Abstract
A typical 26-week Tg.rasH2 mouse carcinogenicity study usually has 4 dose groups, composed of 25 mice/sex, which include 1 control and 3 test article–treated groups. In every study, there is a protocol required full tissue list of 49 tissues which is examined microscopically in all animals of these 4 dose groups. Based on retrospective analysis of the historical control data collected from studies conducted in Tg.rasH2 mice from 2004 to 2012, we propose that a full tissue list be examined as usual in the control and high-dose groups; however, in the low- and mid-dose groups, only select tissues should be examined. The select tissue list is generated after analyzing common tumors, uncommon tumors seen grossly, uncommon tumors not seen grossly, organ weight variations with accountable microscopic lesions, and target organs identified in the high-dose groups. The proposed changes to the International Conference on Harmonization S1 guidance may lead to an increased need for 26-week Tg.rasH2 mice studies. The time savings resulting from processing and evaluating a select tissue list rather than a full tissue list from low- and mid-dose groups of Tg.rasH2 mouse studies will further accelerate early completion of these studies without compromising the quality and integrity.
Introduction
In the last decade, the Tg.rasH2 mouse has become a popular model for performing 26-week carcinogenicity studies, replacing about a quarter of all 2-year carcinogenicity studies performed in conventional mouse models (Jacobs and Hatfield 2012; Nambiar, Turnquist, and Morton 2012; Paranjpe, Elbekai, et al. 2013). We recently published the largest historical control database for both neoplastic and nonneoplastic lesions in Tg.rasH2 mice (Paranjpe, Elbekai, et al. 2013; Paranjpe, Shah, et al. 2013). Based on extensive experience with this assay in our laboratory, a number of improvements have been made including reducing the number of animals in the positive control group from 25 mice/sex to 10 mice/sex, leading to a substantial reduction in animal use, and thereby contributing to the 3Rs (replace, reduce, and refine) of animal research (Shah et al. 2012).
The proposed changes to the International Conference on Harmonization (ICH) S1 guidance based on solid scientific reasoning indicate that the number of 2-year rat studies will eventually be significantly reduced, as the carcinogenic potential of drugs can be predicted based on rat studies of 6-month duration (Federal Register 2013; Reddy et al. 2010; Sistare et al. 2011). The proposed changes to the ICH S1 guidance may lead to an increased need for 26-week Tg.rasH2 mice studies. Since the Tg.rasH2 mouse assay plays a critical role in carcinogenicity testing, it is important to continue to refine the Tg.rasH2 assays to best meet regulatory and industry testing needs.
A retrospective analysis of Tg.rasH2 mouse studies conducted at our facility was undertaken to determine if a reduction could be made in the number of tissues examined without compromising the quality and integrity of these studies. In each of these Tg.rasH2 mouse studies, 49 tissues were examined microscopically for every animal in the control and test article–treated groups. This tissue list is the same as the one used by the International Life Science Institute Health and Environmental Science Institute’s (ILSI HESI) research program to characterize the transgenic mouse models for carcinogenicity assessment (Robinson and MacDonald 2001). With minor variations, this full tissue list is similar to the ones recommended by the National Toxicology Program and the Society of Toxicologic Pathology and is commonly used in the U.S. and EU carcinogenicity studies (McConnell 1983; Bregman et al. 2003; Hayes et al. 2011). The value of examining this full tissue list in lower dose groups has been discussed before for 2-year rodent studies (McConnell 1983) and for nonrodent toxicity studies (Weddle, Mahrt, and Schmidt 2012). Based on the retrospective analysis of our extensive database of Tg.rasH2 mouse studies conducted at our facility, we propose that a full tissue list be examined as usual in the control and high-dose groups but only select tissues be examined in low- and mid-dose groups. This approach will save time without compromising the integrity of the studies. This article details the list of select tissues recommended for examination in the low- and mid-dose groups and the approach we took to create the list.
Materials and Methods
Animals
CByB6F1-Tg(HRAS)2Jic (+/− hemizygous c-Ha-ras) mice, obtained from Taconic Farms (Germantown, NY), were used in all studies. The knock-in Tg element (human prototype c-Ha-ras gene with its own promoter/enhancer) is injected into C57BL/6 × BALB/c (Bagg Albino) F2 zygotes, which are crossed back to C57BL/6J forming C57BL/6JJic-Tg(HRAS)2Jic. The CByB6F1-Tg(HRAS)2Jic (+/− hemizygous c-Ha-ras) is the offspring from a cross of the C57BL/6JJic-Tg(HRAS)2Jic hemizygous male mice with the BALB/cByJJic female mice. Each mouse was genotyped by Taconic to verify the presence of the transgene before being placed on study. Animals were assigned to groups using a computer-generated randomization program. On the first day of treatment, animals were 6 to 10 weeks of age and weighed at least 20 or 15 g (males and females, respectively).
Housing and Environmental Conditions
Housing and environmental conditions were similar in all studies. Animals were single housed in polycarbonate cages with hardwood bedding chips in environmentally controlled rooms. Animals were verified to be free of illness prior to being placed on a study. All animals had ad libitum access to feed (Harlan TEKLAD Global Diet, Madison, WI) and water.
Regulatory Requirements
The numbers of animals, procedures, and experimental design for each study were reviewed and approved by the BioReliance Institutional Animal Care and Use Committee. All procedures followed the specifications recommended in The Guide for the Care and Use of Laboratory Animals and were conducted in an Association for Assessment and Accreditation of Laboratory Animal Care–accredited facility. All procedures involving but not limited to quarantine and acclimation, randomization, application of a unique identification system, housing, provision of food and water, administration of test article, recording of clinical signs, necropsy, and tissue processing were followed in strict accordance with the Good Laboratory Practice Regulations, Standard Operating Procedures, and the study protocol for each study.
Retrospective Analysis
Retrospective analysis was performed on twenty-six, 26-week Tg.rasH2 mouse studies completed at our facility in the period between 2004 and 2012. Generally, each study included 1 control group and 3 test article–treated dose groups. Together, we analyzed the data for 5,370 mice that included 710 mice/sex assigned to control, 710 mice/sex assigned to low dose, 605 mice/sex assigned to mid dose, and 660 mice/sex assigned to high-dose groups. Per current protocols, all tissues in the full tissue list presented in Table 1 were collected from all control and test article–treated animals in each study. Tissues were fixed in 10% neutral buffered formalin, embedded in paraffin, sectioned, stained with hematoxylin and eosin, and evaluated microscopically. Pathology evaluations of each study were peer reviewed.
Full tissue list.

The select tissue list for the low- and mid-dose groups was generated after analyzing the historical control data for (a) common tumors, (b) uncommon tumors seen grossly, (c) uncommon tumors not seen grossly, (d) organ weight variations with accountable microscopic lesions, and (e) target organs identified in the high-dose groups.
Results
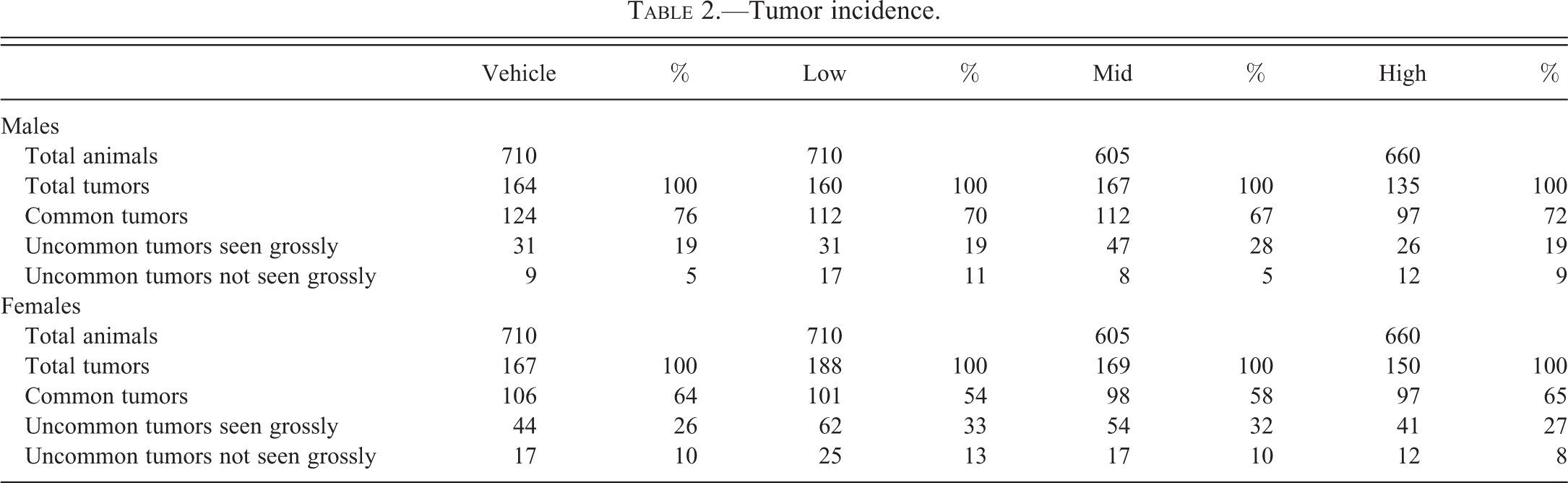
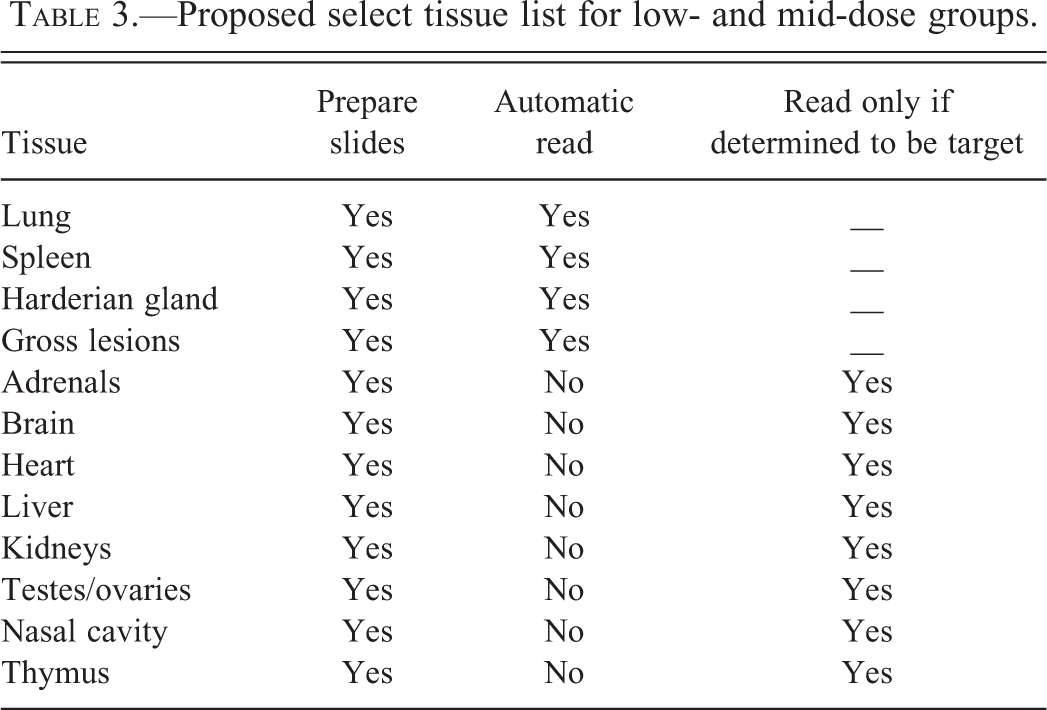
The incidence of tumors is provided in Table 2, which includes information pertaining to total tumors and percentages of common tumors, uncommon tumors seen grossly, and uncommon tumors not seen grossly. Table 3 presents a proposed select tissue list of 12 tissues that will be processed to slide for the low- and mid-dose groups. Of these 12 tissues, 4 tissues (lungs, spleen, Harderian glands, and any gross lesions) will be automatically examined for every animal in the low- and mid-dose groups. The remaining 8 tissues (adrenals, brain, heart, kidneys, liver, nasal cavity, testes/ovaries, and thymus) will be evaluated microscopically only if any of these tissues were determined to be target organs based on evaluation of the control and high-dose groups.
Tumor incidence.
Proposed select tissue list for low- and mid-dose groups.
Discussion
The breadth of the historical control data was a key in determining the appropriate tissues for the select list in the low- and mid-dose groups. It is important that each facility conducting these studies use their data to determine this list, ensuring that any facility and pathologist-specific variations are taken into account (Guidance for Industry: U.S. FDA 2001; Haseman, Huff, and Boorman 1984; Keenan et al. 2009; Takaoka et al. 2003). In creating a select tissue list for low- and mid-dose groups in 26-week Tg.rasH2 mouse studies, we took into consideration (a) common tumors, (b) uncommon tumors seen grossly, (c) uncommon tumors not seen grossly, (d) organ weight variations with accountable microscopic lesions, and (e) target organs identified in the high-dose groups. These important criteria are discussed further:
Common Tumors
Included in this category are lung tumors (adenomas and carcinomas), splenic hemangiosarcomas, and Harderian gland tumors (adenomas and carcinomas) in both sexes (Paranjpe, Elbekai, et al. 2013). These tumors represent up to 76% of all tumors in males and up to 65% of all tumors in females in the data collected in our studies. While most of the lung adenomas and all of the carcinomas were noted grossly as nodules or masses, there were certainly several smaller adenomas that were embedded deep in the parenchyma that were not seen grossly. For every splenic hemangiosarcoma, there was a corresponding focus, nodule, or a mass noted at necropsy. Harderian gland carcinomas were noted grossly easily; however, there were several instances where adenomas did not have corresponding gross lesions detected at necropsy. Regardless of whether or not there is a gross lesion we decided that the lungs, spleens, and Harderian glands need to be examined in all animals based on the common occurrence of these tumors.
Uncommon Tumors Seen Grossly
Included in this category are nonsplenic hemangiosarcomas, nonvascular malignant tumors, and other benign tumors. All nonsplenic hemangiosarcomas were seen grossly, except for those in the nasal cavities. Examples of those noted grossly include the ones involving male and female reproductive systems, skin/subcutis, and muscles. The nasal cavity tumors are discussed under uncommon tumors not seen grossly in Uncommon Tumors Not Seen Grossly section below. Examples of malignant tumors that were seen grossly include lymphomas in multiple tissues (spleen, lymph nodes, thymus), squamous cell carcinoma of the stomach, and sarcomas affecting multiple tissues (skin/subcutis, bones, muscles). The examples of benign tumors seen grossly include papillomas at various sites, testicular interstitial cell tumors, uterine deciduomas, and ovarian teratomas. All of these tumors we noted (nonsplenic hemangiosarcomas, nonvascular malignant tumors, and other benign tumors) had been scattered across groups and none reached statistical significance in any one of our studies. Uncommon tumors seen grossly represent up to 28% of all tumors in males and up to 33% of all tumors in females.
Uncommon Tumors Not Seen Grossly
Included in this category are benign and malignant tumors that would have been missed if a particular tissue was not examined microscopically. The noteworthy examples of benign tumors include thymomas and mesotheliomas. Mesotheliomas were usually noted in the anterior mediastinal area around the lungs and thymus. The lung tissue is already included in the select tissue list. We added thymus to the list of select tissues to be processed, so that neither the mesotheliomas nor the thymomas would be missed. The noteworthy examples of malignant tumors that were not seen grossly include tumors of the nasal cavity such as adenocarcinomas and hemangiosarcomas. Therefore, we decided to include the nasal cavity in the select tissue list.
Uncommon tumors not seen grossly represent up to 11% of all tumors in males and up to 13% of all tumors in females. Similar to uncommon tumors seen grossly, these tumors are once again scattered among groups and none of these tumors reached statistical significance in any of the 26 studies that we have completed. However, we do not want to underestimate the significance of these scattered tumors and rule out the possibility of their presence related to the test article. Therefore, if any of these tumors are observed in the high-dose groups, we can compare their incidence with the control dose groups and the historical control database, which will allow us to evaluate the impact of these tumors on a particular study. After evaluating the impact of such tumors, we can always decide to read a particular tissue for a particular tumor in low- and mid-dose groups. However, such occurrences will be rare.
Organ Weight Variations with Accountable Microscopic Lesions
We routinely weigh adrenals, brain, heart, kidneys, liver, spleen, and testes/ovaries for all animals in every study. If any of these tissues demonstrated variations in the organ weights with accountable microscopic changes, those tissues would be automatically included in the select tissue list for low- and mid-dose groups. Based on our experience with this model, we decided that all tissues weighed need to be included in the select tissue list.
Target Organs Identified in the High-dose Groups
Any target tissues identified in the preceding 28-day dose range finding study will be automatically included in the select tissue list for low- and mid-dose groups of the 26-week studies. Similarly, target organs identified in the high-dose group of a 26-week study will also be automatically included in the select tissue list for low- and mid-dose groups. It needs to be pointed out here that we have always identified the target tissues in the high-dose groups of each sex for every 26-week study and never in the low- or mid-dose groups alone.
The slides for the select tissue list for low- and mid-dose groups will be processed at the same time as the full tissue list for the control and high-dose groups. Based on the discussion above, we have created a list of 12 tissues that includes lungs, spleen, Harderian glands, gross lesions, adrenals, brain, heart, kidneys, liver, testes/ovaries, nasal cavity, and thymus. Of these 12 tissues lungs, spleen, Harderian glands, and gross lesions will be automatically read in low- and mid-dose groups for each sex. If any of the remaining tissues become target organs, those tissues can be read without any lapse of time since the slides will have been already prepared for those tissues. We feel that by this approach, we will be well equipped to deal with all tumors and target organs. However, this does not rule out the possibility of the tissues not included in the select tissue list becoming target organs. In such a situation, those tissues can always be processed after reading the control and high doses. However, such occurrences will be rare.
The value of examining the full tissue list in lower dose groups has been discussed before for 2-year rodent and nonrodent studies, and alternative tissue lists have been proposed for 2-year rodent studies (McConnell 1983; Weddle, Mahrt, and Schmidt 2012). However, the very high incidence of spontaneous tumors in 2-year rodent models can certainly pose problems in developing and adopting a select tissue list with lesser number of tissues than that for control and high-dose groups. The percentages of total spontaneous tumors in 2-year B6C3F1 and CD-1 mice as well as F344 rats have been reported to be over 65% (Baldrick and Reeve 2007; Haseman, Hailey, and Morris 1998; NTP 2011). In comparison, the percentage of spontaneous tumors in Tg.rasH2 mice in our studies is 23.05% in males and 23.47% in females (Paranjpe, Elbekai, et al. 2013). Therefore, we feel the select tissue list can be developed and adopted more easily in Tg.rasH2 mice compared to the 2-year conventional rodents. Adoption of a select tissue list for low- and mid-dose groups of Tg.rasH2 studies will further accelerate early completion of Tg.rasH2 mouse studies without compromising the quality and integrity of these studies.
Footnotes
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
The author(s) received no financial support for the research, authorship, and/or publication of this article.
