lining several cells thick where the affected tubule is dilated or cystic Tubular basophilia: lacks crowding of nuclei; generally limited to tubules from a solitary nephron
Comment: The basophilic tubules characteristic of CPN represent simple tubule hyperplasia (Alden et al. 1992; Hard et al. 1995). Simple tubule hyperplasia can be spontaneous as in CPN or can be chemically induced and may be a consequence of single cell degeneration with compensatory regeneration.
Hyperplasia, Tubule, Atypical (Figures 64 and 65)
Species: rat, mouse
Synonyms: atypical tubular hyperplasia, atypical renal cell hyperplasia
Pathogenesis/cell of origin: Origin from renal tubule epithelium, usually proximal tubule; origin from distal nephron also possible
Diagnostic features Complex proliferation confined to an individual tubule; may consist of 1 to 5 solid profiles representing convolutions of a single tubule Occurrence is solitary More than a single cell layer forming a solid tubule, papillary projections, or a lining several cells thick where the affected tubule is dilated or cystic. Tubule lumen may be partially or completely obliterated Tubule size may be increased due to lumen dilation or increased cell number Typically, cytoplasm well developed, with well-defined cell borders; usually basophilic with a glassy sheen but occasionally eosinophilic or clear; nucleoli usually prominent Cellular and nuclear pleomorphism is present with marked variability in cell and nuclear size Nucleus to cytoplasm ratio is increased Integrity of the single tubule structure is maintained, with no evidence of compression of adjacent parenchyma Expansive nature indicated by close encirclement of fibroblasts or capillaries peripherally; no vascular ingrowth
Differential diagnoses Regeneration, tubule: cells are basophilic but cell number is not increased. Regeneration, tubule in CPN: Cells without well-defined cell borders or glassy sheen; nucleoli are usually not prominent; may be surrounded by thickened basement membrane; growth may appear complex but there is no margination or peripheral alignment of fibroblasts; (Hard and Seely 2006) Hypertrophy, tubule: Cells are enlarged; single layer is retained; cell number is not increased. Adenoma: Proliferation extends beyond integrity of a single tubule; usually fibrovascular ingrowth evident; if solid, size exceeds 5 to 6 tubule profiles of tubule and may show some complexity in structure that is not in accord with the convolutions of a single tubule (Shinohara and Frith 1980).
Comment: Atypical tubule hyperplasia is generally accepted as a preneoplastic lesion and on a continuum with adenoma (Bannasch 1984; Frith, Terracini, and Turusov 1994; Hard 1984; Hard 1985a). Peripheral encirclement of fibroblasts or capillaries around the hyperplastic focus appears to be a particularly useful indicator of expansive growth associated with atypical tubule hyperplasia (Hard and Seely 2005). The term dysplasia is not recommended for this lesion as it denotes a specific diagnostic entity of anomalous kidney development in some species (Hard et al. 1999; Picut and Lewis 1987; Seely 1999).
Hyperplasia, Oncocytic (Figure 66): Collecting Duct
Species: rat
Synonyms: none
Pathogenesis/cell of origin Origin from collecting tubule epithelium
Diagnostic features Composed of few to several tubule profiles consistent with convolutions of a single tubule entity Oncocytic hyperplasia is a monomorphic population of cells with finely granular cytoplasm staining pale to faintly eosinophilic with centrally located nuclei and indistinct nucleoli
Differential diagnoses Oncocytoma: Small, solid masses extending beyond the integrity of a single tubule entity with larger size, compression of surrounding tubules and/or altered growth pattern Adenoma, chromophobic: Cell borders prominent and well defined
Special procedures Ultrastructurally, the main cytoplasmic feature of oncocytes is a dense crowding of atypical mitochondria Immunohistochemically, oncocytes stain positively for cytochrome-c-oxidase
Comment: Oncocytic hyperplasia is a proliferative lesion that is difficult to separate from oncocytoma. Either lesion appears to be a benign end-stage process that does not progress into carcinoma, and metastases have not been reported (Bannasch et al. 1998a; Nogueira and Bannasch 1988; Montgomery and Seely 1990). Some oncocytomas have abnormal or irregular morphologies which makes differentiation from oncocytic hyperplasia easier, but any oncocytic lesions larger than 3 glomeruli in profile should be considered an oncytoma rather than hyperplasia. Oncocytic hyperplasia and oncocytomas should be scored separately from other types of renal tubule hyperplasia or adenoma, because of their different renal segment of origin, distinctive morphology, and implications for risk assessment.
Miscellaneous Lesions
Extramedullary Hematopoiesis: Interstitium Adjacent to Pelvis
Species: rat, mouse
Synonyms: none
Pathogenesis/cell of origin Erythroid and/or myeloid progenitor cells Occur with agents that affect red blood cell production, but the specific nature of the stimuli in rodents varies (Ben-Ishay 1977) EMH in the kidney is less frequently encountered in rodents than in the spleen or liver May occur under conditions of hypoxia, with administration of certain cytokines or with exogenous erythropoietin
Diagnostic features Heterogenous clusters of differentiated erythroid and/or myeloid cells located in the adipose tissue and interstitium adjacent to the renal pelvis Do not invade or replace existing renal tissue
Differential diagnosis lymphoma or leukemia: homogenous group of cells with invasive characteristics more often extending into and replacing renal parenchyma
Comment: The major toxicologic significance of renal extramedullary hematopoiesis is in the suggestion of the presence of a regenerative anemia and/or erythroid or myeloid stimulation (Dixon, Heider, and Elwell 1995; Pospisil et al. 1998).
Hyperplasia, Juxtaglomerular: Cortex
Species: rat, mouse
Synonyms: juxtaglomerular hypertrophy
Pathogenesis/cell of origin juxtaglomerular cells This change occurs in rodents following hypertension, sodium depletion, adrenalectomy, and especially after treatment with ACE inhibitors and angiotensin II antagonists (Doughty et al. 1995; Owen et al. 1995) This is an adaptive response in the afferent and efferent arteriolar superficial cortex after reduced renal blood flow in order to maintain glomerular filtration by vasoconstriction of the efferent cells
Diagnostic features Hyperplasia and hypertrophy of the juxtaglomerular cells Renal secreting cells of the JGA are modified arteriolar smooth muscle cells; hypertrophy is visible as an onion skin-like morphology of the JGA in advanced cases Increased granularity of cells which often demonstrate increased renin immunostaining and prominent toluidine blue staining in thin sections
Differential diagnosis: none
Comment: Angiotensin II receptor or ACE antagonism stimulates increased renal renin production by hypertrophy of existing granulated cells, metaplasia of smooth muscle cells to renin-synthesizing cells, and cell proliferation (as noted by marked increases in BrdU labeling) in juxtaglomerular cells (Owen et al. 1995; Ozaki et al. 1994). Ultrastructurally, hypertrophic cells contain abundant rough endoplasmic reticulum and free ribosomes, and prominent Golgi complexes associated with numerous cytoplasmic coated vesicles (Dominick et al. 1990). Ultrastructural changes also suggest stimulated renin synthesis by a regulated pathway, renin secretion by exocytosis and cytoplasmic solubilization of granules under conditions of excessive stimulation (Jackson and Jones 1995).
Proliferative Lesions: Neoplasia
Adenoma (Figures 67–70): Kidney
Species: rat, mouse
Synonyms: renal tubule adenoma, renal cell adenoma, benign renal epithelioma, benign renal cell tumor
Pathogenesis/cell of origin Basophilic cell adenomas are presumed to originate mainly from proximal tubules, while some clear cell tumors and acidophilic cell adenomas have been shown to originate from the collecting ducts, at least in the rat Origin of the oncocytic variant from collecting tubules has been demonstrated in rats (Nogueira and Bannasch 1988; Zerban et al. 1987)
Diagnostic features Solitary, circumscribed, or irregular, nodular growth; usually located in the outer zones of the kidney (i.e., cortex and outer stripe of outer medulla; Nogueira et al. 1989) Demarcated with compression of the surrounding parenchyma Cells are well differentiated Cellular or nuclear pleomorphism may be present. Cell staining is often basophilic, but eosinophilic, amphophilic, oncocytic, clear cell, chromophobic, or mixed cell variants can occur occasionally Oncocytic variants have finely granular, pale to faintly eosinophilic, cytoplasm and centrally located nuclei with indistinct nucleoli and form small solid monomorphic masses in the outer zones of the cortex Oncocytic cells stain positively for cytochrome-c-oxidase (Mayer et al. 1989) and ultrastructurally, the main cytoplasmic feature is a dense crowding of atypical mitochondria (Krech et al. 1981) Larger than 5 to 6 contiguous tubular profiles and extends beyond confines of original tubule structure; Growth pattern is solid, tubular, cystic, lobular, papillary, cystopapillary, or mixed Mini-lumens may be present Early vascular ingrowth may be present No evidence of multiple areas of necrosis or hemorrhage
Differential diagnoses Atypical tubule hyperplasia: Proliferation does not exceed integrity of a single tubule; usually less than 6 contiguous profiles of proliferation representing convolutions of a single tubule; vascular ingrowth absent (Bannasch and Ahn 1998b; Dietrich and Swenberg, 1991; Hard 1990; Mitsumori et al. 2002) Carcinoma: Larger than a few millimeters; evidence of cell and/or nuclear pleomorphism, multiple areas of necrosis or hemorrhage; usually conspicuous mitotic activity; invasion or metastasis may be present (Hard 1984; Alden et al. 1992; Hard et al. 2001a) Regenerative tubules in CPN: Bland cells and nuclei; no vascular ingrowth; often surrounded by thickened basement membrane with no margination of fibroblasts (Hard and Seely 2005) Oncocytoma: clusters of large, lightly eosinophilic, granular cells usually without glandular formation
Comment: There is neoplastic progression of proliferative responses from atypical tubule hyperplasia through to adenoma and carcinoma (Dietrich and Swenberg 1991). Basophilic adenomas of lobular organization are by far the most common type in rats and mice. In the mouse, the growth pattern is often papillary or cystopapillary (Shinohara and Frith 1980; Alden et al. 1992; Hard et al. 2001b). An amphophilic, vacuolar type of adenoma is of sporadic occurrence in the rat and has been determined to be of spontaneous origin (Hard et al. 2008), but chromophobe adenoma can be induced by chemical treatment (Bannasch and Ahn 1998b).
Oncocytoma (Figures 71 and 72): Kidney
Species: rat
Synonyms: oncocytic adenoma, acidophilic adenoma, oxyphilic adenoma
Pathogenesis/cell of origin Origin from collecting tubules has been demonstrated in this species (Nogueira and Bannasch 1988); it is preceded by oncocytic hyperplasia
Diagnostic features Small, solid masses in outer zones of kidney Monomorphic population of oncocytes Compression of surrounding tubules with tubular contortion and/or altered growth pattern within the mass Oncocytic cells have finely granular, pale to faintly eosinophilic, cytoplasm and centrally located nuclei with indistinct nucleoli May be encapsulated, but this is inconsistent Oncocytic cells stain positively for cytochrome-c-oxidase (Mayer et al. 1989) and ultrastructurally, the main cytoplasmic feature is a dense crowding of atypical mitochondria (Krech et al. 1981)
Differential diagnoses Oncocytic hyperplasia: oncocytic lesion consists of small number of tubule profiles consistent with convolutions of a single tubule entity Chromophobe adenoma: cell borders prominent and well defined Renal adenoma: well-defined borders; variable morphology, but lack of oncocytic differentiation
Comment: Oncocytoma appears to be a benign end-stage lesion that does not progress into carcinoma, and metastases have not been reported (Bannasch et al. 1998a; Nogueira and Bannasch 1988; Montgomery and Seely 1990). Oncocytic hyperplasia is a proliferative lesion that is difficult to separate from oncocytoma. Some oncyocytomas have abnormal or irregular morphologies which makes differentiation easier, but any oncocytic lesions larger than 3 times the size of glomeruli should be considered an oncocytoma. Additional criteria for oncocytoma diagnosis include total circumferential compression with or without evidence of partial encapsulation, altered growth pattern, and/or loss of normal tubule appearance within the lesion. Oncocytic hyperplasia and oncocytomas should be scored separately from other types of renal tubule hyperplasia or adenoma, because of their different renal segment of origin, distinctive morphology, and lack of clinical significance for risk assessment.
Carcinoma (Figures 73–75): Kidney
Species: rat, mouse
Synonyms: adenocarcinoma, renal tubule carcinoma, renal cell carcinoma, malignant renal epithelioma
Pathogenesis/cell of origin Presumed to originate mainly from proximal tubules (Hard 1998c); origin from distal nephron is also possible, particularly for clear cell tumors (Nogueira et al. 1989)
Diagnostic features Circumscribed or irregular, fleshy, nodular growths Growth is usually by expansion with the exception of anaplastic variants, which can be infiltrative and expansive (Montgomery and Seely 1990; Hard 1990; Hard 1984, Hard 1985a) Evidence of cell, and sometimes nuclear, pleomorphism Usually multiple necrotic or hemorrhagic areas and well-developed vascular arborization Scattered or conspicuously increased mitotic activity
Rat Cell staining is usually basophilic, but eosinophilic, amphophilic, clear cell, chromophobic, or mixed cell variants can occur Cell arrangement can be tubular, lobular, papillary, solid, or mixed Chromophobic cell tumors are characterized by finely vacuolated cytoplasm and prominent cell borders
Mouse Cell staining is usually basophilic, but can be eosinophilic, clear or mixed (Seely 1999) Cell arrangement can be solid, papillary, anaplastic, or mixed (Nogueira et al. 1989; Shinohara and Frith 1980; Sass 1998) Anaplastic variants appear to be more common than in the rat
Differential diagnoses Adenoma: only a few millimeters, relatively well differentiated, lacks multiple areas of degeneration; no, or only slight, cell pleomorphism and no evidence of invasion or metastasis (Frith, Terracini, and Turusov 1994) Nephroblastoma: characterized by presence of highly basophilic, densely packed, blast cells Secondary metastases arising from neoplasia in distant epithelial organs: Usually infiltrate between and sequester tubules and glomeruli toward periphery of tumor mass; glandular structures may be present; may be more than one metastasis in available kidney sections; growth may appear to have arisen from the kidney surface or invaded through the renal hilus, or be perivascular)
Comment: Renal cell tumors and tubular precursor lesions may occur spontaneously, particularly in old animals, but a significant increase in the incidence and number produced by a test compound could indicate the potential for carcinogenicity. There is neoplastic progression of proliferative responses that develop from atypical tubule hyperplasia through to adenoma and carcinoma. Anaplastic variants appear to be more common in the mouse than in the rat. In rats, basophilic carcinomas of lobular organization are by far the most common type. Clear cell (the most frequent type in humans) and papillary types occur in the rat, but much less frequently. Clear cell carcinomas lack staining because of high content of lipid and/or particulate glycogen. Anaplastic and sarcomatoid variants are rare (Alden et al. 1992; Hard et al. 2001a). Tumors attaining dimensions approaching 2 cm have a relatively high rate of metastasis to the lungs. The Eker rat model has a predisposition to renal tumors including carcinoma and has a genomic mutation in the TSC2 gene (Hino et al. 1999; Laping et al. 2007).
An amphophilic, vacuolar type of carcinoma in the rat occurs sporadically and has been shown to be spontaneous (Hard et al. 2008). Like oncocytoma, these tumor types should be placed in a separate category from the basophilic tumor type.
Chromophobe adenoma/carcinoma is a variant of renal tubule tumor that has clear-to-granular cytoplasm with very well-defined cell borders and central nuclei (Bannasch and Ahn 1998b). This neoplasm can occur as a result of chemical treatment.
Nephroblastematosis (Figure 76): Kidney
Species: rat
Synonyms: nephroblastomatosis, blastemal rest, nephrogenic rest
Pathogenesis/cell of origin Origin from a remnant of developing metanephric blastema is presumed
Diagnostic features Small, solitary, basophilic cell mass consisting of densely crowded blast cells with ill-defined cytoplasm and basophilic nuclei Invariably located in outer stripe of outer medulla infiltrating between pars recta tubules May be a few signs of early organoid differentiation into epithelial rosettes Mitoses in blast cells occasional and also in adjacent pars recta tubules
Differential diagnoses Foci of lymphocyte accumulation: Foci are more discrete and composed of mononuclear inflammatory cells; no early epithelial rosette formation Nephroblastoma: Nephroblastomas are larger, usually include organoid differentiation such as primitive, basophilic tubules; invasion of the cortex and occasionally renal pelvis; often have clusters of blast cells around mature ducts which may be extensions of the renal pelvis lining
Comment: This spontaneous lesion appears to be encountered sporadically in specific colonies of rats, suggesting a predisposing genetic basis. Foci of nephroblastematosis appear to have the potential to develop into nephroblastomas as they enlarge, and may be regarded as preneoplastic lesions; the distinction between nephroblastematosis and nephroblastoma appears arbitrary and would probably rest on size and degree of organoid differentiation (Beckwith, Kiviat, and Bonadio 1990; Mesfin 1999). Mitotic activity in the pars recta tubules appears to be an autocrine response to the blastema.
Nephroblastoma (Figures 77 and 78): Kidney
Species: rat, mouse
Synonyms: embryonal nephroma, Wilms’ tumor (human nomenclature)
Pathogenesis/cell of origin Origin from the metanephric blastema is presumed
Diagnostic features
Rat Circumscribed, fleshy growth; discrete clusters of highly basophilic blast cells, sometimes surrounding mature ducts; blast cells can also be arranged in trabecular, alveolar, papillary, or rarely, fascicular or cylindromatous patterns (Cardesa and Ribalta 1998; Hard 1985b; Hard and Grasso 1976; Turusov, Alexandrov, and Timoshenko 1980) Organoid differentiation is usually present as epithelial rosettes, primitive basophilic tubules, attempted glomerulus formation, or mature epithelial ducts Stroma varies from delicate areolar tissue to well-developed fibrous tracts; mitotic activity frequent in blast cell clusters and primitive tubules.
Mouse Basophilic, undifferentiated blast cells in clusters or dispersed in loose reticular sheets; primitive tubule-like structures sometimes present Stroma may be conspicuous
Differential diagnoses: Renal mesenchymal tumor: Consists of neoplastic connective tissue elements such as fibroblast-like spindle cells, which sequester preexisting renal tubules; entrapped tubules frequently exhibit simple hyperplasia Papillary variant of renal tubule tumor: Papillary extensions of neoplastic tubule epithelial cells into epithelial sacs mimic, but should not be confused with, attempted glomeruli formation
Comment: Nephroblastomas have been induced in the rat only with genotoxic chemicals, usually by transplacental, prenatal exposure (Turusov, Alexandrov, and Timoshenko 1980; Mesfin and Breech 1992; Mesfin and Breech 1996; Jasmin and Riopelle 1970; Hottendorf and Ingraham 1968; Hard and Noble 1981; Hard and Grasso 1976; Hard 1985b; Cardesa and Ribalta 1998). This neoplasm has probably been overdiagnosed in the rat because of confusion with renal mesenchymal tumor (Seely 2004). Nephroblastoma should be regarded as a malignant neoplasm even though metastases are infrequent. The pathognomonic features of rat nephroblastoma are a highly basophilic blastema and attempted organoid differentiation along the epithelial pathway into nephric elements. Tumor growth is often associated with chronic inflammatory reaction in the surrounding compressed parenchyma. Nephroblastoma in the mouse is exceedingly rare and only a few cases have been recorded. This tumor appears to be morphologically similar to that observed in the rat, but some cases have shown a predominance of loose, reticular sheets of blastemal cells with little organoid differentiation. Organoid differentiation into structures resembling primitive avascular glomeruli has been described in one case. In contrast to experience with the rat, there is no unequivocal evidence of chemical induction of this tumor in mice (Seely 1999). The human term of Wilms’ tumor is a synomym for human nephroblastoma, but should not be used for rodent nephroblastoma.
Renal Mesenchymal Tumor (Figure 79): Kidney
Species: rat
Synonyms: stromal nephroma, interstitial cell tumor of kidney, mixed malignant tumor of kidney, malignant mesenchymal cell tumor
Pathogenesis/cell of origin Cell of origin from foci of atypical fibroblast-like cells in the interstitium of the outer stripe of outer medulla (Hard and Butler 1970a; Sunter and Senior 1983) With growth, the foci infiltrate around and sequester preexisting tubules, which tend to survive within the tumor mass
Diagnostic features Single or multiple lesion with poor demarcation, irregular shape, and infiltrative growth; malignant Growth is by infiltration replacing parenchyma. Cyst formation may be prominent Small tumors have a fibrous texture Large tumors are multi-loculated, cystic, gelatinous, and haemorrhagic Heterogeneous connective tissue cell composition but predominantly spindle cells with some stellate cells and smooth muscle fibers; occasionally, rhabdomyoblasts, striated muscle, cartilage, osteoid, or hemangiosarcomatous areas may be present (Dezso et al. 1990; Hard 1998b; Hard and Butler 1970b) Stellate cells resemble primitive mesenchyme or myxomatous tissue Collagen deposition in atypical clumps is a characteristic feature and reticulin is prominent Dense whorls of “onion skin-like” spindle cells layers may encircle tubule profiles; mitotic figures quite frequent in spindle cell areas Fibrosarcoma-like sheets of fibroblastic cells may be present featuring herring-bone growth pattern Tubule profiles, or nests of transitional epithelium, and these epithelial structures may become hyperplastic or metaplastic
Differential diagnoses Nephroblastoma: Embryonal tumor consisting of epithelial blast cells, primitive tubules, and sometimes, primitive glomerular bodies; in contrast to renal mesenchymal tumor, which is a neoplasm of connective tissue, this is basically an epithelial neoplasm Liposarcoma: Grossly similar to renal mesenchymal tumor, but consists of a mixture of mature fat cells, lipoblasts, and poorly differentiated mesenchymal cells; sequestered, preexisting renal epithelial elements tend to become cystic and atrophic Renal sarcoma: Dense monomorphic population of basophilic spindle cells with almost no collagen deposition and no sequestering of preexisting tubules
Comment: Renal mesenchymal tumor has frequently been misdiagnosed as nephroblastoma because of the presence of preexisting tubules which not only survive within the tumor tissue, but can become hyperplastic and/or metaplastic (Seely 2004; Turusov, Alexandrov, and Timoshenko 1980). This is probably due to signaling influences of the mesenchymal tumor cells. In the rat, renal mesenchymal tumor is a rare spontaneous malignant tumor and has been induced only by potent genotoxic chemicals. Metastases from spontaneous tumors are rare, but observed in tumors induced experimentally. There is no real distinction between benign and malignant forms, as this tumor has the potential to progress to a life-threatening size. The hallmark of renal mesenchymal tumour is the heterogeneous spectrum of connective tissue cell types that represent the multipotentiality of the stem cell of origin. Sequential studies have shown origin from foci of atypical fibroblast-like cells in the interstitium of the outer stripe of outer medulla (Hard and Butler 1970a). Mesenchymal tumors are extremely rare in the mouse and have not been well characterized.
Renal Sarcoma: Kidney
Species: rat, mouse (less common)
Synonyms: renal fibrosarcoma
Pathogenesis/cell of origin Cell of origin and histogenesis are not known
Diagnostic features Composed of a monomorphic population of densely packed, fibroblast-like, highly basophilic spindle cells; collagen deposition is absent or not conspicuous (Montgomery and Seely 1990) Preexisting tubules and glomeruli may be seen at the periphery but are absent from most of the tumor mass Fascicular pattern sometimes present; mitotic figures sporadic to frequent Growth is infiltrative, replacing parenchyma Entrapped preexisting tubules and glomeruli may be seen at the periphery but are absent from most of the tumor mass Cells are arranged in densely packed sheets or forming interlacing bundles, associated with fine collagen fibers Fascicular pattern is sometimes present Nuclei are large, fusiform, often possessing several nucleoli Mitotic figures are present Areas of hemorrhage and necrosis may be present
Differential diagnoses Renal mesenchymal tumor: Less cellularly dense with sequestered preexisting epithelial elements scattered throughout; collagen deposition is conspicuous Liposarcoma: Composed of admixture of mature and immature fat cells and undifferentiated mesenchyme
Comment: Renal sarcoma is a spontaneous, but rare tumor in laboratory rats and mice. A similar tumor can be induced experimentally with polyoma virus in rats (less than 7 days old) and mice, and early foci arise within the outer stripe of outer medulla (Flocks et al. 1965; Ham and Siminovitch 1961; Prechtel, Zobl, and Georgii 1967). Renal sarcoma has also been induced by intrarenal administration of 20 methylcholanthrene. Virus-induced renal sarcomas develop rapidly after inoculation, and progressively invade and replace the renal tissue (Stevenson and Von Haam 1962).
INHAND Nomenclature: Lower Urinary Tract
Nonproliferative Lesions of the Lower Urinary Tract
Aplasia, Ureteral: Ureter
Species: rat, mouse
Synonyms: agenesis
Pathogenesis/cell of origin Congenital absence of ureter
Diagnostic features Noted macroscopically as a missing ureter Generally unilateral, kidney missing as well
Comment: This is an uncommon to rare finding in the rodent. Agenesis more commonly refers to the macroscopic absence of the ureter, while aplasia is the more common histologic term.
Dilation, Pelvis: Renal Pelvis
Species: rat, mouse
Synonyms: drug-induced pelvic dilation, iatrogenic renal pelvic dilation, congenital pelvic dilation
Pathogenesis/cell of origin Can occur secondary to parenchymal damage and inflammation from outflow obstruction Pathogenesis remains obscure in cases where there is no relationship to crystalluria, pyelonephritis, or obstruction Acquired and congenital forms: often difficult to distinguish other than by age Unilateral or bilateral In rats, it is usually found on right side, as a result of compression of the ureter by the overlying spermatic or ovarian artery Can be transient, especially in congenital forms (Sellers et al. 1960) Associated with the administration of some drugs, especially in reproductive toxicology studies or juvenile toxicity studies (Fujita et al. 1979)
Diagnostic features Dilatation of the renal pelvis with or without evidence of inflammation or parenchymal degeneration Lining urothelium may be hyperplastic Varies from mild-to-marked dilatation of the renal pelvis and may be accompanied by calculi, hemosiderin pigment, or drug crystals In chronic cases may get compression of parenchyma with tubule atrophy, decreased glomeruli and glomerulosclerosis, and may progress to complete cortical atrophy (Gobe and Axelsen 1987) In severe cases, may get renal tubular damage, sclerosis and scarring of cortex and medulla due to secondary effects of outflow obstruction
Differential diagnosis Incidental dilatation of the renal pelvis: no evidence of damage from outflow obstruction
Comment: Pelvic dilation is the correct histologic diagnosis while the correct macroscopic term for this lesion is hydronephrosis. These terms are inclusive of both congenital and iatrogenic forms of pelvic dilation.
Dilation (Figure 80): Ureter
Species: rat, mouse
Synonyms: dilatation, ureter, hydroureter (macroscopic term)
Pathogenesis/cell of origin Congenital, inherited, or acquired Often noted with pelvic dilation/hydronephrosis
Diagnostic features Can be noted macroscopically as thickened ureter In the rat, it is most often observed on the right-hand side May be unilateral or bilateral
Comment: Although more commonly congenital, this condition may occur as a sequela to drug-induced or spontaneous urolithiasis. Ethanol-induced hydroureter was observed during in vivo exposure in mouse embryos in order to study Fetal Alcohol syndrome (Gage and Sulik 1991).
Infiltrate, Inflammatory Cell: Ureter, Urethra, Renal Pelvis
Species: rat, mouse
Synonyms: ureteritis, cystitis, urethritis, pyelitis
Pathogenesis/cell of origin Inflammatory infiltrates are commonly noted in the submucosa of the renal pelvis in rodents as a spontaneous background finding of no biologic significance, but when due to ascending bacterial infection, may be severe Inflammation of the ureters and renal pelvis usually results from ascending lesions of the urinary bladder. Pelvic inflammation may also be a result of descending infection from pyelonephritis Focal to diffuse lymphocytic infiltrates are commonly observed in subepithelial areas lining the renal pelvis
Diagnostic feature The type of inflammation depends upon the main component of the lesion Inflammation may be acute, subacute, chronic, necrotizing, or granulomatous fibrinous, necrotic, or ulcerative Hemorrhage often occurs with inflammation. Inflammation may be observed as a part of a neoplastic process Hyperplasia of the urothelium often accompanies inflammation In acute to chronic cases, chronic inflammatory cells are present in the subepithelial sites and acute suppuration noted in the lumen
Differential diagnoses None. Should be easily differentiated from lymphoma, which is a monomorphic population of lymphoid cells
Comment: Inflammation of the ureter or renal pelvis occurs most commonly as an ascending bacterial infection, but may also result from descending infections (pyelonephritis) or as sequela to urolithiasis or hydroureter (Seely 1999). Bacteria involved in the pathogenesis are usually those that are commonly isolated from other parts of the rodent urinary tract including E. coli, Proteus, or Staphylococcus species, and so on. In rodents, particularly rats, foci of extramedullary hematopoiesis may be observed in the fatty tissue present in the renal hilus which may resemble inflammation on low power. Renal papillary necrosis is most often seen with pyelitis/pyelonephritis (Burek et al. 1988). Inflammation of the renal pelvis may extend into the collecting ducts and ascend further into the renal parenchyma (Montgomery 1998; Duprat and Burek 1986). A case with inflammation and necrosis of the renal pelvic urothelium occurred with a 2-week administration of trimethyl imidazopyrazolopyrimidine (Macallum and Albassam 1994).
Erosion: Urinary Bladder, Renal Pelvis
Species: mouse, rat
Synonyms: attenuation, urothelium
Pathogenesis/cell of origin Superficial necrosis or degeneration of the urothelium without exposure of the basement membrane
Diagnostic features Focal to multifocal incomplete loss of urothelium without loss of basement membrane or exposure of subepithelial tissue
Differential diagnoses Ulceration: Necrosis of the urothelium which progresses through the entire epithelial layer and is associated with acute inflammation and hemorrhage
Comment: Associated with many of the same causes of ulceration to include toxicants, inflammation, Trichosomoides crassicauda, crystalluria, and calculi.
Ulceration (Figure 81): Urinary Bladder, Renal Pelvis
Species: rat, mouse
Synonyms: necrosis, urothelium
Pathogenesis/cell of origin Necrosis of the urothelium which progresses through the entire epithelial layer and is associated with acute inflammation and hemorrhage Many of the causes of ulceration also result from similar reasons as noted for inflammation
Diagnostic features In general, focal to multifocal complete loss of the urothelium which exposes the subepithelial tissue Associated with acute inflammation and hemorrhage; in chronic cases, fibrosis is evident in subepithelial tissue
Differential diagnosis Erosion: Superficial necrosis or degeneration of the urothelium without exposure of the basement membrane
Comment: Ulceration of the renal pelvis is an uncommon lesion. It may be seen with toxicants or as a secondary event associated with inflammation or the presence of calculi.
Vacuolation, Urothelium: Urinary Bladder
Species: rat, mice
Synonyms: none
Pathogenesis/cell of origin Is considered as a nonspecific lesion noted following exposure to a number of chemicals toxic or carcinogenic to the bladder epithelium Also noted as an autolytic change
Diagnostic features Variably sized vacuoles usually within superficial epithelial cells of the urothelium
Differential diagnoses Inclusions: may be clear when washed out with tissue processing, but usually some eosinophilic inclusions remain
Inclusions, Urothelium (Figure 82): Urinary Bladder
Species: mouse
Synonyms: inclusion bodies, inclusions, transitional epithelium
Pathogenesis/cell of origin May represent degenerative cell components, lipid, or accumulation of the test chemical or metabolite
Diagnostic features Eosinophilic or clear inclusions (washed out from tissue processing) noted in the superficial (umbrella cells), less commonly in the intermediate or basal cell layers No other evidence of cellular pathology
Differential diagnoses Vacuolation: clear spaces rather than solid and found throughout the urothelium rather than confined to umbrella cells
Comment: Inclusions in the urinary bladder are extremely common in mice and may occur in controls, but may also be associated with drug administration. They do not represent an association with preneoplasia or neoplasia (Cohen 2002). In one report of inorganic arsenic administration, small, eosinophilic inclusions were found to represent intramitochondrial granules (Suzuki et al. 2008). Similar inclusions have been seen with exposure to other metals and may play a protective role.
Calculi (Figures 83 and 84): Urinary Bladder, Ureter, Renal Pelvis
Species: rat, mouse
Synonyms: urolithiasis, concretion, calculosis
Pathogenesis/cell of origin Can be formed spontaneously or induced by chemicals
Diagnostic features Single or multiple, size varies; color varies depending on composition Usually lying free in lumen Microscopically, concentric layering of basophilic or eosinophilic material, depending on composition Often noted in association with chronic inflammation and transitional cell hyperplasia Range in size from sand-like to large calculi which may obstruct the neck of the urinary bladder The presence of calculi may be associated with necrosis and/or ulceration of the epithelium
Differential diagnoses Crystals—smaller, discrete particles; no concentric layering
Comment: As with tubular crystalluria, compounds or their metabolites in high concentrations and of low solubility may precipitate in the pelvis, ureters or bladder, depending on urinary pH and mineral composition (Yarlagadda and Perazella 2008). Calculi may be produced by a number of chemicals or their metabolites, or calculi can be formed by precipitation of normal urinary constituents by altering the composition and physiology of the urine (Cohen 2002). Xenobiotic-induced calculi are often a complex mixture of minerals and can consist of drug/mineral complexes or separately of magnesium phosphate, calcium phosphate, or calcium oxalate. In the rat, calculi are usually composed of calcium phosphate and to a lesser degree magnesium ammonium phosphate (Hard et al. 1999; Montgomery and Seely 1990). Similar calculi (struvite) in mice have also been reported (Wojcinski et al. 1992). Calculi may arise spontaneously in rodents, and those not associated with xenobiotics are more common in the bladder than in the renal pelvis (Peter, Burek, and Van Zwieten 1986). Rough surface features of calculi and constant irritation to the adjacent pelvic urothelium often leads to concurrent urothelial hyperplasia and the proliferative response may result in an increased incidence of neoplasia in rodents in chronic studies (Clayson 1974; Bach and Bridges 1985b). In aged rats, mineral deposits may form spontaneously under the renal papillary epithelium and be associated with degeneration, ulceration, or inflammation of the overlying urothelium. These can be found along the urothelial basal lamina, at the base of the papilla or along the fornix, and sometimes are noted freely in the pelvis (Greaves 2007). The presence of chronic physical irritation from calculi has been associated with the development of bladder neoplasia (Fukushima and Murai 1999; Jull 1979).
Crystals: Urinary Bladder, Ureter, Renal Pelvis
Species: rat, mouse
Synonyms: urolithiasis
Pathogenesis/cell of origin Crystalluria is a normal finding in rodent urine. Under predisposing conditions, crystals may precipitate out of the urine and result in local irritation.
Diagnostic features Crystals may or may not be evident in tissue sections by microscopic examination because they may be washed out by tissue preparation May result in urinary bladder epithelial hyperplasia and mucosal inflammation
Differential diagnosis Calculi, calculosis—large concretions usually mineralized
Comment: Strain-related differences in urine composition may affect the amount of urine protein, lithogenic ions, citrate (inhibitor of lithogenesis), and/or volume (Tannehill-Gregg et al. 2009). Strain differences have been shown in studies with peroxisome proliferator-activated receptor (PPAR) agonists and bladder tumor development with urolithiasis as an inciting event. Crystals are nearly always present in the urine of rats, even in controls, and are usually composed of magnesium ammonium phosphate. The precipitation of calcium phosphate and relationship of sodium saccharin to bladder cytotoxicity, increased cell proliferation, and carcinogenicity have been shown to be a rat-specific phenomenon (Cohen 1999).
Edema: Urinary Bladder
Species: rat, mouse
Synonyms: none
Pathogenesis/cell of origin
Usually accompanies inflammation, is a vascular disturbance, or of unknown etiology
Diagnostic features The presence of an amorphous slightly eosinophilic fluid in the subepithelial connective tissue layer of the bladder
Differential diagnoses Artifact from formalin injection
Comment: The pathogenesis of edema is similar to that in other tissues and results from increased capillary leakage in the submucosal vessels as a result of inflammatory cytokine influence or pharmacologic effects by drugs.
Infiltrate, Inflammatory Cell (Figures 85 and 86): Urinary Bladder
Species: rat, mouse
Synonyms: cystitis, inflammation
Pathogenesis/cell of origin Inflammation of the bladder may result from a number of factors including bacterial infections, urinary solids, toxicants, or the presence of tumors
Diagnostic features The presence of inflammatory cells or other characteristics of inflammation such as congestion, hemorrhage, necrosis, and ulceration Inflammatory cells may be present in any area of the bladder or observed in the bladder lumen. Inflammation may be acute, subacute, chronic, necrotizing, granulomatous, or ulcerative
Differential diagnoses Lymphoma/leukemia/histiocytic sarcoma: homogenous infiltrate of mononuclear cells, not usually well differentiated; uncommon in the rodent bladder as compared to other tissues
Comment: The spectrum of inflammatory lesions in the urinary bladder of mice has been previously well characterized (Frith 1979).
Necrosis: Urinary Bladder
Species: rat, mouse
Synonyms: none
Pathogenesis/cell of origin Results from cytotoxicity to the urothelium Observed with urinary solids or as a response to chemical toxicants
Diagnostic features Cellular eosinophilia, fragmentation, and nuclear pyknosis and karyorrhexis
Differential diagnoses: postmortem autolysis
Comment: Epithelial cell necrosis results in regenerative hyperplasia which may be reversible (Gopinath, Prentice, and Lewis 1987).
Hemorrhage: Urinary Bladder
Species: rat, mouse
Synonyms: none
Pathogenesis/cell of origin Hemorrhage can occur from inflammation, ulceration, and the presence of calculi or tumors
Diagnostic feature Blood-tinged urine on gross necropsy The presence of extravasated red blood cells Usually noted with some other lesion such as inflammation, calculi, or neoplasia
Differential diagnoses Congestion
Comment: Hemorrhage is a hallmark of acute injury to the urinary bladder from any cause. With subacute injury it may be accompanied by hemosiderin pigment or the presence of macrophages with intracellular phagocytosed debris.
Angiectasis: Urinary Bladder
Species: rat, mouse
Synonyms: dilatation, blood vessel
Pathogenesis Spontaneous lesion or secondary to vasodilating chemicals
Diagnostic features Dilation of blood vessels Solitary or multiple Usually noted in submucosal vessels
Differential diagnosis Hemangioma: an organized network of vascular channels and spaces lined by variably differentiated endothelial cells and lumina that are usually filled with erythrocytes versus angiectasis which are usually just a cluster of vessels lined by well-differentiated endothelium
Comment: Angiectasis is noted more often in mice than in rats. It has little pathological significance.
Dilation: Urinary Bladder
Species: rat, mouse
Synonyms: none
Pathogenesis/cell of origin Dilation can arise spontaneously without an obvious cause or results from sustained increased urine volume secondary to diuretics or from blockage of the urethra
Diagnostic features Dilation of the bladder lumen; usually appears empty since contents are washed out during histologic preparation The bladder wall is thin and the epithelium is reduced in thickness Inflammation may or may not be present
Differential diagnoses formalin artifact
Comment: Occasionally formalin is injected into the urinary bladder at the time of necropsy to better inflate and fix the bladder. Overinflation of the bladder in this way may result in misinterpretation of artifact as dilation (Cohen et al. 2007).
Mineralization (Figure 87): Urinary Bladder
Species: rat, mouse
Synonyms: dystrophic calcification, nephrocalcinosis
Pathogenesis/cell of origin Mineralization may be observed in areas of previous degeneration and/or necrosis Uncommon lesion in rodents
Diagnostic features Observed as basophilic, granular deposits in connective tissue or muscle wall
Differential diagnoses Calculi: limited to the lumen
Comment: As with mineralization in other rodent tissues, bladder mineralization is often associated with disturbances in calcium:phosphorus ratios (Ritskes-Hoitinga and Beynen 1992; Nicoletta and Schwartz, 2004).
Uropathy, Obstructive: Urinary Bladder
Species: mouse
Synonyms: obstructive syndrome, mouse urological syndrome (MUS)
Pathogenesis/cell of origin Inflammatory material and proteinaceous plug which blocks outflow of urine from the bladder Bacterial infection commonly noted The etiology seems multifactorial; hormonal, fighting among males, urethral plugs from secondary sex glands, infectious agents, self-mutilation, dehydration, and injury from wire caging
Diagnostic features Grossly may observe paraphimosis, cutaneous ulceration, distended urinary bladder, and hydroureter/hydronephrosis Ulcerative to inflammatory balanoposthitis Proteinaceous material containing inflammatory cells; spermatozoa or desquamated urothelial cells forming a proteinaceous plug in the urethra In chronic cases, bladder calculi may be present. Bacteria present in most cases Dilatation of the bladder, hydroureters, and hydronephrosis are often present
Differential diagnoses Cutaneous ulceration/inflammation of the preputial gland and/or area Calculi: in the case of obstructive uropathy, if calculi are present they are a result of, rather than a cause of the obstruction and are not generally found within the urethra
Comment: Obstructive uropathy was reported in 1962 (Sokoloff and Barile 1962) and again in 1965 (Babcock and Southam 1965). It is still considered a frequent cause of spontaneous mortality in long-term studies (Everitt, Ross, and Davis 1988). Better colony management and single housing of male mice has reduced the incidence of obstructive uropathy in mice. An increased incidence has been seen with wire caging (Bendele 1998). It has been reported to be prevented by antibiotics and ochratoxin A (Bendele and Carlton 1986).
Diverticulum: Urinary Bladder
Species: rat, mouse
Synonyms: none
Pathogenesis/cell of origin Congenital or related to persistent urethral obstruction or from chronic inflammatory lesions.
Diagnostic features Downgrowth of epithelial lined out-pocketings into muscular wall or adventitia
Differential diagnosis Carcinoma, transitional cell: cellular atypia and clustering along the lining
Comment: Diverticuli are rarely observed as spontaneous lesions. They may be predisposed to inflammation. May be mistaken as transitional cell carcinomas but epithelium is normal appearing without dyplasia or atypia (Frith, Terracini, and Turusov 1994).
Hypertrophy, Urothelium: Urinary Bladder
Species: rat, mouse
Synonyms: hypertrophy, transitional cell
Pathogenesis/cell of origin Urothelium undergoes increase in cytoplasmic volume due to unknown mechanism
Diagnostic features: Increased cell size
Differential diagnoses Cytomegaly
Comment: Urothelial hypertrophy due to the direct effect from a PPAR agonist has been reported in vivo in the rat (Oleksiewicz et al. 2005). Urothelial hypertrophy is a possible precancerous lesion (Lawson, Dawson, and Clayson 1970).
Proteinaceous Plug (Figures 88 and 89): Urinary Bladder, Urethra
Species: rat, mouse
Synonyms: none
Pathogenesis/cell or origin May occur as a common postmortem finding and represent an agonal secretion from the accessory sex glands occurring during euthanasia, especially in rats.
Diagnostic features Eosinophilic proteinaceous material in male rodent bladder or urethra In mice, may be intermingled with exfoliated urothelial cells Occasionally contains spermatozoa Can be very large and partially fill lumen of urinary bladder
Differential Diagnoses calculi: largely contain mineral rather than protein and do not contain spermatozoa; often associated with secondary inflammation or ulceration
Comment: This is an incidental finding of no clinical or toxicologic importance and its notation should be at the discretion of the pathologist. It is not considered a precursor of urinary calculi (Hard et al. 1999).
Nematodiasis (Figure 90): Urinary Bladder
Species: rat: Trichosomoides crassicauda (naturally occurring); schistosomiasis (experimentally produced)
Synonyms: parasitism, Trichosomoides crassicauda
Pathogenesis/cell of origin Transmission of the parasite may occur from improperly cleaned cages and soiled bedding material resulting in exposure to ova and larvae from the urine of other infected rats
Diagnostic features Sections of nematodes in the bladder lumen or attached to the mucosa Adult female worms are 0.2 mm in diameter and have a body cavity with cuticle; intestine and reproductive glands containing ova may be visible in section; males are much smaller than females (1/8th of their length) May be accompanied by erosion or urothelial hyperplasia, but inflammation of the bladder is uncommon Concomitant urolithiasis is occasionally present Migrating larvae may be present in other organs including kidney and lung and may be associated with granulomatous inflammation
Differential diagnoses Calculi
Comment: Because of improved laboratory animal housing practices, parasitism with the urinary bladder roundworm, Trichosomoides crassicauda is rarely encountered, except in wild rodent populations. Chronic infection with the rat bladder worm may predispose animals to transitional cell carcinoma or other urothelial tumors (Barthold 1986). Papillary hyperplasia and overt papillomas of the bladder commonly accompany this parasite, but are thought to arise from the associated urolithiasis that occurs, rather than from direct effects of the nematode (Serakides et al. 2001).
Obstruction: Urethra
Species: rat, mouse
Synonyms: occlusion, urethra
Pathogenesis/cell of origin May occur secondary to urolithiasis, proteinaceous plugs, or with stricture following chronic inflammation in the urethra
Diagnostic features Proteinaceous plug often noted containing desquamated epithelial cells, inflammatory cells, and spermatozoa Occurs with calculi Obstruction may be difficult to observe and caused by a relatively small region of hyperplasia/neoplasia or calculi anywhere along the urethra
Differential diagnoses Obstructive uropathy: outflow obstruction occurs in the bladder of mice as part of the mouse urologic (urinary) syndrome and is not due to calculi
Comment: Blockage by proteinaceous plugs or calculi is seen with obstruction of the urethra.
Infiltrate, Inflammatory Cell: Urethra
Species: rat, mouse
Synonyms: urethritis
Pathogenesis/cell of origin Usually occurs from some kind of trauma to the penis/prepuce in which bacterial-associated inflammation extends into the urethra
Diagnostic features Acute to chronic inflammatory lesions
Differential diagnoses Hematogenous neoplasms: uniform population of less differentiated cell population
Comment: Inflammation in the urethra is generally an ascending infection from the distal urogenital tract and unrelated to xenobiotic treatment.
Proliferative Lesions of the Lower Urinary Tract
Hyperplasia, Urothelium (Figures 83, 86, 91–96): Ureter, Urinary Bladder, Urethra, Renal Pelvis
Species: rat, mouse
Synonyms: hyperplasia, transitional cell
Pathogenesis/cell of origin Arises from the urothelial lining of the ureter, bladder, and a portion of the urethra Regenerative hyperplasia results from erosion, ulceration, and or necrosis of the epithelium
Diagnostic features: An increased number of cells with or without cellular atypia Frequently observed with inflammation of the bladder May be recorded as focal, diffuse, and characterized as simple, nodular, or papillary Variable numbers of mitoses may be evident Solitary growth Focal, multifocal, or diffuse multicellular thickening of urothelial lining is present Cells may be of normal size, smaller, or larger than normal Increase in number of cells containing PAS-positive granules may be present Chronic inflammation or calculi may be present Growth pattern may be simple, papillary, or nodular Simple: Linear uniform thickening of the lining lacking prominent outward or inward focal growth. Several degrees of severity may be observed (see comments). Papillary: Delicate exophytic projection of lining into the lumen supported by a simple branching fibrovascular core. Short micropapillae are lined by epithelial cell layers of varying thickness. Nodular: Solid round or oval nests of transitional cells protruding outward into the lumen (exophytic), or inward as a downgrowth (endophytic). May occur in conjunction with either simple or papillary hyperplasia
With Atypia
Cellular and nuclear pleomorphism, hyperchromatism, and enlarged nucleoli are present
Arrangement of cells is irregular
Growth pattern is nodular, or may be papillary.
Cytoplasm is basophilic
Mitotic figures may be present
Without Atypia
Cells are uniform retaining the differentiated and relatively organized character of regular urothelium
Cytoplasm may be basophilic.
Differential diagnoses Papilloma, urothelial: Consists of uniform epithelial cells, exhibits complex branching of stalks; lesion is solitary, but may be multiple and larger in size Carcinoma, transitional cell (urothelial): Metastasis or invasion is present, demarcation is poor, and/or pronounced cellular atypia is present; hemorrhage and necrosis may be present. Carcinoma of the renal pelvis is uncommon in rodents.
Comment: The lining of the normal renal pelvis in rat consists predominantly of cuboidal epithelium over the papilla but continues as urothelium for the remainder of the renal pelvis. Urothelial hyperplasia can arise at any part of the renal pelvis surface. Hyperplastic lesions of the ureter are often confined to the distal third. They range from focal to diffuse. The various forms of urothelial hyperplasia occur as responses to bacterial infection, urinary tract toxins, and carcinogens, calculi, or in association with renal papillary necrosis. Chemical carcinogens which target the urinary tract can induce both forms of hyperplasia with and without atypia. The atypical form is regarded as a preneoplastic lesion. Hyperplasia must be carefully distinguished from an apparent increased cellularity secondary to folding and/or tangential sectioning when the bladder is fixed in a nondistended state. Hyperplasia has also been reported as a consequence of extensive handling in rats (Cohen et al. 1996). Hyperplasia often appears to extend downward into the submucosa, resulting from chronic cystitis (Shinohara and Frith 1981). Nodular or hyperplastic apparent downgrowths into the bladder can be misdiagnosed as neoplasms even when they do not have cellular atypia (Cohen 2002). Hyperplasia in these cases may be entirely reversible if the inciting agent is removed. Spontaneous transitional cell hyperplasia in the mouse is rare but there is a potential for its induction by chemicals causing injury to these cells. In addition, it could be a response to infective agents causing pyelonephritis, and may be secondary to lower urinary tract obstruction. Atypical hyperplasia should always be distinguished from urothelial hyperplasia without atypia because, when associated with bladder carcinogens, the atypical form is considered a precursor of epithelial bladder cancer. Atypical urothelial hyperplasia has been associated with genotoxic urothelial carcinogens such as N-butyl-N(4-hydroxybutyl) nitrosamine OH-BBN (Bach and Gregg 1988). Urothelial hyperplasia can show several degrees of severity. With increasing degree of hyperplasia, proliferative activity also increases. Nodular hyperplasia is comparable morphologically to von Brunn’s nests or cystitis cystica in humans. Although the areas of nodular hyperplasia often appear to have no connection with the surface epithelium, serial sections usually reveal such a connection. The distinction between papillary hyperplasia and nodular hyperplasia can be difficult in some cases, so modifiers are not always necessary or descriptive. Hyperplasia may regress if the etiologic stimulus is removed. Acute or chronic inflammation often accompanies hyperplasia in the urinary bladder. In rare cases, simple, papillary, and nodular hyperplasias exhibit squamous metaplasia with or without keratinization. Glandular metaplasia occurs far less commonly.
Metaplasia, Squamous Cell (Figures 97 and 98): Ureter, Urinary Bladder, Urethra, Renal Pelvis
Species: rat, mouse
Synonyms: none
Pathogenesis/cell of origin Urothelium which undergoes squamous metaplasia or replacement by squamous epithelium
Diagnostic features Often associated with hyperplasia The presence of focal to multifocal flattened squamous cells Variable keratinization or sometimes only keratohyaline granules present Squamous metaplasia may be minimal or markedly extensive Desquamation of keratin Replacement of urothelium by squamous cells Focal, multifocal, or diffuse metaplasia Nuclei are mostly round, oval, or flattened Cellular axes are parallel to basal membrane Sometimes nuclear polymorphism and cellular atypia are present Surface cells may be highly keratinized, nonkeratinized, or contain only keratohyaline granules Desquamation of cornified material is present
Differential diagnoses: Squamous cell papilloma/carcinoma: Growth pattern is exophytic; fibrovascular stroma is delicate; proliferative thickening of the squamous epithelium is prominent and mitotic figures may be present
Comment: Squamous metaplasia of the urinary bladder may occur as a response to the administration of bladder toxicants and carcinogens, vitamin A deficiency, or chronic inflammation. Keratinization can be so extensive that, on occasion, the entire lumen of the urinary bladder becomes filled with desquamated cornified material. Squamous metaplasia of the renal pelvis is much less common than in the urinary bladder. Squamous metaplasia may be followed by the development of transitional cell carcinomas with squamous differentiation and squamous cell papillomas and carcinomas.
Metaplasia, Glandular (Figure 98): Ureter, Urinary Bladder, Urethra
Species: rat, mouse
Synonyms: glandular cell metaplasia, mucinous hyperplasia, mucinous metaplasia
Pathogenesis/cell of origin Urothelium which undergoes glandular metaplasia
Diagnostic features Cuboidal to columnar epithelial cells Formation of glandular structures With or without mucus production No invasion or any signs of malignancy and absence of mitotic figures
Differential diagnoses Adenoma/adenocarcinoma: Cellular atypia, mitotic figures may be present, or evidence of invasion Metaplasia, squamous cell: Squamous cell structures and keratinization are present
Comment: Glandular metaplasia is a rarely observed spontaneous lesion. Urothelial hyperplasias can undergo focal or total metaplasia. When this occurs the transitional epithelium is usually replaced by squamous epithelium. Less commonly, columnar or glandular epithelium replaces the urothelium of the bladder, resulting in columnar or glandular metaplasia. This type of metaplasia may be observed within nodular hyperplasia. In rare instances, the glands are lined with highly columnar, mucus-secreting cells reminiscent of goblet cells.
Papilloma, Transitional Cell (Figures 99–102): Ureter, Urinary Bladder, Urethra
Species: rat, mouse
Synonyms: papilloma, urothelium
Pathogenesis/cell of origin Papillomas are benign neoplasms which arise from transitional cell epithelium
Diagnostic features Exophytic growth pattern into the bladder lumen which is pedunculated Less commonly, inverted (endophytic) pattern can be recognized as flattened lesions or have epithelial growth downward into the stalk of the mass of the urinary bladder (inverted papilloma) Cells are uniform and well differentiated Little cellular atypia and no evidence of invasion Mitotic figures are rare or absent Finger-like fibrovascular stalks lined by regular transitional epithelium which may be complex or branching, and with a fibrovascular core May be associated with inflammation Solitary or may be multiple Glandular metaplasia or squamous metaplasia may be present Squamous metaplasia may exhibit keratin formation Cytoplasm may be slightly more basophilic than in normal cells The individual cells are slender and are parallel to each other at right angles to the basement membrane
Differential diagnoses Hyperplasia, urothelial: Multifocal or diffuse, distinctly smaller in size, with simple thickening of the urothelial lining Carcinoma, transitional cell (urothelial): Metastasis or evidence of invasion; demarcation is poor and cellular atypia is present; mitotic figures may be numerous and hemorrhage or necrosis may be present
Comment: Tumors of the lower urinary tract have been shown to progress through hyperplasia to papilloma to noninvasive and invasive carcinomas (Cohen 1989). A new term in the human literature for this lesion is urothelial papilloma and over time will likely become the preferred term for this entity, but historically the term transitional cell papilloma has been used for this lesion and is still technically accurate and appropriate to use, and easily distinguishes the lesion from squamous cell papilloma. Since the biological behavior of the inverted type of urothelial papillomas seems to be the same as the common type of exophytic papilloma, both can be grouped as papilloma. However, distinguishing downgrowth of the inverted papilloma from true invasion may be difficult by light microscopy. Sometimes, exophytic papillomas can show a focal endophytic growth pattern which resembles that of inverted papilloma. The transitional cell papilloma is thought to be an intermediate step in the progression from normal urothelium to final development of carcinoma. Chemically induced papillomas of the urinary bladder usually consist of a solid mass of transitional epithelial cells with scant connective tissue, while spontaneously occurring papillomas tend to have a thin cover of urothelium over a fibrovascular stromal core.
Papilloma, Squamous Cell: Ureter, Urinary Bladder, Urethra
Species: rat, mouse
Synonyms: none
Pathogenesis/cell of origin May develop directly from transitional epithelium which undergoes squamous metaplasia
Diagnostic features Generally an exophytic growth pattern and single Usually highly differentiated squamous epithelium Absence of invasion into adjacent tissue Variable keratinization and desquamation of keratin into bladder lumen Ramifying squamous proliferations resting on a vascularized stalk of connective tissue Prominent thickening of the highly differentiated squamous epithelium Cytologic features similar to squamous cell metaplasia Cellular atypia and mitotic figures are not frequent Keratinized squamous epithelium may contain only keratohyaline granules or may be highly keratinized
Differential diagnoses Metaplasia, squamous cell: No or minimal exophytic growth, no prominent thickening of the squamous epithelium; typically, no vascularized stalk of connective tissue Carcinoma, squamous cell: Cellular atypia, loss of cellular polarity, frequent mitotic figures, or evidence of invasion is present
Comment: Squamous cell papilloma of the urinary bladder is a rare spontaneous tumor in rodents. It is composed exclusively of tumor cells with squamous differentiation lacking cellular abnormalities. The neoplastic squamous epithelium may show severe keratinization and in some instances the lumen of the urinary bladder contains large amounts of desquamated keratin material. Chemically induced bladder papilloma may be completely squamous but spontaneously occurring squamous cell papilloma has not been reported (Hard et al. 1999).
Carcinoma, Transitional Cell (Figures 103–113): Ureter, Urinary Bladder, Urethra, Renal Pelvis
Species: rat, mouse
Synonyms: carcinoma, urothelium
Pathogenesis/cell of origin Arises from the urothelial (transitional cell epithelium) lining Can arise spontaneously or from exposure to chemical carcinogens, urinary solids, or infections (Boorman and Hollander 1974)
Diagnostic features
Usually a solitary neoplasm but rarely can be multiple
May become large and poorly demarcated.
Invasion into the bladder wall
Well-differentiated or highly anaplastic cellular morphology
Mitoses variable
Abnormal mitotic figures may be present
Mixed cellular components may be noted
Growth pattern is papillary projecting into the lumen from a slender stalk, or solid originating from a broad base as a sessile growth
Urothelium is arranged in cords and solid sheets (sometimes in a polar arrangement), or hollow nests or in trabecular fashion
Stroma is a delicate connective tissue framework
Vascularization may be prominent
Hemorrhage and necrosis may be present
Great majority of cells within a mass represent transitional cells, but squamous differentiation may be present
In sheets of urothelium, cellular polarity of the transitional cells is lost
Nuclear polymorphism may be present
Nuclei may be bizarre or spindle shaped
Inflammatory cells, especially lymphocytes and mast cells, may be present
Metastasis can occur occasionally
Some of these malignant neoplasms may occur as noninvasive lesions with similar morphology to invasive tumors
Differential diagnoses Papilloma, urothelial: No evidence of invasion or cellular pleomorphism; fibrovascular core with complex branching Carcinoma, squamous cell: Great majority of cells are squamous cells; keratinization and pearl formation may be present Carcinoma, renal: No evidence of nontubule-derived cells or urothelial differentiation
Comment: Spontaneous carcinomas of the lower urinary tract and renal pelvis are uncommon tumors in most strains of rodents (Cohen 1998). Highly malignant tumors invade readily but metastases are uncommon. When observed they may be in regional lymph nodes and the lungs (Frith et al. 1995). Mixtures with squamous cells occasionally occur. The predominant cell type determines the diagnosis, i.e., transitional or squamous cell carcinoma. While the newer term in the human literature is urothelial carcinoma and in time will likely become the preferred term for this entity in preclinical toxicity studies, the term transitional cell carcinoma has been used for many years and is still appropriate. Typically these tumors are associated with a prominent inflammatory reaction. Transitional cell carcinomas may project into the lumen as exophytic growths or they may grow into the subepithelial tissue as endophytic solid growths. The diagnosis of carcinoma can be made on morphology alone. The identification of invasion of well-differentiated carcinomas of papillary type is difficult. This usually requires a careful search of the connective tissue stalk of large papillary lesions, as well as of the urinary bladder wall. Urinary bladder carcinomas commonly invade locally but only occasionally metastasize to regional lymph nodes and the lungs. A broad spectrum of variants of urothelial carcinomas can be observed: variants with pseudoglandular differentiation, with squamous cell differentiation, with glandular (mucous) differentiation, with both squamous cell and glandular differentiation, and with intracellular mucus production.
In advanced stages of carcinogenesis, carcinomas can be observed which show a mixed histological pattern and cellular differentiation. In such cases, classification is based on the different components present. Mixed carcinomas consist either of mixed transitional and squamous cells, mixed transitional and glandular cells or mixed transitional, squamous and glandular cells. Induced transitional cell carcinomas of the ureter are extremely rare in the mouse. Spontaneous transitional carcinomas of the mouse urinary bladder or urethra are also rare. They have been induced experimentally by a few genotoxic chemicals, notably N[4-5-nitro-2-furyl)-2-thiazolyl]formamide (FANFT) and OH-BBN. Bladder tumors induced by chemical carcinogens in mice are usually flat, poorly differentiated, invasive carcinomas in contrast to the exophytic papillary bladder tumors induced in rats. OH-BBN is a particularly potent bladder carcinogen for the mouse, rapidly inducing highly aggressive transitional cell carcinomas that can metastasize to the lungs (Akagi et al. 1973). Spontaneous transitional cell carcinomas of the renal pelvis have not been reported in control mice, but they have been induced by genotoxic chemicals such as dimethylnitrosamine and OH-BBN. The OH-BBN studies in NON/Shi mice illustrate the potential of this tumour to metastasize to the lungs (Akagi et al. 1973; Fukushima et al. 1976). In a study with tetrachloroazobenzene (TCAB) in B6C3F1 mice, urethral transitional cell carcinomas and carcinomas of the urethral glands arose which were invasive into surrounding tissues (Singh et al. 2010) Transitional cell carcinomas have been induced by a variety of chemicals (Frith 1986; Jokinen 1990; Gaillard 1999; Cohen 2002). Transitional cell carcinomas appear to metastasize less frequently in the mouse than does the squamous cell type. Transitional cell carcinomas of the renal pelvis in rats are poorly demarcated growths that tend to proliferate within the renal pelvis at first, and then invade and deform the renal parenchyma. Mixtures with squamous cells occasionally occur. Typically these tumors are associated with a prominent inflammatory reaction. In general, mesenchymal tumors of rodents occur considerably less frequently than epithelial tumors (Gaillard 1999).
Carcinoma, Squamous Cell (Figures 113–115): Ureter, Urinary Bladder, Urethra
Species: rat, mouse
Synonyms: epidermoid carcinoma
Pathogenesis/cell of origin: Arises from transitional cell epithelium which has either undergone squamous differentiation and then developed into neoplasia or from transitional epithelium which has undergone neoplastic transformation to transitional cell carcinoma and then developed squamous differentiation
Diagnostic features Usually single neoplasms which are higly invasive Well differentiated to poorly differentiated Cellular atypia and pleomorphism may be pronounced The squamous epithelium is commonly highly keratinized with formation of keratin pearls or may contain keratohyaline granules only Mitotic figures may be frequent The fibrocollagenous stromal component may be well developed Squamous cells are arranged in cords, sheets, or irregular nests Inflammatory cell infiltrate is often present Invasion of the surrounding tissue is present Metastasis may be common Well-differentiated cells show intercellular bridges, normal keratinization, minimal nuclear atypia, and a low mitotic index Poorly differentiated cells exhibit cellular and nuclear atypia, abnormal keratinization (parakeratosis), frequent mitoses, and abnormal mitotic figures Mineralization may be present, especially in the rat Vascularization is very prominent in the mouse
Differential diagnoses Carcinoma, transitional cell: transitional cell differentiation; limited areas of squamous differentiation Stroma usually consists of delicate framework Papilloma, squamous cell: Absence of invasion, minor cellular atypia; vascularized ramiform stalk of connective tissue
Comment: Areas of squamous differentiation may be present in urothelial tumors including transitional cell carcinoma. The diagnosis squamous cell carcinoma should be reserved for tumors where the great majority of cells are of the squamous type. Spontaneous squamous carcinomas are extremely rare, but this tumor can be induced experimentally in the renal pelvis following the introduction of polynuclear hydrocarbons into the kidney or renal pelvis, and in hydronephrotic NON/Shi mice after treatment with OH-BBN. In the urinary bladder, it has been induced by dibutylnitrosamine and OH-BBN (Akagi et al. 1973; Fukushima et al. 1976). The majority of squamous cell carcinomas are poorly differentiated and more invasive than transitional cell carcinomas. They metastasize readily. However, squamous cell carcinomas are encountered less frequently in association with carcinogens than the transitional cell counterpart, but appear to metastasize more frequently.
Adenocarcinoma: Ureter, Urinary Bladder, Urethra
Species: rat, mouse
Synonyms: adenocarcinoma, urothelium
Pathogenesis/cell of origin Arises from urothelium that has undergone glandular metaplasia and neoplastic transformation
Diagnostic features Single neoplasms which may become large Glandular component lined by one or more layers of cuboidal to columnar cells Moderate cellular atypia and invasive growth pattern Glandular structures may contain mucus Nuclei may be located eccentrically Mitotic figures may be present
Differential diagnoses Carcinoma, transitional cell: transitional cell differentiation; limited areas of squamous differentiation; Stroma usually consists of delicate framework Glandular metaplasia: No cellular atypia or mitotic figures and no evidence of invasion
Comment: Bladder neoplasia has been produced by both DNA reactive and non-DNA reactive agents (Cohen 2002). According to NTP reports, bladder carcinogens that have been tested are more likely to employ nongenotoxic mechanisms of tumorigenesis and tend to involve transitional and squamous epithelial tumors (Wolf 2002). Primary adenocarcinomas of the urinary bladder are those with glandular structure throughout. Transitional cell carcinomas with occasional foci of glandular metaplasia are excluded from the category of true adenocarcinoma. Histological differentiation of primary and metastatic adenocarcinoma of the urinary bladder may be difficult. The three most common adenocarcinomas that secondarily involve the bladder of the rat are those from the seminal vesicles, prostate, and uterus (Kunze and Chowaniec 1990). Features helpful for differentiating a primary adenocarcinoma of the bladder include: foci of transitional epithelium; areas of transition from normal urothelium to neoplastic cuboidal and columnar epithelium; and association of proliferative changes in the adjoining epithelium. Histogenetically, adenocarcinomas are probably derived from metaplastic glands present in transitional cell carcinomas or from hyperplasia with areas of columnar or glandular metaplasia, including so-called cystitis glandularis.
Mesenchymal Proliferative Lesion (Figure 118): Urinary Bladder, Urethra
Species: mouse
Synonyms: decidual-like reaction, vegetative lesion, benign mesenchymal tumor
Pathogenesis/cell of origin The cell of origin has not been clearly identified, but one suggestion is derivation from juxtacoelomic tissue contributing not only to parts of genital organs but also to mesenchymal portions of the urinary bladder near the trigone area in both genders (Karbe et al. 2000)
Diagnostic features Occurs mainly in submucosal tissue, usually in caudal half of bladder, often near trigone Solitary or multiple May protrude into bladder lumen May be highly vascularized with well-developed vascular channels Usually associated with a chronic, mononuclear inflammatory cell infiltrate at the periphery and deposits of hemosiderin-laden macrophages. Necrosis is usually absent Is distinctly demarcated, but nonencapsulated The growth pattern is solid or polypoid Areas of coagulative necrosis may be present Cells of two types are present, large pleomorphic epithelioid cells in compact islands and surrounded peripherally by spindle cells Epithelioid cells have distinct borders, eosinophilic, homogeneous or fibrillar cytoplasm, and eosinophilic granules may be present Nuclei of epithelioid cells are eccentric, often with pleomorphic or bizarre outline, and prominent nucleoli Spindle cells are fibrocyte-like or smooth muscle-like. Mitotic figures may be present Does not invade surface urothelium or beyond serosa, and overlying epithelium remains intact Local invasion of smooth muscle and submucosa by spindle cell component may be present
Solid subtype
Single or multiple submucosal proliferations
Polypoid subtype
Polypoid mass is bulging or protruding into the lumen
Mass has a fibrovascular stroma covered by urothelium
Close to the surface, suburothelial foci of solid mesenchymal proliferative lesions are present
Differential diagnoses Leiomyoma/leiomyosarcoma: definitive smooth muscle differentiation Chronic inflammation: no mitotic figures or invasion, well-differentiated fibroblasts Sarcoma or fibrosarcoma: fibroblastic differentiation; invasive and development within the bladder wall rather than polypoid protrusion into the lumen
Comment: Mesenchymal tumors of the mouse urinary bladder have been reported with vascular and smooth muscle differentiation (Butler, Cohen, and Squire 1997). The definitive nature of this lesion is controversial, with views ranging from classifying these as mesenchymal cell proliferations to benign tumors or decidual-like reactions. In the past, the tumor has been incorrectly identified as a leiomyoma/leiomyosarcoma or even an undifferentiated sarcoma. The general consensus among pathologists currently is that mesenchymal proliferative lesions do not represent true neoplasia. The pathogenesis of these lesions is still uncertain. Some similarities with lesions of the uterus and seminal vesicles have been reported, leading to the suggestion they may represent decidual-like reactions (Karbe et al. 1998; Karbe 1999; Karbe et al. 2000). This lesion is primarily spontaneous and strain-specific, being found most frequently in Swiss-derived mice. Thus, it has been seen predominantly in Swiss Webster, NMRI and CD1 strains, but not as yet in B6C3F1 mice. In particular, mesenchymal tumors were characterized morphologically from a number of studies involving the Swiss Webster CD-1 mouse (Halliwell 1998; Cohen 2002). With immunohistochemistry the lesions are cytokeratin negative and slightly desmin postitive. Actin positivity is equivocal (Halliwell 1998). Smooth muscle differentiation is evident by transmission electron microscopy (Jacobs et al. 1976). Identical bladder lesions have been described in association with surgically implanted glass or paraffin pellets, particularly in the vicinity of sutures used to close the bladder after implantation (Bonser and Jull 1957). Whether these lesions can be induced by chemical agents is not presently known, although an increased incidence has been noted with various combinations of endogenously administered estrogens and progesterone (McConnell 1989).
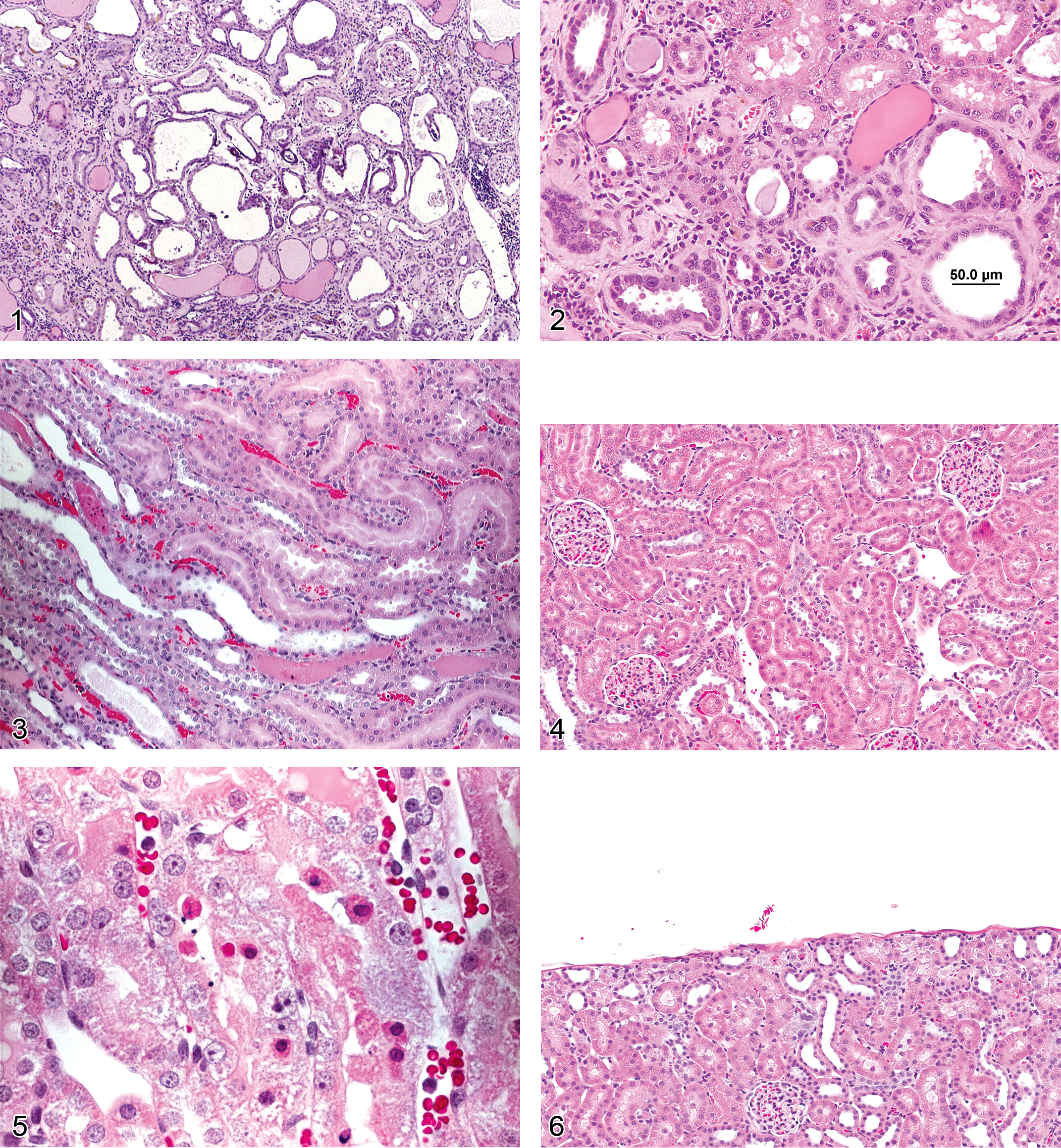
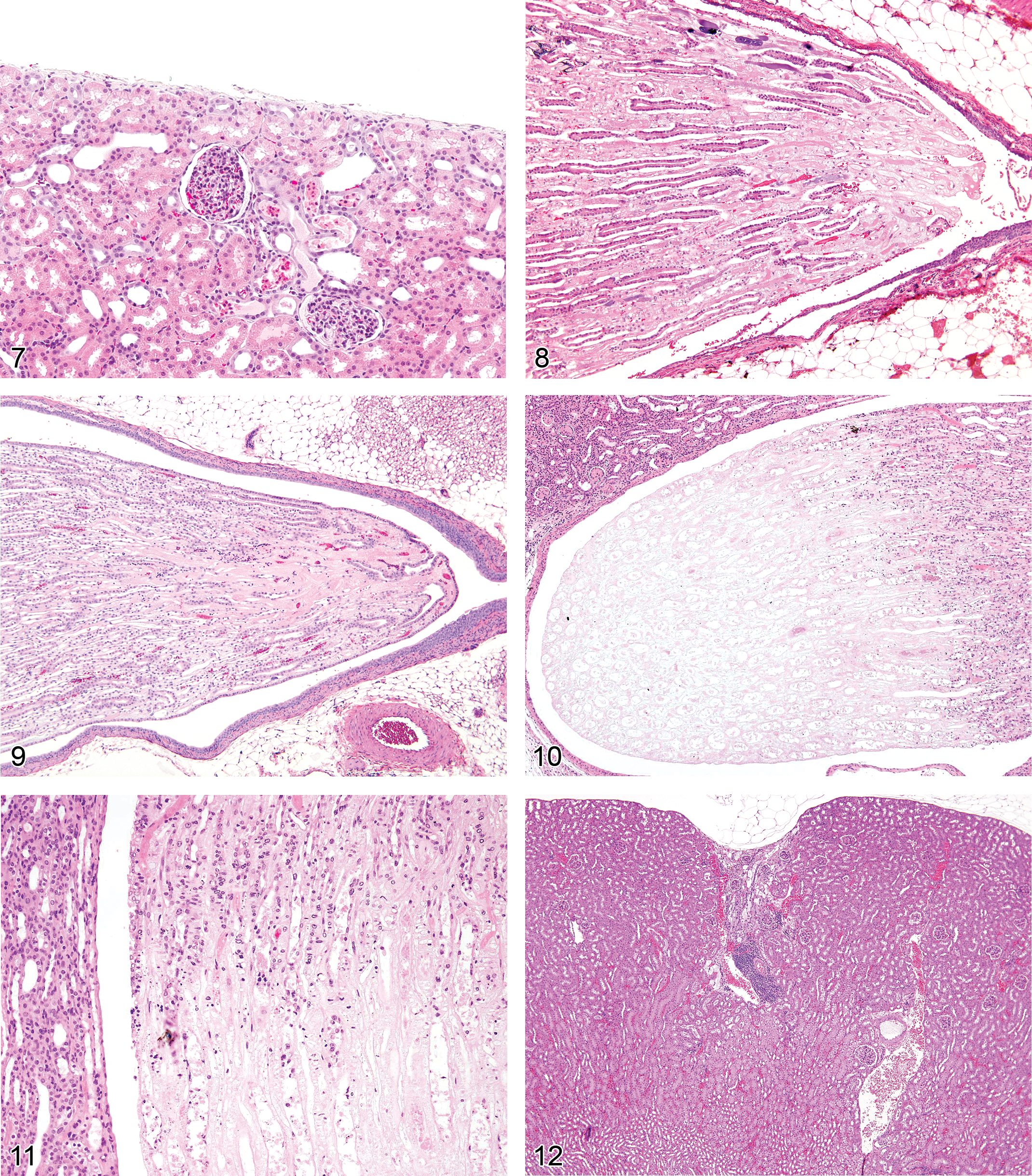
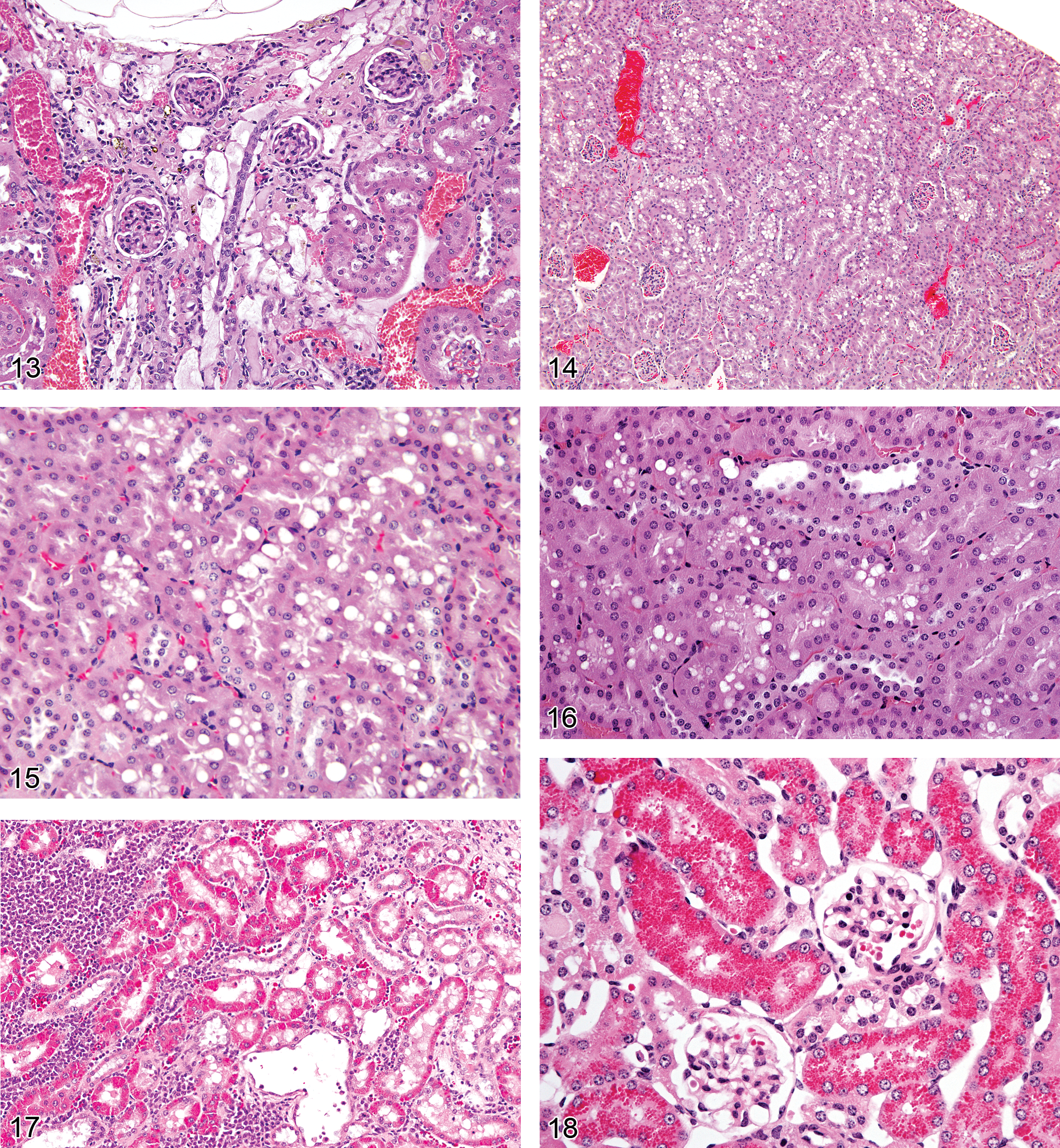
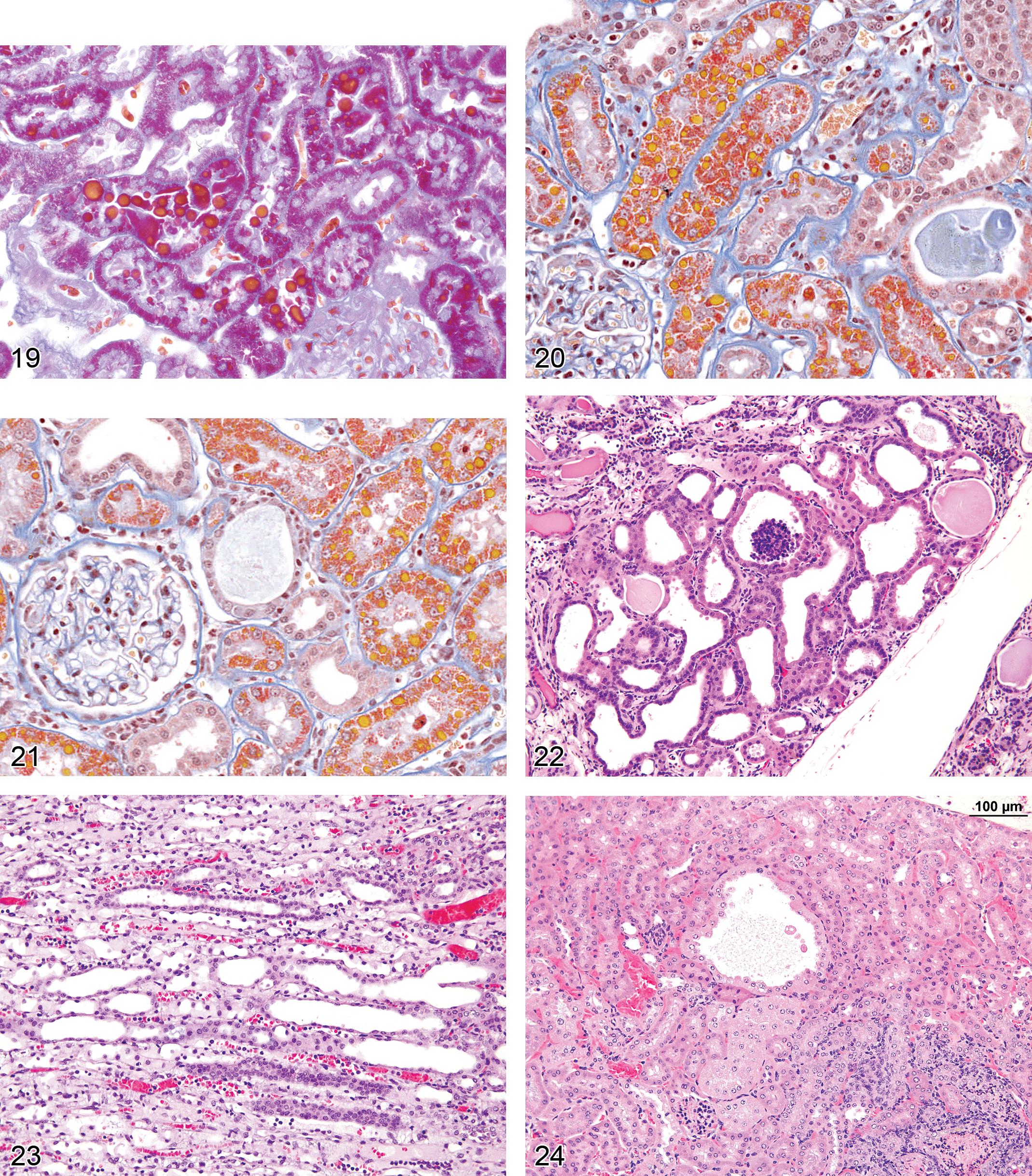
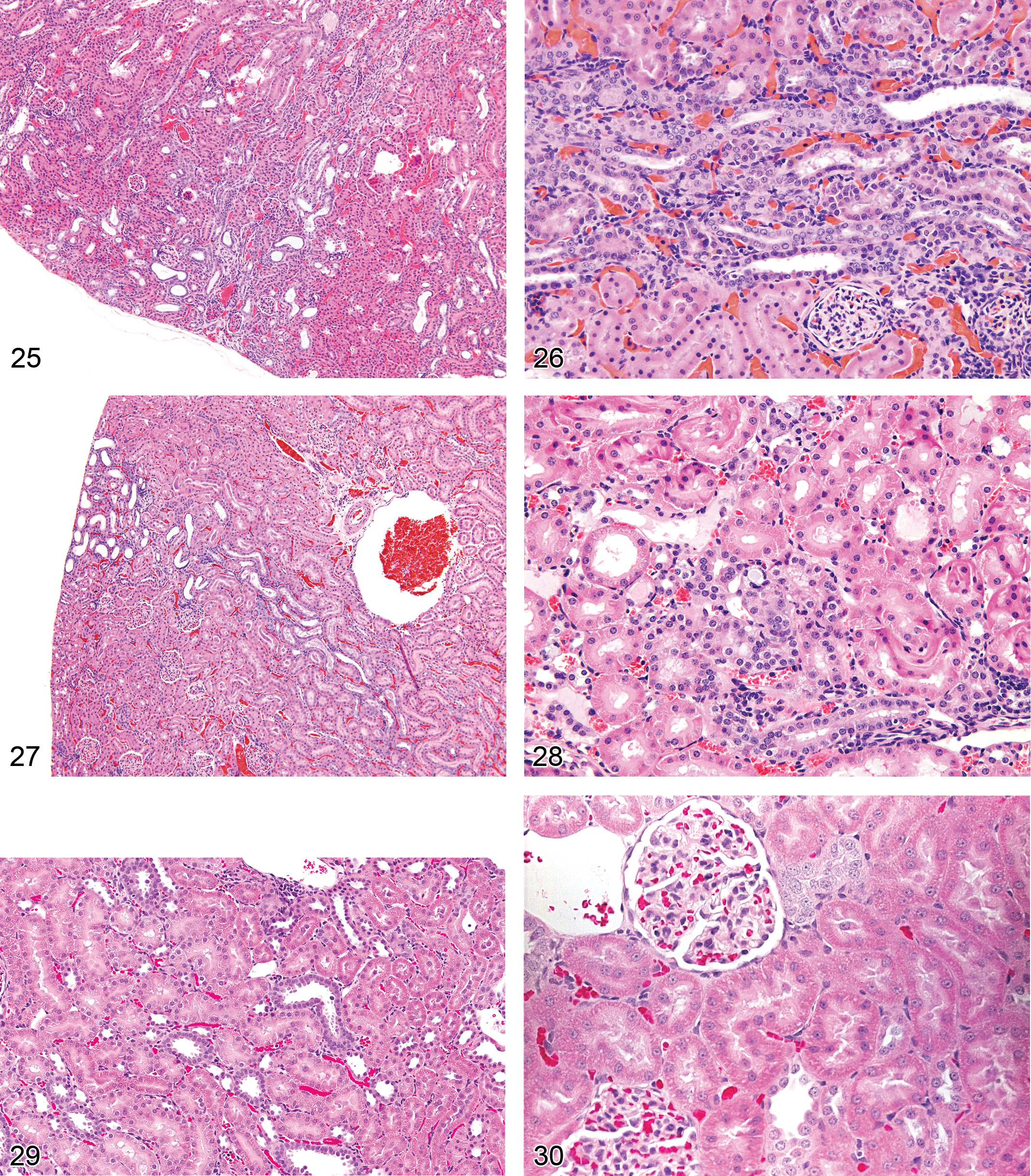
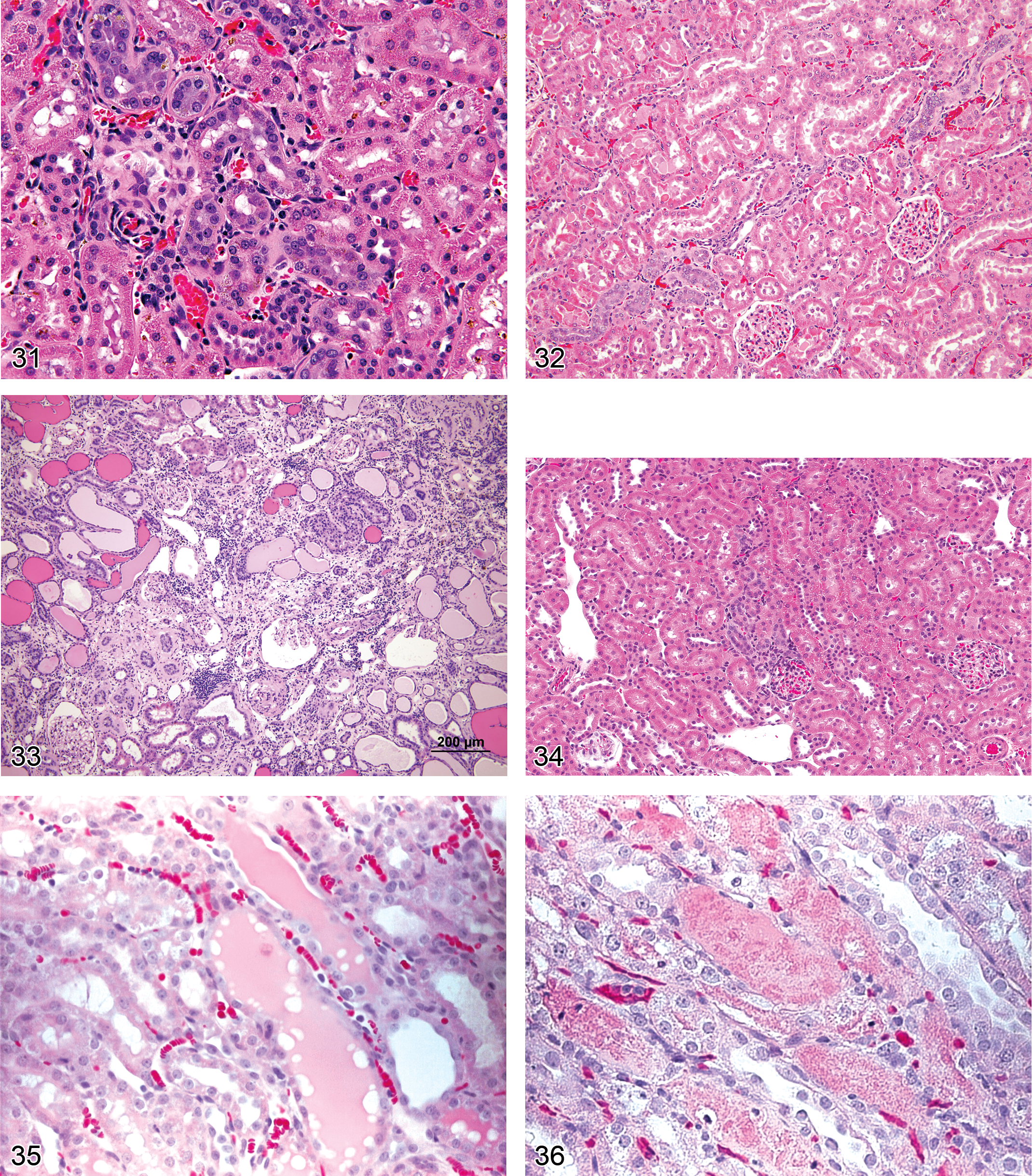
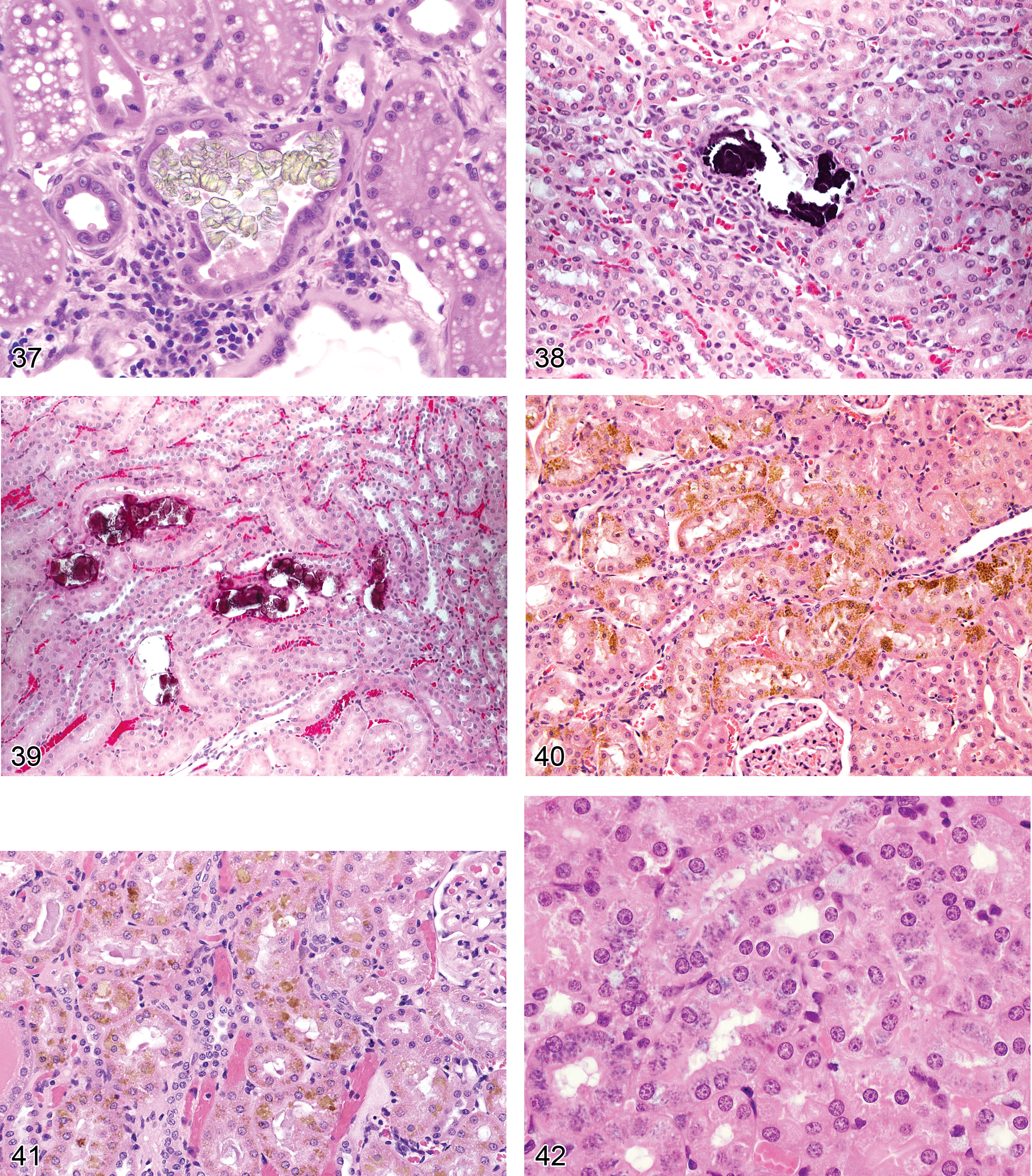
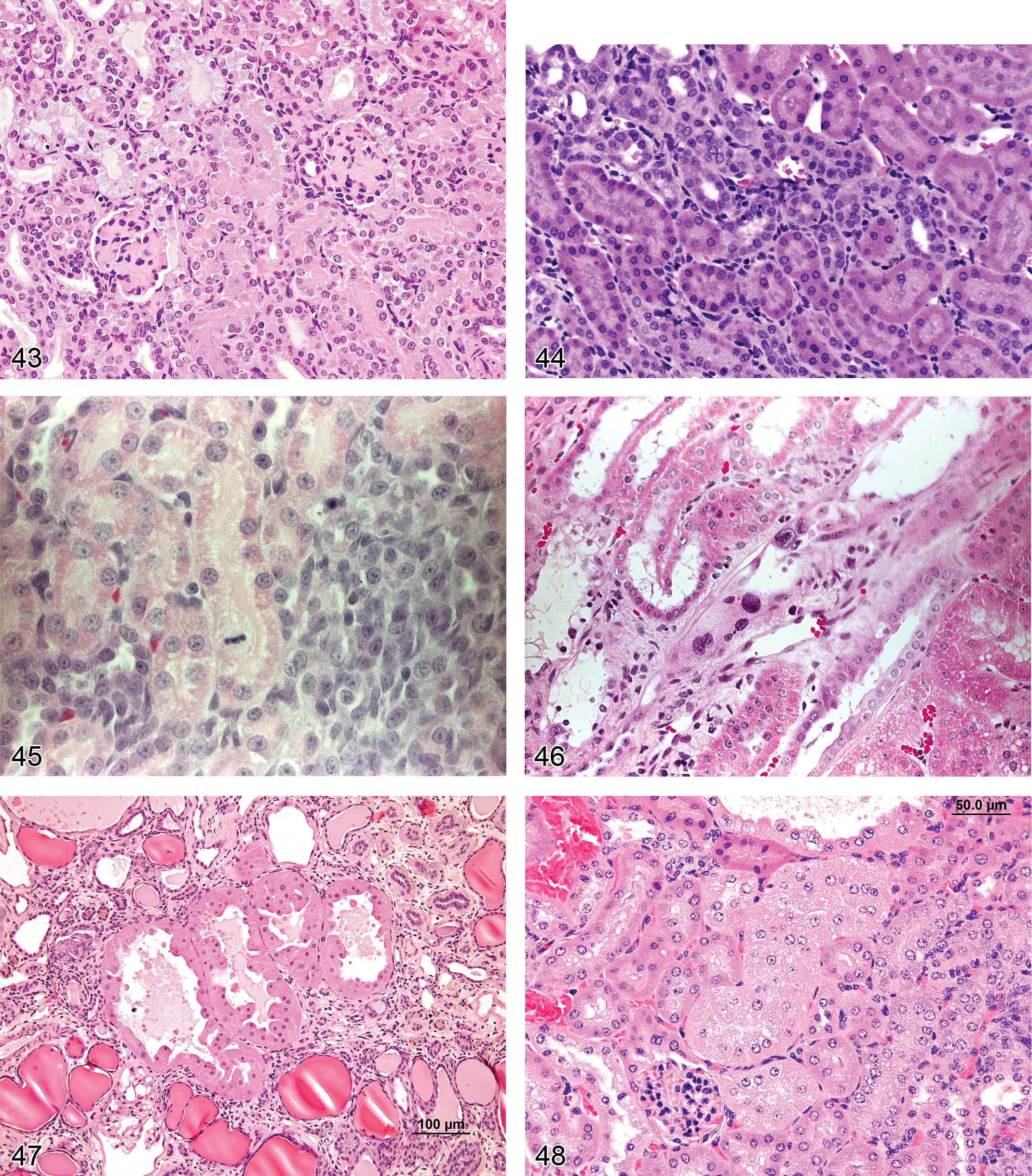
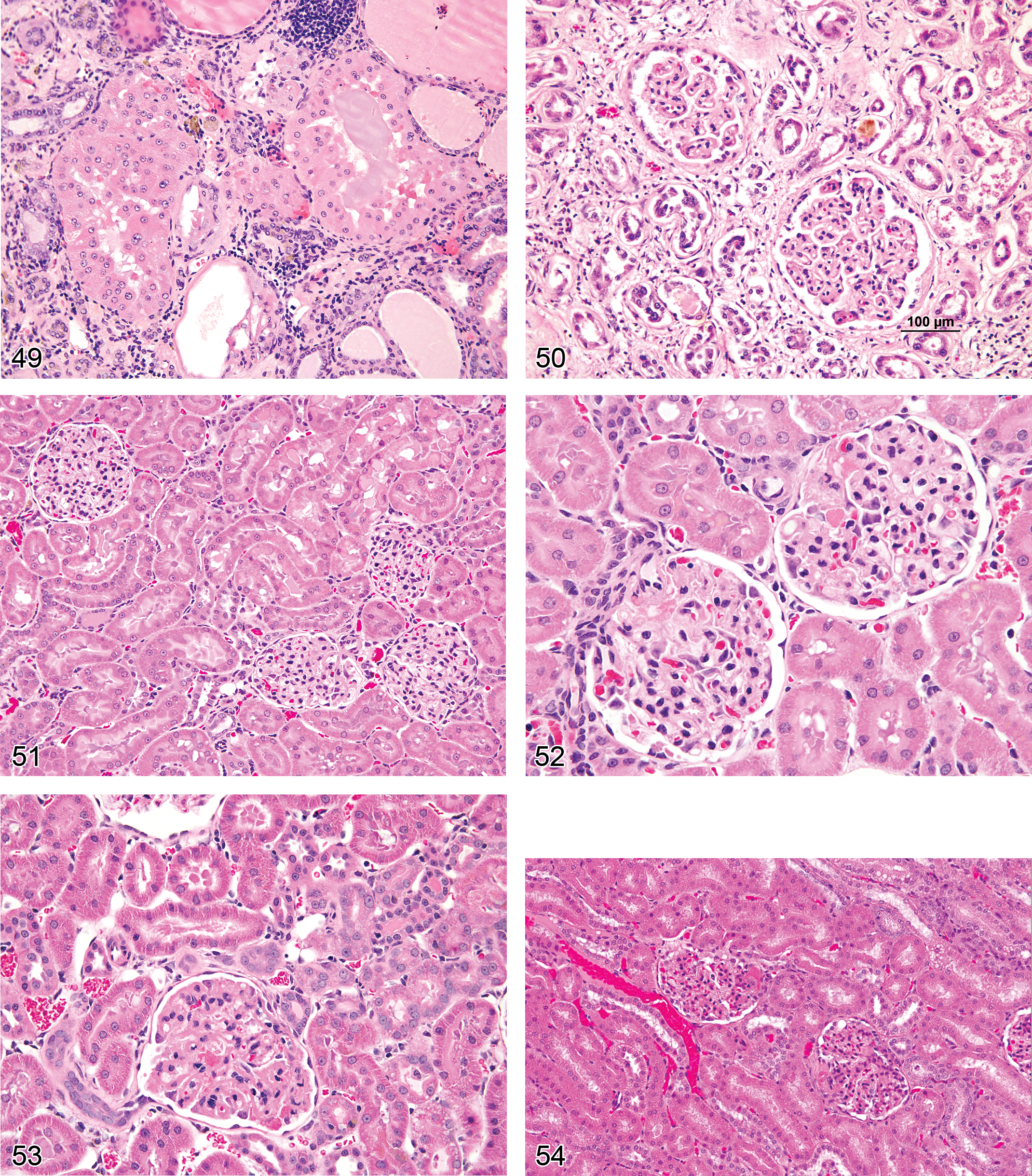
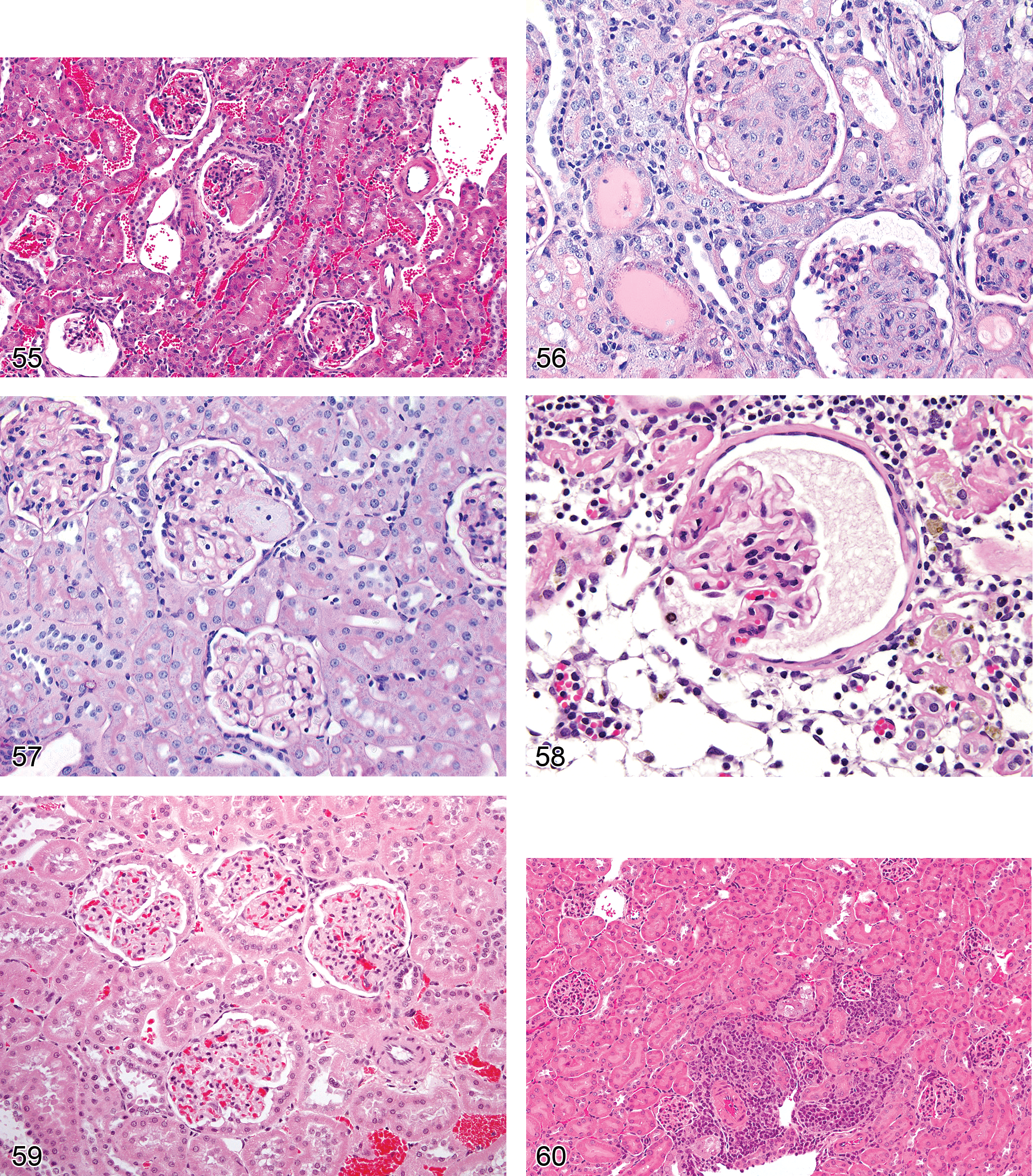
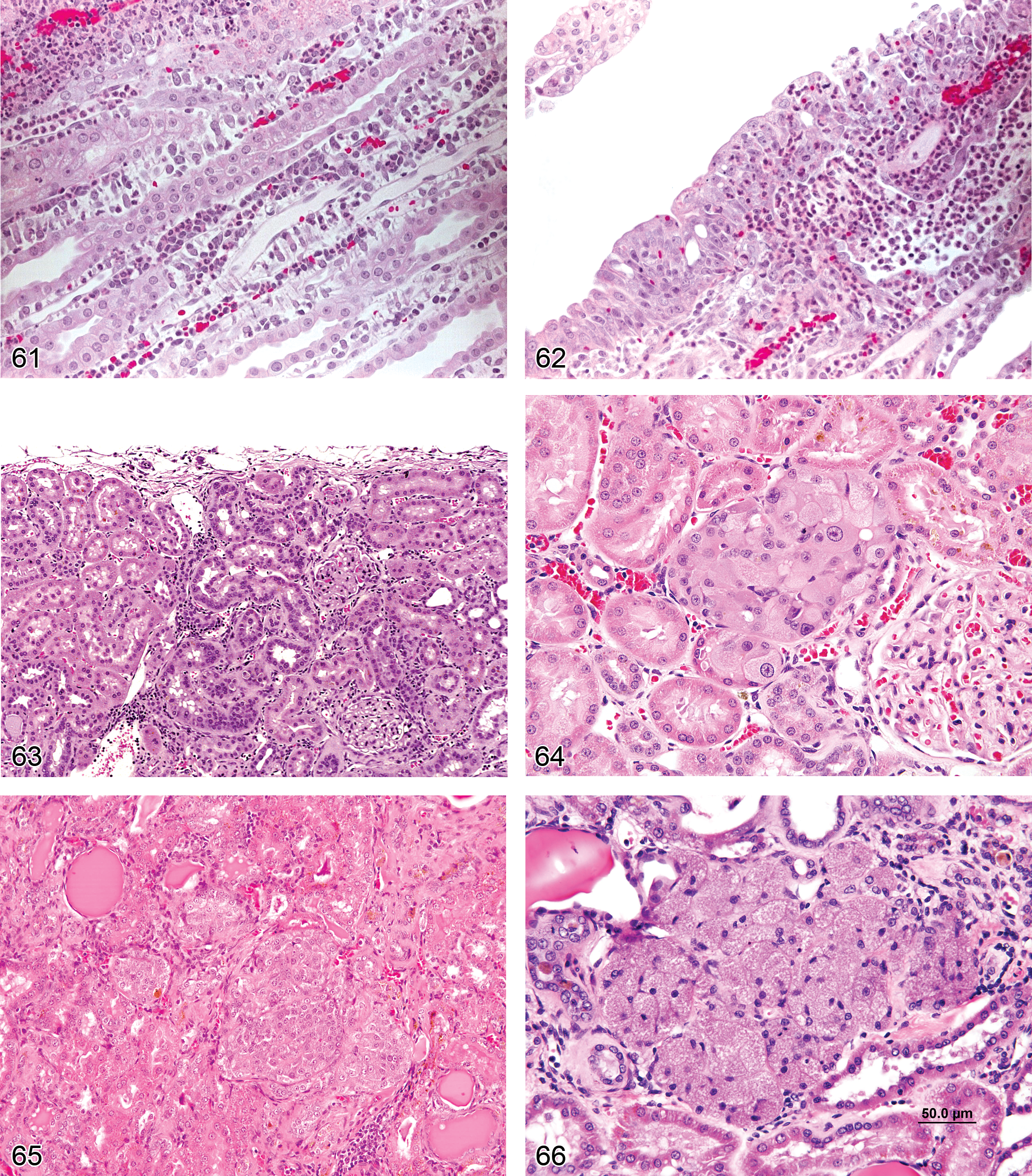
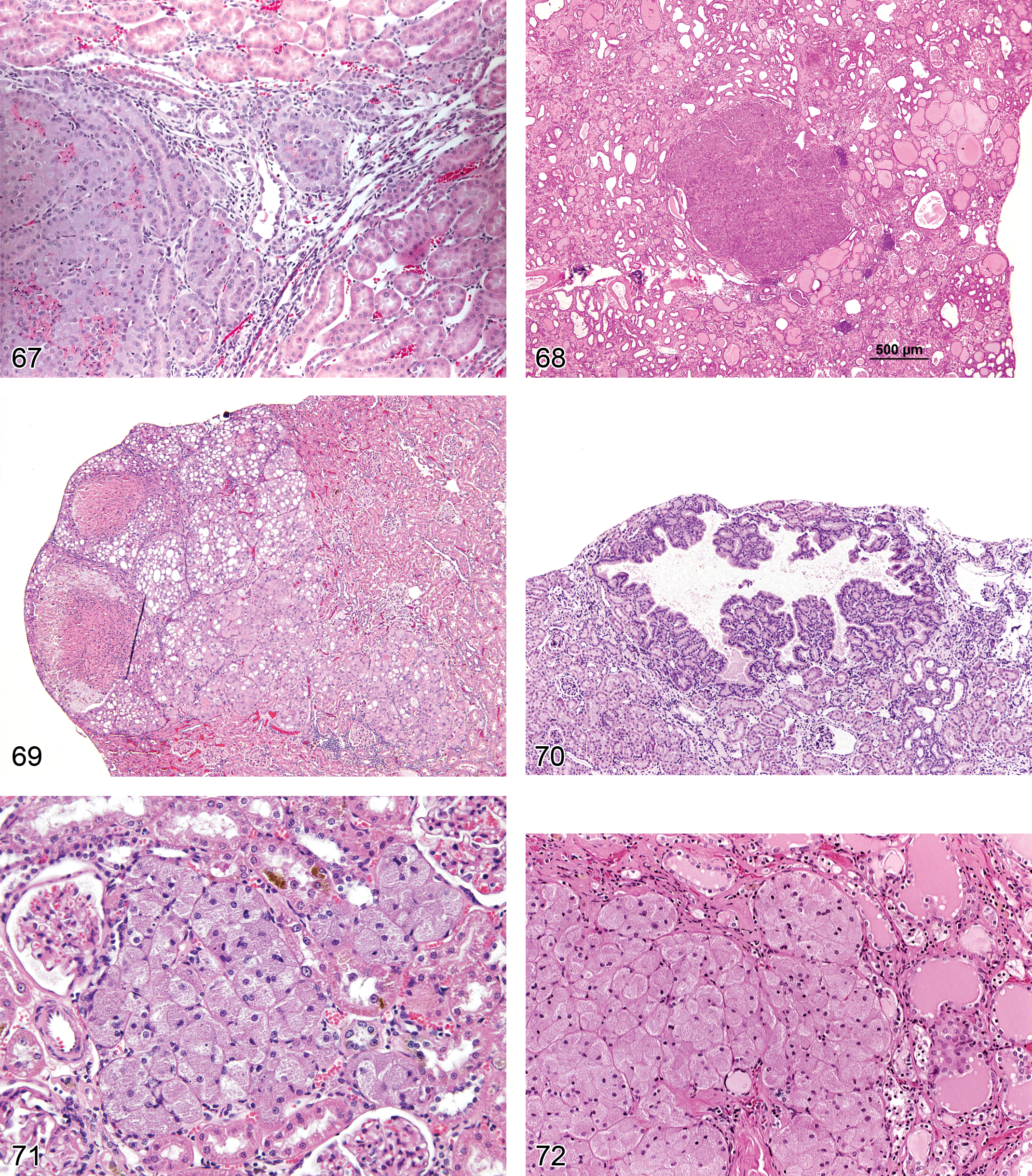
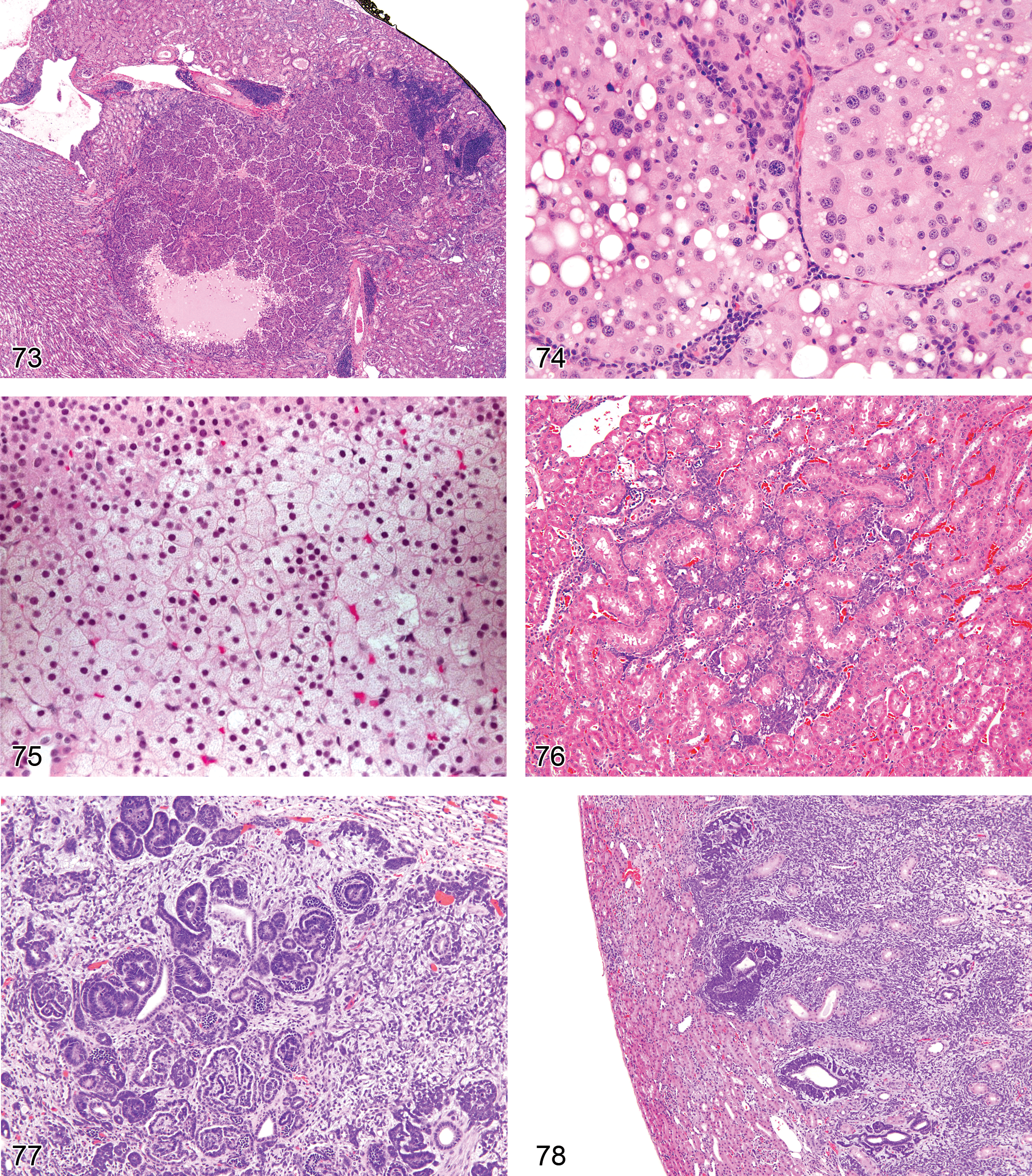
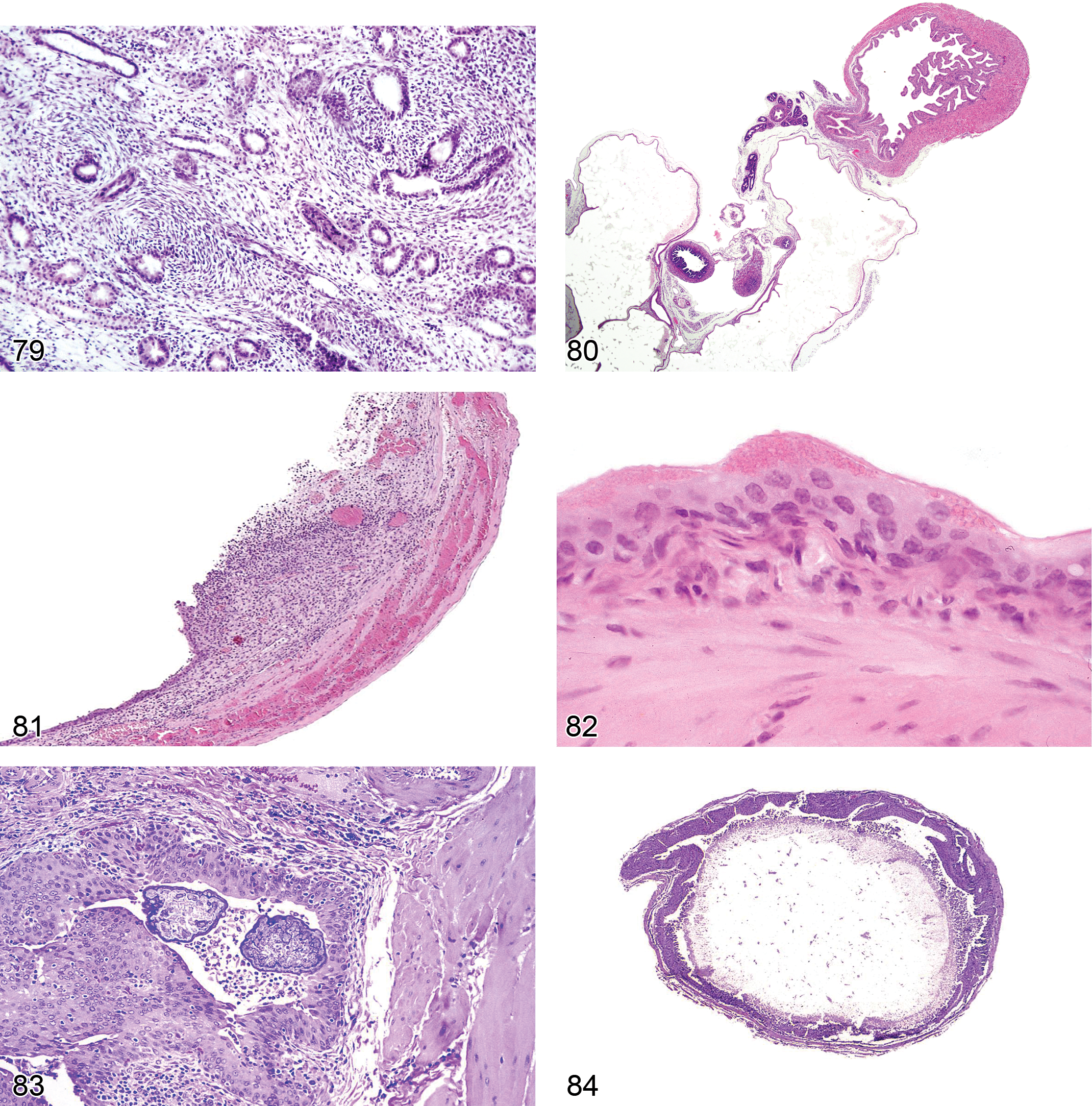
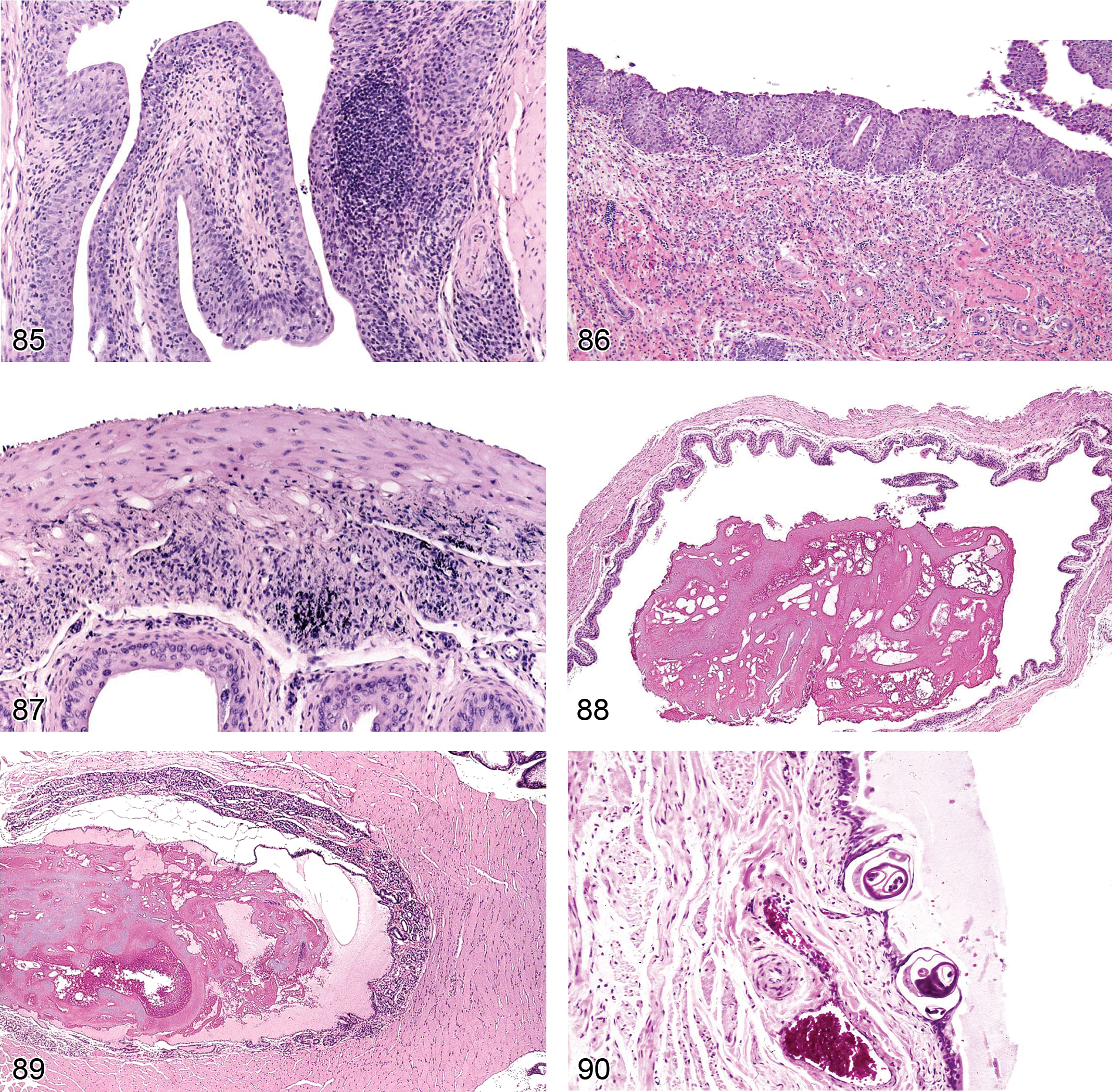
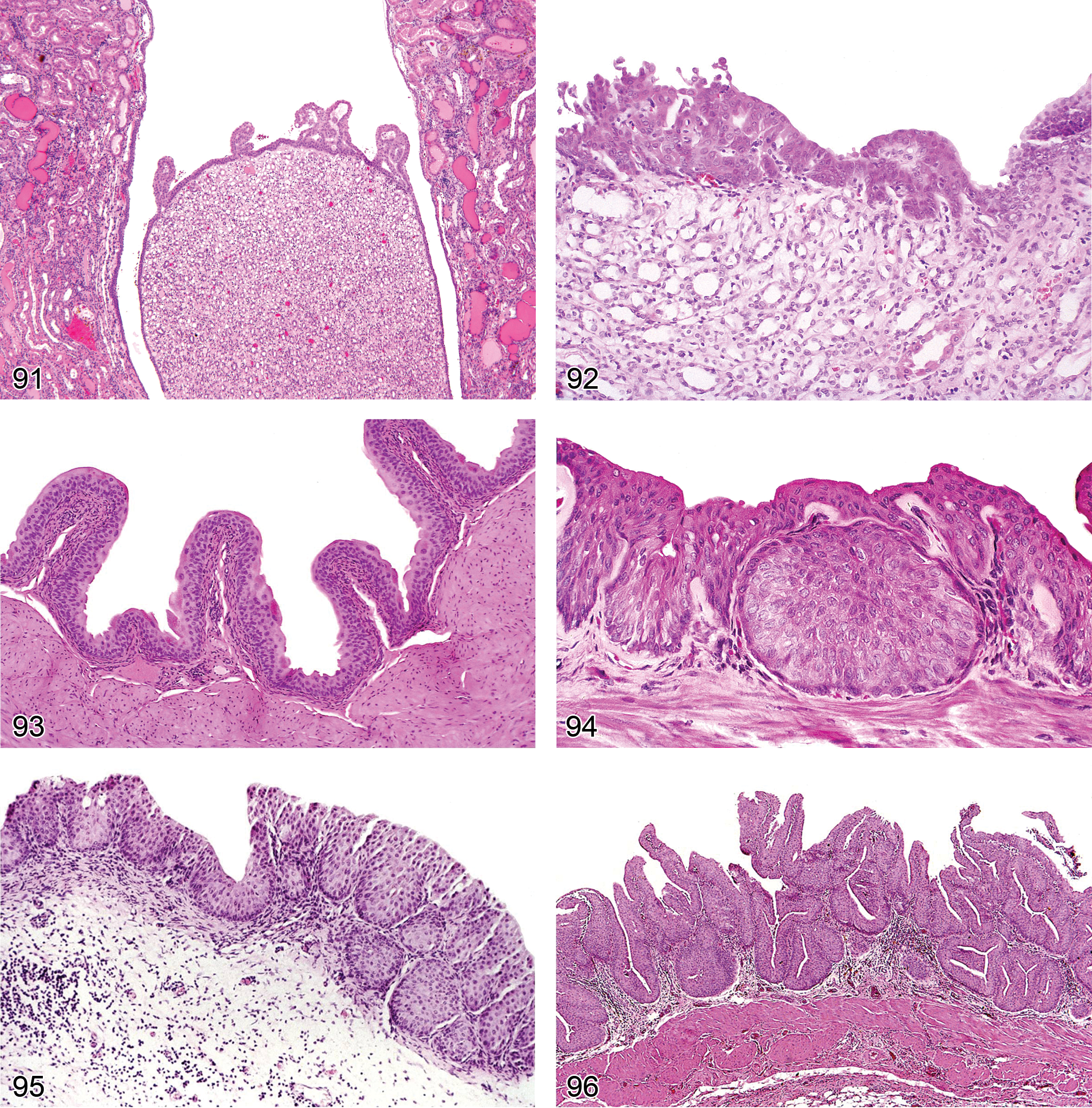
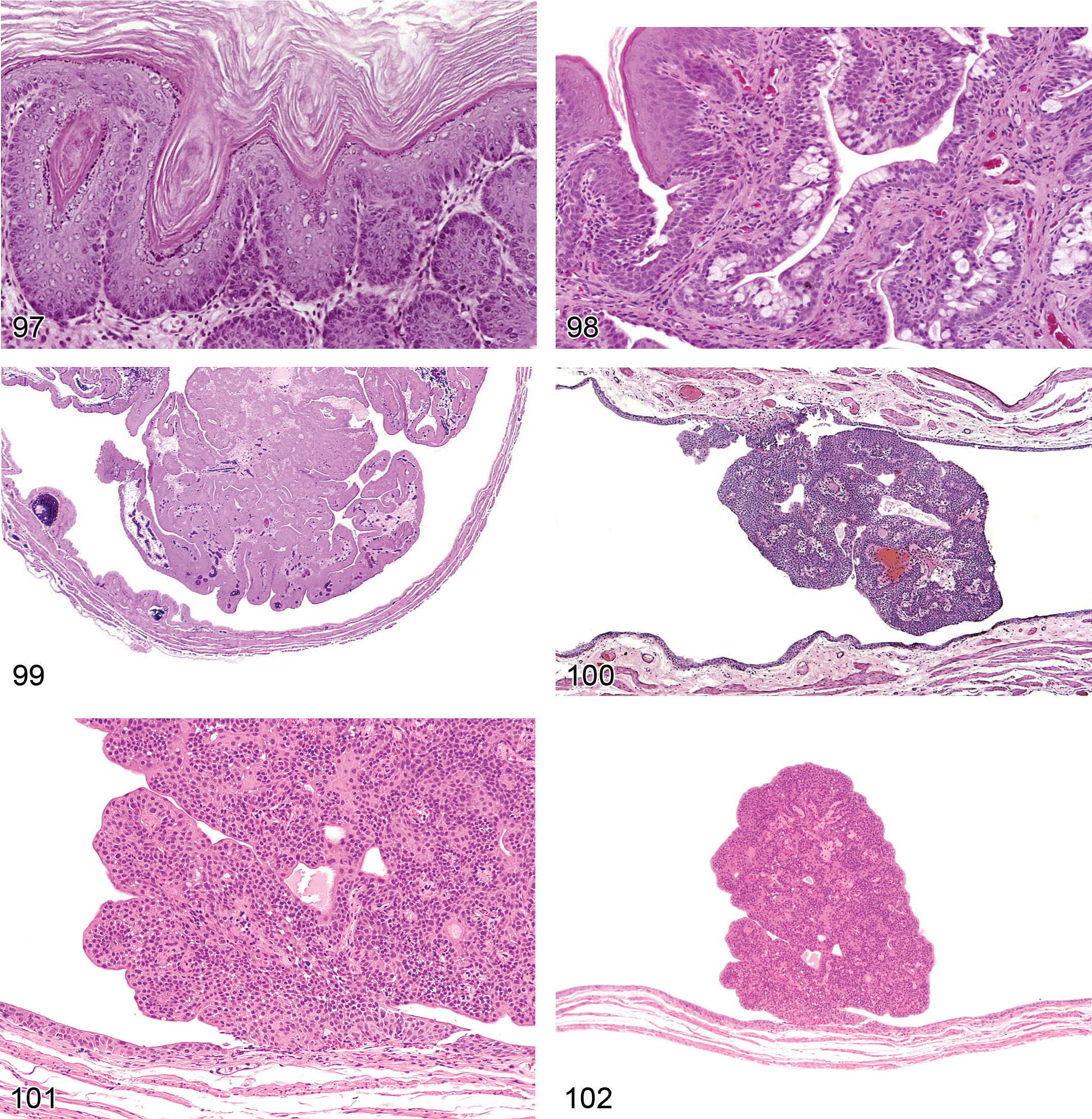
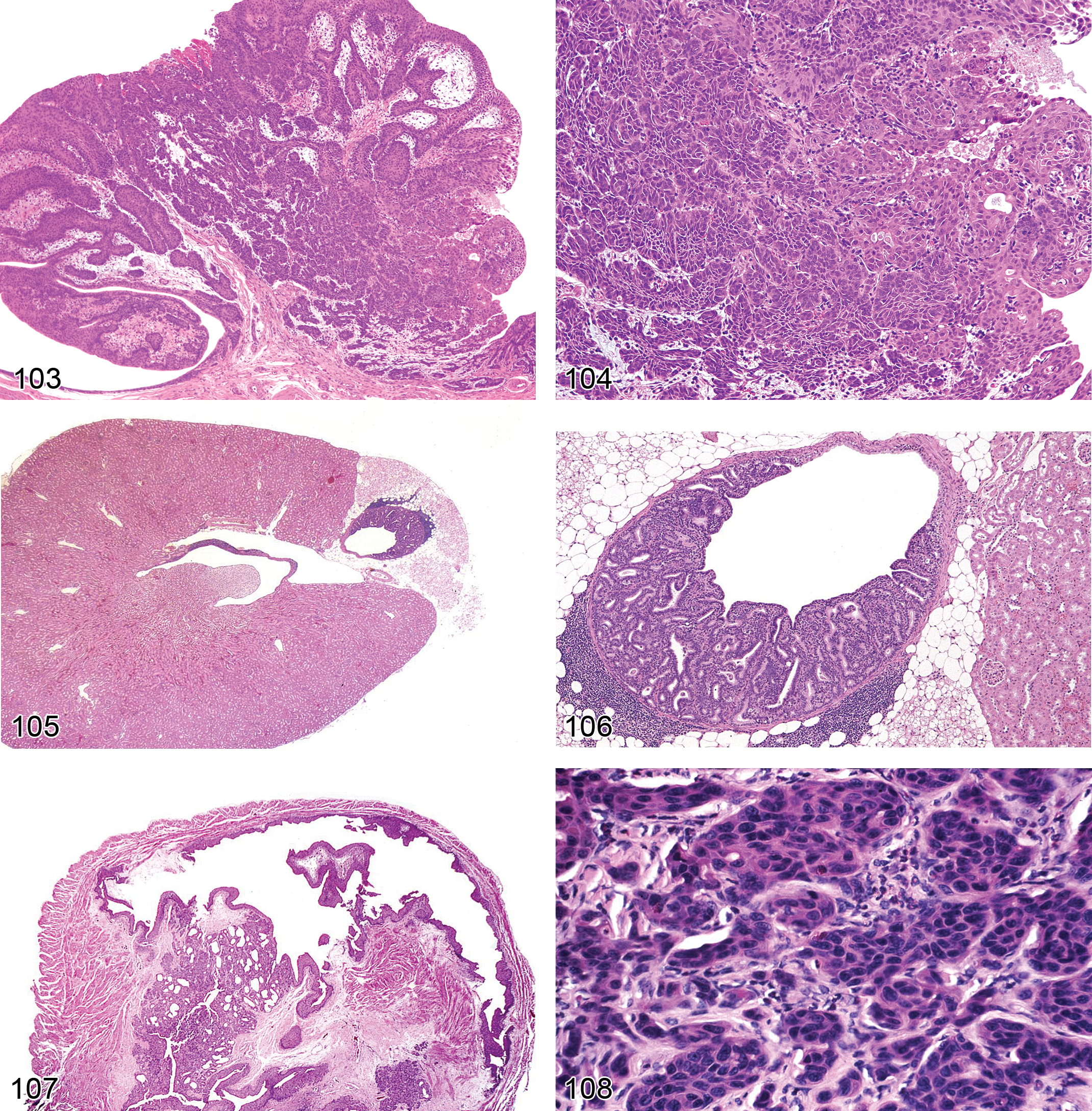
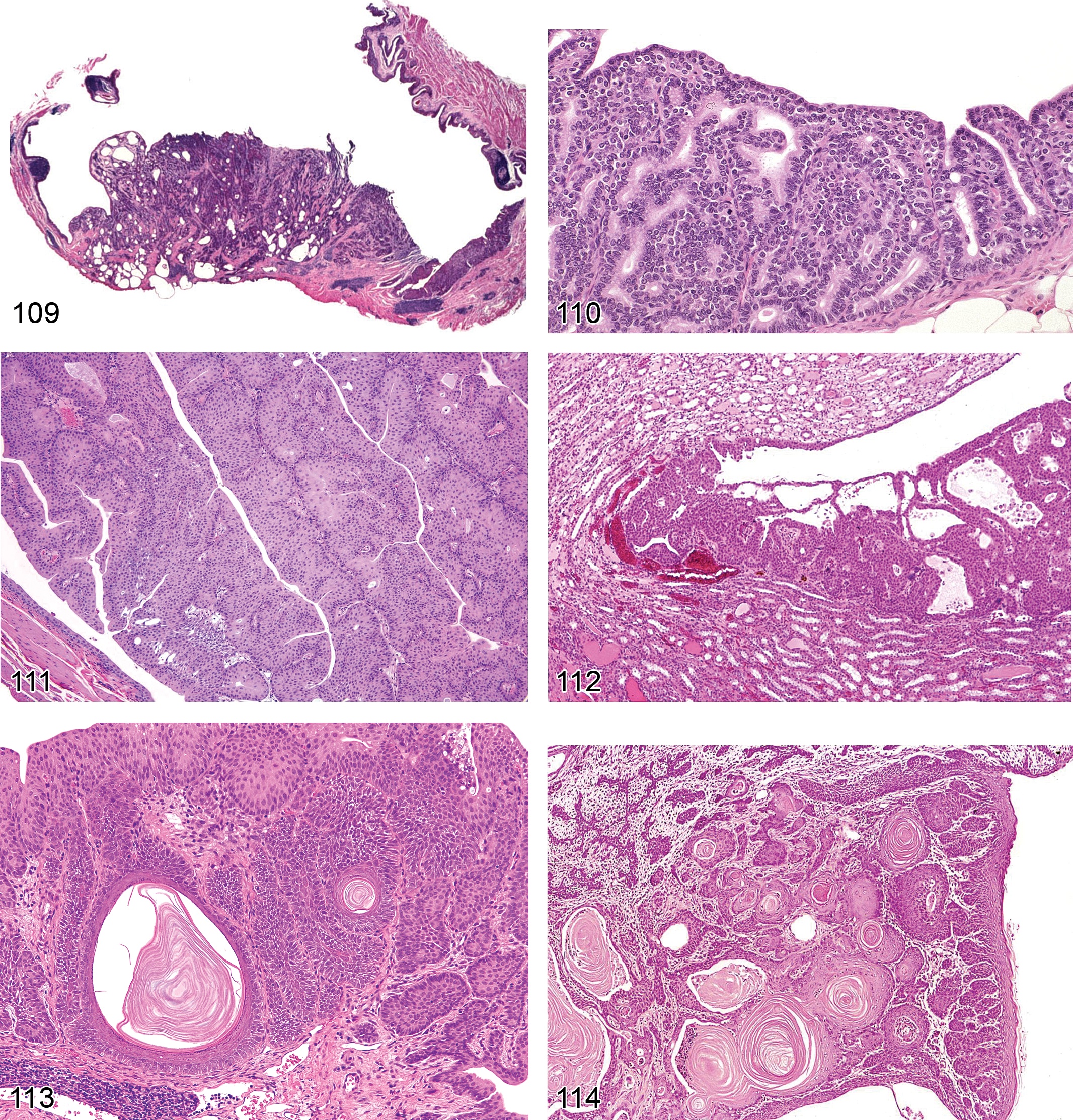
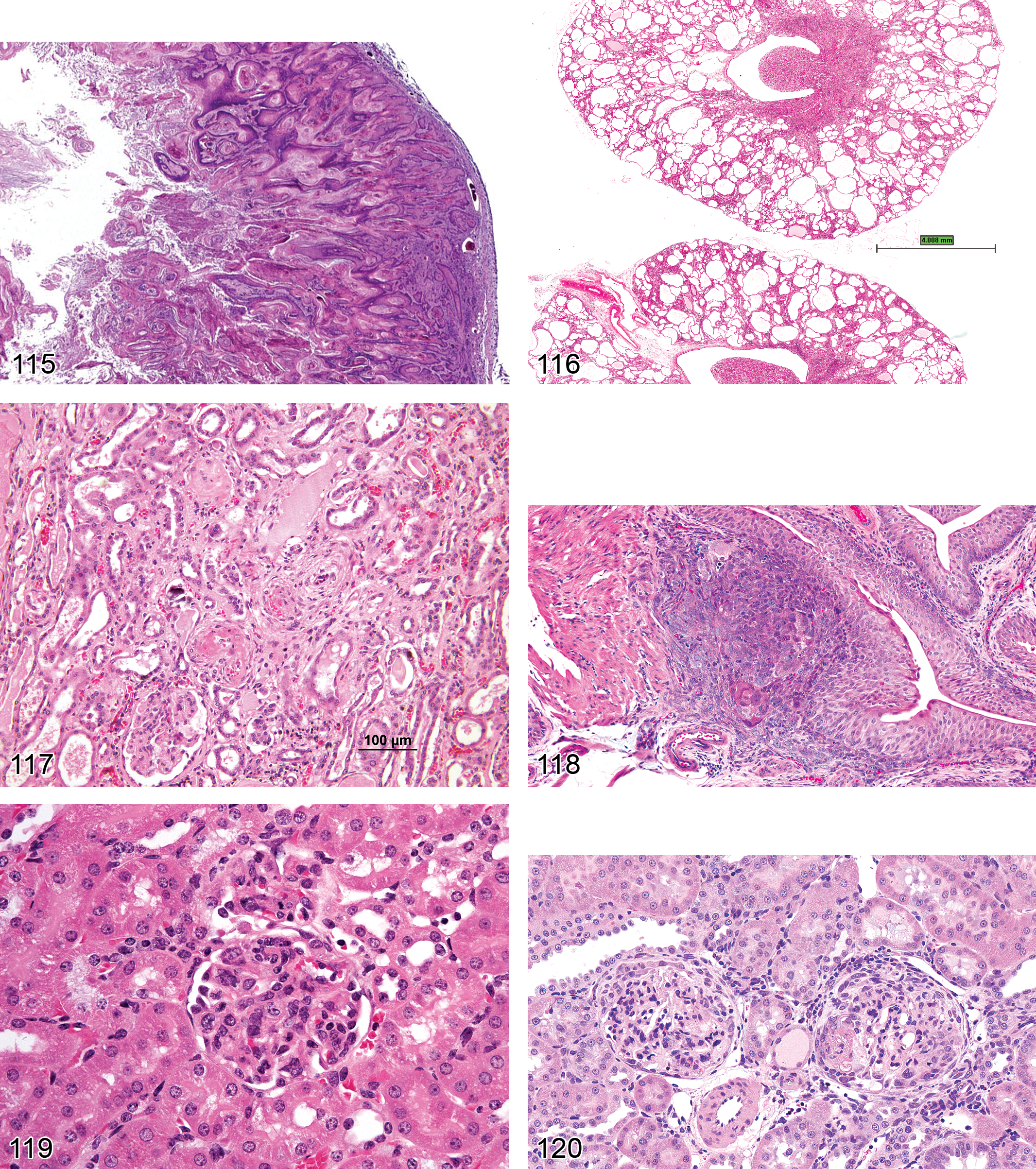
Footnotes
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The authors received no financial support for the research, authorship, and/or publication of this article.
Acknowledgments
The authors wish to express their sincere appreciation for the assistance of John Vahle, Peter Mann, Tracy Gales, Beth Mahler, Beverly Maleef, Wolfgang Kaufmann, Charlotte Keenan, Rupert Kellner, and Sam Cohen for their help and contributions in the preparation of this work.
