Abstract
Cardiac troponins serve as serum biomarkers of myocardial injury. The current study examined the influence of age on serum concentrations of cardiac troponin I (cTnI). An ultrasensitive immunoassay was used to monitor cTnI concentrations in Sprague-Dawley (SD) rats and Erythrocebus patas monkeys of different ages. The mean cTnI concentrations were highest in 10-day-old rats compared to 25-, 40-, and 80-day-old SD rats. Cardiomyocyte remodeling was apparent in hearts from 10-day-old SD rats as evident by hypercellularity, irregularly shaped nuclei, and moderate numbers of myocytes undergoing mitosis and apoptosis. The mean concentration of cTnI in 5 newborn monkeys was considerably higher than that of three 1-year-old monkeys. Evidence of cardiomyocyte remodeling was also observed in these newborn hearts (loss of myofibrils and cytoplasmic vacuolation). Commercial animal serum samples were also analyzed. The concentrations of cTnI detected in fetal equine and porcine serum were considerably higher than that found in adult equine and porcine serum samples Likewise, fetal bovine serum had higher cTnI concentrations (>2,400 pg/ml) than did adult caprine and laprine samples (2.5–2.7 pg/ml). The present study found age-related differences in cTnI concentrations, with higher levels occurring at younger ages. This effect was consistent across several animal species.
Introduction
Troponins are proteins critical to the regulation of striated muscle contraction. The amino acid sequence of 2 cardiac troponin proteins, cardiac troponin I (cTnI) and cardiac troponin T (cTnT), are unique to the cardiac myocyte (Mair 1997), which enables the development of sensitive and specific immunoassays (Katus et al. 1989; Bodor et al. 1992). Because the cardiac myocyte is the sole source of the cardiac troponins, monitoring the serum or plasma concentrations of either of these 2 troponins provides a specific means for detecting myocardial injury. The quantity of cardiac troponin that reaches the blood varies according to the type, duration, and degree of myocyte injury. Initially, monitoring the blood cTnI or cTnT concentrations became an exclusive biochemical means for detecting acute coronary syndromes (Alpert et al. 2000). Further experience in adult human patients indicated that they could also be used as biomarkers to detect various types of nonischemic cardiac injury (Gaze and Collinson 2005). The antibodies used in the commercial cardiac troponin assays also recognize nonhuman epitopes (Apple et al. 2008) and as a result both cTnI and cTnT have been used as biomarkers of cardiac injury in animals (O’Brien 2008).
Given the value of cardiac troponins as biomarkers for cardiac injury, whether they can be as useful to detect myocardial alterations in younger patients as in adults is an area of considerable interest. Age has been found to be an important factor that influences baseline cardiac troponin concentrations. For example, the baseline concentrations of cTnI are higher in normal adults older than 60 years than they are in those younger than 60 years of age (Venge et al. 2003). The range of normal values of cardiac troponin is only beginning to be determined in neonatal and pediatric populations. In seemingly healthy infants, the baseline cardiac troponin concentrations have been reported to be higher than those in adults (Kanaan and Chiang 2004; Lipshultz et al. 2008; Bennett et al. 2011). In adults, the presence of either cTnT or cTnI has been accepted as evidence of myocardial damage, but in children whether the cardiac troponins are equivalent biomarkers is not certain.
A possible concern is troponin ontology. In this instance, cTnI is not fully expressed within the human myocardium until 9 months of age (Sasse et al. 1993). As a result, cTnI may be limited as a useful myocardial injury biomarker at least through the neonatal period (Sasse et al. 1993; El-Kuffash and Molloy 2008). In the present study, we determined the effect of age on cTnI concentration in rats from 10 to 80 days old and in Erythrocebus patas (patas) monkeys from birth to 1-year-old as measured with a recent generation ultrasensitive immunoassay.
Materials and Methods
Studies with Rats
All procedures involving rats were approved by the Institutional Animal Care and Use Committee Center under the Food and Drug Administration’s White Oak Animal Program.
Serum obtained from Sprague-Dawley (SD) rats of the following sexes and ages were evaluated: 80 days (10 males), 40 days (10 males), 25 days (8 males), and 10 days (8 males [2 separate and 2 pooled from 3 animals each] and 3 females). Rats were purchased from Taconic (Hudson, NY) and housed individually. Neonates (10 days) were kept with their mothers. Animals 25 days or older were given standard rodent chow and water ad libitum.
These serum samples came from untreated control animals that were part of an in-house study conducted in the Division of Drug Safety laboratory of the Center for Drug Evaluation, Food and Drug Administration.
Blood samples from the ten 10-day-old rats (4 individual samples, 2 pooled samples of 3 neonates each) were collected following decapitation and from the eight 25-day-old rats (individual samples) by cardiac puncture. The ten 40-day-old rats (individual samples) and ten 80-day-old rats (individual samples) were anesthetized with isoflurane, the inferior vena cava was exposed through a midline incision, and blood was collected through a 19-gauge needle-tipped Venocath catheter inserted into the vena cava. All blood samples were centrifuged immediately following collection (20 min at 3,000× g) and the sera stored at −80°C until analyzed.
At necropsy, both rat and monkey hearts were excised and fixed in 10% neutral buffered formalin. Hearts were embedded in glycol methacrylate plastic resin. Sections (1-µm thick) of the plastic-embedded left ventricular tissue were stained with alkaline toluidine blue. Some cardiac tissues were also embedded in paraffin (5-µm sections) and stained with hematoxylin and eosin (H&E). All stained sections (2–4 sections/heart) were examined by a research pathologist. This light microscopic evaluation was blinded to age and any related biomarker data.
In SD rats, programmed cardiomyocyte cell death has been shown to affect the postnatal myocardium (Kajstura et al. 1995). This phenomenon was no longer detected by 21 days of age (Kajstura et al. 1995). In the present study, it was felt that both biochemical (apoptosis) and microscopic examination of cardiac morphology could help in identifying potential causes of age-related differences in cTnI concentrations. As a result, the terminal deoxynucleotidyl transferase deoxyuridine triphosphate nick-end labeling (TUNEL) assay was applied to sections of formalin-fixed, paraffin-embedded rat cardiac tissues. The procedure for detecting cardiac apoptosis is described elsewhere (Zhang et al. 2008). Apoptosis was detected with the CardioTACS in situ apoptosis detection kit (Trevigen, Inc., Gaithersburg, MD). Prepared myocardial sections were evaluated by light microscopy.
Studies with Monkeys
Patas monkeys were maintained under conditions approved by the American Association for Accreditation of Laboratory Animal care using protocols approved by the Institutional Animal care and Use committee of Bioqual, Inc., or the National Cancer Institute (NCI) Animal Care and Use Committee. Their maintenance and breeding have been previously described in detail (Divi et al. 2010) and are summarized here.
Groups of 3 females and 1 male patas were housed together until the female was ready to deliver. Fetal monkeys taken by cesarean section within 7 days of due date were designated “at birth” and blood was obtained either by cardiac puncture or via the femoral artery. Animals kept for longer periods were born naturally and mother reared, and for these animals (2 months to 1-year-old) blood was collected through the femoral artery. Blood was collected from a total of 11 monkeys that ranged from neonates (5), 2–6 months (3), and 1 year (3) of age. Samples were centrifuged immediately after collection (20 min at 3,000× g) and the sera stored at −80°C until analyzed.
Commercial Serum Samples from Other Animal Species
Individual samples of serum derived from various animal species were purchased from commercial sources. Fetal serum was obtained from AppliChem, Inc. (bovine and equine; Boca Raton, FL), Animal Technologies, Inc. (bovine, calf bovine and porcine; Tyler, TX), Hyclone Laboratories, Inc. (bovine; South Logan, UT), and Biofluids (bovine; Rockville, MD). Serum samples from adult animals were procured from Gibco-BRL (equine; Carlsbad, CA), AppliChem, Inc. (equine and leporine), Sigma-Aldrich Co. LLC (caprine; St Louis, MO), and Animal Technologies (porcine; Tyler, TX). It was not known whether the samples came from a single animal or were from a pool of several animals. Upon receipt, all samples were stored at −80°C until assayed.
cTnI Analysis
Cardiac troponin-I was monitored using an ultrasensitive digital assay based upon single molecule counting technology (Erenna System; Singulex, Alameda, CA; limit of detection [LoD] = 0.2 pg/ml, lower limit of quantitation [LloQ] = 0.8 pg/ml with ≤10% coefficient of variation [CV] ) as previously described (Todd et al. 2007; Schultze et al. 2008). The assay utilized frozen samples (−80°C) that were thawed and tested in 15-µl sample volumes for duplicate analysis. The volume of each sample was increased to 50 µl with calibrator diluent and subsequently added to 150 µl of assay buffer that contained microparticles coated with biotinylated cTnI capture antibody. This mixture was incubated for 1 hr in a 96-well plate. The microparticles are magnetically separated, washed, and incubated with 20-µl fluorescent dye-labeled detection antibody for 30 min. These microparticles are then washed 5 times via magnetic separation, 20 µl of elution antibody is added, and the eluted detection antibody separated from the microparticles by means of a 384-well filter plate. This eluate was passed through the Erenna Immunoassay System, and detection antibodies were quantified using single-molecule counting instrumentation. All serum samples and calibrators were assayed in duplicate, and individual results are reported as the mean of the 2 measurements. The human National Institute for Standards and Technology cTnI standard was used as the reference material for calibration. This human reference material, used with the Erenna assay, has been demonstrated previously to facilitate accurate determination of cTnI in rat, dog, and monkey serum (Schultze et al. 2008).
Ideally, monitoring of cTnI should be done in samples of blood obtained directly from a vascular site. In the present study, intravenous samples were collected from 40- and 80-day-old rats, patas monkeys, and some commercial samples. However, because of size, samples from neonatal rats and 25-day-old rats were collected by decapitation and cardiac puncture, respectively. Some commercial samples were obtained by cardiac puncture. At present, information concerning the influence of sample collection methodology on the concentration of cardiac troponin is not available. However, it is possible that samples obtained by means such as cardiac puncture could influence the amount of cardiac troponin detected by the Erenna immunoassay. All sera samples were frozen at −80°C immediately following centrifugation. The time frame for gathering the various samples was approximately 8 months. Basit et al. (2007) have shown that no significant changes in cardiac troponin concentrations occur in samples that have been frozen at −80°C for up to a year. All samples were analyzed at the same time.
Statistical Methods
The Tukey–Kramer multiple comparisons test was used to assess the significance of differences among groups in rat and monkey serum concentrations of cTnI. Alpha was set at .05 and all tests were two tailed. The InStat (GraphPad software, Inc, San Diego, CA) statistical software package was used for all analyses.
Results
SD Rats
cTnI Concentrations
The concentrations of cTnI measured in rats 10 to 80 days of age are shown in Figure 1. cTnI was detected in all rats regardless of age. The highest cTnI concentrations (34.1 ± 11.2 pg/ml) were found in the youngest animals (10 days of age). The mean concentration at this age was significantly higher than that detected at 25, 40, or 80 days (p < .05). The mean value detected at 25 days was 12.4 ± 5.0 pg/ml compared to 7.0 ± 4.6 pg/ml and 5.8 ± 3.1 pg/ml at 40 and 80 days of age, respectively. The concentrations of cTnI at 25, 40, or 80 days of age were not significantly different from each other.
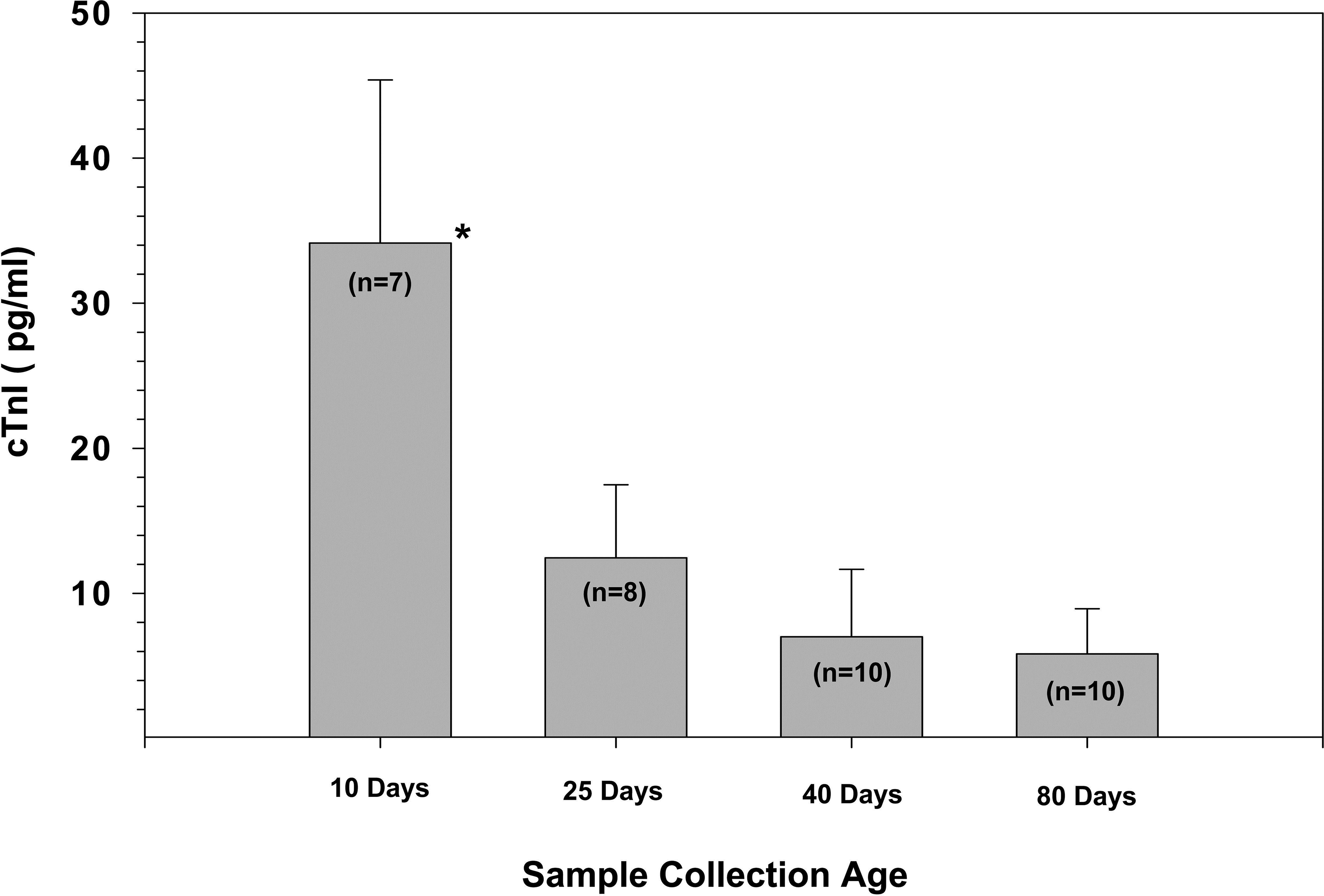
Age comparison of serum cardiac troponin I concentrations in Sprague-Dawley rats at 10, 25, 40, and 80 days of age. Significance was determined using a Mann–Whitney t-test with α set at p < .05. Results are shown as M ± SD, with cTnI concentration in pg/ml. cTnI = cardiac troponin I.
Cardiac Pathology
Myocyte morphology differed between adult and neonatal hearts. Heart sections from 80-day-old rats stained with H&E showed regularly arranged cardiac myocytes, with abundant eosinophilic cytoplasm and regular cross striations. Also noted were centrally located and light-stained nuclei in the cytoplasm (Figure 2A). In contrast, myocytes from 10-day-old rats had a very dense appearance, with basophilic cytoplasm that contained only a rarely observable cross striation, with nuclei of nonuniform shape and size that were deeply stained with hematoxylin. In addition, various forms of cytoplasmic vacuoles were observed in 10-day-old hearts (Figure 2C). The TUNEL assay staining was negative in 80-day-old adult hearts (Figure 2B), while blue-stained apoptotic nuclei accompanied by nuclear and cytoplasmic condensation were prominent features in almost all of the myocytes from 10-day-old hearts (Figure 2D). Fewer cytoplasmic vacuoles and apoptotic cells were noted in the myocardium of 25-day-old rats (photo not shown). By 40 days, cytoplasmic vacuolization was minimal (photo not shown).
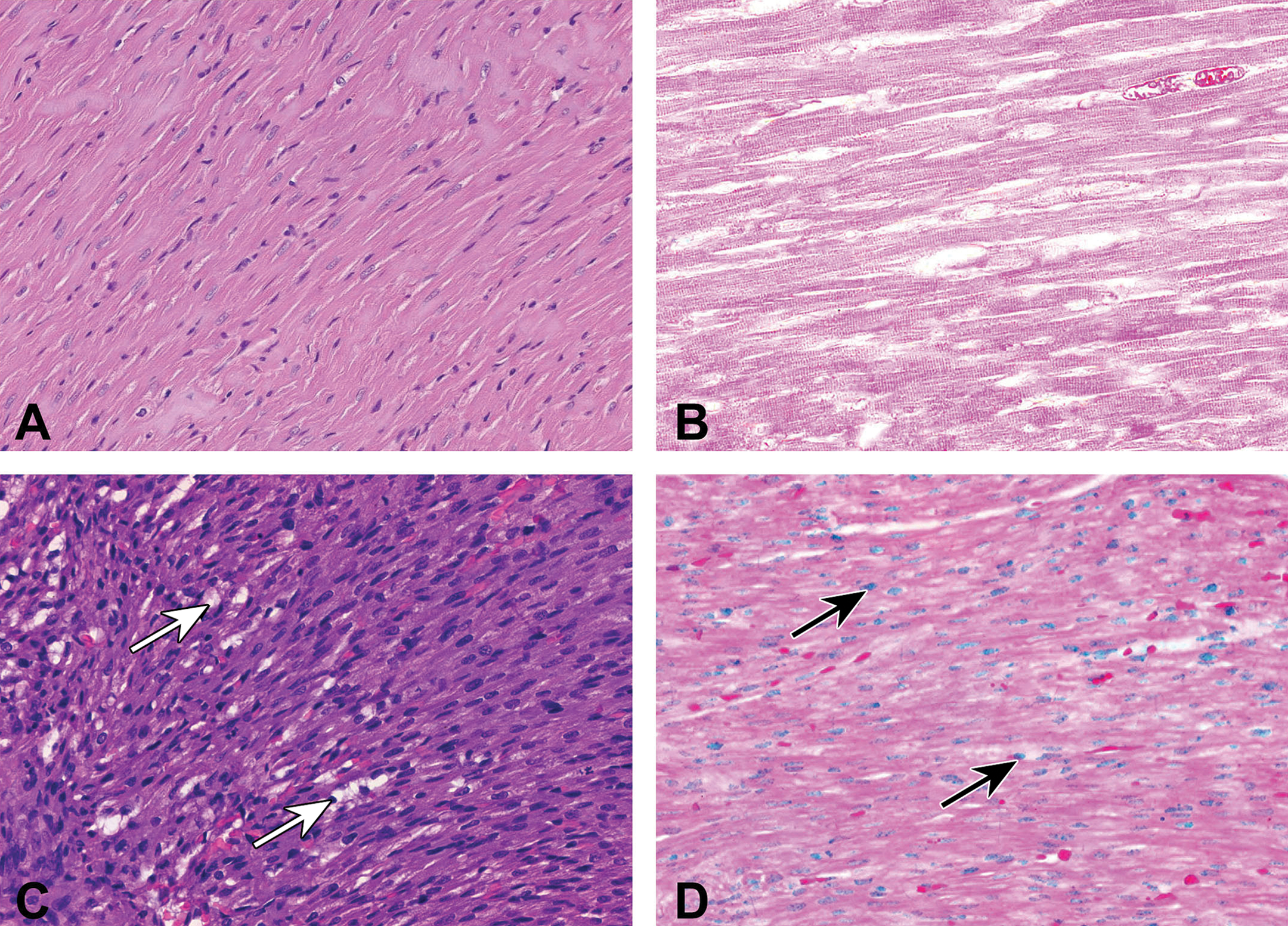
Light micrographs showing cardiac morphology and incidence of apoptosis in the left ventricle from 80-day-old (A and B) and 10-day (C and D) Sprague-Dawley rats. Arrows indicate examples of cytoplasmic vacuolation (C) and positive TUNEL staining for apoptosis (D). Glycol methacrylate-embedded, H&E and TUNEL-stained, 1-μm thick plastic sections. Original magnification 200×. TUNEL = deoxyuridine triphosphate nick-end labeling; H&E = hematoxylin and eosin.
Patas Monkeys
cTnI Concentrations
cTnI was detected in all 11 patas monkeys evaluated. Baseline concentrations of cTnI were highest in blood taken from monkeys at birth (Figure 3). The mean cTnI concentration of 571 ± 596 pg/ml was considerably higher than that found in either 2- to 6-month-old animals (4.4 ± 3.4 pg/ml) or 1-year-old animals (0.76 ± 0.05 pg/ml; p < .05; Figure 3). However, because of the great variability in the cTnI concentrations found in the birth samples, this difference was not statistically different from that of the older animals.
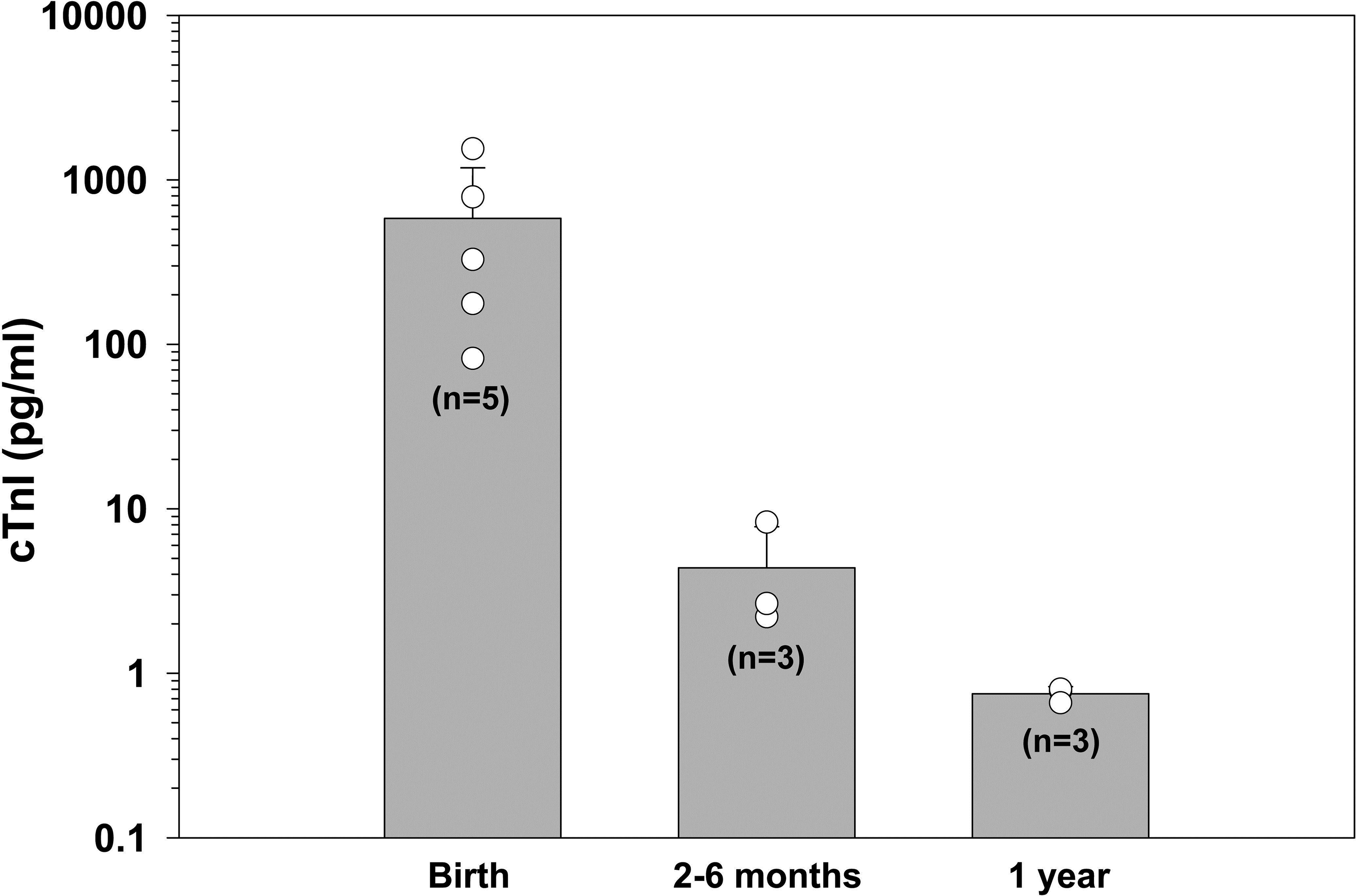
Age comparison of serum cardiac troponin I concentrations in patas monkeys at birth, 2, 6 months, and 1 year of age. Significance between birth and 1 year of age was determined using a Mann–Whitney t-test with α set at p < .05. Results are shown as M ± SD with cTnI concentration in pg/ml. cTnI = cardiac troponin I.
Cardiac Pathology
Hearts from monkeys at birth (n = 6) and 1-year-old (n = 3) were available for evaluation. Light microscopic examination of ultrathin toluidine blue-stained slides from 1-year-old monkeys showed normal myocyte nuclear, myofibrillar, and cytoplasmic morphology (Figure 4A and B). In contrast, the presence of numerous and varied sized cytoplasmic vacuoles and loss of myofibrils were prominent morphologic alterations observed in the hearts of newborn patas monkeys (Figure 4C and D).
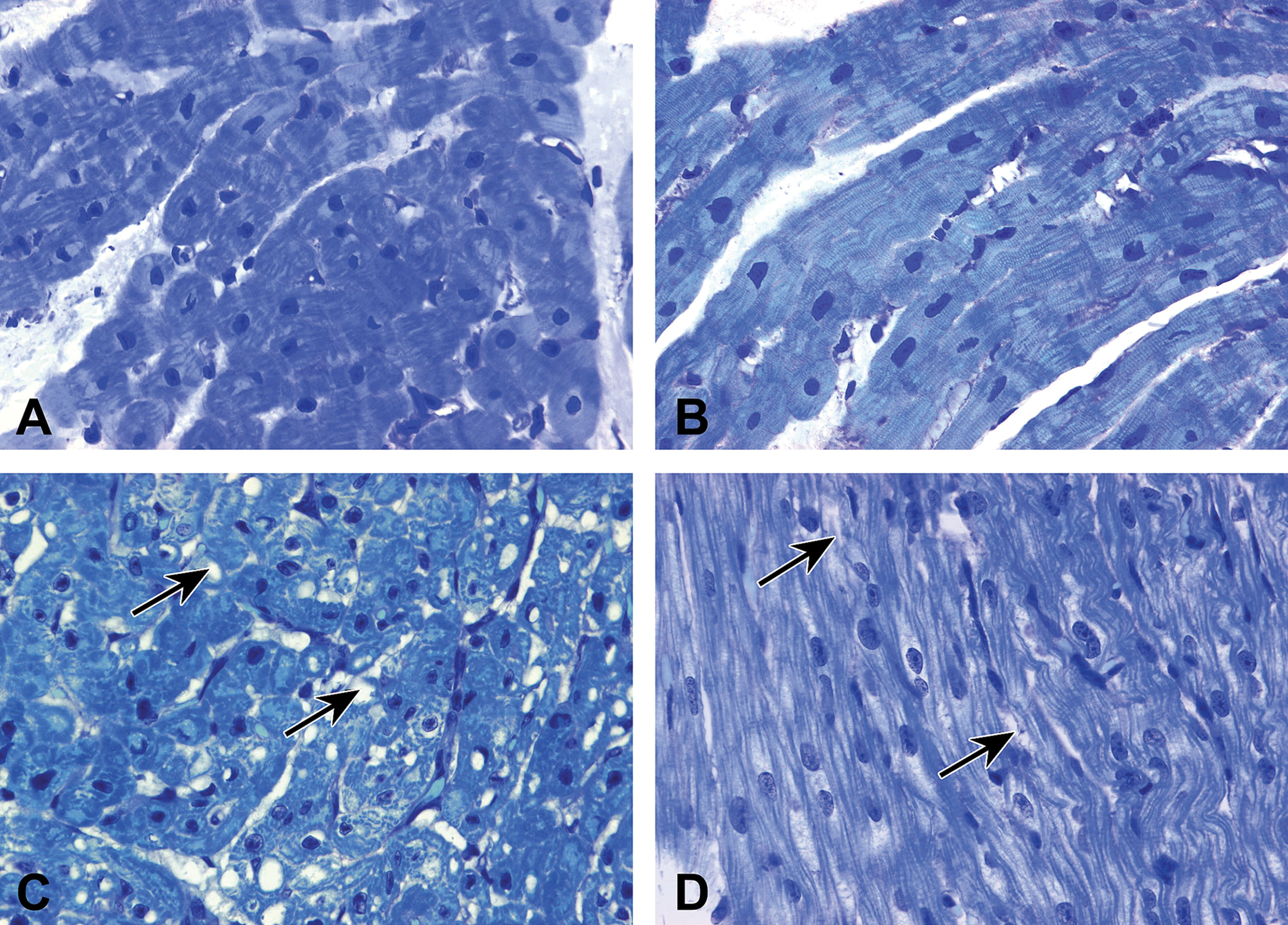
Light micrographs from left ventricle showing cytoplasmic vacuolization (C, arrows) and myofibrillar loss (D, arrows) in newborn patas monkeys. Compare with 1-year-old patas monkeys (A and B). Glycol methacrylate–embedded, alkaline toluidine blue, 1-μm thick plastic sections. Original magnification 630×.
Animal Sera from Other Animal Species
cTnI Levels
cTnI was measured in sera from a variety of other animal species using commercially obtained samples. Blood samples were collected in various ways. In most instances, fetal samples were obtained by cardiac puncture. cTnI was detected in all samples analyzed. When fetal and adult samples were compared, the highest concentrations of cTnI were found in the fetal sera, particularly the bovine samples (656–54,752 pg/ml; Table 1). In contrast, except in one instance, cTnI concentrations ranged from 1.2 to 5.8 pg/ml in sera of adult animals (Table 1).
Cardiac troponin I concentrations in various commercially available animal sera.
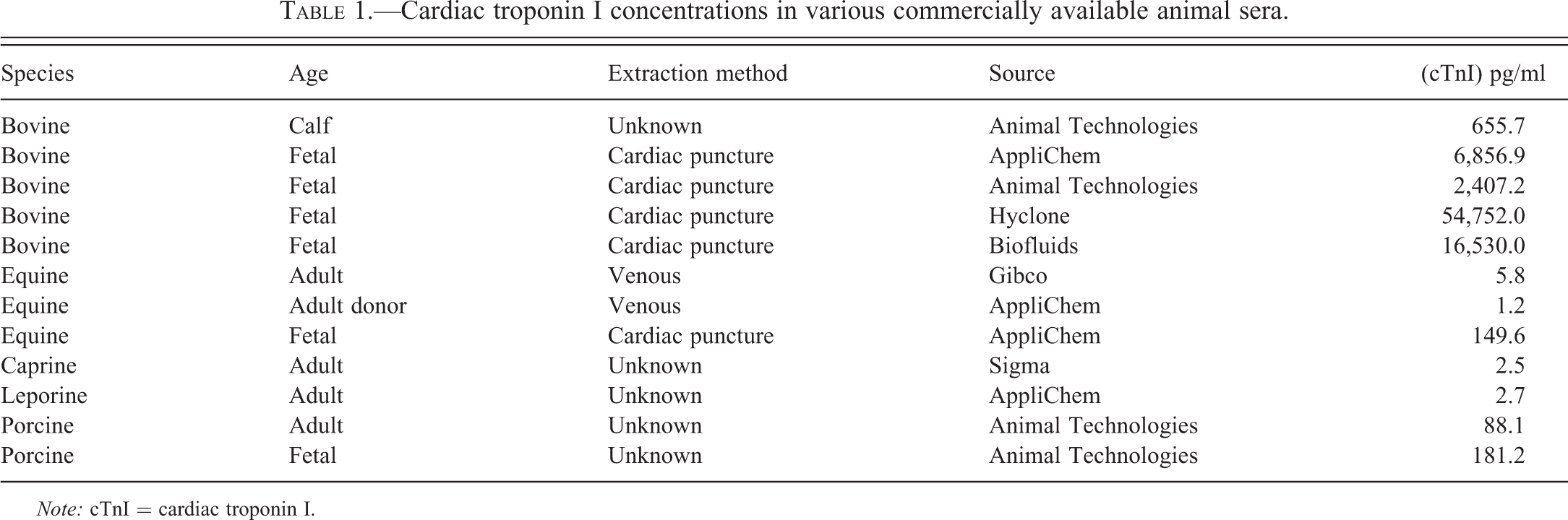
Note: cTnI = cardiac troponin I.
Discussion
Masson et al. (2013), using a high sensitivity cTnT immunoassay, found that age influenced normal baseline plasma concentrations of cardiac troponin. They reported higher baseline cTnT concentrations in elderly individuals (73 ± 5 years). Cardiac troponin has also been detected in the blood of apparently healthy neonates. Initially, data obtained from studies monitoring cardiac troponin concentrations in very young populations were contradictory. Some studies report similar cTnI concentrations in young and old populations (Hirsch et al. 1997), whereas other studies have found distinct differences between baseline cardiac troponin concentrations in healthy adult and infant populations (Kanaan and Chiang 2004). Subsequent studies (reviewed by El-Kuffash and Molloy 2008) have determined that normal baseline cTnT concentrations are considerably higher (by a factor of 10) in preterm infants than in apparently healthy full-term newborns. In both instances (term and preterm infants), normal troponin concentration were higher than that the reported cTnT reference range for adults (Alpert et al. 2000). Normal cTnI concentrations in healthy term and preterm infants can also vary widely depending on which immunoassay is used (El-Kuffash and Molloy 2008).
Reports of age-related differences in cardiac troponin concentrations are less common in preclinical studies. The present study, which included 2 species and an ultrasensitive immunoassay to monitor serum levels of cTnI, examined the effect of specific age (10–80 days for rats and newborn to 1-year patas monkeys) on cTnI concentrations. Our findings clearly show that cTnI is present at all ages but that the highest concentrations were detected in the youngest (neonatal) rats. In the second species in this study, the mean concentration of cTnI found in the newborn monkeys was considerably higher than that noted in animals at 1-year-old. There was, however, a high degree of interindividual variability in cTnI levels noted in the newborn monkeys, possibly reflecting the maturity of the infant or the circumstances of the cesarean section performed. Previously, Trevisanuto et al. (1998) reported high interindividual variability among apparently homogeneous young human infants.
Another example of age as a factor in levels of cardiac troponin was reported in chicks. Maxwell, Robertson, and Mosely (1995a) found that increases in cTnT levels were frequent and often severe in newly hatched chicks. The individuals with elevated cTnT concentrations often developed a cardiomyopathy associated with the usually fatal broiler ascities syndrome (Maxwell, Robertson, and Mosely 1995a). The earlier reports in chickens and the findings with rats and monkeys in the present study are all consistent with previous clinical observations that demonstrate higher concentrations of cardiac troponins in pediatric populations compared to adults (Trevisanuto et al. 1998; Soldin et al. 1999; Baum et al. 2004; Lipshultz et al. 2008).
There has been some question as to whether monitoring cTnI is as useful as monitoring cTnT at early biological ages. Clark, Newland, and Thorburn (2001) imply that cTnT is less affected by developmental changes than is cTnI. The troponin I isoform that predominates in fetal heart tissue is adult slow-twitch skeletal muscle troponin I (ssTnI; Saggin et al. 1989). Sabry and Dhoot (1989) detected this developmental isoform of ssTnI in neonatal cardiac tissue from both rats and chickens. In humans, this isoform is downregulated during maturation, so that by 9 months of age only cTnI is present (Sasse et al. 1993). Theoretically, such a lack of early full early expression could limit the overall applicability of cTnI as a biomarker to detect changes in neonatal myocardial integrity. The antibodies used in the Erenna immunoassay are specific for cTnI and are unlikely to cross-react with ssTnI because the N-terminal sequence recognized in cTnI is absent in skeletal troponin (Todd et al. 2007).
In the present study, the concentrations of cTnI, as monitored by the Erenna immunoassay system, were compared in normal SD rats ranging in age from 10 to 80 days. Although cTnI was detected in all rats concentrations in the very youngest animals (10 days) were significantly higher than those found in animals that were 25, 40, or 80 days old. At 40 days, the cTnI concentrations declined and were essentially similar to the normal concentrations reported for SD rats in previous studies (Herman et al. 2011). Newborn patas monkeys had significantly higher concentrations of cTnI than those in animals that were 2 months or older. These observations suggest that cTnI could provide appropriate cardiac biomarker information over a spectrum of ages, including the very young.
Clinically, cardiac troponin concentrations vary with age and appear to spike shortly after birth. Almeida et al. (2011) detected low concentrations of cTnI in human cord blood. However, in otherwise normal neonates’ maximal increases in cTnI concentrations peaked between 48 and 72 hr after birth and declined by 50% by the 10th day of life (Almeida et al. 2011). The finding that elevated serum troponin concentrations were more common in normal neonatal blood than in umbilical cord blood is consistent with other studies that found maximal cardiac troponin concentrations were maximal between 48 and 96 hr after birth (El-Kuffash and Molloy 2008; Lipshultz et al. 2008).
Studies to determine whether a similar cardiac troponin pattern occurs in animals are more limited. Maxwell, Robertson, and Mosely (1995b) noted that the highest cTnT levels monitored in chickens occurred within 24 hr after birth. In the present study, mean cTnI concentrations detected in rats at 10 days of age (34.2 ± 11.3 pg/ml) were significantly higher than that detected in animals that were 25, 40, or 80 days old. The youngest age from which blood samples could be consistently collected was 10 days, so whether higher cTnI concentrations would be present at a younger age is not known. The concentrations of cTnI detected in the 40- and 80-day-old rats are similar to the normal concentrations of cTnI reported previously in adult male SD rats (Herman et al. 2011).
Serum concentrations of cTnI were evaluated at birth in patas monkeys. At this time, serum levels of cTnI were considerably higher than those found in samples from animals 2 months or older. The concentration of cTnI detected in the newborn monkeys was considerably higher than that of the youngest rats perhaps because of the difference in the initial age at which the samples were collected (10 days for rats and birth for monkeys). Because we tested monkeys at only one early time point (birth), we could not determine whether this concentration of cTnI represented the maximal levels of cTnI in Patas monkeys. cTnI concentrations varied so widely that the mean concentration did not differ significantly from the much lower and less variable concentrations detected at 2 months and at 1 year of age. Schultze et al. (2008), using similar immunoassay methodology, also reported low cTnI concentrations in normal young cynomolgus monkeys (5.3 and 4.4 pg/ml for males and females, respectively). Thus, the inverse relationship between age and cTnI concentrations appears to occur in both humans and animals.
The exact mechanism responsible for higher postnatal cardiac troponin concentrations remains to be elucidated. Lipshultz et al. (2008) surmised that the elevated concentrations in some healthy newborns might be associated with myocardial remodeling, occult myocardial injury, or both (Lipshultz et al. 2008). White (2011) has identified at least 6 different pathogenic processes that could increase troponin concentrations (4 of which are subsequently mentioned). Ischemia-induced myocardial necrosis is a frequently identified mechanism that elevates cardiac troponin concentrations. Ischemia or other changes in cell viability may also increase cardiac troponin levels by altering cell wall permeability and releasing proteolytic troponin degradation products (White 2011). The development and release of membranous blebs may also cause troponin to be released from cardiac cells as occurs in ischemic myocytes in tissue culture (White 2011). We did not determine the state of oxygenation in the rat or monkey hearts in the present study. However, the elevated cardiac troponin concentrations noted in the neonatal rats and newborn monkeys likely occurred in the absence of ischemia because myocyte necrosis was not a common morphological observation in either neonatal rats or newborn monkeys. Normal myocyte turnover may also cause cardiac troponin release (White 2011). Bergmann et al. (2009) reported a small turnover of cardiac cells each year which may responsible for the presence of cardiac troponins in healthy people. It is conceivable that this process could occur at a higher rate for a limited time at a very young postnatal age.
The transition from prenatal to neonatal involves multiple physiological changes and adaptations that directly affect myocardial structure and function (Almeida et al. 2011). One of the consequences of these changes may be to enhance programmed cell death (apoptosis; Kajstura et al. 1995). The frequency of myocardial apoptosis has been reported to be age related. In mice, apoptotic activity is higher during the first 2 weeks of life than it is in adult mice (Fisher, Langille, and Srivastava 2000). We found a similar result in the present study. In this instance, the morphological characteristics of neonatal rat and newborn monkey hearts differed from those of older animals. In the youngest hearts, we also noted increased numbers of mitotic cells and apoptotic cells. These types of cells were not apparent in older rats and monkeys. Apoptosis, in which the integrity of the cell membrane is maintained, occurs in conjunction with the activation of caspases. These enzymes can cleave structural proteins that could promote the release of cardiac troponins (Narula et al. 1996).
Cultured isolated rat cardiomyocytes are commonly used as a model to identify potentially cardiotoxic substances. Several studies have evaluated the potential utility of cardiac troponins as biochemical biomarkers of injury in isolated cultured cardiomyocytes exposed to certain substances (Li et al. 2004; Adamcová et al. 2007; Hessel, Atsma, et al. 2008; Hessel, Michielsen, et al. 2008). An important component of the media used to culture the isolated myocytes has been animal serum. For example, Hessel, Atsma, et al. (2008) and Hessel, Michielsen, et al. (2008) used fetal bovine serum as part of the culture media in their studies. In the present study, we measured cTnI concentrations in serum samples from several species collected by different means and obtained from several different commercial sources. cTnI was detected in all samples. However, as noted with the younger animals, samples from fetal animals had considerably higher cTnI concentrations did samples from adults. Because Li et al. (2004), Adamcová et al. (2007), and Hessel, Atsma, et al. (2008) and Hessel, Michielsen, et al. (2008) used different versions of the cTnI assay, it is not possible to determine whether the amount of serum added to the culture media had any effect on the ultimate cTnI concentrations The present study demonstrated that cTnI concentrations are not uniform at all ages. An important observation was that high levels of cTnI appear in the serum early in life. The age-related differences appear to be consistent across species as noted in animals as diverse as the rat and the monkey. The elevated concentrations of cTnI appear to be associated with ongoing maturation processes that are causing intracellular modifications to immature myocytes. Thus, the higher concentrations of cTnI we detected in the present study could reflect myocyte turnover and apoptosis, both of which appear to occur at a higher rate in the youngest animals. High normal concentrations could be a factor when monitoring cTnI with an ultrasensitive assay and levels would certainly need to be assessed before cTnI could be considered a reliable biomarker for either in vivo or in vitro cardiotoxicity studies.
The present study detected higher levels of cTnI in 10-day-old rats and newborn patas monkeys than older animals. However, because of limited sampling times we were not able to determine whether the concentrations detected represented the overall maximal levels that occurred in these animals. The predominant form of troponin I in the young myocardium is reported to be ssTnI. We were not able to monitor levels of ssTnI to see how levels of this protein compared with that of cTnI. We were also not able to determine whether older ages exerted any influence on serum concentrations of cTnI.
Footnotes
The findings and conclusions in this article have not been formally disseminated by the Food and Drug Administration and should not be construed to represent any agency determination or policy.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
