Abstract
A male CD-1 mouse from a segment I reproduction toxicity study died at the age of fifteen weeks. At necropsy, the thymus was abnormally enlarged and virtually obliterated the thoracic cavity. Histopathological examination of thymus revealed abundant cell population consisting of mature lymphocytes and few lymphoblasts arranged in cords and sheets. Neoplastic cells completely obliterated the thymic architecture, and loss of corticomedullary junction was evident. Microscopically, neoplastic cells were round with moderate cytoplasm. Nuclei were spherical and hyperchromatic with centrally placed nucleoli. Mitotic figures and tingible body macrophages imparting “starry sky appearance” were seen. Multifocal necrotic areas were also observed. Proliferating neoplastic cells invaded the thymic capsule, surrounding adipose tissue and neighboring organs such as lungs, heart and fat pad of aorta, esophagus, trachea, and thoracic vertebrae. No evidence of systemic metastasis was observed in other organs. Immunostained neoplastic cells were positive for CD3 antigen. Gross, histopathology, and immunohistochemistry results were suggestive of thymic T-cell lymphoma.
Most rodent tumors are age-related, and data on their occurrence during the first year are scarce (Son and Gopinath 2004). Lymphomas are one of the most common tumors in many strains of mice (Haines, Chattopadhyay, and Ward 2001; Ward 2006); they are the fifth or sixth most common target site of carcinogens (Ward 2006). Lymphomas are the most prominent tumor in the hematopoietic system (Maita et al. 1988), occurring at an incidence of 8.8% in male and 22.0% in female CD-1 mice (Sher, Jensen, and Bokelman 1982; Maita et al. 1988). Incidences of 5–24% in CD-1 and B6C3F1 mice were reported recently (Haseman, Boorman, and Huff 1997; Ward 2006). These occurrences were influenced by several factors such as strain, stock, sex, and age (Maita et al. 1988; Gopinath 1994; Son and Gopinath 2004; Ward 2006). Neoplasms of the thymus in mice are most commonly T-cell lymphomas originating from thymic lymphocytes (Maronpot, Boorman, and Gaul 1999; Pearse 2006a). Occurrence of thymic lymphomas was reported in female CD-1 mice at a rate twice greater than in males (Maita et al. 1988). Almost all reported thymic lymphomas are systemic, and the literature on the locally invasive, nonsystemic, thymic T-cell lymphomas in the male CD-1 mouse is meager. Therefore, an attempt has been made to elucidate the morphological features of this tumor.
The animals from this study were housed in an environmentally controlled experimental room with a temperature of 19–26°C, a relative humidity of 34–69%, and a twelve hours light: twelve hours dark cycle and were kept in polycarbonate cages with rice husk bedding and stainless steel grill. The mouse was provided ad libitum autoclaved standard pelleted laboratory animal feed (Netherlands BV, Kreuzelweg 53, 5961 NM Horst, The Netherlands) and purified water (reverse osmosis). Proximate analysis of feed and contaminants analysis of water supplied were within the permissible limits. The experimental animals were maintained as per the recommendations of the Committee for the Purpose of Control and Supervision of Experiments on Animals guidelines for the Laboratory Animal Facility of India. The study protocol was approved by the Institutional Animal Ethical Committee.
A male CD-1 (Crl: CD-1) mouse from the low-dose treatment group in a segment I reproduction toxicity study died at the age of fifteen weeks. The mouse received the test item orally for a period of forty-five days. The mouse died on day 46 of the experiment with a history of dyspnoea. At necropsy, the mouse showed an abnormally enlarged thymus that virtually obliterated the thoracic cavity (Figure 1 ). The entire heart and visceral surfaces of the lungs were masked by the enlarged thymus. The enlarged thymus occupied almost three-fourths of the thoracic cavity starting from thoracic inlet to diaphragm. The whitish-colored thymus was shaded with yellow and a light pink color. It was soft in consistency, and no demarcation between the lobes was observed. Thymus was measured 1.2 × 2.3 × 1.4 cm and weighed about 1.013 g. Thymus was collected along with other protocol organs in 10% neutral buffered formalin. Tissues were trimmed seventy-two hrs postfixation, processed and then embedded in paraffin, sectioned at 3–5 μ thickness, and stained with hematoxylin and eosin (HE).
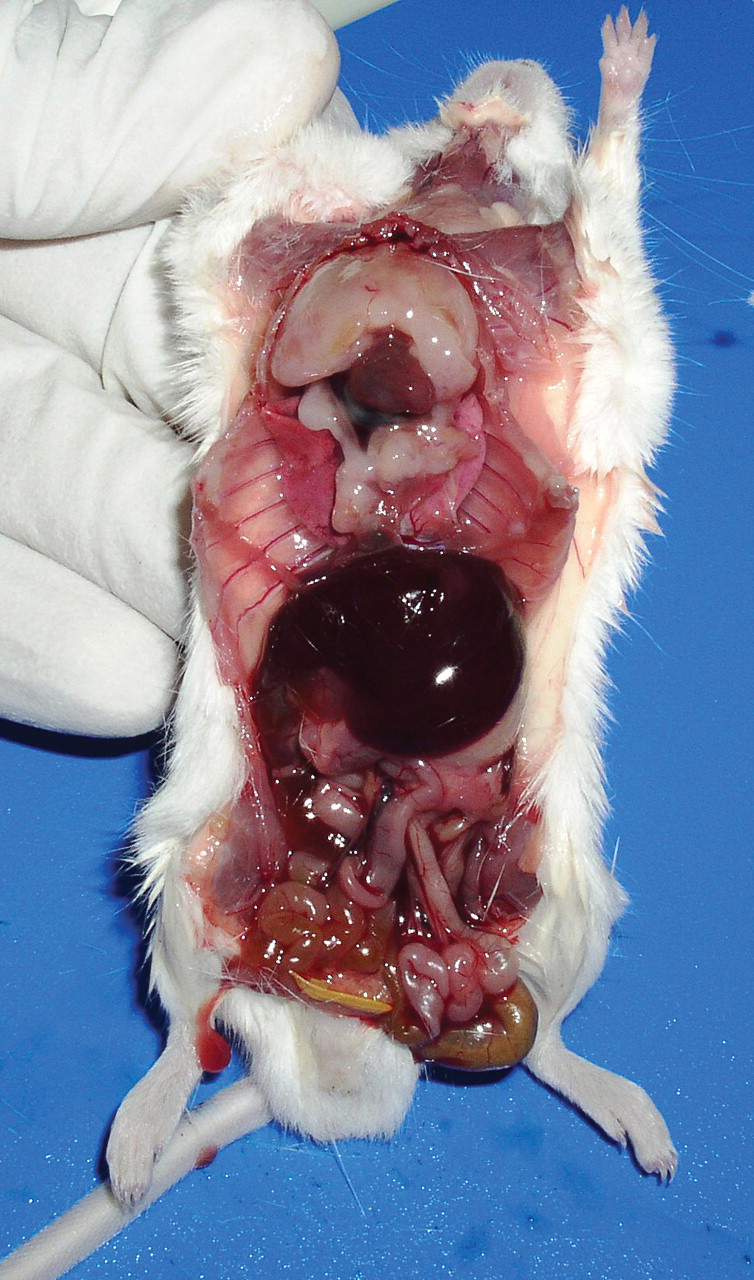
Enlarged thymus adhered with lungs and heart and obliterated thoracic cavity.
Immunohistochemical (IHC) stainings were performed in paraffin-embedded tissue sections. Briefly, a few sections were incubated with primary antibody T-cell marker [Rabbit polyclonal to CD3 (ab5690)] followed by Horseradish Peroxidase conjugated secondary antibody (Goat anti-rabbit) and counterstained with hematoxylin. Some of the sections were incubated with B-cell marker [Rabbit monoclonal {EP459Y} to CD20 (ab78237)] and epithelial cell marker [Mouse monoclonal {PCK-26} to pan cytokeratin (ab6401)] followed by Streptavidin Peroxidase conjugated secondary antibodies (Goat anti-rabbit and goat anti-mouse) and counterstained with hematoxylin.
Microscopically, abundant cell population consisting of mature lymphocytes and few lymphoblasts (Figure 2) arranged in cords and sheets were observed in thymus. Neoplastic cells completely obliterated the thymic architecture, and loss of corticomedullary demarcation was evident. Proliferating cells invaded the thymic capsule (Figure 3), neighboring adipose tissue, and thoracic organs as well. Neoplastic cells were round with moderate cytoplasm. Nuclei were spherical and hyperchromatic with prominent central nucleoli. Multiple mitotic figures and tingible body macrophages imparting “starry sky appearance” were observed (Figure 4). Increased angiogenesis and multifocal areas of necrosis comprised of neoplastic cells were seen (Figure 5). Neighboring adipose tissue and adjacent organs such as lungs, heart, aorta, esophagus, trachea, and thoracic vertebrae were invaded with neoplastic cells. In the lungs, peribronchial invasion of neoplastic cells entirely covered the bronchial tree bilaterally (Figure 6). Neoplastic cells invaded in the pericardium and myofibrils of both the auricles and ventricles (Figure 7). Infiltration of neoplastic cells was observed in the peri-aortic adipose tissue (Figure 8). Neoplastic cell infiltration also extended into peripheral fat cushion of the esophagus, trachea, and thoracic vertebrae. All other organs examined microscopically did not reveal any evidence of metastasis. Immunohistochemistry results of the thymus (Figure 9), peribronchial tree of lungs, and heart showed abundant expression of CD3 stained neoplastic T-lymphocytes. Also, few CD20 positive B-lymphocytes (Figure 10) and scattered clusters of cytokeratin stained epithelial cells (Figure 11) were observed in the thymus.
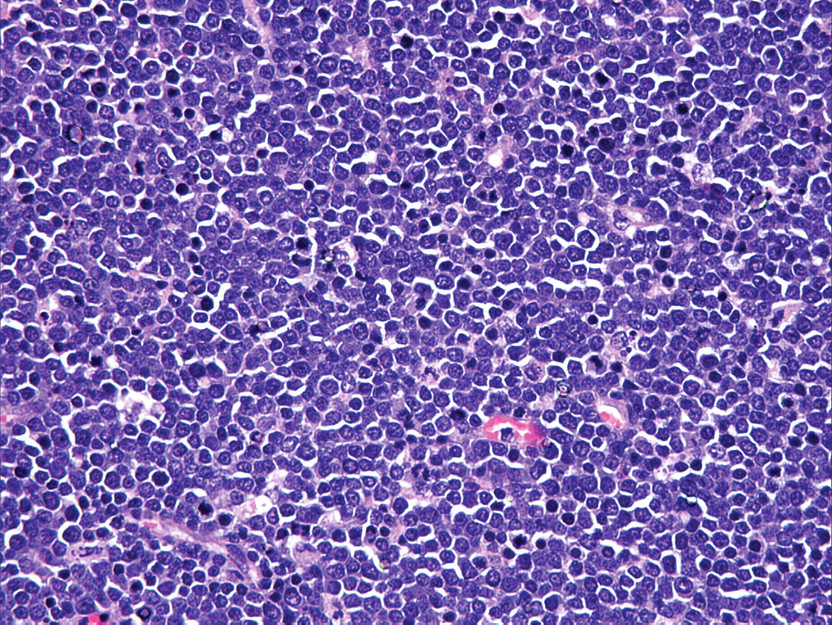
Thymus: Neoplastic cells consisting of mature lymphocytes and few lymphoblasts. HE, 400×.
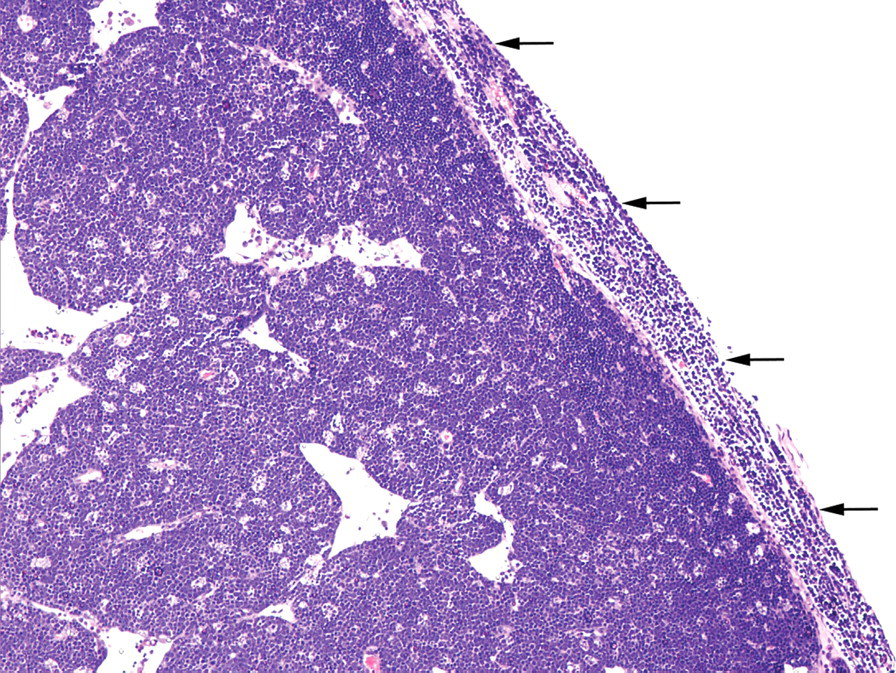
Thymus: Proliferating neoplastic cells invading thymic capsule (arrow). HE, 200×.
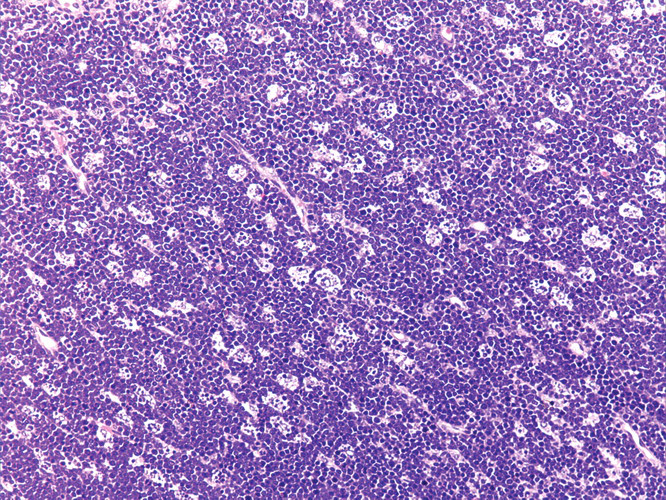
Thymus: Note the presence of tingible body macrophages imparting “starry sky appearance.” HE, 200×.
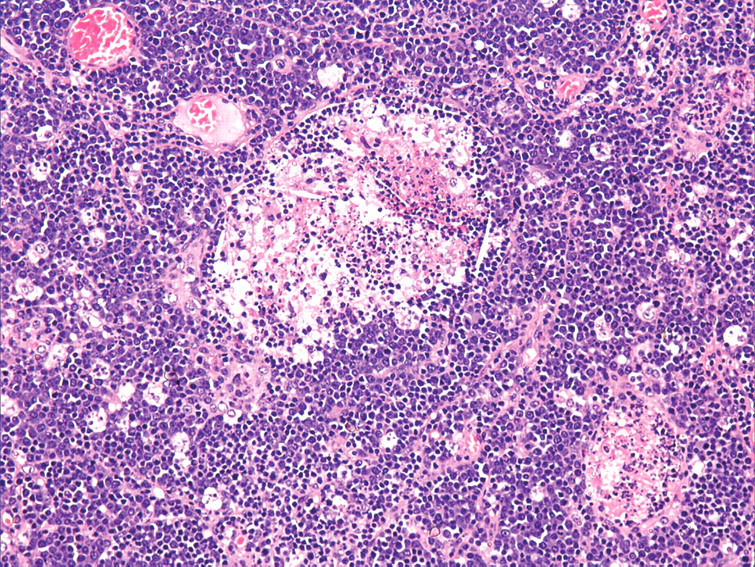
Thymus: Focal area of necrosis composed of neoplastic cells. HE, 200×.
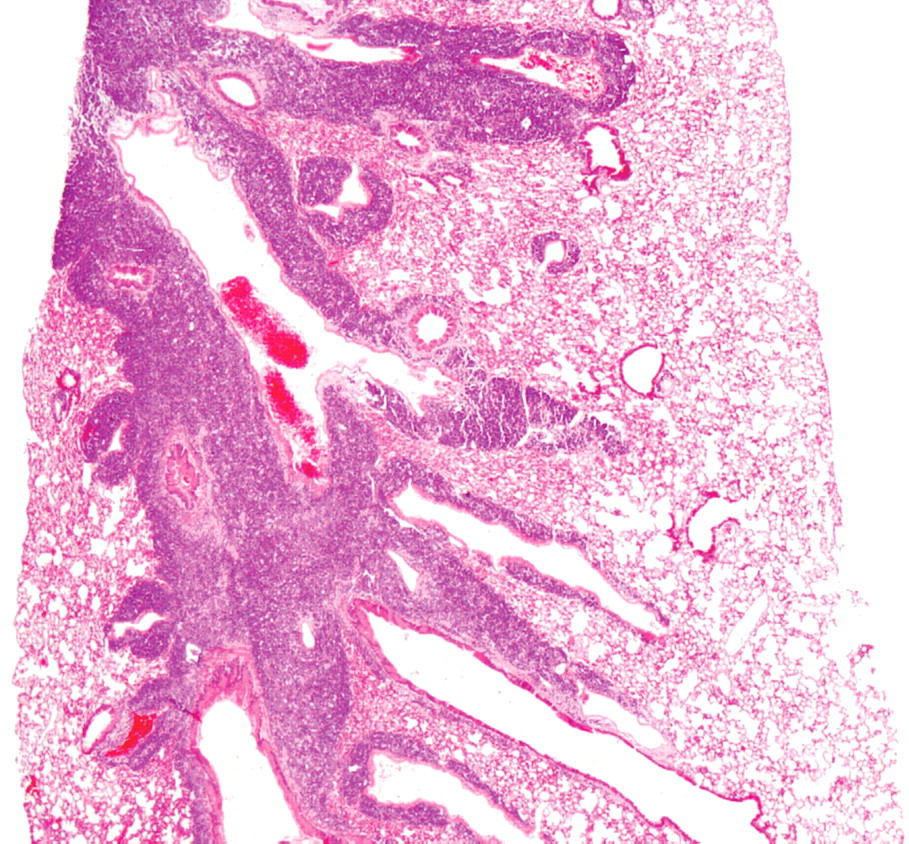
Lung: Neoplastic cells infiltration around the bronchial tree. HE, 25×.
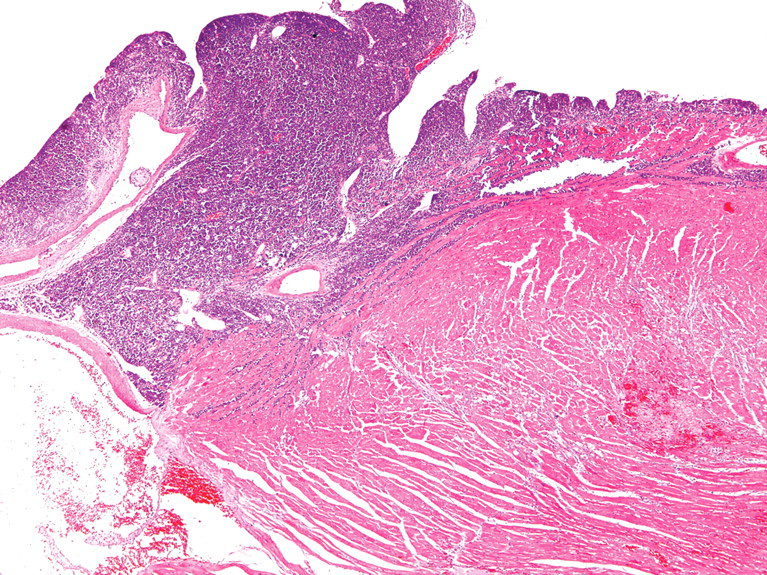
Heart: Neoplastic cells infiltration in pericardium, auricles, and ventricles. HE, 40×.
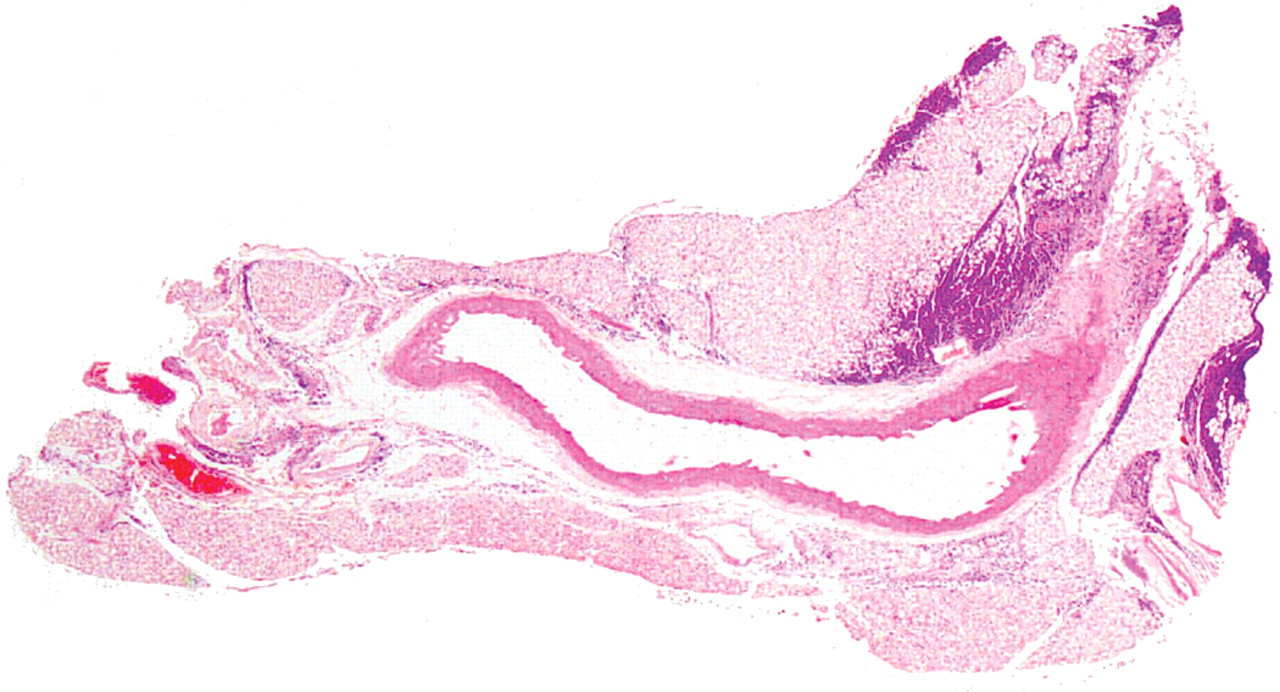
Aorta: Neoplastic cells infiltration in the peri-aortic adipose tissue. HE, 25×.
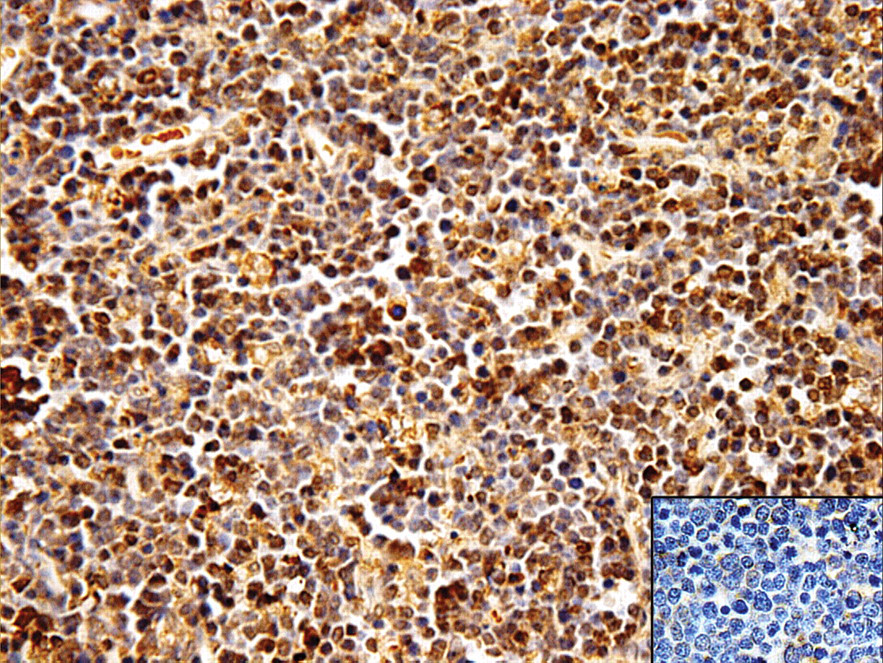
Thymus: Neoplastic T-cells expressing CD3 antigen. (Inset) Negative control image. IHC, hematoxylin, 400×.
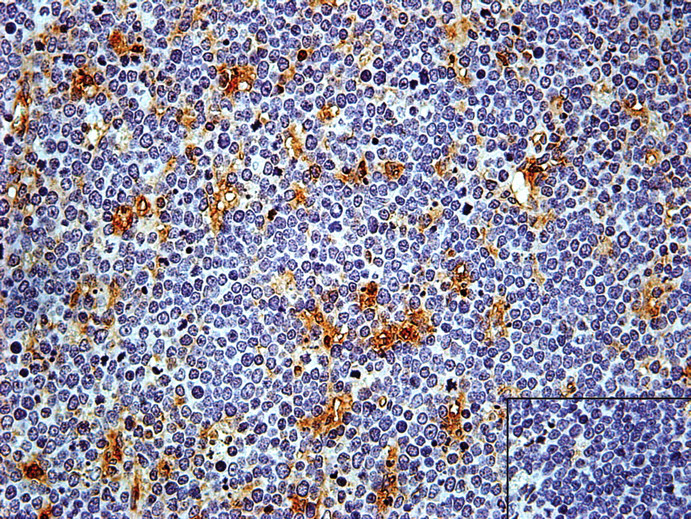
Thymus: Note the presence of CD20 stained B-lympho-cytes. (Inset) Negative control image. IHC, hematoxylin, 400×.
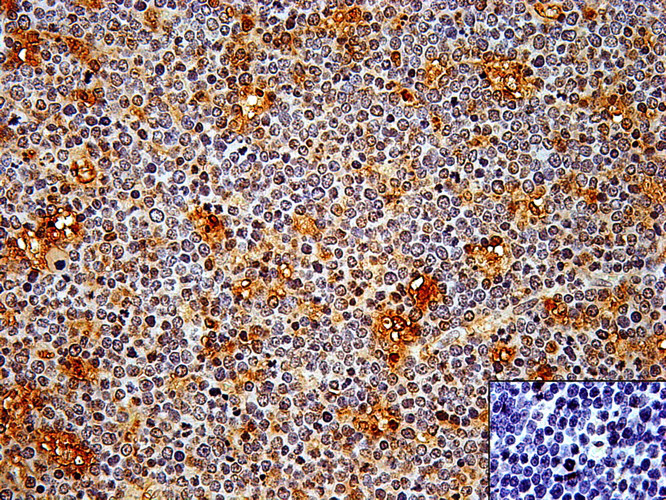
Thymus: Scattered clusters of cytokeratin-stained epithelial cells. (Inset) Negative control image. IHC, hematoxylin, 400×.
Mouse thymus develops from the endoderm of the 3rd and 4th pharyngeal pouches (Pearse 2006a). As development progresses, the thymus along with the thyroid and parathyroid, sharing the same pharyngeal pouch origin, migrate caudally (Pearse 2006a). They separate around day 15, when the thymus migrates into the thorax (Pearse 2006a). Once migration is complete, the epithelial cells organize into a loose meshwork separated by the developing vasculature. Thymus becomes a lymphoepithelial organ following rapid turnover of lymphocyte precursors from developing hematopoietic tissues on gestational days 11–12 (Pearse 2006a). By days 14–15, the thymus rapidly develops, and by 18 days of gestation, it reaches its maximum size (Maronpot, Boorman, and Gaul 1999). In mice, the thymus reaches its greatest size at sexual maturity, which in turn depends on nutritional status, steroid hormone levels, and stress. Thymic involution then begins and occurs rapidly between 35 to 80 days (Maronpot, Boorman, and Gaul 1999) and more slowly between 4 to 9 months of age. The changes of involution included loss of thymus size, mass, cortical T cells, epithelial cells, and stroma. Abnormally enlarged size of the thymus observed after involution period is due to either hyperplasia or neoplasia. Most commonly encountered neoplasms in thymus are lymphoma and thymoma.
Lymphoid hyperplasia may be unilateral or bilateral and focal or diffuse, occurs in mice greater than 3–6 months old, and is more common in females (Maronpot, Boorman, and Gaul 1999; Pearse 2006b). It consists of proliferations of lymphoid cells frequently in the vicinity of the corticomedullary junction (Pearse 2006b). Epithelial and lymphoid cell hyperplasia may occur together, especially when thymus undergoes involution (Pearse 2006b). Atypical hyperplasia is the preneoplastic lesion to the thymic lymphoma where the normal architecture is replaced by sheets of large, atypical lymphocytes and fewer admixed small lymphocytes (Maronpot, Boorman, and Gaul 1999; Pearse 2006b). This can be differentiated from lymphoma by the heterogeneous cell population, variable mitotic index, and failure of lymphocytes to extend beyond the capsule of the thymus (Maronpot, Boorman, and Gaul 1999; Pearse 2006b).
Lymphoma grows quickly, obliterates thymic architecture, penetrating the capsule, and disseminates to other sites. In some cases the initial lesion appears to be cortical atrophy characterized by loss of cortical lymphocytes followed by enlargement of the thymus due to lymphocytic proliferation, which progresses to generalized involvement of the thymus (Pearse 2006b). Microscopically, in thymic T-cell lymphoma, neoplastic cells obliterating the normal thymic architecture and infiltrating in adjacent adipose tissues and organs corroborate the earlier findings (Pearse 2006b). Neoplastic cells with moderate cytoplasm arranged in cords and sheets observed were appealing. Morphology of the neoplastic cells, mitotic figures, and tingible body macrophages observed agreed with reported literature (Pearse 2006b). No parallel findings explaining the multifocal areas of necrosis composed of neoplastic cells were cited. However, this could be because of the extensive cellular turnover in the neoplastic tissue. Most of the spontaneous and induced lymphomas in the mouse arise unilaterally in the thymus (Frith, Pattengale, and Ward 1985; Pearse 2006b), which is not in accordance with the present case where the neoplasm is observed bilaterally.
Maita et al. (1988) and Maronpot, Boorman, and Gaul (1999) reported abdominal organs (liver and kidney) and lymphatic tissues (lymph nodes and spleen) as preferential sites of metastasis of systemic lymphoma. However, in the present case, lymphoma observed primarily in the thymus invaded locally only to the neighboring organs, ruling out systemic involvement. Expression of CD3 antigen by neoplastic cells in the thymus indicates the tumor from T-cell origin (Maronpot, Boorman, and Gaul 1999). The invasive component in the peribronchial tissue and heart was composed of entirely lymphoid cells, which were positive only for CD3 antigen. The presence of nonneoplastic CD20 stained B-cells and cytokeratin stained epithelial cells in the thymus were suggestive of reactive lymphocytes and entrapped epithelial cells. The earliest occurrence of lymphoma is identified at the ninth week of a study (Son and Gopinath 2004). But thymic lymphoma in a male CD-1 mouse at fifteen weeks of age has not been reported earlier. Hence, a rare case of nonsystemic, locally invasive, thymic T-cell lymphoma in a male CD-1 mouse has been reported.
Footnotes
Acknowledgments
The authors wish to extend their thanks to the Ranbaxy Research Laboratories for the provision of a facility to carry out their work.
