Abstract
Cystic degeneration of the adrenal cortex is a common age-related finding in the Sprague-Dawley (SD) rat strain occurring more frequently in females. Compression of the adjacent cortex, a common hallmark of benign adrenal cortical tumors, often accompanies foci of cystic degeneration, creating a diagnostic challenge. Accurately differentiating these relatively common degenerative changes from proliferative lesions is critical in safety assessment studies. Cystic degeneration typically arises in the zona fasciculata of the adrenal cortex and often causes compression along the margin of the lesion. The degenerating cells are large, with abundant eosinophilic cytoplasm, or contain clear cytoplasmic vacuoles. Mitotic figures are generally uncommon. In many cases, cystic degeneration appears to arise in areas of hypertrophy in the zona fasciculata. In contrast, adrenal cortical hyperplasia and adrenal cortical adenoma are frequently comprised of smaller cells that cause compression of adjacent cortex, and in some cases mitotic figures are observed. Cytological detail and growth patterns should be considered more useful criteria than compression alone for separating degenerative cystic lesions from proliferative lesions in the adrenal cortex of SD rats.
Introduction
Spontaneous cystic degeneration characterized by cell loss and formation of cystic spaces often resulting in compression of the surrounding cortical tissue is used to describe severe forms of vacuolization in the adrenal cortex of rats (Hamlin and Banas 1990). The lesion is more common in Sprague-Dawley (SD) rats than Fischer 344 (F344) rats. Congestion and/or hemorrhage are common features, leading to the use of the term peliosis (Dhom et al. 1981); however, more recently cystic degeneration is considered the preferred terminology. The lesion is more common in female rats and the incidence increases in male rats treated with estrogens (Gopinath, Prentice, and Lewis 1987). Increased incidence of cystic degeneration following exposure to polyhalogenated aromatic hydrocarbons was reported in several recent National Toxicology Program (NTP) studies using female SD rats. In our experience, certain stages of cystic degeneration may present a diagnostic challenge because the lesions are compressive and may have an appearance similar to proliferative lesions. The goal of this review is to describe and illustrate cystic degeneration in the SD rat and establish defined criteria to distinguish this commonly reported lesion from hyperplasia, hypertrophy, focal vacuolation, and adenoma of the adrenal cortex. Cortical carcinomas were not included in this report because the lesions do not create a diagnostic challenge.
Materials and Methods
Animals were fed Certified Rodent Diet #8728CM (Harlan Laboratories, Inc.) and water ad libitum. Animals were individually housed in stainless steel cages. All study procedures were performed in compliance with the Animal Welfare Act, the Guide for the Care and Use of Laboratory Animals, and the Office of Laboratory Animal Welfare. Moribund animals or those found dead during the course of the study were necropsied as soon as possible. At the end of the 104-week period, surviving animals were euthanized and necropsied according to the protocol and amendments. Protocol specified tissues were taken from all animals, preserved with 10% neutral buffered formalin and processed routinely for histopathological evaluation.
Primary evaluation of adrenal glands from control male and female SD rats from two 104-week carcinogenicity studies (250 animals) conducted by Covance Laboratories, Inc., was performed by 2 veterinary pathologists, followed by an independent evaluation by a third veterinary pathologist. These studies were used to evaluate the criteria for common degenerative and proliferative lesions of the adrenal cortex. Both adrenal glands from most animals were available for examination. Disagreements in diagnoses were resolved by a consensus between the 3 pathologists. The combined incidences of adrenal cortical lesions in control rats from these 2 studies are represented in Table 1 and also in the Results section.
Incidence (%) of spontaneous adrenal cortical lesions in control Sprague-Dawley rats from two 104-week carcinogenicity studies.
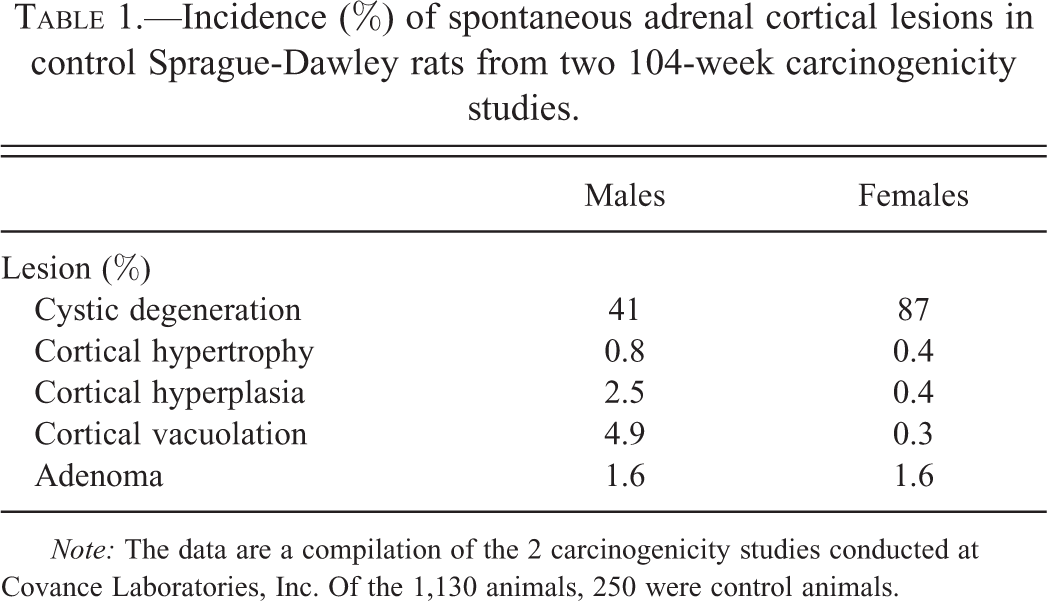
Note: The data are a compilation of the 2 carcinogenicity studies conducted at Covance Laboratories, Inc. Of the 1,130 animals, 250 were control animals.
After the initial review of the first 2 studies, an additional 2-year study conducted by the NTP in female SD rats was also reviewed because of an increased incidence of cystic degeneration, cytoplasmic vacuolization, and hyperplasia attributed to exposure to the test article (NTP 2006b). The goal of this second review was to determine whether cystic degeneration induced by the test chemical was morphologically similar to spontaneously occurring lesions and to review differences between cytoplasmic vacuolation and cystic degeneration. Adrenal glands (usually two) from 261 female SD rats were examined. The adrenal gland had already been evaluated as a target tissue by a study pathologist and quality assessment pathologist as part of the NTP Pathology Peer Review process. There were few diagnostic disagreements between the reviewing pathologists, and selected lesions from the NTP study were also reviewed by a second pathologist.
Results
Common benign proliferative and/or degenerative changes noted in the adrenal cortex in the two studies conducted at Covance Laboratories, Inc., were cystic degeneration, cortical hypertrophy, cortical hyperplasia, cortical vacuolation, and cortical adenoma. Cystic degeneration was a common finding with a higher incidence in females than males (Table 1). Extramedullary hematopoiesis and inflammatory cell infiltrates were also noted but were not considered part of the degenerative or proliferative cortical lesions included in this report.
Cystic Degeneration
The incidence of cystic degeneration in the two studies conducted at Covance Laboratories, Inc., was 41% and 87% in males and females, respectively. These lesions were well circumscribed and appeared to arise in the zona fasciculata. Cystic degeneration in the adrenal cortex of animals sacrificed at early time points were characterized by foci of enlarged vacuolated cells (Figure 1A), typically located toward the medulla, or multifocal dilated spaces within the cortex (Figure 1B). At scheduled or unscheduled sacrifices at later time points, lesions were characterized by large blood filled spaces or large proteinaceous fluid cysts resulting from cortical cell loss in these areas (Figure 2). In some cases, the resulting loss of cortical cells and subsequent pooling of blood/proteinaceous fluid caused an extensive area of the cortex to be replaced by one or more cavities (Figure 3) that occasionally contained fibrin thrombi (Figure 4). Occasional lesions at later time points were well-circumscribed foci composed of hypertrophied and vacuolated cells causing slight some compression of the adjacent cortex (Figures 5A and inset) or presented as a poorly demarcated lesion of hypertrophied and vacuolated cells (Figure 5B and inset).
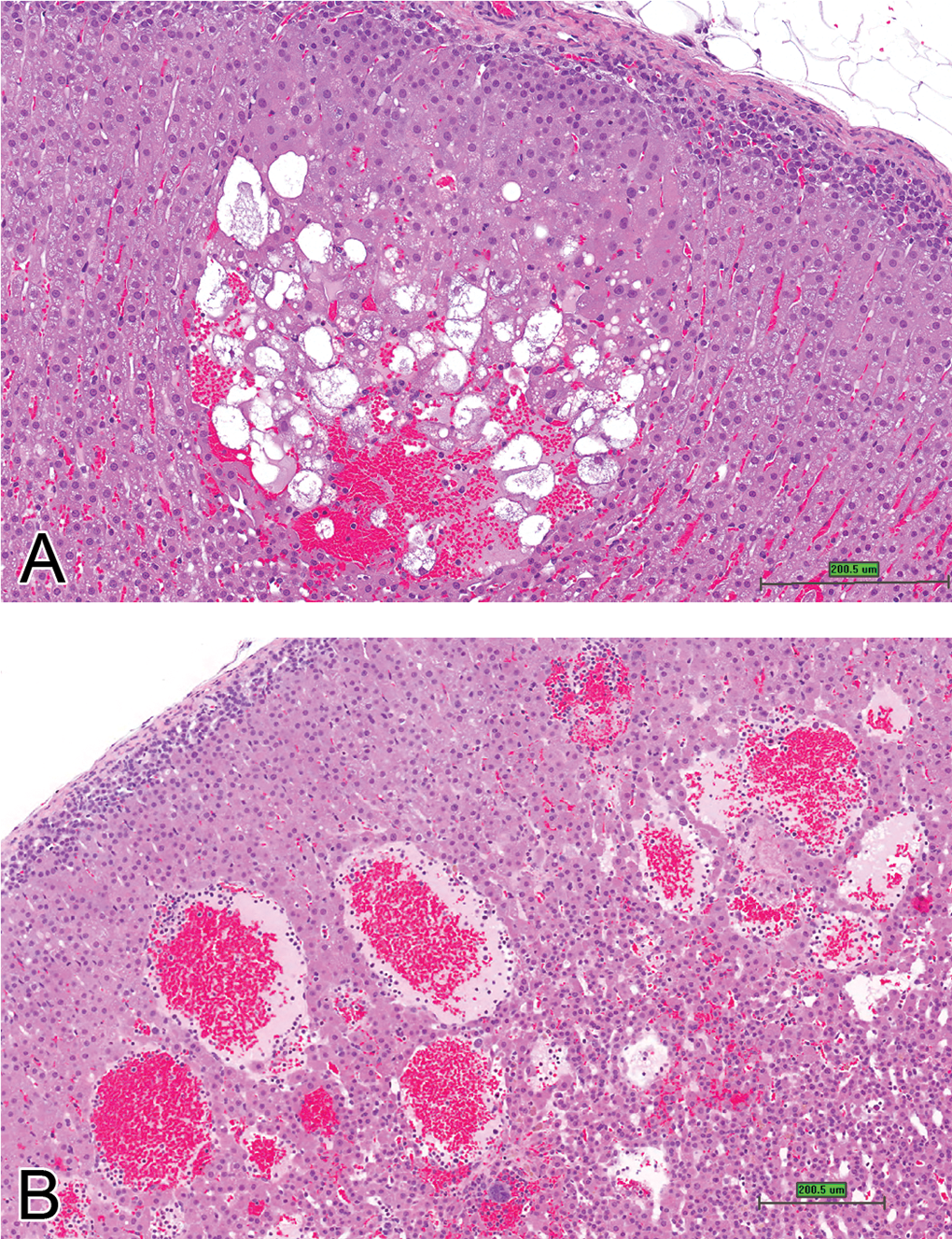
Adrenal cortex, depicting early cystic degeneration within a focus of hypertrophy located in the zona fasciculata (A) and in (B) mainly dilated sinuses within zona fasciculata (H&E stain, scale bar is approximately 200 μm).
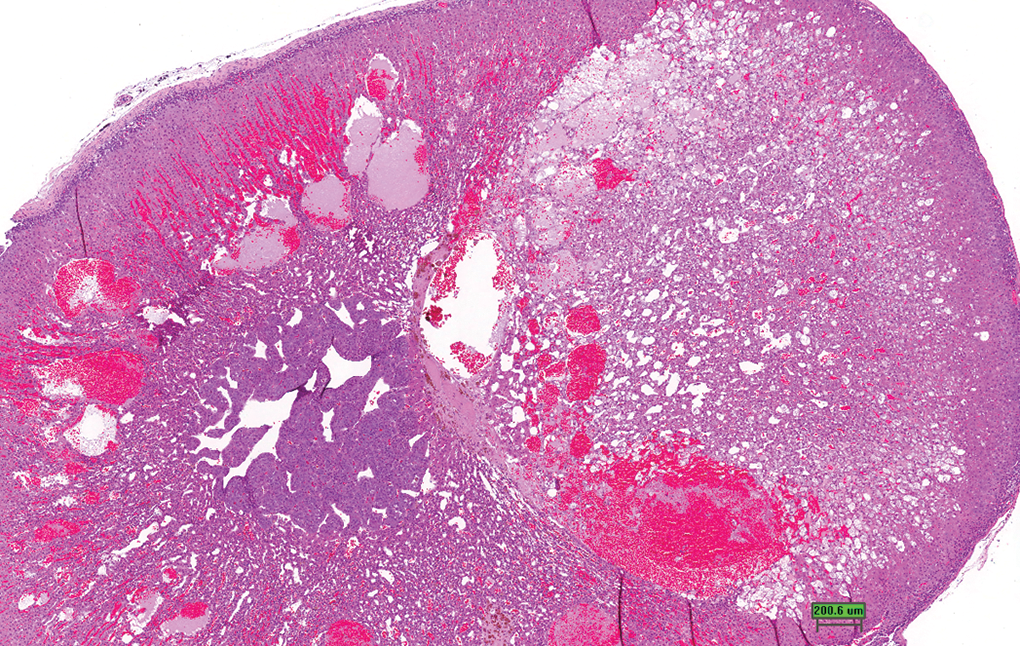
Adrenal gland. Cystic degeneration. More extensive loss of cortical cells and dilated sinuses containing blood or proteinaceous fluid. Note compression of the inner cortical cells by large focus of cystic degeneration (H&E stain, scale bar is approximately 200 μm).
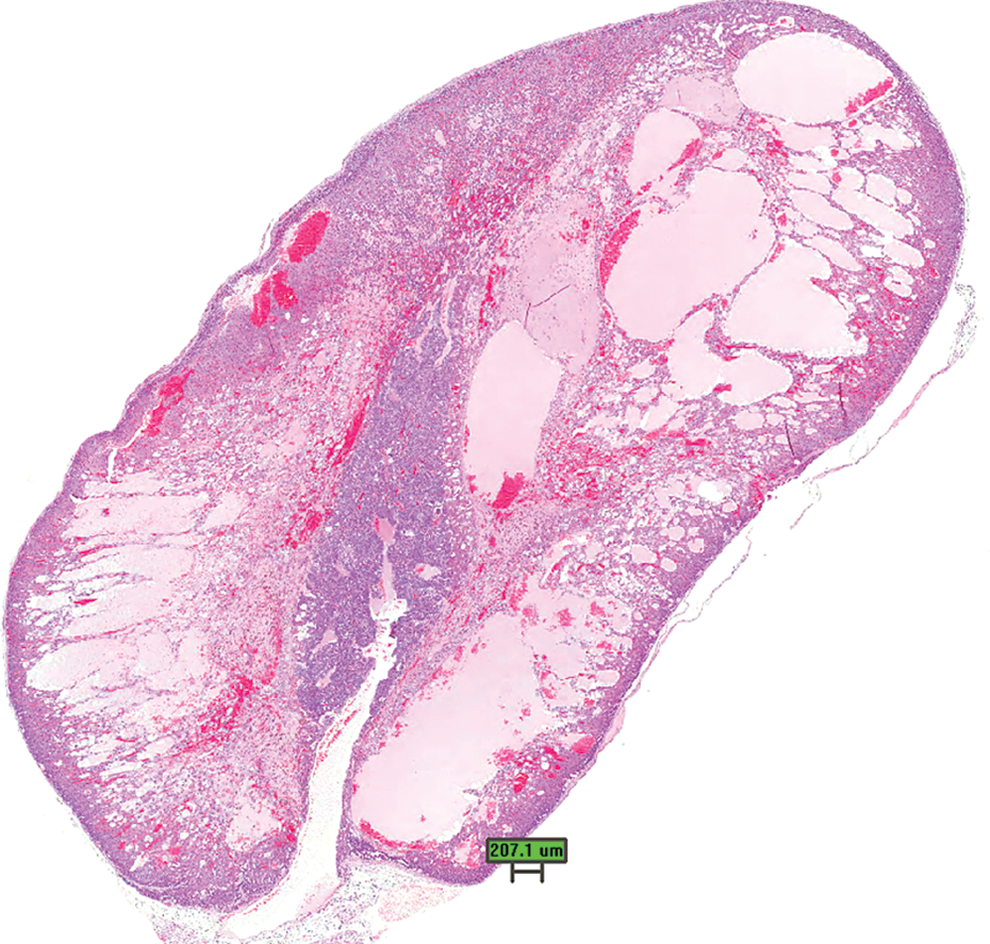
Adrenal gland. Late stages of cystic degeneration, observed in animals sacrificed at scheduled sacrifices or later time points. Marked loss of cortical cells with formation of large cystic spaces containing blood or proteinaceous fluid (H&E stain, scale bar is approximately 200 μm).
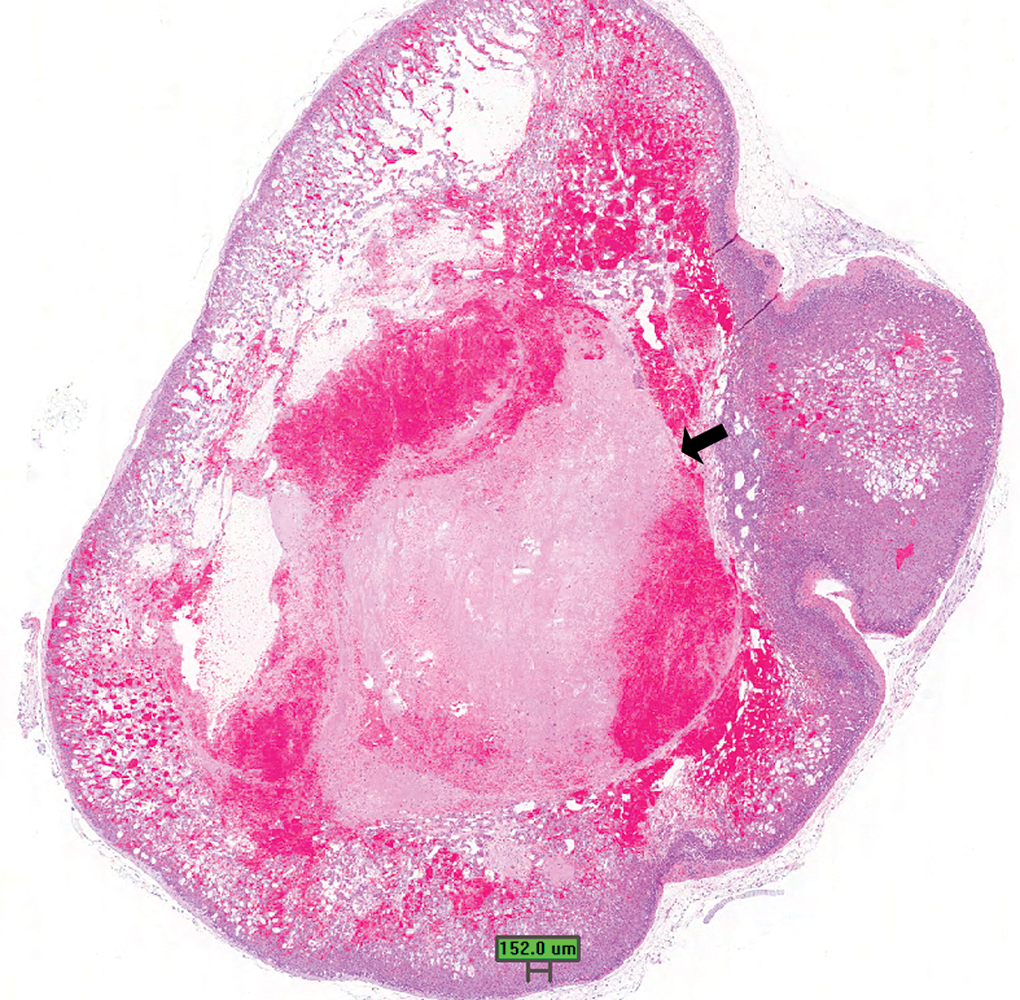
Adrenal gland. Another presentation of well-developed cystic degeneration lesion with large fibrin thrombus (arrow; H&E stain, scale bar is approximately 150 μm).
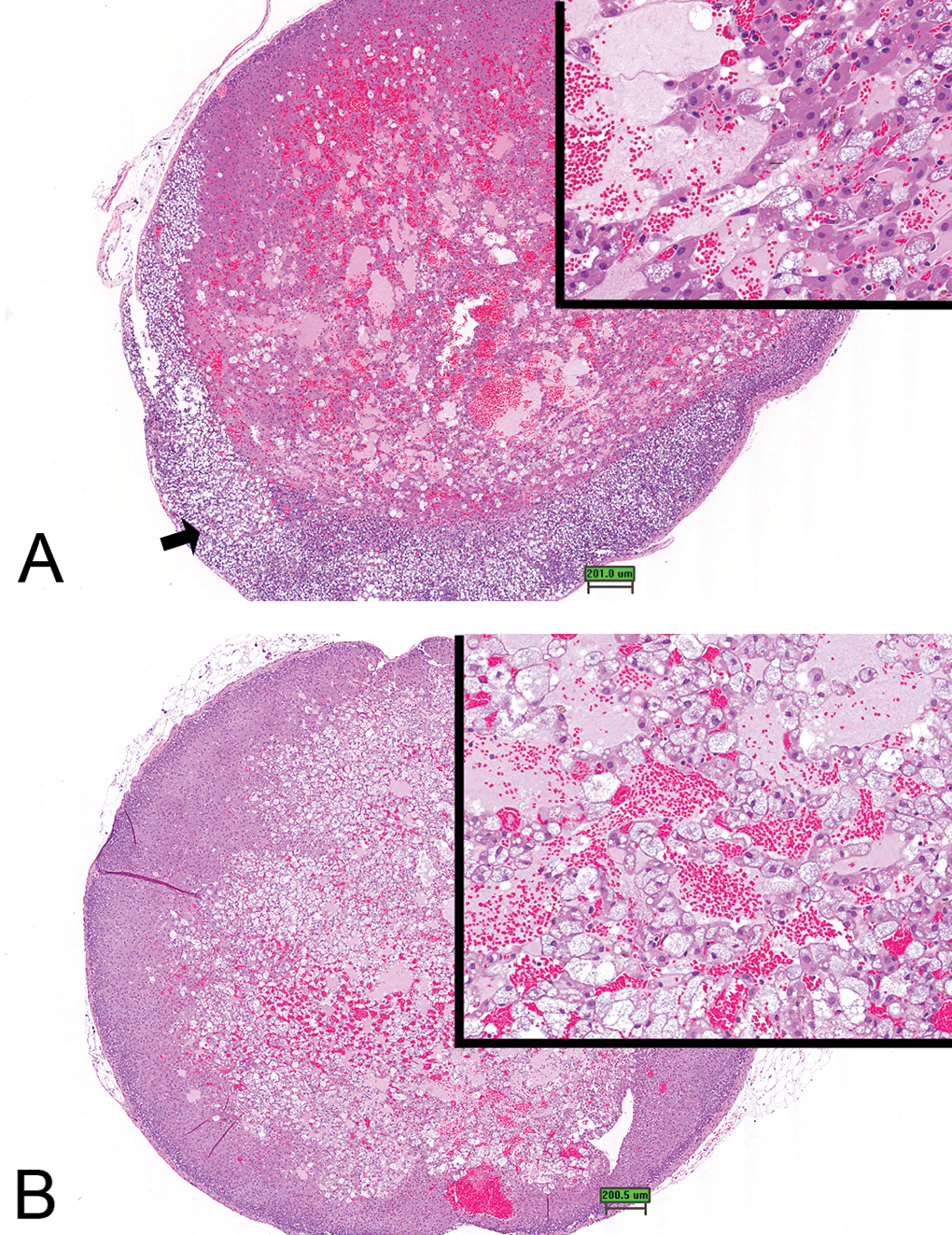
Adrenal cortex. (A) Well circumscribed, large focus of cystic degeneration. Note vacuolated and hypertrophied cells, cystic spaces, and loss of cortical cells within this presentation of cystic degeneration (inset). Also note vacuolation and atrophy of zona glomerulosa cells (arrow). (B) Cystic degeneration in adrenal cortex. Note nonencapsulated, poorly demarcated lesion with cystic spaces within the cortex. Inset—higher magnification of cells within focus (H&E stain, scale bar is approximately 200 μm).
Adrenal Cortical Hypertrophy
These lesions were characterized by well-defined triangular to crescent-shaped foci arising in the zona glomerulosa or zona fasciculata that were composed of sheets of large cells with finely vacuolated, eosinophilic, or granular cytoplasm. Focal hypertrophy arising in the zona glomerulosa often has a triangular shape, with cells that tend to stain less intensely than the surrounding cortex (Figure 6A). In contrast, hypertrophic foci located in the zona fasciculata are composed of large plump cortical cells containing abundant eosinophilic cytoplasm (Figure 6B). Compression of the surrounding cortex was occasionally noted in larger foci. Foci of hypertrophy occurred bilaterally or unilaterally, and were present as single, multiple or diffuse lesions in the adrenal cortex. Adrenal cortical hypertrophy was present in 0.4% of evaluated females and 0.8% males.
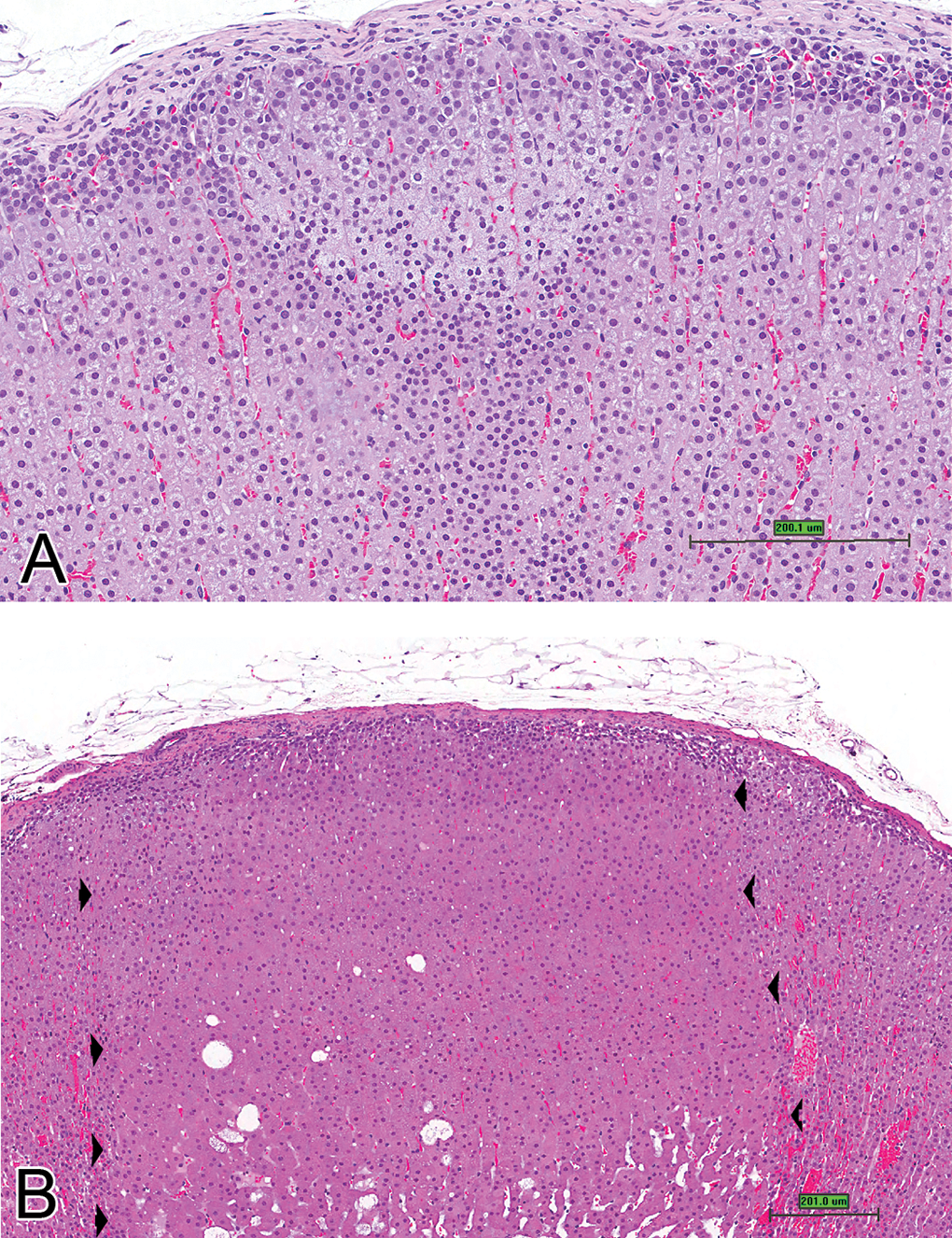
Adrenal cortex. (A) Cortical hypertrophy arising in zona glomerulosa. Note paler staining cells within focus. (B) Cortical hypertrophy. Arrow heads delineate focus within zona fasciculata. Note larger cells with abundant eosinophilic cytoplasm within focus (H&E stain, scale bar is approximately 200 μm).
Adrenal Cortical Hyperplasia
Hyperplasia is typically diffuse or focal and is located mainly in the zona fasciculata with occasional encroachment of the zona reticularis. In this review, focal hyperplasia was more common than hypertrophy, with an incidence of 0.4% in females and 2.5% in males. Cells in these foci were generally smaller with more basophilic cytoplasm compared with cells in adjacent cortex. The normal architecture of the zona fasciculata was generally maintained with cells arranged perpendicular to the capsule. A characteristic feature of hyperplasia was bulging of the cords into the adjacent parenchyma along the periphery of the lesion. Compression of adjacent parenchyma was generally minimal, and mitoses were not commonly noted (Figure 7A and 7B).
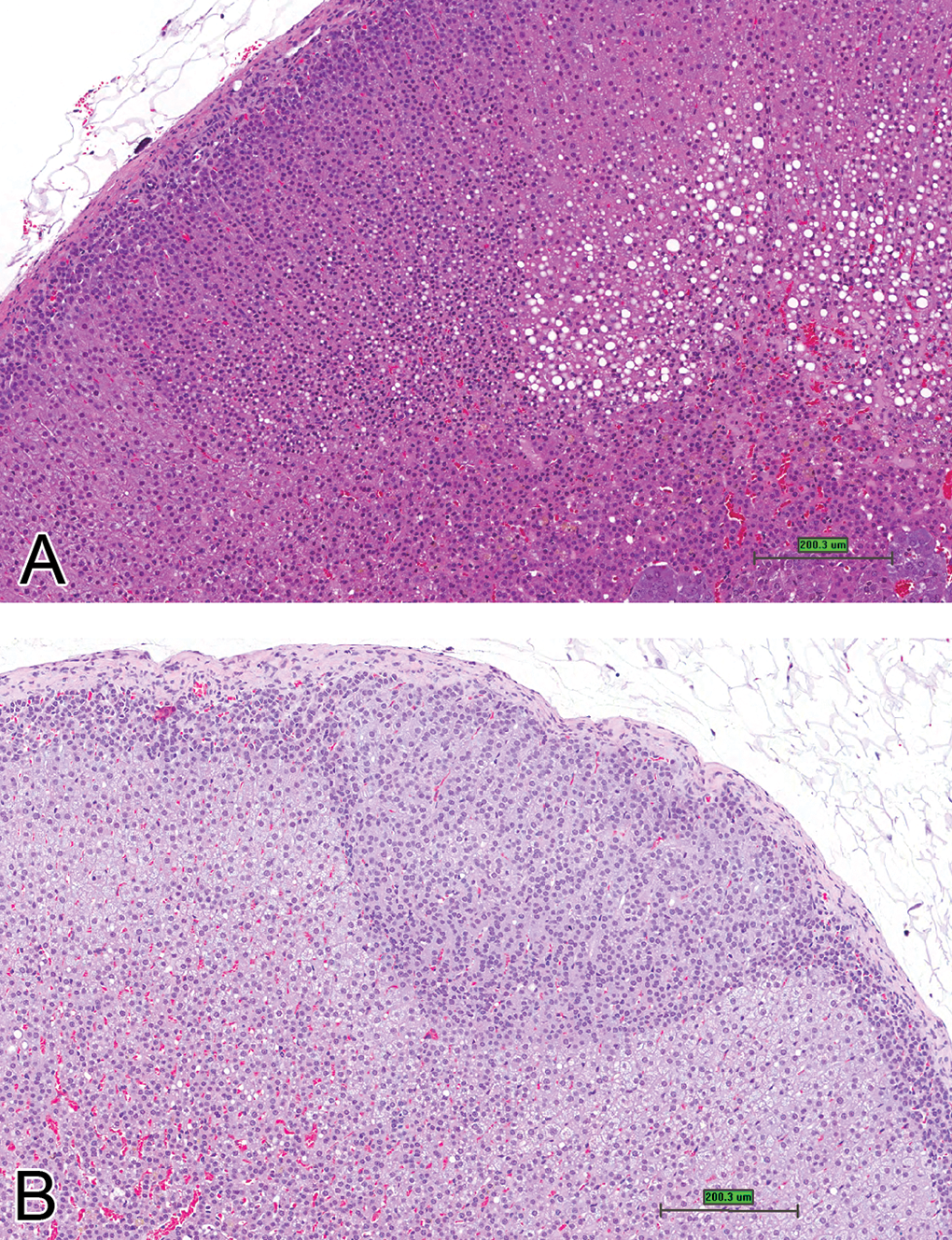
Adrenal cortex. A and B. Foci of cortical hyperplasia. Note more basophilic, smaller cells compared with adjacent cortical cells, arising within zona glomerulosa (H&E stain, scale bar is approximately 200 μm).
Adrenal Cortical Vacuolation
Lesions were focal, multifocal (Figure 8), or diffuse, and characterized by multiple small, or single large, clear cytoplasmic vacuoles in cortical cells. All three zones of the cortex were affected; however, the zona fasciculata was the most commonly affected region, with vacuoles in the zona reticularis generally being small. Cytoplasmic vacuolation is easily distinguished from cystic degeneration because of the tendency of vacuolation to be fairly uniformly distributed across the lesion, lack of association of cytoplasmic vacuolation with hypertrophied cells, and absence of degeneration and hemorrhage. As with cystic degeneration, there appeared to be a sex predilection for this lesion with males having a higher incidence (4.9%) than females (0.3%).
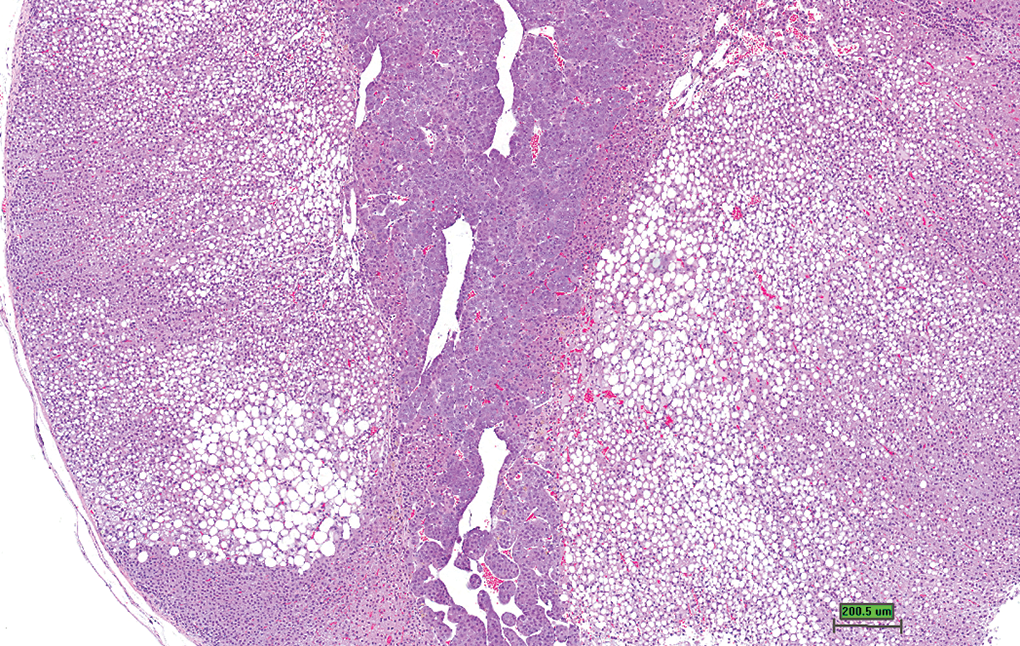
Adrenal cortex. Cortical vacuolation. Low magnification of adrenal gland with multifocal to diffuse cortical vacuolation (H&E stain, scale bar is approximately 200 μm).
Adrenal Cortical Adenoma
Adenomas were generally well circumscribed, occurred in the deeper zones of the zona fasciculata and zona reticularis, and usually resulted in compression of the adjacent parenchyma (Figure 9A). The cells were increased in number, enlarged or sometimes smaller, and were arranged in distorted cords with disruption of normal cortical architecture and minimal cellular atypia (Figure 9B). In larger adenomas, the cords were several cells thick. Mitoses were present in low numbers (Figure 9C). The incidence of adenomas was similar in males and females (1.6%).
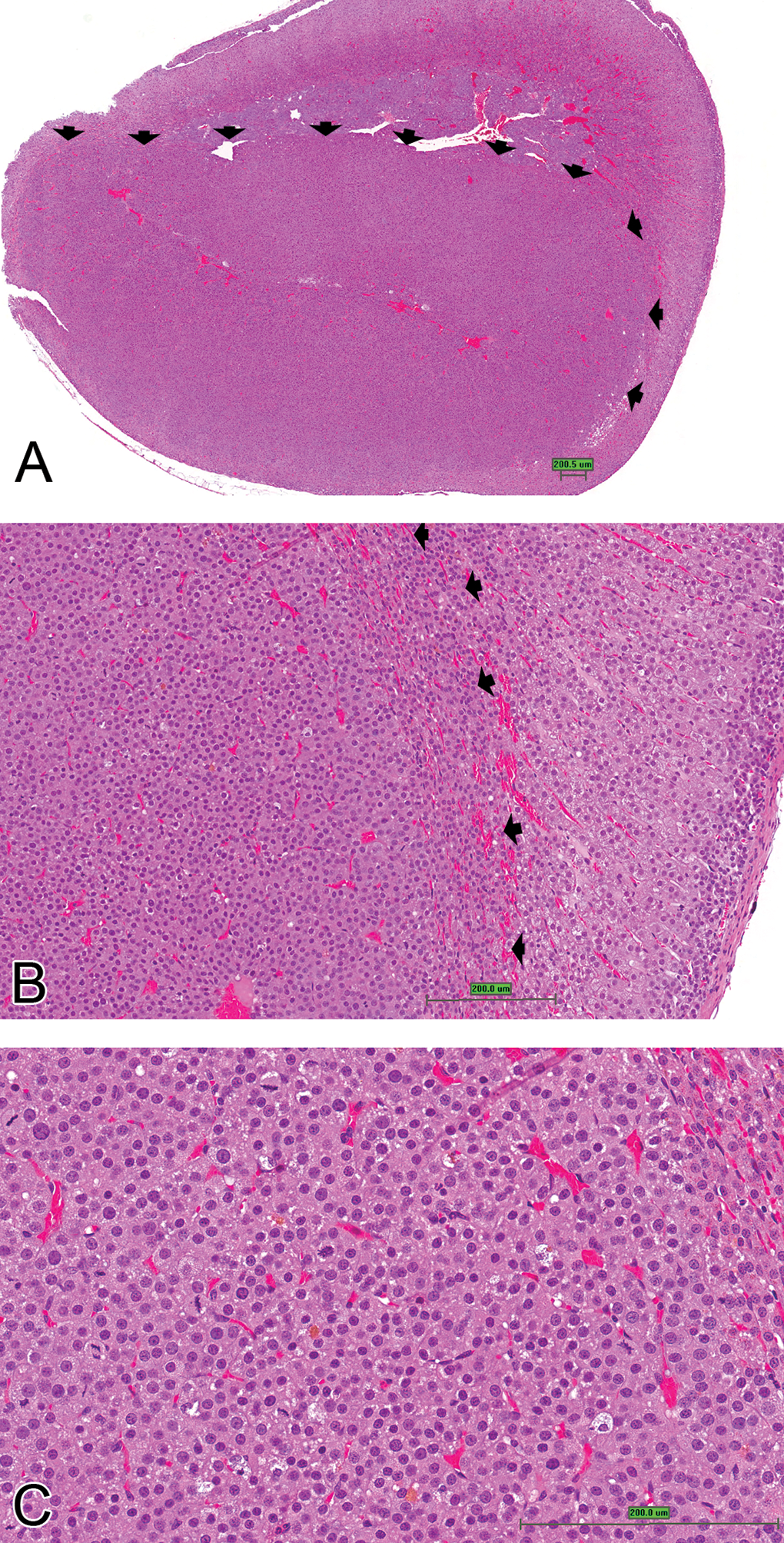
Adrenal cortical adenoma. (A) Well-circumscribed adenoma (arrowheads delineate adenoma) located deep within zona fasciculata. (B) Higher magnification of 9A. Note compression of adjacent cortical cells (arrow heads). (C) Higher magnification of 9A. Note thick cords as well as presence of mitotic figures (H&E stain, scale bar is approximately 200 μm).
A review of the adrenal glands from the NTP (2006b) study revealed similar morphological features in the adrenal cortex as noted in the 2 Covance studies. The NTP study was a multiple dose study with cystic degeneration present in 9/52 controls, increasing to 25/53 and 16/53 in the 2 highest exposure groups. We were unable to discern morphological differences between cystic degeneration found in treated animals versus the concurrent controls, or compared to the 2 Covance studies. In all exposure groups, hypertrophy was diagnosed in greater than 85% of rats. The location of hypertrophic lesions within the adrenal cortex was not specified in the NTP studies; in many animals, both hypertrophy and cystic degeneration were diagnosed when it was possible to definitively separate the lesions.
Discussion
Cortical hypertrophy, hyperplasia, vacuolation, cystic degeneration, and adrenal cortical adenoma were commonly diagnosed findings noted and discussed in this review of the adrenal cortex. The adrenal gland is the most susceptible of the endocrine tissues to compound-induced lesions, with the zona fasciculata and reticularis being the most frequently affected zones (Gopinath, Prentice, and Lewis 1987). Several compounds such as alpha-(1,4-dioxide-3-methylquinoxalin-2-yl)-N-methylnitrone; oxymetholone; 2,3′,4,4′,5-pentachlorobiphenyl and 2,3,7,8-tetrachlorodibenzo-p-dioxin cause widespread vacuolization of adrenal cortical cells in rats and dogs (Yarrington et al. 1985; Gopinath, Prentice, and Lewis 1987; NTP 1999, 2006b, 2010b). Increased adrenal cortical cytoplasmic vacuolation has been reported in 5 of the 8 studies designed specifically to evaluate the toxic equivalency factors for polyhalogenated aromatic hydrocarbons in female SD rats (NTP Technical Reports 520, 521, 525, 526, 529, 530, 531, and 559). Cystic degeneration was also increased in 2 of the NTP studies (NTP 2006a, 2006b). Seven of the eight studies listed the adrenal gland as a target due to the presence of nonneoplastic findings. Cytoplasmic vacuolation in these studies was diffuse in nature, distinguishing this lesion from cystic degeneration. Other important features of cytoplasmic vacuolation that can be used to distinguish this lesion from cystic degeneration include the presence of cytoplasmic vacuoles in relatively normal appearing cortical cells, a tendency for the vacuoles to vary in size but without parenchymal destruction, and no propensity for the vacuoles to be more common and larger at the base of the lesion.
Cystic degeneration has been reported with exposure to pentachlorodibenzofuran in female SD rats (NTP 2006a) and to a mixture of dioxin compounds (NTP 2006b). In the studies reviewed for this report, these lesions frequently occurred in foci of hypertrophy. The cause of cystic degeneration is uncertain. It has been suggested that estrogenic compounds could produce similar lesions (Gopinath, Prentice, and Lewis 1987); however, in studies where ethinyl estradiol (NTP 2010a) and genistein, a naturally occurring isoflavone that interacts with the estrogen receptor (NTP 2007), were administered to SD rats for 2 years, there was no association with increased incidences of cystic degeneration. The relationship of cystic degeneration to pituitary tumors found in rats at this age is also unclear. Although all animals in the studies examined for this report that had pituitary adenoma or hyperplasia also had cystic degeneration, not all animals with cystic degeneration of the adrenal cortex had observable pituitary tumors or hyperplasia. Long-term administration of ACTH to SD rats, with resulting decreased stimulation for adrenocorticotropic hormone (ACTH) in the pituitary gland caused a decreased incidence of cystic degeneration in the adrenal gland of these rats (Imazawa et al. 2000). Lesions similar to cystic degeneration have been seen in association with anti-vascular endothelial growth factor compounds, suggesting that the integrity of the vessels in the adrenal cortex may be part of the underlying factor in their development (Kolomecki, Stepien, and Naraebski 2000). Compounds such as 3-chloro-4-(dichloromethyl)-5-hydroxy-2(5H)-furanone (Komulainen et al. 1997) have been associated with adrenal cortical adenomas in 2-year studies in Wistar rats. An increase in nonproliferative lesions and/or increased incidence of cystic degeneration was however not reported.
Cystic degeneration appears to occasionally arise in areas of hypertrophy localized to the zona fasciculata. We suggest that the focal lesions of hypertrophied cells with few or no cytoplasmic vacuoles be diagnosed as hypertrophy, whereas instances where foci of hypertrophied cells show degenerative and cystic changes or prominent vacuolation, especially at the base of the lesion, be diagnosed as cystic degeneration. We do not favor using 2 diagnoses for 1 lesion in carcinogenicity studies. In our current review, most foci of hypertrophy appear in the zona glomerulosa with involvement of zona fasciculata. Hypertrophy in the zona glomerulosa was diagnosed because this lesion was separate both morphologically and by location. Focal hypertrophy arising in the zona glomerulosa often has a triangular shape, with cells that tend to stain less intensely than the surrounding cortex. In contrast, hypertrophic foci located in the zona fasciculata are composed of large plump cortical cells containing abundant eosinophilic cytoplasm. Even small lesions of hypertrophy may cause slight compression. As the lesions become larger they may contain a few cytoplasmic vacuoles, and we hypothesize that as the lesions become larger they progress to cystic degeneration.
In most cases, cystic degeneration does not create a diagnostic challenge. However, those that present as a circumscribed or well-demarcated lesion with compression of the adjacent cortical parenchyma may be confused with adenomas. However, one must keep in mind that while cystic degeneration may include the presence of large cells and mild compression, there are typically fewer cells present due to cellular dropout, often with large cystic spaces, in contrast to adenomas, which are truly proliferative lesions composed of sheets of large numbers of neoplastic cells. The compression observed in cases of cystic degeneration is due to the presence of large vacuolated or hypertrophied cells and cystic spaces formed by the degeneration and dropout of cortical cells. As a result, cystic degenerative lesions have fewer cells in contrast to adenomas and lack cellular atypia.
In conclusion, while cystic degeneration is a common, spontaneously arising nonneoplastic lesion in the adrenal cortex of Sprague-Dawley rats, it can be found as a test article–related effect, and may often pose a diagnostic challenge due to its occasional association with cellular hypertrophy and compression. Therefore, further study of the pathogenesis and progression of this lesion and its relevance in toxicity and carcinogenicity bioassays is warranted.
Footnotes
Abbreviations
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors. Images used in this article were not from NTP studies.
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported (in part) by the Intramural Research Program of the National Institutes of Health (NIH), National Institute of Environmental Health Sciences (NIEHS). This article may be the work product of an employee or group of employees of the National Toxicology Program (NTP), NIEHS, NIH; however, the statements, opinions, or conclusions contained therein do not necessarily represent the statements, opinions, or conclusions of NTP, NIEHS, NIH, or the U.S. Government.
