Abstract
This article presents the historical control data of spontaneous tumors in Tg.rasH2 published in 2013 (2004–2012) and compares and contrasts it to more recent data collected from 2013 to 2018, reporting differences in the average percentage incidences or incidence ranges as well as the incidence of new tumors. In 2013, we published a comprehensive review of spontaneous tumors in Tg.rasH2 mice used in 26-week carcinogenicity studies, which included data from control dose groups from 26 studies and a total of 710 mice per sex. The total database, now including the more recent data, has nearly doubled the number of animals, completing to date a total of 52 studies in males and 51 studies in females for a total of 1,615 male mice and 1,560 female mice, respectively. In this article, we compare the data collected from 2004 to 2012 against the data collected from 2013 to 2018 and the overall tumor incidence change.
All studies presented in this article were conducted at BioReliance in Rockville, Maryland, now known as MilliporeSigma. The original publication reporting data from 2004 to 2012 demonstrated that pulmonary tumors (adenomas and carcinomas), splenic and other hemangiosarcomas, and Harderian gland tumors were the most common tumors in Tg.rasH2 mice of both sexes (Paranjpe et al. 2013). We also demonstrated that the combined incidence of all spontaneous tumors in Tg.rasH2 mice is generally low and below 25% in each sex. The incidence of spontaneous tumors in Tg.rasH2 mice at 26 weeks is much lower than the incidence of spontaneous tumors in conventional 2-year B6C3F1 and CD1 mice (Jacobs and Hatfield 2013; Nambiar, Turnquist, and Morton 2012; Paranjpe et al. 2013). Because of lower incidence of tumors and shorter duration of assays, the use of this model in carcinogenicity studies has almost doubled from 2013 to 2018. While we present the entire data on tumors observed during two different time intervals (2004–2012 and 2013–2018), in this article, we mainly discuss statistically significant differences in the most common tumors: primary lung tumors, hemangiosarcomas in multiple organs including spleen, Harderian gland tumors, and other tumors.
Material and Method
Experimental Design
The 26-week studies were conducted in Tg.rasH2 mice. Animals were assigned to groups using a computer-generated randomization program based upon each individual study design with a minimum of 25 animals per sex in each control group. Occasionally, studies contained a vehicle control as well as a negative control group (such as water). On the first day of treatment, the mice were 6 to 10 weeks of age and weighed at least 20 or 15 g (males and females, respectively). Individual body weights for the mice in each dose group and sex were within ±20% of the mean body weight.
Animals
CByB6F1-Tg(HRAS)2Jic (+/− hemizygous c-Ha-ras) mice, obtained from Taconic Biosciences (Germantown, New York), were used in all studies. The knock-in Tg element (human prototype c Ha-ras gene with its own promoter/enhancer) was injected into C57BL/6 _ BALB/c F2 zygotes, which were crossed back to C57BL/6J forming C57BL/6JJic-Tg(HRAS)2Jic. The CByB6F1 Tg(HRAS)2Jic (+/− hemizygous c-Ha-ras) is the offspring from a cross of the C57BL/6JJic-Tg(HRAS)2Jic hemizygous male mice with the Bagg Albino (BALB)/cByJJic female mice. Each mouse was genotyped by Taconic to verify the presence of the transgene before being placed on study.
Housing and Environmental Conditions
Housing and environmental conditions were similar in all studies. Animals were single housed in polycarbonate cages with hardwood bedding chips in environmentally controlled rooms. Animals were verified to be free of illness prior to being placed on a study. All animals had
Regulatory Requirements
The numbers of animals, procedures, and experimental design for each study were reviewed and approved by the BioReliance Institutional Animal Care and Use Committee. All procedures followed the specifications recommended in The Guide for the Care and Use of Laboratory Animals and were conducted in an Association for Assessment and Accreditation of Laboratory Animal Care–accredited facility. All procedures involving but not limited to quarantine and acclimation, randomization, application of unique identification system, housing, provision of food and water, administration of test article, recording of clinical signs, necropsy, and tissue processing were followed in strict accordance with the good laboratory practice regulations, standard operating procedures, and protocol for each study.
Retrospective Analysis
The database was comprised of 51 studies that contained both male and female Tg.rasH2 mice, as well as one study that contained only male Tg.rasH2 mice, for a total of 1,615 control males and 1,560 control females. All studies followed the same general study design. In the 51 studies, with both sexes, 43 were dosed by oral gavage, 3 were given drug in feed, 3 were dosed subcutaneously, and the remaining 2 were dosed intravenously. Males on the male-only study were dosed intravenously. The earliest studies were completed in 2004 and the most recent study was completed in 2018.
There were generally 25 animals in each control group. From 2004 to 2012, there were 26 studies that totaled 710 control mice per sex. From 2013 to 2018, there were 26 studies in males and 25 studies in females that totaled 905 control male mice and 850 control female mice. Variations in the number of animals were due to differences in the study design, addition of an extra control group, and/or the number of animals assigned to each dose group; otherwise, the study designs remained very similar for all studies.
All animals were humanely euthanized by CO2 overdose and were subjected to a complete necropsy. Protocol required tissues were saved in 10% neutral buffered formalin, processed routinely, stained with hematoxylin and eosin, and evaluated microscopically by a board-certified veterinary pathologist. A pathology peer review by another board-certified veterinary pathologist was also performed on each study.
The tumor data presented in this article include three tables. Tables 1 and 2 contain data from 2004–2012 (published in 2013) to 2013–2018 (updated data for 2018) for the males and females, respectively. Each table demonstrates the number of studies performed per sex, the number of animals examined per sex, number of tumors observed by tissue, incidence range, percentage range for the study, and average percentage of tumors. For each organ/tumor type, the proportion observed from 2004 to 2012 was compared to the proportion observed from 2013 to 2018 using a two-sided Fisher’s exact test. All calculations were performed using the SAS System reference on stats, version 9.2 (SAS®, 2008). The
Historical Control Data from 2004–2012 versus 2013–2018 for Male Tg.rasH2 Mice.
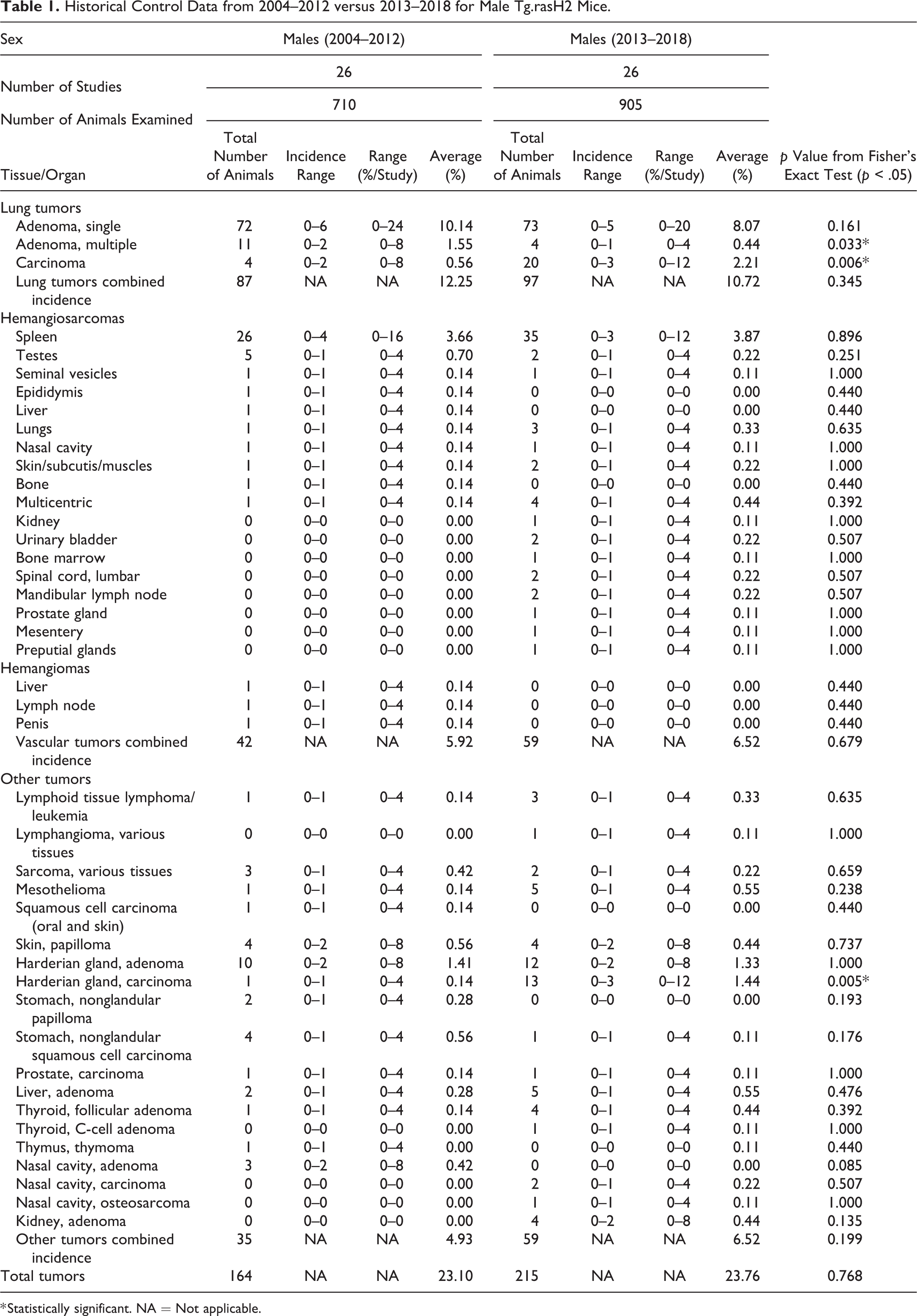
* Statistically significant. NA = Not applicable.
Historical Control Data from 2004–2012 versus 2013–2018 for Female Tg.rasH2 Mice.
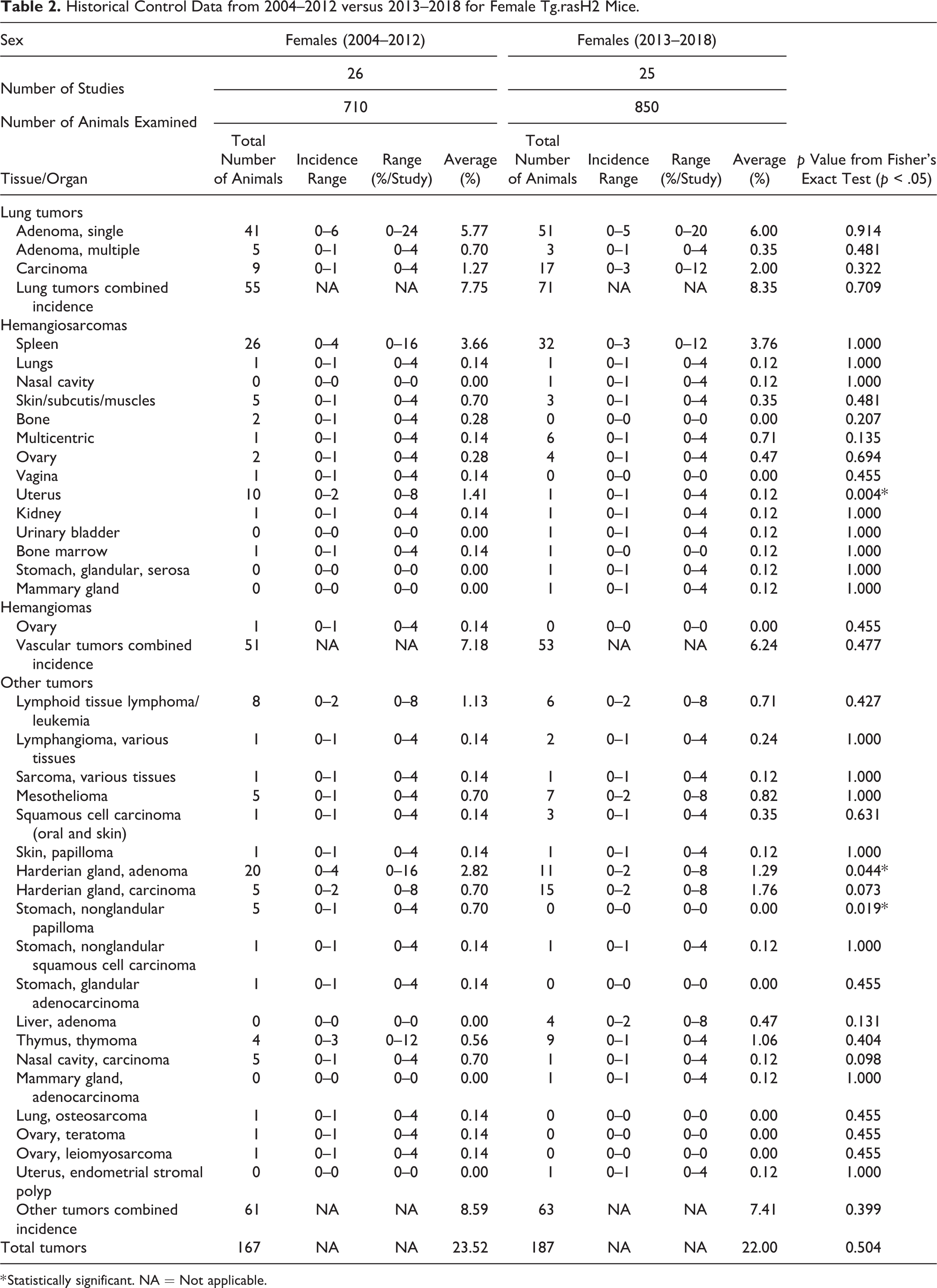
* Statistically significant. NA = Not applicable.
Historical Control Data from 2004–2018 for Male and Female Tg.rasH2 Mice.
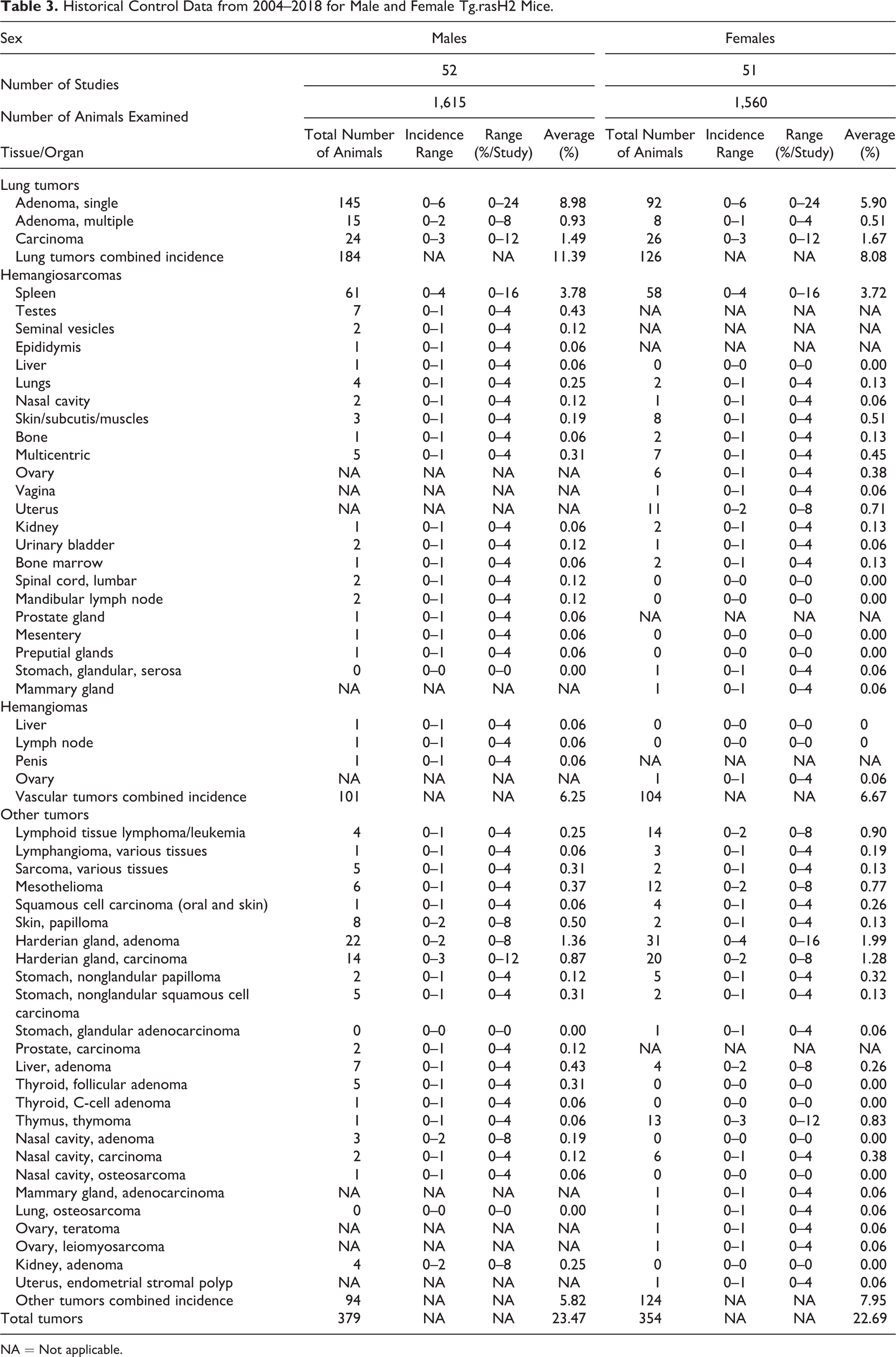
NA = Not applicable.
Results
Males
Tumors in lungs
There were no significant differences in single adenomas (only one tumor present in the section) when the data from 2004–2012 to 2013–2018 were compared (Table 1). The incidence range per study was 0 to 6 in 2013 versus 0 to 5 in 2018.
There was a statistically significant decrease in multiple adenomas (2 or more tumors in the same section) when data collected from 2013 to 2018 were compared to the data collected from 2004 to 2012. The incidence range per study was 0 to 2 in 2013 versus 0 to 1 in 2018.
There was a statistically significant increase in the carcinomas from 2013–2018 compared to 2004–2012. The incidence range per study was 0 to 2 in 2013 versus 0 to 3 in 2018.
For the purpose of tumor data statistics, the combined incidence of all pulmonary tumors (single adenomas, multiple adenomas, and carcinomas) was analyzed together in each study. When we compared the combined incidence of all pulmonary tumors from 2004–2012 to 2013–2018 for each sex, there were no statistically significant differences.
Hemangiomas and hemangiosarcomas
In males, there were no statistically significant differences in the incidence of splenic hemangiosarcomas, hemangiomas, and hemangiosarcomas observed in various tissues as well as the combined incidence of all hemangiomas and hemangiosarcomas from 2004–2012 to 2013–2018. The historical control incidence ranges for all these tumors were very similar.
Other tumors
Under the category of other tumors (nonvascular and nonpulmonary), there was a statistically significant increase in the Harderian gland carcinomas from 2013–2018 compared to 2004–2012.
Apart from Harderian gland tumors, there were a variety of other tumors (nonvascular and nonpulmonary) observed in various organs. There were no statistically significant differences in the combined incidence of other tumors.
New tumors 2013–2018, not reported in 2004–2012
Compared to the data published in 2013 (2004–2012), there were several new tumors that were observed in males from 2013 to 2018: hemangiosarcomas in the kidney (1), urinary bladder (2), bone marrow (1), lumbar spinal cord (2), mandibular lymph node (2), prostate gland (1), mesentery (1), and preputial glands (1); lymphangioma, various tissues (1), thyroid c-cell adenoma (1), nasal cavity carcinoma (2), nasal cavity osteosarcoma (1), and kidney adenoma (4). However, none of these tumors were statistically significant.
Females
Tumors in lungs
There were no statistically significant differences in the incidence of single adenomas, multiple adenomas, or carcinomas of the lung or that of combined incidence of all pulmonary tumors (Table 2). The incidence ranges remained very similar in both time periods.
Hemangiomas and hemangiosarcomas
In females, there were no statistically significant differences in the incidence of splenic hemangiosarcomas, hemangiomas or in the combined incidence of all hemangiosarcomas. The historical control incidence ranges for all these tumors were very similar. However, there was a statistically significant decrease in the incidence of uterine hemangiosarcomas from 2013–2018 compared to 2004–2012.
Other tumors
Under the category of other tumors (nonvascular and nonpulmonary), there was a statistically significant decrease in the Harderian gland adenomas and papillomas in the nonglandular stomach from 2013–2018 compared to 2004–2012. There were no statistically significant differences in the combined incidence of other tumors.
New tumors 2013–2018, not reported in 2004–2012
Compared to the data published in 2013 (2004–2012), there were several new tumors that were observed in females from 2013 to 2018: nasal cavity hemangiosarcoma (1), urinary bladder hemangiosarcoma (1), glandular stomach serosa hemangiosarcoma (1), mammary gland hemangiosarcoma (1), liver adenoma (4), mammary gland adenocarcinoma, (1) and uterus endometrial stromal polyp (1). However, none of these tumors was statistically significant.
Table 3 contains the combined data from all studies conducted from 2004 to 2018. This table shows that the combined incidence of all tumors in both sexes remains below 25%.
Discussion
Each mouse was genotyped by Taconic to verify the presence of the transgene before being placed on study (Urano et al. 2008; Urano et al. 2014). Also, when we performed these studies along with urethane as a positive control group, the mice in both sexes of the positive control groups developed close to 100% of the lung and splenic tumors in all studies. This clearly indicates robust genetic stability of the Tg.rasH2 mice (Paranjpe et al. 2013).
When the incidence of tumors from 2004–2012 was compared to 2013–2018, the following differences were noted: In the lungs of male mice, there was a statistically significant decrease in multiple adenomas and a statistically significant increase in carcinomas. However, when the combined incidence of all pulmonary tumors was compared, there were no statistically significant differences. In males, there was also a statistically significant increase in the Harderian gland carcinomas. In female mice, there was a statistically significant decrease in the incidence of uterine hemangiosarcomas. However, when the combined incidence of all hemangiosarcomas was compared, there were no statistically significant differences. In females, there was also a statistically significant decrease in Harderian gland adenomas and nonglandular stomach papillomas.
Because humans have poorly developed or rudimentary Harderian glands, any changes in the Harderian glands of rodent carcinogenicity studies have little or no significance (Albert et al. 1986).
The exact causes for the statistically significant increases or decreases in a few of the tumors noted in males and females is unknown, but it may represent a biological drift as well as a diagnostic drift over a period of 14 years. However, in general, the Tg.rasH2 model appears to be very stable based on tumor incidence range which is important for nonclinical carcinogenicity studies.
In order to maintain consistency, all studies followed similar diagnostic criteria and each study was peer reviewed. However, the possibility of a diagnostic drift, a very important factor, cannot be completely ruled out (Ward et al. 1995). The historical control ranges noted from 2004–2012 to 2013–2018 were very similar and there were no statistically significant differences in tumors for each category (all lung tumors, all hemangiosarcomas, all other tumors, and the total tumors). Thus, these differences described above in individual tumors are likely due to the very slight biologic variability in the animals. Eventually, the combined tumor incidence ranges at the end of all studies from 2004 to 2018 remained below 25%.
This is the first time that the historical control data for spontaneous tumors on any rodent used in carcinogenicity studies are published by the same laboratory in two data sets at an interval of five years. The Tg.rasH2 model, which can be used for both genotoxic and nongenotoxic compounds, has gained popularity and its use has increased over the years, and currently more than 75% of mouse carcinogenicity studies are conducted in Tg.rasH2 mice (Jacobs and Brown 2015). Our database also shows similar findings.
The Tg.rasH2 model has been proven to predict neoplastic findings relevant to human cancer risk assessment, produces fewer nonbiologically significant neoplastic outcomes, and is thus preferable to a 2-year rodent study (Morton et al. 2014). Historical control data on the Tg.rasH2 model previously have been published by others (Nambiar, Turnquist, and Morton 2012; Morse et al. 2015), however, we have presented the largest historical control data for spontaneous tumors (lung tumors, hemangiosarcomas, other tumors, and total tumors) noted in Tg.rasH2 males and females from 2004 to 2018. This data demonstrates that the combined incidence of tumors in both males and females remained below 25% and that the Tg.rasH2 model appears to be very stable based on tumor incidence range, with the differences in individual tumors likely due to very slight biologic variability in the animals. This further supports the use of the Tg.rasH2 model, particularly for nonclinical carcinogenicity studies.
Footnotes
Author Contributions
All authors (MP, JB, PM, MM, RE, CB, DP) contributed to conception or design; data acquisition, analysis, or interpretation; drafting the manuscript; and critically revising the manuscript. All authors gave final approval and agreed to be accountable for all aspects of work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential, real, or perceived conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
