Abstract
In mice, hyaline droplets in renal proximal tubules have been associated with histiocytic sarcoma but have not been reported with lymphoma. Tissues from CD-1 mice in a 2-year carcinogenicity bioassay were examined microscopically. Twenty-five mice with hyaline droplets in renal tubules were identified. Immunohistochemistry to detect IgA, IgG, IgM, lysozyme, albumin, CD3, and CD79a was performed on kidneys of 21 affected mice. Hyaline droplets were present in the kidneys of 11 mice with lymphoma (1 male, 10 female), of which 1 female also had histiocytic sarcoma. Hyaline droplets were also present in 7 other mice with histiocytic sarcoma, 2 with chronic progressive nephropathy, 3 with renal cortical tubular necrosis, and 2 with granulocytic leukemia. Five of the 11 lymphomas were CD3+, indicating a T lymphocyte origin. Hyaline droplets in mice with lymphoma did not stain for IgA, IgG, or IgM, except in one questionable case. Results of other immunohistochemical stains were inconclusive. Although the droplet composition could not be determined immunohistochemically, the study findings indicate that renal tubular hyaline droplets may be associated with lymphoma in mice.
Keywords
Introduction
Hyaline droplets accumulate in the renal proximal tubules of rodents secondary to a variety of conditions. In male rats, spontaneous or induced α2u-globulin overproduction can cause hyaline droplet formation (Greaves 2007; Doi et al. 2007). In mice and rats, chronic progressive nephropathy (CPN) can induce hyaline droplets composed primarily of albumin (de Rijk et al. 2003).
Another condition associated with hyaline droplets in renal tubules of rodents is histiocytic sarcoma (Hard and Snowden 1991; Seely 1999; Wolf and Hard 1996). The neoplastic histiocytes produce large amounts of lysozyme. The lysozyme accumulates in the tubular epithelium to such an extent that the epithelial cells’ ability to catabolize this protein is exceeded.
Less commonly, hyaline droplets have been associated with other neoplasms. F344 rats in one report had a rat fibrosarcoma-derived transplantable tumor (Yamate et al. 1998), and F344 rats in another report had a meningioma-derived transplantable tumor (Yamate et al. 1994). The tumors in these two reports did not produce lysozyme, and the pathogenesis of the droplet formation was not determined. Hyaline droplet nephropathy associated with eosinophilic crystals composed of Ym1/Ym2 chitinase-like proteins has been described in a mouse model of acute myelogenous leukemia (Marchesi et al. 2003). Hyaline droplets in renal tubules have also been reported in association with myelomonocytic leukemia in humans (Schwarze 1975).
To the authors’ knowledge, eosinophilic hyaline droplets have not previously been associated with lymphoma in rodents. We describe a study in which there was an association between lymphoma and renal tubular hyaline droplets in mice, particularly in females.
Materials and Methods
A 2-year bioassay to investigate the carcinogenic potential of a small molecule on CD-1 mice (Charles River Laboratories, Portage, MI) was conducted at Covance Laboratories (Madison, WI). All procedures were in compliance with the Animal Welfare Act, the Guide for the Care and Use of Laboratory Animals, and the Office of Laboratory Animal Welfare. Mice were housed individually in animal rooms that were set to maintain a temperature of 18 to 26°C and a relative humidity of 30% to 70%, with a 12-hour light/12-hour dark cycle. Mice were fed certified rodent diet #8728C (Harlan Teklad, Indianapolis, IN) ad libitum and were allowed free access to water. Mice were 4 to 7 weeks old at the start of the study.
The test article was administered daily via oral gavage at doses of 0 (control), 30 (low-dose), 80 (mid-dose), and 200 (high-dose) mg/kg/day. Each dosage group had 65 males and 65 females at the start of the study (a total of 520 animals). Necropsies were performed on all animals, including those that died spontaneously or were euthanized in a moribund condition prior to the end of the 730-day period. Representative samples of multiple tissues from each mouse were fixed in neutral buffered formalin, embedded in paraffin, sectioned at approximately 5 µm, placed on glass slides, and stained with hematoxylin and eosin (H&E).
The pathologic findings in the study were interpreted by one pathologist (LWD), with a peer review performed by second pathologist (JHD). Following the interpretation of study results, kidneys from 21 of the 25 animals that had hyaline droplets in renal tubules were processed for immunohistochemistry (IHC). Kidneys with no apparent hyaline droplets from animal 26 (a female mouse in the control dosage group) were also processed for IHC to serve as a negative control. Antibodies against mouse immunoglobulin (Ig)A (NB7501, Novus Biologicals, Littleton, CO), mouse IgG (CLCC30200, Cedarlane, Burlington, NC), mouse IgM (NB7499, Novus Biologicals), human lysozyme (macrophage marker) (A0099, Dako, Carpinteria, CA), mouse albumin (ab19194-1, Abcam, Cambridge, MA), human CD3 (T lymphocyte marker) (ab16669, Abcam), and human CD79a (B lymphocyte marker) (LS-B1061, LifeSpan BioSciences, Seattle, WA) were applied to the slides utilizing horseradish peroxidase polymer detection (Biocare Medical, Concord, CA), diaminobenzidine chromogen, and hematoxylin counterstain. Periodic acid-Schiff (PAS) staining (87007, Richard-Allan Scientific, Kalamazoo, MI) and chromotrope-aniline blue (CAB) staining (K006, Poly Scientific, Bay Shore, NY) as described by de Rijk et al. (2003) were performed on the kidneys of animal 5, a representative low-dose female with lymphoma.
Results
Incidences of selected diagnoses by dosage group are presented in Table 1. Hyaline droplets were present in the kidneys of 25 mice, 11 of which were diagnosed with lymphoma (Figure 1). The hyaline droplets were present in all treatment groups, including controls. Other conditions that were associated with hyaline droplets included histiocytic sarcoma (n = 8), CPN (n = 2), granulocytic leukemia (n = 2), and subacute cortical tubular necrosis (n = 3) (Table 2; Figures 2 and 3). These numbers do not add up to 25 because several animals had zero or two diagnoses (e.g., animal 11 was diagnosed with both lymphoma and histiocytic sarcoma). Hyaline droplets were round, eosinophilic, and approximately 1–2 µm in diameter. In most of the affected kidneys, droplets were most prevalent in the P2 segment of proximal tubules, occasionally extending into the P3 segment. All affected animals died or were euthanized in a moribund condition prior to the completion of the study, with the exception of three females (animals 9, 19, and 25). The hyaline droplets were not associated with death or moribund condition and were not considered adverse.
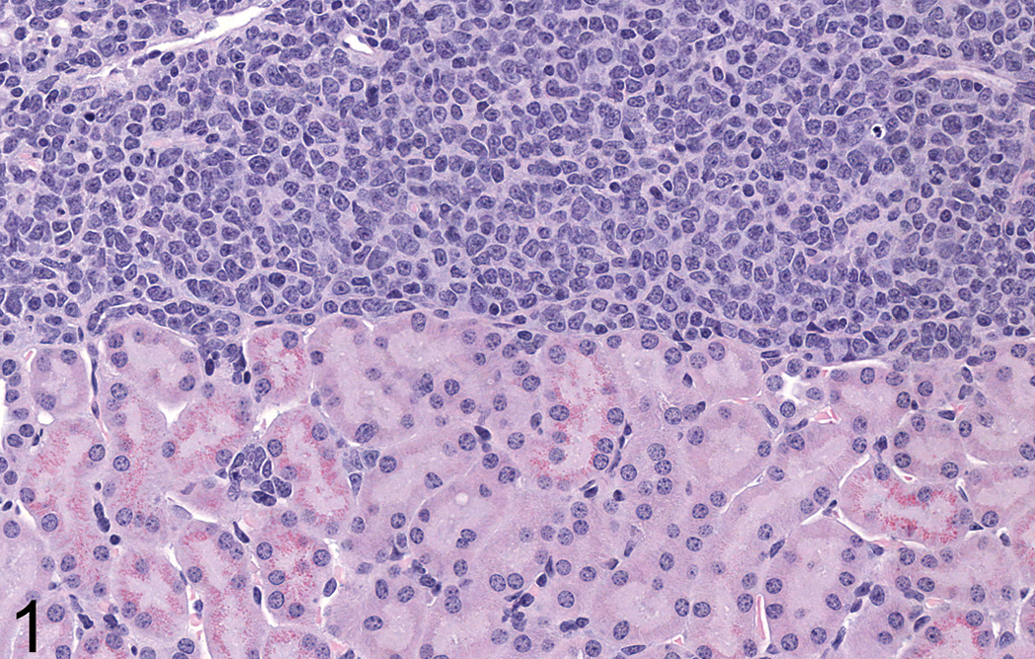
Lymphoma in kidney of animal 4 (control female). Large numbers of neoplastic lymphocytes are present along the capsular surface. Hyaline droplets are present along the brush border of occasional tubules. Original magnification 360×. H&E.
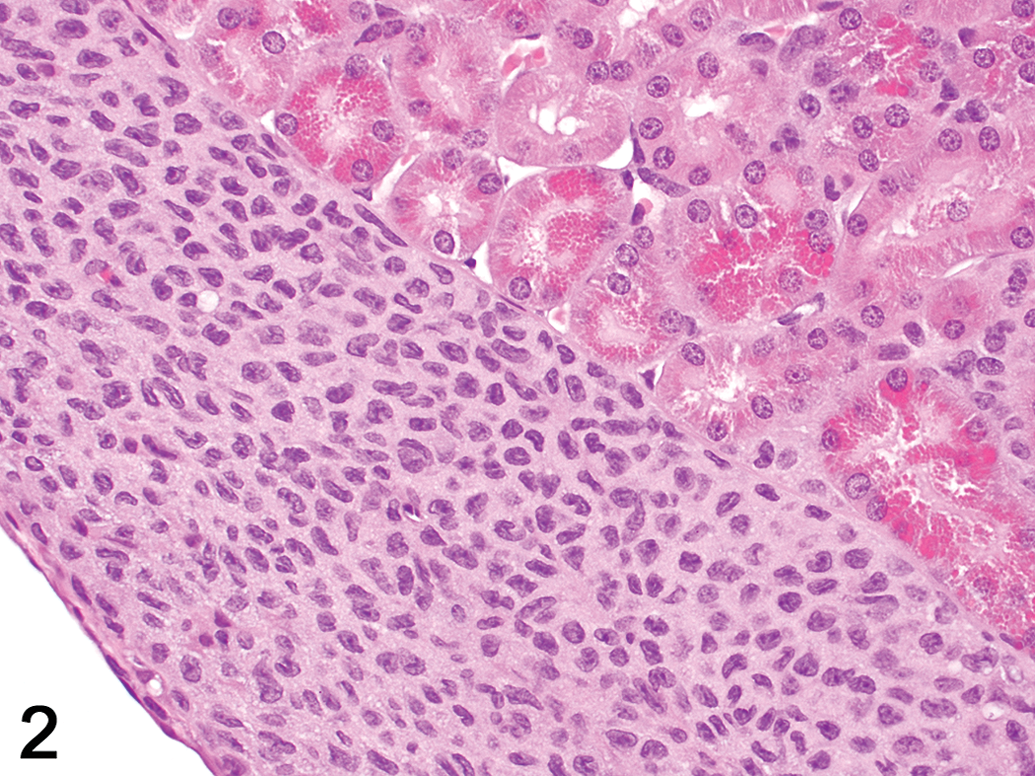
Histiocytic sarcoma in kidney of animal 16 (high-dose female). Neoplastic cells infiltrate the subcapsular space. Large numbers of brightly eosinophilic hyaline droplets are present in the epithelium of many proximal tubules. Original magnification 400×. H&E.
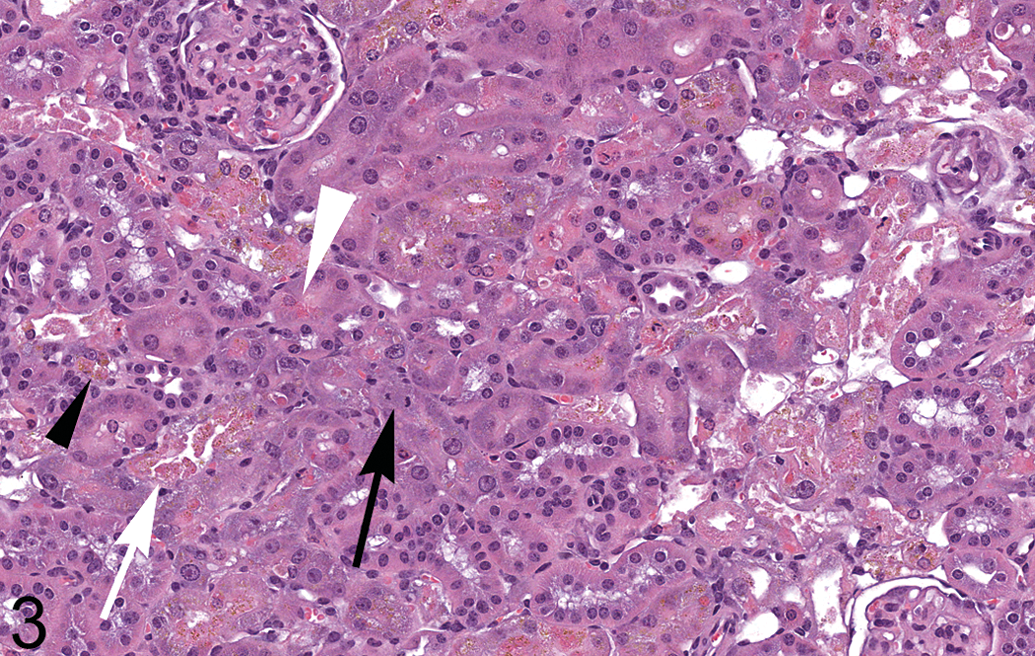
Subacute cortical tubular necrosis in kidney of animal 22 (control female). Hyaline droplets (white arrowhead), brown pigment (black arrowhead), intraluminal cellular debris (white arrow), and regenerative epithelium with basophilic cytoplasm and a mitotic figure (black arrow) are present. Original magnification 250×. H&E.
Incidence of selected diagnoses by dosage group.
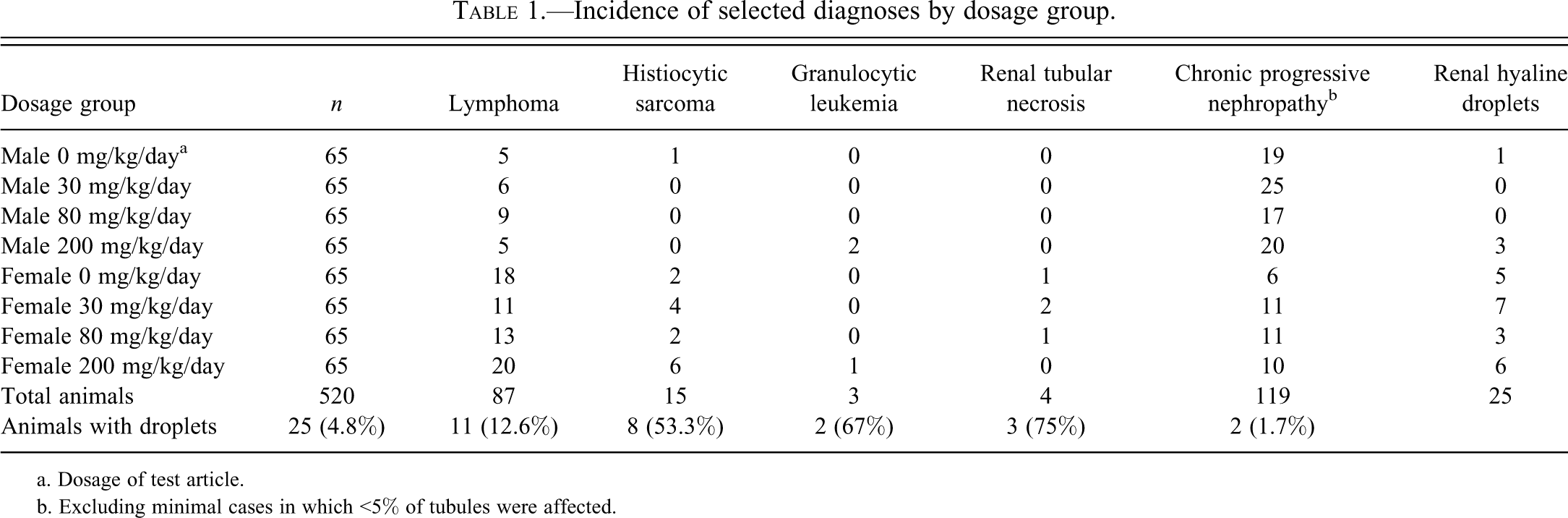
a. Dosage of test article.
b. Excluding minimal cases in which <5% of tubules were affected.
Individual animal data: mice with renal tubular hyaline droplet formation.
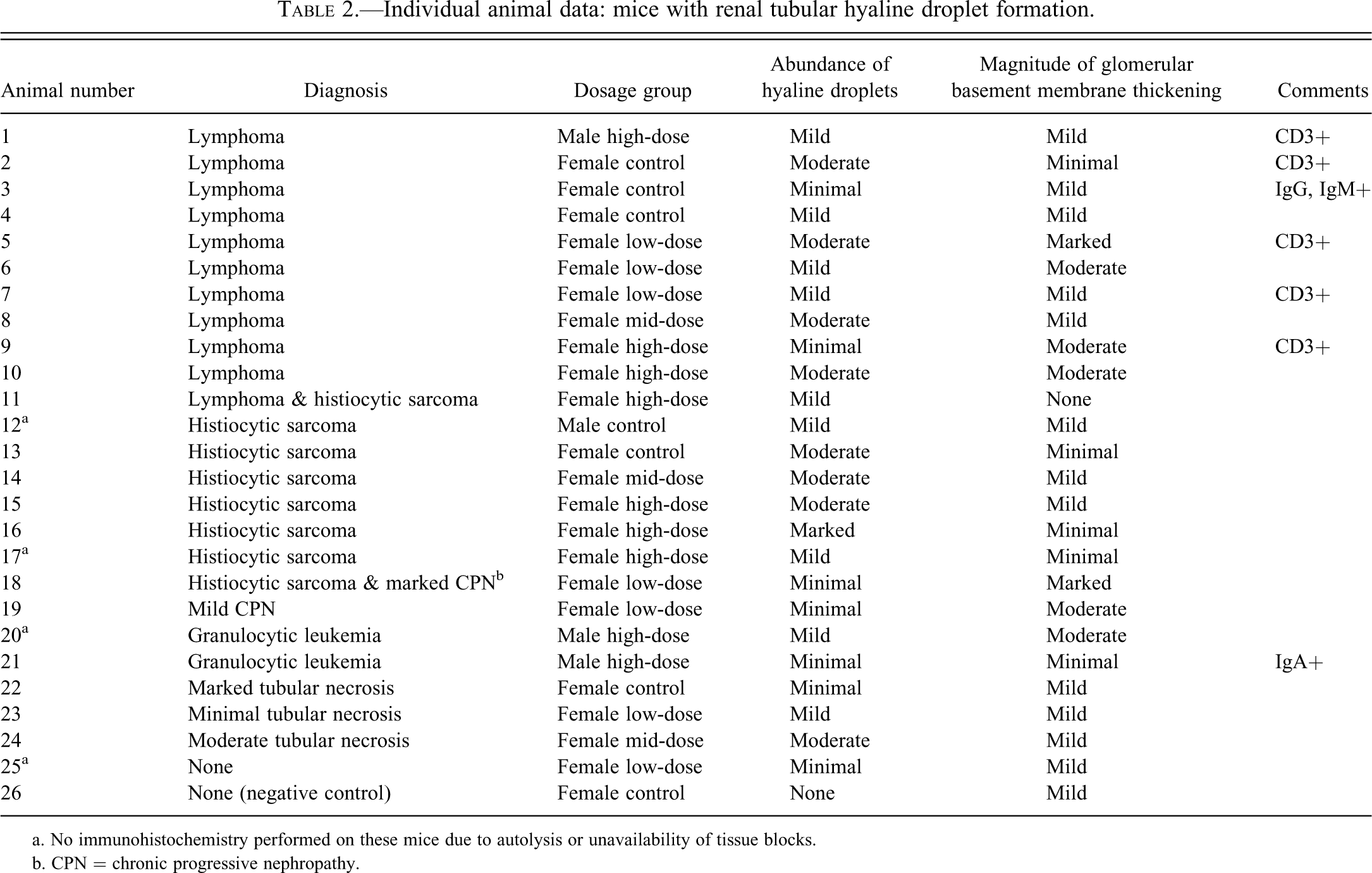
a. No immunohistochemistry performed on these mice due to autolysis or unavailability of tissue blocks.
b. CPN = chronic progressive nephropathy.
The hyaline droplets in the renal tubules of animal 5 stained red with CAB staining (Figure 4), indicating their protein composition (de Rijk et al. 2003), while the droplets were negative for the PAS reaction (not shown), indicating their lack of polysaccharide content (Hard and Snowden 1991).
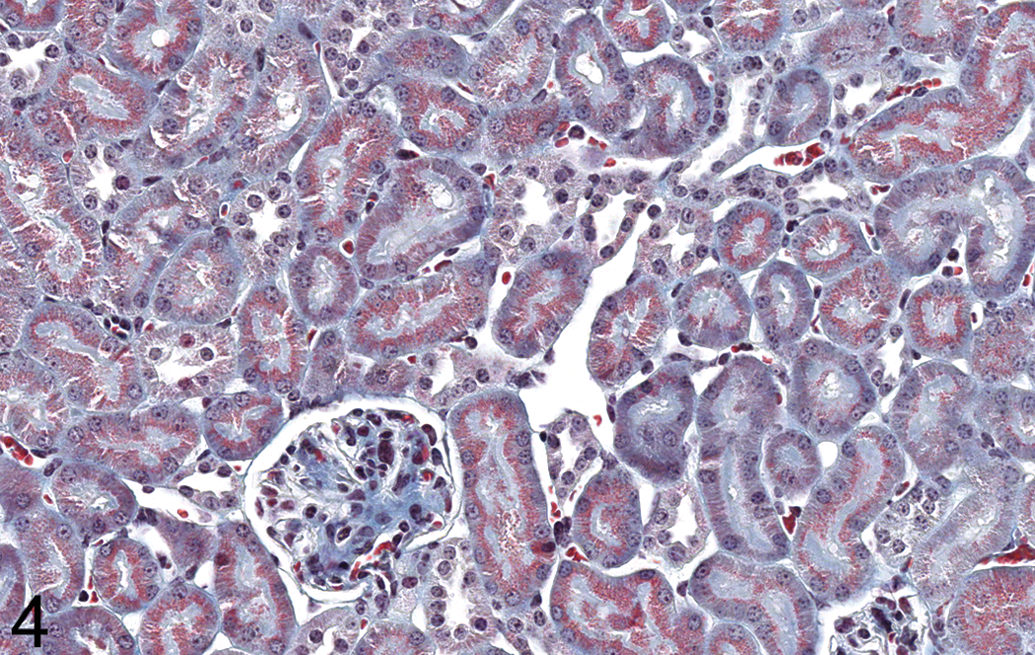
Kidney, animal 5 (low-dose female). Hyaline droplets in proximal tubules stain red with chromotrope-aniline blue staining procedure in this lymphoma-affected mouse, indicating their protein composition. Original magnification 400×.
Minimal to marked thickening of glomerular basement membranes was present in most study animals with and without hematopoietic neoplasms (Table 2; Figure 5). There was no appreciable difference in incidence or magnitude of this change between mice with hematopoietic neoplasms and without. The most severe instances of CPN were generally associated with marked glomerular basement membrane thickening. No correlation between glomerular basement membrane thickness and the abundance of tubular hyaline droplets was noted.
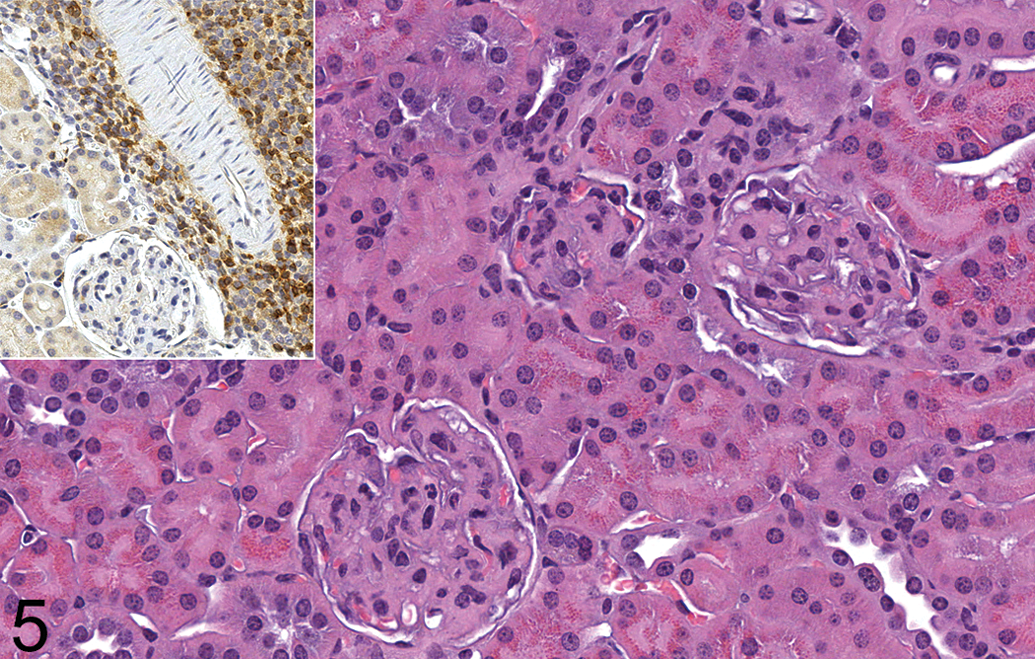
Kidney, animal 5 (low-dose female). Moderate numbers of hyaline droplets are present in proximal tubular epithelium. Basement membranes of the glomeruli are thickened. H&E. Original magnification 400×. Inset: Some neoplastic lymphocytes stain positively for CD3. Original magnification 290×.
Hyaline droplets were immunohistochemically negative for IgA, IgG, and IgM in all kidneys (Figure 6), except in 2 animals. Droplets along tubular brush borders stained positively for IgG and IgM in animal 3 (a female with lymphoma), but the significance of this finding is questionable because the tissue was moderately autolyzed and most of the positive staining occurred in the P3 segment of tubules rather than in the P2 segment. Droplets were positive for IgA in animal 21, a male with granulocytic leukemia. The significance of this finding is unknown. There were no kidneys in which the neoplastic cells were definitively positive for IgA, IgG, or IgM. (No neoplastic cells were present in the kidneys of animal 13, a female with histiocytic sarcoma.) Glomerular basement membranes in mice with lymphoma or histiocytic sarcoma did not stain with immunoglobulins to any appreciable magnitude greater than in the negative control mouse.
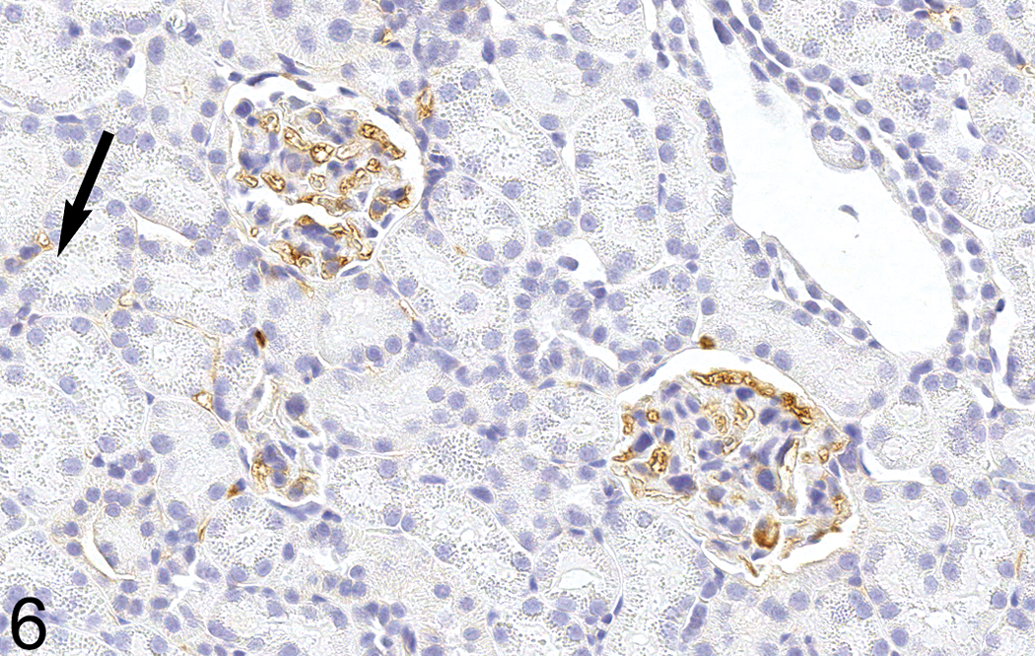
Kidney, animal 2 (control female). Hyaline droplets (arrow) do not stain for IgA in this lymphoma-affected mouse. Glomerular basement membrane staining is similar to that observed in animal 26 (negative control, not shown). Original magnification 400×.
Staining for CD3 revealed moderate to strong positive staining of large numbers of neoplastic cells in lymphoma-affected animals 1, 2, 5, 7, and 9 (Figure 5 inset). This indicated that the neoplastic cell population was composed at least partially of T lymphocytes in these mice.
Results of IHC staining of the kidneys for albumin, lysozyme, and CD79a were inconclusive. There was extensive background staining of numerous structures for albumin and lysozyme. (Extensive background staining also was present in the positive control tissue for albumin, mouse liver, but little background staining was noted in the positive control tissue for lysozyme, mouse lymph node.) CD79a staining did not reveal neoplastic or non-neoplastic resident B lymphocytes in any examined kidney, even though B lymphocytes were detected in the positive control tissue (mouse small intestine).
Discussion
Results of this study indicate that hyaline droplets within renal proximal tubules may be associated with lymphomas in mice, especially in females. Unfortunately, the inconclusiveness of the IHC results hindered our ability to characterize the lymphoma-associated hyaline droplets further. Factors such as prolonged formalin fixation can hinder IHC staining, a problem also encountered in a study of hyaline droplets in tumor-bearing Wistar rats (Walsh 1991).
There are several possible explanations for the lymphoma-associated hyaline droplets in renal tubules. The neoplastic lymphocytes may be producing excessive amounts of a protein that is absorbed by the tubular epithelium from the glomerular filtrate. As with lysozyme in the case of histiocytic sarcoma, excessive amounts of this protein would overwhelm the epithelial cells’ ability to catabolize it. Immunoglobulins or immunoglobulin subunits are a possibility, although the droplets did not stain for immunoglobulin in any case of lymphoma in our study, except in one questionable instance. Also, excessive immunoglobulin production would not be expected in the T-cell lymphomas, although lymphomas composed of both B and T lymphocytes have been described in mice (Hao et al. 2010). Alternatively, the neoplastic lymphocytes may be secreting immunoglobulins that form immune complexes that deposit in glomeruli, leading to complement activation, endothelial dysfunction, and serum protein leakage. This mechanism seems unlikely in this study, because glomerular basement membranes in animals with lymphoma were not appreciably thicker than in the other mice. Finally, the neoplastic lymphocytes may have been intermingled with large numbers of non-neoplastic histiocytes (histiocyte-associated lymphoma), a subset of lymphoma described in mice (Hao et al. 2010). These non-neoplastic histiocytes could produce excessive lysozyme that would accumulate in tubular epithelial cells.
In conclusion, hyaline protein droplets in renal proximal tubules were associated with 11 cases of lymphoma as well as 8 cases of histiocytic sarcoma in this 2-year study of CD-1 mice. Although the composition of the droplets could not be determined, the study findings suggest that lymphoma should be included in the differential diagnosis of hematopoietic neoplasms associated with tubular hyaline droplets. The incidence of the lymphoma-associated hyaline droplets was greater in females than in males in this study, but this may reflect the greater overall incidence of lymphoma in females than in males in the study and in published data (Baldrick and Reeve 2007). A previous report found zero instances of hyaline droplet accumulation in 60 B6C3F1 mice with lymphoma (Hard and Snowden 1991); perhaps this phenomenon is specific to CD-1 mice. Future investigations may be able to characterize the droplets further and provide more information on the pathogenesis of this condition.
Footnotes
Acknowledgments
We would like to thank Jacqueline O’Connor for excellent assistance with immunohistochemistry, and Holly Jakel and the histology staff of Covance Laboratories for histologic slide preparation and for timely shipment of slides and tissue blocks.
