Abstract
Short-term (26 weeks) Tg.rasH2 mouse carcinogenicity studies have been conducted as an alternative model to the conventional 2-year mouse carcinogenicity studies, using urethane as a positive control material. In these studies, urethane was used at a dose of 1,000 mg/kg/dose, administered intraperitoneally on days 1, 3, and 5. Urethane consistently produces lung adenomas and carcinomas and hemangiosarcomas of the spleen, proving validity of the assay. We conducted 3 pilot studies at 3 different sites of Charles River Laboratories using a lower dose of urethane (500 mg/kg/dose), administered on days 1, 3, and 5, followed by a 12-week observation period. Our results demonstrate that a lower dose can be used successfully with fewer number of animals per sex to prove the validity of the assay. However, based on our cumulative experience with this model, we propose to eliminate positive control dose groups in future Tg.rasH2 carcinogenicity studies.
Introduction
The initial 26-week Tg.rasH2 carcinogenicity studies using urethane as a positive control material were conducted from 2003 to 2004. 1 -9 Most common spontaneous tumors in Tg.rasH2 are lung adenoma (single or multiple) and carcinoma and splenic hemangiosarcomas. 9 These same tumors also develop in Tg.rasH2 mice when administered urethane as a positive control. 1 Another positive control material that has been used by many laboratories is N-methyl-N-nitroso-urea (NMU). 10
Published data show that urethane was used at a dose of 1,000 mg/kg/dose in Tg.rasH2 carcinogenicity studies. 2,-7,9 When commercial good laboratory practice studies started in 2003 to 2004, the initial 3 studies were conducted in Tg.rasH2 with urethane as a positive control material, 1 control group and 3 test article-treated groups. In addition, control and high dose groups of CByB6F1 wild-type mice were included for comparison. There were 25 mice assigned to all dose groups of the study, including the positive control groups. All tissues were examined in the positive control group, vehicle and test article-treated groups, as well as control and high dose groups of CByB6F1 wild-type mice, at the end of the 26-week study period. However, there was considerable early mortality in the positive control groups because of an extensive increase in the tumors induced by urethane. After initial 3 studies, the animals in positive control groups were reduced to 15/sex for the duration of the study (26 weeks) because of almost 100% incidence of spontaneous tumors in Tg.rasH2 mice (lung tumors and splenic hemangiosarcomas). Also, as the confidence in Tg.rasH2 studies using urethane grew, in all subsequent studies control and high dose groups of CByB6F1 wild-type mice were dropped. Thereafter, a pilot study was conducted 1 using 10 mice per sex for durations varying from 8 to 16 weeks. As a result of this experiment, we noted that at 10 to 12 weeks the mice in positive control groups demonstrated 100% incidence of lung tumors and splenic hemangiosarcomas. Subsequently, we changed the protocols and performed studies with 10 mice per sex and examined only lungs and spleens grossly and microscopically. These Tg.rasH2 studies showed 100% incidence of lung and splenic tumors. From around 2015 and onward, we conducted Tg.rasH2 studies with urethane as a positive control material and the mice were sacrificed depending on clinical signs of rapid and shallow breathing, distended abdomen, and prostrate conditions between 10 and 12 weeks. At necropsy, only lungs were examined for gross lesions. Gross lesions, even if present in any other organs, were not recorded grossly and not examined microscopically. The lungs were harvested from 10 mice of each sex and examined microscopically. The remaining carcass and all tissues other than lung were saved in 10% neutral buffered formalin. We noted 100% incidence of lung tumors in each of the studies.
Historical control data using urethane had been established in laboratories such as Millipore Sigma (previously known as BioReliance). Several other laboratories and Charles River Laboratories (CRL) had been successfully using NMU as a positive control material. 10 Drawbacks with NMU include its unstable nature and potentially challenging procurement due to limited vendors. Examples of common microscopic lesions that are considered to be NMU induced are lymphomas, skin papillomas, and stomach papillomas, among others. In order to establish the historical control data using urethane and Tg.rasH2 mice, 3 experiments at 3 different sites of CRL were conducted. In the original study (study 1) after the first dose of 1,000 mg/kg/dose of urethane was administered, the mice demonstrated untoward clinical signs such as sedation of significant duration/severity, decreased activity, ataxia, impaired limb function, low carriage, and loss of skin elasticity, particularly in females. Subsequently, the next 2 doses were reduced to 500 mg/kg/dose of urethane. The other 2 studies conducted included 3 intraperitoneal injections of urethane at 500 mg/kg/dose on days 1, 3, and 5. The results of all 3 studies combined show conclusively that the lower dose at 500 mg/kg/dose and lower number of mice can be used successfully to prove the validity of the assay.
Material and Methods
Three studies conducted are designated as studies 1, 2, and 3. Ten male and 10 female Tg.rasH2 and CByB6F1 mice for each study were obtained from Taconic Biosciences. Ten males and females randomized separately were assigned to groups upon receipt. Age at the initiation of dosing was 8 and 1/2 to 12 weeks and average weight at initiation depending on sex was 17.8 and 27.1 g, respectively, for females and males. Urethane obtained from Sigma-Aldrich, Inc was dissolved in 0.9% sodium chloride injection USP obtained from MWI Veterinary Supply Company. Formulations of the urethane positive control material were prepared by mixing the appropriate amount of vehicle in 0.9% sodium chloride to achieve nominal concentration of 100 mg/mL. Urethane was scheduled to be administered on days 1, 3, and 5 of the study. Animals were acclimatized for 2 weeks and were identified by subcutaneously implanted electronic identification chips. Animals were fed ad libitum using Block Lab Diet (Certified Rodent Diet #5002 or #5CR4, PMI Nutrition International, Inc.). Ultraviolet-treated reverse osmosis deionized water was available ad libitum to each animal via an automatic watering system. Veterinary care, environmental conditions, cage side observations, observations for morbidity, mortality, injury, and the availability of food and water were conducted as per the protocol for each study. Postmortem study evaluations were performed on all animals euthanized at the scheduled necropsy. Animals were euthanized by carbon dioxide inhalation followed by Standard Operating Procedure (SOP) approved method to ensure death. Necropsy examinations were performed under procedures approved by a veterinary pathologist. Macroscopically lungs and spleens were examined from all 60 mice on the study. Microscopic examination of lungs was performed on all 60 lungs, irrespective of presence or absence of any gross lesions. Microscopic examination of spleens was performed on 39 spleens that had grossly visible lesions. All sections were routinely processed and hematoxylin and eosin-stained paraffin sections were examined. The first study (study 1) was started with 1,000 mg/kg/dose on day 1. However, because of untoward clinical signs in female mice such as sedation of significant duration/severity, decreased activity, ataxia, impaired limb function, low carriage, and loss of skin elasticity, the dose was subsequently lowered to 500 mg/kg/dose for days 3 and 5. The other 2 studies (studies 2 and 3) were conducted with the dose of 500 mg/kg/dose for all days (days 1, 3, and 5). All studies were terminated after 12 weeks of duration. A total of 60 CbyB6F1 mice used as controls were similarly divided into 30 males and 30 females and were further assigned to 10 mice/sex to each group. As there were only rare tumors noted in CbyB6F1 mice, findings pertaining to control mice are not tabulated in the results.
The general study design for each study is shown in Table 1.
Study Designs.
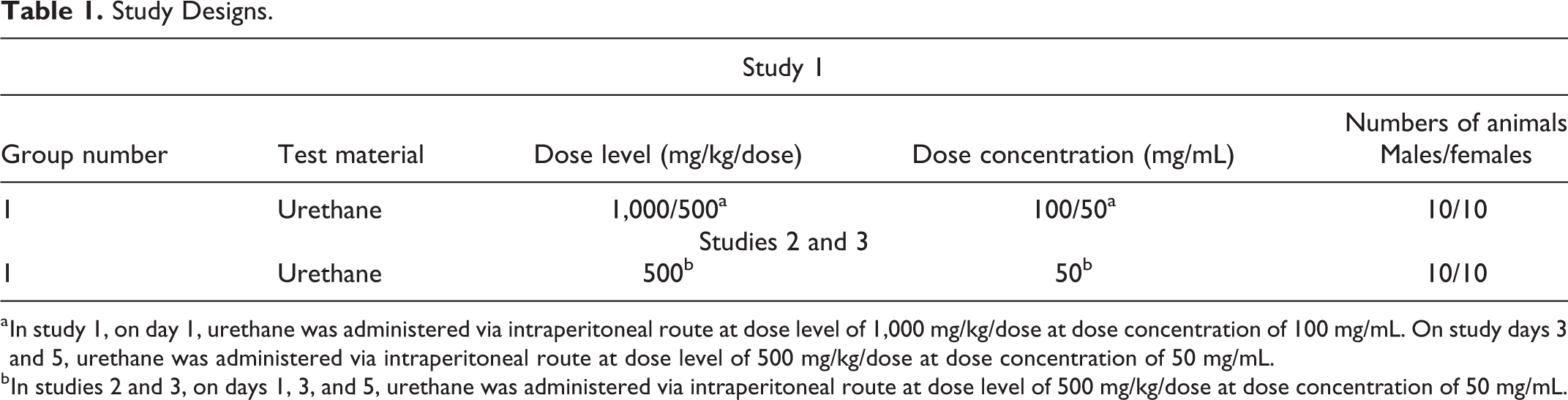
a In study 1, on day 1, urethane was administered via intraperitoneal route at dose level of 1,000 mg/kg/dose at dose concentration of 100 mg/mL. On study days 3 and 5, urethane was administered via intraperitoneal route at dose level of 500 mg/kg/dose at dose concentration of 50 mg/mL.
b In studies 2 and 3, on days 1, 3, and 5, urethane was administered via intraperitoneal route at dose level of 500 mg/kg/dose at dose concentration of 50 mg/mL.
Results
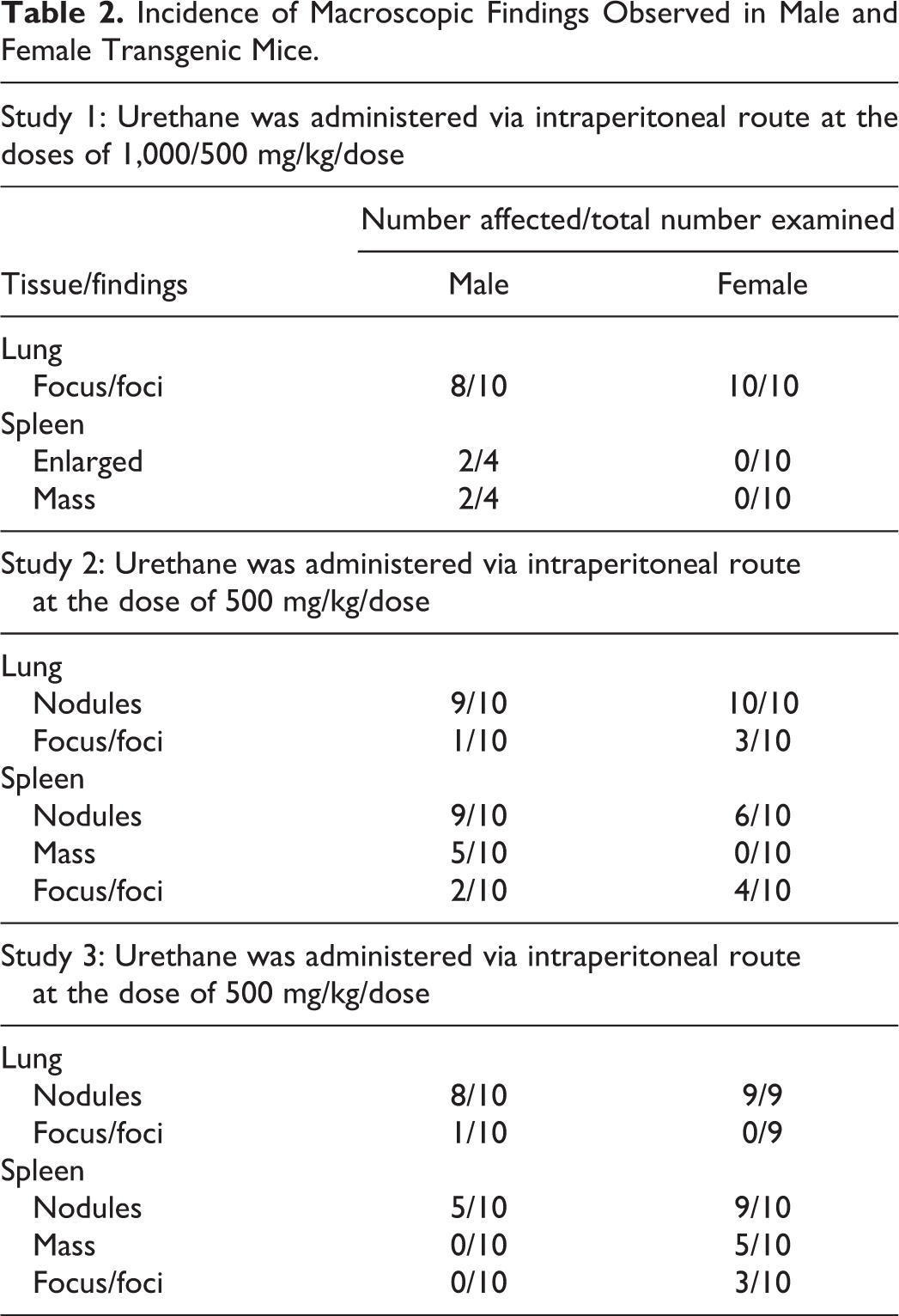
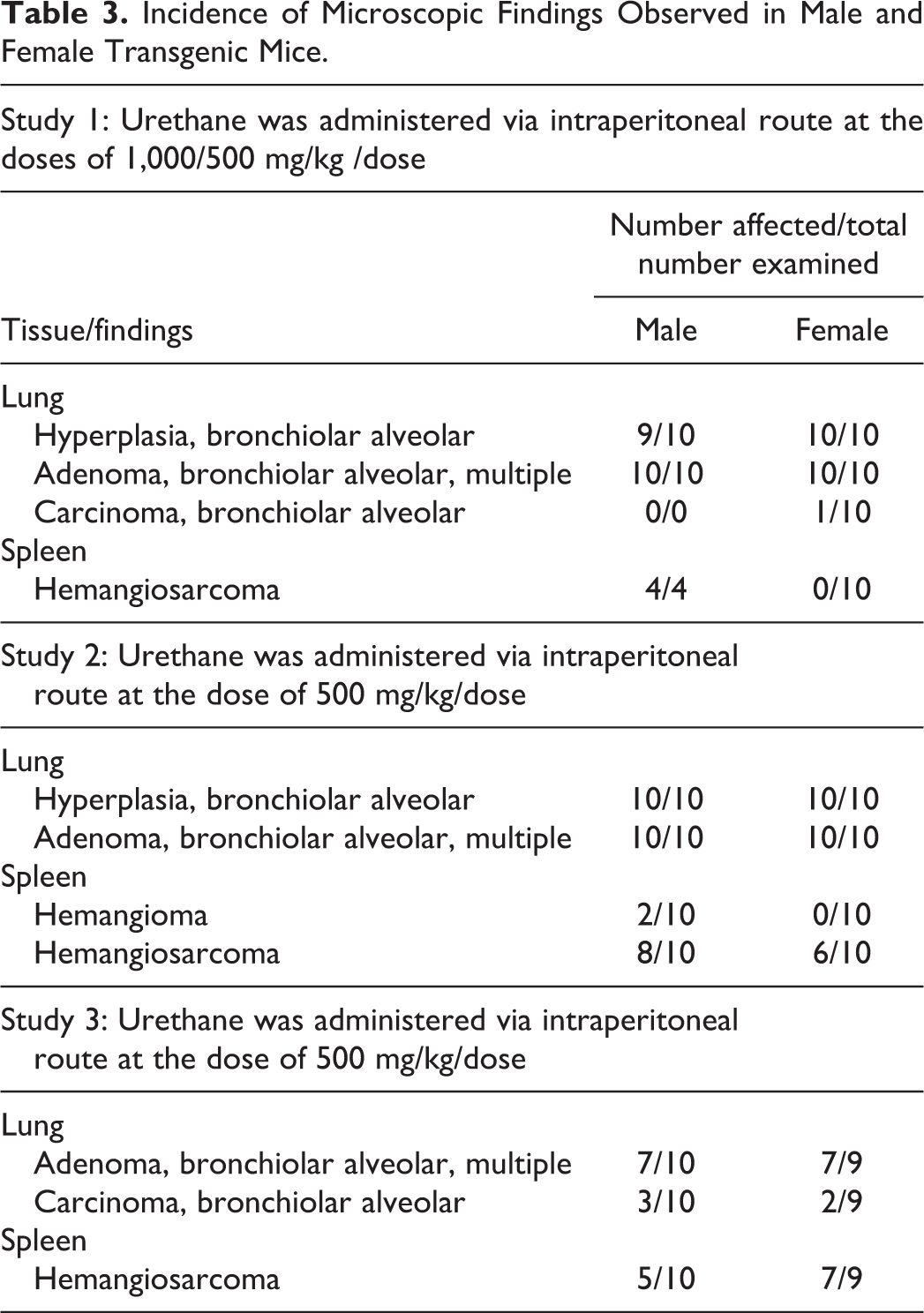
In none of the 3 studies was there any mortality or any clinical signs/symptoms at the end of the study. Macroscopic and microscopic lesions noted in lungs and spleen of each study conducted in Tg.rasH2 mice are presented in Tables 2 and 3.
Incidence of Macroscopic Findings Observed in Male and Female Transgenic Mice.
Incidence of Microscopic Findings Observed in Male and Female Transgenic Mice.
In study 1, on day 1, 1,000 mg/kg/dose of urethane was injected in all 10 males and 10 females. Urethane being an anesthetic exhibits signs of sedation immediately after injections. However, per animal welfare and Institutional Animal Care and Use Committee perspective, the signs exhibited were considered deep and exaggerated. The clinical signs exhibited included sedation of significant duration/severity, decreased activity, ataxia, impaired limb function, low carriage, and loss of skin elasticity noted particularly in females. As these signs were considered to be exaggerated, the dose was dropped to 500 mg/kg/dose in both sexes for next 2 doses on days 3 and 5. For the subsequent 2 studies (studies 2 and 3), all mice were injected with 500 mg/kg/dose for all dose groups on days 1, 3, and 5. At 500 mg/kg/dose, exaggerated clinical signs were not observed. However, mild signs of sedation of less severity and duration were noted.
In 56 of the 60 mice treated with urethane in the 3 studies, there were lung nodules, foci, or masses noted grossly. Microscopically, bronchioalveolar hyperplasia was noted in 39 mice, multiple adenomas were noted in 54 mice, and carcinomas were noted in 6 mice. Thus, in each study the combined incidence of adenomas and carcinomas is 100%
In 39 of the 60 mice treated with urethane in the 3 studies, there were splenic enlargement, nodules, masses, and foci noted grossly. Microscopically, hemangiosarcomas and hemangiomas were noted in 33 mice. Thus, incidence of hemangiomas or hemangiosarcomas noted in these 3 pilot experiments was lower compared to lung tumors.
In contrast to the Tg.rasH2 mice, of 60 CByB6F1 lungs examined, only 4 had alveolar bronchiolar adenomas, whereas none had any splenic hemangiosarcomas.
Discussion
The 3 studies performed here clearly indicate that in all 60 Tg.rasH2 mice treated with urethane (500-1,000 mg/kg) presented with lung tumors at the end of the 12-week period, diagnosed as alveolar bronchiolar adenoma, multiple, and alveolar bronchiolar carcinomas. A prior published paper 1 showed similar 100% presence of lung tumors in Tg.rasH2 mice dosed with urethane as a positive control material, at much higher dose of 1,000 mg/kg/dose/dose. In these 3 experiments, the percentage of splenic hemangiosarcomas was much lower, most likely because of lower dose of 500 mg/kg/dose in these experiments, compared to higher doses used at 1,000 mg/kg/dose in previous experiments. 1
We now have abundant experience with Tg.rasH2 mouse model with urethane as a positive control material. 8,9 After we started using the Tg.rasH2 mouse as an alternative model to perform carcinogenicity studies, all studies have included positive control groups. When the Tg.rasH2 model became an acceptable alternative model for carcinogenicity assessment, 25 Tg.tasH2 mice/sex were assigned to each of the positive control groups. The number of animals in the urethane-treated groups was eventually reduced to 10 mice/sex.
Based on the results of the 3 pilot experiments combined with our experience, we have 2 options. One option is to maintain single positive control group per test facility for every 2 years. Most ideal option is that there is really no need to include positive control groups in any of the 26-week Tg.rasH2 studies as genotypic certification of these mice is provided by the supplier Taconic. 11
Footnotes
Acknowledgments
The authors wish to acknowledge Luc Chouinard (
Author Contributions
Paranjpe, M. substantially contributed to conception or design, contributed to acquisition, analysis, or interpretation, drafted the manuscript, and critically revised the manuscript for important intellectual content; Rudmann, D., Sargeant, A., Morse, M., Yonpiam, R., Bonnette, K., and Albretsen, J. contributed to acquisition, analysis, or interpretation and critically revised the manuscript for important intellectual content; Papagiannis, C. substantially contributed to conception or design, contributed to acquisition, analysis, or interpretation, drafted the manuscript, and critically revised the manuscript for important intellectual content. All authors gave final approval and agree to be accountable for all aspects of the work in ensuring that questions relating to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
